1 Cancer Molecular Research Laboratory (CMRL), Faculdade de Medicina de São José do Rio Preto - FAMERP, 15090-000 São José do Rio Preto, SP, Brazil
2 Department of Structural and Functional Biology, Institute of Biosciences, São Paulo State University (UNESP), 18618-689 Botucatu, SP, Brazil
3 Department of Cell Systems and Anatomy, Joe R and Teresa Lozano Long School of Medicine, UT Health San Antonio, San Antonio, TX 78229, USA
†These authors contributed equally.
Abstract
Melatonin, a highly conserved indoleamine produced by the pineal gland and also in the mitochondria of many, perhaps all, extrapineal tissues, has emerged as a powerful antioxidant molecule. This review explores its role in counteracting lipid peroxidation (LP), a process that damages cellular membranes through the oxidative degradation of lipids. LP is involved in numerous pathological conditions, including neurodegenerative diseases, cancer, cardiovascular disorders, and aging. The article discusses how melatonin prevents, mitigates, or even reverses LP-induced cellular damage by acting as both a direct free radical scavenger and as an indirect regulator of antioxidant enzymes. A key point is melatonin’s amphiphilic nature, which enables it to access both lipid and aqueous cellular compartments, allowing for broad protection and supporting its diverse antioxidant, cytoprotective, and regulatory functions within the cell. Melatonin and its metabolites, such as N1-acetyl-N2-formyl-5-methoxykynuramine and N1-acetyl-5-methoxykynuramine, interact with reactive oxygen and nitrogen species (ROS and RNS), effectively reducing the LP chain reaction. This series of protective actions is known as the melatonin antioxidant cascade. This highlights that melatonin not only inhibits the initiation and propagation phases of LP but may also contribute to the repair of oxidized membrane components. We further summarize the experimental and clinical evidence supporting melatonin’s therapeutic potential in conditions in which LP plays a central role. Its ability to cross the blood–brain barrier and its synthesis in multiple tissues, combined with its low toxicity and minimal side effects, make it a promising therapeutic candidate. Additionally, melatonin modulates mitochondrial function and membrane fluidity, offering additional protection against oxidative stress. This positions melatonin not just as a passive antioxidant, but as an active therapeutic agent against oxidative damage. We advocate for deeper exploration of melatonin-based therapies in LP-driven diseases, proposing it as a multifunctional molecule with significant clinical value.
Keywords
- melatonin/metabolism
- melatonin/pharmacology
- lipid peroxidation/physiology
- antioxidants/pharmacology
- reactive oxygen species/metabolism
- oxidative stress
Melatonin, a pleiotropic indoleamine, plays a crucial role in maintaining redox homeostasis by directly scavenging reactive oxygen species (ROS) and enhancing endogenous antioxidant defenses. Beyond its well-established circadian function, melatonin regulates oxidative stress, inflammation, apoptosis, and autophagy, positioning it as a promising therapeutic candidate in various pathophysiological contexts, including cancer, neurodegenerative disorders, cardiovascular disease, and aging [1, 2].
One of the key processes influenced by melatonin is lipid peroxidation (LP), an oxidative chain reaction triggered by ROS that targets unsaturated fatty acids in cellular membranes, producing reactive aldehydes such as malondialdehyde (MDA) and 4-hydroxynonenal (4-HNE) [3, 4]. While excessive LP compromises membrane integrity and mitochondrial function, contributing to genomic instability and inflammation, regulated LP can act as a signaling mechanism, modulating adaptive pathways such as apoptosis and ferroptosis [5, 6].
Melatonin exerts protective effects by neutralizing ROS, reducing the formation of cytotoxic aldehydes, and preventing protein and DNA adducts that disrupt cellular homeostasis [7]. Importantly, it not only suppresses harmful LP but also fine-tunes redox-dependent signaling, preserving the physiological functions of lipid-derived mediators. This dual action distinguishes melatonin from traditional antioxidants, which may blunt essential oxidative signaling.
In cancer, where LP has a paradoxical role—promoting both tumor progression and ferroptotic cell death—melatonin emerges as a modulator capable of tipping the balance toward protective outcomes. By restoring redox balance and regulating ferroptosis, melatonin offers a unique therapeutic avenue for oxidative stress–related diseases [8, 9].
Melatonin (N-acetyl-5-methoxytryptamine) is an endogenous indoleamine synthesized in a circadian manner in the pineal gland and in a non-circadian manner in many other cells [10]. In the pineal, its secretion is tightly regulated by the suprachiasmatic nucleus and modulated by the light/dark cycle. Peripheral tissues where melatonin synthesis has been documented include the gastrointestinal tract, adrenal cortex, retina, immune cells, and others. The presence of melatonin synthesis in multiple tissues underscores its broad involvement in autocrine and paracrine signaling pathways [11, 12]. More recently, compelling evidence has demonstrated that mitochondria represent the primary intracellular source of melatonin in many tissues [13, 14]. Unlike pineal synthesis, which depends on photoperiod regulation, mitochondrial melatonin production responds directly to metabolic demands, bioenergetic status, and the cellular redox environment, acting as a relevant local defense mechanism, complementary to other antioxidant systems [9, 13, 15].
The biosynthetic pathway of melatonin begins with the essential amino acid tryptophan, which is converted into 5-hydroxytryptophan by the enzyme tryptophan hydroxylase (TPH), followed by decarboxylation into serotonin by aromatic L-amino acid decarboxylase (AADC). The rate-limiting step occurs through the activity of serotonin N-acetyltransferase (SNAT), also known as arylalkylamine N-acetyltransferase (AANAT), which catalyzes the conversion of serotonin into N-acetylserotonin. Subsequently, the enzyme hydroxyindole-O-methyltransferase (HIOMT)—currently designated as acetylserotonin-O-methyltransferase (ASMT)—methylates N-acetylserotonin to produce melatonin [9, 13, 15]. This biosynthesis depends on the availability of serotonin and cofactors such as zinc, an essential element that also participates in antioxidant defense, underscoring that melatonin production is interconnected with broader metabolic and redox pathways.
Although widely recognized for its pivotal role in circadian rhythm regulation, melatonin exerts effects that extend beyond chronobiology. It is a multifunctional molecule that contributes to cellular homeostasis, protection against oxidative stress, and modulates physiological and pathological processes, acting in synergy with classical antioxidant mechanisms. From an antioxidant perspective, melatonin exerts a dual mechanism of action: it directly scavenges reactive oxygen species (ROS) and reactive nitrogen species (RNS), and it also induces the expression of endogenous antioxidant enzymes [16, 17]. Melatonin also inhibits lipid peroxidation (LP), stimulates antioxidant enzymes, and reduces metal toxicity; it stabilizes mitochondrial activity and suppresses inflammatory signaling [18]. Moreover, melatonin and its metabolites act collectively to provide versatile antioxidant protection: their combined presence amplifies the neutralization of ROS, facilitates metal chelation, radical adduct formation, and repair of oxidatively damaged biomolecules, positioning the melatonin family as a natural defense system against oxidative stress [19].
Its lipophilic nature facilitates diffusion across membranes, allowing melatonin to neutralize free radicals in mitochondria and other cellular compartments, thereby reducing lipid peroxidation, preventing DNA damage, and preserving mitochondrial integrity [9]. In addition to this direct antioxidant action, melatonin modulates gene expression of crucial antioxidant enzymes such as cytosolic Cu/Zn-superoxide dismutase (SOD1), mitochondrial MnSOD (SOD2), glutathione peroxidase (GPx), and catalase (CAT). These enzymes act in concert to detoxify ROS: SODs catalyze the dismutation of superoxide anions into hydrogen peroxide, which is subsequently degraded into water and oxygen by GPx and CAT [16, 20]. By influencing these pathways, melatonin complements and amplifies the activity of the endogenous antioxidant network, rather than acting as an isolated regulator.
