1 Fidia Ophthalmic Research, 95123 Catania, Italy
2 Department of Biology, University of Pisa, 56123 Pisa, Italy
§Retired.
Abstract
Oxidative stress, caused by the formation of free radicals, such as reactive oxygen species (ROS), leads to cell and tissue degradation, contributing to various diseases and aging. While oxygen is essential for aerobic organisms, it inevitably causes oxidative stress. Antioxidants protect against damage from free radicals, and oxidative stress arises when an imbalance occurs between free radical production and antioxidant defenses. However, when investigating whether an excess of antioxidants, almost eliminating oxidative stress, could benefit aging and disease susceptibility, it was observed that a basic level of oxidative stress appears necessary to maintain the correct homeostasis of tissues and organs and life in general. Therefore, this review aimed to compile the most significant and recent papers characterizing and describing the dual role of oxygen as a molecule essential for life and as a precursor of oxidative stress, which can be detrimental to life. We conducted targeted searches in PubMed and Google browsers to gather all relevant papers. We then focused on the eye, an organ particularly vulnerable due to its high metabolic activity combined with direct exposure to light and environmental pollutants, which produces a substantial number of free radicals (mainly ROS). We present a curated selection of relevant literature describing the main ocular pathologies of the posterior and anterior segments of the eye, highlighting oxidative stress as a significant contributing factor. Additionally, we report how endogenous and exogenous antioxidants can mitigate the development and progression of these diseases. Finally, we consider a frequently overlooked aspect: the balance between oxidants and antioxidants in maintaining the homeostatic equilibrium of tissues and organs. It is widely recognized that when oxidants overwhelm antioxidants, oxidative stress occurs, leading to negative consequences for the organism's homeostasis. However, we emphasize that a similarly dangerous situation can arise when the presence of antioxidants overwhelms the production of free radicals, drastically reducing their amount and adversely affecting aging and longevity. Unfortunately, no specific studies have addressed this particular situation in the eye.
Keywords
- oxidative stress
- antioxidants
- eye disease
- anterior segment
- retina
The relationship between oxygen and life is indeed paradoxical. Oxygen is vital for the aerobic life of nearly all metazoans, yet it also significantly contributes to their aging and susceptibility to ailments due to oxidative stress.
Oxygen is crucial for aerobic respiration, the process through which most higher organisms, including humans, extract energy from food [1]. During this process, oxygen acts as the final electron acceptor in the electron transport chain located in the mitochondria. This reaction produces ATP, the primary energy currency of the cell, starting from glucose and other nutrients [2]. Oxygen is also involved in numerous biosynthetic pathways, detoxification reactions, defense mechanisms, and metabolic processes essential for maintaining healthy cellular functions and growth [3].
However, despite its critical role, oxygen can also be detrimental, being responsible for what is known as oxidative stress. Reactive oxygen species (ROS), such as superoxide anions, hydrogen peroxide, and hydroxyl radicals, are produced as a byproduct of normal mitochondrial function and other cellular processes. ROS are essential for cell signaling and immune functions at low or moderate levels [3]. However, oxidative stress occurs when ROS levels exceed the neutralizing capacity of antioxidant defense systems. This imbalance leads to the oxidation of cell components, such as lipids, proteins, and DNA, disrupting normal cell function and increasing the risk of mutations and cellular death [4].
Oxidative stress has been closely linked to the aging process. It is believed to contribute to the gradual decline in physiological function observed during aging by damaging cellular components and accumulating cellular senescence. Accordingly, the rate of living theory suggests that the faster an organism’s metabolism, the shorter its lifespan, partly due to increased oxidative stress from higher oxygen turnover. Thus, organisms with high metabolic rates generate more ROS, potentially leading to accelerated aging and reduced lifespan [5]. Many chronic conditions, such as cardiovascular diseases, diabetes, cancer, and neurodegenerative diseases (like Alzheimer’s and Parkinson’s), are associated with oxidative damage catalyzed by excessive ROS production [6].
The body combats oxidative stress with an array of antioxidant mechanisms, both enzymatic (e.g., superoxide dismutase, catalase, glutathione peroxidase) and non-enzymatic (e.g., vitamin C, vitamin E, glutathione). The efficacy of these defense systems plays a significant role in moderating the aging process and influencing lifespan [7].
Therefore, the dual nature of oxygen as both essential for life and a contributor to aging and disease highlights a fundamental biological paradox. Research on balancing the beneficial roles of oxygen in metabolism with its damaging effects through controlled oxidative stress and enhanced antioxidant defenses may provide avenues for extending healthy lifespans and reducing the burden of age-related diseases. This ongoing exploration continues to be a critical study area in biology, medicine, and aging research.
