1 Graduate School, Tianjin Medical University, 300000 Tianjin, China
2 Department of Geriatric Orthopedics, The Second Hospital of Tangshan, 063000 Tangshan, Hebei, China
3 Department of Minimally Invasive Spine Surgery, Tianjin Hospital, 300211 Tianjin, China
4 Department of Spine Surgery, Tianjin Hospital, 300211 Tianjin, China
Abstract
Background: Spinal cord injury (SCI) is considered a central nervous
system (CNS) disorder. Nuclear factor kappa B (NF-
Keywords
- spinal cord injury
- nuclear factor kappa B
- dynamin-related protein 1
- microglia polarization
- mitochondrial fission
A serious condition damaging the central nervous system (CNS) is spinal cord injury (SCI) [1]. Trauma (including motor vehicle accidents, sport injuries and falls) and spinal disease (including spinal tuberculosis, tumors and amyotrophic lateral sclerosis) can lead to SCI [2, 3, 4, 5]. SCI causes to serious social, economic and psychological damage to patients [6]. Pathological processes of SCI comprise irreversible primary damage and reversible secondary damage [7, 8]. Primary SCI can cause tissue damage, which impairs cellular and organ dysfunction [9]. Alleviating inflammatory response during secondary damage is crucial for the treatment of SCI.
Microglia are specialized CNS macrophages that monitor the microenvironment through extending and retracting branches [10, 11, 12]. In response to neuropathological stimuli, microglia migrate to the site of injury to eliminate damaged neural cells [13, 14, 15]. Microglial activation after injury can prevent neuronal cell death. A chronic microglial response can however lead to further damage owing to the excessive production of pro-inflammatory molecules [16, 17]. Polarizing microglia into M1 or M2 states can ameliorate neuronal inflammation and degeneration, promoting the recovery of neuronal function after SCI [18]. Remodeling microglia polarization is therefore crucial to reducing inflammatory responses and restoring motor functions after SCI.
Nuclear factor kappa B (NF-
Mitochondrial fragmentation and dysfunction are closely linked
to the progression of CNS disease [23] and are key contributors to the neuronal
injury observed in SCI [24]. The fragmented and dysfunctional mitochondria
release from microglia into the neuronal environment also increases neuronal cell
damage [25]. Dynamin-related Protein 1 (DRP1) is a
mitochondrial fussion’s extensive regulator in neurons. The inhibition of DRP1
leads to the collapse of mitochondria into perinuclear clusters, perturbing
mitochondrial homeostasis [26, 27]. Targeting DRP1 has shown promise for treating
Alzheimer’s disease [26]. We explored whether the effect of NF-
We obtained serum specimens from twenty SCI patients at the Second Hospital of Tangshan. This included patients with cervical spondylotic myelopathy (CSM), cervical fracture with SCI, mixed spondylosis (including cervical spondylotic radiculopathy and CSM), conus medullaris injury or cervical SCI without fracture and dislocation. Samples were compared to 20 control samples obtained from patients with fractures or thumb injuries. Written informed consent was acquired by each subject. The research was accepted by the Ethics Committee of the Second Hospital of Tangshan (approval number: 20220026).
Mouse BV2 microglia (No. CL-0493A; Procell, Wuhan, China) were kept in Dulbecco’s Modified Eagle’s Medium (DMEM) with 10% fetal bovine serum (FBS). We validated all cell lines by Short Tandem Repeat (STR) profiling and confirmed negative for mycoplasma. Cells were cultivated at 37 °C. We stimulated BV2 microglial cells with lipopolysaccharide (LPS) and BV2 cells with LPS (1 µg/mL, serotype O111:B4) for 1 d in the existence or absence of JSH-23 (No. HY-13982; MedChemExpress, Monmouth Junction, NJ, USA) using 50 µM.
Animal experiments were conducted. Procedures were permitted by the
Institutional Animal Care and Use Committee of Yi Shengyuan Gene Technology Co.,
Ltd. (protocol number YSY-DWLL-2023288). Sprague-Dawley rats (eight to ten week
old; 200–250 g) were housed in Specific pathogen Free (SPF) animal facilities
with free access to water and food, a 12-hour L/D cycle, and regulated
temperature (22–26 °C) and humidity (40–70%). We
randomized all rats into three groups (n = 6 per group): Sham, SCI, and
SCI+NF-
Hindlimb motor function was evaluation using Basso-Beattie-Bresnahan (BBB) scores [29]. According to the presence of motor function defects, the BBB rating scale from 0–21 scores (Supplementary Table 1), and 0–7 assessed the hindlimb joint movement, 8–13 assessed the gait and coordination of the hindlimbs, and 14–21 assessed the fine movements of the claws. Scores were recorded for each group at 1, 3 and 7 days after SCI. Functional scores were assigned by two observers independently.