At physiological concentrations, melatonin predominantly exerts its antioxidant activity via gene regulation, whereas pharmacological levels potentiate its direct radical-scavenging effects. Recent studies reinforce that, beyond its recognized antioxidant activity, melatonin helps to preserve the structural integrity of cellular components, particularly membranes, mitochondria, and DNA [17, 20, 21]. A key example of this property is illustrated by the “antioxidant cascade” of melatonin, in which the indoleamine not only scavenges hydroxyl and peroxyl radicals to prevent the initiation and propagation of LP, but also generates metabolites such as cyclic 3-hydroxymelatonin (C3OHM), N1-acetyl-N2-formyl-5-methoxykynuramine (AFMK), and N1-acetyl-5-methoxykynuramine (AMK). These products retain significant radical-scavenging activity, thereby extending melatonin’s protective role against oxidative damage through successive reactions [22]. As shown in Fig. 1 (Ref. [10]), this cascade interrupts LP chain reactions and neutralizes reactive intermediates, highlighting melatonin’s unique advantage over classical antioxidants [10].
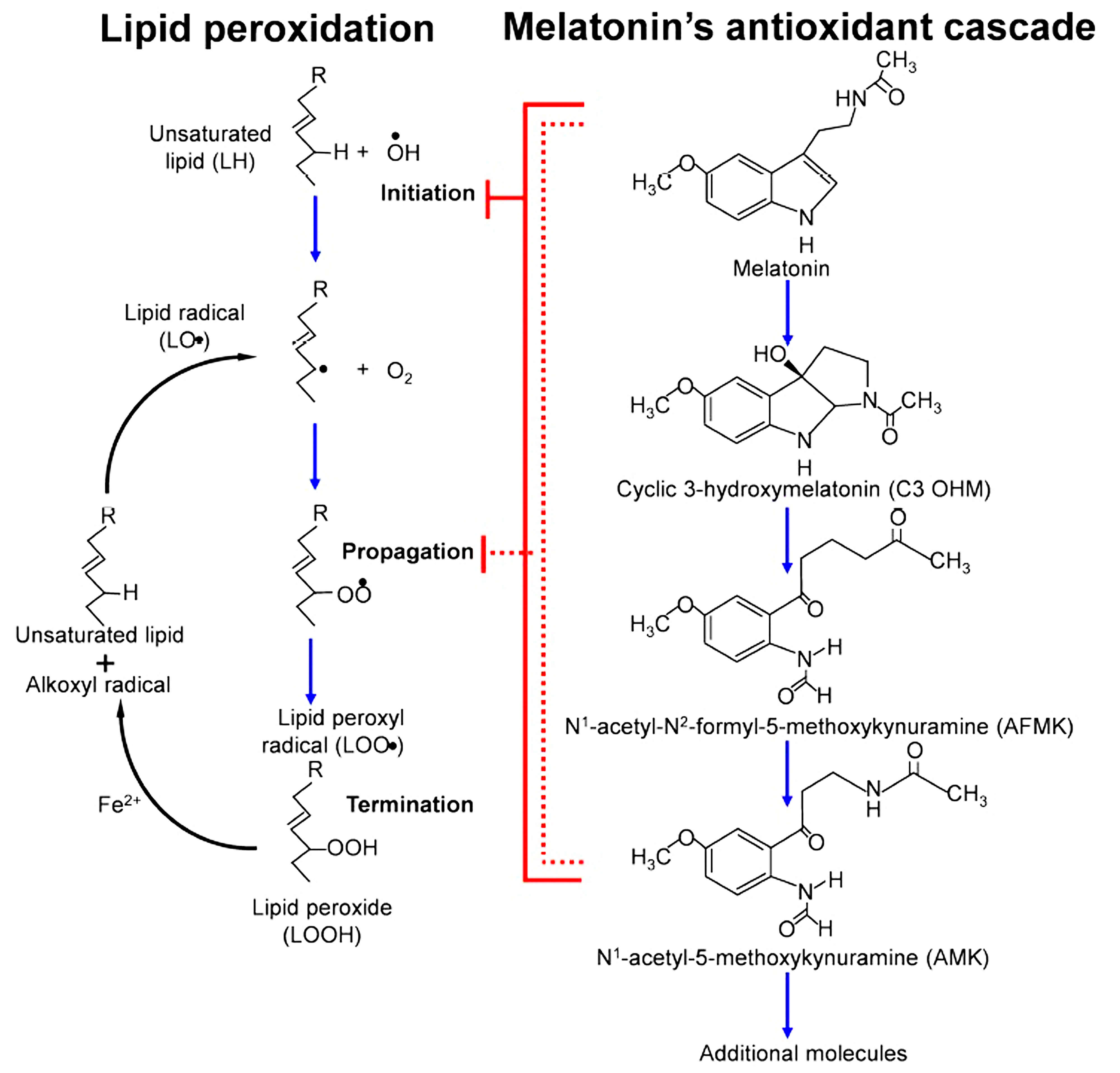 Fig. 1.
Fig. 1.
Mechanism of lipid peroxidation (LP) and melatonin’s antioxidant cascade. On the left, the initiation, propagation, and termination steps of LP are shown, leading to the generation of lipid radicals and lipid hydroperoxides (LOOH). On the right, melatonin’s antioxidant cascade is represented, where melatonin not only directly neutralizes reactive oxygen and nitrogen species (ROS/RNS) but also generates bioactive metabolites such as cyclic-3-hydroxymelatonin (C3OHM), N1-acetyl-N2-formyl-5-methoxykynuramine (AFMK), and N1-acetyl-5-methoxykynuramine (AMK). These metabolites further contribute to the suppression of oxidative stress, amplifying melatonin’s protective role. Illustration adapted with permission from Reiter et al. [10], Dual sources of melatonin and evidence for different primary functions; published by Frontiers Media S.A., 2024.
Its ability to contribute to the limitation of LP helps maintain membrane fluidity and selective permeability, which are essential for cell function and signaling. At the mitochondrial level, melatonin cooperates in preventing dysfunction by mitigating ROS accumulation, thereby preserving the integrity of the respiratory chain and ATP synthesis efficiency. Furthermore, it supports genomic stability by protecting both nuclear and mitochondrial DNA from oxidative damage—either by directly scavenging radicals or by enhancing DNA repair mechanisms [20]. These protective mechanisms are fundamental for maintaining cellular homeostasis, particularly under conditions of oxidative stress, and have been associated with melatonin’s protective effects on aging, neurodegeneration, and cancer [9, 21].