The eye, due to its constant exposure to potentially harmful light radiation and the intense metabolic activities occurring in its tissues, is subjected to continuous insults by free radicals, progressively damaging most of its structures, from the ocular surface up to the retina and the optic nerve (Fig. 1). There is a well-documented link between high concentrations of free radicals, resulting from a combination of genetic and environmental causes, and the occurrence of eye diseases affecting both the anterior and posterior segments. In this context, it is noteworthy to report two recent findings that link the expression of specific microRNAs (miRNAs) as potential biomarkers. The first study [8] investigated the link between miRNA expression and the oxidative stress signaling pathways in diabetic retinopathy (DR), a complication of diabetes characterized by damage to the retina due to prolonged high blood sugar levels, leading to oxidative stress and inflammation. miRNAs, small non-coding RNAs that regulate gene expression post-transcriptionally, play critical roles in various cellular processes, including the response to oxidative stress. In the context of DR, chronic hyperglycemia leads to increased ROS production, contributing to retinal damage and disease progression. This study highlights that specific miRNAs are implicated in regulating oxidative stress pathways in DR. Thus, changes in the expression levels of these miRNAs can influence the progression and severity of diabetic retinopathy. The research identified key signaling pathways affected by miRNAs, providing insights into potential therapeutic targets for DR. Furthermore, it underscored the complex interactions between miRNA expression and oxidative stress, suggesting possible avenues for developing miRNA-based therapies to mitigate the effects of diabetic retinopathy.
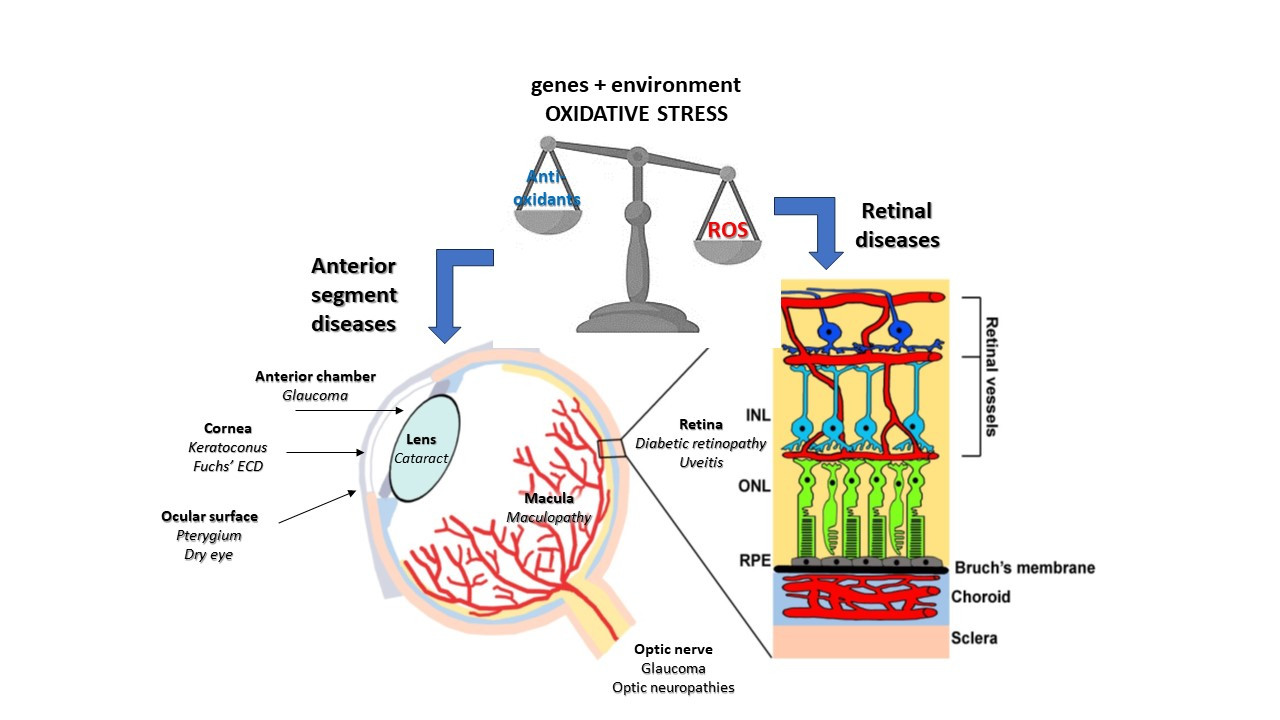 Fig. 1.
Fig. 1.
The effects of oxidative stress on the eye. ROS, reactive oxygen species; ECD, endothelial cell dystrophy; INL, inner nuclear layer; ONL, outer nuclear layer; RPE, retinal pigment epithelium.
