1 Institute of Special Animal and Plant Sciences, Chinese Academy of Agricultural Sciences, 130112 Changchun, Jilin, China
2 The Scientific Research Center, China-Japan Union Hospital of Jilin University, 130033 Changchun, Jilin, China
Abstract
Background: Articular cartilage has limited self-repair capacity, and
current clinical treatment options for cartilage defects are inadequate. However,
deer antler cartilage possesses unique regenerative properties, with the ability
to rapidly repair itself. This rapid self-repair process is closely linked to the
paracrine factors released by deer antler stem cells. These findings present
potential for the development of cell-free therapies for cartilage defects in
clinical settings. The aim of this study was to investigate a novel method for
repairing cartilage. Methods: A rat model with articular cartilage
defects was established through surgery. Hydrogels loaded with exosomes (Exos)
derived from antler stem cells (ASC-Exos) were implanted into the rat cartilage
defects. The extent of cartilage damage repair was assessed using histological
methods. The effects of ASC-Exos on chondrocytes and rat bone marrow mesenchymal
stem cells (BMSCs) were evaluated using cell viability assays, proliferation
assays, and scratch assays. Additionally, the maintenance of the chondrocyte
phenotype by ASC-Exos was assessed using real-time fluorescence quantitative PCR
(qPCR) and western blot analysis. The protein components contained of the Exos
were identified using data-independent acquisition (DIA) mass spectrometry.
Results: ASC-Exos significantly promoted the repair of cartilage tissue
damage. The level of cartilage repair in the experimental group (ASC-Exos) was
higher than that in the positive control (human adipose-derived stem cells,
hADSC-Exos) and negative control (dulbecco’s modified eagle medium) groups
(p
Keywords
- deer antler stem cells
- exosomes
- cartilage regeneration
- cartilage defects
Articular cartilage is a critical component of the locomotor system, covering bone tissue at joints with its dense, glassy appearance. The primary constituents of articular cartilage are chondrocytes and the extracellular matrix (ECM). Chondrocytes account for approximately 2% of the total volume of articular cartilage, while the remaining 98% consists of a porous extracellular matrix. Since cartilage lacks a direct blood supply, chondrocytes rely on the diffusion of nutrients for their survival and function [1]. Therefore, cartilage repair is challenging once it is damaged.
Current clinical treatments for cartilage damage include conservative and surgical methods. Traditional approaches are no longer effective in promoting cartilage regeneration and repair, making it essential to find advanced and effective alternatives.
Recent studies have shown that in cartilage repair, mesenchymal Stem Cells (MSCs) release paracrine factors, such as growth factors, chemokines, and cytokines, to regulate local inflammation, apoptosis, and proliferation, rather than differentiating directly into chondrocytes to participate in tissue repair [2, 3]. Implanting bioscaffolds containing MSCs paracrine secretions into cartilage defects effectively promotes repair and regeneration, and this method has been validated in animal models [4]. This approach avoids issues like immune compatibility, tumorigenicity, embolism formation, and infection propagation associated with living cell transplantation. Additionally, long-term storage of paracrine substances does not affect their efficacy, and it can be produced in large quantities for practical clinical applications [5]. MSCs paracrine substances activate cellular and vascular pathways to nourish cartilage [4], Moreover, MSCs participate in local immunoregulatory mechanisms, inhibiting dendritic cell maturation, T cell proliferation, B cell activation, and antibody secretion, while affecting macrophage polarization [6], which can reduce the risk of rejection and disease transmission.
Exosomes (Exos), measuring 30–150 nm, are extracellular vesicles secreted by almost all cells [6, 7]. Recent studies have shown that MSC-derived Exos play a key role in regulating cell migration, proliferation, differentiation, and extracellular matrix synthesis [8]. Exos contain lipids, metabolites, nucleic acids (mRNAs and microRNAs), and proteins that act as carriers for intercellular communication, regulating cell growth, differentiation, and extracellular matrix synthesis. Exos have been demonstrated to be effective in ameliorating cartilage diseases. Studies found that MSC-derived Exos up-regulate the expression of cartilage maintenance gene COL2a1 and proteoglycans and down-regulate the expression of hypertrophy markers MMP-13 and Runx2 [8, 9]. These studies suggest that MSC-Exos maintain the cartilage phenotype and inhibit cartilage osteogenesis. It has also been found that Exos derived from human MSCs are involved in the inhibition of chondrocyte apoptosis through the miR-206/G Protein-Coupled Receptor Kinase Interacting ArfGAP 1 (GIT1) axis [8]. The application of MSC-Exos significantly reduce the production of inflammatory cytokines in chondrocytes, and is also able to improve the function and structure of cartilage [4, 10, 11]. MSC-Exos have excellent tolerability, very low immunogenicity, and very low risk of toxicity, while their storage and transportation requirements are lower than those of cells. Therefore, Exosomes are considered an ideal “cell-free” tissue engineering strategy to replace MSCs. However, to obtain large-scale purified Exos and to improve their utilization efficiency, biosafety, and therapeutic efficacy, further in-depth research and exploration are required.
Antler Stem Cells (ASCs), a novel source of stem cells, have great potential for the repair of cartilage defects. Deer antlers, a unique mammalian organ, can completely regenerate from the stump of pedicle, making them an ideal model for complete regeneration in mammals [12]. Studies have shown that the proliferation and differentiation of pedicle periosteal cells form the antler blastemal [13]. Antler pedicle periosteal cells can be induced to differentiate into somatic cells in vitro, express some MSCs markers, and be identified as stem cells called ASCs [14]. The cartilage formation of deer antlers resembles mammalian cartilage formation, involving the differentiation of mesenchymal cells into prechondrocytes, chondrocytes, and ultimately hypertrophic chondrocytes [15]. Under natural conditions, antler cartilage not only repairs itself but also grows at a very impressive rate (up to 2 cm/d) [16]. This rapid regeneration is attributed to the stimulatory effects of biomolecules secreted by ASCs, which regulate the microenvironment, promote angiogenesis and metabolic activity, stimulate chondrocyte proliferation and growth, regulate chondrogenesis and osteogenesis, and control cartilage phenotype transformation [13].