Rather than functioning alone, melatonin interacts with multiple signaling and defense pathways, exerting modulatory actions across apoptosis, immunoregulation, and mitochondrial processes. It can exert anti-apoptotic effects in normal tissues, while promoting apoptosis in malignant cells, highlighting its context-dependent role [23]. In the hematopoietic system, for example, melatonin protects progenitor cells from chemotherapy-induced apoptosis by stimulating Th2 cells to release IL-4, which in turn activates stromal cells to produce GM-CSF, thereby promoting cellular regeneration. In tumor cells, such as MCF-7, melatonin induces cell cycle arrest via the p53/p21WAF1 pathway, balancing mitosis and apoptosis—demonstrating its dual role as a cytoprotective agent in normal cells and an antiproliferative factor in cancer cells [23]. Additionally, Florido et al. (2022) [24] detailed the mechanisms through which melatonin induces reactive ROS production in cancer cells, including interactions with calmodulin to activate iPLA2, inhibition of the AKT pathway leading to NRF2 degradation, modulation of mitochondrial sirtuin 3 (SIRT3), and stimulation of the mitochondrial respiratory chain via reverse electron transport. By increasing ROS while reducing antioxidant defenses, melatonin selectively promotes apoptosis in tumor cells, highlighting its potential as a complementary anticancer therapy.
Beyond its antioxidative and apoptotic regulation, melatonin contributes to
immunomodulation. It regulates the function of diverse immune cells, including T
and B lymphocytes, macrophages, neutrophils, and dendritic cells [25]. This
indoleamine finely adjusts the balance between pro-inflammatory and
anti-inflammatory cytokines, being capable of either enhancing immune responses
against pathogens or suppressing chronic inflammation [26]. Mechanistically, this
occurs through modulation of key intracellular signaling pathways such as NRF2,
NF-
A further aspect of melatonin’s biological role is its ability to regulate apoptosis—a key process for maintenance of tissue integrity and the elimination of damaged or potentially malignant cells. Melatonin promotes apoptosis in tumor cells by modulating key regulatory proteins such as Bax, Bcl-2, caspases, and p53, while also affecting mitochondrial membrane permeability and triggering cytochrome c release [9, 14]. Likewise, melatonin exerts anti-apoptotic and cytoprotective effects in normal cells subjected to oxidative or toxic stress, preserving cell viability and preventing irreversible damage [28]. This duality emphasizes its complementary value within endogenous defense mechanisms, particularly in conditions of oxidative stress.
Adding to its multifaceted role, recent studies have highlighted melatonin’s capacity to regulate mitochondrial biogenesis and function. By enhancing mitochondrial efficiency and reducing mitochondrial ROS production, melatonin further strengthens its antioxidant and cytoprotective effects [9, 29]. Therefore, melatonin emerges as an important component of the antioxidant and regulatory network, acting alongside classical enzymatic defenses and trace elements such as zinc.
In summary, melatonin is a versatile molecule, endowed with a broad spectrum of bioactive properties that contribute to maintaining homeostasis and protecting against physiopathological insults, in concert with other endogenous mechanisms. Its therapeutic potential has been extensively explored in various medical fields, with significant relevance in the treatment of neurodegenerative diseases, cancer, cardiovascular disorders, sleep disturbances, and chronic inflammatory conditions. This underscores the importance of advancing research focused on elucidating its molecular mechanisms and expanding its clinical applications [20].
Based on extensive experimental evidence, melatonin is considered a highly promising natural modulator of LP, a critical process mediated by ROS/RNS that compromises membrane integrity and generates toxic byproducts such as MDA and 4-HNE (Fig. 2). Melatonin exerts its antioxidant effect both directly, by neutralizing radicals such as •OH, ROO•, and ONOO⁻, and indirectly, by enhancing the expression and activity of SOD, CAT, GPx, while inhibiting pro-oxidant enzymes such as NADPH oxidase (NOX) and iNOS. Its metabolites, including AFMK and AMK, retain significant antioxidant capacity which sometimes exceeds that of melatonin, providing a cascading protective effect against LP progression. Furthermore, melatonin attenuates ferroptosis by modulating ROS production and iron metabolism.
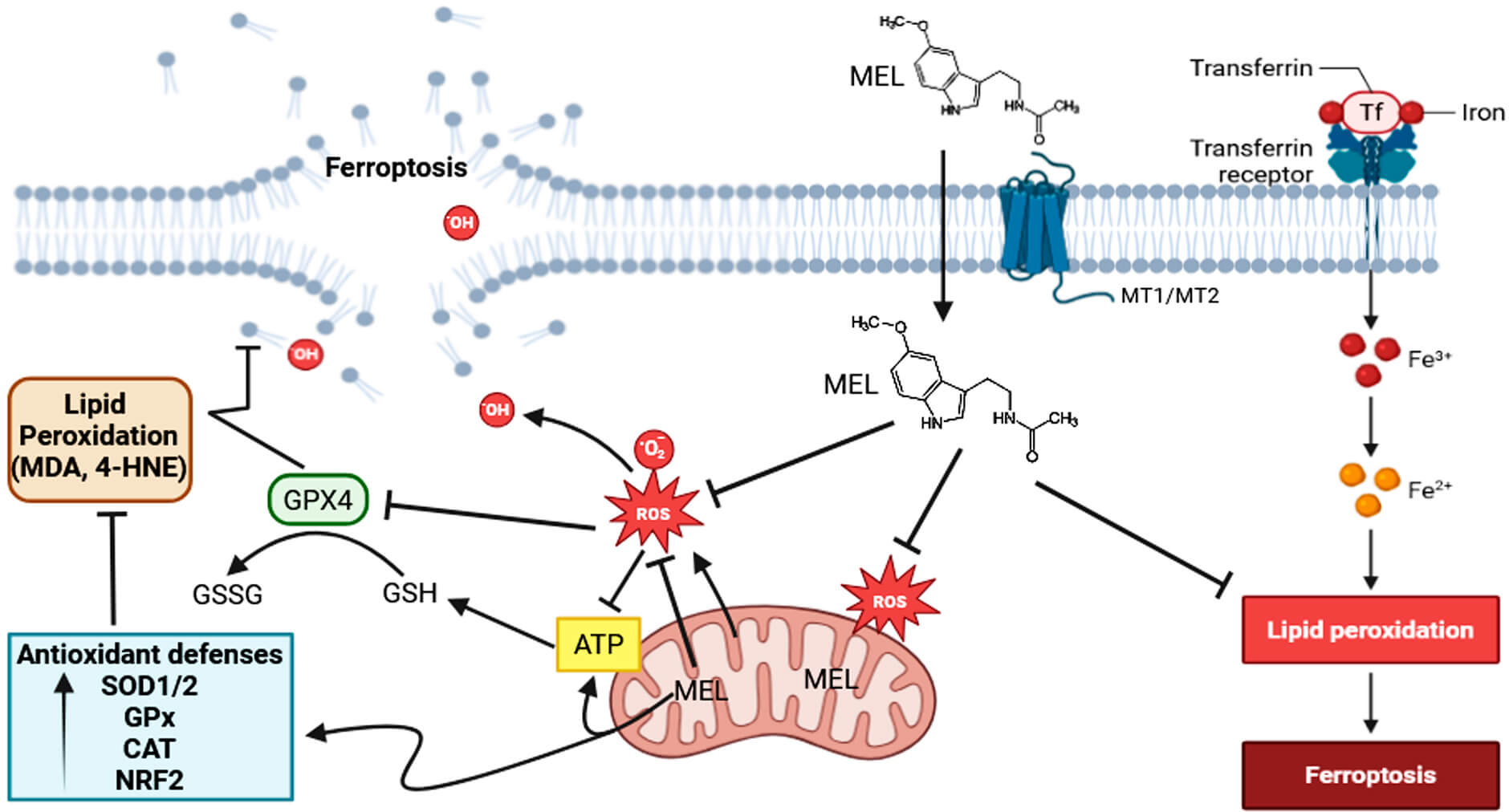 Fig. 2.
Fig. 2.
