1 Discipline of Surgical Specialties, Adelaide Medical School, University of Adelaide, The Queen Elizabeth Hospital, Woodville South, SA 5011, Australia
2 Robinson Research Institute, University of Adelaide, Adelaide, SA 5006, Australia
3 College of Medicine and Public Health, Flinders Health and Medical Research Institute, Flinders University, Bedford Park, SA 5042, Australia
4 SAHMRI Women and Kids, South Australian Health and Medical Research Institute, Women's and Children's Hospital, North Adelaide, SA 5006, Australia
Abstract
Lactation is associated with long-term reduced risk of breast cancer. However, there is a transient increased risk of breast cancer in the 5 to 10 years postpartum and this is associated with a high incidence of metastasis and mortality. Breastmilk is a physiological fluid secreted by the mammary glands intimately connected with breast cells and the microenvironment that may affect postpartum breast cancer development and progression. This study aims to investigate the effect of breastmilk on interactions between breast cancer cells and macrophages in vitro.
Human breastmilk from healthy donors (n = 7) was pooled and incubated with breast cancer (MCF-7 and MDA-MB-231) and macrophage (RAW264.7) cell lines to assess cell proliferation, viability, migration, and expression of key genes associated with epithelial-mesenchymal transition (EMT) and macrophage phenotype. Indirect co-culture studies assessed the effect of breastmilk on interactions between breast cancer cells and macrophages.
Breastmilk increased the proliferation and viability of breast cancer cells, reduced EMT markers, and reduced cell migration in MDA-MB-231 cells. Breastmilk decreased mRNA expression of interleukin 1B (IL1B) and interleukin 10 (IL10) in macrophages. Reduced EMT marker expression was observed in breast cancer cells co-cultured with macrophages pre-treated with breastmilk. Macrophages co-cultured with breast cancer cells pre-treated with breastmilk exhibited increased expression of a pro-inflammatory cytokine tumor necrosis factor A (TNFA) and pro-inflammatory nitric oxide synthase 2 (NOS2), and reduced expression of cytokines IL10 and transforming growth factor B1 (TGFB1) which are associated with the alternatively-activated macrophage phenotype.
Breastmilk has the potential to promote breast cancer proliferation, however, it can also reduce breast cancer progression through inhibition of breast cancer cell migration and regulation of macrophage polarisation. These findings suggest that breastmilk has potential to shape the tumour microenvironment in postpartum breast cancer.
Keywords
- breastmilk
- breast cancer
- postpartum
- lactation
- cytokines
- epithelial-mesechymal transition
- macrophages
Breast cancer is the most frequent type of malignant tumour in women, accounting
for more than 2.3 million occurrences worldwide in 2023 and it is the most
diagnosed cancer during pregnancy [1, 2]. Given the present trend of delayed
childbearing, there is an anticipated rise in the incidence of breast cancer
cases identified during pregnancy or in the few years after giving birth [3].
While definitions may vary, postpartum breast cancer is typically characterised
as breast cancer detected within 5 to 10 years after childbirth and it is
estimated to account for approximately 35% to 55% of all breast cancer cases in
women
Analysis of breastmilk provides an opportunity to non-invasively study the intricate cellular interactions within the microenvironment of the mature mammary gland that affect breast cancer risk and progression [10]. Human breastmilk is an extraordinary and specifically customised fluid secreted by the mammary glands that provides nourishment and immunological protection for infants [11]. Breastmilk contains a wide range of bioactive substances in addition to nutrients, including stem cells, microbes, leukocytes and immune-related factors, hormones, cytokines, and microRNAs [12]. Studies have demonstrated that various constituents of breastmilk have roles in distinct biological functions associated with immune activity and cancer [13, 14, 15]. For example, the production of proinflammatory cytokines such as tumour necrosis factor alpha (TNFA), interleukin 1B (IL1B) and interleukin 6 (IL6) in colon cancer cells was inhibited by 5% supernatant breastmilk [13]. In the context of breast cancer, it has been reported that exosomes derived from breastmilk harbouring elevated levels of transforming growth factor B2 (TGFB2) promote proliferation of cancerous and non-cancerous breast cells and promote epithelial-mesenchymal transition (EMT) [14]. However, it has also been reported that human breastmilk possesses the ability to impede the migration of mouse mammary Met-1 tumour cells [15]. Based on these studies, the effect of breastmilk on breast cancer cells may be either tumour-promoting or tumour-inhibiting, potentially dependent on the types of cells exposed to breastmilk and the tumour microenvironment.
Tumour microenvironments comprise a wide variety of inflammatory cells and cytokines that have roles in regulating cancer development and progression [16]. Breast cancer development is a multi-factorial process influenced by crosstalk between malignant cells and components of the microenvironment including inflammatory cytokines and immune cells [17, 18]. Macrophages, which are phagocytic cells of the immune system, are present in almost all tissues. They exhibit impressive flexibility and adaptability, responding to signals in their vicinity [19]. Tumour-associated macrophages (TAMs) are critical cells of the tumour microenvironment and have a substantial impact on several facets of carcinogenesis, including cell proliferation, metastasis, and invasion [20]. Furthermore, in breast cancer, TAMs are the most frequent infiltrating immune cells in the tumour microenvironment and show an indispensable function in promoting EMT through their interactions with tumour cells [21, 22]. Polarisation is a pivotal characteristic of macrophages, and their ability to adopt distinct phenotypes in response to environmental signals is recognised as a crucial factor in regulating cancer cells [23]. Macrophages can undergo a transformation into the M1 phenotype, during which they produce pro-inflammatory cytokines that actively participate in anti-tumour processes [24]. In contrast, the alternatively-activated macrophage M2 phenotype, which can be promoted by cancer cells, releases oncogenic cytokines such as IL10 and TGFB that contribute to immune suppression, facilitating the evasion of cancer cells from immune defences [25]. For example, it has been shown that secretion of IL6 from breast cancer cells led to enhanced monocytic THP-1 cell line polarisation into M2 macrophages, thereby promoting increased invasiveness in MDA-MB-231 cells [26].
According to the role of macrophages in breast cancer development and the significant impact of breastmilk on several inflammatory diseases, it is possible that breastmilk may be involved in crosstalk between cancer cells and macrophages within the mammary gland that affects cancer progression [27, 28]. In this study, we investigate the effect of breastmilk on the activity of breast cancer and macrophage cell lines in vitro, and the cell-to-cell interactions between these cell types. This study provides new insights into the role of breastmilk in inhibiting postpartum breast cancer development and progression.
