1 The First School of Clinical Chinese Medicine, Hunan University of Chinese Medicine, 410208 Changsha, Hunan, China
2 Department of Neurology, The First Affiliated Hospital of Hunan University of Chinese Medicine, 410006 Changsha, Hunan, China
3 The Key Laboratory of Hunan Province for Integrated Traditional Chinese and Western Medicine on Prevention and Treatment of Cardio-Cerebral Diseases, College of Integrated Traditional Chinese and Western Medicine, Hunan University of Chinese Medicine, 410208 Changsha, Hunan, China
†These authors contributed equally.
Abstract
Ischemic stroke leads to significant neuronal damage, and impaired angiogenesis remains a critical factor limiting post-stroke recovery. Ginkgolide B (GB), a key component of Ginkgo biloba extract, has shown potential neuroprotective effects, but its pro-angiogenic mechanisms remain unclear.
To investigate the effects of GB, we established an oxygen–glucose deprivation/reperfusion (OGD/R) model using bEnd.3 cells. Potential molecular targets of GB were explored through a combination of network pharmacology analysis, protein–protein interaction (PPI) network construction, pathway enrichment, and molecular dynamics simulations. Based on these predictions, a series of in vitro assays—including Cell Counting Kit-8 (CCK-8), 5-ethynyl-2′-deoxyuridine (EdU) incorporation, wound-healing, Transwell migration, and Matrigel tube formation tests—were performed to evaluate cell viability, proliferation, migration, and angiogenic activity. Western blotting was conducted to detect AKT serine/threonine kinase 1 (AKT1), vascular endothelial growth factor (VEGF), and Angiogenin (Ang) expression and clarify the role of the AKT1/VEGF/Ang pathway.
Bioinformatics analysis identified 19 potential targets, among which AKT1, Matrix Metalloproteinase 9 (MMP9), and Prostaglandin-Endoperoxide Synthase 2 (PTGS2) exhibited the highest relevance. GB showed no evident cytotoxicity at concentrations up to 40 μM and mitigated the OGD/R-induced reduction in cell viability. At this concentration range, GB also enhanced endothelial proliferation, migration, and tube formation in bEnd.3 cells. Mechanistic studies revealed that MK2206 inhibition of AKT1 markedly suppressed AKT1 expression (p < 0.01), impaired angiogenic capacity, and aggravated ischemic–hypoxic injury, whereas GB treatment significantly increased VEGF and Ang expression (p < 0.01), likely via AKT1 upregulation (p < 0.01).
GB promotes angiogenesis and exerts neuroprotective effects by activating the AKT1/VEGF/Ang signaling pathway, suggesting its potential therapeutic value for ischemic stroke–related injuries.
Keywords
- Ginkgolide B
- ischemic stroke
- angiogenesis
- AKT1
Ischemic stroke (IS) is a common and severe neurological disorder caused by impaired cerebral blood flow, resulting in localized ischemic–hypoxic necrosis and subsequent neurological deficits [1]. It is the primary cause of long-term disability and ranks second among global causes of death [2]. The sustained deprivation of oxygen and nutrients results in neuronal injury, which constitutes the key pathological feature of IS. Collateral circulation serves as an important compensatory mechanism that helps restore regional perfusion and oxygen supply, thereby improving clinical outcomes [3]. Angiogenesis, a principal component of tertiary collateral circulation, drives the formation of new microvessels from existing vasculature. This process supports the ischemic penumbra by providing oxygen and nutrients, thus promoting repair after ischemic injury [4, 5]. Both clinical and experimental studies have shown that enhanced angiogenesis contributes to tissue recovery, vascular remodeling, and improved perfusion in peri-infarct regions, ultimately facilitating neurological restoration [6, 7, 8]. For these reasons, promoting post-ischemic angiogenesis has emerged as a potential therapeutic strategy to improve outcomes after cerebral infarction [9, 10, 11]. In particular, early and robust angiogenic responses can stabilize local blood flow, limit neuronal loss, and reduce neurological deficits in the acute phase [12, 13].
Traditional Chinese medicine (TCM) has gained attention in recent years for its potential in preventing and treating cardiovascular and cerebrovascular diseases. Its therapeutic effects are often attributed to multi-target actions and generally favorable safety profiles. Ginkgo biloba leaves, a classical medicinal herb, contain bioactive compounds capable of modulating the onset and progression of ischemic stroke through multiple mechanisms [14, 15, 16]. Among these, diterpene lactones are the main pharmacologically active constituents, with Ginkgolide B (GB) being the most representative and demonstrating significant pharmacological activity [17]. Previous research has shown that GB exerts neuroprotective effects, improves endothelial function, and possesses anti-inflammatory and antioxidant activities [18, 19, 20, 21], and exerts therapeutic effects in various free radical–related diseases, including IS [22, 23], ischemic heart disease [24], chronic inflammation [25], and aging [26].
The AKT Serine/Threonine Kinase 1 (AKT1) signaling pathway, also referred to as the PI3K/AKT pathway, regulates a range of cellular processes, including survival, proliferation, metabolism, angiogenesis, migration, and apoptosis [27, 28, 29, 30]. Evidence indicates that AKT1 is central to the pathophysiology of cardiovascular and cerebrovascular disorders, such as ischemic stroke [31], atherosclerosis [32], and hypertension [33]. Despite its established roles, it remains unclear whether GB can promote post-stroke angiogenesis by modulating AKT1 signaling, and this question has not been systematically addressed.
Building on these findings, this study aims to clarify how GB modulates the AKT1 signaling pathway to promote post-stroke angiogenesis. By elucidating these mechanisms, we hope to provide new insights into cerebrovascular repair and to identify therapeutic targets that may inform future treatment strategies.
The mouse brain microvascular endothelial cell line (bEnd.3, lot number CL-0598) was obtained from Wuhan Punosai Life Science Co., Ltd. Cells were maintained in high-glucose Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 5% fetal bovine serum (FBS) and 1% penicillin–streptomycin at 37 °C in a humidified incubator containing 5% CO2, and were subcultured upon reaching 80–90% confluence. All cell lines were validated by STR profiling and tested negative for mycoplasma.
Ginkgolide B (GB; purity 98% by HPLC) was provided by the U.S. National Institute for Control of Pharmaceutical and Biological Products (Beijing, China). The powder was dissolved in dimethyl sulfoxide (DMSO; D2650) to prepare a 100 µM stock solution, which was stored at –20 °C protected from light and freshly prepared prior to use.