In this study, we aimed to investigate the role of exosomes secreted by Antler Stem Cells (ASC-Exos) in cartilage repair through both in vivo and in vitro experiments.
The method for obtaining antler stem cells (ASCs) followed the procedures outlined by Wang, and previous studies have identified the cells [17]. Specifically, antler tissue was collected from three 8-month-old sika deer immediately post-slaughter, minced into small pieces, and digested in dulbecco’s modified eagle medium (DMEM) containing 150 U/mL collagenase (Gibco, 11965092, Grand Island, NE, USA). ASCs were successfully isolated through this process and cultured in DMEM supplemented with 10% fetal bovine serum (FBS) (Gibco, 10100147C, USA) and 1% penicillin-streptomycin solution (Gibco, 15140163, USA) at 37 °C, under saturated humidity with 5% CO2 concentration. The study also utilized other cell lines obtained from Procell (Wuhan, China), including rat bone marrow mesenchymal stem cells (rat-BMSC, product number: CP-R131), human adipose-derived stem cells (hADSC, product number: CP-H202), and rat chondrocytes (product number: CP-R087). All cells were tested for mycoplasma to ensure that they were not contaminated with mycoplasma, and human cells were identified by short tandem repeats (STR).
Cells were seeded in T75 cell culture flasks and expanded in DMEM medium supplemented with 10% FBS (Exos removed) at 37 °C and 5% CO2. Once the cell density reached 80%, the medium was collected and centrifuged at 4 °C, 300 g for 10 minutes to remove cells. The supernatant was collected, transferred to a new centrifuge tube, and centrifuged at 4 °C, 2000 g for 20 minutes to remove cell debris. The supernatant was then filtered through a 0.22 µm filter (Merck, SVGP010, South San Francisco, CA, USA) and centrifuged at 4 °C, 10,000 g for 30 minutes. The resulting supernatant was transferred to an ultracentrifuge tube and ultra-centrifuged at 4 °C, 100,000 g for 60 minutes. After centrifugation, the supernatant was discarded, and the exosomes were resuspended in PBS. The exosomes were centrifuged again at 100,000 g for 60 minutes at 4 °C. After resuspension in Phosphate Buffered Saline (PBS), the exosomes were transferred into sterile eppendorf (EP) tubes and stored in a refrigerator at –80 °C for subsequent experiments
A 20 µL sample was dropped onto a copper grid with 2 nm pores and left at room temperature for 3 minutes. The liquid was then blotted away with filter paper. Next, 30 µL of 3% phosphotungstic acid solution (Merck, 228559, USA) was added for negative staining (5 minutes at room temperature). After removing the staining solution, the copper grid was carefully transferred to a transmission electron microscope to observe and record the exosome morphology.
Refer to Li’s method [18] for identification of exosomes. Alix, CD63, and Tumor Susceptibility Gene 101 (TSG101) were detected by immunoblotting using the antibodies Alix (1:1000, ab275377, Abcam, Cambridge, UK), CD63 (1:1000, AF1471, Beyotime, Jiangsu, China), and TSG101 (1:1000, AF8259, Beyotime, China), Golgi Matrix Protein 130 (GM130) (1:1000, ab52649, Abcam, UK).
Load the pre-treated exosome samples into the sample chamber of the Flow Nano Analyzer model type N30 (Nano FCM Inc., Xiamen, China), start the instrument, and initiate data collection.
Eight-week-old male Wistar rats (250 g) were obtained from Liaoning Changsheng Biotechnology Co., Ltd. (Shenyang, China). All experiments followed the guidelines of the Ethics Committee for Animal Experiments of the Institute of Special Animal and Plant Sciences. To create cartilage defects, the knee joints were exposed by incising the skin and muscle, and full-thickness cylindrical defects (2 mm in diameter, 1 mm in depth) were drilled. Thirty rats were randomly assigned to five groups: (1) Control (Untreated), (2) DMEM (negative control), (3) ASC-Exos (200 µg/mL), (4) hADSC-Exos (200 µg/mL, positive control), and (5) Sham -operated. In groups 1, 2, 3, and 4, cartilage defects were created. After anesthesia with 1.5% sodium pentobarbital (30 mg/kg), exosomes mixed with hydrogel (SunP Gel G1, Shangpu Bio, Shanghai, China) were implanted into the defects of groups 3 and 4. DMEM mixed with hydrogel was implanted into the defects of group 2. Postoperative antibiotics (gentamicin 80,000 U, intramuscularly, once daily for three days) were administered to prevent infection. Samples were collected after 6 weeks.
At the end of the experiment, the rats were euthanized by CO2, and the muscle tissues around the rat knee joints were removed. The right and left knee joints of six rats from each group were selected. Photos of the rat knee joint cartilage were taken with a high-resolution camera (Canon, Tokyo, Japan) and the overall morphology repair effect was evaluated according to the International Cartilage Repair Society (ICRS) morphological assessment criteria [19].
After embedding the tissue, sections were prepared and stained with hematoxylin (Beyotime, C0107, China) for 10 minutes. The excess stain was subsequently rinsed off for 10 minutes, followed by a 2-minute rinse with distilled water. The sections were then stained with eosin (Beyotime, C0109, China) for 15 seconds. For mounting, the sections were sequentially placed in 70%, 80%, 90% ethanol, and then in anhydrous ethanol for 15 seconds each. Subsequently, they were placed in xylene I and xylene II for 10 minutes each, and finally mounted with neutral gum.
