1 Department of Urology, The Affiliated TCM Hospital of Southwest Medical University, 646000 Luzhou, Sichuan, China
2 Department of Urology, Institute of Urology, West China Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
3 Department of Urology, West China School of Public Health and West China Fourth Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
4 Department of Urology, The Third People’s Hospital of Chengdu, Southwest Jiaotong University, 610043 Chengdu, Sichuan, China
†These authors contributed equally.
Abstract
Partial bladder outlet obstruction (pBOO) causes deposition of extracellular matrix (ECM), promotes bladder fibrosis, and decreases bladder compliance.
To investigate the effect of β-adrenoceptor (ADRB) on the ECM deposition of pBOO rat model and explore its underlying mechanism, human bladder smooth muscle cells (hBSMCs) were exposed to the pathological hydrostatic pressure (100 cm H2O) for 6 h, reverse transcription-polymerase chain reaction (RT-PCR) and western blotting were employed. Then the rats of sham operation and pBOO model were treated with vehicle or ADRB agonists for 3 weeks, and the alterations of the bladder were observed via Masson staining and immunohistochemical analysis.
100 cm H2O hydrostatic pressure significantly upregulated the expression of collagen I (COL1), collagen III (COL3) and fibronectin (FN), and downregulated the expression of ADRB2 and ADRB3 of hBSMCs at 6 h. The agonists of ADRB2 and ADRB3, Formoterol and BRL 37344, decreased COL1 and FN expression of hBSMCs under 100 cm H2O for 6 h compared with the cells exposed to hydrostatic pressure only. As the classic downstream pathways of ADRB, the EPAC pathway inhibited COL1 and FN expression of hBSMCs via regulating SMAD3 and SMAD2 activities, respectively. In pBOO rats, Procaterol (ADRB2 agonist), and Mirabegron (ADRB3 agonist) inhibited the formation of collagen and decreased the expression of FN and COL1 in the bladders of pBOO rats.
The bladder fibrosis of pBOO and deposition of hBSMCs ECM under hydrostatic pressure were regulated by ADRB2, and ADRB3 via EPAC/SMAD2/FN and EPAC/SMAD3/COL1 pathways, these findings pave an avenue for effective treatment of pBOO.
Keywords
- pathological hydrostatic pressure
- pBOO
- hBSMCs
- ECM
- β-adrenoceptor
Partial bladder outlet obstruction (pBOO) is a common symptom leading to the dysfunction of the bladder. pBOO decreases bladder compliance [1] accompanied by bladder wall hypertrophy [2], and hypercontractility [3]. Chapple CR et al. [4] found that 40–50% of patients with pBOO showed the storing urinary dysfunction symptom of urgency, urinary frequency and urge incontinence. With the development of pBOO, bladder fibrosis [5] and increased bladder pressure [6] would lower the bladder contractile ability and finally impair renal function [7]. Thus, the patients with pBOO have the symptoms of dysuria and urinary retention and usually accompany chronic renal failure during the decompensatory period.
In addition to the dysfunction of the bladder, the decrease in bladder compliance is associated with the deposition of the extracellular matrix (ECM)[8]. Previous studies found the expression of collagen increased in the bladder wall of a rat pBOO model [9, 10]. The accumulation of ECM containing collagen leads to progressive damage of the bladder to high intravesical pressure resulting from the bladder’s structural and functional changes by pBOO [11]. Collagen I (COL1) and collagen III (COL3) are the two predominant collagen types of bladders, and the ratio of COL1 and COL3 is approximately 3:1 [12]. Therefore, the COL1 dominates the collagen in the bladder wall and significantly influences the bladder detrusor. Besides the COL1, Wu J et al. [13] revealed that fibronectin (FN), an adhesive protein of ECM, is a key mediator of bladder fibrosis, and the high expression of FN and COL1 is associated with bladder dysfunction. In this study, we mainly detected the expression of collagen and FN in the bladder and human bladder smooth muscle cells (hBSMCs) under different conditions.
Currently, a large number of studies focus on studying the relationship between
In this study, we planned to illustrate the relationships between ADRBs and ECM deposition of rat bladder with pBOO in vivo and hBSMCs under pathological hydrostatic pressure in vitro, and explored its underlying mechanism.
Primary hBSMCs (Cat. No. 4310, ScienCell, San Diego, CA, USA) were cultured with special smooth muscle cell medium (SMCM, ScienCell), supplemented with 10% fetal bovine serum (FBS, Gibco, Thermo Fisher Scientific, Waltham, MA, USA), penicillin (100 U/mL) and streptomycin (100 µg/mL) in a humidified atmosphere with 5% CO2. Mycoplasma testing has been done for the hBSMCs in cell culture, and hBSMCs were identified by immunofluorescence. Two to six passages of primary hBSMCs were used for all experiments. Selective ADRB1 agonist, dobutamine hydrochloride (D0676), ADRB2 agonist, formoterol fumarate dihydrate (F9552), ADRB3 agonist, BRL 37344 (B169), ADRB3 antagonist SR 59230A (S8688), selective inhibitor of PKA-H89 dihydrochloride hydrate (B1427), specific inhibitor of EPAC ESI-09 (SML0814) were purchased from Sigma (St. Louis, MO, USA). Selective ADRB1 antagonist Metoprolol (ab120711) and ADRB2 antagonist ICI 118551 (ab120808) were obtained from Abcam (Pudong, Shanghai, China). Selective SMAD2 inhibitor LY2109761 (HY-12075) and SMAD3 inhibitor SIS3 (HY-13013) were purchased from MCE (Shanghai, China). The solvent of all the above drugs was Dimethyl sulfoxide (DMSO), so the control group was also treated with DMSO accordingly. ADRB2 agonist Procaterol was obtained from Otsuka (Guangdong, China), and ADRB3 agonist Mirabegron was purchased from Astellas (Tokyo, Japan). hBSMCs were preincubated for 60 min with ADRB agonist before the application of pressure or preincubated for 60 min with the selective ADRB antagonist, protein kinase A (PKA) or exchange protein directly activated (EPAC) inhibitor before the treatment of ADRB agonist.
hBSMCs cultured in 6-well plates at 80–90% confluence were exposed to hydrostatic pressure using a custom-designed motorized pressure apparatus, which was placed in a CO2 incubator with the adapted condition for cell growth [21]. hBSMCs in the CO2 incubator can be exposed to the hydrostatic pressure of 0–100 cm H2O. After pilot experiments, 100 cm H2O is the optimal condition for the treatment of hydrostatic pressure. hBSMCs in the control group were exposed to a static condition of 0 cm H2O. The pressure sensor in the chamber conducted the pressure data to the computer every hour and constant pressure can be obtained by regulating the inlet and outlet valves of the chamber. Moreover, the parameters of culture medium, such as PH, PCO2 and PO2 can be monitored by the Abbott i-stat 300 hand-held Blood Gas Analyzer (Abbott, Princeton, NJ, USA).