Based on the study by Zhou et al. [34], 3-n-Butylphthalide (NBP) was
used as a positive control. NBP (
Puromycin (lot number P8230) was purchased from Beijing Soleibao Biotechnology
Co., Ltd. (Beijing, China). Fetal bovine serum (FBS; lot 164210-50),
penicillin–streptomycin solution (lot PB180120), 0.25%
trypsin–Ethylenediaminetetraacetic Acid (EDTA) (lot PB180229), high-glucose DMEM
(lot PM150210), glucose-free DMEM (lot PM150270), PBS buffer (lot PB180327), and
serum-free cryopreservation solution (lot PB180438) were all purchased from Wuhan
Procell Co., Ltd. (Wuhan, China). Matrigel matrix (lot 356234) was purchased from
BD Biosciences, (San Jose, CA, USA), and Transwell 24-well plates (lot 3422) were
purchased from Corning (Corning, NY, USA). Bicinchoninic Acid (BCA) protein
assay kit (catalog E-BC-K318-M) and Cell Counting Kit-8 (CCK-8) (catalog
E-CK-A362) were purchased from Wuhan Elairuite Biotechnology Co., Ltd. (Wuhan,
China).
The following instruments were used in this study: incubator (model 3427), tri-gas incubator (model 3131), ultra-low temperature freezer (900 Series), and refrigerated high-speed centrifuge Fresco17 (Thermo Fisher Scientific, Waltham, MA, USA); low-speed centrifuge (Changsha Yingtai Instrument Co., Ltd. Changsha, China, model TD5A); constant-temperature water bath (Crystal Instruments, Silicon Valley, CA, USA, model SYG-1210); inverted microscope (ZEISS, Oberkochen, Germany, model AxioVert.A1); laminar flow cabinet (Suzhou Antai Air Technology Co., Ltd., Sujing Group, Suzhou, China, model SW-CJ-1F); multifunctional microplate reader (BioTek, VT, USA, model Cytation3); 0.22 µm Polyvinylidene Fluoride (PVDF) membrane (Millipore, MA, USA, catalog ISEQ00010); analytical microbalance (SHIMADZU, Kyoto, Japan, model AUW220D); power supply for general electrophoresis (Shanghai Yamei Biotechnology Co., Ltd., Shanghai, China, model UPS-600); and vertical electrophoresis chamber and transfer module (Bio-Rad, Hercules, USA, models Mini-PROTEAN® TetraCell and Mini Trans-Blot Module).
The chemical structure of GB was retrieved from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) using the keyword “Ginkgolide B”, and both its SMILES or 3D structure were downloaded (Fig. 1a). Potential targets of GB were obtained from the PharmMapper (http://www.lilab-ecust.cn/pharmmapper/) and TargetNet (http://targetnet.scbdd.com/) databases. After removing duplicates, all targets were mapped to their corresponding gene names using the UniProt database (https://www.uniprot.org/).
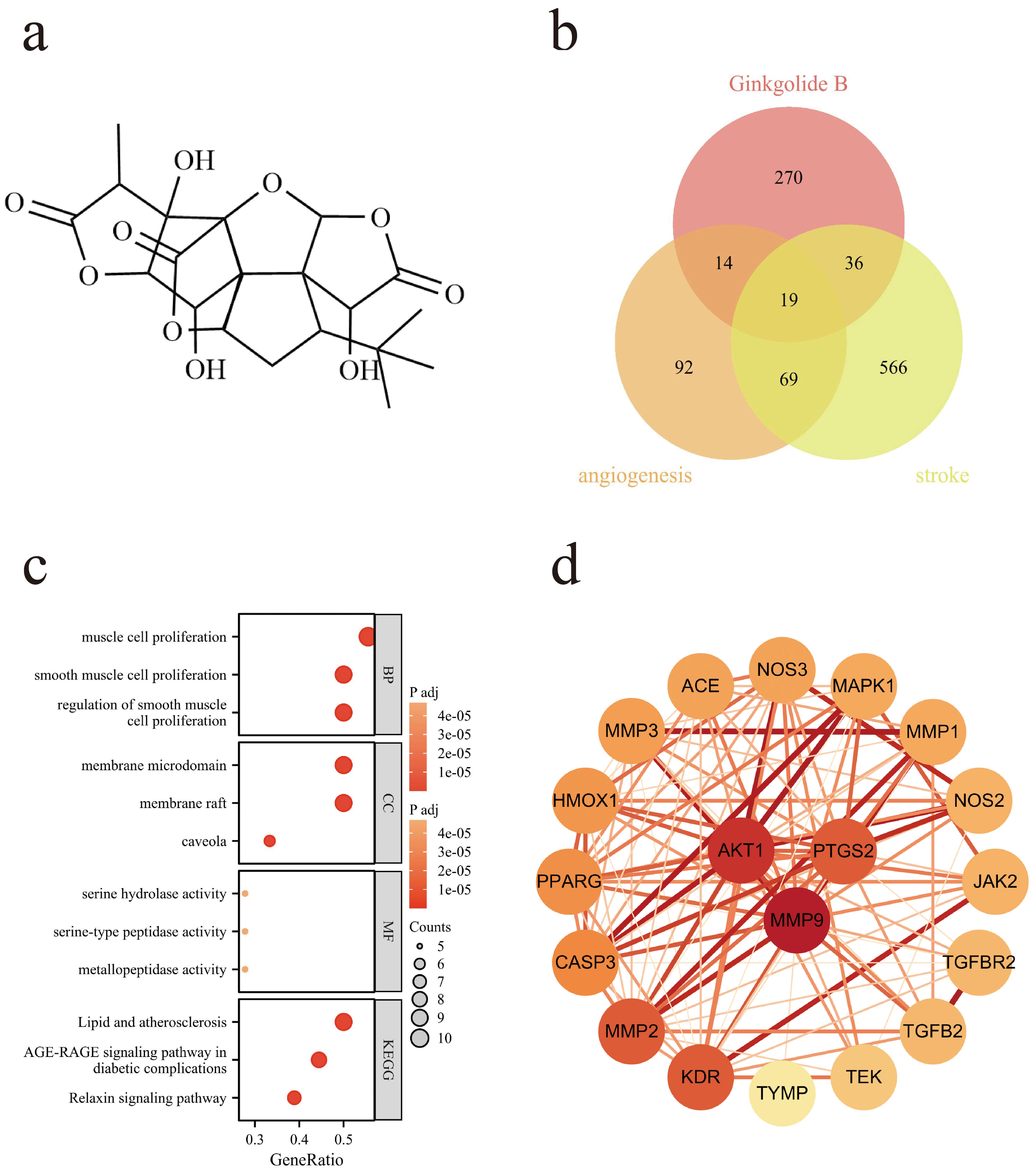 Fig. 1.