For Safranin-O/fast green staining (Solarbio, G1371, Beijing, China), the sections were rehydrated and stained with Weigert’s staining solution for 3–5 minutes, differentiated with a neutral solution for 15 seconds, and washed with distilled water for 1 minute. Subsequently, the sections were immersed in fast green staining solution for 5 minutes, followed by a 2-minute wash with distilled water. Next, the sections were immersed in Safranin O staining solution for 2 minutes, followed by another 1-minute wash with distilled water. To remove excess fast green, the sections were rinsed with acetic acid solution for 2 minutes and washed with distilled water for 2 minutes. Finally, the sections were briefly treated with 95% ethanol for 2 seconds, dehydrated with anhydrous ethanol for 2 seconds, clarified with xylene for 10 minutes, and sealed with neutral resin.
Histological observations were evaluated based on the International Society for Cartilage Repair’s scoring criteria for cartilage defect repair [19].
Washed the tissue sections in PBS for 5 minutes, followed by replacing PBS with
1% antigen retrieval solution. Heated in a water bath for 8 minutes. Rinsed the
cooled sections with PBS three times for 3 minutes each. Encircled the tissue
with an immunohistochemical pen. Follow-up experiments with ready-to-use rapid
immunohistochemistry MaxVision TM kits (MXB, KIT-5001, Shanghai, China). Applied
Reagent A and Reagent B, then added 1 drop of Collagen II (COLII) primary
antibody (1:200, GB11021, Servicebio, Wuhan, China) to each section and incubated
overnight at 4 °C. Subsequently, applied Reagent C and Reagent D,
followed by incubating with 3,3
Added 5 µL of PKH26 fluorescent (Merck, PKH26PCL, USA) dye to the exosome
suspension, mixed thoroughly, and incubated at 37 °C for 10 minutes.
Centrifuged the cells at 300 g for 5 minutes to collected the cell pellet. Rinsed
the cells several times with PBS, then resuspended them in serum-free medium and
seeded them into a culture flask. Once the cell density in 6-well plates reached
60%, added 100 µL of fluorescent-stained exosomes to each well. Gently
agitated the plates to ensure even distribution of exosomes, then incubated for
12 hours. Stained the cell nuclei with 4
Cell counting kit-8 (CCK-8) assay: Rat chondrocytes and MSCs were thawed and
cultured in petri dishes until they reached 80% confluence. The cells were then
harvested, counted, and seeded into 96-well plates at a density of 3000 cells per
well. Cultures were maintained at 37 °C with 5% CO2 and saturated
humidity for 5 days. Cell proliferation was measured daily using the CCK-8 assay.
For the assay, 10 µL of CCK-8 (Beyotime, C0038, China) was added to each
well and incubated with the cells for 2 hours. Proliferation was quantified by
measuring the absorbance at 450 nm using an Enzyme-Linked Immunosorbnent Assay (ELISA) plate reader. Data are
presented as mean
5-Ethynyl-2
Culture inserts (Ibidi, 80209, Munich, Germany) were placed in each well, and 70
µL of 3
We co-cultured exosomes with chondrocytes for 7 generations to observe the exosomes’ impact on maintaining chondrocyte markers. RNA and protein were extracted from passages 1, 4, and 7 for subsequent experiments.
Total RNA from rat chondrocytes was extracted using the RNA Easy Fast Cell Kit (TIANGEN, DP451, China). For qPCR analysis, a 20 µL reaction system (10 µL SYBR, 2 µL primers, 2 µL cDNA, and 6 µL H2O) was prepared using the TB Green Premix Ex Taq II (Takara, RR820A, Shiga, Japan), following the manufacturer’s instructions. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) served as the internal reference gene. Primer sequences used in qPCR are listed in Table 1. Gene expression levels were quantified using cyclic thresholds (Ct) values and calculated via the 2-ΔΔCt method. All reactions were performed in three replicates.
| Gene | Primer |
|---|---|
| Sox-9-F | GAGTTTGACCAATACCTGCCG |
| Sox-9-R | GCCTGTTGCTTTGACATCCAC |
| COL II-F | GACCTGAAACTCTGCCACCC |
| COL II-R | TGCTCTTGCTGCTCCACCA |
| Aggrecan-F | CCCCAAATCCCTCATACTCA |
| Aggrecan-R | AGGTGGCTCCATTCAGACAA |
| GAPDH-F | AAGAAGGTGGTGAAGCAGG |
| GAPDH-R | AAGGTGGAAGAATGGGAGTT |
Refer to Li’s method for identification of exosomes [18]. We used a primary
antibody collagen II (Servicebio, GB11021, 1:2000, China), aggrecan (Abcam,
ab313636, 1:1000, UK), Sox9 (Abcam, ab185966, 1:1000, UK),
After exosome was stored in liquid nitrogen, it was sent to Guangzhou Kidio Biotechnology Co. for Data-Independent Acquisition (DIA) proteomic quantitative analysis.
Statistical analyses were performed using Prism 8 (GraphPad Software, GraphPad
Software, Inc., San Diego, CA, USA). All data were assessed for normal
distribution. One-way ANOVA with Dunnett’s multiple comparison test was employed
for comparisons involving more than more than two groups. Repeated measures ANOVA
was used for data collected at various time points in the CCK-8 assay. All
quantitative data are presented as mean
Electron microscopy results showed that the particle size range of both types of exosomes predominantly ranged between 50–100 nm range (Fig. 1A). Western blotting detected positive expression of Alix, CD63, and TSG101, and negative expression of GM130 (Fig. 1B), which are characteristic surface markers of hADSC-Exos and ASC-Exos. The presence of these specific markers, coupled with the observed size distribution, confirms the identity of the isolated exosomes, thereby ensuring the reliability and purity of the sample isolation process. Furthermore, NTA analysis revealed that both types of exosomes primarily exhibited particle sizes in the range of 50–100 nm, further validating their purity (Fig. 1C).