Melatonin attenuates lipid peroxidation and ferroptosis through multiple antioxidant and regulatory mechanisms. Melatonin (MEL) exerts protective effects against ferroptosis by modulating reactive oxygen species (ROS) production and iron metabolism. Upon entering the cell via membrane receptors (MT1/MT2), melatonin directly scavenges ROS, including hydroxyl radicals (•OH) and superoxide anions (O2•⁻), reducing oxidative stress and mitochondrial dysfunction. MEL also enhances mitochondrial ATP production and maintains redox homeostasis. Through upregulation of antioxidant defense systems—including superoxide dismutase (SOD1/2), glutathione peroxidase (GPx), catalase (CAT), and the NRF2 signaling pathway—melatonin promotes the attenuation of lipid peroxidation. GPX4 reduces lipid hydroperoxides, thereby inhibiting the accumulation of toxic lipid peroxidation products (e.g., MDA, 4-HNE) and preventing ferroptotic cell death. Additionally, melatonin interferes with iron metabolism by modulating transferrin receptor expression and iron uptake (Fe3+/Fe2+), thus limiting the Fenton reaction and downstream lipid peroxidation. Collectively, these actions highlight melatonin’s role in maintaining membrane integrity and preventing iron-dependent lipid damage and ferroptosis. GPX4, glutathione peroxidase 4; MDA, malondialdehyde; 4-HNE, 4-hydroxynonenal; NRF2, nuclear factor erythroid 2–related factor 2; GSH, reduced glutathione; GSSG, oxidized glutathione; ATP, adenosine triphosphate. The Fig. 2 was created in BioRender.com. Chuffa, L. (2025) https://BioRender.com/fd436c4.
Upon entering the cell via PEPT ½ oligopeptide transporters, GLUT4 receptor and/or diffusion, melatonin scavenges ROS, enhances mitochondrial ATP production, maintains redox homeostasis, and promotes antioxidant defense via SOD1/2, GPx, CAT, and NRF2 signaling. GPX4 reduces lipid hydroperoxides, preventing accumulation of toxic LP products and ferroptotic cell death. Melatonin also regulates transferrin receptor expression and iron uptake (Fe3+/Fe2+), limiting the Fenton reaction and subsequent LP, thereby maintaining membrane integrity and protecting against iron-dependent oxidative damage.
LP can trigger regulated cell death pathways, particularly apoptosis and ferroptosis, depending on context, location, and intensity. During apoptosis, LP products like 4-HNE modify regulatory proteins such as caspases and members of the Bcl-2 family and facilitate cytochrome c release from mitochondria [30, 31]. Cardiolipin, a highly unsaturated mitochondrial phospholipid, is especially sensitive to LP; its oxidation promotes Bax translocation and mitochondrial permeabilization, initiating intrinsic apoptosis [32]. LP can also act as a resolution signal, promoting non-inflammatory clearance of damaged cells, which is essential for tissue remodeling, embryogenesis, and immune responses [12]. When deregulated, LP leads to pathological cell death such as ferroptosis, characterized by iron-dependent accumulation of lipid peroxides and GPX4 failure [33]. Melatonin counteracts ferroptosis by reducing iron uptake, scavenging ROS, and reinforcing GPX4 activity, serving as a crucial regulator of ferroptotic sensitivity in cancer and degenerative diseases [34].
Mitochondria are both sources and targets of ROS, and their imbalance triggers
oxidative stress, apoptosis, necrosis, and chronic inflammation [35, 36].
Melatonin acts as a central protector against LP, both directly and indirectly.
Its high mitochondrial concentration (~100
In HepG2 liver cancer cells, melatonin increases ROS and triggers mitochondrial dysfunction and cell death, while protecting neighboring healthy cells [41]. Similarly, Florido and co-workers [24] demonstrated that specific concentrations of melatonin can generate ROS in cancer cells, promoting programmed cell death and enhancing chemotherapy sensitivity, without affecting normal tissues. Chok et al. [42] further showed that melatonin induces ROS-mediated autophagy in colorectal cancer cells, causing endoplasmic reticulum stress and selective tumor cell death. These findings indicate that melatonin acts as a context-dependent redox modulator, preserving normal cell integrity while exploiting the oxidative vulnerability of cancer cells, supporting its potential as an adjuvant therapeutic agent in oncology.
In addition to being synthesized locally in the mitochondria, melatonin is an amphiphilic molecule capable of freely crossing biological membranes, allowing its rapid and efficient distribution to different cellular compartments, including the nucleus, mitochondria, and plasma membranes [9]. Melatonin released into the blood is metabolized through different pathways. In the liver, it undergoes enzymatic metabolism predominantly via CYP1A2, producing 6-hydroxymelatonin, which is subsequently conjugated [9]. However, at the tissue level and especially in the mitochondrial microenvironment, melatonin is also metabolized through non-enzymatic pathways that occur in direct response to its interaction with ROS and RNS [13, 15]. While melatonin possesses a notable ability to neutralize multiple reactive species, it functions alongside other endogenous and exogenous antioxidants, such as glutathione and vitamins C and E, contributing to a coordinated defense against oxidative stress [9, 15]. A distinctive feature of melatonin, compared to some classical antioxidants, lies in its ability to perform sequential free radical scavenging. The metabolites generated during detoxification—such as AMK and cyclic 3-hydroxymelatonin—retain antioxidant activity, often exceeding that of native melatonin [13, 15]. This cascade establishes melatonin as a central player in preserving cellular and mitochondrial integrity, particularly in high-ROS tissues, including tumor cells.
Beyond its direct radical scavenging activity, melatonin operates through a unique cascade defense system that plays a key role in preserving the integrity of cellular membranes, protecting nuclear and mitochondrial DNA, and maintaining mitochondrial homeostasis [14]. This property is especially relevant in tissues highly dependent on oxidative metabolism, such as the brain, heart, liver, and also in stressed cells, including tumor cells, where redox modulation plays a critical role in survival, proliferation, and progression [9, 13].
Tanabe and colleagues (2015) [17] demonstrated that melatonin protects granulosa
cells in ovarian follicles by crossing lipophilic membranes, reducing LP,
preventing DNA damage, and maintaining mitochondrial integrity. Such protection
includes the regulation of mitochondrial function, preservation of
steroidogenesis, and inhibition of the activation of apoptotic enzymes caspases
3/7. In addition to its direct antioxidant functions, Mayo et al. (2002)
[16] showed that, at physiological concentrations, melatonin acts indirectly by
hormonal modulation of the expression of antioxidant enzymes, increasing mRNA
levels of Cu-ZnSOD (cytoplasmic), MnSOD (mitochondrial), and GPx even in the
absence of oxidative stress. This regulation involves receptor-dependent protein
synthesis and modulation of mRNA stability, as well as interference with
transcription factors such as AP-1, NF-
This dual antioxidant role has significant implications in aging, neurodegenerative diseases, and the maintenance of cellular function, especially in tissues exposed to high levels of ROS. In summary, melatonin stands out as a multifunctional molecule capable of preserving cellular, mitochondrial, and genomic integrity, modulating both the immediate response and adaptive antioxidant mechanisms, consolidating its role as a natural regulator of oxidative stress.