Fig. 1.
Network pharmacology analysis of GB targets. (a) Chemical structure of GB. (b) Venn diagram showing the overlap among predicted GB targets, ischemic stroke-related genes, and angiogenesis-related genes. (c) GO and KEGG enrichment analyses of the overlapping targets. (d) PPI network of the 19 overlapping targets. GB, Ginkgolide B; PPI, Protein–protein interaction.
Genes related to angiogenesis were retrieved from the human GeneCards database (https://www.genecards.org) using the keyword “angiogenesis”. Similarly, genes associated with stroke were obtained by querying the keyword “stroke” in the same database.
Data from Sections 2.4.1 and 2.4.2 were first combined. R software (R Foundation for Statistical Computing, Vienna, Austria) was then used to identify unique and overlapping targets among the groups. The results were visualized using the ggplot2 and VennDiagram packages to highlight potential GB targets involved in post-stroke angiogenesis.
Protein–protein interaction (PPI) analysis of GB-related targets was conducted using the STRING database (https://cn.string-db.org/). The resulting network was visualized in Cytoscape software (Cytoscape Consortium, San Diego, CA, USA) to facilitate further analysis.
Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses of GB-related targets were conducted using the ggplot2 package in R. Biological processes (BP), cellular components (CC), molecular functions (MF), and KEGG pathways were analyzed to explore the potential mechanisms by which GB may exert its effects in cerebral infarction.
Molecular docking and molecular dynamics (MD) simulations were performed to assess the interactions between GB and key angiogenesis-related proteins. Docking was carried out on the CB-Dock2 platform [35, 36], which detects potential binding pockets using the CurPocket algorithm [37] and evaluates binding affinities with the AutoDock Vina scoring function. The crystal structures of AKT1 (PDB ID: 3O96), ANG (PDB ID: 1ANG), and VEGF (PDB ID: 4DEQ) were retrieved from the Protein Data Bank. GB was converted to mol2 format and docked into the five top-ranked binding pockets, and the conformation with the lowest binding energy was selected for further analysis. The resulting complexes were visualized in Discovery Studio 2021(BIOVIA, San Diego, CA, USA) and PyMOL v2.6 (Schrödinger, LLC, New York, NY, USA).
MD simulations were carried out using GROMACS v2022.03 (GROMACS Development Team) [38, 39]. The protein structures were parameterized with the AMBER99SB-ILDN force field, and ligand topologies were generated using the Generalized Amber Force Field (GAFF). Partial atomic charges were assigned via the restrained electrostatic potential (RESP) method implemented in Gaussian 16W. Each protein–ligand complex was solvated in a cubic Transferable Intermolecular Potential with 3 Points (TIP3P) water box, keeping at least 1.2 nm between the solute and the box edge, and counterions (Na⁺) were added to neutralize the system. Energy minimization was performed using the steepest descent algorithm. Short-range electrostatic and van der Waals cutoffs were set to 1.0 nm, and long-range electrostatics were treated with the particle mesh Ewald (PME) method. All bonds involving hydrogen atoms were constrained using the LINCS algorithm. The system was equilibrated under constant number of particles, volume, and temperature (NVT) and constant number of particles, pressure, and temperature (NPT) ensembles for 100 ps each to stabilize temperature and pressure. Subsequently, a 50 ns production run was performed under NPT conditions at 300 K and 1 bar, using a 2 fs integration step. Trajectories were recorded every 10 ps for further analysis.
Following the simulations, we analyzed the trajectories to examine the structural stability and binding dynamics of the protein–ligand complexes. The root-mean-square deviation (RMSD), radius of gyration (Rg), root-mean-square fluctuation (RMSF), solvent-accessible surface area (SASA), and hydrogen bond number were calculated to assess conformational stability and overall compactness. To further characterize the energetic landscape, Gibbs free energy surfaces were constructed using the g_sham module in GROMACS. Binding free energies were then estimated with the MM/PBSA approach implemented in gmx_MMPBSA, using snapshots extracted from the final 20 ns of the production trajectories.
To induce oxygen–glucose deprivation (OGD), the culture medium was replaced with glucose-free DMEM, and bEnd.3 cells were incubated under hypoxic conditions (1% O2, 5% CO2, 94% N2) at 37 °C for 6 hours. After hypoxic exposure, the medium was changed to complete DMEM, and the cells were returned to normoxic conditions for 24 hours to allow reperfusion.
The study included six experimental groups: control, model, 10 µM GB, 20 µM GB, 40 µM GB, and NBP. The control group was maintained under normoxic conditions, whereas the model group was subjected to OGD/R as described above. For GB treatment, cells underwent OGD for 6 hours and were then incubated with GB at final concentrations of 10, 20, or 40 µM during the 24-hour reperfusion period. The positive control group received 10 µM NBP under the same post-OGD/R conditions.
To determine the safe concentration range of GB, bEnd.3 cells were cultured in a control group (0 µM) and treatment groups with varying GB concentrations (0, 5, 10, 20, 40, 80, and 160 µM). After 24 hours, cell viability was measured using the CCK-8 assay. Briefly, 10 µL of CCK-8 reagent was added to each well containing 100 µL of fresh medium, and the plates were incubated at 37 °C with 5% CO2 for 2 hours. Absorbance at 450 nm was recorded using a multifunctional microplate reader to identify the optimal GB concentration for subsequent experiments.
After OGD/R treatment, bEnd.3 and control cells were seeded at 1.8
To standardize imaging positions across samples, five evenly spaced horizontal
reference lines were drawn on the underside of each 6-well plate. bEnd.3 cells
were seeded at a density of 2
Wound closure (%) =
Where W0 represents the initial wound width and Wt denotes the wound width measured after 24 hours.