The research was approved by the Central Adelaide Local Health Network (CALHN) Human Research Ethics Committee (HREC) (CALHN Reference Number: 19523). Informed consent was received from each participant. Breastmilk was obtained from 7 healthy women, with no current medical conditions, no history of mastitis, who were not pregnant and had been breastfeeding their infants for at least one month. Samples of breastmilk were collected using sterile tubes (62.547.254, Sarstedt, Nümbrecht, Germany). Breastmilk samples were kept at –20 °C immediately after being expressed and then transported to a –80 °C freezer (Thermo Fisher Scientific, Carlsbad, CA, USA) within 48 hours. Following the collection, breastmilk samples were combined into a single pool. A 0.22 µM filter (SLGP033R, Millex-GP, Darmstadt, Germany) was subsequently applied to the pooled breastmilk sample in order to eliminate any cells. The resulting filtered breastmilk pool was used for all experiments as whole milk in this investigation.
The human hormone-sensitive breast cancer cell line MCF-7 HTB-22™, human triple negative breast cancer (TNBC) cell line MDA-MB-231 HTB-26™ and the mouse macrophage cell line RAW264.7 TIB-71™ were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). MCF-7 and RAW264.7 cells were cultured in RPMI 1640 medium (11835-030, Gibco, Waltham, MA, USA) containing 10% foetal bovine serum (FBS) (S-FBS-EU-015, Serana, Pessin, Germany) and 1X Penicillin/Streptomycin (15240062, Gibco, Waltham, MA, USA). MDA-MB-231 was cultured in DMEM-high glucose (D5796, Sigma-Aldrich, St. Louis, MO, USA) with 10% FBS (S-FBS-EU-015, Serana, Pessin, Germany) and 1% Penicillin/Streptomycin (15240062, Gibco, Waltham, MA, USA). The cells were incubated at standard culture conditions at 37 °C in 5% CO2 and 95% humidity. MCF-7 and MDA-MB-231 cells were subcultured using Trypsin-EDTA (25200056, Gibco, Waltham, MA, USA) when they reached around 80% confluency. RAW264.7 cells were subcultured using Accutase (00-4555-56, Invitrogen™, Carlsbad, CA, USA) after reaching 60–70% confluency. All cell lines were confirmed by STR profiling and tested negative for mycoplasma using a 4’,6-diamidino-2-phenylindole (DAPI) staining [29].
Cell lines were seeded in a 24-well plate (662160, Greiner, Kremsmünster, Austria) in medium containing 5% FBS. After a 24-hour incubation period, the media were replaced and supplemented with a varying percentage of whole breastmilk (0, 0.5, 1, 2, and 5%). The number of viable and dead cells were determined at 24, 48 and 72 hours using trypan blue staining (T8154, Sigma-Aldrich, St. Louis, MO, USA) and counted manually via haemocytometer (8100103, Hirschmann, Eberstadt, Germany). This experiment was carried out in two independent replicates, each with two technical replicates.
To provide further validation of cellular proliferation, the MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) assay was used. For this, cell lines were seeded in a 96-well plate (655180, Greiner, Kremsmünster, Austria) and allowed to adhere for 24 hours in medium containing 5% FBS. Subsequently, the media was refreshed, supplemented with whole breastmilk (0, 0.5, 1, 2, and 5%), and the cells were incubated for 24, 48, and 72 hours. At each time point, 5 µL of 10 mg/mL sterile MTT (M5655, Sigma-Aldrich, St. Louis, MO, USA) assay reagent added into the wells. After 4 hours incubation, culture media were discarded carefully, and each well was dissolved in 100 µL DMSO (D4540, Sigma-Aldrich, St. Louis, MO, USA). The absorbance of each well was measured at 570 nm using a microplate reader (CLARIOstar Plus, BMG labteck, Mornington, VIC, Australia) to quantify cell viability. The MTT assay was performed in two independent replicates, each with four technical replicates.
To extract RNA from the cell lines, RNeasy Mini Plus kit (74104, Qiagen, Hilden, Germany) was used according to the manufacturer’s instructions. RNA yield was quantified using the spectrophotometer ND 1000 Nanodrop (Thermo Fisher Scientific, Carlsbad, CA, USA). Following that, complementary DNA (cDNA) conversion was carried out on 1 µg of RNA using the iScript Select cDNA Synthesis Kit (1708891, Bio-Rad, Hercules, CA, USA) according to manufacturer’s instructions. qRT-PCR was performed using specific oligonucleotide primers for genes of interest with iQ SYBR Green Supermix (1725274, Bio-Rad, Hercules, CA, USA) on Applied Biosystems PCR machine (ViiA™, Waltham, MA, USA) to quantify the gene expression levels. Sequences of primers are provided in Supplementary Tables 1,2. The mRNA expression of genes was normalised to housekeeping genes (ACTB and Rpl13a) and the relative gene expression levels were quantified using the 2-ΔΔCq method. All experiments were carried out in three independent replicates, each with three technical replicates.
Cells were seeded into a 24-well plate and incubated overnight to obtain a 90%
confluent cell monolayer. Then, a constant sized artificial gap was created with
a 200 µL plastic tip (Sorelac17374T, Bio-Strategy, Melbourne, VIC,
Australia) in the cell monolayer and washed with PBS two times. After that, media
containing 1% FBS was added, and wells were supplemented with whole breastmilk
(0 or 1%). The degree of movement of the cells filling the gap was captured at
0, 24, and 48-hour time points. The images at each time point were captured using
a Nikon eclipse TS100 inverted microscope (Nikon, Tokyo, Japan) with a Nikon
DS-L3 camera (Nikon, Tokyo, Japan). Images of migration areas were analysed with
Fiji ImageJ software (Version 1.54d, National Institutes of Health (NIH),
Bethesda, MD, USA) [30]. The percent migration was measured by ([initial wound
area] – [wound area at the last time point])/[initial wound area]
RAW264.7 cells were seeded into a 24 mm transwell insert with 0.4 µm pores polycarbonate membrane (3412, Corning, Kennebunk, ME, USA) in medium with 5% FBS, and after 24 hours cells were treated with 1% of whole milk and incubated for 48 hours. Alongside, breast cancer cells were cultured in serum-free media in the bottom chamber of a 6-well plate, 24 hours prior to the experiment. Following that, the transwell inserts containing RAW264.7 cells were transferred into the wells with breast cancer cells and co-cultured for 48 hours. Subsequently, the mRNA expression of EMT markers in MCF-7 and MDA-MB-231 cells was determined. The same experimental setting was used to determine inflammatory markers in RAW264.7 cells. Briefly, breast cancer cells were seeded into a 24 mm transwell insert with 0.4 µm pores polycarbonate membrane in medium containing 5% FBS for 24 hours and then treated 48 hours with 1% of whole milk. Macrophage cells were cultured in the bottom chamber of a 6-well plate in serum-free media 24 hours prior to the co-culture. Then, the transwell inserts containing breast cancer cells were transferred into the wells with RAW264.7 cells and incubated for 48 hours. The mRNA expression of inflammatory markers was determined in macrophages.
