1 Shaanxi Key Laboratory of Ischemic Cardiovascular Diseases & Institute of Basic and Translational Medicine, Xi’an Medical University, 710021 Xi’an, Shaanxi, China
2 Department of Cardiovascular Surgery, The First Affiliated Hospital of Xi’an Jiaotong University, 710061 Xi’an, Shaanxi, China
Abstract
Background: This study aimed to elucidate the molecular mechanism through which C1q/tumor necrosis factor (TNF)-related protein 9 (CTRP9) acts in the formation and differentiation of brown adipose tissue (BAT). Methods: Adenovirus particles encoding CTRP9 and green fluorescent protein were inoculated into the scapula of C57BL/6J mice and fed a high-fat diet for 8 weeks; the body weight, lipid droplet morphology, glucose tolerance, insulin tolerance, and protein expression levels were analyzed. In addition, CTRP9 adenovirus was transfected into brown preadipocytes, and differentiation was induced to identify the effect of CTRP9 overexpression on adipocyte differentiation. Results: CTRP9 overexpression significantly increased the weight gain of mice. Additionally, the CTRP9 overexpression group exhibited significantly increased adipose tissue weight and glucose clearance rates and decreased insulin sensitivity and serum triglyceride levels compared to the control group. Furthermore, CTRP9 overexpression significantly upregulated the adipose triglyceride lipase (ATGL) and perilipin 1 protein expression levels in BAT. The cell experiment results confirmed that CTRP9 overexpression significantly inhibited the adipogenesis of brown adipocytes as evidenced by the downregulation of uncoupling protein 1, beta-3 adrenergic receptor, ATGL, and hormone-sensitive lipase mRNA levels and the significant suppression of uncoupling protein 1, ATGL, and perilipin 1 protein levels in brown adipocytes. Conclusions: The finding of this study demonstrated that CTRP9 promotes lipolysis by upregulating ATGL expression in vivo and inhibits the differentiation of brown preadipocytes in vitro.
Keywords
- CTRP9
- UCP-1
- ATGL
- lipolysis
- brown adipocytes
Obesity, which results from an imbalance between long-term energy intake and
energy consumption, is increasing worldwide [1]. Currently, approximately
one-third of the global population (2.2 billion individuals) is overweight, with
10% classified as obese [2]. Obesity increases the risk of various diseases,
including insulin resistance [3], type 2 diabetes mellitus (T2DM), and
cardiovascular disease [4]. T2DM, a chronic metabolic disease, results from
increased blood glucose levels caused by peripheral insulin resistance, resulting
in islet
Mammalian adipose tissue is primarily categorized into the following two types: white adipose tissue and brown adipose tissue (BAT) [9]. This categorization reflects the diversity in adipose tissue composition and function across mammalian species. White adipocytes store and mobilize lipids [10]. Glucose is transported into adipocytes to synthesize triglycerides (TGs) through a series of reactions [11]. To generate thermal energy, white adipocytes undergo lipolysis, hydrolyzing TGs into free fatty acids (FFAs). Subsequently, these FFAs are released to serve as metabolic fuel, providing energy [12]. In contrast, brown adipocytes, which are rich in mitochondria and form multilocular lipid droplets, specifically consume chemical energy to produce thermal energy [13]. UCP1, which has a critical role in the inner membrane of mitochondria, regulates thermal energy production. Additionally, UCP1 separates mitochondrial respiration from ATP synthesis, promoting the production of cellular thermal energy [14]. Lipid droplets, which comprise a highly hydrophobic ester lipid core, are dynamic intracellular vesicles surrounded by a phospholipid monolayer with several attached proteins [15]. Perilipin 1 is considered a marker for lipid droplets as it is located on the surface of these droplets [16, 17].