For the Transwell migration assay, cells were harvested by trypsinization,
resuspended in serum-free medium, and adjusted to a density of 1.8
For the tube formation assay, growth factor–reduced Matrigel was thawed on ice,
aliquoted at 50 µL per well, and allowed to solidify at 37
°C for 30 minutes. bEnd.3 cells were harvested by trypsinization,
resuspended in serum-free medium, and adjusted to a final concentration of 1.8
Total protein was extracted from each experimental group using RIPA buffer
supplemented with protease inhibitors. Protein concentrations were determined
with a BCA assay kit, and equal amounts of protein were resolved by SDS–PAGE at
160 V for 45 minutes. Proteins were subsequently transferred onto PVDF membranes
at a constant current of 300 mA for 30 minutes. Membranes were blocked with a
rapid protein-free blocking buffer and incubated overnight at 4 °C with
the following primary antibodies: AKT1 (1:1000), VEGF (1:5000), Ang (1:1000), and
Statistical analyses were conducted using SPSS version 26.0 (IBM Corp.,
Armonk, NY, USA). Data are expressed as mean
Potential targets of GB were initially identified using PharmMapper and
TargetNet, yielding 299 and 58 candidates, respectively. Representative targets
from PharmMapper included NR1H2, GSTP1, BCHE, METAP1, and GBA, while TargetNet
examples comprised NOS2, CHRM4, CASP9, NR2F2, and NR3C1. All targets were mapped
to gene symbols via the UniProt database, though eight proteins lacked
annotations. After removing duplicates, a total of 339 unique GB targets were
retained. Genes associated with angiogenesis and stroke were retrieved from
GeneCards (https://www.genecards.org) using a relevance score threshold of
The PPI analysis of the 19 predicted GB targets identified 110 interaction
pairs, with AKT1, MMP9, and PTGS2 exhibiting the highest connectivity (Fig. 1d).
GO and KEGG enrichment analyses were performed with a significance threshold of
p
To evaluate the potential of GB in regulating angiogenesis, molecular docking and molecular dynamics simulations were performed to systematically assess its interactions with key angiogenesis-related proteins. Docking analysis revealed that GB binds to AKT1 with a binding energy of –10.2 kcal/mol, forming hydrogen bonds with Glutamine (GLN79), Asparagine (ASN54), and Tyrosine (TYR326), a carbon–hydrogen bond with TYR272, and a repulsive interaction with ARG273 (Fig. 2a). Comparable intermolecular interaction patterns were also observed with ANG and VEGF (Fig. 2b,c).
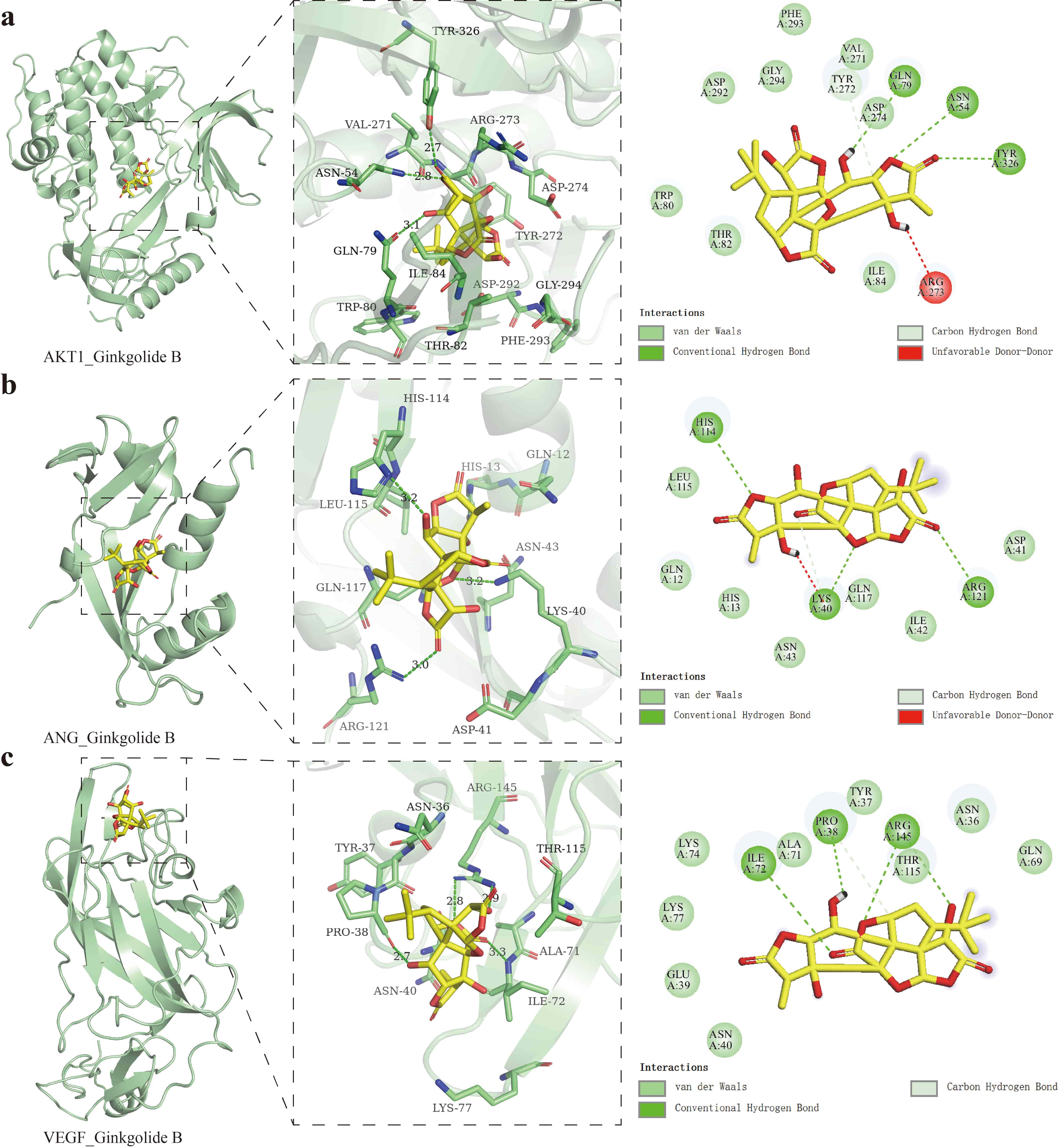 Fig. 2.
Fig. 2.
Molecular docking results. (a) Molecular docking of GB with AKT1. (b) Molecular docking of GB with ANG. (c) Molecular docking of GB with VEGF.