Statistical analyses were performed with the GraphPad Prism 10.1 software
(Dotmatics, Boston, MA, USA). All data are reported as mean
In order to determine impact of breastmilk on the growth of breast cancer cells
and macrophages, cell proliferation and MTT assays were employed. Based on the
cell proliferation findings, the growth of breast cancer cells and macrophages
was enhanced when they were exposed to breastmilk. This enhancement was amplified
with higher concentrations of milk in the culture medium (Fig. 1). Overall, MCF-7
cells demonstrated a higher rate of responsiveness to breastmilk than MDA-MB-231
cells, as indicated by cell proliferation and cell viability results. The results
of the cell proliferation analysis showed that number of live MCF-7 cells treated
with 2% and 5% breastmilk significantly increased at 24 hours compared to
non-treated cells. In contrast, MDA-MB-231 cell proliferation did not
significantly change with any quantity of milk at this time point
(Supplementary Fig. 1A). Following 48 and 72 hours of treatment, the
proliferation of MCF-7 cells was significantly enhanced by a 0.5% concentration
of breastmilk (p = 0.0061 and p = 0.0002, respectively).
Conversely, the proliferation of MDA-MB-231 cells did not exhibit a significant
increase at the same concentration and time points (Fig. 1A,B,D,E). Both breast
cancer cell lines demonstrated a significant rise in number of live cells after
being exposed to breastmilk at concentrations of 1% or higher for either 48 or
72 hours. A statistically significant increase in cell proliferation was observed
in the RAW264.7 macrophage cell line after 24 hours of exposure to 1%
(p = 0.0343), 2% (p = 0.0003) and 5% breastmilk (p
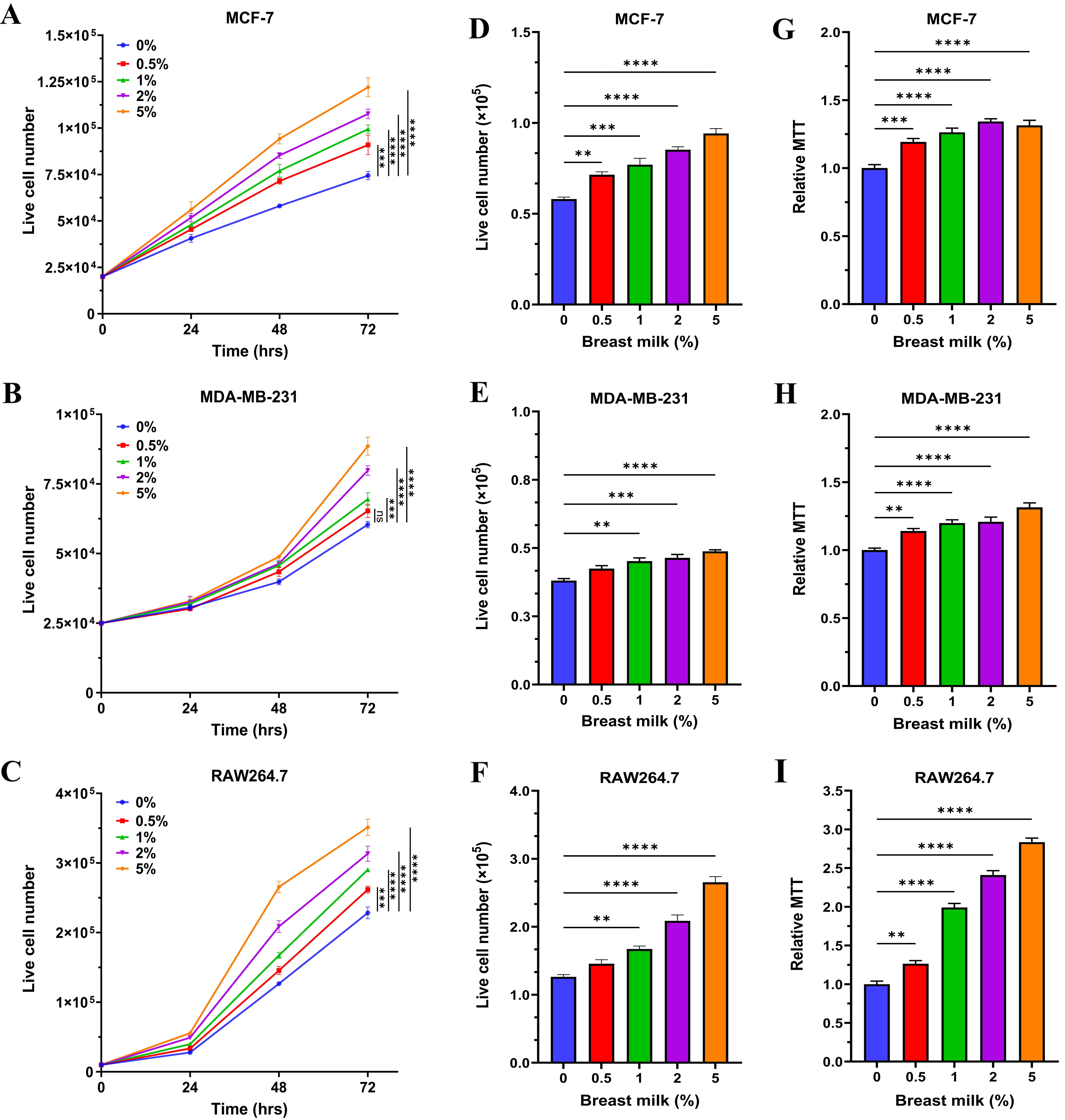 Fig. 1.