Dysregulated LP underlies ferroptosis, a regulated necrosis-like death
characterized by the lethal accumulation of iron-dependent lipid peroxides and
impaired glutathione peroxidase 4 (GPX4) function [33]. Ferroptosis integrates
iron metabolism, PUFA-rich phospholipid oxidation, and antioxidant defenses,
serving as a double-edged sword in cancer biology. Iron promotes ferroptosis via
the Fenton reaction, generating ROS that oxidize polyunsaturated fatty acids
(PUFAs) into cytotoxic aldehydes such as 4-HNE, MDA, and acrolein [43, 44]. These
adducts modify mitochondrial proteins, deplete reduced glutathione (GSH), inhibit
SIRT3, and activate oncogenic pathways contributing to tumor growth and
angiogenesis [44]. LP can also influence tumor immunity: it enhances CD8+ T
cell activity via IFN-
As illustrated in Fig. 2, melatonin counteracts ferroptosis by reducing iron uptake, scavenging ROS, and reinforcing GPX4 activity, thereby confirming it as a crucial regulator of ferroptotic sensitivity in cancer and degenerative diseases [34]. Ferroptosis arises when cellular homeostasis is disturbed, either by heightened susceptibility to ferroptotic signals or by impairment of the antioxidant defense machinery. As shown in Fig. 2, melatonin counteracts ferroptosis by reducing iron uptake, scavenging ROS, and reinforcing GPX4 activity, thereby suggesting that it is a crucial regulator of ferroptotic sensitivity in cancer and degenerative diseases [34]. Melatonin has emerged as a candidate for mitigating iron overload and restoring iron homeostasis. Its protective role involves both direct iron chelation and regulation of iron metabolism, limiting Fenton reaction and attenuating oxidative stress linked to free iron ions. It upregulates hepcidin in hepatocytes via MT1 activation [47], decreases the expression of iron importers (TFR1, DMT1), and enhances iron export in the mouse hippocampus by upregulating ferroportin via MT2–NRF2 signaling, thereby counteracting memory loss induced by sleep deprivation [48]. It also promotes ferritin expression, particularly FTH, through MT2 activation in neuroinflammation and ferroptosis following brain injury [49], while inhibiting ferritinophagy by downregulating NCOA4 in age-related cataract [50]. Considering the involvement of LP and ferroptosis, evidence suggests that melatonin interferes with this process by modulating enzymes linked to PUFA-PL synthesis (e.g., ACSL4, LPCAT3) and by limiting both enzymatic and non-enzymatic LP; the latter was already demonstrated in rat kidney, brain, and liver. As an antioxidant, it preserves long-chain PUFAs such as DHA and AA, while also suppressing pro-ferroptotic enzymes like 12-LOX and 5-LOX [51, 52, 53]. Exogenous melatonin has consistently shown protective efficacy against ferroptosis in numerous cell and animal models, although clinical validation remains necessary. Importantly, melatonin exerts beneficial effects across a wide spectrum of noncancerous conditions, mitigating ferroptosis-related injury in the eyes, brain, cardiovascular system, lungs, liver, kidneys, and bones. The underlying mechanisms, effective dosages, experimental models, and disease-specific pathologies are comprehensively addressed in the study presented by Zhang et al. (2023) [52].
A recent review provided evidence that melatonin counteracts ferroptosis by
modulating multiple protective signaling cascades, including SIRT6/p-NRF2,
NRF2/ARE/HO-1/SLC7A11/GPX4/PTGS2, ERK/NRF2, ferroportin (FPN), Hippo/YAP,
PI3K/AKT/mTOR, and SIRT6/NCOA4/FTH1 pathways, thereby preserving redox balance,
iron homeostasis, and cell survival in diverse disease contexts [54]. While
melatonin is widely recognized for its protective role against ferroptosis in
noncancerous diseases affecting the eyes, brain, heart, lungs, liver, kidneys,
and bones, high pharmacological doses can exert the opposite effect in cancer.
Melatonin’s ability to act as either an antioxidant or a pro-oxidant depends on
the administered dose, the cellular context, and the length of exposure. By
enhancing ROS generation, promoting LP, elevating intracellular iron through
ferritin degradation and TFR1 upregulation, and mediating pathways such as
AKT/GSK-3
Melatonin preserves the fluidity and selectivity of cell membranes by reducing LP, preventing pore formation and functional alterations that would lead to cell dysfunction, inflammation, and cell death [17, 22]. In the mitochondrial environment, melatonin preserves respiratory function and prevents the opening of the mitochondrial permeability transition pore (mPTP), which, when activated by LP or Ca2+ overload, promotes the activation of the intrinsic apoptosis pathway [55]. Mitochondrial melatonin production may be upregulated by oxidative stress, independently of the light-dark cycle. Mitochondrial melatonin acts in an autocrine manner, reinforcing in situ antioxidant defense and reducing vulnerability to LP. In situations of SIRT3 dysfunction—a mitochondrial deacetylase that activates SOD2—such as in aging, obesity, or high-fat diets, melatonin administration restores SIRT3 activity, increasing antioxidant capacity and reducing lipid peroxide formation [35, 56].
As already pointed out, melatonin exhibits a dual and context-dependent redox
behavior, acting as an antioxidant in normal cells while displaying pro-oxidant
properties in certain malignant cells [24, 57]. This dualism is closely linked to
cellular metabolic state, mitochondrial function, and the regulation of
intracellular metal ions, which together dictate its functional outcome. In
normal cells, melatonin primarily scavenges reactive oxygen and nitrogen species,
reduces LP, and maintains mitochondrial membrane potential (
As shown in Fig. 3, melatonin protects cardiovascular tissues by reducing LP and preserving membrane integrity, thereby preventing ferroptosis-associated cardiomyocyte death. A comprehensive review by Maity et al. (2023) [61] highlighted melatonin’s protection against obesity- and diabetes-induced cardiovascular injury through multiple mechanisms: reducing oxidative stress and inflammation, inhibiting LP and cholesterol deposition, promoting anti-inflammatory macrophage polarization, and preserving cardiac integrity. In animal models of high-fat diet–induced metabolic stress, melatonin mitigates mitochondrial and endoplasmic reticulum stress, helping maintain cardiac and adipose tissue homeostasis. However, these protective effects are markedly compromised when SIRT1 expression is reduced, underscoring the dependence of melatonin’s redox-regulatory functions on intact SIRT1 signaling [62]. In models of drug-induced toxicity, such as doxorubicin-treated cardiomyocytes, melatonin reversed mitochondrial dysfunction, apoptosis, and ferroptosis marker alterations (ACSL4 and GPX4), emphasizing its role in suppressing oxidative stress-mediated cell death [63]. Mechanistically, melatonin also regulates the NRF2/SLC7A11/GPX4 axis, reducing LP and ferroptosis in macrophages exposed to oxidized LDL, and modulates inflammatory signaling and oxidative damage in acute injury models, such as heat stroke-induced myocardial injury [64]. Melatonin supplementation protected rat cardiac tissue from exercise-induced oxidative stress by reducing LP and protein oxidation, boosting the activity and expression of key antioxidant enzymes (e.g., CAT, GPx, SOD), and restoring redox homeostasis, particularly in the recovery phase after exhaustive exercise [65].
