1 Department of Biology, The Peace College, 24420 Charsadda, Khyber Pakhtunkhwa, Pakistan
2 Department of Biosciences, University of Wah, 47000 Wah Cantt, Punjab, Pakistan
3 Agronomy Department, University of Florida, Gainesville, FL 32608, USA
Abstract
The cultivation of halophytes is an alternative approach to sustain agricultural productivity under changing climate. They are densely equipped with a diverse group of metabolites that serve multiple functions, such as providing tolerance to plants against extreme conditions, being used as a food source by humans and ruminants and containing bioactive compounds of medicinal importance. However, some metabolites, when synthesized in greater concentration above their threshold level, are considered antinutrients. Widely reported antinutrients include terpenes, saponins, phytate, alkaloids, cyanides, tannins, lectins, protease inhibitors, calcium oxalate, etc. They reduce the body's ability to absorb essential nutrients from the diet and also cause serious health problems. This review focuses on antinutrients found both in wild and edible halophytes and their beneficial as well as adverse effects on human health. Efforts were made to highlight such antinutrients with scientific evidence and describe some processing methods that might help in reducing antinutrients while using halophytes as a food crop in future biosaline agriculture.
Keywords
- halophytes
- antinutrients
- bioactive compounds
- medicinal importance
- sustainable agriculture
Climate change-induced drought and salinity, along with population explosion, are contributing to an imbalance and unsustainable food patterns of Earth inhabitants. One strategy to fulfill food demand was the development of genetically modified crops with strong resistance against pesticides, and their consumption has improved human health [1]. The long-lasting consumption of plant-based food and nutrition has proven to reduce the risk of major chronic human diseases, such as myocardial infarction, major types of strokes, breast, prostate, and colorectal cancer, vertebrae and hip fractures, etc. [2, 3]. Plant-based diets (fruits, vegetables, nuts, seeds, whole grains, legumes, herbs, and spices) contain a plethora of bioactive compounds implicated to exhibit strong antioxidant capacity and induce cellular mechanisms to provide defense and reduce inflammation. Despite the fact that some issues have been raised about the presence of various compounds present in plant-based food, if ingested in excess amounts can affect human health, also reducing the intake, absorption, and utilization of other essential nutrients. Such compounds are categorized as antinutrients [4].
This study highlights the major antinutrients in edible and wild halophytes and compiles scientific evidence on their impact on human health. Moreover, it also examines their negative impacts. Then some processing methods were deciphered to reduce the composition of antinutrients in edible halophytes. The aim of the current study is to highlight the available scientific records of halophyte crops in order to evaluate the maximum nutritional value specifically and point out whether the antinutritional factor poses significant health risk when used as human food.
The chemical properties and effects on the utilization of nutrients are two major factors responsible for the classification of antinutrients. On the basis of chemical properties, four major groups have been recognized: (i) proteins (protease inhibitors, lectins), (ii) glycosides (saponins, cyanogens, glucosinolates), (iii) phenols (tannins, gossypol), and (iv) miscellaneous (antimetals, antivitamins) [5]. Other criteria for their classification are: (i) as elemental forms that reduce nutrient intake in organisms, (ii) as compounds occurring in food ingredients that affect the physiology, immunity, and reproductive ability of living organisms [6].
Plant-derived oxalate (C2O42-) is negatively charged ion that bind with positively charged ions (i.e., Na+, K+, Ca2+, Fe2+, Zn2+, and Mg2+) to form salt crystals. On the basis of solubility, oxalate crystals are categorized into two groups; Na, K, and NH4-oxalate being water-soluble, whereas Ca, Mg, Fe, and Zn forming oxalate crystals are partially or completely insoluble [7]. Due to their salt-forming ability, oxalate causes kidney stones and earns the name antinutrient. Oxalate is synthesized both in plants and in humans as a byproduct of cellular metabolism.
Salinity (800 mM NaCl) and metal stress (10 mg/kg Cd) stimulated the accumulation of oxalate (183.6%) in Kochia scoparia halophyte [8]. Similarly, different degrees of salinity (low and high level) also induced the formation of different shapes of oxalate crystals, as reported by Cuadra and Cambi [9]. Halo xerophytic species with druses were found at high salinity and Ca level, whereas polyhedral crystals of oxalate were found in species found in low salinity and low Ca concentration [9].
The mechanism followed by oxalate is a multifactorial and complex process. In the kidney, oxalate secretion is mediated by Solute Carrier Family 26 Member 6 (SLC26A6), located on the proximal side of the renal tubule of the nephron. The abnormal expression of SLC26A6 and hyperoxaluria were two factors that aid in the increased secretion of oxalate in renal epithelial cells [10]. The hyperaccumulation can trigger mitochondrial dysfunction and induce mitochondrial reactive oxygen species (ROS) and cytochrome c release into the cytoplasm. The ROS causes lipid peroxidation (injure cell membrane), whereas cytochrome c triggers various signaling pathways such as caspase activation that cause apoptosis. The apoptotic cell bodies and debris release into the tubular lumen and become part of the crystal aggregates [11].
Early studies have described the potential biological role of oxalate on human health, i.e., oxalate stimulates the absorption of Cl, H2O, and Na by the proximal tubule via SLC26A6 transporter protein, activate the release of phagocytes through H2O2 burst by oxalate oxidase enzyme [12]. Finally, oxalate is also required for the synthesis of uracil and orotic acid which are essential components of RNA involved in protein synthesis [13]. However, high intake of oxalate might cause kidney stones, low plasma level of Fe and Ca, reduced blood Ca level by forming insoluble calcium oxalate as well. Furthermore, nephrolithiasis, acute and chronic kidney disease, anemia, hyperphosphatemia, and hyperkalemia are disorders related to oxalate consumption [14, 15].