Fig. 1.
Breastmilk stimulates the proliferation of breast cancer cells
and macrophages. (A–C) The effect of breastmilk on the growth of three cell
lines over time and (D–F) at the 48 hour time point quantified using Trypan
blue. (G–I) The effect of breastmilk on cell viability determined by 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) assay
relative to non-treated controls (0% breastmilk) after 48 hours. (A,D,G) MCF-7
cells. (B,E,H) MDA-MB-231 cells. (C,F,I) RAW264.7 cells. Data presented as
mean+SEM of two independent experiments. Data analysed by one-way ANOVA followed
by Tukey’s multiple comparison test (ns, not significant;
**p
The MTT assay confirmed the observed responses to breastmilk, indicating that as the concentration of breastmilk increased, cell viability increased across all cell lines. At the 24-hour time point in the MTT assay, the viability of MCF-7 cells was significantly enhanced at 2% and 5% concentrations of breastmilk (p = 0.0067 and p = 0.0013, respectively) compared to the non-treated group, while MDA-MB-231 cells showed a significant increase in viability only at the 5% concentration (p = 0.0006). In the same frame of time, the viability of RAW264.7 cells significantly increased when treated with the 1% and higher concentrations of milk (Supplementary Fig. 1B). As indicated by the MTT outcomes, the viability of each cancer cell line increased dramatically in all concentrations of breastmilk in comparison to non-treated cells, following 48 hours treatment and thereafter (Fig. 1G–I and Supplementary Fig. 1C). Collectively, these results suggest that breastmilk has the capacity to accelerate the growth rate of macrophages and breast cancer cells. All subsequent experiments used a concentration of 1% breastmilk and a 48-hour time point.
To further study effect of breastmilk on breast cancer cells, gene expression of key M1 and M2 proteins in macrophages and EMT markers in breast cancer cells was investigated. MCF-7, MDA-MB-231, and RAW264.7 cells were treated with 1% of breastmilk for a duration of 48 hours. As demonstrated in Fig. 2A, breastmilk did not alter the mRNA expression of either mesenchymal or epithelial markers in MCF-7 cells. However, the mRNA expression of the E-cadherin gene (CDH1), an epithelial marker, was substantially elevated in MDA-MB-231 cells treated with breastmilk in comparison to the control group (p = 0.013). Additionally, the presence of breastmilk resulted in a significant downregulation of N-cadherin (CDH2) (p = 0.049), a mesenchymal marker compared to cells that were not treated (Fig. 2B). Alteration of gene expression of M1 and M2 proteins in RAW264.7 cells was observed with treatment with breastmilk (Fig. 2C). When compared to non-treated cells, breastmilk-treated macrophages exhibited a significant downregulation of the M1 cytokine IL1B and the M2 cytokine IL10, with the latter revealing more pronounced reduction in mRNA expression (p = 0.003 and p = 0.00019, respectively). These findings demonstrate that breastmilk has a capacity to reduce gene expression linked with EMT in breast cancer cells and lower cytokine expression in macrophages.
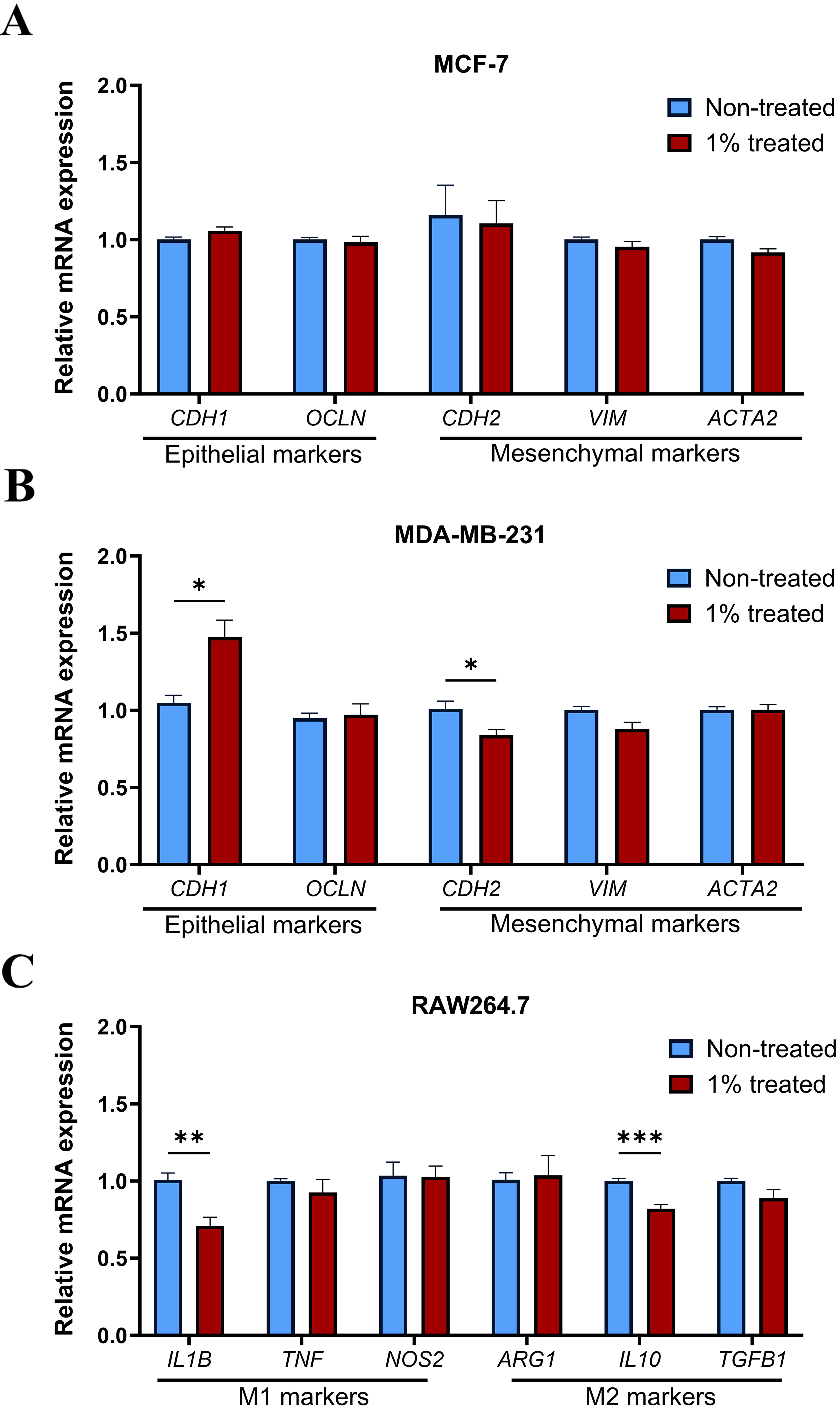 Fig. 2.