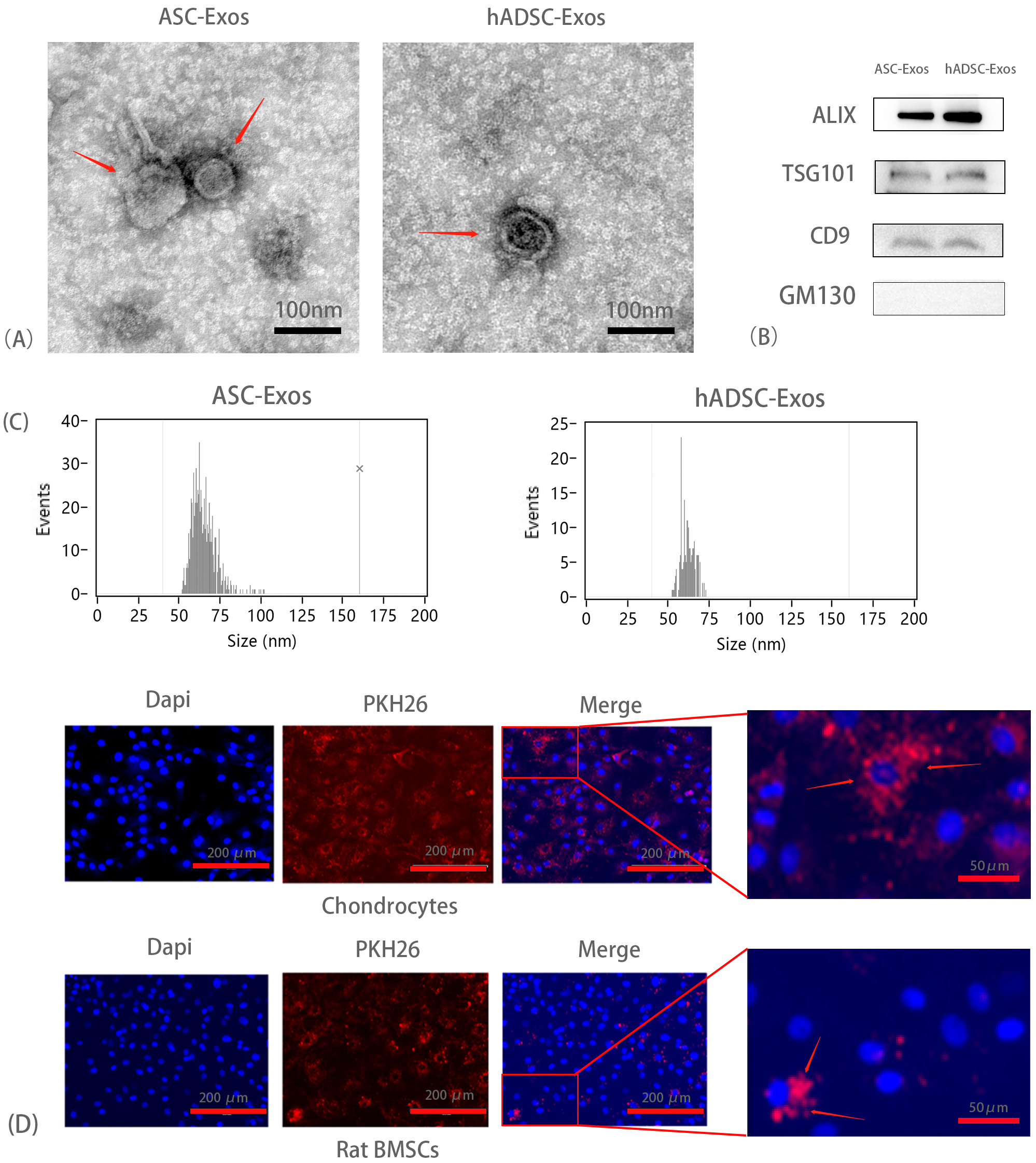 Fig. 1.
Fig. 1.
Identification of exosomes derived from antler stem cells (ASC-Exos) and exosomes derived from human adipose-derived stem cells (hADSC-Exos). (A) Electron microscopy results depicting the particle size distribution of ASC-Exos and hADSC-Exos, predominantly within the 50–100 nm range. Scale bar = 100 nm. (B) Western blot assay results demonstrating positive expression of the specific surface markers Alix, CD63, TSG101 and GM130. (C) The results of Nanoparticle Tracking Analysis (NTA) detection for Exosomes’ particle size. (D) Assessment of ASC-Exos uptake by Chondrocytes and Rat Bone Marrow Stromal Cells, conducted in triplicate (n = 3). Scale bar = 200 µm or 50 µm. TSG101, Tumor Susceptibility Gene 101; GM130, Golgi Matrix Protein 130.
To investigate the uptake of ASC-Exos by chondrocytes and rat BMSCs, ASC-Exos were labeled with PKH26 and co-cultured with chondrocytes and rat BMSCs for 24 hours. The uptake of ASC-Exos was subsequently visualized using a fluorescence microscope. A significant amount of red granular material was observed aggregated around the nuclei of both chondrocytes and rat BMSCs, indicating successful uptake of ASC-Exos after 24 hours of co-culture (Fig. 1D).