The second study explores the complex interactions between miRNAs, ocular hypertension, and primary open-angle glaucoma (POAG) [9]. The researchers highlight that miRNAs play a significant role in the pathogenesis of POAG: by influencing oxidative stress pathways, miRNAs contribute to cellular damage in the eye. The authors identify specific miRNAs that are differentially expressed in ocular hypertension and POAG, suggesting that these miRNAs may serve as biomarkers or therapeutic targets. In addition to miRNAs, the study examined genes involved in oxidative stress, inflammation, and apoptosis networks. These genes are crucial in the progression of ocular diseases. The authors present evidence that the interplay between these genetic and molecular factors contributes to the development and progression of POAG from ocular hypertension. Overall, the article emphasizes the importance of miRNAs and related gene networks in mediating oxidative stress, inflammation, and apoptosis. This research might open new avenues for potential therapeutic interventions targeting these molecular pathways.
Starting from the posterior segment of the eye, the retina stands as the light-sensitive neural tissue lining the back surface of the eye. This tissue enjoys immune privilege, housing a defense system comprising retinal immune cells and the complement system [10]. Under normal aging conditions, the retina undergoes low-grade activation capable of eliminating products of oxidative metabolism. Nonetheless, there are cases in which an unbalanced equilibrium between oxidative metabolism and antioxidant defense may cause pathologic situations in the eye.
Glaucoma is a neurodegenerative disease characterized by the progressive loss of retinal ganglion cells (RGCs) and optic nerve damage in which oxidative stress may play a significant role in its pathogenesis and progression. In glaucoma, there is a complex relationship between intraocular pressure (IOP) and oxidative stress. The elevated IOP typically found in hypertensive glaucoma can be, on the one side, the reason for mechanical damage and loss of RGC, and on the other side may contribute to enhanced oxidative stress through impaired blood flow, inflammatory response, and mitochondrial dysfunction [11, 12]. Mitochondrial dysfunction is central to the role of oxidative stress in glaucoma. RGCs and other retinal cells in glaucoma exhibit mitochondrial abnormalities, including reduced membrane potential and impaired respiratory chain function, leading to increased ROS production [13]. Moreover, glaucomatous eyes often have impaired antioxidant defense mechanisms; thus, decreased levels of endogenous antioxidants, such as glutathione, superoxide dismutase, and catalase, leave cells vulnerable to oxidative damage. This imbalance between ROS production and antioxidant capacity contributes to the accumulation of oxidative stress in the glaucomatous eye [14]. Oxidative stress also induces inflammatory responses in the retina and optic nerve. Activation of inflammatory pathways produces proinflammatory cytokines and chemokines, exacerbating neuronal damage and contributing to RGC apoptosis and optic nerve degeneration [15]. Moreover, oxidative stress promotes neurodegeneration by damaging cellular components. ROS-mediated DNA damage can lead to mutations and cell death, while oxidative modification of proteins impairs their function and promotes apoptotic signaling pathways. Lipid peroxidation, initiated by ROS, disrupts cell membranes and compromises neuronal integrity [16]. Conversely, oxidative stress can also influence IOP levels in glaucoma by impairing the function of the trabecular meshwork, the tissue responsible for regulating the outflow of aqueous humor from the eye [17]. Dysfunction of the trabecular meshwork can lead to increased resistance to aqueous outflow and elevated IOP. Oxidative stress may directly induce damage to RGCs and their axons, thus disrupting neurovascular coupling and impairing the production of nitric oxide (NO), a key regulator of aqueous humor dynamics [18]. Dysregulation of NO signaling can contribute to alterations in IOP homeostasis.
If mitochondrial dysfunction can directly affect retinal cells in glaucoma, in additional retinal dystrophies characterized by pathological vessel proliferation, dysregulated ROS production due to the anomalous growth of leaky vessels in the posterior segment of the eye leads to retinal cell degeneration [19]. The oxygen-induced retinopathy (OIR) model is commonly used to mimic retinopathy of prematurity (ROP) in preterm infants, but pathologically, it displays similar features to adult retinopathies, including proliferative diabetic retinopathy (PDR) and age-related macular degeneration (AMD). OIR is generated through early exposure of neonatal mice to 75% oxygen, followed by a return to room air. Hyperoxia leads to the regression of blood vessels from the central retina, in which endothelial cell apoptosis triggered by oxidative stress is implicated in vaso-obliteration. Ischemia-induced microvascular abnormalities create a hypoxic condition, activating the proangiogenic pathway and forming new blood vessels that may protrude into the vitreous. The OIR retina provides an excellent model for studying proliferative diseases because its pathological angiogenesis resembles human diseases in which retinal cell degeneration can be indirectly driven by hypoxia-induced retinal vessel growth [20]. Among proliferative retinopathies mimicked by the OIR model, ROP is characterized by abnormal vascularization due to preterm birth [21]. The exposure of the still incompletely vascularized retina to the relatively hyperoxic extrauterine environment induces the downregulation of proangiogenic factors, interrupting retinal vascularization [22]. Indeed, a newborn faces oxidative stress at birth because of the rapid transition from the low-oxygen environment in the womb to the higher oxygen levels outside. During this period, the newborn is vulnerable to oxidative stress due to the underdeveloped efficiency of its antioxidant defenses. In the subsequent weeks, the increasing metabolic demands of the oxygen-deprived retina cause ongoing hypoxia, which stimulates the production of proangiogenic factors and results in harmful blood vessel growth.