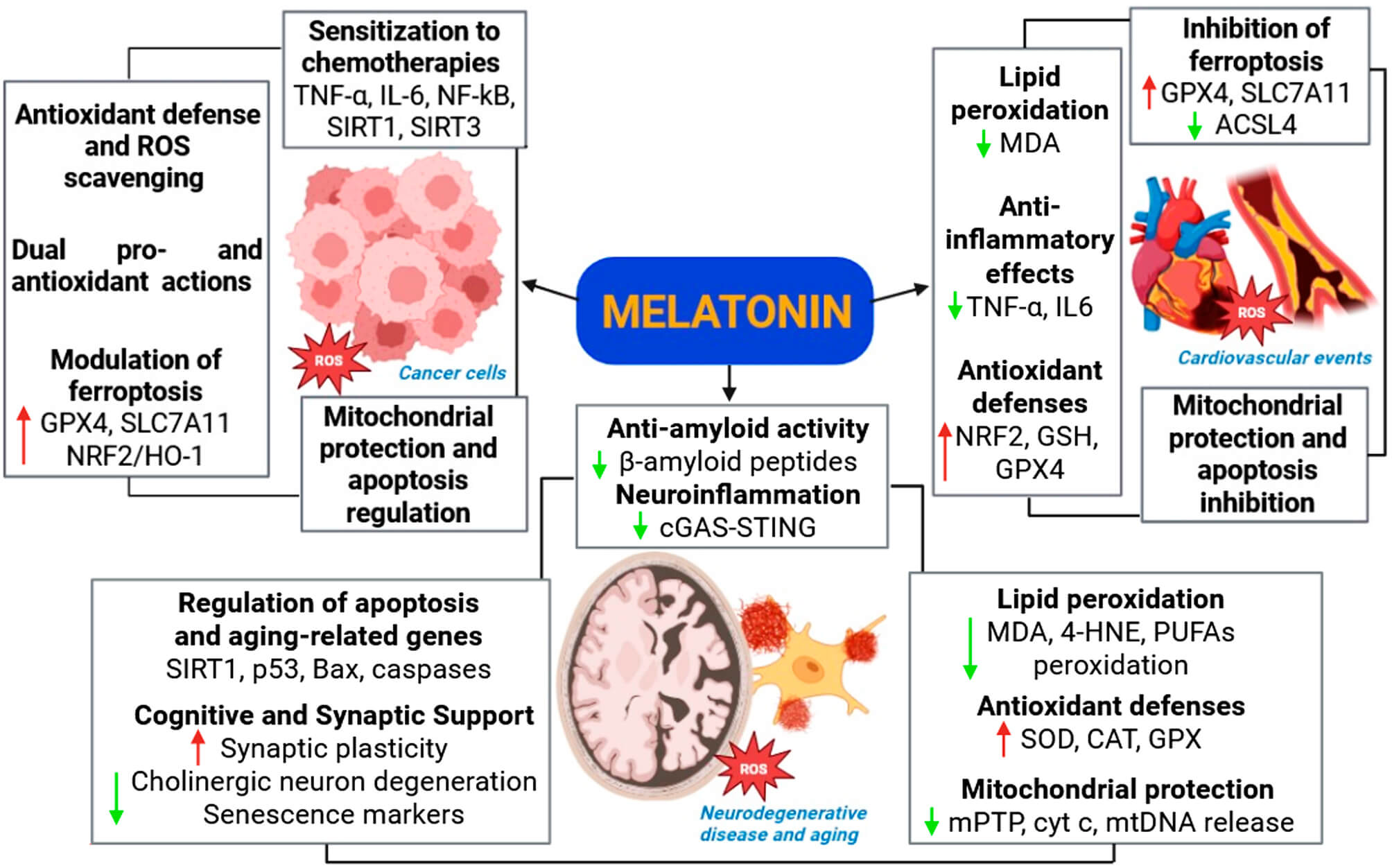 Fig. 3.
Fig. 3.
Multifaceted roles of melatonin in modulating lipid
peroxidation and oxidative stress across cancer, neurodegenerative diseases, and
cardiovascular events. Melatonin exerts antioxidant, anti-inflammatory, and
mitochondrial-protective effects in various pathological contexts. In cancer,
melatonin regulates antioxidant defenses, modulates ferroptosis, and sensitizes
cells to chemotherapy via regulation of pro-inflammatory and apoptotic pathways.
In neurodegenerative diseases and aging, melatonin reduces
In summary, melatonin exerts a dual functional mechanism: directly scavenging radicals and modulating gene expression and enzyme activity to maintain cellular integrity under oxidative stress. These mechanisms are critical in metabolically active tissues, including the brain, heart, and tumor cells, positioning melatonin as a versatile agent for mitigating LP-associated pathologies, including cancer, cardiovascular, and neurodegenerative diseases [20, 29, 56].
Aging is associated with the progressive production of ROS, predominantly generated in the mitochondria during aerobic respiration. This excess of free radicals is an important causal factor in functional cellular decline and the onset of neurodegenerative diseases [58, 66]. Among the main targets of oxidative stress are membrane lipids, especially in PUFAs which are abundant in neuronal membranes and in myelin. These changes alter neuronal physiology and promote neurotoxicity [67]. With aging, there is a reduction in membrane fluidity, especially in the mitochondria, as a result of the accumulation of oxidized lipids, which compromises bioenergetic function and favors mitochondrial dysfunction [58, 66]. This process affects the central nervous system particularly severely due to its high energy demand, elevated lipid content, and relatively limited antioxidant activity. The oxidation of neuronal lipids is now recognized as an early marker and potential promoter of neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease [13, 15].
Melatonin counteracts these processes by lowering iron overload, preventing excessive LP, and reinforcing neuronal antioxidant defenses (Fig. 3). Through these mechanisms, it helps preserve mitochondrial bioenergetics, maintains neuronal integrity, and protects cognitive function, highlighting its therapeutic potential in aging-related neurodegeneration. A recent review further documented that melatonin exerts strong antioxidant and anti-inflammatory effects by scavenging free radicals, limiting LP, and enhancing mitochondrial stability [18].
Melatonin, whose endogenous synthesis declines significantly with age [59] can prevent or attenuate LP of the few molecules capable of preventing and interrupting LP. Produced primarily in the mitochondrial matrix of neurons [68], melatonin acts both as a direct antioxidant, neutralizing reactive species [58], and as a regulator of the expression of antioxidant enzymes such as SOD, CAT, and GPx [13, 20]. When administered peripherally, its high lipophilicity facilitates penetration into cell and mitochondrial membranes, supporting protection of lipid bilayers against the initiation and propagation of LP [12]. Melatonin also preserves mitochondrial integrity by preventing the opening of the mPTP and the release of cytochrome c and mitochondrial DNA, events that activate apoptosis and inflammatory responses via the cGAS-STING pathway [68]. These mechanisms contribute to limiting chronic neuroinflammation characteristic of neurodegenerative diseases (Fig. 3).
It is important to highlight that LP by-products such as 4-HNE may not only
damage membranes but also form adducts with neuronal proteins, promoting the
formation of pathological aggregates such as
In summary, melatonin protects the brain against deleterious effects associated with LP, promoting membrane stability, mitochondrial integrity, and the inhibition of neuroinflammation [58]. Its multifunctional antioxidant, anti-inflammatory, and anti-amyloid actions position it as a relevant molecule in the prevention and treatment of aging-associated neurodegenerative diseases [13, 20, 57].
Melatonin therapy can be administered orally, by injection, topically, or in nanoencapsulated form. Various pharmaceutical formulations of melatonin have been explored with the aim of improving its bioavailability, stability, and therapeutic efficacy, especially in conditions related to oxidative stress. Oral administration is widely used, particularly in the treatment of sleep disorders and in antioxidant protocols, due to its practicality and safety. Clinical studies have shown that co-supplementation with melatonin, magnesium, and zinc improves sleep quality in elderly individuals, highlighting the role of micronutrients supporting melatonin synthesis and function [71]. However, this route presents limited bioavailability (9% to 33%) because of the first-pass hepatic metabolism [72, 73].