MD simulations were performed to assess the stability and conformational
dynamics of GB bound to AKT1, ANG, and VEGF (Fig. 3). RMSD analysis (Fig. 3a)
indicated that the AKT1–GB and ANG–GB complexes remained below 0.35 nm,
reflecting structural stability, whereas the VEGF–GB complex exhibited larger
fluctuations (
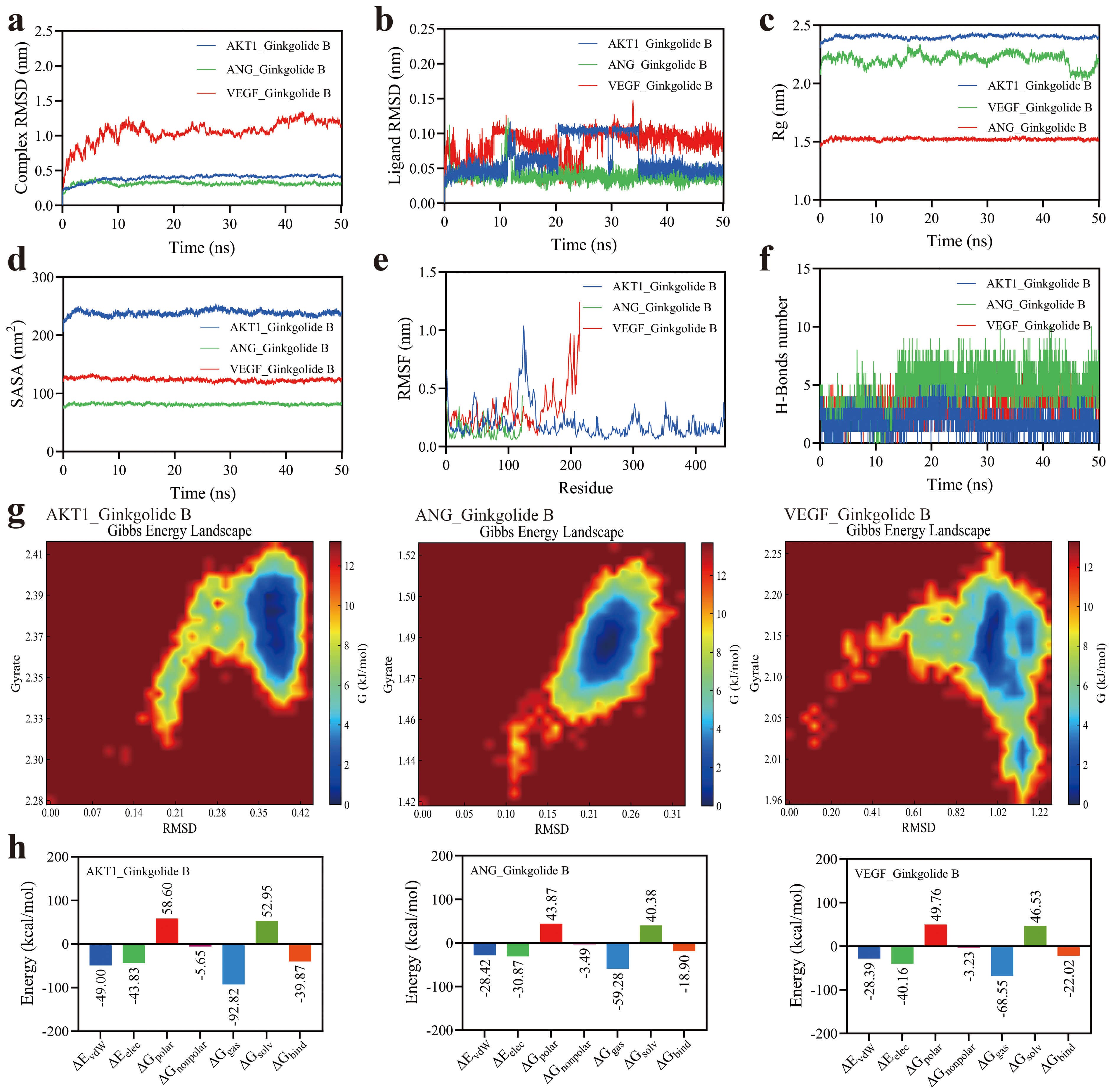 Fig. 3.
Fig. 3.
MD simulation analysis of the GB–protein complexes over 50 ns. (a) RMSD of the complexes. (b) RMSD of GB ligands. (c) Rg of proteins. (d) SASA of proteins. (e) RMSF of proteins. (f) Number of hydrogen bonds between GB and proteins. (g) Gibbs free energy landscape of the complexes. (h) Binding free energy of GB–protein interactions calculated by MM/PBSA. RMSD, root-mean-square deviation; Rg, radius of gyration; SASA, solvent-accessible surface area; RMSF, root-mean-square fluctuation.
Solvent-accessible surface area (SASA) analysis (Fig. 3d) supported these
observations, with AKT1 exhibiting the largest accessible surface
(~220 nm2), ANG the smallest (~90 nm2),
and VEGF intermediate (~120 nm2). RMSF analysis (Fig. 3e)
revealed localized flexibility, with AKT1 residues 100–150 showing minor
fluctuations (RMSF
Gibbs free energy landscape analysis (Fig. 3g) demonstrated that the ANG–GB
complex occupied the deepest energy basin, reflecting the most stable
conformation. AKT1 adopted moderately flexible but stable states, whereas VEGF
exhibited a scattered energy distribution with multiple metastable states.
Molecular mechanics/Poisson–Boltzmann surface area (MM/PBSA) calculations (Fig. 3h) revealed that GB had the strongest binding affinity for AKT1
(
The safe concentration range of GB was determined using the CCK-8 assay. In normal bEnd.3 cells, treatment with GB at 5, 10, 20, and 40 µM had no significant effect on cell viability, whereas higher concentrations (80 and 160 µM) caused marked cytotoxicity (Fig. 4a,b). In the OGD/R model, cell viability was significantly reduced; treatment with GB at 10, 20, or 40 µM effectively attenuated this injury in a dose-dependent manner. Based on these results, 10, 20, and 40 µM were selected for subsequent experiments to investigate the stress-repair effects of GB and its underlying mechanisms in OGD/R-induced bEnd.3 cell injury.