Among adult neovascular diseases, PDR represents the most advanced stage of DR, characterized by thin and weak blood vessels whose bleeding can cause scar tissue, leading to retinal detachment [23]. Several factors contribute to the pathogenesis of PDR; however, oxidative stress and inflammatory processes are major. In PDR, hyperglycemia is a significant amplifier of oxidative stress, causing dysregulation of cell metabolism and limiting antioxidant defenses [24]. Epigenetic changes induced by hyperglycemia suppress the body’s antioxidant defense mechanisms, resulting in an imbalance between ROS removal and production. The excessive build-up of ROS leads to mitochondrial damage, cell death, inflammation, lipid peroxidation, and structural and functional changes in the retina. Current treatments for PDR focus only on the later stages, particularly the phase involving abnormal blood vessel growth. However, oxidative stress-related abnormalities present numerous potential therapeutic targets for developing safe and effective treatments for DR. Therefore, it is crucial to address the early ischemic stages, including oxidative stress.
In addition to DR, neovascular age-related macular degeneration (nAMD) is characterized by the abnormal growth of choroidal vessels that penetrate the subretinal space through Bruch’s membrane. This choroidal neovascularization (CNV) leads to fluid leakage, which causes significant retinal damage and subsequent cell death [25]. AMD remains the most commonly diagnosed condition, followed by PDR, with both disorders exhibiting similar underlying mechanisms, such as disrupted angiogenic balance and inflammation triggered by oxidative stress. In proliferative retinopathies, blood accumulates between the neurosensory retina and the retinal pigment epithelium (RPE) or between the RPE and the choroid. Blood breakdown products subsequently damage photoreceptors and the RPE, leading to poor vision and blindness. Antioxidants are crucial for protecting cells from oxidative stress, helping to prevent the progression of diseases, and maintaining vision in many age-related eye conditions, especially AMD. Consequently, incorporating micronutrients with strong antioxidant properties can be a promising approach for preventing and treating eye diseases linked to aging [26]. Much work is being performed to address antioxidant therapy for AMD. However, anti-vascular endothelial growth factor (VEGF) therapy remains a major treatment for most eye diseases associated with VEGF-related vessel proliferation [27, 28]. Yet, despite the encouraging outcomes observed with antioxidant compounds in animal studies, two extensive multicenter trials, namely the Age-Related Eye Disease Study (AREDS) and its follow-up AREDS2, did not demonstrate compelling evidence of antioxidant supplementation effectively benefiting individuals with AMD [29, 30].
Oxidative stress also plays a crucial role in the pathogenesis of several hereditary retinal dystrophies, including Leber congenital amaurosis (LCA), Leber hereditary optic neuropathy (LHON), Stargardt disease, and retinitis pigmentosa (RP).
In LCA and LHON, the mechanisms and contribution of oxidative stress to disease pathogenesis may differ between these conditions. In LCA, oxidative stress may exacerbate retinal cell dysfunction and degeneration caused by genetic mutations, whereas in LHON, oxidative stress is more directly linked to mitochondrial dysfunction and RGC degeneration [31].
LCA is a group of rare inherited retinal disorders characterized by severe visual impairment or blindness at birth or within the first few months of life. Oxidative stress is thought to play a role in the pathogenesis of LCA, particularly in cases where mutations affect genes involved in phototransduction, visual cycle, or retinal development. Specifically, RDH12 gene mutations, associated with a subset of LCA, have been shown to cause oxidative stress. RDH12 is responsible for reducing all-trans-retinal (atRAL) to all-trans-retinol (atROL) in the visual cycle, and mutations in this gene lead to the accumulation of atRAL, which is highly reactive and induces oxidative stress [32, 33]. In LCA, mutations in various genes may lead to dysfunction or degeneration of photoreceptor cells and RPE, which are highly susceptible to oxidative damage due to their high metabolic activity, constant exposure to light, and abundant polyunsaturated fatty acids. Oxidative stress may exacerbate the underlying genetic defects in LCA by causing further damage to retinal cells through mechanisms such as lipid peroxidation, protein oxidation, DNA damage, and mitochondrial dysfunction. Therapeutic strategies to reduce oxidative stress, such as antioxidant supplementation or gene therapy to enhance antioxidant defenses, are being explored as potential treatments for LCA [33].