To achieve sustained in vivo release of ASC-Exos, hydrogels containing ASC-Exos (50 µg/mL), hADSC-Exos, and DMEM were prepared for topical administration to cartilage defects. After 6 weeks, samples were macroscopically assessed to evaluate the impact of ASC-Exos on cartilage repair (Fig. 2A). In the blank control group, the drilled defects exhibited less regenerated tissue with a rough surface and a clear boundary between the new and original cartilage. In the DMEM group, new tissue filled the defect, but the surface remained rough, and there was an obvious interface between the new and original cartilage. The hADSC-Exos group showed new tissue with a relatively smooth surface, but with a distinct boundary between the new and original cartilage. In contrast, the ASC-Exos group displayed new tissue similar to the sham-operated group, with a complete and smooth surface that integrated well with the surrounding cartilage. According to the International Cartilage Repair Score (ICRS) criteria, the ASC-Exos group achieved a higher macroscopic score compared to the hADSC-Exos, DMEM, and blank control groups, and approached that of the sham-operated group (Fig. 2B).
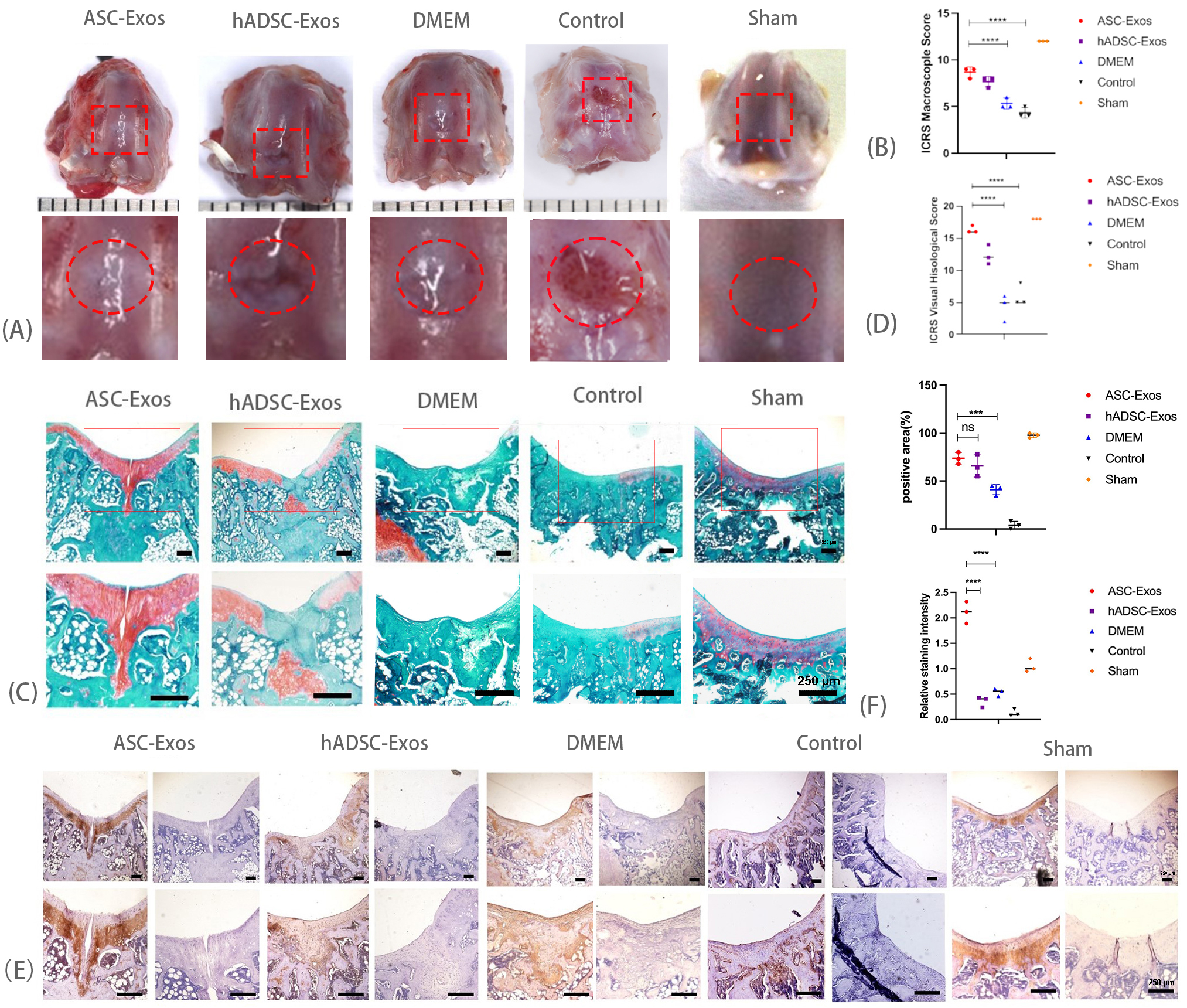 Fig. 2.
Fig. 2.
Histologic evaluation of cartilage defect. (A) Macroscopic
observation of cartilage defects 6 weeks after surgery. (B) Macroscopic ICRS
scores of different groups. Data are expressed as mean
For histological evaluation, Safranin O/fast green staining was employed to assess cartilage regeneration (Fig. 2C). The blank control and DMEM groups exhibited the most severe cartilage defects, characterized by irregular and rough surfaces. In the hADSC-Exos group, the new tissue almost completely filled the defects but showed little red staining, indicating a non-smooth surface. Conversely, the regenerated cartilage in the ASC-Exos group displayed intense red staining and a smooth surface, resembling natural cartilage in the sham-operated group. According to the ICRS for histology, the ASC-Exos group achieved higher scores compared to the hADSC-Exos, DMEM, and blank control groups, and approached scores observed in the sham-operated group (Fig. 2D). These findings suggest that ASC-Exos effectively promoted cartilage regeneration.
Immunohistochemical staining was employed to assess the expression of COL II in the newly formed tissues. The ASC-Exos group exhibited strong positive staining for type II collagen, similar to the sham-operated group. In contrast, the DMEM and hADSC-Exos groups showed comparatively weaker staining for type II collagen. Surprisingly, the intensity of type II collagen staining was even higher in the ASC-Exos group than in the sham group. On the other hand, the area of type II collagen positivity in the ASC-Exos group was not significantly different from that in the hADSC-Exos group, but was significantly higher than that in the DMEM group. This enhanced expression of type II collagen in the ASC-Exos group suggests that ASC-Exos promoted the formation of the extracellular matrix (Fig. 2E,F).