Lectins or phytohemagglutinin are a diverse group of sugar-binding proteins found in a wide variety of plant species with the highest abundance in the legume family. Structurally, they are classified into merolectins, hololectins (most plant lectins), chimerolectins, and super lectins [5]. On the basis of carbohydrate specificity, Ga/GaINAc-binding, GaINAc-binding, Fructose-binding, Sialic acid-binding, and Glucose/Mannose-binding lectins have been identified [16]. The final categorization is related to plant families, i.e., legumes, Type II Ribosome-Inactivating Protein (RIP), monocot mannose binding, chitin-binding, Cucurbitaceae phloem, and Amaranthaceae [16].
Lectins are an integral component of receptor proteins found in the plasma membrane of plant cells and involved in providing defense against biotic and abiotic stress. The jacalin-related lectin family is the third-largest lectin family in Oryza sativa. The jacalin-related lectin gene (OsJRL 40) confers salt tolerance in Oryza sativa L. Under high salt stress, the overexpression of OsJRL 40 lectin protein in roots and internodes enhanced antioxidant enzyme activities and regulated Na+ K+ homeostasis thus conforming salt tolerance in O. sativa [17]. The sensor and mediator mechanism of lectin at the plant cell surface is depicted in Fig. 1.
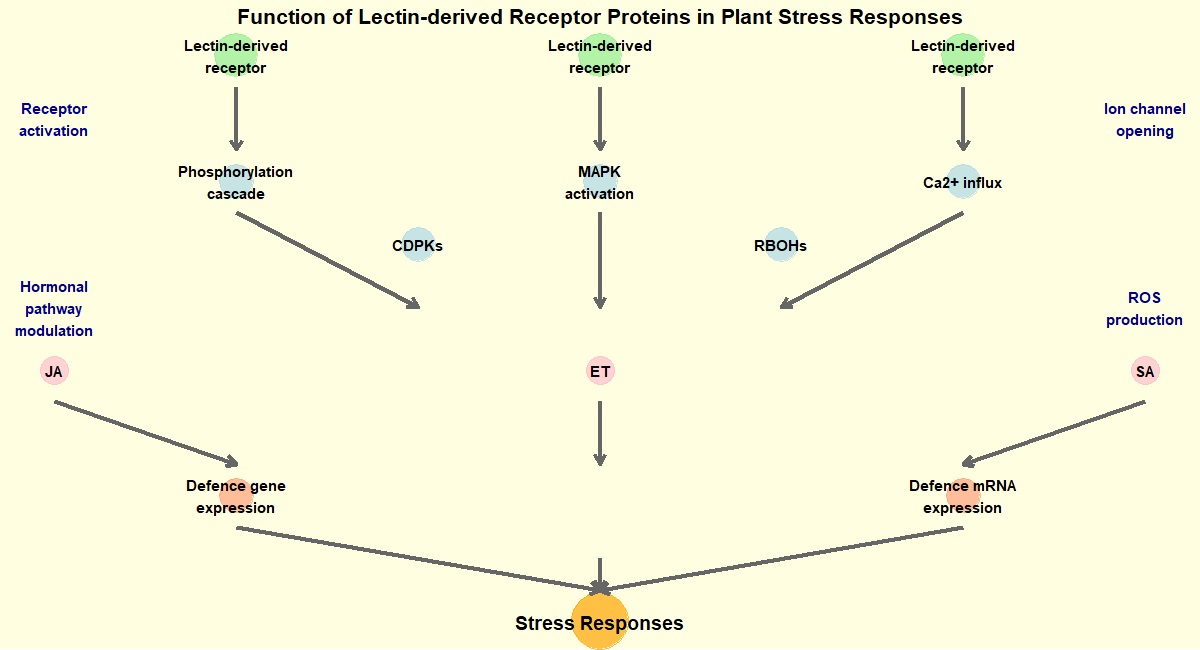 Fig. 1.
Fig. 1.
Function of Lectin derived receptor protein in stress responses of plants. MAPK, mitogen activated protein kinase; CDPKs, calcium dependent protein kinase; RBOHs, respiratory burst oxidase homologs; JA, jasmonic acid; ET, ethylene; SA, salicylic acid.
It is worth mentioning that lectins resist the activity of digestive enzymes, thereby interacting with intestinal epithelial cells and altering their permeability [18]. They also reversibly bind to specific oligosaccharide moieties and cause erythrocyte agglutination. As a sugar-binding protein, lectins cause agglutination of erythrocytes, hypertrophy, and hyperplasia of the pancreas, induce intestinal atrophy, alter intestinal mucosa permeability, and thus reduce nutrient absorption by affecting carbohydrate digestion [5, 19, 20]. The positive effects of lectins are also listed as lectins are immunity boosters and enhance vaccine efficacy [21, 22], i.e., concanavalin A; is used as a potent cancer therapy agent, lectins possess anti-infectivity properties against SARS-CoV-2 [23]. Therapeutic value against myeloid leukemia have also been reported [24].
Glucosinolates are a diverse class of 120 sulfur-containing plant secondary
metabolites, commonly known as
Phytoestrogens are non-steroidal phenolic secondary metabolites structurally similar to estradiol. Isoflavones, lignans, stilbenes, ellagitannins, and coumestrol are major phytoestrogens [33]. Isoflavones are flavonoids (i.e., genistein, daidzein, glycitein, and biochanin), lignans are polyphenolic, with the highest concentration in flax (Linum usitatissimum) seeds as secoisolariciresinol diglucoside, in cereals as matairesinol, also include pinoresinol, lariciresinol, and syringaresinol. The three halophytic grasses; Spartina maritima, Spartina patens, and Puccinellia maritima contained flavones, flavonols, and lignans [34]. Their extracts were particularly rich in trihydroxy methoxyflavone, apigenin, and tricin derivatives. Phytoestrogen has a great influence on the endocrine systfem. It has been reported previously that the utilization of phytoestrogens led to a goitrogenic effect and reduced level of insulin in non-alcoholic fatty liver patients [35]. Some other biological importance of phytoestrogens includes reducing the risk of cardiovascular disease in menopausal women and reducing the risk of myocardial infarction and prostate cancer [36, 37, 38].