Total RNA was extracted using the RNeasy Mini Kit (Qiagen, Hilden, Germany). RNA was eluted in 40 µL nuclease-free water and stored at –80 °C. RNA concentration was determined by the Spectrophotometer (IMPLEN Nanophotometer, München, Germany). Then, cDNA was synthesized using Thermo Scientific Revert Aid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific). Real-time quantitative PCR was performed by using Bio-Rad CFX ManagerTM software version 3.1 (CFX96, Bio-Rad, Hercules, CA, USA) and the specific primers were listed in Table 1. The housekeeping gene GAPDH was used as an internal control. The polymerase chain reaction (PCR) reactions were performed using IQTM SYBR Green Supermix reagent (Bio-Rad) under the following conditions: 95 °C for 3min and 40 cycles of 95 °C for 10 s, and 55 °C for 30 s. The real-time data were analyzed by the 2-ΔΔCt method. Experiments were performed in triplicate.
| Gene | Primer sequence | |
| ADRB1 | ||
| Forward | ATCGAGACCCTGTGTGTCATT | |
| Reverse | GTAGAAGGAGACTACGGACGAG | |
| ADRB2 | ||
| Forward | GCCTGTGCTGATCTGGTCAT | |
| Reverse | AATGGAAGTCCAAAACTCGCA | |
| ADRB3 | ||
| Forward | GACCAACGTGTTCGTGACTTC | |
| Reverse | GCACAGGGTTTCGATGCTG | |
| COL1A1 | ||
| Forward | GAGGGCCAAGACGAAGACATC | |
| Reverse | CAGATCACGTCATCGCACAAC | |
| COL2A1 | ||
| Forward | TGGACGCCATGAAGGTTTTCT | |
| Reverse | TGGGAGCCAGATTGTCATCTC | |
| COL3A1 | ||
| Forward | GGAGCTGGCTACTTCTCGC | |
| Reverse | GGGAACATCCTCCTTCAACAG | |
| COL4A1 | ||
| Forward | GGGATGCTGTTGAAAGGTGAA | |
| Reverse | GGTGGTCCGGTAAATCCTGG | |
| FN1 | ||
| Forward | AGGAAGCCGAGGTTTTAACTG | |
| Reverse | AGGACGCTCATAAGTGTCACC | |
| GAPDH | ||
| Forward | ACAACTTTGGTATCGTGGAAGG | |
| Reverse | GCCATCACGCCACAGTTTC | |
PCR, polymerase chain reaction.
Membrane proteins were extracted as per the protocol of a special membrane
protein kit from Thermo Scientific (Men-PERTM Plus, Waltham, MA, USA,
89842). The treated cells were scraped off the petri dish and further cleaned
with cell wash solution. Next, the cells were incubated with permeabilization
buffer for 10 min at 4 °C and centrifuged for 15 min at 16,000 g.
Furthermore, the cells were incubated with solubilization buffer for 30 min at 4
°C and centrifuged for 15 min at 16,000 g. Finally, the solubilized
membrane and membrane-associated proteins were extracted in the supernatant. The
proteins were detected using primary antibodies against three subtypes of ADRBs.
Total proteins were extracted using the RIPA buffer (Biosharp, Hefei, China)
containing protease inhibitors and phosphatase inhibitors. Proteins (20 µg)
were separated by electrophoresis using a 10% sodium dodecyl
sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel (Bio-Rad) and
transferred onto polyvinylidene fluoride (PVDF) membranes. Then, the membranes
were blocked with 5% skimmed milk for 1 h and incubated with the primary
antibodies at 4 °C overnight followed by secondary anti-rabbit IgG or
anti-mouse IgG (MBL, Nagoya, Japan) for 1 h at RT. The primary antibodies used
for western blotting were as follows: ADRB1 (ab3442, Abcam, Cambridge, UK;
1:1000), ADRB2 (ab13989; Abcam; 1:1000), ADRB3 (ab76249; Abcam; 1:1000), SMAD2
(ab40855; Abcam; 1:2000), SMAD3 (ab40854; Abcam; 1:1000), p-SMAD2 (phospho S467;
ab53100; Abcam; 1:500); p-SMAD3 (phospho S423+S425; ab52903; Abcam; 1:2000);
Collagen I (ab34710; Abcam; 1:1000); Collagen III (ab7778; Abcam; 1:5000);
Fibronectin (ab2413; Abcam; 1:1000); anti-
A total of 24 eight weeks old female Sprague-Dawley rats (weight 200–250 g, purchased from Dasuo Experimental Animals Limited Company, Chengdu, China) were divided into four groups randomly: sham-operated group, pBOO group, pBOO+Procaterol group, pBOO+Mirabegron group. The steps of pBOO rat model are as flows: Rats were anesthetized with isoflurane (5% in oxygen). A urethral catheter (1 mm diameter) was placed through the urethra of anesthetized female rats, and then 3-0 silk was used to ligate around the proximal urethra to simulate pBOO, the catheter was carefully removed finally. The sham group underwent the same procedure except for urethral ligation. All rat groups were treated one day after the operation. The rats of the sham-operated group and pBOO group were intragastrically administered with normal saline, and rats of the BOO+Procaterol group or BOO+Mirabegron group were intragastrically administered with Procaterol at 0.01 mg/kg/d or Mirabegron at 5.0 mg/kg/d respectively (the dosage was determined based on the equivalent dose of clinical application). Then the bladder tissue was harvested after rats were sacrificed with CO2 3 weeks later.
The bladder tissue of rats was fixed with 4% paraformaldehyde, bedded in
paraffin, then cut into 4 µm sections. The sections were stained using
Masson’s trichromatic staining kit (Solarbio,
Beijing, China). The images of collagen accumulation of fibrotic lesions were
obtained at 100
The paraffin sections of bladder tissue were immersed in the distilled water, washed with PBS–T, and then blocked with 3% peroxide-methanol at room temperature (RT) for endogenous peroxidase ablation. Sections were incubated in primary antibody COL1 (ab34710; Abcam, Cambridge, UK; 1:100), FN (ab2413; Abcam; 1:50) respectively overnight at 4 °C. Then the sections were washed with PBS and incubated in rabbit IgG antibody-horseradish peroxidase complex (ab6721; Abcam; 1:10,000) for 30 min at RT, after which the sections were reacted with diaminobenzidine and were preserved for additional analysis.
The results were presented as means
To investigate whether the pathological hydrostatic pressure in vitro
stimulated the ECM expression of hBSMCs, cells were cultured in a hydrostatic
apparatus with sustained hydrostatic pressure of 40 cm H2O. The expression
of ECM genes including COL1, COL2, COL3, COL4, FN was detected. Firstly, COL2 and
COL4 expression was relatively low in hBSMCs, indicating these two proteins may
not the predominant components of hBSMC ECM.