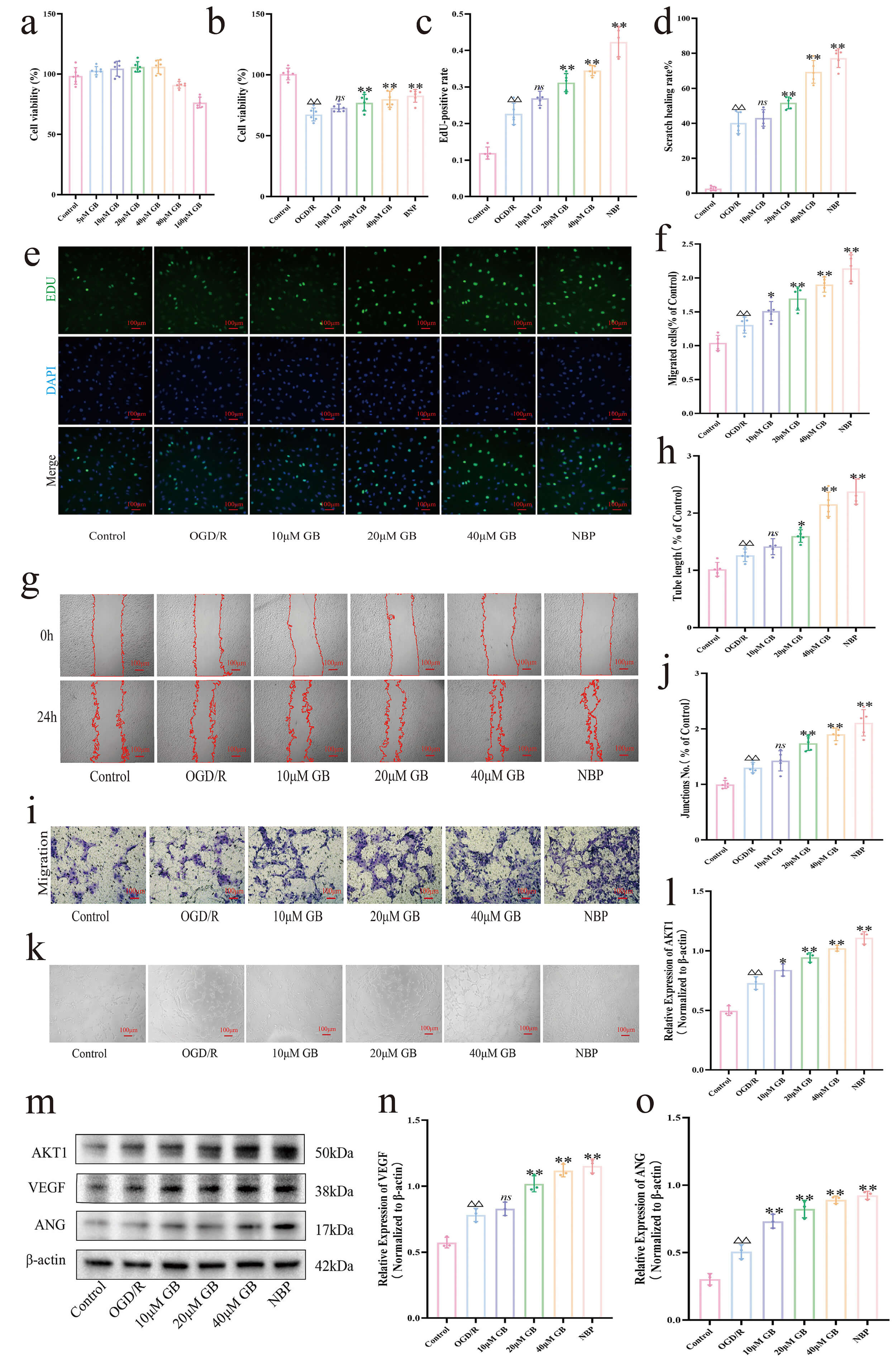 Fig. 4.
Fig. 4.
GB promotes angiogenesis in vitro. (a) CCK-8 assay
evaluating the effects of different concentrations of GB on bEnd.3 cell viability
(n = 6). (b) CCK-8 assay assessing bEnd.3 cell viability following GB
intervention at different concentrations (n = 6). (c) Quantification of
EdU-positive cells after GB treatment (n = 5). (d) Quantitative analysis of
horizontal migration of bEnd.3 cells (n = 5). (e) Representative EdU
intake assay (Scale bar, 100 µm, 100
To evaluate the effect of GB on endothelial cell proliferation, EdU assays were
performed in bEnd.3 cells. Results showed that treatment with GB at 20, and 40
µM, as well as with NBP, for 24 h significantly increased the proportion of
EdU-positive cells relative to the control (p
Significant differences in endothelial cell proliferation were observed among
the treatment groups (Fig. 5a,d). OGD/R treatment alone increased the proportion
of EdU-positive cells relative to the control (p
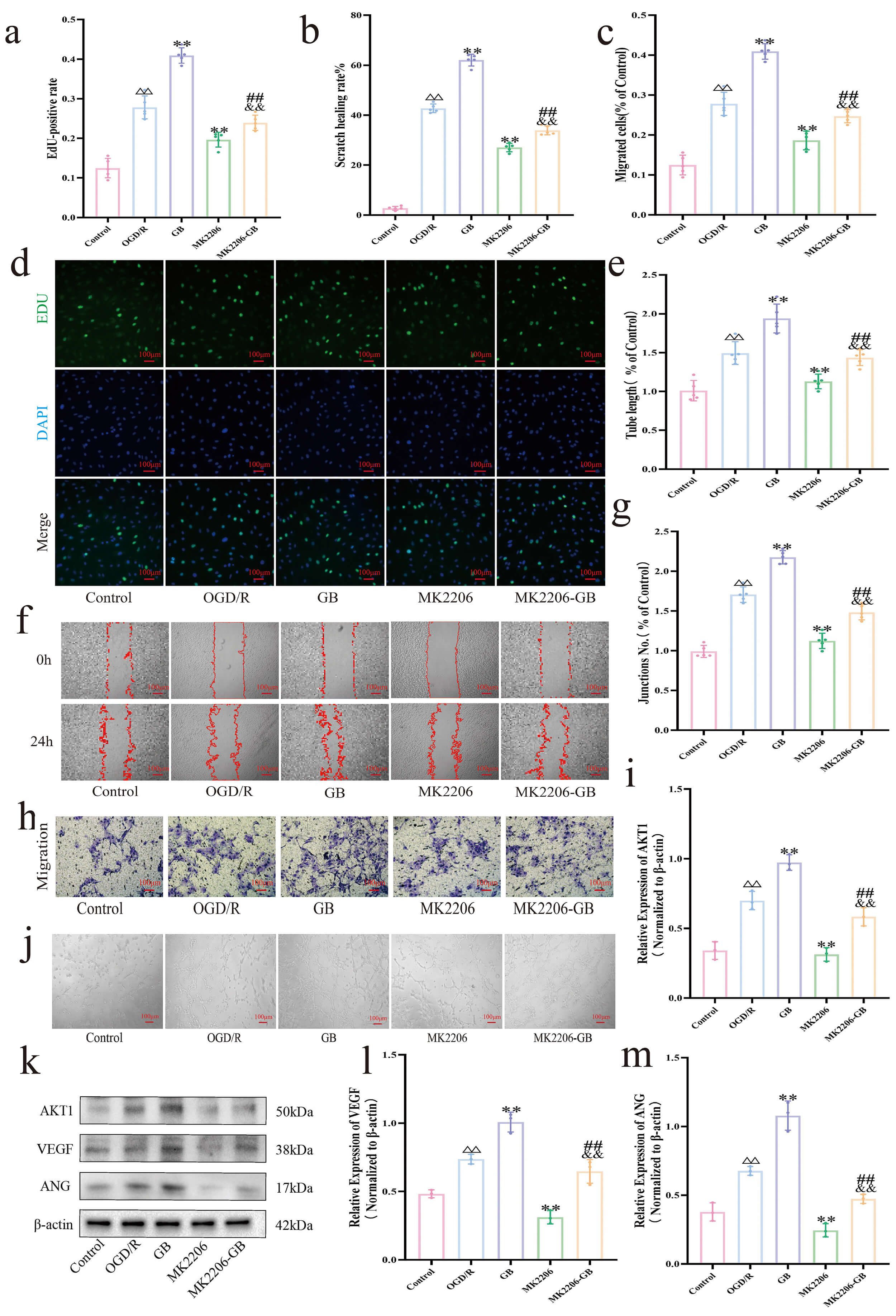 Fig. 5.