Phytate or phytic acid is myo-inositol-1, 2, 3, 4, 5, 6-hexakis dihydrogen
phosphate. Structurally, it is composed of six phosphate groups attached to an
inositol ring. It is a major form of phosphorus reservoir in seeds (60–90%) of
the total phosphorus content). Srivarathan et al. [39] determined
phytate contents in three edible halophytes which were listed in the following
order; Sesuvium sp. (45.8 mg PA/g dry weight (DW))
Tannins are water polyphenolic metabolites with high molecular weight ranging from 500–3000 Da. The three well-known recognized categories of tannins include hydrolysable tannins (i.e., gallotannins, ellagitannins), condensed tannins/proanthocyanidins (i.e., catechins, epicatechins, epigallocatechin, epicatechin-3-gallate, and epigallocatechin-3-gallate), and complex tannins. The complex tannins are associated with proteins or carbohydrates. A wide variety of halophytes have been reported to contain various forms of tannins, i.e., the shoot extract of Glaucium flavum was analyzed for the presence of tannin compounds in different solvents [50]. The condensed tannins were enriched (19.83 mg Catechin Equivalents per Dry Weight (CE/DR) in the ethyl acetate extract of G. flavum. Similarly, seven principal phenolics were identified in three different fractions, i.e., Kaempferol (1.557 µg/g DR) in the petroleum fraction, and the highest level of caffeic acid (2.810 µg), chlorogenic acid (2.526 µg), catechin hydrate (30.664 µg), isoquercetin (27.395 µg)/g DR, and trans-hydroxy-cinnamic acid (20.904 µg/g RD) in the ethanolic fraction [50]. In another research, Limonium Spathulatum leaves are highly enriched with caffeic acids, coumaric acids, ferulic acids, gallic acids, syringic acids, catechin, epigallocatechin gallate, and naringin respectively [51].
The in vivo study has shown that tannins negatively affect the
activities of
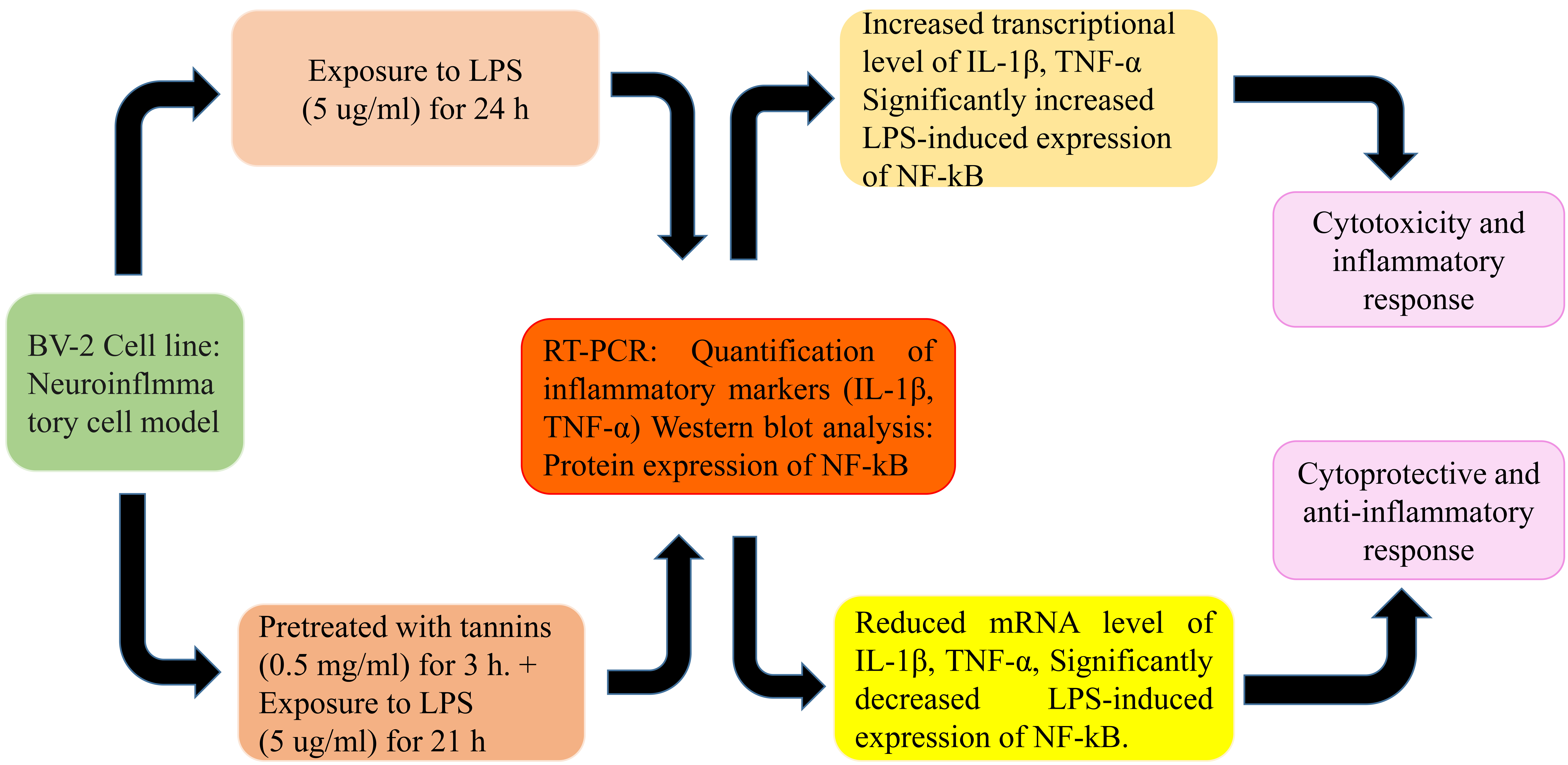 Fig. 2.