We stained sections with hematoxylin (1:2 diluted in purified water) for two min and excess stain was removed through treatment with 0.3% acid alcohol. Sections were rinsed with tap water and counterstained with 1% eosin for 2 min. To stain nissl, spinal cord sections were dewaxed, hydrated and stained with toluidine blue (1%) for 40 min at 60 °C. Bright-field images were acquired from 3 random fields on a SOPTOP OD630K microscope (Yuyao, China).
Commercially available terminal deoxynucleotidyl
transferase-mediated 2
We fixed Mouse BV2 microglia in paraformaldehyde (4%), permeabilised in Triton X-100 (0.5%) and blocked in Bovine Serum Albumin (BSA, 2%). Cells were labelled at 4 °C overnight with anti-CD206 (ab300621, 1:100 dilution, Abcam, Cambridge, UK) and anti-CD86 (ab220188, 1:100 dilution, Abcam, Cambridge, UK) antibodies and stained with fluorescent conjugated secondary antibodies (ab150077, 1:250 dilution, Abcam, Cambridge, UK) for 2 h. Fluorescent images were captured by means of a BH2-RFC (OLYMPUS, Tokyo, Japan) microscope and examined by Image Pro Plus software (Media Cybernetics, MD, USA).
Spinal cord tissues were fixed in glutaraldehyde (2.5%) for 4 h at 4 °C and then 1% osmic acid for 2 h at 4 °C. We dehydrated samples using graded ethanol (30%–100%), rinsed three times in propylene oxide and embedded in Epon812 resin (Spi-Chem, Shanghai, China). Samples were sliced into sections of 70 nm, and stained with uranyl acetate (3%) and then lead citrate (2.7%) for 8 min. Segments were imaged using TEM (HT7700, HITACHI, Tokyo, Japan).
For polarization assays, treated BV2 cells were re-suspended in PBS and stained with conjugated anti-F4-80 (F21480A03, LIANKE BIO, Hangzhou, China), anti-CD86 (F2108601, LIANKE BIO, Hangzhou, China) or anti-CD163 (ab282114, Abcam, Cambridge, UK) antibodies at 4 °C for 1 h. Intracellular reactive oxygen species (ROS) levels were detected following staining with DCFH-DA (CAS No.:S0033M, Beyotime, Shanghai, China) for 20 min at 37 °C. Mitochondrial membrane potential (MMP) was measured through staining with JC-1 (CAS No.:C2006, Beyotime, Shanghai, China) for 20 min at 37 °C. Flow cytometry was performed using a BD FACSCalibur™ (BD Biosciences, New York, NJ, USA). Data analysis was done with FlowJo software (Kentucky, New York, NJ, USA).
Serum levels of interleukin-6 (IL-6), IL-1
We extracted total RNA from tissues and BV2 cell samples utilizing Redzol
reagent (No. FTR-50; SBS Genetech Co., Ltd. Shanghai, China). cDNA synthesis was
performed using Surescript™ First-Strand cDNA Synthesis Kits (No.
QP056; GeneCopeia, Rockville, MD, USA). RT-PCRs were performed by means of 2
| Genes | Primers |
|---|---|
| Inos | F-5 |
| R-5 | |
| Il-1 |
F-5 |
| R-5 | |
| Il-6 | F-5 |
| R-5 | |
| Il-4 | F-5 |
| R-5 | |
| Gapdh | F-5 |
| R-5 |
Cells were lysed and proteins were quantified via Bicinchoninic Acid (BCA)
assays. We separated proteins using 10% SDS-PAGE gels and transported onto PVDF
membranes. Membranes were probed with anti-dynamin-related protein 1 (DRP1,
ab184247; dilution 1:300, Abcam, Cambridge, UK), anti-pDRP1 (phospho S616;
ab314755; dilution 1:350, Abcam, Cambridge, UK), anti-NF-
For IHC, samples were dewaxed, hydrated, blocked in goat serum (5%) for 20 min and stained with 0.2 mg/mL anti-CD206 (ab300621, dilution 1:2000, Abcam) and anti-CD86 (ab220188, dilution 1:2000, Abcam) antibodies at 4 °C for 1 d. Then, sections were stained with the relevant secondary antibody for 20 min, stained with Diaminobenzidine (DAB, ZSGB-BIO, Beijing, China) and counterstained with 1% hematoxylin for 2 min (Beyotime, Shanghai, China). Three random imagesd were captured for each treatment.