Secondly, the pressure of 40 cm H2O
cannot upregulate the expression of hBSMC ECM (p
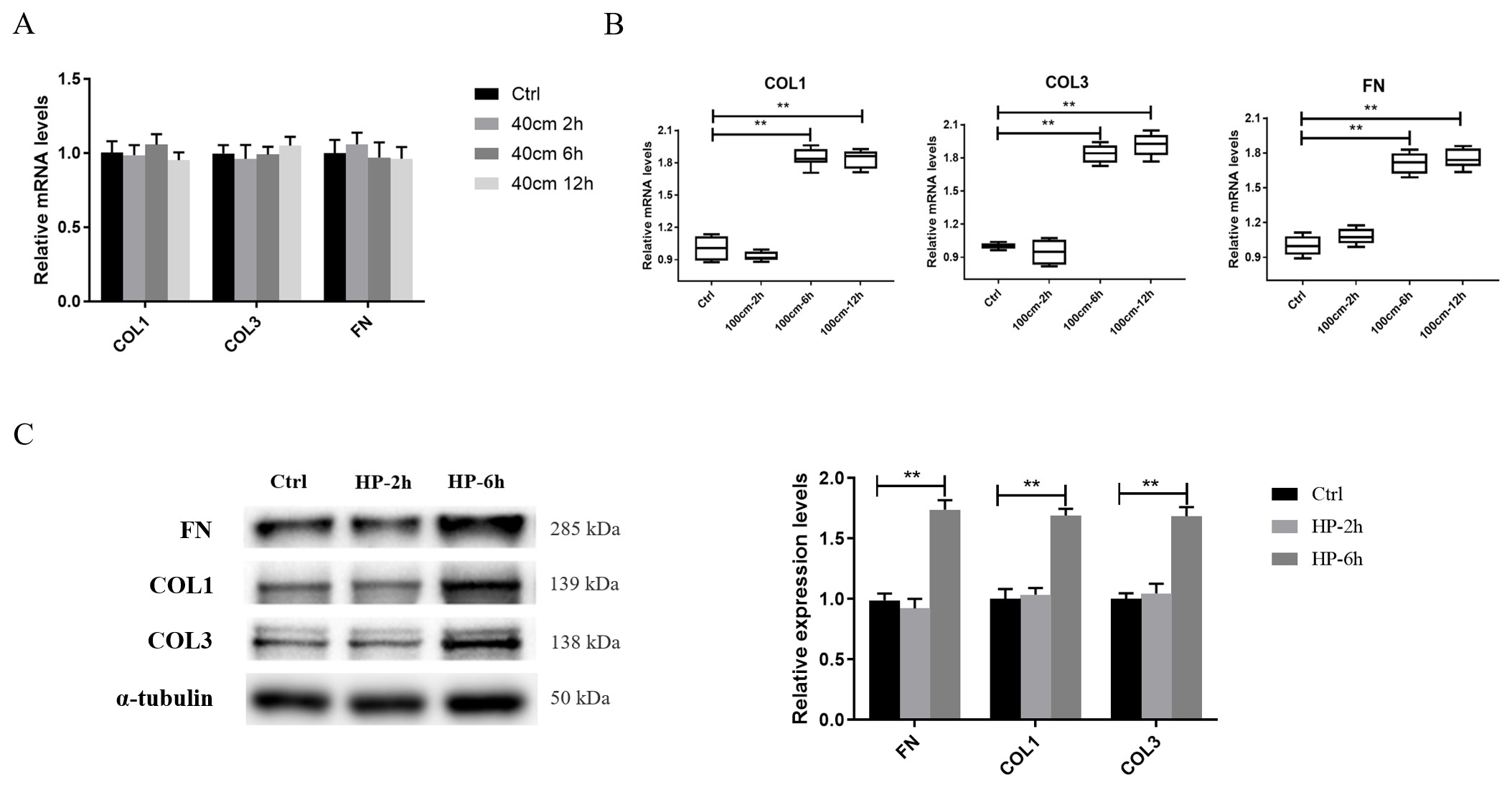 Fig. 1.
Fig. 1.
100 cm H2O hydrostatic pressure increases COL1, COL3 and FN
expression in hBSMCs. (A) Hydrostatic pressure of 40 cm H2O cannot
upregulate the expression of hBSMC ECM compared to the control (0 cm H2O).
(B) The mRNA expression of COL1, COL3 and FN increased in hBSMCs under 100 cm
H2O hydrostatic pressure for 6 h and 12 h. (C) The protein levels of COL1,
COL3 and FN in hBSMCs were determined by western blotting and the grey
intensities of the brands were shown. Ctrl, control group; controlHP, hydrostatic pressure; FN, fibronectin; hBSMCs, human bladder
smooth muscle cells. Three independent
experiments have been performed. **, p
To explore the relationship between the expression of ADRBs and hydrostatic
pressure in hBSMCs, the mRNA levels of three subtypes of ADRBs were detected in
hBSMCs exposed to the hydrostatic pressure of 100 cm H2O for 6 h. The
results showed that the mRNA expression of ADRB1 did not change significantly,
and the expression of ADRB2 and ADRB3 significantly decreased at 6 h under
hydrostatic pressure when compared to the control group (Fig. 2A, p
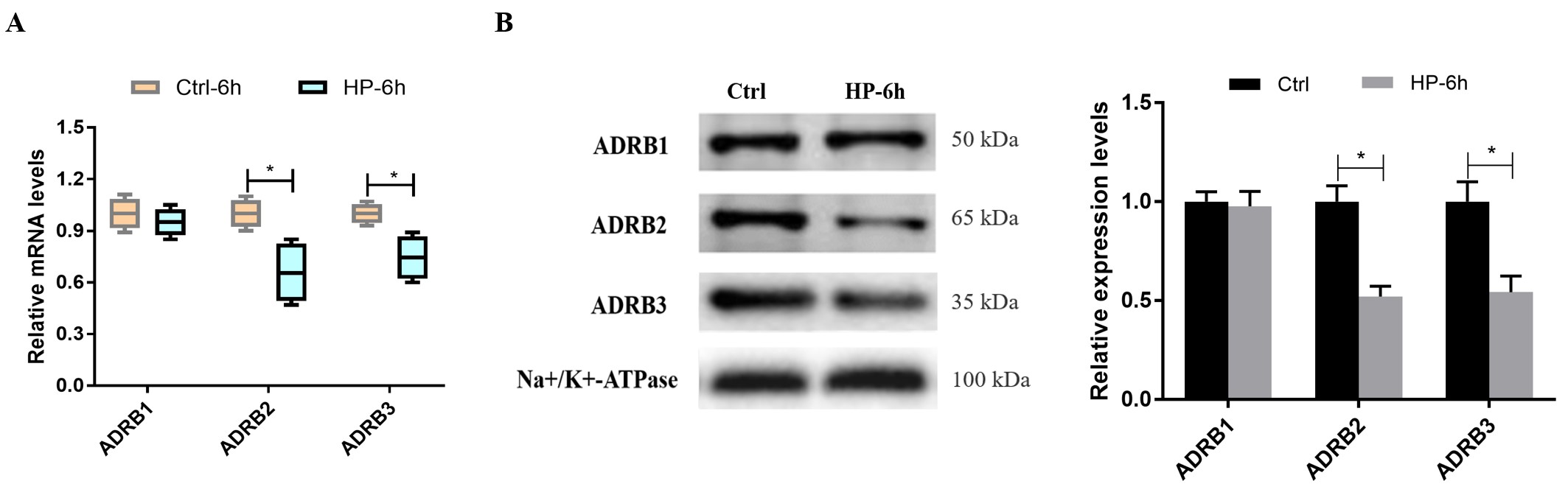 Fig. 2.
Fig. 2.
Hydrostatic pressure suppresses the expression of ADRB2 and
ADRB3. (A) The mRNA expression of ADRB2 and ADRB3 was decreased at 6 h in hBSMCs
under hydrostatic pressure when compared to the control. (B) The protein levels
of ADRBs were identified by western blotting, and Na+/K+-ATP was used for
normalization. HP, hydrostatic pressure. Three independent experiments have been
performed. *, p
To investigate the relationship among ADRBs, hBSMCs ECM and hydrostatic pressure, the agonists or antagonists of ADRBs were used to study their effects on hBSMCs ECM under hydrostatic pressure, and the optimal concentrations of these reagents were determined as per their half maximal inhibitory concentration (IC50) or concentration for 50% of maximal effect (EC50). According to our previous study [24, 25], the optimal concentration of ADRB1 antagonist Metoprolol 200 nM (IC50 for ADRB1 120 nM), ADRB2 antagonist ICI 118551 10 nM (IC50 for ADRB2 1.2 nM), ADRB3 antagonist SR 59230A 100 nM (IC50 for ADRB3 40 nM), ADRB1 agonist Dobutamine 500 nM [26], ADRB2 agonist Formoterol 500 nM [27], ADRB3 agonist BRL 37344 100 nM [28] were selected for the next experiments.