Oxidative stress is also involved in the development of LHON, primarily through its impact on mitochondrial dysfunction, which ultimately leads to cell death via apoptosis, autophagy, and inflammation [31]. LHON is marked by the degeneration of RGCs and the optic nerve, manifesting as a sudden and typically severe bilateral loss of central vision. This condition is linked to three main mitochondrial DNA (mtDNA) mutations that cause missense mutations in NADH dehydrogenase, a crucial part of a large multienzyme complex responsible for ATP production through electron transfer and oxidative phosphorylation. LHON’s pathological characteristics include small-caliber axonal demyelination and loss, fiber swelling, and abnormal mitochondria. Proposed mechanisms of pathogenesis include reduced ATP synthesis, increased oxidative stress, and impaired glutamate transport, all contributing to RGC dysfunction and, ultimately, leading to apoptotic cell death. Due to the intricate nature of the structural proteins and pathways involved in mitochondrial functions, modern medicine has seen limited progress in developing effective therapies for mitochondrial diseases.
RP is the leading cause of inherited blindness among retinal diseases, with some rare but severe forms beginning as early as 4 years of age due to mutations in the gamma subunit of phosphodiesterase 6 (PDE6). Research in humans and mice has demonstrated that RP initially manifests as the progressive death of photoreceptors, which then triggers the degeneration of cone photoreceptors. Subsequently, alterations in downstream neurons, the surrounding RPE, and blood vessels accompany this process. In RP, prolonged oxidative stress can result in oxidative damage, further hastening the death of cone photoreceptors [34].
Alongside RP, Stargardt macular degeneration is another genetic eye disorder that results in central vision loss due to lipid buildup in the macula. The visual process begins with the photoisomerization of 11-cis-retinal to its all-trans-form, which is then converted back to 11-cis-retinal to regenerate rhodopsin or cone opsins. Despite being crucial for vision, retinal can be highly toxic to cells because of its reactive aldehyde group and ability to diffuse out of the outer segment compartment. To mitigate this toxicity, retinal is sequestered within a protein and reduced to retinol. The ATP-binding cassette transporter ABCA4 is vital for eliminating toxic byproducts, and mutations that impair its function cause an accumulation of retinoid compounds in photoreceptors and RPE cells [35]. These mutations lead to a range of conditions classified as Stargardt disease, which is marked by central vision impairment and the accumulation of lipofuscin and bis-retinoid compounds in the macula. In both RP and Stargardt disease, additional genetic mutations that cause metabolic disturbances or reduce cellular resilience to oxidative stress contribute to the progressive degeneration of photoreceptors and RGCs, ultimately leading to blindness [36].
Transitioning from the retina toward the anterior eye segment, oxidative stress also plays a significant role in the pathogenesis of various ocular surface diseases, the uvea, and the lens, contributing to tissue damage, inflammation, and disease progression.
Conditions such as dry eye disease (DED), blepharitis, and ocular surface inflammation are characterized by disruption of the tear film, inflammation of the ocular surface tissues, and symptoms such as dryness, burning, itching, and redness. Oxidative stress is implicated in the pathogenesis of ocular surface diseases through several mechanisms. Tear film instability and hyperosmolarity in DED can lead to increased production of ROS and oxidative damage to corneal and conjunctival epithelial cells. Inflammation and immune responses on the ocular surface generate ROS as byproducts, contributing to tissue damage and perpetuating the inflammatory cycle [37]. Environmental factors such as ultraviolet (UV) radiation, air pollution, and cigarette smoke exposure can induce oxidative stress and exacerbate ocular surface inflammation. Antioxidant defense mechanisms in tears and ocular surface tissues help counteract oxidative stress, but their efficacy may be overwhelmed in conditions of chronic inflammation or environmental stress. Therapeutic strategies for ocular surface diseases often include interventions to reduce oxidative stress, such as topical antioxidant eye drops, omega-3 fatty acid supplementation, and lifestyle modifications to minimize environmental exposures [38].
Uveitis refers to inflammation of the uveal tract, which includes the iris,
ciliary body, and choroid, although inflammation can also involve adjacent ocular
structures, such as the retina and optic nerve. Oxidative stress is a key
contributor to the pathogenesis of uveitis, amplifying inflammatory responses and
causing tissue damage in the affected eye [19]. Inflammatory cells infiltrating
the uveal tissues release ROS and reactive nitrogen species (RNS), leading to
oxidative damage to endothelial cells, fibroblasts, and other resident cells.