Data were presented as the mean
Pro-inflammatory and anti-inflammatory factors in serum samples from control and
SCI patients were measured via ELISA. Serum IL-6, IL-1
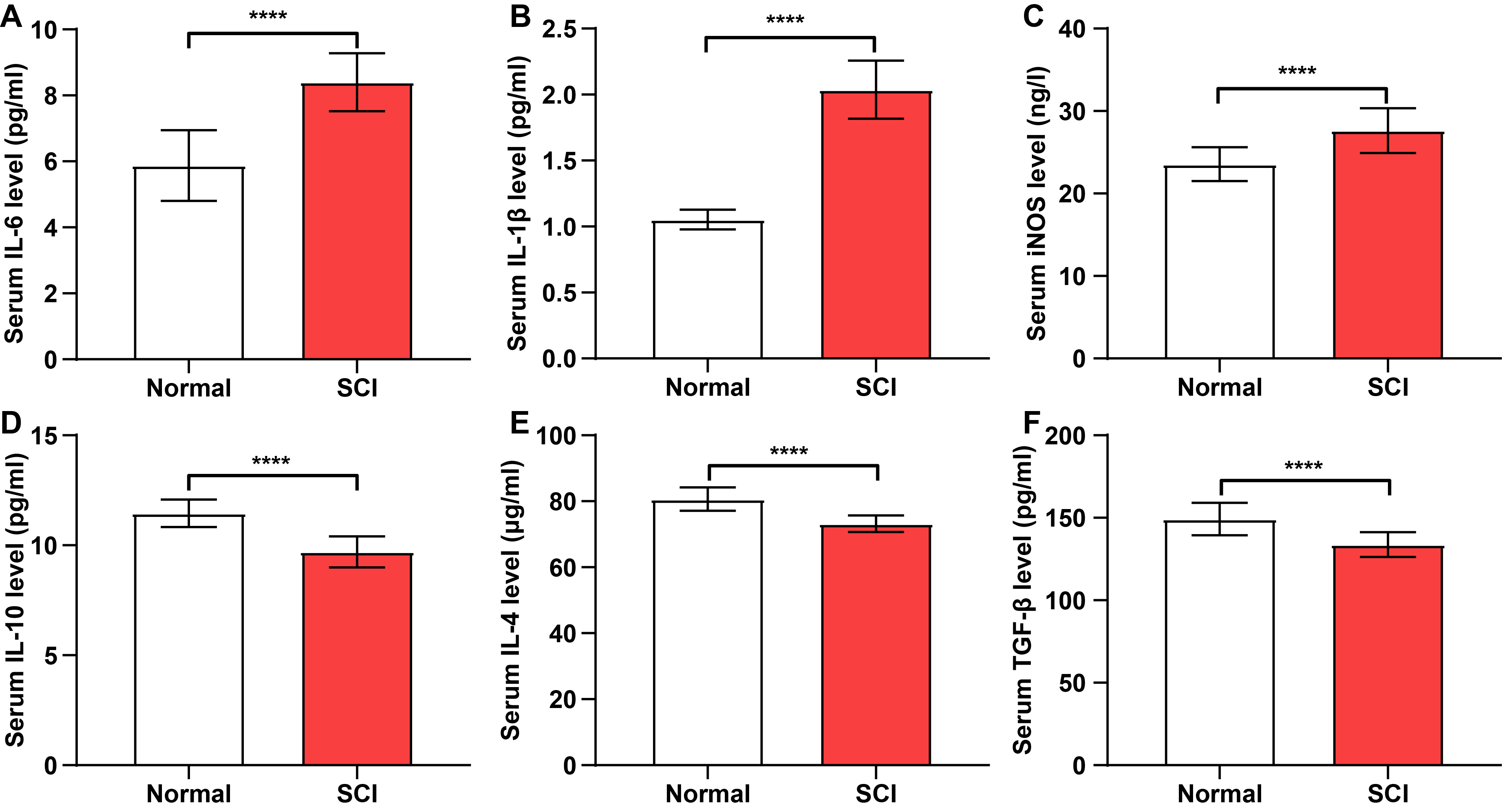 Fig. 1.
Fig. 1.