Firstly, hBSMCs were pretreated with 200 nM of Metoprolol, 10 nM of ICI
118551, or 100 nM of SR 59230A for 1 h, and then exposed to 100 cm H2O
hydrostatic pressure for 6 h. In Fig. 3A, the expressions of COL1,COL3 and FN in hBSMCs treated with hydrostatic pressure after
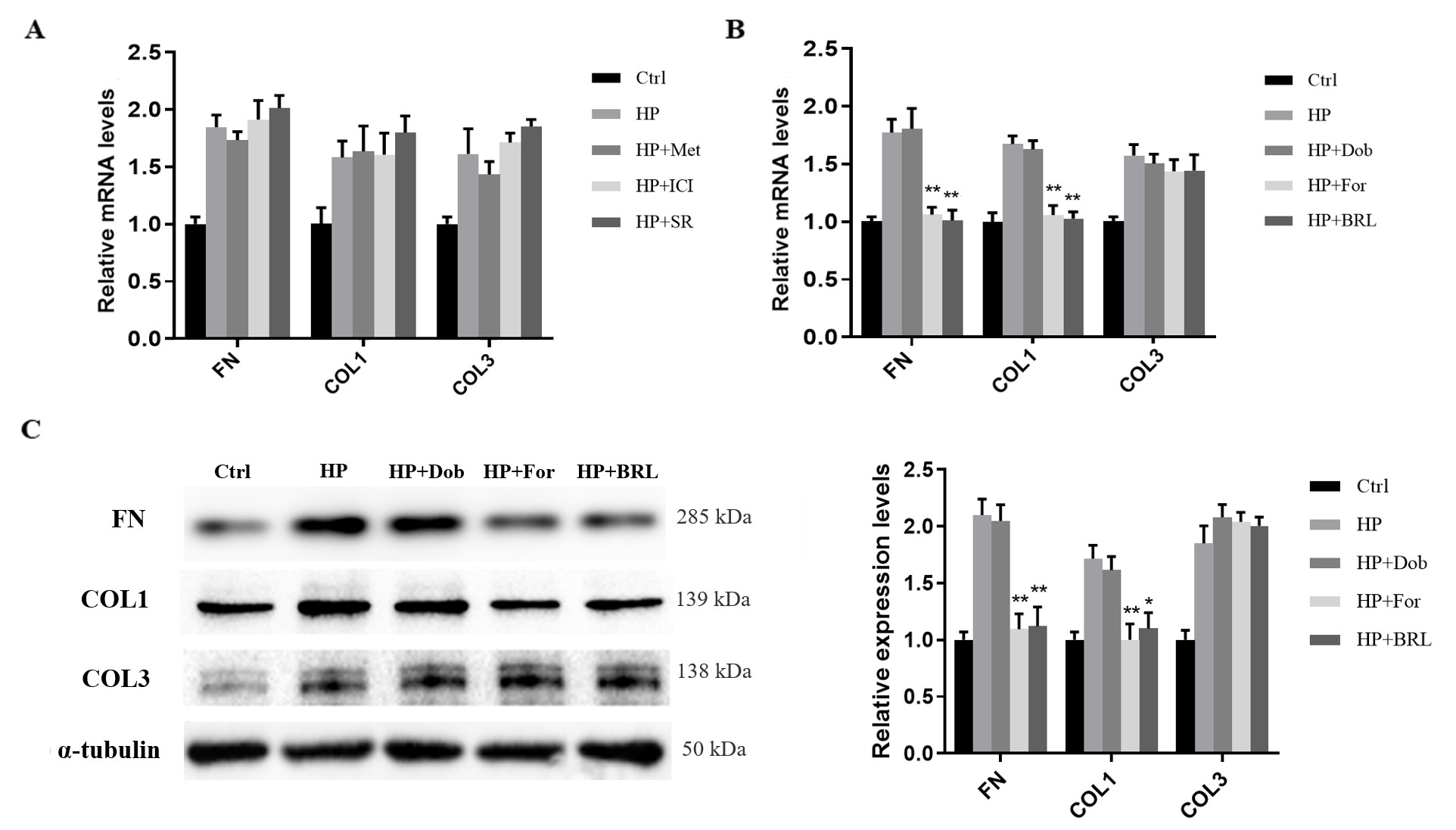 Fig. 3.
Fig. 3.
Formoterol or BRL 37344 significantly suppresses the expression
of COL1 and FN in hBSMCs under hydrostatic pressure. (A) The antagonists of
different ADRBs had no effect on the COL1, COL3 and FN expression at the mRNA
level of hBSMCs under hydrostatic pressure for 6 h. (B) The agonists of ADRBs,
Formoterol and BRL 37344, remarkably decreased the COL1 and FN mRNA expression of
hBSMCs under hydrostatic pressure for 6 h when compared to the hydrostatic group.
(C) COL1 and FN protein levels were determined by western blotting. HP,
hydrostatic pressure; Met, Metoprolol; ICI, ICI 118551; SR, SR 59230A; Dob,
Dobutamine; For, Formoterol; BRL, BRL 37344. Three independent experiments have
been performed. *, p
To further identify the effects of Formoterol and BRL 37344 on hBSMCs ECM,
hBSMCs were pretreated with the antagonists ICI 118551 or SR 59230A for 1 h, and
then incubated with Formoterol or BRL 37344 for another one hour followed by
hydrostatic pressure. ICI 118551 and SR 59230A could respectively inhibit the
effects of Formoterol and BRL 37344 on hBSMCs ECM, namely the suppressive effects
of Formoterol and BRL 37344 on hBSMCs ECM under hydrostatic pressure can be
reversed by their corresponding antagonists (p
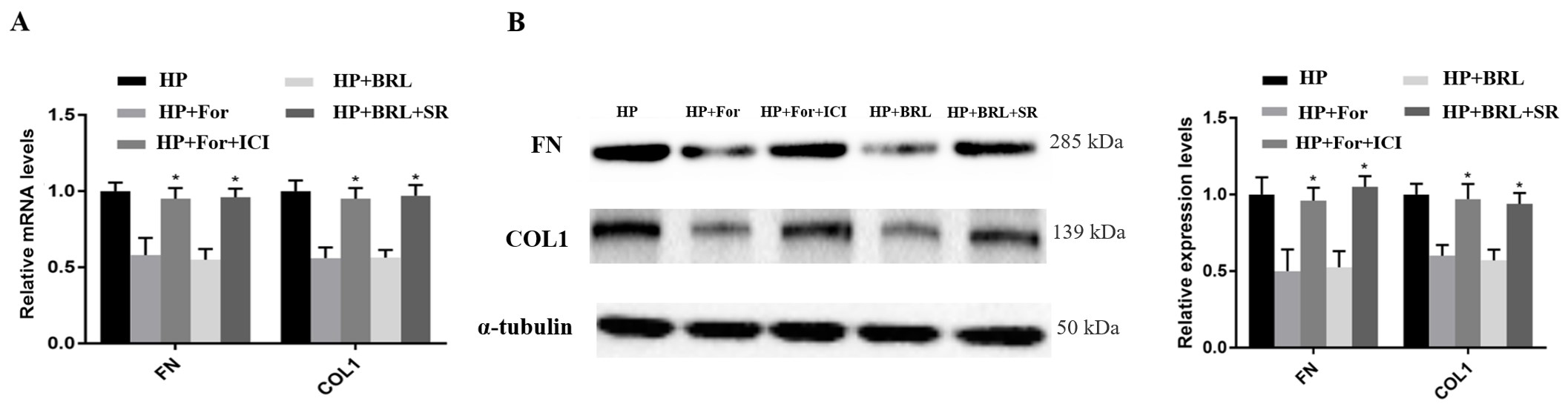 Fig. 4.