Fig. 2.
Breastmilk regulated expression of genes associated with breast
cancer cell epithelial-mesenchymal transition (EMT) and macrophage polarisation.
(A) Represents mRNA expression of EMT markers in MCF-7 cells with no difference
between treated and non-treated groups. (B) Expression of EMT markers changed
upon treatment with milk in MDA-MB-231 cells with upregulation of CDH1
and downregulation of CDH2 compared to non-treated cells. (C) Breastmilk
reduced both M1 and M2 markers in RAW264.7 cells compared to the control group
with more significant reduction in IL10. Data presented as mean+SEM of
three independent experiments each with triplicate wells. p-values were
calculated using unpaired student’s t-test
(*p
To explore the influence of breastmilk on breast cancer cell-macrophage interactions that affect EMT, breast cancer cells were co-cultured indirectly with RAW264.7 cells that had been treated with breastmilk (Fig. 3A). Breastmilk-treated macrophages substantially increased the expression of an epithelial marker CDH1 (p = 0.004) and reduced the expression of mesenchymal markers CDH2, vimentin (VIM) and alpha-smooth muscle actin (ACTA2) (p = 0.0006, 0.004, and 0.0012 respectively) in MCF-7 cells compared to macrophages not treated with breastmilk (Fig. 3B). Macrophages treated with breastmilk also reduced expression of mesenchymal-associated genes CDH2 and VIM (p = 0.002 and 0.00003 respectively) in MDA-MB-231 cells (Fig. 3C). Taken together, this data suggests that breastmilk modifies the activity of macrophages such that they exert inhibitory effects on breast cancer cell EMT.
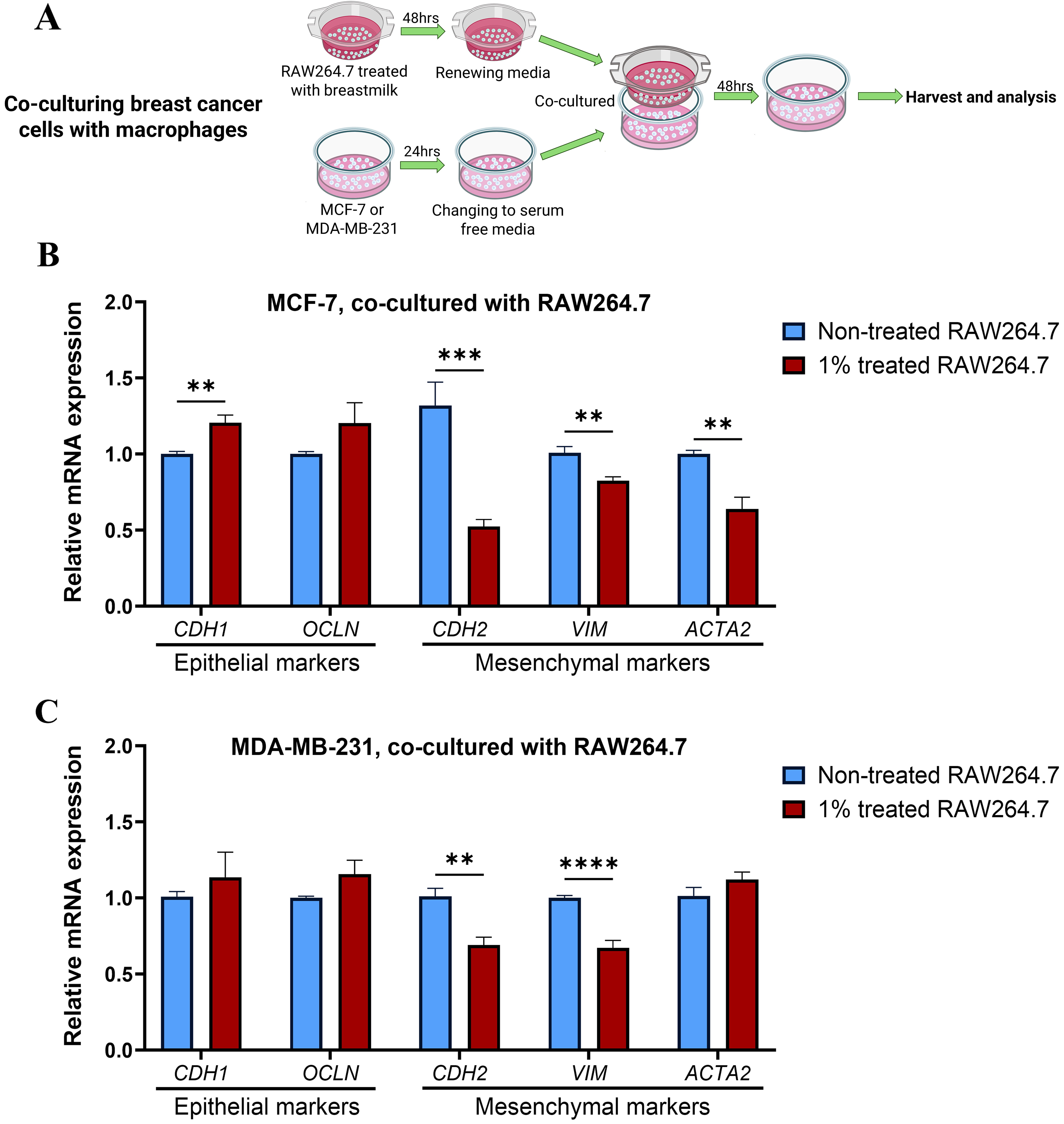 Fig. 3.
Fig. 3.