Pro-inflammatory cytokine levels are elevated in the serum of
spinal cord injury (SCI) patients. (A–F) Concentrations of
interleukin-6 (IL-6), interleukin-1beta (IL-1
Rat models were established. H&E staining of spinal cord
tissue in control rats showed no signs of inflammatory cell infiltration,
hemorrhage, or necrosis (Fig. 2A). Conversely, spinal cord tissue in SCI rats
showed hemorrhage, nerve cell edema, necrosis and inflammatory cell infiltration,
confirming establishment of the SCI model (Fig. 2A). Consistent
with previous data, NF-
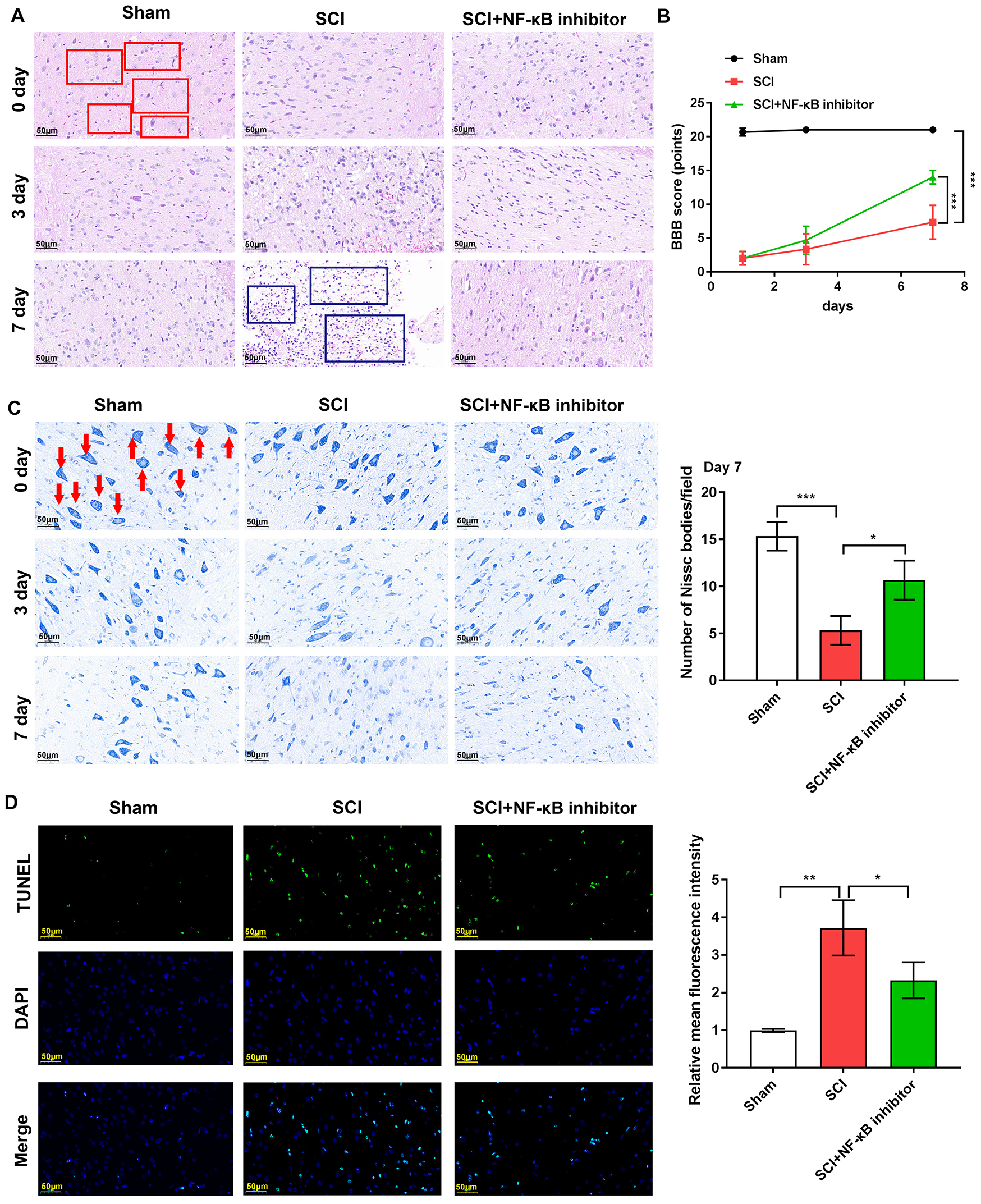 Fig. 2.
Fig. 2.
Nuclear factor kappa B (NF-