Fig. 2.
Cytoprotective and anti-inflammatory action mechanism
of tannins. LPS, lipopolysaccharide; IL-1
During the storage process, melanosis appears in white leg shrimp caused by polyphenol oxidase, thus depreciating their market value. The polyphenol extract of Portulaca oleracea contained melanosis inhibiting agents (isoorientins, isovitexin, and apigenin) and showed the highest inhibiting effect against dipolyphenol oxidase [61].
Saponins are plant secondary metabolites composed of a carbohydrate moiety (mono/oligosaccharide) and a lipid-soluble aglycone that is steroidal or triterpenoid in nature, contributing to plant defense against herbivores [33]. The highest saponin levels are present in Atriplex sp. (1246 mg), followed by Sesuvium sp. (635 mg) and Suaeda sp. (247 mg) OE/100 g dry weight [39]. In the methanolic extract of Bassia indica, about 25 different compounds were identified, categorized into lignans, steroids, amide alkaloids, coumarins, nucleic acid derivatives, phenolic glycosides, flavonoids, and triterpene oleanane saponins [47]. The saponin derivative displayed anti-inflammatory activity. Similarly, a new class of pentacyclic triterpenoid saponins was identified from the whole plant of Tripolium pannonicum, collectively named pannosides A-E [62]. Saponins showed anticarcinogenic properties, hypoglycemic, immune stimulatory, neurogenesis, and hypocholesterolemic effects [46, 47, 63]. The high dose of saponin glycosides over an extended period causes excessive secretion of saliva, diarrhea, and vomiting [64]. Saponins also inhibit the absorption of lipids in the intestine, suppress the accumulation of adipose tissue, and promote fecal excretion of bile acids and triglycerides [65]. Similarly, the increase and decrease in the generation of free radicals by H2O2 have also been reported [66]. The adverse effects of saponins utilization include hypocholesterolemic effect, hemolysis, indigestion disorder [67], affect mineral and vitamin absorption [68], hypoglycemic effect [69], anti-inflammatory, antitumor therapy [47], regulate blood glucose level and lipid profile in diabetes type 2 patients [63].
Raffinose family oligosaccharides (RFOs) are indigestible oligosaccharides that
include raffinose (trisaccharide), stachyose (tetrasaccharide), verbascose
(pentasaccharide), and adjugose (hexasaccharide) [70, 71]. Raffinose is formed
when galactinol donates galactose to sucrose, creating an
This class of antinutrients are proteinous in nature and exhibit enzyme
inhibition activity. They form enzyme inhibitor complexes, blocking the active
site or altering the enzyme configuration, thus reducing catalytic activity.
Examples are protease inhibitors,
A nitrogen-containing plant secondary metabolite that yields hydrogen cyanide (HCN) upon hydrolysis. The basic structure is composed of a central C atom attached to CN, R1 (alkyl or aryl group), R2 (alkyl group or H-atom), and mono or disaccharide through glycosidic linkage [84, 85]. About 25 to 75 types have been reported in the literature; however, major types include amygdalin, laminarin, linustatin, lotaustralin, prunasin, and taxiphyllin. The detailed study about the chemical diversity of cyanogenic glycosides has been discussed by Yulvianti et al. [86] in their literature review. Salsola is one of the largest genus of the family Amaranthaceae. Different types of cyanogenic glycosides have been reported in this genus. The LC-MS analysis of root extract of Cenchrus ciliaris exhibited the presence of cyanogenic hyperoside [87].
It is a monounsaturated omega-9 fatty acid composed of 22 carbon atoms, found in rich soil seeds mostly in the members of Brassicaceae. The seeds of Cakile maritima contain a significant amount of oil rich in erucic acid (20–22%). In another study, it is reported the least fraction % of erucic acid in three halophyte spp., Atriplex griffithii (2.5%), Haloxylon ammodendron (4.6%), and Salsola yazdiana (6.8%), respectively [88, 89]. The oxidation of triglyceride containing erucic acid in fats releases energy in mitochondria of muscle cells. However, in cardiac muscles, fats containing erucic acids are less oxidized, and their accumulation is associated with cardiotoxic potential, as the prolonged intake of erucic acid might be linked with myocardial lipidosis [13]. Many studies have also reported the beneficial effect of erucic acid. The in vitro analysis showed strong antibacterial activity of erucic acid against Borrelia burgdorferi and Borrelia garinii, with minimal bactericidal concentration Minimal Bactericidal Concentration (MBC)90 for stationary phase 0.75 and 0.70 mg/mL respectively. Similarly, the replication of influenza viral A strains was suppressed by the activity of erucic acid as expressed as IC50 in the range of 0.49–1.24 mM respectively [90].
Tecticornia is an Australian indigenous halophyte belonging to
subfamily Salicornioideae of family Amaranthaceae. This family
includes important food crops such as Spinacia oleracea, Beta
vulgaris, and Chenopodium quinoa. Different species of Samphire have
been reported to contain antinutrients in high concentration, i.e., T.
halocnemoides 1 contains hydrolysable tannins (1.2 mg Tannic Acid Equivalent/gm
DW), total saponins (158.7 mg Ortho-diphenols Equivalent/100 g DW) in T.