Oxidative stress activates nuclear factor kappa B (NF-
Cataract is a common age-related condition characterized by clouding of the crystalline lens, leading to visual impairment or blindness if left untreated. Oxidative stress is a major contributor to cataract formation, particularly in age-related and oxidative stress-induced cataracts [42]. Accumulation of ROS and oxidative damage to lens proteins, lipids, and DNA disrupts lens transparency and homeostasis, leading to protein aggregation, lens fiber cell disorganization, and, ultimately, cataract formation. Oxidative modification of lens proteins, such as oxidation of sulfhydryl groups and formation of protein-protein crosslinks, alters the structural and functional properties of lens proteins, impairing their solubility and transparency. Environmental factors such as UV radiation, smoking, and exposure to heavy metals exacerbate oxidative stress and increase the risk of cataract development. Antioxidant defense systems in the lens, including glutathione and antioxidant enzymes such as superoxide dismutase and catalase, help maintain redox balance and protect against oxidative damage. Dietary antioxidants, supplementation with vitamins C and E, and lifestyle modifications to reduce oxidative stress (e.g., smoking cessation, UV protection) may help prevent or delay the progression of cataract formation [43].
Myopia is a condition where visual images are focused in front of the retinal plane while the eye is at rest, resulting in blurred distance vision. It is primarily a consequence of excessive axial elongation during the growth of the eye. This elongation can disturb the choroid, leading to a significant decrease in its thickness, as well as a reduction in blood flow and perfusion [44]. The resultant hypoxic state triggers a series of events that induce oxidative stress in eye tissues, including (1) mitochondrial dysfunction, as reduced oxygen levels impair mitochondrial function, ultimately leading to increased ROS production [45]; (2) activation of hypoxia-inducible factor (HIF), a protein that responds to hypoxia and regulates the expression of genes involved in oxidative stress pathways [46]; (3) hypoxia-induced inflammation, which can promote ROS production [46]; (4) endoplasmic reticulum stress, as hypoxia disrupts protein folding in the endoplasmic reticulum, thereby leading to oxidative stress [47].
In this context, it is noteworthy to mention the dual effect of atropine, which is often used to slow myopia progression in children, on extracellular matrix (ECM) production by human scleral (HSF) and choroidal (HCF) fibroblasts. Atropine stimulates ECM production by HSF, likely increasing the rigidity of the sclera and inhibiting its elongation, while it decreases ECM production by HCF, probably enhancing blood perfusion to the surrounding eye tissues, thus reducing oxidative stress and inhibiting axial elongation [48].
In addition to the paradox of oxygen being both necessary and detrimental for life, another paradox is the effect of an excess of antioxidants on lifespan, a focal point of ongoing research and debate in biology and medicine. Emerging evidence suggests that the relationship between antioxidants, lifespan, and susceptibility to pathological damage is more nuanced than previously thought. Experimental studies, as reported by Halliwell [49], indicated excessive antioxidant supplementation may harm health and lifespan.
In a pivotal study [50], the author explores the common belief that oxidative damage leads to aging and that antioxidants can help maintain youthfulness, presenting recent experiments that dispute this idea. For example, scientists discovered that roundworms genetically modified to lack specific antioxidants and had lifespans comparable to normal worms despite significant oxidative damage. This suggests that aging might not be caused by oxidative chemical reactions that damage cells; instead, some free radicals might be advantageous. Siegfried Hekimi and Wen Yang [51] proposed that a certain level of free radicals could activate an organism’s internal repair systems. In their 2010 experiment, they genetically altered a group of roundworms to produce high amounts of specific free radicals. Unexpectedly, these mutant worms outlived normal worms. However, their increased lifespan disappeared when the researchers administered antioxidants to the mutant worms.
Similarly, studies in mice, as reported by Salmon et al [52], revealed that increased free radicals correlated with a longer lifespan, suggesting that antioxidants may not always be beneficial. Transgenic mice experiments suggested that lifespan and the development of pathology could be uncoupled [52]. Mice with genetic modifications in their antioxidant defense systems generally showed no change in lifespan. However, as the oxidative stress theory predicted, these genetically altered mice exhibited changes in the progression and severity of age-related diseases. Specifically, increased oxidative stress accelerates the development of pathologies, while reduced oxidative stress slows it down.