Fig. 4.
ICI118551 and SR 59230A rescue the inhibitory effects of
Formoterol and BRL 37344 on FN and COL1 in hBSMCs, respectively. (A) The
suppressive effects of Formoterol and BRL 37344 on the mRNA expression of FN and
COL1 of hBSMCs under hydrostatic pressure were rescued by ICI118551 and SR
59230A. (B) The protein levels of COL1 and FN were determined by western blotting
and the grey intensities of bands were shown. HP, hydrostatic pressure; ICI, ICI
118551; SR, SR 59230A; For, Formoterol; BRL, BRL 37344. Three independent
experiments have been performed. *, p
To unravel the regulatory mechanism of ADRBs, inhibitors of PKA and EPAC, H89
250 nM (IC50 = 140 nM) and ESI09 5 µM (EPAC1 IC50 = 3.2 µM,
EPAC2 IC50 = 1.4 µM) were used to treat hBSMCs for 1 h before the
treatment with Formoterol or BRL 37344 for another 1 h. Furthermore, the cells
were exposed to 100 cm H2O cm hydrostatic pressure for 6 h. The mRNA
expression of FN and COL1 increased in hBSMCs pretreated with ESI09 when compared
to the groups of HP+For or HP+BRL, however, the mRNA expression of FN and COL1
did not change significantly in hBSMCs pretreated with H89 (Fig. 5A, p
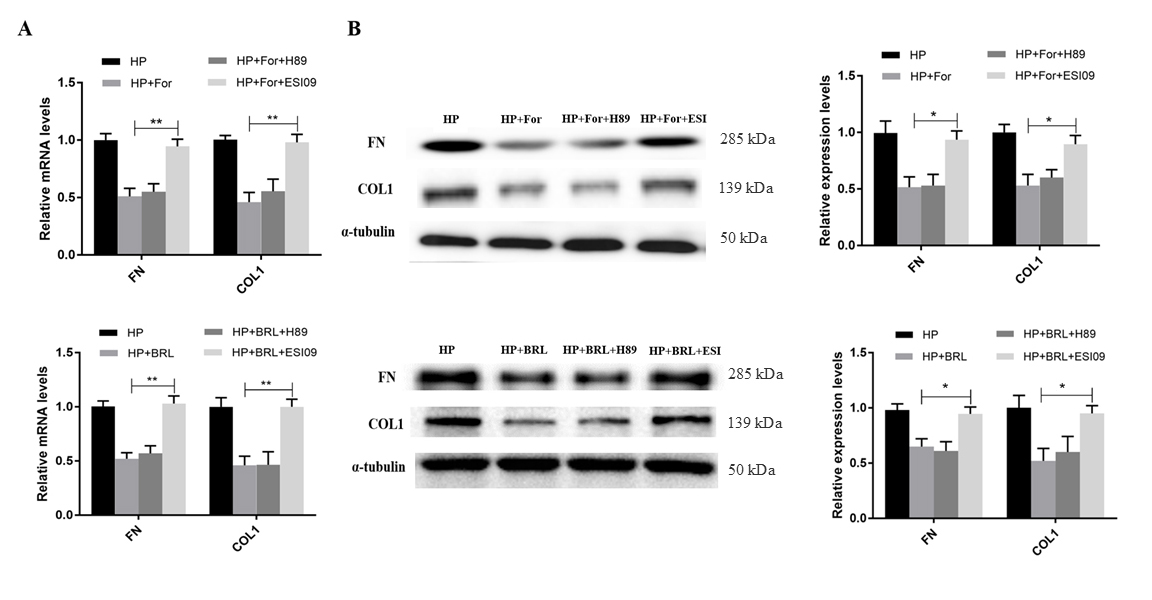 Fig. 5.
Fig. 5.
EPAC regulates the expression of COL1 and FN of hBSMCs under
hydrostatic pressure. (A) The mRNA levels of COL1 and FN in
hBSMCs pretreated with ESI09 significantly increased when compared to that of
hBSMCs treated with Formoterol or BRL37344. (B) The protein levels of COL1 and FN
in hBSMCs were determined by western blotting. HP, hydrostatic pressure; For,
Formoterol; BRL, BRL 37344; ESI, ESI09. Three independent experiments have been
performed. *, p
To further study the factors regulating COL1 and FN expression, the roles of
well-known regulators of collagen and fibronectin, SMAD2 and SMAD3 were
investigated. The protein levels of p-SMAD2 and p-SMAD3 were increased in hBSMCs
under hydrostatic pressure when compared to the control group (Fig. 6A,
p
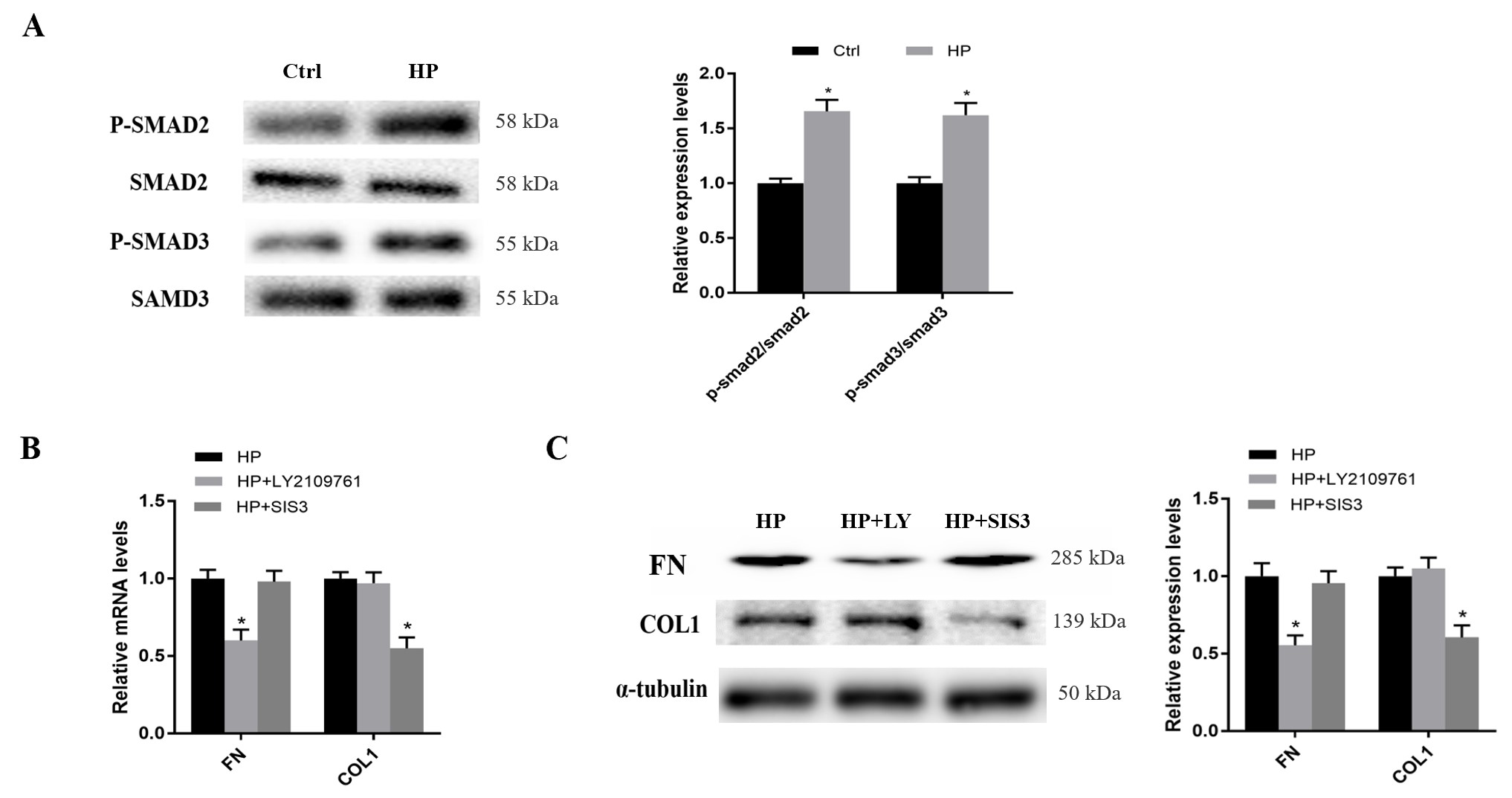 Fig. 6.