C1q/tumor necrosis factor-related proteins (CTRPs) were reported to be expressed in adipose tissue by Wong et al. [18]. Currently, 15 CTRPs (CTRP1–15) have been identified [18]. CTRP9, also called C1QTNF9, which belongs to the family of CTRPs [19], is the closest adiponectin paralog and is evolutionarily conserved. This conservation is evident in their shared structural domain, which includes an N-terminal signal peptide, a collagen-like domain, a short variable domain, and a C-terminal C1q-like globular domain [20]. CTRP9 was reported to regulate the pathological processes associated with obesity [21], lipid metabolism [22], cardiovascular disease [23], myocardial ischemia [24], apoptosis [25], and pulmonary artery pressure [26]. Thus, these critical roles of CTRP9 in pathological and physiological conditions have piqued the interest of the scientific community [27]. However, the effect of CTRP9 on brown adipocyte formation and lipid metabolism has not been elucidated. This study used the adenovirus overexpression constructs to evaluate the impact of CTRP9 overexpression on lipid metabolism in brown adipocytes obtained from the high-fat diet (HFD)-induced obesity mouse model.
Male mice aged 6 weeks (body weight: 20–22 g) were procured from the Laboratory Animal Center of Xi’an Jiaotong University. Each cage accommodated a maximum of three animals. The mice were maintained in a hygienic, controlled, and pathogen-free environment under the following conditions: access to food and water, ad libitum; relative humidity, 50%–80%; temperature, 20 °C–25 °C; circadian cycle, 12 h light–dark cycle.
The mice were categorized randomly into the following two groups (10
mice/group): Ad-GFP group, injected with the green fluorescent protein
(GFP)-encoding adenoviral constructs; Ad-CTRP9 group, injected with the
CTRP9-encoding adenoviral constructs. Mice were administered with 1
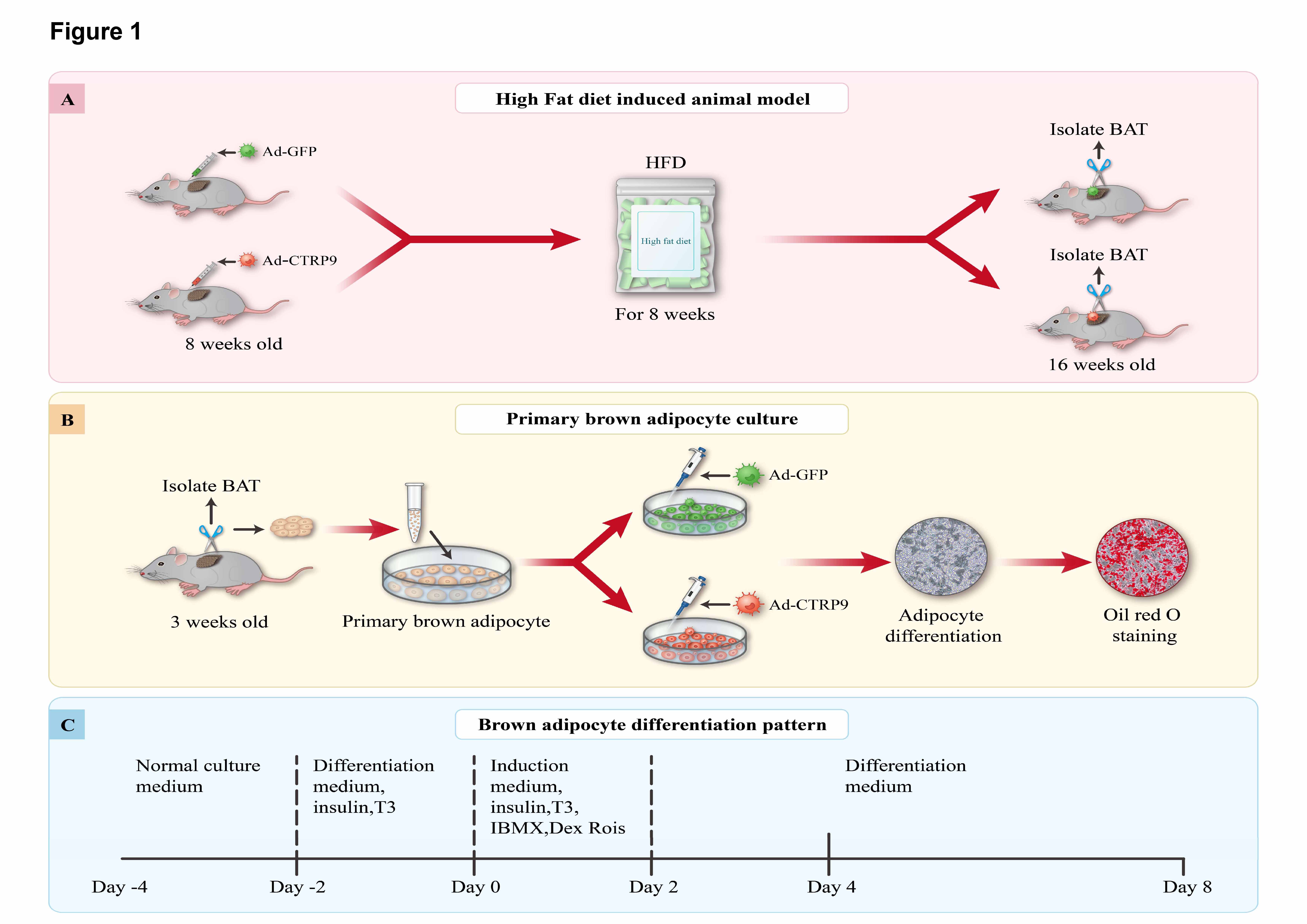 Fig. 1.