halocnemoides 3, Phytate contents (6.0 mg PA/g DW) in T. halocnemoides
2, T. halocnemoides 4, and T. halocnemoides 6, trypsin
inhibitor (0.3 Trypsin Units Inhibited/mg DW) in T. halocnemoides 1 and
T. halocnemoides 4 respectively [91]. Similarly, three edible
halophytes; Mesembryanthemum crystallinum, Crithmum maritimum,
and Triglochin maritima were characterized for the presence of bioactive
compounds. Twenty-five phenolic compounds were identified, apigenin with minor
one in Crithmum maritimum, and iso-orientin in Triglochin
maritima respectively [92]. The analysis of leaf extract of Limonium
spathulatum showed that it contained toxic factors such as trypsin inhibitor
(72–83%), alpha-amylase inhibitor (9–29.6%), gallocatechin, epigallocatechin,
astragalin, naringenin [19]. The phytochemical composition of seven cultivated
halophytes were determined for their nutritional purpose, the Sarcocornia
fruticosa and Mesembryanthemum nodiflorum were predominant in
flavonoids, while phenolic acids were detected in C. maritimum, and
Salicornia ramosissima [93]. The flavonols compound; rhamnetin hexosyl
pentoside for the first time was identified in S. fruticosa [93]. In one
study the antinutritional components of twenty-two halophytes were determined and
grouped into three antinutritive categories: high (
In ancient times, wild halophytes were used traditionally as herbs, vegetables, and for ethnomedicinal purposes. In recent years, climate change and population explosion have posed serious threats to agriculture productivity. Therefore around the globe, scientists are performing preliminary trials on edible and wild halophytes to develop novel crops that sustain the productivity under saline conditions in response to climate change. One such aim is based on determining the nutritive value of halophytes. Nutritive value reflects the digestibility and efficiency of nutrients used for animal production in terms of meat, milk, and wool production etc. It is mainly composed of crude protein, minerals, and metabolites [96]. The nutritive value of some selected halophytes are listed in Table 1 (Ref. [97, 98, 99, 100, 101, 102, 103, 104, 105, 106]). The diversity of halophytes (2500 to 3000 spp.) with a wide range of salinity tolerance (250–500 mM NaCl) in the form of euhalophytes, recretohalophytes, and pseudohalophytes provide an opportunity to use them as food crops. Therefore, their exploitation as food and nonfood crops could be a winning strategy in the saline world. A variety of halophytes have been reported to be used as food crops, i.e., Atriplex hortensis, Aster trifolium, M. crystallinum, Crithmum maritimum, Salsola korarovii, Mertensia maritima, C. quinoa, Salicornia sp., Inula carithmoids. Samphire may have the potential to be used as a functional food ingredient. These species have the highest value of fiber (46.8 gm/100 gm DW), Fe (41.5 mg/100 gm DW), Mg (1.28 gm/100 gm DW), Na (16.7 gm/100 gm DW), and fatty acid (palmitic, stearic, oleic, linoleic acid, alpha linoleic acid) [91].
| Species | Location and experiment type | Parts used | Proximate composition | Mineral contents | Reference |
| Arthrocnemum indicum, Halocnemum strobilaceum | Sebkha of Sidi el Heni (Tunisia). Wild halophytes. | Young shoots. | Moisture: 80.1, 77.4, total ash: 3.4–7.4, crude protein: 3.1–3.3, total fat: 2.6–3.3, total available carbohydrates: 3.2–3.1, total dietary fiber: 7.6–8.0 (g/100g FW). | Fe: 0.47–0.45, Cu: 0.29–0.71, Mn: 0.16–0.34, Zn: 0.48–0.76, Mg: 59.37–72.52, Ca: 109.50–174.92, Na: 122.01–518.62, K: 32.81–43.05 (g/100g FW). | [101] |
| Arthrocnemum macrostachyum | Mediterranean coastal land of Egypt. Wild halophytes. | Above ground parts. | Moisture: 16.68, total ash: 9.58, crude fibre (%): 17.55, lipid (%): 1.45, crude protein (%): 6.88, sucrose (mg/g DW): 2.87. | Na+: 27.81, K+: 64.21, Ca2+: 47.27, Mg2+: 13.25 (mg/g DW). | [104] |
| Cladium mariscus | Ria Formosa Lagoon near Faro, Southern Portugal. Wild halophytes. | Oven dried seeds. | Ash: 3.52, crude protein: 6.55, total lipids: 0.98, carbohydrates: 88.96 (g/100 g DW). Metabolizable energy: 390.81 (Kcal/100 g DW). | Ca: 138.57, K: 1164.31, Mg: 116.52, Na: 152.24, Fe: 3.37, Mn: 2.57, Zn: 0.86, Cu: 0.35, Cr: 0.07, Ni: 0.07 (mg/100 g DW). | [97] |
| Salicornia europaea | Urmia lake Rahmanloo and Gharagheshlagh, Iran, Middle East. Wild halophytes. | Powder samples of roots and shoots. | Protein: 17.2%, oil: 4.5%, dry matter: 38.5%. | Shoot mineral contents | [98] |
| Cl: 16.87, Na: 28.52, K: 22.77, Ca: 1.46, Mg: 5.92, Fe: 666.00, Mn: 28.94, Zn: 113.48, Cu: 24.37. | |||||
| Salicornia ramosissima, Sarcocornia perennis alpini | Morraceira island (Mondego estuary), Atlantic coast. Cultivated in salt marsh. | Aerial parts. | Moisture: 87.8–89.7 |
Na: 177–204 |
[100] |
| Salicornia bigelovii | ICBA-UAE. Mubarak Valley-Egypt. Field experiment (Nov 2019–Sept 2020). | Shoots and seeds. | Water content: 3.4–1.4%, fat: 19.1%, protein: 18.1% total carbohydrate content: 37.6–47.4%, total ash content: 4.2–6.2%, crude fibre: 8–9.4. | Genotypes grown at ICBA were characterized by higher Na+, Ca2+, P3, and lower concentrations of K+, Mg2+, Fe2+, Mn2+, Zn2+ compared to the ones obtained at Mubarak Valley. | [103] |
| Sarcocornia fruticosa | iBET, Oeiras, Portugal. Soilless cultivation system. | Whole plant parts. | Moisture: 86.6, total ash: 5.70, protein: 4.44, total fat: 0.30, carbohydrates: 0.06, total dietary fibre: 2.90 (g/100g FW). Energy (Kcal/100g FW): 26.30. | Salt: 2.80, Chloride: 4.84 (g/100 g FW). Na: 1120, Ca: 36.10, K: 400, Mg: 55.60, Cu: 0.110, Fe: 1.42, I |
[102] |
| Suaeda maritima | Cadiz Bay Natural Park, Spain. Grown in Saltmarsh. | Edible tips. | Moisture: 88.2 (g/100 g FW), ash: 35.4, proteins: 7.92, crude fat: 4.37, total dietary fibre: 19.0 (g/100 g DW). | Na: 230 |
[99] |
| Chenopodium murale, Heliotropium digynum, Salsola imbricata, Tribulus pentandrus | ICBA Campus-Dubai, UAE. Grown in wild habitat. | Aerial parts. | Moisture: 70.5–88.5, total ash: 6.57–6.98, total carbohydrates: 0.7–16.5, proteins: 3.09–5.72, fat: 0.36–0.7 (g/100 g), crude fibre (%): 0.071–0.018, energy (Kcal/100 g): 22–95. | P: 137–154, N: 29–9157, Zn: 36.7–642, Ca: 3753–6100, K: 438–6665 (mg/kg). | [105, 106] |
FW, fresh weight; DW, dry weight.