Breastmilk-treated macrophages inhibit EMT in breast cancer
cells. (A) Schematic diagram of co-culturing breast cancer cells with
milk-treated macrophages. (B) The figure represents significant changes in
expression of both epithelial and mesenchymal related genes in MCF-7 after
co-culturing with macrophages supplemented with milk. (C) Illustrates how 1%
milk supplementation in culture media of macrophages could alter the mRNA
expression of CDH2 and VIM genes in MDA-MB-231 cells through
the co-culture experiment. Data presented as mean+SEM of three independent
experiments each with triplicate wells. Unpaired student’s t-test was
used to compare treated groups and non-treated (**p
In the previous section, breast cancer cells were co-cultured with macrophages that had been treated with breastmilk in order to investigate how macrophage exposure to breastmilk might affect their interaction with breast cancer cells. In this section, the converse experiment was undertaken, where breast cancer cells were treated with breastmilk and then cultured with macrophages in order to explore the influence of breastmilk on breast cancer cell-macrophage interactions that affect macrophage cytokine expression (Fig. 4A). Breastmilk-treated MCF-7 cells reduced the expression of M2-associated cytokines IL10 and TGFB1 (p = 0.00009 and 0.0001 respectively) compared to MCF-7 cells not treated with breastmilk (Fig. 4B). Breastmilk-treated MDA-MB-231 cells increased the expression of TNFA and NOS2 (p = 0.00007 and 0.0014 respectively) compared to MDA-MB-231 cells not treated with breastmilk (Fig. 4C). Taken together, this data suggests that exposure of breast cancer cells to breastmilk affects cancer-macrophage interactions that lead macrophages to reduce tumour-promoting activity and adopt a tumour suppressing M1 phenotype.
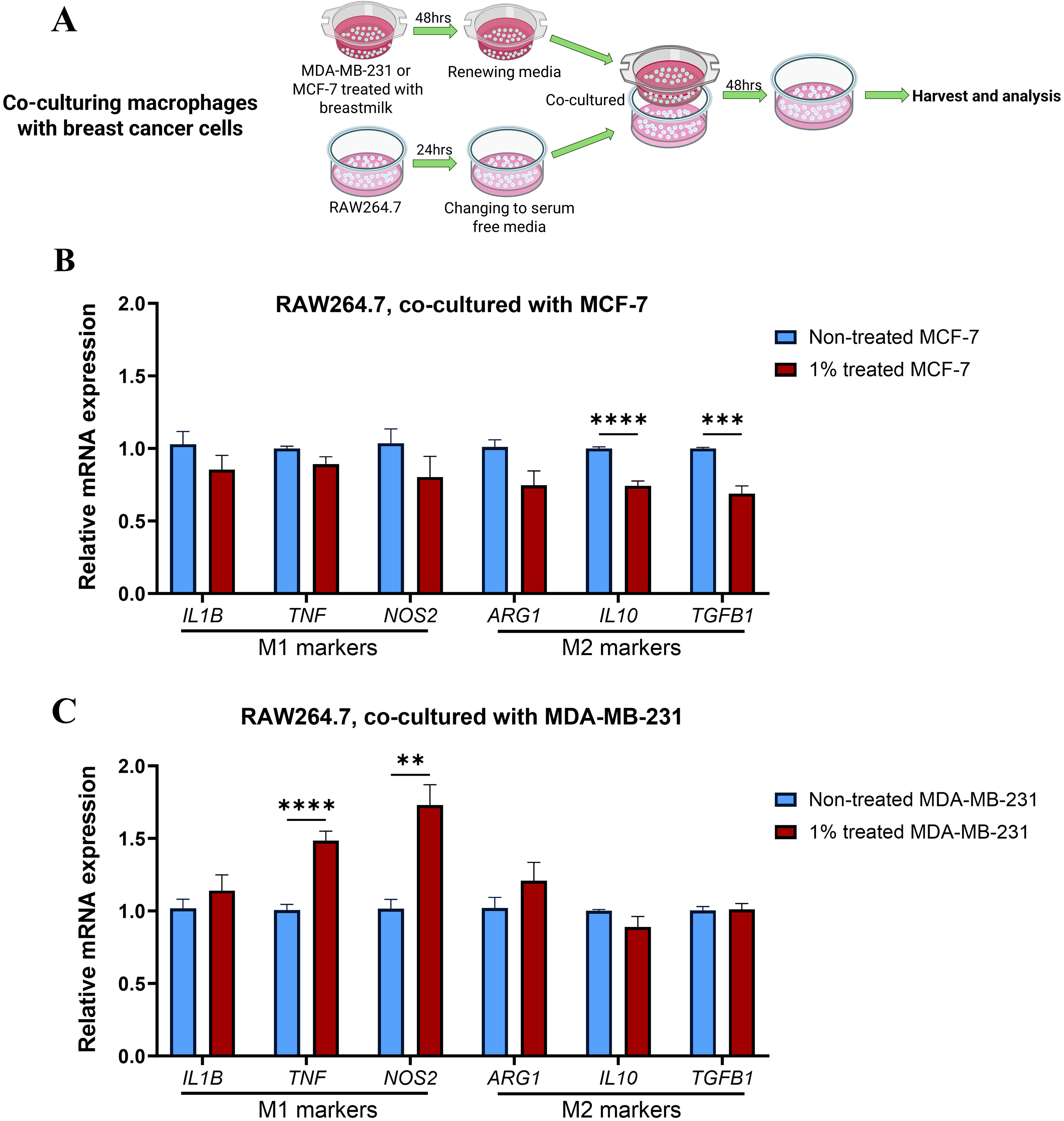 Fig. 4.
Fig. 4.
Breastmilk-treated breast cancer cells modulate macrophages
towards M1 phenotype. (A) Schematic diagram of co-culturing macrophages with
milk-treated breast cancer cells. (B) Shows differential expression of
inflammatory markers when RAW264.7 cells co-cultured with pre-treated MCF-7 cells
with 1% of breastmilk. IL10 and TGFB1 showed a significant
decrease in their expression compared to non-treated group. (C) Co-culturing
RAW264.7 cells with treated MDA-MB-231 (1% breastmilk) resulted in a significant
increase in the expression of TNF and NOS2 genes. Data
presented as mean+SEM of three independent experiments each with triplicate
wells. Unpaired student’s t-test was used to compare treated groups and
non-treated (**p
To further investigate the role of breastmilk in breast cancer progression, the
migratory properties of breast cancer cells incubated with breastmilk was
assessed using a migration assay. Breastmilk did not affect migration of MCF-7
cells in comparison to cells that were not incubated with breastmilk (Fig. 5A).