Fig. 1.The experimental flowchart and brown adipocyte differentiation
protocol. (A) Male mice were randomly divided into two groups, and their BAT was
injected with 1
Mice were euthanized through the intraperitoneal injection of pentobarbital sodium (150 mg/kg body weight). The experimental procedures adhered to the guidelines for animal experiments at Xi’an Jiaotong University, which were derived from the Guide for the Care and Use of Laboratory Animals (NIH Publication, 8th Edition, 2011). The experimental procedures were approved by the Laboratory Animal Administration Committee of Xi’an Jiaotong University (Institutional Animal Care and Use Committee; Permit No. 2022-1323).
Brown preadipocytes were isolated from the interscapular BAT of C57BL/6J mice
aged 3 weeks using collagenase I digestion (Fig. 1B) [28]. The isolated cells
were seeded in 6-well plates (1
Plasma lipids were analyzed following a previously reported protocol [29]. High-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), TG, and plasma total cholesterol (TC) were examined using commercial assay kits (Biosino Bio-Technology & Science Inc., Beijing, China) [30]. Plasma FFAs were measured using an enzyme-linked immunosorbent assay kit (Shanghai Jingkang Biotechnology Co., Ltd., Shanghai, China).
GTT and ITT were performed with mice that were allowed to fast for 4–6 h, based on previously reported protocols [31]. Mice were intraperitoneally injected (i.p.) with 25% (w/v) glucose (1.5 g glucose/kg body weight) or insulin (0.75 U/kg body weight). In GTT, the glycemic levels were measured before the glucose challenge and post at 15, 30, 60, 90, and 120 min using a OneTouch® Glucometer (Lifescan, Milpias, CA, USA).
Frozen tissue sections were washed twice with phosphate-buffered saline (pH 7.2), fixed with 4% paraformaldehyde for 30 min, and stained with oil red O solution at room temperature for 30 min. Next, the sections were washed twice with distilled water and imaged under an optical microscope. The image analysis system (WinROOF, Mitani Co., Tokyo, Japan) was utilized to quantify the oil red O-stained area.
qRT-PCR analysis was performed as previously described [32]. Briefly, total RNA
was extracted from the BAT using TRIzol Plus (Invitrogen, Carlsbad, CA, USA). The
isolated RNA was reverse-transcribed into complementary DNA using a
SuperScript® III First-Strand Synthesis System (Invitrogen,
Carlsbad, CA, USA). qRT-PCR analysis was performed using TaKaRa TP800 (TaKaRa
Biology Inc., Shiga, Japan). The relative gene expression levels were determined
using the 2
Cell or tissue lysis buffer was used to lyse brown adipocytes, and the resulting
supernatant was subjected to Western blotting using primary antibodies against
UCP-1 (No. MAB6158, R&D, Minneapolis, MN, USA), adiponectin (No. AF1119, R&D,
Minneapolis, MN, USA), perilipin 1 (No. 3467, CST, Boston, MA, USA), adipose
triglyceride lipase (ATGL) (No. T58408, Abmart, Shanghai, China), CTRP9 (No.