A recent study determined the nutritional composition and antinutrients of the profile of three important Australian indigenous edible halophytes, Atriplex sp., Suaeda sp., and Sesuvium sp. The highest fiber content (41.5 g/100 g DW) and folate (303 µg/100 g DW) were determined in Atriplex, whereas all AIEH contained considerable amounts of essential elements and trace elements (Ca, Fe) and protein (20.1 g/100 g DW) [39]. The main fatty acids were linoleic acid, alpha-linoleic acid, and palmitic acid. The highest ratio of vitamin C (157 mg/100 g DW), betaine and isobetaine were identified in Sesuvium. However, the antinutrients were lower than Spinacia oleracea, relative to Atriplex and Suaeda respectively [39].
In another study, it investigated the in vitro protein digestibility, bioaccessibility and intestinal absorption of minerals and trace elements in saltbush and samphire. The in vitro digestibility of samphire protein was higher than saltbush protein [107]. The in vitro bioaccessibility of Mg, Fe, and Zn was higher in freeze-dried halophyte powder, however, samphire test food digesta had the highest intestinal Fe absorption rate as compared to saltbush digesta exhibited the lowest (37.7 vs 8.9 ng/mL ferritin).
The leaves of halophytic species L. spathulatum contained the most abundant Cl, Na, Ca, Fe and Zn, neither alpha-amylase inhibitor nor phytic acid and toxic metals (Pb, Cd). The ethanol and hydroethanolic extracts had the highest capacity to chelate Fe and Cu and to inhibit lipid peroxidation which were associated with high flavonoids contents. So, it is suggested to be used in herbal products with the aim as food additive for preventing lipid oxidation of lipid-rich foods [19]. The Cladium mariscus L. seeds may be considered safe to use as a food ingredient. It contained a rich and diverse profile of metabolites having strong antioxidant and anti-inflammatory properties and had moderate capacity to inhibit enzymes related to neuroprotection and diabetes [97].
Study conducted in a greenhouse setting showed that the addition of 100 to 200 mM NaCl in irrigation water along with an increase in the proportion of N provided as NH4+ up to 100% vs decrease in the proportion of N provided as NO3 up to 0% significantly reduced the concentration of antinutrients oxalate in the shoot of M. crystallinum and Enchylaena tomentosa with concurrent reduction in shoot biomass [108]. Although application of N source in the form of NH4 hampers the accumulation of shoot oxalate concentration, it also adversely affects the biomass of edible halophytes. On the other hand, the application of N in the form of NO3 promoted the accumulation of oxalic acid as NO3 inhibits oxalic acid oxidase activity, preventing the breakdown of oxalic acid, resulting in the accumulation of oxalic acid in leaves and stem. Plants grown in harsh conditions trigger the synthesis of various bioactive compounds to cope with stress. Low organic carbon and relatively high CaCO3 also affect the concentration of antinutrients, i.e., two halophyte species, Arthrocnemum macrostachyum and Halocnemum strobilaceum naturally grown along the Nile Delta coast showed the highest content of phenolic compounds, tannins, and saponins contents [104]. A study conducted in China at the national level showed that leaf N concentration decreased significantly with increasing latitude, which was mainly driven by the mean annual temperature and mean annual precipitation. The leaf P concentration increased remarkably with increasing longitude, which was induced by the variation in soil total P content [109].
The accumulation of cyanogenic glycosides in floral parts of many plant species is influenced by the pattern of latitude. In one research work a correlation was established between latitudinal trends and level of floral cyanogenesis. It was investigated that whole floret cyanogenic content varied widely about tropical rain forest and temperate species, while two sub-tropical species; Macadamia tetraphylla, and Grevillea robusta had high floral cyanogenic glycoside content [110]. Harvesting seeds at the immaturity stage also affects the antinutrients composition of halophytes, seeds of C. maritima collected at immaturity stage showed decreased amounts of erucic acid as compared to mature seeds [111]. Salinity induced the accumulation of raffinose in sugar beet leaves when treated with 150 mM NaCl [72].
Variation in seasons is another factor that influences the antinutrients composition of halophytes. The shoots of Suaeda fruticosa change color from green to red-violet in response to varied seasons. It has been shown that red-violet shoots of S. fruticosa were richer in O-glycosylated and acylated flavonoids than green shoots [101]. Cadmium contaminated (30 mg/kg Cd) saline soil (400 mM NaCl) significantly increased the accumulation of oxalate (28.76%) in K. scoparia [8]. In recent research work, it has been reported that mixed crop systems alter only the flavonoid composition of Salicornia europaea and did not influence the nutritional profile when cultivated with salt sensitive Solanum lycopersicum, respectively [112].