However, when MDA-MB-231 cells were incubated with breastmilk in their culture
medium, wound healing was significantly impaired by more than 50% compared to
untreated cells (p
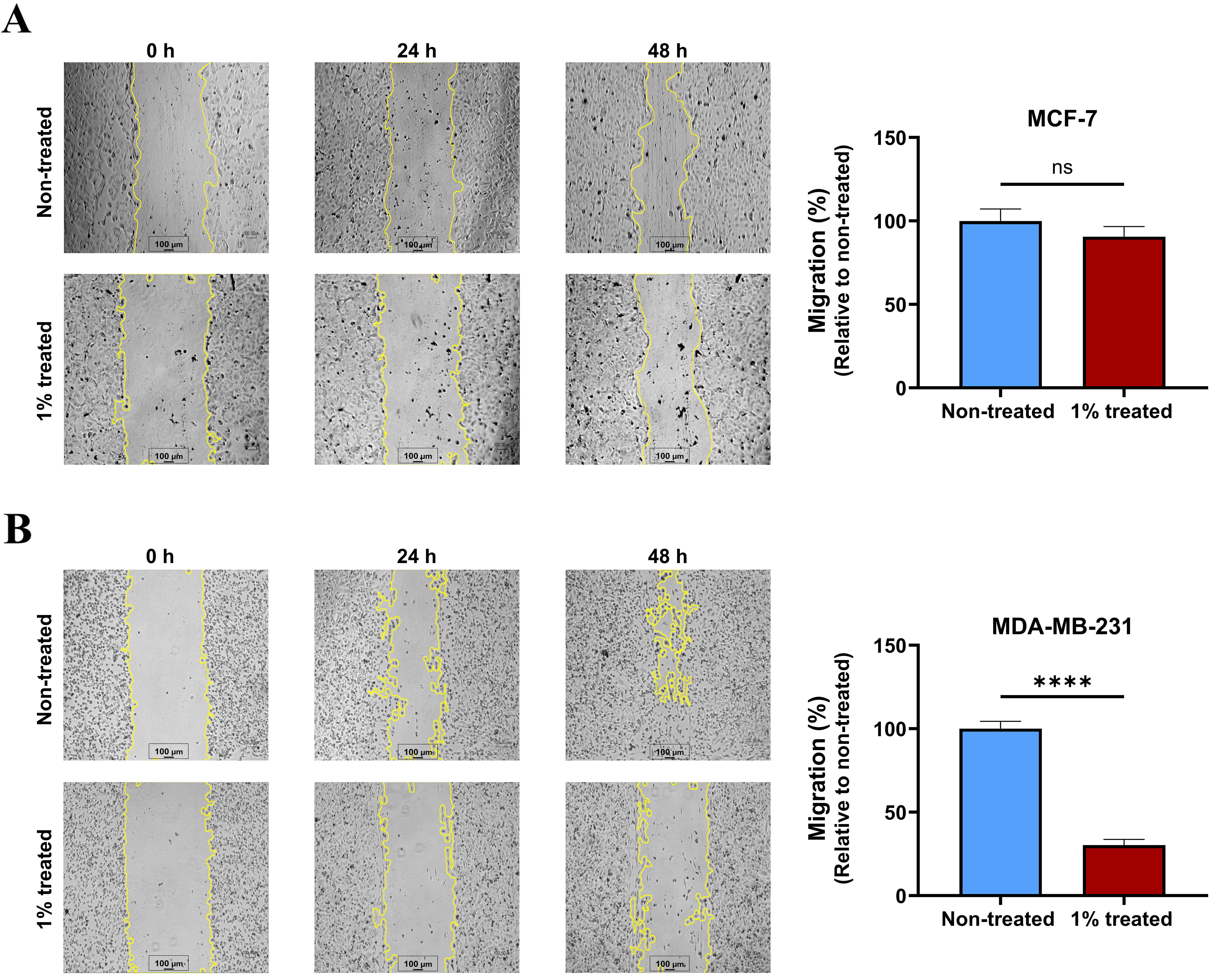 Fig. 5.
Fig. 5.
Breastmilk reduced migration of MDA-MB-231 cells. (A) Displays
wound healing photos captured every 24 hours, as well as the healing area
assessed in MCF-7 cells after 48 hours with and without breastmilk
supplementation. (B) Quantification of wound healing in MDA-MB-231 revealed that
cells treated with milk had a significantly lower capacity to close the wounded
area compared to cells that were not treated. Data presented as mean+SEM of three
independent experiments each with duplicate wells. Unpaired student’s t-test was
used to compare treated groups and non-treated (ns, not significant;
****p
There is substantial epidemiological evidence that breastfeeding reduces the incidence of breast cancer and may also prevent the development of postpartum breast cancer in some capacity [31, 32]. Breastmilk is a complex fluid containing over 1000 proteins including many physiologically active substances [33]. As breastmilk originates inside of the mammary glandular epithelium, it has the capacity to directly interact with cells in the ducts and lobules of the breast during the postpartum period and affect breast cell activity. The secretory characteristics of lactating breast epithelial cells could provide insight into associations between breastfeeding and breast cancer risk [34]. Therefore, investigation of the effect of breastmilk on breast cancer cells could provide valuable data on how lactation affects cancer development and progression during the postpartum period.
Due to the lack of in vitro models of postpartum breast cancer, we used a hormone dependent and independent breast cancer cell line to investigate the effect of breastmilk on breast cancer cell activity. The hormone-sensitive breast cancer cell line MCF-7 is well recognised and extensively studied in breast cancer research [35]. The hormone-independent triple negative breast cancer cell line MDA-MB-231 displays more metastatic properties than MCF-7, in accordance with the high aggressiveness of postpartum breast cancer [36]. Due to the absence of an inherently malignant human macrophage cell line, we used the RAW264.7 cell line, which is widely recognised as a primary murine macrophage cell line in cancer studies [37].