Locomotion function was evaluated using BBB scores. For the SCI group, BBB
scores were 2.00
Nissl bodies were reduced in the rat spinal cord tissues
following SCI. NF-
The effects of NF-
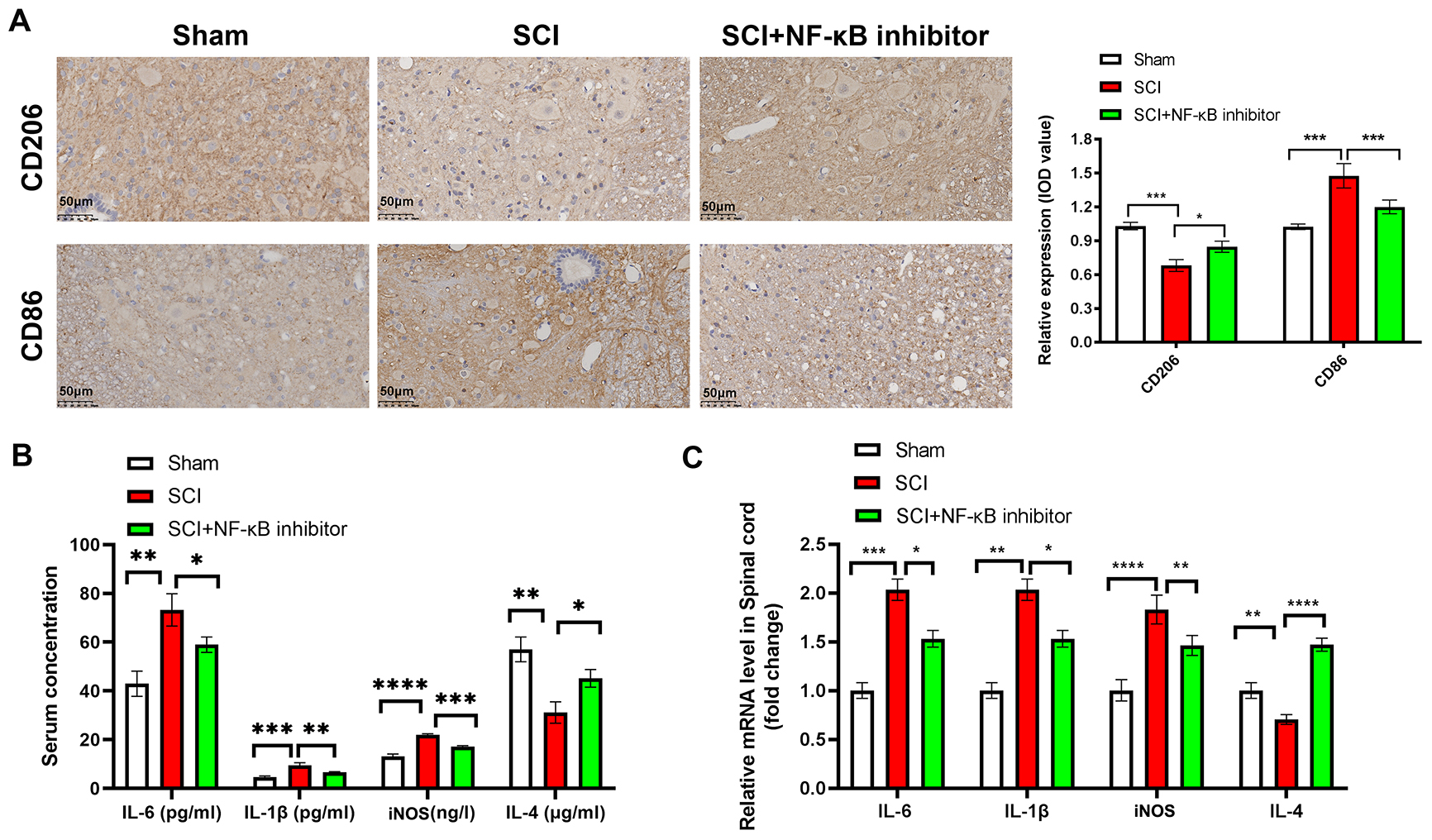 Fig. 3.
Fig. 3.
NF-
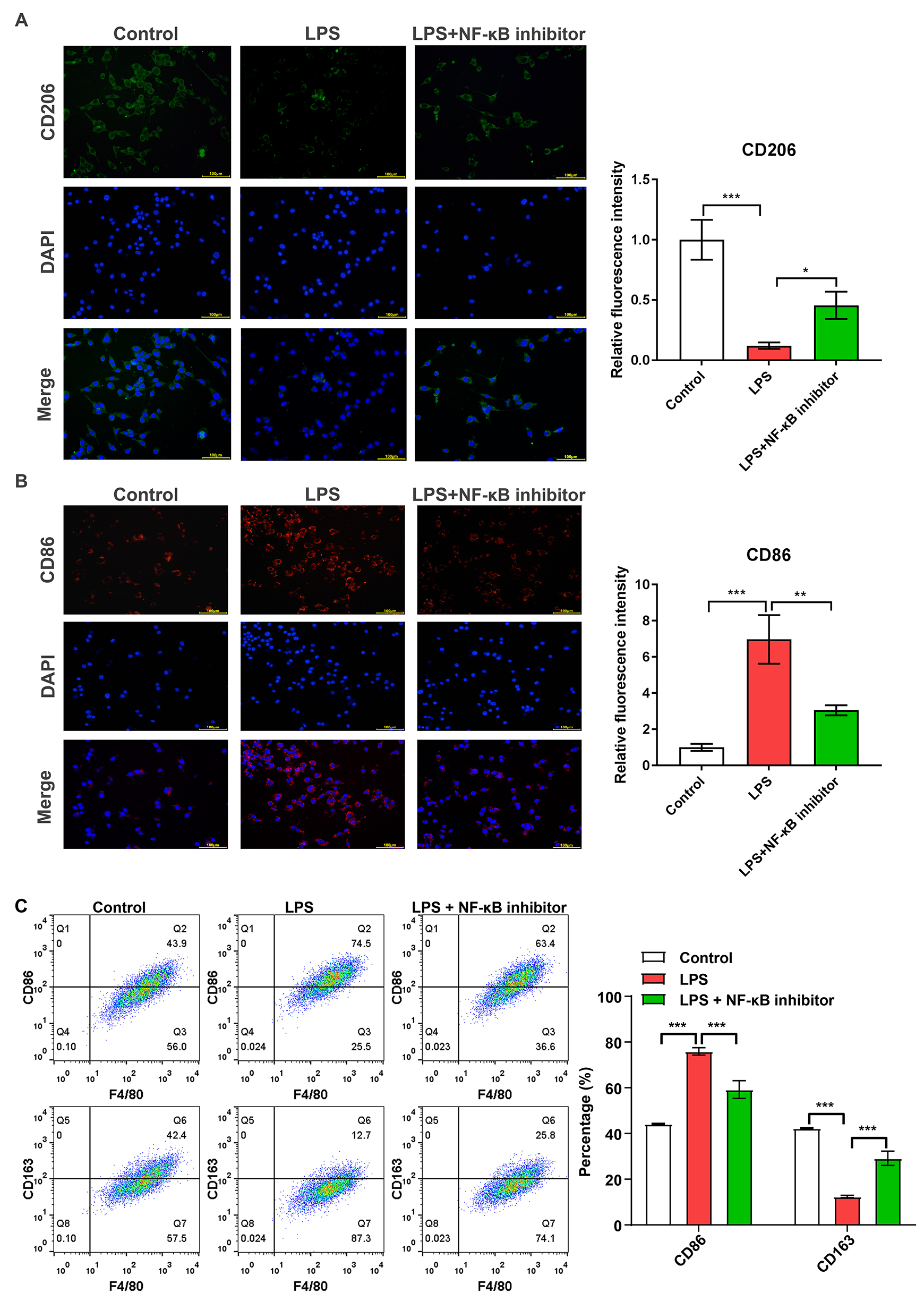 Fig. 4.
Fig. 4.
NF-
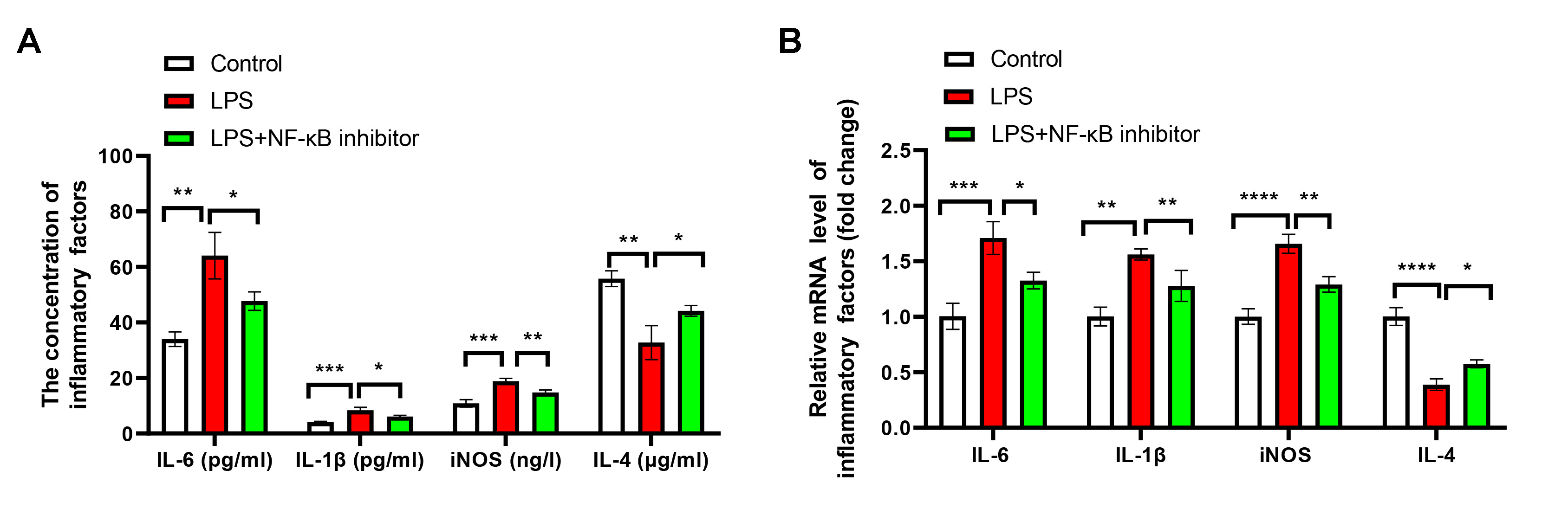 Fig. 5.
Fig. 5.