In experimental models, injectable administration—mainly via intraperitoneal or intravenous routes—has been used to achieve faster and higher plasma levels, being particularly effective in acute scenarios, such as in models of cerebral or hepatic ischemia-reperfusion [74]. Topical melatonin formulations, in turn, have shown efficacy in skin diseases and wound healing processes, benefiting from melatonin’s ability to cross the epidermal barrier and exert localized antioxidant and immunomodulatory effects [73]. More recent advances include the use of nanoencapsulated melatonin formulations, which enable controlled drug release, greater chemical stability, and preferential targeting to intracellular compartments, especially mitochondria. Nanoencapsulated melatonin has proven more effective in neutralizing reactive oxygen and nitrogen species, in addition to showing greater antitumor and neuroprotective activity, as evidenced by studies in experimental models [9, 73].
Several preclinical experimental models have demonstrated the efficacy of melatonin as an antioxidant agent and cellular protector against various pathophysiological conditions characterized by oxidative stress and LP. These studies cover hepatic, neurodegenerative, oncological, and metabolic models, strengthening the understanding of its molecular mechanisms and potential clinical applications (Table 1, Ref [66, 74, 75, 76, 77]).
| Experimental model | Mechanism of action related to lipid peroxidation | Melatonin dose | Peroxidation markers | Reference |
| Rats with CCl4-induced liver damage | Reduction in membrane fluidity, ↓ MDA/4-HDA (−93.4%), ↓ carbonylation and hepatic necrosis | 10 mg/kg (i.p.) | MDA, 4-HDA | Aranda et al., J Pineal Res. 2010 [76] |
| SAMP8 and SAMR1 mice (aging model) | Preservation of mitochondrial fluidity, ↓ cathepsin D, restoration of GSH/GSSG | 10 mg/kg/day | MDA, 4-HNE | García et al., Neurobiol Aging. 2011 [66] |
| Diabetic rabbits (alloxan-induced) | ↓ free radicals, ↑ GSH/GSSG, ↑ GPx, GR and GST; ↓ hydroxyl free radicals (HFR) | 1 mg/kg (i.p.) | HFR, GSH | Winiarska et al., J Pineal Res. 2006 [77] |
| Rats with cerebral ischemia-reperfusion | ↑ SOD, GPx, catalase and NRF2; ↓ MDA, ↓ infarct volume | 5 mg/kg (i.p.) | MDA | Sabbaghziarani et al., IBRO Neurosci Rep. 2024 [74] |
| HUVECs exposed to homocysteine | ↑ cell viability, ↓ TBARS (MDA), ↓ caspases, ↑ Bcl-2 | 10 µM (in vitro) | TBARS (MDA) | Aykutoglu et al., Mol Biol Rep. 2020 [75] |
| Endothelial cells treated with vitamin E or melatonin | Melatonin more effective in ↓ LPO and apoptosis than vitamin E | 10 µM melatonin vs. 50 µM vitamin E | TBARS, caspases, Bax/Bcl-2 | Aykutoglu et al., Mol Biol Rep. 2020 [75] |
| Rats treated with NAC vs. melatonin (induced diabetes) | Melatonin superior: ↓ hydroxyl radicals, ↑ GSH/GSSG, ↑ antioxidant enzymes | 1 mg/kg melatonin vs. 10 mg/kg NAC | HFR, GSH/GSSG, GPx, GR | Winiarska et al., J Pineal Res. 2006 [77] |
MDA, malondialdehyde; 4-HDA, 4-hydroxy-2-decenal; 4-HNE, 4-hydroxynonenal; GSH,
reduced glutathione; GSSG, oxidized glutathione; GPx, glutathione peroxidase; GR,
glutathione reductase; GST, glutathione S-transferase; SOD, superoxide dismutase;
NRF2, nuclear factor erythroid 2-related factor 2; TBARS, thiobarbituric acid
reactive substances; LPO, lipid peroxidation; NAC, N-acetylcysteine.
Aykutoglu et al. [75] (Table 1) demonstrated that endothelial cells (HUVECs) exposed to homocysteine showed increased cell viability, reduced lipid peroxidation (TBARS/MDA) levels, and lower caspase activation, associated with an upregulation of Bcl-2 expression. Moreover, treatment with melatonin (10 µM) was more effective than vitamin E (50 µM) in reducing LP and apoptosis, documenting the superior antioxidant and antiapoptotic potential of melatonin.
In the hepatic context, Aranda et al. (2010) [76] (Table 1) investigated the effect of melatonin in rats exposed to carbon tetrachloride (CCl4), a potent hepatotoxic agent that induces LP. Treatment with melatonin (10 mg/kg, intraperitoneally) promoted a significant reduction in MDA and 4-HDA levels, classic biomarkers of lipid damage. Furthermore, melatonin preserved the fluidity of hepatic cell membranes and prevented morphological alterations such as necrosis and cellular ballooning. Even at pharmacological doses, no side effects were observed, confirming its high safety profile. These findings demonstrate melatonin’s protective action on tissues highly susceptible to oxidative injury by xenobiotics.
In models of brain aging, García et al. (2011) [66] (Table 1) used mice from the SAMP8 (accelerated senescence) and SAMR1 (normal aging) strains to evaluate the chronic effects of melatonin (10 mg/kg/day). Treated animals showed significant improvement in mitochondrial membrane fluidity, reduction in LP levels, and lower expression of cathepsin D, a lysosomal protease associated with neuronal apoptosis. Restoration of the GSH/GSSG ratio was also observed, indicating recovery of redox homeostasis. These data suggest that melatonin exerts neuroprotective effects associated with preservation of mitochondrial function and attenuation of oxidative aging.
In metabolic disorders, especially in the context of experimental diabetes, Winiarska et al. (2006) [77] (Table 1) compared the efficacy of melatonin (1 mg/kg) to N-acetylcysteine (NAC, 10 mg/kg) in alloxan-induced diabetic rabbits. Melatonin proved more effective in reducing hydroxyl radicals, restoring the GSH/GSSG ratio, and activating antioxidant enzymes such as GPx, GR, and GST. On the other hand, NAC only promoted an increase in GSH levels without significantly impacting other oxidative stress markers. This highlights the broader and mitochondria-targeted action of melatonin, especially relevant for tissues with high redox disruption, such as those often affected in renal and hepatic contexts.
In the setting of acute neurological injury, Sabbaghziarani et al. (2024) [74] (Table 1) used a cerebral ischemia-reperfusion model in Wistar rats subjected to middle cerebral artery occlusion (MCAO). Administering melatonin (5 mg/kg), NAC (50 mg/kg), or a combination of the two revealed that melatonin alone induced the antioxidant NRF2 pathway, increased SOD, GPx, and catalase enzymes, and significantly reduced infarct volume. Although NAC showed antioxidant effects, its action was less robust and limited to increasing total glutathione. The combined treatment produced the best results, with marked reduction in lipid peroxidation (MDA), decreased brain damage, and improved functional recovery, suggesting therapeutic synergy.
These experimental models confirm that melatonin consistently acts as a modulator of LP, with direct and indirect effects on mitochondrial biogenesis, enzymatic antioxidant pathways, apoptosis, and cellular plasticity. Its ability to cross biological barriers, such as the blood–brain barrier, and its mitochondrial affinity confer significant advantages over classical antioxidants. These findings support the translational potential of melatonin as a therapeutic agent in clinical conditions marked by oxidative stress, such as hepatic diseases, neurodegenerative disorders, cancers, and metabolic disorders.