Given that there is limited research on the effective amount of breastmilk required to impact cancer cell activity, we first opted to investigate the concentration and timeframe of optimal cell proliferation and viability to inform further investigations. Results from these experiments revealed that breastmilk exhibits stimulating effects on proliferation and viability of cells in vitro across different time points and concentrations. This was not surprising as breastmilk contains many nutrients, hormones, and growth factors [38]. These results align with previous research conducted in various illness contexts, indicating that breastmilk has the potential to promote cell proliferation [39, 40]. For example, the study conducted by Meng et al. [41] revealed that application of breastmilk-derived extracellular vesicles to C2C12 myoblast cells and mouse quadricep muscle resulted in significant improvements in myotube diameter and myofiber growth stimulation. These results suggest that breastmilk may have broad stimulatory effects in supporting growth of breast cancer cells. However, we sought to further investigate the role of breastmilk in tumour-macrophage interactions and breast cancer progression. In the following experiments, a 1% concentration of breastmilk over a 48-hour period was used, as these conditions were sufficient to elicit a notable response in cancer cells and macrophages.
The effect of breastmilk on expression of epithelial and mesenchymal markers in breast cancer cells was evaluated, as prior research suggested breastmilk may influence cancer migratory capability [14]. While there was no discernible effect of breastmilk on EMT marker expression in MCF-7 cells, changes in gene expression consistent with reduced capacity for EMT was observed in MDA-MB-231 cells. Expression of the gene encoding CDH2, which causes loss of adhesive properties in epithelial cells and acquisition of migratory and invasive characteristics was reduced following treatment with breastmilk [42]. Additionally, despite the fact that CDH1 expression is typically comparatively low in MDA-MB-231 cells and is not considered a significant EMT marker in these cells, breastmilk-treated MDA-MB-231 cells exhibited elevated expression, suggesting a possible shift towards an epithelial cell phenotype [43]. Consistent with this observation, breastmilk inhibited migration of MDA-MB-231 cells and did not affect migration of MCF-7 cells. This finding is similar to studies in mouse mammary Met-1 tumour cells, which is a triple-negative mammary cancer cell line, where breastmilk inhibited migration [15]. While the specific components of breastmilk with this inhibitory activity are unknown, one candidate is alpha-casein, which is abundant in breastmilk and has been shown to reduce migratory features in triple-negative breast cancer cells [44].
Breastmilk also had inhibitory effects on cytokine expression in the RAW264.7 macrophage cell line, with a decrease in mRNA encoding IL1B and IL10. This result is different to a study on breastmilk-derived oligosaccharides which demonstrated an increase in TNFA, reactive oxygen species, IL1B, IL2, IL6, prostaglandin E2, and nitric oxide in RAW264.7 cells, without impacting cell proliferation [45]. One potential explanation for this disparity is that the present study used whole breastmilk which contains a wider range of physiologically active constituents that might potentially influence the functioning of macrophages differently to oligosaccharides on their own.
More intriguing findings were discovered when the impact of breastmilk on communication between breast cancer cells and macrophages was investigated through co-culture studies. The mammary gland consists of a variety of cell types that create complex networks of interactions, which are crucial at various phases of tumor formation [46]. In our study on crosstalk between macrophages and breast cancer cells, we found that macrophages that had been exposed to breastmilk had a profound impact on breast cancer cell EMT markers. Upregulation of an epithelial marker (CDH1 in MCF-7 cells) and downregulation of mesenchymal markers (CDH2, VIM, and ACTA2 in MCF-7 cells; CHD2 and VIM in MDA-MB-231 cells) was observed. Overall, these studies suggest that breastmilk-exposed macrophages reduce aggressiveness of breast cancer cells.
One of the key aspects in cancer development and metastasis is the phenotypic transformation of macrophages which is influenced by cancer cells within the tumour microenvironment [47]. Here, observe that breast cancer cells exposed to breastmilk alter their communication with macrophages such that M1 pro-inflammatory markers (TNFA and NOS2 in MDA-MB-231 cells) are upregulated and M2 alternatively-activated markers (IL10 and TGFB1 in MCF-7 cells) are downregulated. TGFB1 is recognised as a key cytokine that dampens inflammatory macrophage activity and increases breast cancer susceptibility [48, 49]. Therefore, breastmilk may drive macrophages towards an M1 phenotype and away from an M2 phenotype, consistent with an anti-tumour immune response [50]. These studies infer that breastmilk has the potential to suppress breast cancer development and progression through regulating macrophage phenotype and function.
There are some limitations in our research. In our present study, we pooled breastmilk from seven breastfeeding mothers with no current medical conditions to investigate the impact of healthy breastmilk on breast cancer cell activity. Pooled samples enabled the study of the overall effects of breastmilk which was necessary to establish general trends. However, as breastmilk is a highly customised fluid, it is possible that variability in the components of breastmilk between individual women that could lead to variable results in the in vitro assays employed here. Future studies will delve more deeply into the effect of individual breastmilk samples, as well as specific components of breastmilk, on breast cancer cell and macrophage activity. Overall, more investigations are warranted to explore more in depth the impact of the diverse array of biological components in breastmilk that affect breast cancer development and progression.
This study suggests that breastmilk exhibits both pro-tumour and anti-tumour effects in breast cancer development and progression. Breastmilk promotes tumour development through increasing breast cancer cell proliferation. On the other hand, breastmilk exerts anti-tumour effects by reducing EMT of breast cancer cells through direct effects on breast cancer cells and also through indirectly modulating the activity of macrophages in the tumour microenvironment. In addition, breastmilk skews macrophage polarisation towards the M1 anti-tumour phenotype through influencing the activity of breast cancer cells that direct the function of tumour-associated macrophages. Thus, breastmilk could be considered as a fluid that shapes the tumour microenvironment in postpartum breast cancer.
The data and material supporting the findings of the current study are available from the corresponding author upon reasonable request.
EMT, epithelial-mesenchymal transition; TNFA, tumour necrosis factor alpha; IL1B, interleukin 1B; IL6, interleukin 6; IL10, interleukin 10; ACTA2, alpha-smooth muscle actin; OCLN, occludin; VIM, vimentin; ARG1, arginase 1; NOS2, nitric oxide synthase 2.
RM, AG, LEG and WVI designed the research study. RM and HH performed the research. RM analysed the data. RM wrote the first draft of the manuscript and all authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity.
The research protocol was approved by the Central Adelaide Local Health Network (CALHN) Human Research Ethics Committee (HREC) (CALHN Reference Number: 19523), and all of the participants provided signed informed consent.
Not applicable.
This research was funded by National Health and Medical Research Council of Australia APP2011845.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.fbl2909328.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