NF-
The impacts of NF-
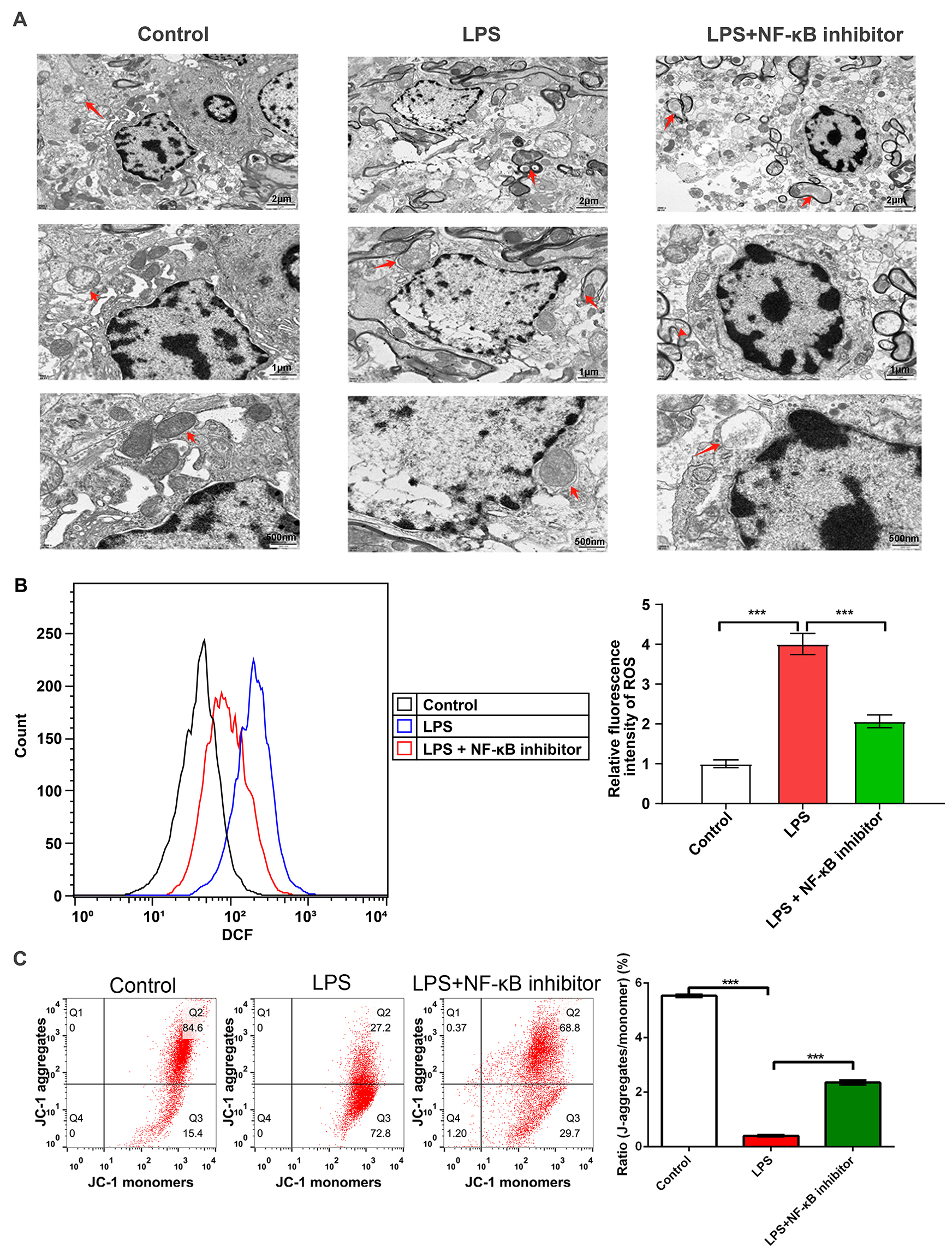 Fig. 6.
Fig. 6.