Fig. 5.
Ginkgolide B promotes angiogenesis and exerts neuroprotective
effects through activation of the AKT1/VEGF/Ang signaling pathway. (a)
Quantification of EdU-positive cells (n = 5). (b) Quantitative analysis of
horizontal migration of bEnd.3 cells (n = 5). (c) Quantitative analysis of
vertical migration of bEnd.3 cells (cells passing through Matrigel-free Transwell
chambers, n = 5). (d) Representative EdU intake assay (Scale bar, 100
µm, 100
The migratory capacity of bEnd.3 cells was evaluated using wound healing and
Transwell assays (Fig. 4d,f,g,i). OGD/R treatment significantly increased cell
migration relative to the control (p
Migration analysis across treatment groups (Fig. 5b,c,f,h) showed that OGD/R
significantly enhanced cell migration compared with the control (p
The effect of GB on angiogenic activity under OGD/R conditions was evaluated
using a tube formation assay in bEnd.3 cells (Fig. 4h,j,k). OGD/R significantly
increased both the number of branch points and the total branch length compared
with the control (p
Tube formation analysis across treatment groups (Fig. 5e,g,j) showed that
OGD/R significantly increased both the number of branch points and total branch
length compared with the control (p
VEGF and Ang are key mediators of endothelial cell angiogenesis, and their
expression is regulated by AKT1. Western blot analysis (Fig. 4l–o) showed that
OGD/R significantly upregulated VEGF, Ang, and AKT1 protein levels compared with
the control (p
Protein expression analysis across treatment groups (Fig. 5i,k–m) showed that OGD/R significantly upregulated VEGF, Ang, and AKT1 compared with the control (p
In this study, we combined network pharmacology with molecular simulations to
investigate the potential mechanisms by which GB may promote angiogenesis
following IS. Analysis of the PharmMapper and TargetNet databases identified 339
potential GB targets. Additionally, 194 angiogenesis-related genes and 690
stroke-related genes were retrieved from GeneCards, with intersection analysis
yielding 19 candidate targets. PPI network analysis highlighted AKT1, MMP9, and
PTGS2 as central nodes. GO and KEGG enrichment analyses revealed that these
targets are primarily involved in inflammation, oxidative stress, apoptosis, and
angiogenesis, with significant enrichment in PI3K–Akt, MAPK, HIF-1, VEGF, and
TNF/NF-
In vitro, GB at safe concentrations (
Previous studies have shown that AKT1 activation promotes angiogenesis in ischemic regions by regulating VEGF [40, 41, 42], thereby facilitating vascular reconstruction following cerebral infarction [43, 44, 45]. VEGF, a central angiogenic mediator, maintains vascular network stability after stroke by controlling the dynamic balance between tip and stalk cells [46]. While Ang supports endothelial cell survival, neovessel maturation, and vascular stabilization [47]. Under OGD/R conditions, the observed increase in angiogenic responses mediated by VEGF and ANG likely reflects a tissue stress response to ischemia, aimed at repairing vascular damage. However, excessive oxidative stress and inflammatory cascades following ischemia appear to limit the effectiveness of this endogenous response, such that elevated VEGF and ANG levels are insufficient to fully ameliorate extensive ischemic injury. Previous studies have demonstrated that neovascular formation exhibits pronounced spatiotemporal heterogeneity: early neovessels are often structurally immature, functionally compromised, and prone to leakage, potentially exacerbating tissue damage. Treatment with GB was associated with further increases in AKT1, VEGF, and ANG levels, suggesting that GB may potentiate angiogenic signaling beyond the capacity of intrinsic repair mechanisms. Accordingly, GB treatment appears to support the formation of stable capillaries, maintain endothelial cell function in nascent vessels, enhance local perfusion, and partially mitigate post-ischemic neurological deficits. To examine whether GB’s pro-angiogenic effects depend on AKT1, we used the AKT1 inhibitor MK2206. MK2206 treatment partially reversed the OGD/R- and GB-induced increases in total AKT1 levels, significantly downregulated AKT1 expression, reduced VEGF and Ang levels, and suppressed endothelial cell proliferation, migration, and tube formation, indicating that AKT1 is a key regulator of angiogenesis under ischemic and hypoxic conditions. Conversely, GB treatment upregulated AKT1 and its downstream effectors, partially reversing the inhibitory effects of MK2206, confirming that GB promotes angiogenesis at least in part through AKT1 pathway activation.
The pathological process of ischemic brain injury is highly complex, involving
excitotoxicity, inflammation, angiogenesis, and neurogenesis [48]. Increasing
evidence suggests that GB exerts neuroprotective and pro-angiogenic effects
through multiple targets and pathways. Wei and Tang [49] reported that GB
inhibits platelet activation, reduces
Although GB generally enhanced endothelial cell viability, proliferation, migration, and tube formation, variability was observed across experiments. At 10 µM, increases in migration, proliferation, and branch point formation were inconsistent and, in several cases, did not reach statistical significance. This variability appeared more pronounced under conditions of lower cell confluence or less stable OGD/R treatment, suggesting that experimental context influences GB’s effects. Mild batch-to-batch fluctuations in VEGF and Ang expression were also noted, indicating that upstream signaling may vary with cellular state or culture conditions. Collectively, these findings suggest that GB’s effects are not strictly dose-linear and may involve a threshold-dependent mechanism. Larger-scale and inter-laboratory studies will be important to further evaluate the robustness and reproducibility of these observations.