The cultivation of halophyte crops under saline conditions is considered a key strategy to ameliorate salt stress. Saline soils contain high concentrations of soluble salts, which can include sodium chloride (NaCl), calcium sulfate (CaSO4), magnesium chloride (MgCl2), and sodium sulfate (Na2SO4). These salts can disrupt plant metabolism and nutrient uptake, leading to the generation of antinutrients. The orphan plant Portulaca oleracea is a promising biosaline crop enriched with phytochemicals and cultivated to improve the ecological balance in saline soils. The seeds pretreated with 600 mM salts have a high potential to recover germination ability in distilled water [113]. This plant has the ability to alter its metabolic profile following salt exposure. In one research study, about 132 different metabolites were quantified in the roots and leaves of P. oleracea under 100 to 200 mM salt stress [114]. These metabolites include 35 organic acids, 26 amino acids, 20 sugars, 14 sugar alcohols, 20 amines, 13 lipids and sterols, and 4 other acids. The diverse metabolic responses of P. oleracea highlight its potential in biosaline agriculture, where it can not only thrive in high-salinity environments but also contribute to soil health and agricultural sustainability.
To date, various methods and techniques have been developed to process or reduce the level of antinutrients in edible crops [5]. A general overview of some techniques is enlisted in Fig. 3. The most common methods are thermal and non-thermal treatment. In the former, autoclaving, steaming, boiling, roasting, blanching, and extrusion cooking are well studied, while non-thermal based on soaking, germination, dehulling, milling, and fermentation [115]. Due to heat, the plant cell wall disrupts, and antinutrients are leached out. Extrusion cooking is effective to denature enzyme inhibitors, whereas steam cooking is least effective in reducing the concentration of antinutrients.
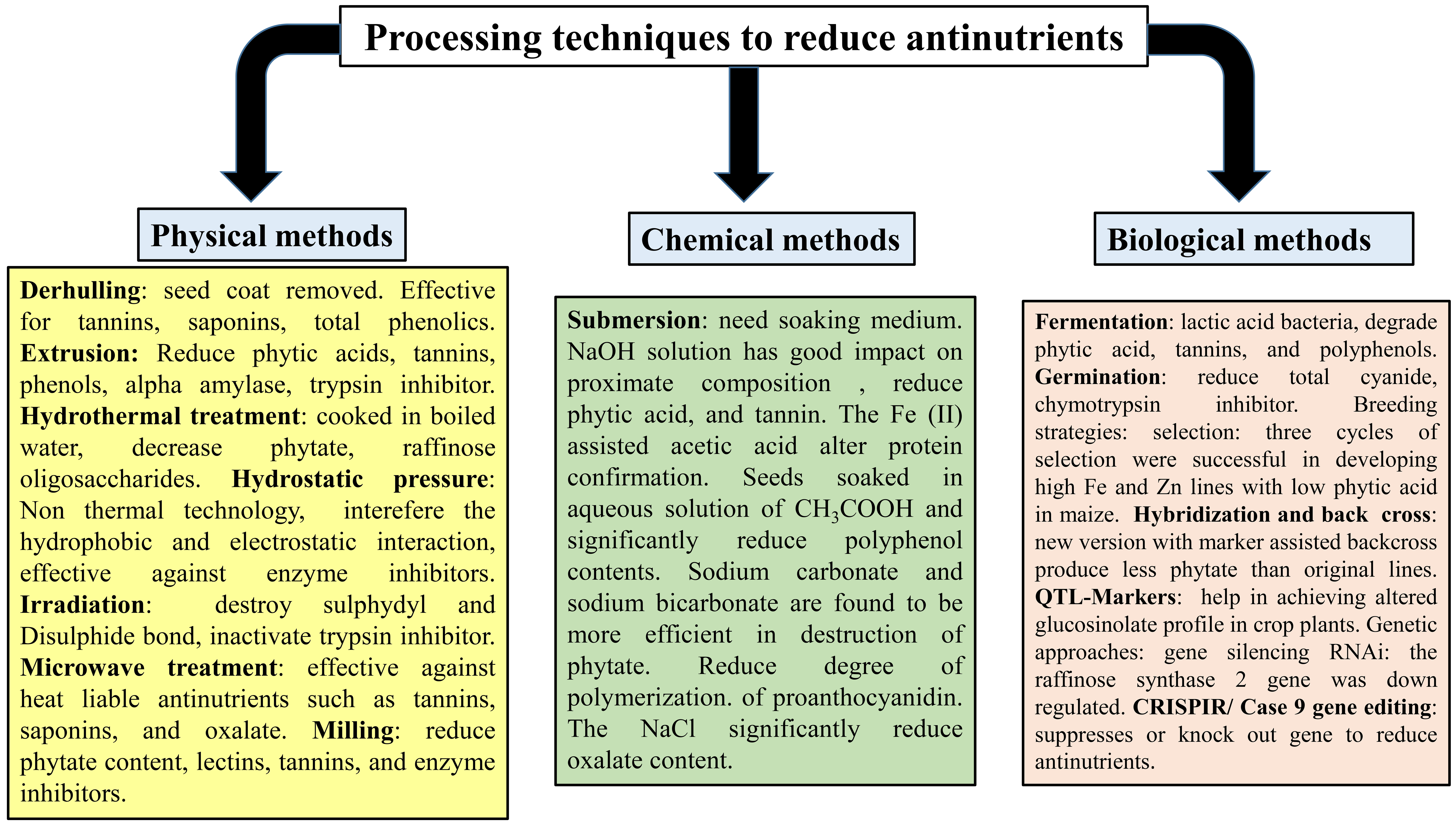 Fig. 3.
Fig. 3.
Schematic presentation of different control measures of antinutrients.