NBP2-46834, Novus, Centennial, CO, USA), and
BATs were excised and fixed using a 10% formalin buffer. The fixed specimens were processed to obtain frozen blocks, which were then sectioned and subjected to hematoxylin–eosin (H&E) and oil red O staining, following standard procedures. To quantify staining intensity, the sections were imaged using a microscope equipped with a digital camera (Nikon, Tokyo, Japan). ImageJ 1.52g software (NIH, Bethesda, MD, USA) was used to measure the diameter and area of adipocytes. The area of the oil red O-stained region was quantified.
Data are presented as the mean
To examine the effect of CTRP9 on HFD-induced BAT lipid accumulation, the BAT of
mice was administered with adenovirus vectors encoding CTRP9 or GFP through
scapular multipoint injection. The body weight of the Ad-CTRP9 group was
significantly higher (p
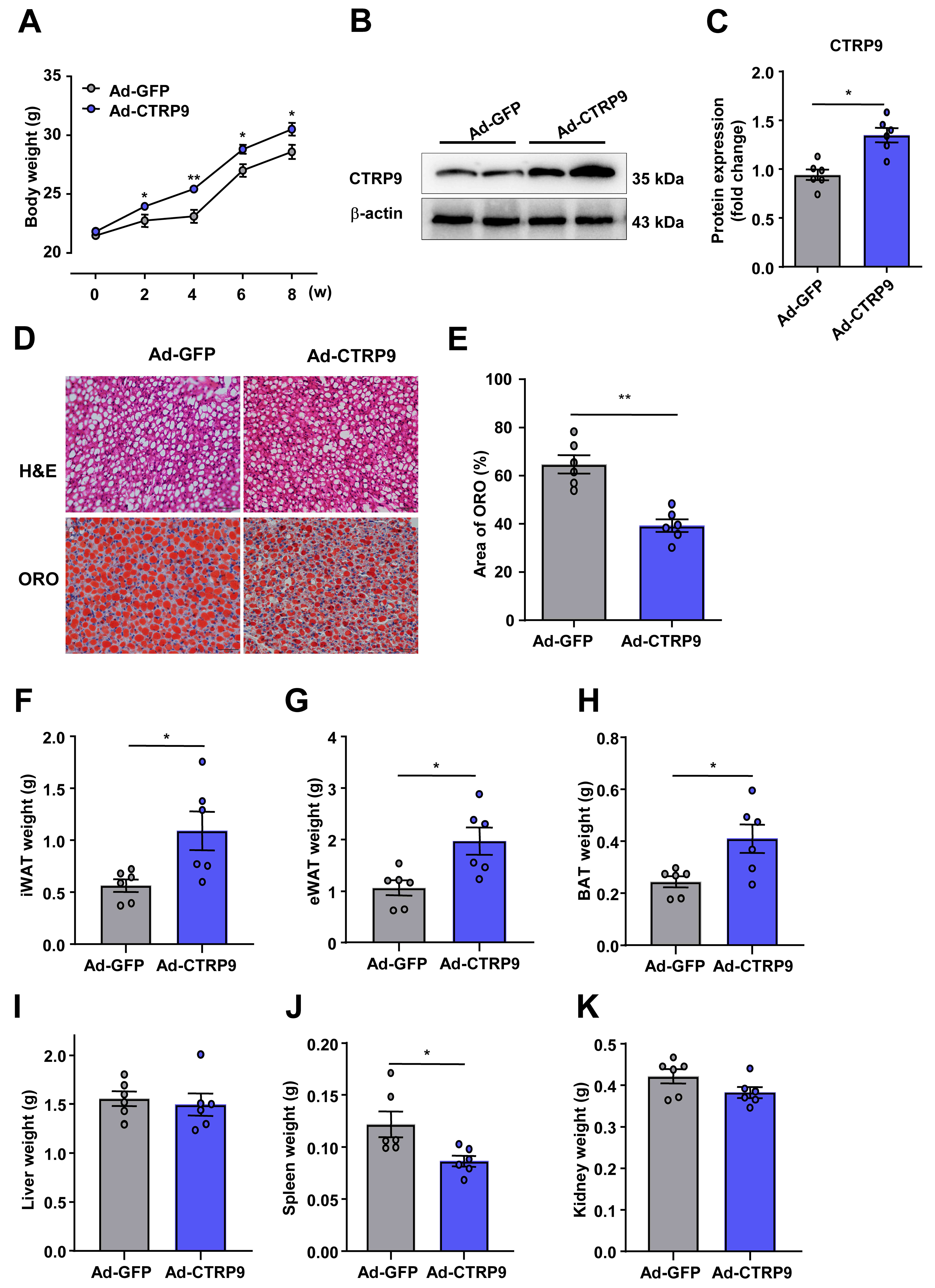 Fig. 2.