These contradictory findings suggest that (a) oxidative stress may have a limited role in the aging process but a significant impact on healthspan (the duration of healthy status of an individual); and/or (b) the influence of oxidative stress on aging may be environment-dependent. In low-stress environments, oxidative damage appears to play a minimal role in aging. Conversely, under chronic stress conditions, including those with pathological phenotypes that reduce optimal health, oxidative stress and damage significantly contribute to aging. In such environments, enhanced antioxidant defenses exhibit anti-aging effects, altering lifespan, age-related pathologies, and physiological functions in line with the oxidative stress theory of aging.
Moreover, in humans, high doses of certain antioxidants, such as vitamin E or beta-carotene, have been associated with increased mortality in certain populations [53]. Another case concerns the use of antioxidants in athletes. Vigorous exercise produces large amounts of ROS due to high oxygen consumption, leading some athletes to consume antioxidants to counteract potential ROS damage. However, no convincing evidence exists that antioxidant supplementation benefits acute physical exercise or training. In fact, exogenous antioxidants can interfere with the beneficial roles of free radicals in cell signaling, potentially hindering performance-enhancing and health-promoting adaptations such as mitochondrial biogenesis, muscle growth, and improved insulin sensitivity. Despite the belief that antioxidants can mitigate oxidative stress and enhance performance, high doses of antioxidants are more likely to have negative effects than benefits [54]. One proposed explanation for this paradoxical effect is that moderate levels of oxidative stress may be necessary for cellular signaling pathways that promote longevity and overall health [52]. By neutralizing all oxidative stress, excessive antioxidant intake may disrupt these beneficial signaling pathways, adversely affecting lifespan. Moreover, antioxidants do not just target harmful free radicals; they also play essential roles in normal cellular functions and signaling pathways. Excessively high levels of antioxidants may interfere with these physiological processes, leading to unintended consequences. Accordingly, recent evidence indicates that calorie restriction and reduced glucose metabolism extend lifespan in various organisms, such as yeast, nematodes, fruit flies, mice, rats, and possibly humans, by increasing mitochondrial oxygen consumption and ROS formation. These ROS are signals to enhance stress resistance and longevity, known as mitochondrial hormesis or mitohormesis. Antioxidants that block ROS signals can negate the benefits of calorie restriction and exercise, challenging Harman’s free radical theory of aging and suggesting ROS as crucial signaling molecules for health and longevity [55, 56].
Finally, it must be considered that excessive antioxidant levels may lead to
reductive stress (RS) [57]. It is the opposite of oxidative stress (OS) and can
occur when conditions push the redox balance of key biological molecules such as
NAD⁺/NADH, NADP⁺/NADPH, and GSH/GSSG towards a more reduced state. When
antioxidant enzyme systems are overexpressed, they produce an excess of reducing
agents that can deplete reactive oxidative species, leading to RS. This creates a