NF-
The p-DRP1/DRP1 and NF-
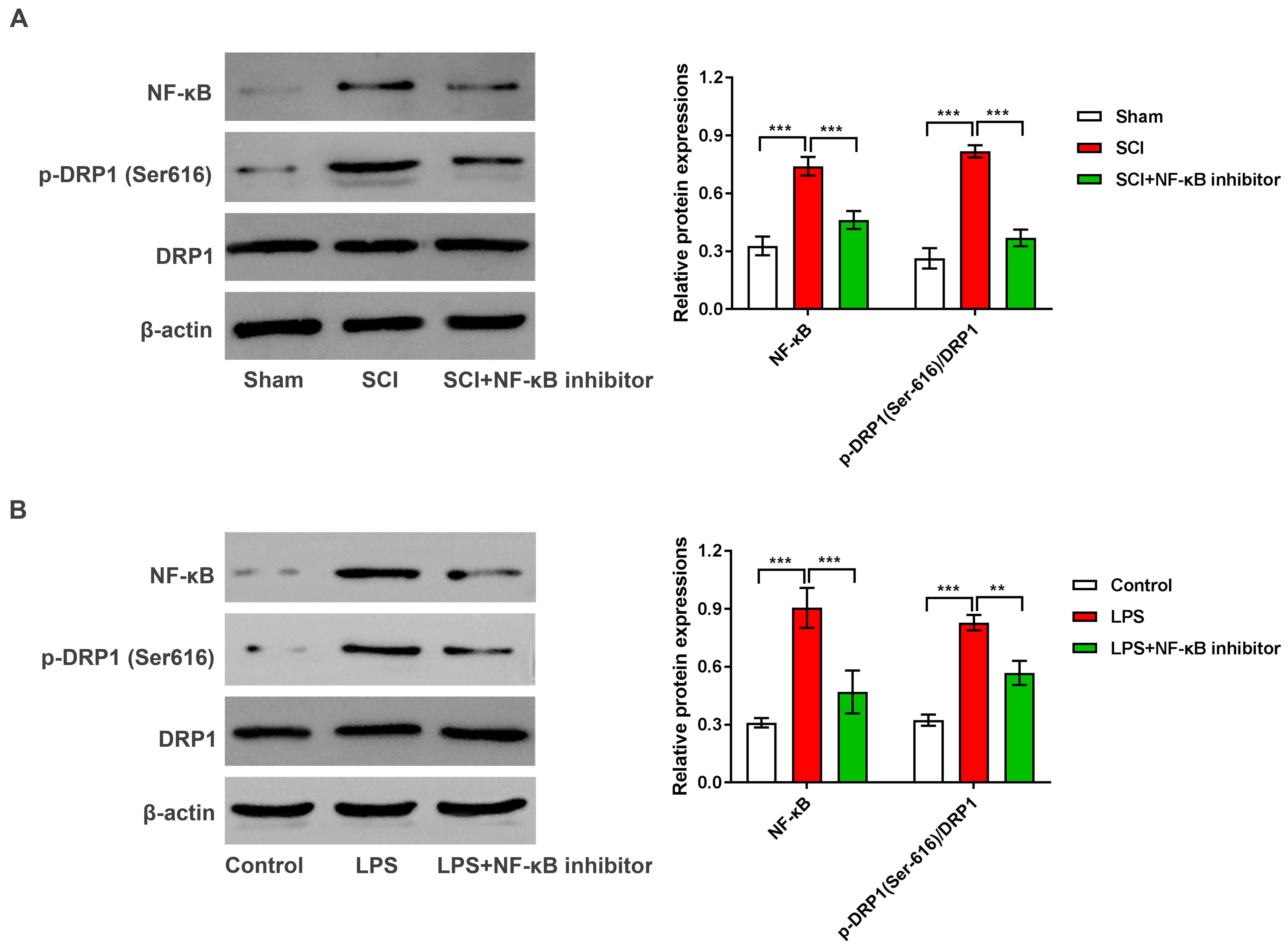 Fig. 7.
Fig. 7.
NF-
Secondary injury processes in SCI promote neuronal apoptosis,
a significant obstacle to the treatment of spinal cord repair. Mechanisms to
prevent tissue and cell damage can serve as a therapeutic strategy to prevent or
minimize secondary damage following SCI, thereby promoting neuronal function. In
our study, we observed the effects of NF-
Microglia functions in neuroinflammation after SCI [30].
Over-activation of pro-inflammatory microglia (M1 type)
following SCI triggers the release of inflammatory mediators
(IL-1
Mitochondria dysfunction is a key pathological feature of SCI [23].
Under physiological conditions, this equilibrium is essential
for maintaining mitochondrial function [37]. Pathological conditions can however
disrupt this balance [38, 39]. DRP1 is a dynamin family Guanosine Triphosphate
(GTPase) that mediates mitochondrial and peroxisomal division in neurons [40] the
activity of which is enhanced by phosphorylation at Ser616 [41]. In this study,
we observed a significant elevation of p-DRP1 in LPS-treated
microglia and in the SCI rat spinal cord reversed by NF-
Previous studies highlight a complex relationship between DRP1 and
NF-
Despite our findings, some limitations of this study should be considered.
Mitochondrial assays exclusively focused on microglia. Future studies
encompassing multi-cellular types, including nerve and immune cells are now
required to confirm our findings. Since, DRP1 is directly phosphorylated by a
number of upstream kinases in neurons, including
Cyclin-dependent Kinase 5 (CDK5),
Calcium-CaM-dependent protein kinase (CAMK), PTEN induced putative kinase 1
(PINK1) and
Protein Kinase B (AKT), further research
are also required to elucidate the target of NF-
We show that inhibition of the NF-
The data that support the study findings are available on request from the corresponding author.
CS and BSX planned the study, CS wrote the original draft. KHZ, CL and XYZ did experiments and acquired the data. BSX examined the data. All authors made editorial changes in the manuscript. Each author read and approved the final draft. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All participants provided written informed consent. This research was permitted by the Ethics Committee of the Second Hospital of Tangshan (approval number: 20220026). Animal experiments were permitted by the Institutional Animal Care and Use Committee of Yi Shengyuan Gene Technology Co., Ltd. (protocol number YSY-DWLL-2023288).
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