Despite the widespread use of melatonin as a sleep aid and therapeutic supplement, recent evidence identifies several likely concerns regarding its safety, efficacy, and regulatory oversight. Studies in pediatric populations have reported non-serious adverse effects such as somnolence, headache, and dizziness, emphasizing the need for careful dose management and monitoring in vulnerable groups [78, 79]. Analyses of commercially available supplements have revealed substantial discrepancies between labeled and actual melatonin content, with some products containing several times the declared dose, raising serious safety concerns [80]. Regulatory data from the CDC’s Morbidity and Mortality Weekly Report reinforce these findings, warning that unregulated melatonin products, especially those marketed for children, may pose health risks due to extreme variability in dosage [81]. Collectively, these studies underscore the importance of cautious clinical use, stringent quality control, and further research to establish safe and effective therapeutic protocols for melatonin administration.
LP is a central process in the onset and progression of numerous chronic diseases, including cancer, neurodegenerative disorders, and cardiovascular conditions. Melatonin emerges as a highly promising molecule in this context, due to its potent antioxidant properties and its capacity to modulate inflammatory and apoptotic pathways. Its amphiphilic nature and ability to cross cellular membranes and target mitochondria—organelles particularly susceptible to oxidative stress—further enhance its therapeutic potential.
Melatonin functions as a multifaceted antioxidant, neutralizing ROS, modulating antioxidant enzymes, and influencing inflammatory signaling, apoptosis, and autophagy. Importantly, it can restore redox balance without disrupting physiological processes mediated by LP, thus maintaining essential cellular signaling and homeostasis. The protective actions of melatonin and its metabolites, such as AFMK and AMK, suggest an efficient antioxidant cascade potentially surpassing the limitations of conventional antioxidants. Its excellent safety profile, including the ability to cross the blood–brain barrier, positions melatonin as an attractive candidate for managing neuroinflammatory, neurodegenerative, and oxidative-stress-associated disorders.
Although preclinical evidence strongly supports melatonin’s antioxidant and lipid-protective actions, clinical research remains limited and fragmented. To enhance its translational potential, future clinical studies should focus on large-scale, well-controlled randomized trials (RCTs) with standardized LP biomarkers (e.g., MDA, oxLDL (oxidized low-density lipoprotein), 4-HNE). Stratification by disease type (metabolic syndrome, oncology, neurodegeneration), route of administration (oral vs. targeted delivery), and dosing regimen will be critical. Biomarker-guided approaches could identify patient populations most likely to benefit, paving the way for personalized therapeutic strategies.
Table 2 (Ref. [82, 83, 84, 85, 86, 87]) summarizes key clinical studies that have investigated melatonin’s effects on sleep, neurodegeneration, cardiovascular risk, and oncology, highlighting dosing strategies, biomarkers, stratification criteria, and relevant clinical outcomes. This compilation illustrates the current state of clinical translation and supports the rationale for precision medicine approaches with melatonin.
| Study/Reference | Population/Context | Intervention/Dose/Duration | Biomarker(s) or stratification criteria | Key findings/Clinical outcomes |
| Optimizing the Time and Dose of Melatonin as a Sleep-Promoting Drug: A Systematic Review (2024) [82] | Individuals with insomnia or healthy volunteers | Multiple clinical trials; 0.5–10 mg; varying administration timing | Insomnia status, timing vs. desired sleep, dose | 4 mg/day optimal for reducing sleep latency; earlier administration (3 h before bedtime) enhances effect |
| Significant potential of melatonin therapy in Parkinson’s disease – meta-analysis (2023) [83] | Patients with Parkinson’s disease | Formulation type, dose, treatment duration | Improvement in motor symptoms (UPDRS) and sleep quality with | |
| Melatonin therapy to improve nocturnal sleep in critically ill patients (2008) [84] | Critically ill patients on mechanical ventilation | 10 mg at 21:00 for 4 nights via enteral tube | Pharmacokinetic profile, blood levels, tolerability | Increased nighttime sleep duration and efficiency; lower doses (1–2 mg) may prevent next-day effects |
| Melatonin levels in the Alzheimer’s disease continuum: a systematic review (2021) [85] | Patients across Alzheimer’s spectrum (preclinical to dementia) | Observational; melatonin measured in blood, CSF, saliva | Alzheimer’s stages (Braak), fluid type, correlation with neuropathology | Melatonin levels decrease with disease severity; inversely correlated with Braak stages; potential biomarker of progression |
| Rethinking Melatonin Dosing: Safety and Efficacy at Higher-than-Usual Levels in Aged Patients (2022/2023) [86] | Older adults with sleep disturbances and metabolic/cardiac comorbidities | High doses 40–200 mg/day long-term | Comorbidities, lab profiles, tolerability | Supraphysiological doses tolerated; improved metabolic and cardiovascular parameters; further safety studies needed |
| Clinical significance of serum melatonin in predicting the severity of oral squamous cell carcinoma (OSCC) (2020) [87] | Patients with OSCC vs. healthy controls | Observational; serum melatonin measured by ELISA | Tumor grade/size, invasion depth, nodal metastasis | Lower serum melatonin in cancer patients; effective for stratifying tumor stages |
Melatonin is clearly a multifunctional molecule with significant potential in the prevention and treatment of diseases associated with oxidative stress, chronic inflammation, and LP. Its antioxidant, anti-inflammatory, and anti-apoptotic properties, combined with its ability to target mitochondria and cross biological barriers, provide a compelling rationale for its therapeutic application in cancer, cardiovascular disorders, neurodegeneration, and metabolic diseases.
Preclinical evidence strongly supports melatonin’s role in modulating LP, preserving mitochondrial integrity, and regulating apoptotic and ferroptotic pathways. Moreover, its metabolites contribute to an efficient antioxidant cascade that may overcome limitations of conventional antioxidants. Clinical studies revealed the diversity of dosing strategies, patient populations, biomarker applications, and observed outcomes, highlighting the translational potential of melatonin.
Importantly, melatonin has an excellent safety profile, especially in adults, even at supraphysiological doses, making it a viable candidate for both adjunctive and standalone therapies. Future research should prioritize personalized approaches, considering dose, timing, route of administration, and patient stratification based on biomarkers and disease type. Integration with advanced delivery systems—such as nanoparticles, extracellular vesicles, or targeted carriers—may further enhance efficacy and adherence.
In conclusion, melatonin represents a promising molecule capable of bridging basic mechanistic insights and clinical applications, positioning it as a potential cornerstone of precision medicine approaches for oxidative-stress-related diseases. Future studies should focus on large-scale, biomarker-guided trials to establish robust clinical protocols and optimize therapeutic outcomes.
OAJV and DAPCZ were primarily responsible for the writing and critical revision of the manuscript, ensuring conceptual clarity and cohesion throughout the text. LGAC and RJR contributed substantially to the critical review and enhancement of visual elements, refining both the scientific content and its presentation. OAJV, DAPCZ, LGAC, and RJR contributed to the conceptualization. RJR served as the original mentor of the project, guiding its development from the initial stages with scientific insight and supervision. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors are grateful to the São Paulo Research Foundation (FAPESP) and the National Council for Scientific and Technological Development (CNPq).
The authors received financial support as follows: LGAC (CNPq Process number 306117/2023-1; FAPESP Process number 2021/12971-7).
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT in order to check spelling and grammar. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