Fig. 6.
SMAD2 and SMAD3 respectively regulate FN and COL1 expression in
hBSMCs under hydrostatic pressure. (A) The protein level of p-SMAD2 and p-SMAD3
increased in hBSMCs under hydrostatic pressure when compared to the control
group. (B) 1 µM of LY2109761 inhibited the mRNA expression of FN and 3
µM of SIS3 inhibited the mRNA expression of COL1 in hBSMCs under
hydrostatic pressure. (C) The protein levels of COL1 and FN in hBSMCs were
determined by western blotting. HP, hydrostatic pressure; LY, LY2109761. Three
independent experiments have been performed. *, p
In order to further clarify the regulatory pathways, the relationships between
PKA, EPAC pathway and SMAD2/3 were further investigated. The protein levels of
p-SMAD2 and p-SMAD3 increased significantly in hBSMCs treated with hydrostatic
pressure+Formoterol+ESI09 when compared to the cells treated with hydrostatic
pressure+Formoterol and hydrostatic pressure+Formoterol+H89 (Fig. 7A, p
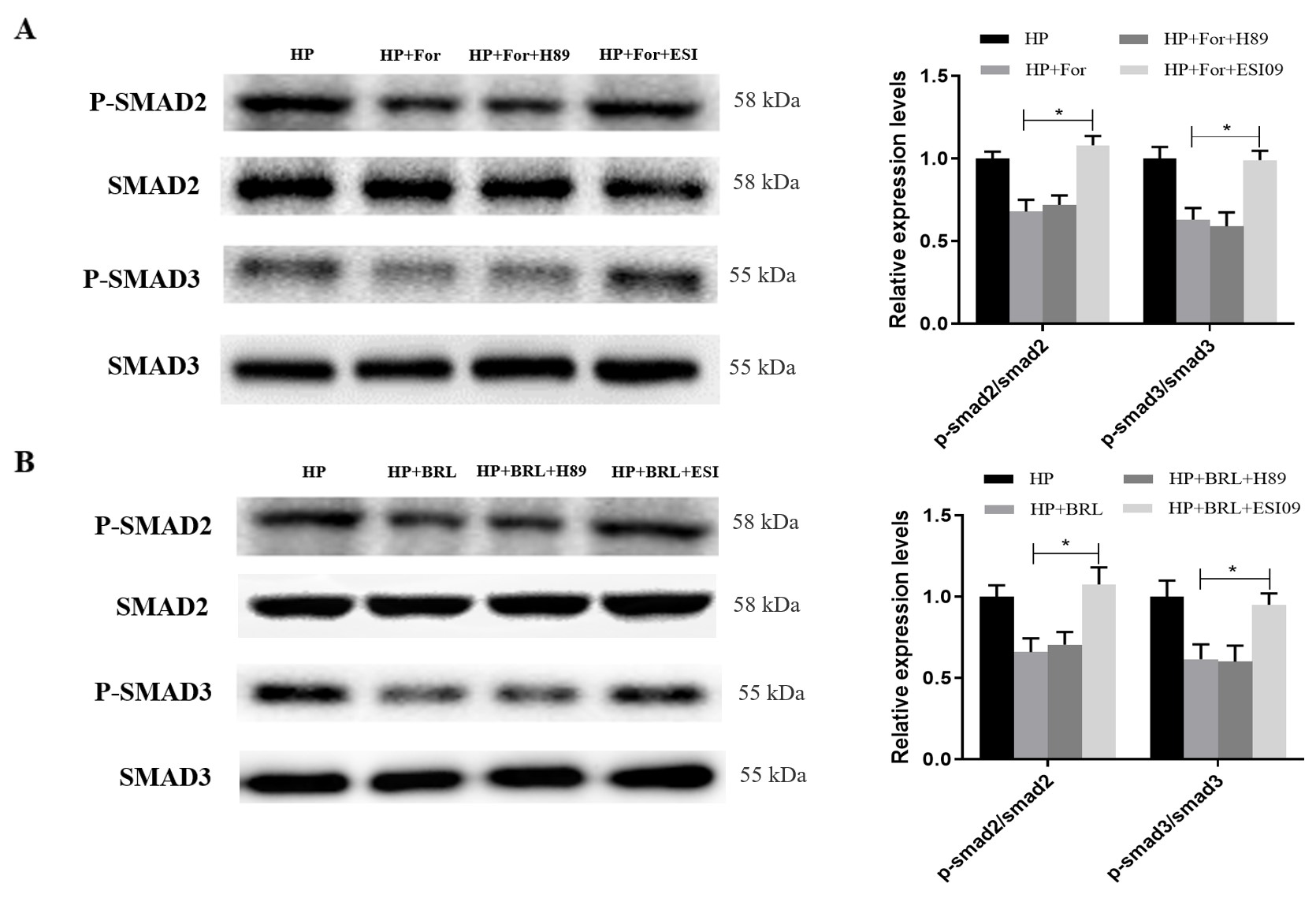 Fig. 7.
Fig. 7.