Fig. 2.Analysis of the CTRP9 expression level in BAT. (A) The body
weight of mice. (B) Protein expression of CTRP9 was determined using Western
blotting analysis. (C) Quantification of Western blotting analysis results
(
To examine the effect of CTRP9 on blood glucose levels, GTT and ITT were
performed to establish the post-glucose challenge levels at 15, 30, 60, and 120
min. Compared with that in the Ad-GFP group, glucose clearance efficiency was
significantly higher (p
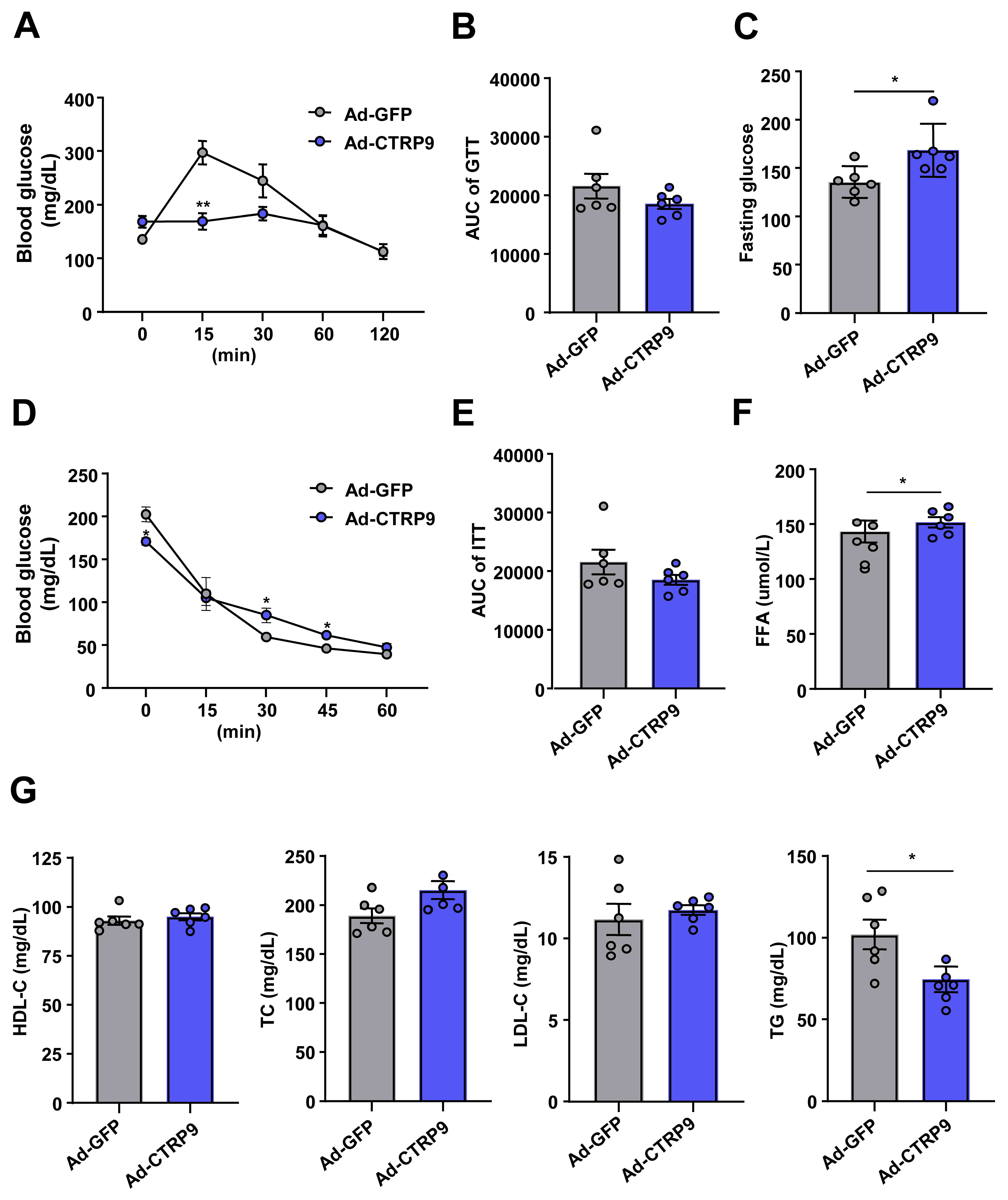 Fig. 3.