The CCK-8 assay results demonstrated that the ASC-Exos group exhibited faster proliferation compared to the hADSC-Exos and the DMEM groups in both cell types (Fig. 3A,B). The findings indicate that ASC-Exos significantly enhance the proliferation of chondrocytes and rat BMSCs. Using the EdU detection kit and inverted fluorescence microscopy, we observed that ASC-Exos treated both chondrocytes and rat BMSCs had the highest percentage of EdU-positive cells compared to the two control groups, respectively. Conversely, the DMEM group displayed the lowest percentage of EdU-positive cells. These results indicate that ASC-Exos significantly increase the proliferation potential of chondrocytes and rat BMSCs, surpassing the effect of hADSC-Exos (Fig. 3C).
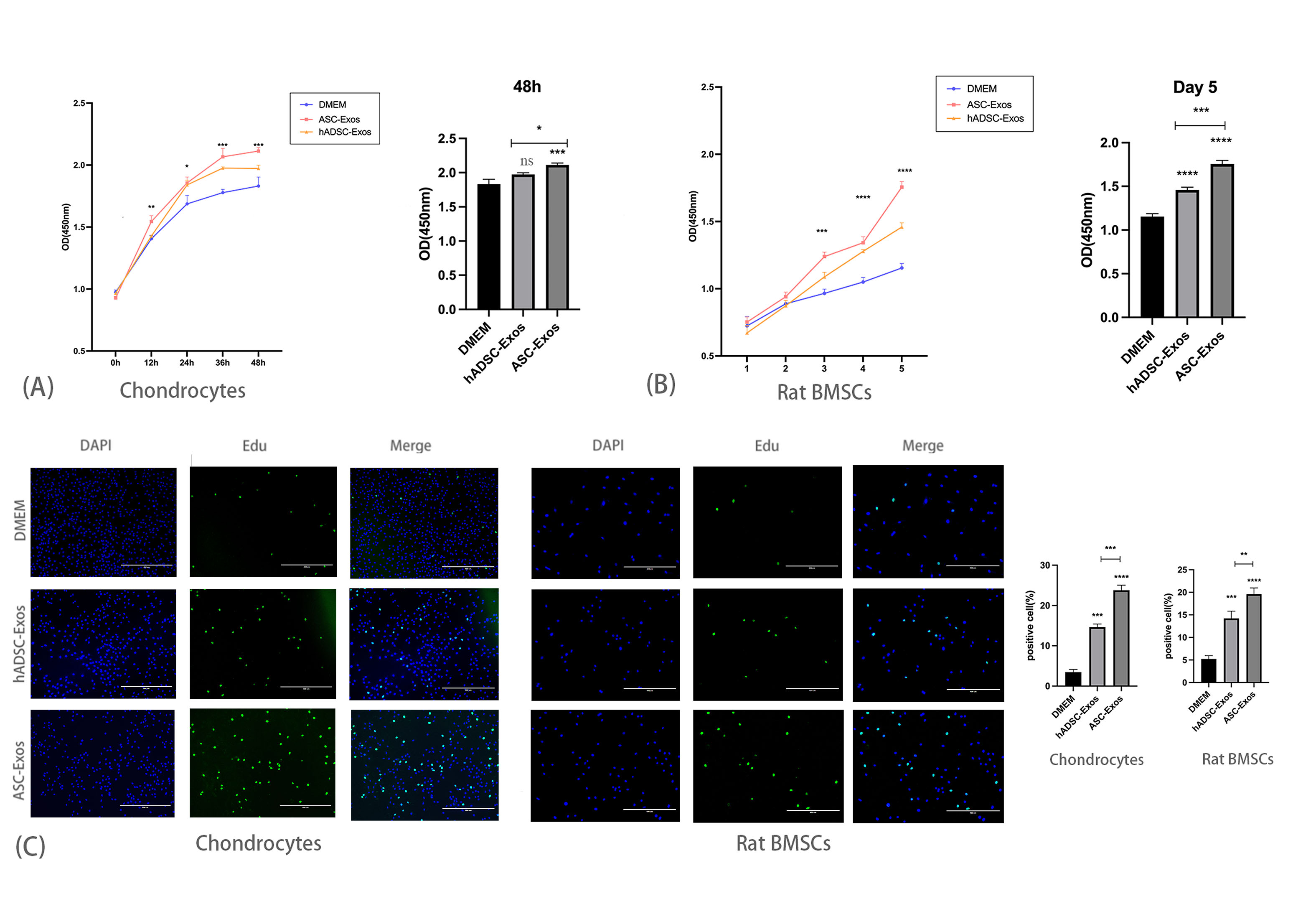 Fig. 3.
Fig. 3.
ASC-exosomes promote the proliferation of rat chondrocytes and
rat bone marrow mesenchymal stem cells (BMSCs). (A) Effect of ASC-Exos on the proliferation of chondrocytes, n = 3,
*p
In a cell migration assay, rat BMSCs treated with ASC-Exos exhibited the highest migration rate compared to those treated with hADSC-Exos and DMEM. This indicates that ASC-Exos significantly enhance the migration of rat BMSCs (Fig. 4A,B).
 Fig. 4.
Fig. 4.