The protein levels of p-SMAD2 and p-SMAD3 in hBSMCs under
hydrostatic pressure were regulated by the EPAC pathway. (A) The protein levels
of p-SMAD2 and p-SMAD3 increased significantly in hBSMCs treated with hydrostatic
pressure+Formoterol+ESI09 when compared to the cells treated with hydrostatic
pressure+Formoterol and hydrostatic pressure+Formoterol+H89. (B) The protein
levels of p-SMAD2 and p-SMAD3 increased significantly in hBSMCs treated with
hydrostatic pressure+BRL 37344+ESI09 when compared to the cells treated with
hydrostatic pressure+BRL 37344 and hydrostatic pressure+BRL 37344+H89. HP,
hydrostatic pressure; For, Formoterol; BRL, BRL 37344; ESI, ESI09. Three
independent experiments have been performed. *, p
In order to further investigate the effect of ADRB2 and ADRB3 on the bladder
wall remodeling of pBOO, the pBOO rats were treated with ADRB2 or ADRB3 agonist.
Considering the cost and accessibility, we chose the ADRB2 agonist Procaterol and
ADRB3 agonist Mirabegron, the commonly two drugs used in the clinic as the
intervention drugs. A total of twenty-four SD rats were divided into four groups
including the sham-operation group, pBOO group, pBOO+Procaterol group, and
pBOO+Mirabegron group. After 3 weeks of treatment, Masson staining of bladder
tissue of different groups showed the collagen fibers in the bladder of
pBOO group were obviously thickened and the proportion was
increased compared to the sham-operation group. The accumulation of collagen in
the bladder of the pBOO+Procaterol group or pBOO+Mirabegron group was
significantly decreased compared to the pBOO group (pBOO+Procaterol vs. pBOO,
p
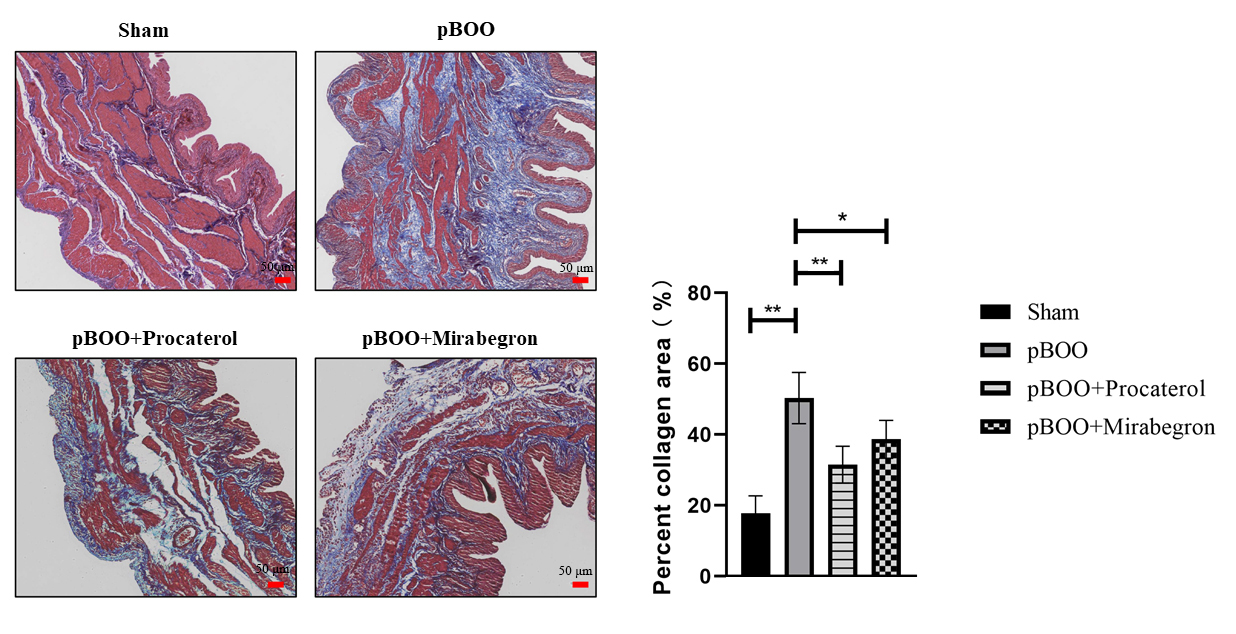 Fig. 8.
Fig. 8.
Procaterol and Mirabegron inhibited collagen deposition in the
bladder of pBOO rats. Masson trichrome staining was used to analyze collagen
(stained blue) and smooth muscle (stained red). The pictures are the
representative images of groups of sham, pBOO, pBOO+Procaterol, and
pBOO+Mirabegron. pBOO, partial bladder outlet obstruction. Scale bar represents 50 µm. n = 6. *, p
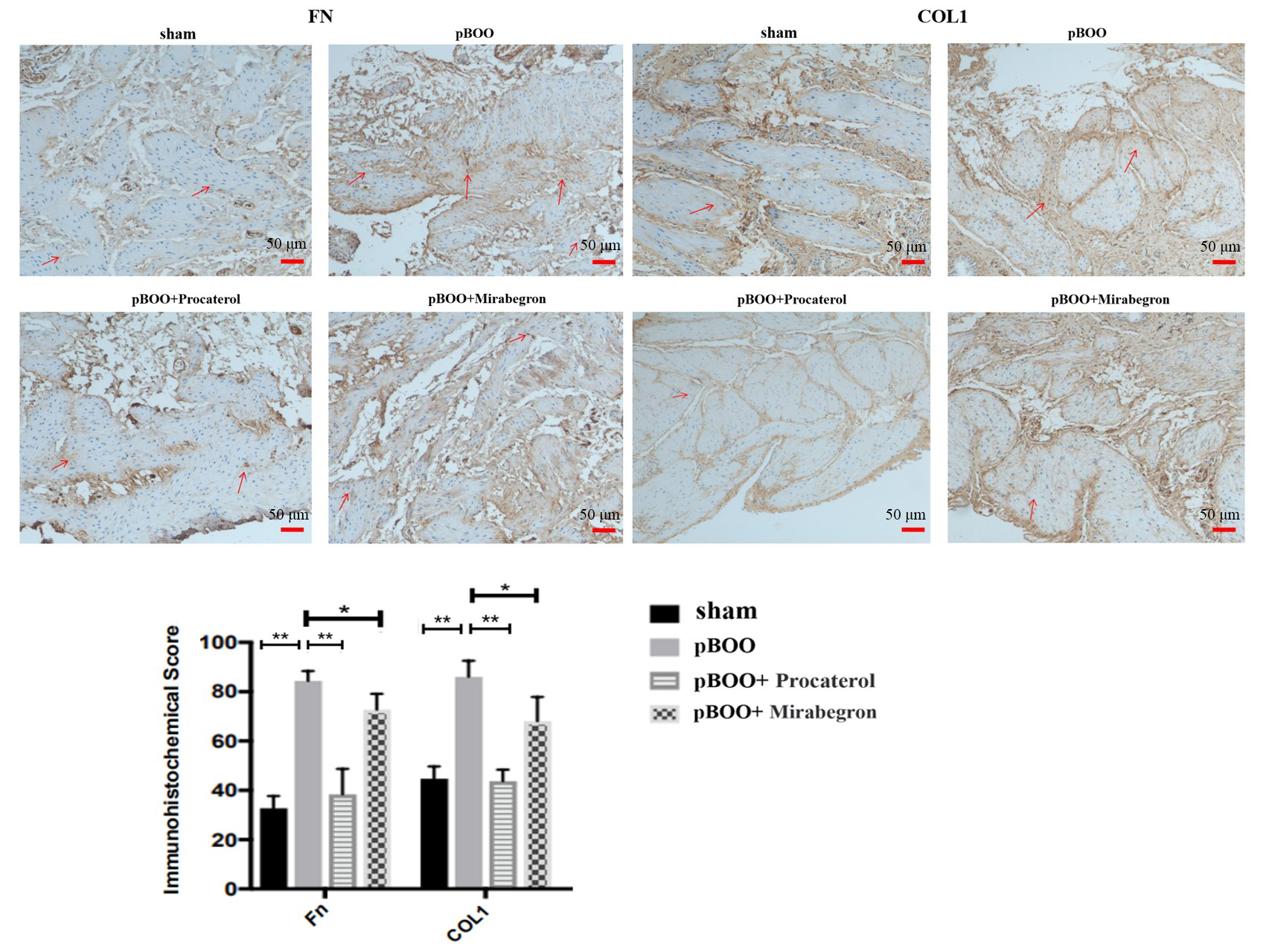 Fig. 9.