Fig. 3.CTRP9 overexpression decreases glucose clearance efficiency in
mice fed a HFD. (A) IpGTT in mice after 12 weeks of being fed a HFD. (B) The AUC
of IpGTT was calculated from the original graph. (C) Fasting blood glucose
concentration was measured after being fed a HFD for 12 weeks. (D) ITT results of
mice. (E) The AUC of the ITT results was calculated. (F) Quantification of plasma
FFAs. (G) Quantification of measured plasma TC, TG, HDL-C, and LDL-C levels. n =
6 for each group. Mean
To assess the molecular mechanism underlying the CTRP9-induced reduction in
lipid droplet content in BAT, BAT was isolated and subjected to Western blotting
and qRT-PCR analyses to determine the protein and gene expression levels,
respectively. In the Ad-CTRP9 group, the expression levels of lipolysis-related
genes, including those encoding hormone-sensitive lipase (HSL) (p
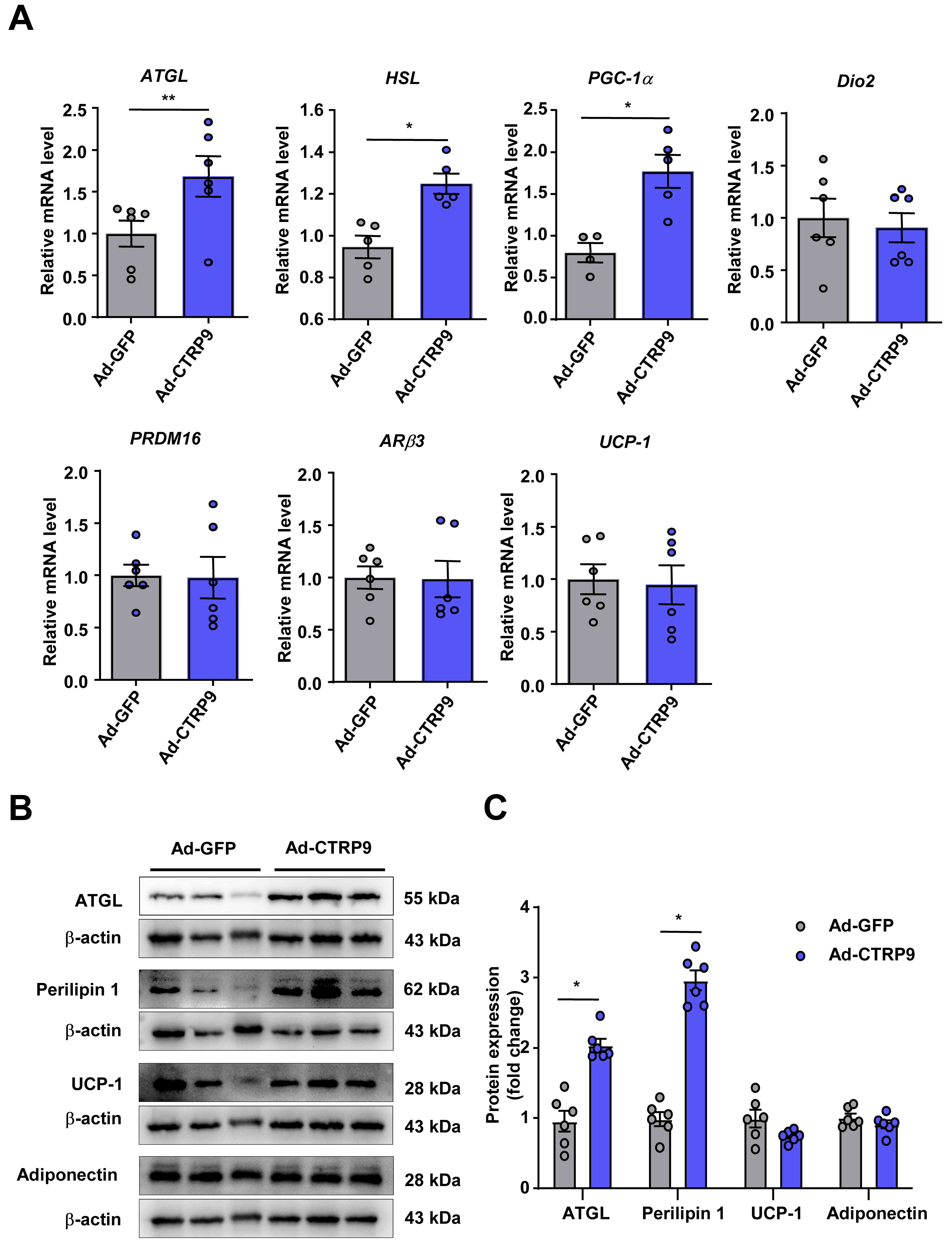 Fig. 4.
Fig. 4.CTRP9 promotes BAT lipolysis induced by a high-fat diet.
(A) The expression levels of adipogenic markers in BAT (UCP-1,
PGC-1
To investigate the potential involvement of CTRP9 in the regulation of brown
adipocyte differentiation, the impacts of CTRP9 were assessed throughout brown
adipogenesis. Brown preadipocytes were isolated from murine BAT and infected with