ASC-exosomes promote the migration of rat BMSCs in
vitro. (A) Migration assay of rat BMSCs. Scale bar = 1000 µm. (B)
Quantitative analysis of migrated rat BMSCs, n = 3, *p
Chondrocytes undergo differentiation and gradually lose their characteristic phenotype in vitro. Maintaining the chondrocyte phenotype is crucial for cartilage repair. In this study, we investigated the impact of ASC-Exos on the expression of chondrogenic genes and proteinsin the first, fourth, and seventh generations of chondrocytes. ASC-Exos treatment significantly up-regulated or maintained the mRNA expression levels of Aggrecan, COL II, and Sox-9, demonstrating more pronounced effects compared to the hADSC-Exos and DMEM groups (Fig. 5A,B).
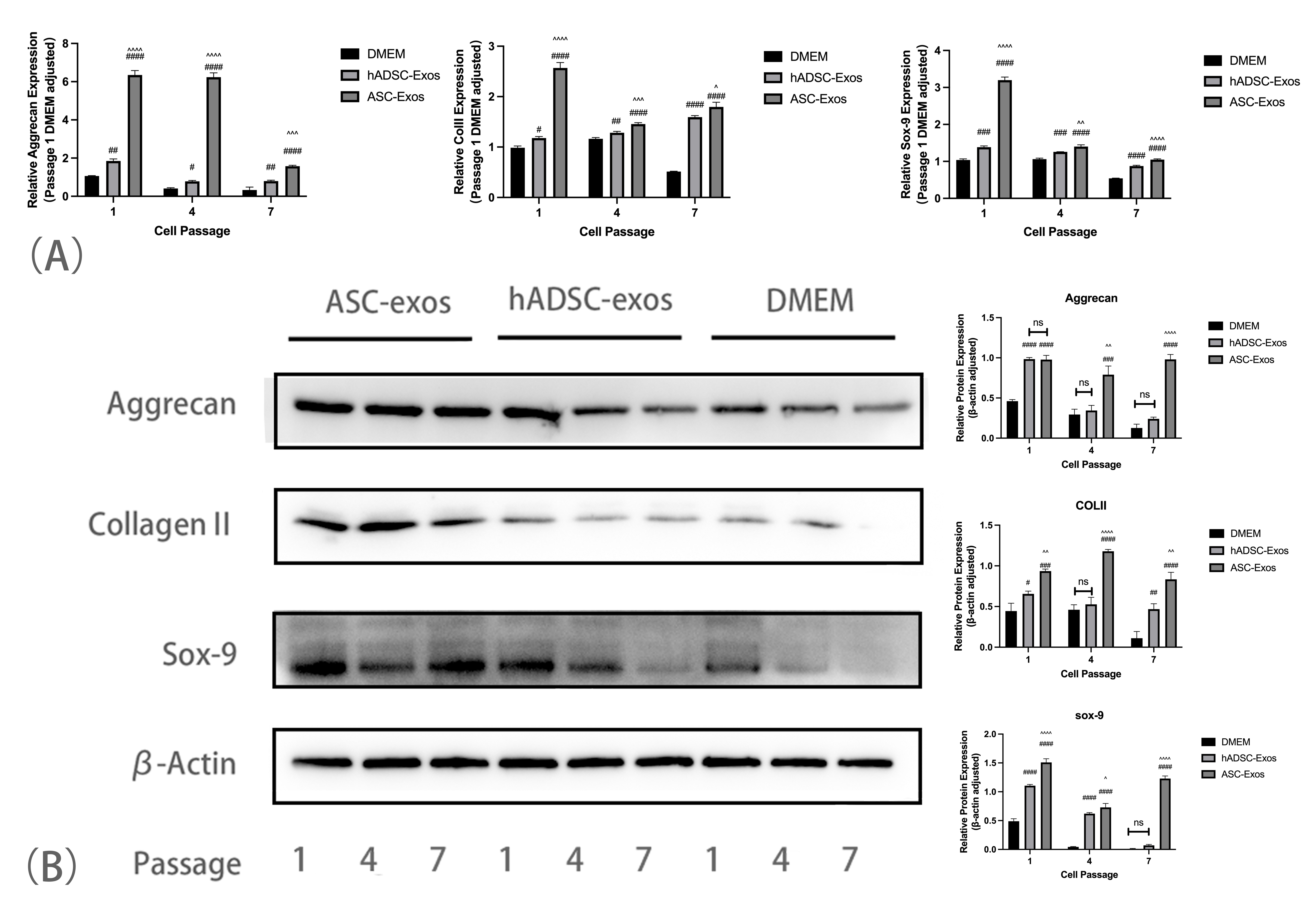 Fig. 5.
Fig. 5.
ASC-exosomes regulate the expression of cartilage-related genes
and proteins. (A) Relative gene expression levels of Aggrecan, Collagen II, and
Sox9 in chondrocytes detected by qPCR. n = 3. #p
We analyzed the protein composition of ASC-Exos using a DIA assay and proteomics data has been deposited in ProteomeXchange (PXD053936). We identified several potential proteins with high relative expression levels, protein function obtained from uniprot (https://www.uniprot.org/) (Table 2). These proteins were found to play roles in promoting cell proliferation and migration, as well as protecting cartilage.
| Protein Name | Abbreviation | Functionality |
|---|---|---|
| Serotransferrin | TF | Stimulate cell proliferation |
| Protein S100A4 | S100A4 | Interacts with cytoskeletal proteins to enhance cell migration |
| Insulin-like growth factor-binding protein 1 | IGF1 | Major anabolic growth factor of articular cartilage; maintains cartilage homeostasis, balances proteoglycan synthesis, and chondrocyte breakdown |
Due to limited blood supply to cartilage [20] and the low proliferative capacity of mature chondrocytes [21], cartilage regeneration is challenging. Previous studies have shown that exosomes derived from stem cells exhibit promising potential in addressing cartilage defects, osteoarthritis (OA), and osteoporosis (OP) [22]. Bone marrow mesenchymal stem cells (BMSCs) are associated with cartilage formation and can differentiate into chondrocytes [23]. During this differentiation process, exosomes promote angiogenesis through the Bone Morphogenetic Protein-2 (BMP-2)/Smad1/Runt-related transcription factor 2 (RUNX2) signaling pathway [24]. Additionally, numerous studies [25, 26] have demonstrated that BMSC-derived exosomes enhance cartilage repair and alleviate OA. BMSC-derived exosomes can maintain cartilage homeostasis and inhibit inflammatory responses by stimulating the production of polysaccharides and collagen [27]. Furthermore, BMSC-derived exosomes promote inflammation resolution and tissue repair through macrophage polarization [24].