feedback loop where chronic RS triggers OS, promoting further RS. An excess of
reducing agents can influence cellular signaling, alter gene expression, affect
protein disulfide bond formation, reduce mitochondrial function, and decrease
cellular metabolism, contributing to various diseases involving NF-
Overall, while antioxidants are crucial in maintaining cellular health and mitigating oxidative damage, the optimal balance and dosage remain under investigation (Fig. 2).
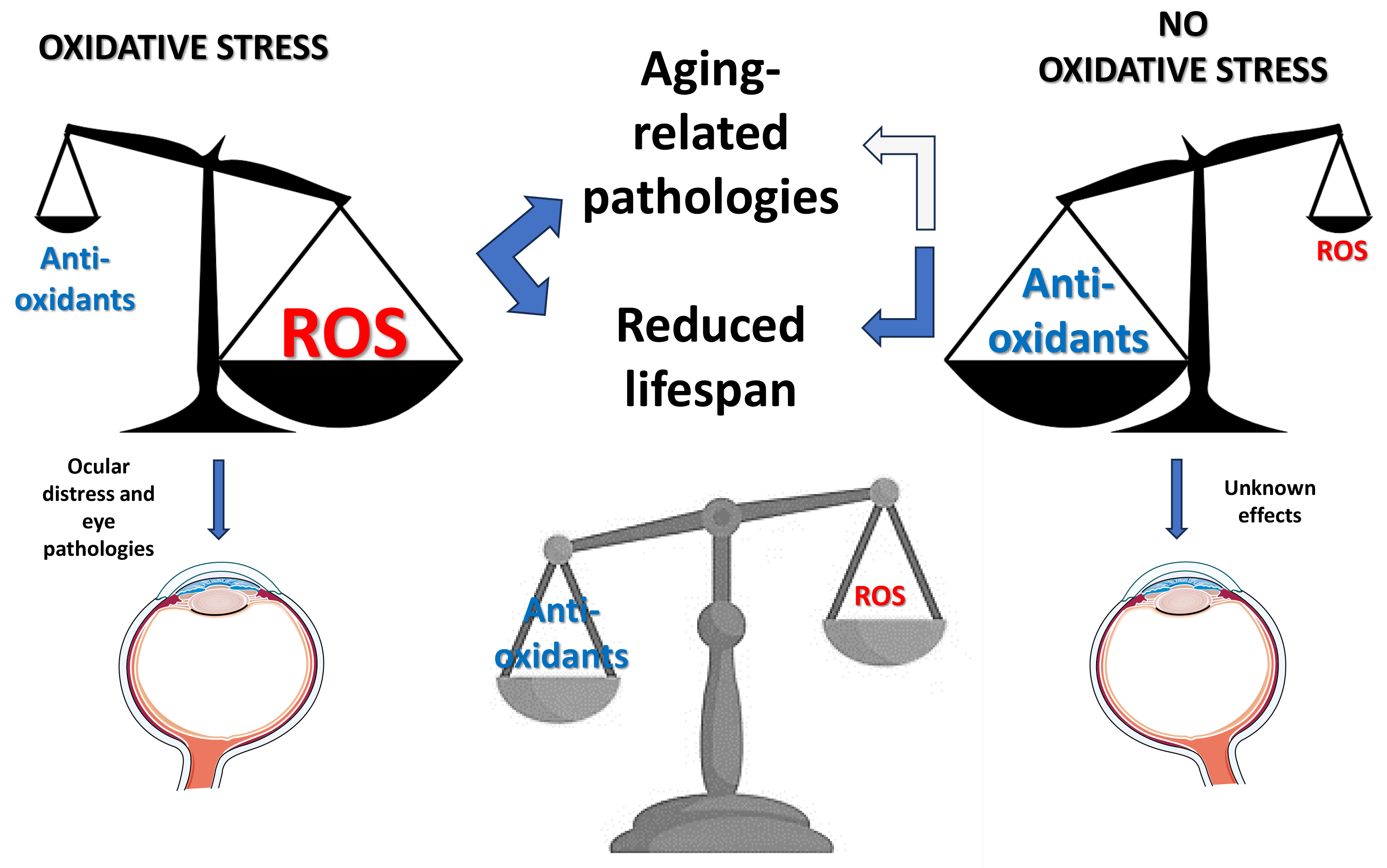 Fig. 2.
Fig. 2.
The supposed effects of oxidative stress on health. Excess oxidative stress (left) alters organ homeostasis, leading to general and specific (eye-included) health problems. The absence or very low levels of oxidative stress (right) also alters organ homeostasis and appears to have negative effects on lifespan, likely including organ pathologies, with unknown specific impact on the eye. An ideal balance between oxidants and antioxidants (bottom) could be required to maintain organ homeostasis and general health, eye included.
Caution should be exercised when interpreting the effects of antioxidant supplementation on lifespan and disease, as further research is needed to elucidate the complex interplay between antioxidants, oxidative stress, health, and longevity.
Despite the retina being part of the neurological system and highly sensitive to the balance between oxidants and antioxidants, no experiments have specifically addressed this balance in the eye concerning its development and disease resistance. It would be an intriguing challenge to determine whether the concept of mitohormesis also applies to the eye or if this organ constitutes an exception. For instance, cognitive disabilities are often linked to retinopathies, such as in the case of glaucoma [58] and diabetic retinopathy [59]. However, current evidence suggests that food supplements with antioxidant and neuroprotective activities can slow the progression of eye diseases in both the posterior [60, 61, 62, 63] and anterior segments [64, 65, 66]. For example, stable transfection of the human OXR1 (hOXR1) antioxidant regulatory gene into the cone-like 661W cell line protected the cells in vitro from oxidative stress induced by hydrogen peroxide. In vivo, subretinal injection of a construct containing the hOXR1 gene into a mutant rd1 mouse model of retinal degeneration improved photoreceptor light response, expression, and localization of photoreceptor-specific proteins, and delayed retinal degeneration [67].
A more intriguing possibility could be to use mutant mouse models of oxidative stress [68], which experimentally induce retinal diseases [69], to evaluate whether the altered balance (in either way) between oxidative processes and endogenous antioxidant defense would influence the development of the induced retinal disease or even the visual cortex plasticity necessary for visual ability recovery after an insult [70].
DR and PB designed the study. DR wrote paragraphs 1, 3, 4. PB wrote paragraph 2. Both authors critically revised the whole manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest. Given his role as Editorial Board members and Guest Editor, Dr. Dario Rusciano had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Peter Brenneisen. The company (Fidia Ophthalmic Research) was not involved in the preparation or publication of the manuscript.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