The soaking method is effective for leaching of water-soluble antinutrients [5]. The lactic acid bacteria (LAB) induced fermentation is effective for degradation of phytate, as LAB is implicated in the production of phytase which degrade phytic acid [116]. Similarly, spontaneous fermentation is also applied to reduce such antinutrients, however, the effect is not the same in response to different food ingredients, i.e., spontaneous fermentation significantly decreased the tannins and phytate content in Phaseolus vulgaris [117], whereas LAB was effective against maize flour. In some cases, fermentation leads to the formation of a newly bioactive compound; gamma-aminobutyric acid in soy sauce [118]. Food materials treated with ionizing radiation such as alpha, beta, gamma, and X-rays for a certain period of time is an effective strategy to reduce the level of antinutrients in most grains and beans. However, higher doses of irradiation might alter the nutritional composition. Some innovative techniques have also been developed which include; physical technologies, electromagnetic technologies, and acoustic technologies [119]. Physical technologies include high-pressure processing (HPP), electromagnetic technologies include dielectric heating treatment such as radiofrequency and microwave radiation. The effects of radiofrequency heating are more powerful than microwave heating. The new cold plasma processing is the fastest emerging technology and may replace thermal processing in reducing antinutrients while saving energy and retaining better nutrients content [120]. Pulse electric field technology (PEF) was primarily developed for food decontamination, but now it has been used to reduce the level of oxalate [121]. Additionally, PEF treated samples always showed a higher yield of extraction of flavonoids, and antioxidant activity within a shorter time [122] (Fig. 3).
Acoustic technology uses an audible frequency range to generate rarefaction and compression in food mediums that cause cavitation bubbles. Ultrasound technique is also helpful in reducing antinutrients. Ultrasound increased bubbles in microcavitation, thereby speeding up mass transfer, increasing the effectiveness of degradation of antinutrients. Ultrasonic processing is an environmentally friendly technology and does not affect the nutritional composition of foods. Supercritical fluid extraction (SFE) is another technique that uses fluid at critical temperature and pressure for the extraction of bioactive compounds from food ingredients. The extraction efficiency and antioxidant capacity of the SFE of Salicornia europaea seed oil using CO2 as a solvent were found to be higher than conventional extraction. Magnetic field treatment is also reported to reduce the antinutrients content in beans, while some antinutrients are increased in other seeds [123].
The most innovative approach is the use of enzyme-based treatments such as
phytase,
Halophytes are a potential novel crop, which is underutilized and have yet to be explored for their full potential. In the recent past, only a few selected species have been domesticated and developed as crop plants. The most prominent examples are C. quinoa, Salicornia, and Atriplex. However, several other halophytes have shown great potential as future crops and have attracted researchers’ attention worldwide. The future prospects of halophytes are discussed as follows:
Halophytes have the potential to be used as food crops to alleviate food insecurity in saline and arid regions. They can be grown in marginal lands where conventional crops cannot be grown. The domestication and cultivation of halophytes such as Salicornia, Suaeda, Atriplex, and Mesembryanthemum have shown promising results in terms of biomass production, nutritional value, and stress tolerance. These halophytes can be used to produce edible oil, protein-rich biomass, and other valuable products such as biofuels, bioactive compounds, and pharmaceuticals. Moreover, halophytes can be used as forage crops for livestock in saline areas, providing a sustainable source of feed in regions where conventional forage crops are limited [127].
Halophytes have the potential to be used for bioenergy production through the production of biomass for biofuels such as biodiesel, bioethanol, and biogas. Salicornia, Suaeda, and Atriplex are promising candidates for biofuel production due to their high biomass production, oil content, and stress tolerance. In addition to bioenergy production, halophytes can also be used for phytoremediation of saline soils and wastewater. Halophytes have the ability to accumulate salts and heavy metals from the soil and water, thereby improving soil fertility and water quality. This makes them valuable tools for mitigating soil salinity, reclaiming degraded lands, and treating wastewater from industries and agriculture [128].
Halophytes are well adapted to saline and arid environments, making them resilient to climate change and extreme weather conditions. As climate change continues to affect global food security and agricultural productivity, halophytes offer a sustainable solution for adapting to changing environmental conditions. By harnessing the genetic diversity of halophytes and breeding for desirable traits such as salt tolerance, drought tolerance, and high yield, we can develop resilient crop varieties that can thrive in saline and water-limited environments. This will not only enhance food security and livelihoods in marginal lands but also contribute to climate change mitigation and adaptation efforts globally [129].
Halophytes represent a unique and diverse group of plants that play a vital role in coastal ecosystems and saline habitats. They provide habitat and food for various wildlife species, including birds, insects, and microorganisms. By conserving halophyte diversity and preserving their natural habitats, we can protect biodiversity and ecosystem services in coastal and saline environments. This requires conservation efforts at local, national, and global levels, including the establishment of protected areas, restoration projects, and sustainable management practices.
In conclusion, halophytes hold great promise as a sustainable source of food, feed, bioenergy, and environmental solutions in saline and arid regions. Saline soils, characterized by high concentrations of salts such as sodium chloride, calcium sulfate, magnesium chloride, and sodium sulfate, pose significant challenges to traditional agriculture, but halophytes, with their ability to thrive in such environments, offer a viable alternative. These plants are equipped with diverse metabolites that provide tolerance to extreme conditions, making them valuable for biosaline agriculture. However, it’s crucial to consider the presence of antinutrients in halophytes, including terpenes, saponins, phytate, alkaloids, cyanides, tannins, lectins, protease inhibitors, and calcium oxalate, which can reduce nutrient absorption and cause health issues in high concentrations. Despite this, many of these compounds also have beneficial bioactive properties. By understanding and managing the levels of antinutrients through processing methods and selective breeding, we can maximize the nutritional benefits of halophytes while minimizing potential adverse effects. The cultivation and utilization of halophytes can significantly contribute to improving ecological balance, enhancing soil health, and providing a stable source of nutritious food.
AU: wrote the initial draft and generated figures; AB: study conception and supervised; AB and NK: design; edited and revised the MS. All authors approved the final version of the MS. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
Given his role as an Editorial Board member and a Guest Editor, Naeem Khan had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Changsoo Kim and Marcello Iriti. The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