To achieve optimal cartilage regeneration, exosomes are often combined with biomaterials. Hydrogels are one of the most commonly used three-dimensional scaffold materials, providing the structural support and moist environment necessary for cell growth, proliferation, and differentiation [28]. Chen et al. [29] found that the combined use of BMSC exosomes and hydrogels can promote BMSC migration, proliferation, differentiation, and repair of osteochondral defects. Moreover, studies have shown that 3D-printed cartilage extracellular matrix (ECM) and methacrylate gelatin (GelMA) can serve as an exosome scaffold, providing a moist three-dimensional environment for BMSC-derived exosomes to support cartilage regeneration in animal models [30].
Deer antler is a unique source of bone marrow mesenchymal stem cells with remarkable regenerative capabilities [17] revealed that exosomes derived from deer antler stem cells can reduce chondrocyte inflammation and promote cartilage repair in arthritis animal models. Our study found that exosomes from deer antler stem cells promote chondrocyte regeneration and repair cartilage defects. We demonstrated that ASC-Exos effectively repair cartilage damage and promote hyaline cartilage regeneration. In vivo studies indicated that ASC-Exos facilitate hyaline cartilage production at defect sites. In vitro experiments showed that interactions between articular chondrocytes and BMSCs favor the maintenance of the chondrocyte phenotype and the synthesis of hyaline cartilage. Consequently, we assessed the effects of ASC-Exos on both rat chondrocytes and BMSCs, finding that ASC-Exos significantly enhanced the proliferation and migration of both cell types. Additionally, ASC-Exos maintained the chondrocyte phenotype. Analysis of the protein components of ASC-Exos suggested that transferrin, S100A4, and insulin-like growth factor-binding protein 1 (IGF1) may play roles in promoting chondrocyte proliferation and migration and maintaining cartilage morphology.
In this study, protein component analysis conducted using the DIA method revealed two highly expressed proteins, S100A4 and Serotransferrin (TF). These proteins are likely key factors in the promotion of chondrocyte proliferation and migration by exosomes. TF is an important iron-binding transporter protein, facilitates the transport of iron from absorption and heme degradation sites to storage utilization sites in the body. Iron is essential for cell growth and metabolism, and iron deficiency leads to abnormal cell proliferation and function. Serum transferrin binds iron and delivers it to cells needing it, thus supporting cell proliferation. The S100A4 protein is a small-molecule (10–12 kDa) calmodulin that is widely found in many various cell types, including tumor cells, immune cells, fibroblasts, and neurons. The S100A4 protein is involved in cell migration and invasion by interacting with actin and non-actin cytoskeletal proteins. In cell migration, S100A4 protein promotes cell polarity and cytoplasmic skeleton reassembly, thereby enhancing cell motility and invasion. Additionally, S100A4 protein facilitates cell adhesion and detachment from the extracellular matrix, thereby facilitating cell movement and spreading. Moreover, our protein analysis identified IGF-1, a growth factor that stimulates chondrocyte proliferation and synthesis of cartilage matrix. IGF-1 initiates signaling pathways upon binding to its receptor, activating cell cycle protein kinases, regulating proliferation and differentiation, and enhancing matrix synthesis and cell adhesion. These actions promote cartilage repair by stimulating chondrocytes proliferation, increasing cell numbers for cartilage repair, and enhancing matrix synthesis to form hyaline cartilage. IGF-1 can promote chondrocytes to synthesize and secrete cartilage matrix molecules, such as COLII, chondroitin sulfate and proteoglycans, thus increasing the volume and elasticity, which may be the key to enable cartilage defect repair to form hyaline cartilage. IGF-1 also promotes chondrocyte differentiation to mature chondrocytes, thereby improving the effect of cartilage repair outcomes. Some studies have shown that IGF-I promotes the repair of large cartilage defects and reduces chronic synovial inflammation in animal models [31]. These proteins may allow chondrocytes and rat MSCs to migrate to the damaged area and promote cell proliferation at the damaged site, accelerating the chondrocyte repair process.
Although the mechanism by which ASC-Exos facilitate cartilage regeneration remains unknown, this study provides a valuable foundation for future research. Further investigations into ASC-Exos and their mechanism of action may uncover crucial insights into the processes underlying cartilage regeneration and develop more effective therapeutic approaches for treating cartilage injuries and diseases.
ASC-Exos have a significant effect on cartilage damage repair, which may be attributed to their promotion of chondrocyte and BMSCs proliferation and migration, as well as the maintenance of chondrocyte phenotype. This effect may be mediated by the presence of TF, S100A4, and IGF1.
All raw data for this study are available from the corresponding author. Raw data of proteomics data has been deposited in ProteomeXchange (PXD053936) and will be made available on the date of publication.
JZ and HMS designed the research study. JZ and JWZ performed the research. YMW, XSL, DTW, ZGY and JPL provided help and advice on all research. JZ, JWZ and YDJ analyzed the data. JZ and YMW wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The animal study was reviewed and approved by the Animal Ethics Committee of Institute of Special Animal and Plant Sciences (Permit Number: ISAPASAEC-2022-61B).
We sincerely appreciate Prof. Adem for revising the language of this manuscript.
This research was funded by Natural Science Foundation of Jilin Province of China, grant number: YDZJ202401459ZYTS.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