Fig. 9.
Procaterol and Mirabegron suppressed the expression of COL1 and
FN in the bladder of pBOO rats by immunohistochemistry. The expression of FN and
COL1 was significantly decreased in the bladder of the pBOO+Procaterol group or
pBOO+Mirabegron group compared to the pBOO group (brown is positive). Red arrows represent the positive expressions of FN or COL1 in tissues. The scale
bar represents 50 µm. n = 6. *, p
The pBOO remodels the bladder structure, increases intravesical pressure, and decreases bladder compliance. ECM is an important component of the bladder wall. The remodeling of the bladder wall structure is closely related to the expression of ECM. In vitro studies have shown that mechanical stress can cause the deposition of ECM of the smooth muscle cell [31, 32], which has an important impact on bladder function. Studies have shown that the ECM/cell ratio is an important indicator of bladder function. A higher proportion of ECM would cause more serious damage to bladder function [33, 34].
Among the changes in bladder ECM induced by pBOO, collagen was the predominant component [35]. The more collagen deposition in the bladder was accompanied by severe lower urinary tract symptoms [36]. It was found that the collagen content of the bladder wall in the rat pBOO model was significantly higher than that in the sham group [37, 38]. A previous study reported that the expression of collagens, particularly COL1 and COL3, was significantly upregulated in the bladder of patients with pBOO [9]. In addition, the content of collagen in the bladder wall was correlated with bladder compliance. One study showed that the content of collagen in the bladder wall of LUTS patients was significantly higher than that of the control group [39]. However, the expression of FN in the bladder of pBOO remains unclear. In the study of bladder fibrosis, it was found that the expression of FN and COL1 in the bladder of female SD rats with interstitial fibrosis was significantly higher than that of rats in the sham group [40]. Moreover, it was reported that the bladder of patients with interstitial cystitis/bladder pain syndrome without Hunner’s disease showed increased expression of fibrosis-associated proteins, including p-SMAD2, vimentin and FN [41]. Therefore, there is a clear relationship between FN and bladder fibrosis/dysfunction.
In this study, we mainly detected the expression of collagen and FN in hBSMCs under different conditions. As per our method of the pBOO rat model, the data showed that the expression of COL1, COL3 and FN in hBSMCs exposed to hydrostatic pressure increased at 6 h when compared to the control group. The increase of collagen expression under hydrostatic pressure was consistent with the previous results in a rabbit pBOO model [42]. As an important component of ECM, FN expression increases under hydrostatic pressure, which is also consistent with the previous results from diabetic rats with damaged bladder detrusors [13].
Early intervention can improve bladder dysfunction and delay the progression of
pBOO [43]. At present, no special drug can improve the deposition of ECM in the
bladder wall caused by pBOO, hence, it is important to investigate the mechanism
involved in the deposition of ECM caused by pBOO. Evidence showed that miR-29 can
counteract bladder over-deformation by affecting the deposition of ECM and the
hardness of the bladder wall [44]. In addition, it was found that pirfenidone
could inhibit the increase of bladder mass, muscle hypertrophy and collagen
deposition in the bladder wall induced by pBOO in rats [45], and muscarinic
cholinergic receptor inhibitor could reduce the expression of collagen in the
bladder wall of pBOO model rats, and improve the urodynamic parameters [46].
Although these studies have explored the deposition of ECM in the bladder wall of
pBOO, the specific mechanism is still unclear. As a transmembrane receptor
closely related to the M receptor,
As per the changes in ADRBs expression in hBSMCs under hydrostatic pressure, selective inhibitors and agonists were used for pretreatment. The results showed the expression of COL1 and FN decreased in hBSMCs pretreated with ADRB2 or ADRB3 agonists before 100 cm H2O hydrostatic pressure for 6 h when compared to the hBSMCs exposed to hydrostatic pressure only, nevertheless, the expression of COL3 did not change significantly. Although the results showed COL1 and COL3 were not inhibited by ADRB2 or ADRB3 agonists simultaneously, the ADRB2 and ADRB3 agonists decreased the collagen deposition and ECM accumulation of pBOO rat bladder by Masson staining and immunohistochemistry test in animal experiments. Thus, the treatment of ADRB2 or ADRB3 agonists could inhibit the expression of COL1 and FN, and further improve bladder dysfunction under pBOO.
The role of two downstream pathways of ADRB, PKA and EPAC [17], in regulating
the ECM of bladder smooth muscle under pBOO is still not clear. The present study
found that the expression of COL1 and FN was regulated by the EPAC pathway rather
than PKA signaling under hydrostatic pressure. In addition, we found that SMAD2/3
was associated with the expression of ECM [49, 50]. The inhibition of SMAD2/3
activity under hydrostatic pressure can reduce the expression of COL1 and FN.
Intriguingly, SMAD2 and SMAD3 have different roles in regulating ECM under
hydrostatic pressure. SMAD2 regulates the expression of FN and SMAD3 regulates
the expression of COL1, however the level of p-SMAD2 and p-SMAD3 is regulated by
the EPAC pathway. Previous study had found that
ADRB2 and ADRB3 agonists, especially the former can influence the collagen deposition in the pBOO bladder and accumulation of hBSMCs ECM under pathological hydrostatic pressure. The expression of FN and COL1 in hBSMCs was regulated by EPAC/SMAD2 and EPAC/SMAD3 pathways, respectively. By regulating the expression of FN and COL1 in hBSMCs, ADRB2 and ADRB3 agonists can contribute to improving bladder dysfunction caused by pBOO. These findings revealed the underlying mechanism of ADRBs involved in regulating bladder function, and further provide new potential therapeutic targets for the treatment of bladder outlet obstruction.
The datasets used or analyzed during the current study are available from the first author or corresponding author on reasonable request.
JL and GC performed the experiments; GC, HS, QH and KX analyzed the data; JL and HS drafted the manuscript; JL, BL and JA conceived and designed the experiments; BL and JA polished the manuscript and provided constructive suggestions to this paper; GC, KX and QH contributed to the tables and figures in the present paper. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experiments were performed in adherence with the National Institutes of Health Guidelines on the Use of Laboratory Animals and approved by the West China Hospital Committee on Animal Care (No. 2018220A).
Thanks to all the peer reviewers who made comments and suggestions on this study.
This work was supported by the National Natural Science Foundation of China (82070784); and from Office of Science & Technology and Talent Work of Luzhou (2020-JYJ-51).
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.fbl2909336.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