While this study systematically elucidated the mechanisms by which GB promotes
angiogenesis via the AKT1/VEGF/Ang signaling pathway (Fig. 6), several limitations should
be acknowledged. First, angiogenesis is a dynamic, time-dependent process, yet
the present study did not fully characterize the dose–response relationship or
temporal dynamics of GB’s effects. Second, in vitro models cannot
completely replicate the complex microenvironment of cerebral infarction or the
dynamic regulation of the blood–brain barrier, and the pro-angiogenic and
neuroprotective effects of GB were not validated in vivo. Third, this
investigation focused primarily on a single signaling pathway, without examining
other critical regulators such as Notch, HIF-1
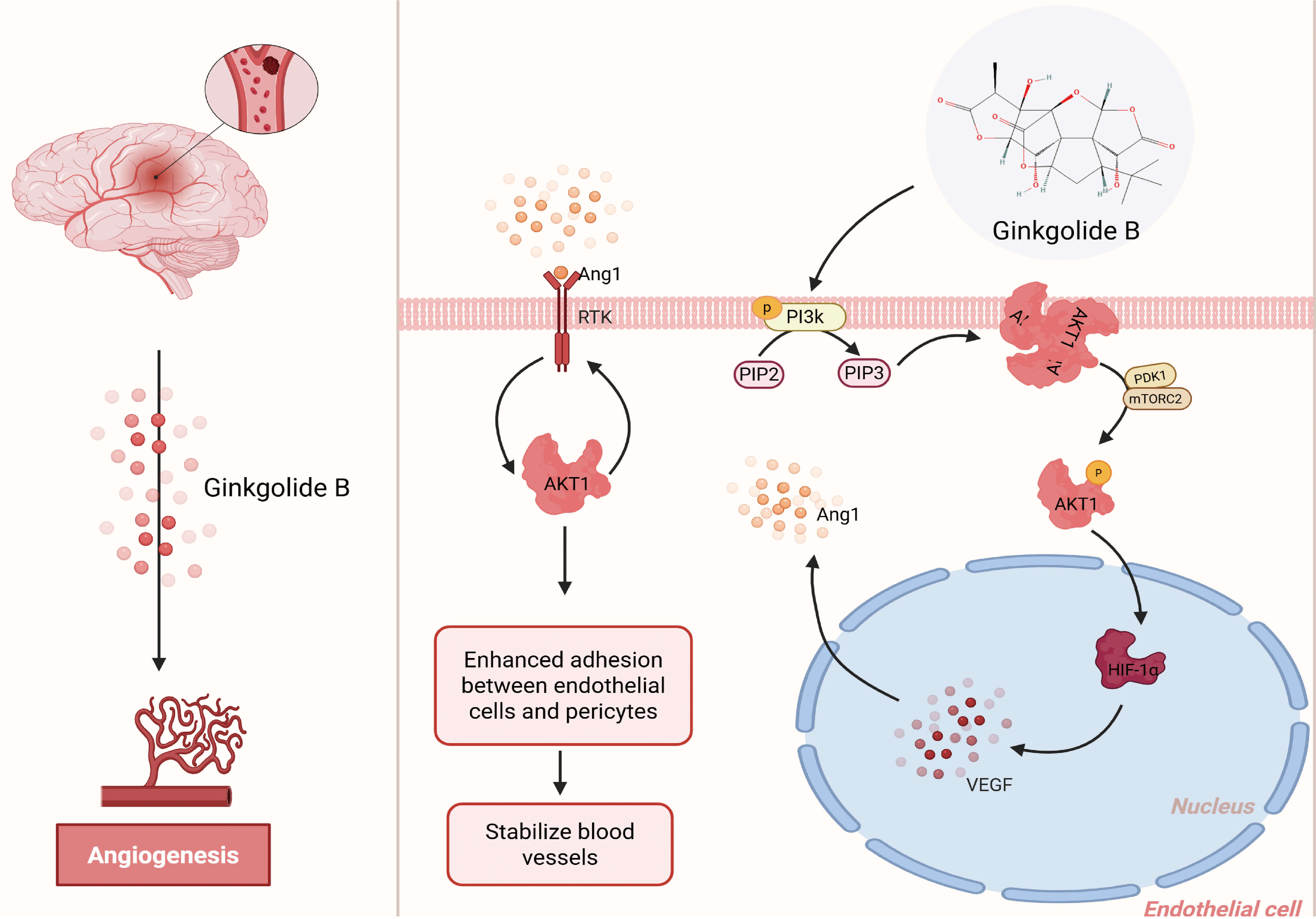 Fig. 6.
Fig. 6.
Schematic representation of the angiogenic mechanism of GB
following ischemic brain injury. GB activates the PI3K/AKT signaling pathway in
endothelial cells, which subsequently promotes HIF-1
In conclusion, by combining network pharmacology, molecular simulations, and cellular experiments, this study elucidates the mechanisms by which GB promotes angiogenesis through the AKT1/VEGF/Ang signaling pathway. These results provide direct evidence for GB’s role in vascular repair following ischemic stroke and lay a foundation for its further development and potential clinical translation.
GB, Ginkgolide B; OGD/R, oxygen–glucose deprivation/reperfusion; PPI, protein–protein interaction; IS, Ischemic stroke; TCM, traditional Chinese medicine; AKT1, The AKT Serine/Threonine Kinase 1; FBS, fetal bovine serum; NBP, 3-n-Butylphthalide; VEGF, Vascular Endothelial Growth Factor; CCK-8, Cell Counting Kit-8; ANG, Angiogenin; BP, biological processes; CC, cellular components; MF, molecular functions; MD, molecular dynamics; GAFF, Generalized Amber Force Field; EM, Energy minimization; PME, particle mesh Ewald; RMSD, root-mean-square deviation; Rg, radius of gyration; RMSF, root-mean-square fluctuation; SASA, solvent-accessible surface area; NVT, constant number of particles, volume, and temperature; NPT, constant number of particles, pressure, and temperature; BDNF, brain-derived neurotrophic factor; CKB, creatine kinase B; RESP, the restrained electrostatic potential.
The datasets generated and analyzed during the current study, including raw experimental data, are available from the corresponding author upon reasonable request.
YCL, LL and QM designed the research study. YCL, LL, QM, MHH, XFG and SYY performed the research. LJL, YC and DSZ provided help and advice on the experiments. LL analyzed the data. YCL drafted the manuscript. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work the authors used ChatGpt-3.5 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