adenoviral constructs encoding CTRP9 (Fig. 1B). The differentiation rates of
brown adipocytes in the CTRP9-transfected and control groups were approximately
30% and 90%, respectively. These findings suggest that CTRP9 overexpression
inhibits brown adipocyte differentiation (Fig. 5A). The protein levels of CTRP9
were examined using Western blotting analysis. On day 8, the mRNA and protein
levels of CTRP9 were markedly upregulated (p
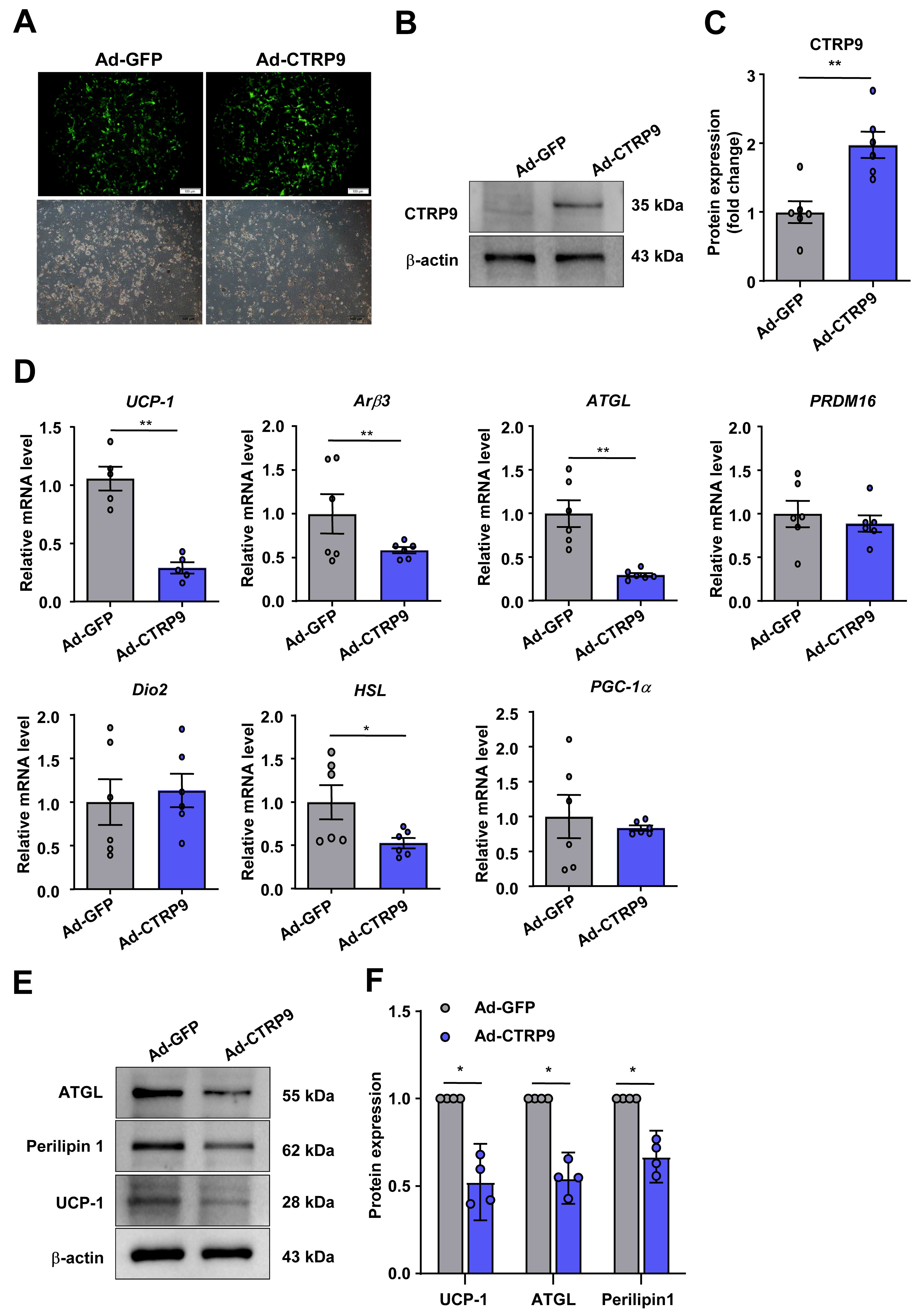 Fig. 5.
Fig. 5.CTRP9 inhibits brown preadipocyte differentiation. (A)
Morphological observation of lipid droplet formation in adipocytes.
Magnification, 200
Western blotting and qRT-PCR analyses were conducted to assess the adipogenic
markers of brown adipocytes (Fig. 5D–F). At the end differentiation time point
of brown adipocytes, the mRNA expression levels of UCP-1, beta-3 adrenoceptor
(
This study used the local injection of adenovirus constructs to overexpress CTRP9 in BAT and demonstrated that CTRP9 promotes diet-induced obesity, which contributes to decreased adipocyte size and lipid droplet number in BAT, resulting in the significant upregulation of ATGL and perilipin 1 protein expression levels. In vitro analysis indicated that CTRP9 served as a negative regulator of brown adipocyte differentiation. After administering Ad-CTRP9 in brown preadipocytes, the adipogenesis-related genes and proteins were significantly downregulated compared to the Ad-GFP group.
Peterson et al. [21] established CTRP9-overexpressing transgenic mice to investigate lipid metabolism in the systematic circulation. CTRP9 overexpression prevented obesity, insulin resistance, and liver steatosis, which were not observed in wild-type mice. This beneficial effect of CTRP9 overexpression was mediated through the reduction in food intake and the augmentation of basal metabolism [18]. The deficiency in CTRP9 exacerbated HFD-induced glucose and lipid metabolism disorders, accompanied by increased body weight and the upregulation of FFAs and serum TGs [33]. CTRP9 was locally overexpressed in the mouse scapular BAT. Thus, the body weight in the Ad-CTRP9 group was higher than in the Ad-GFP group. This novel phenomenon presents a notable divergence from findings in previous research [21, 33]. Conversely, CTRP9 overexpression promoted lipolysis by upregulating the expression levels of ATGL and HSL in BAT. Lipolysis is a sequential process occurring at the cytosolic lipid droplets, starting with the gatekeeper enzyme ATGL catalyzing the hydrolysis of TGs into diglycerides [34]. Hence, the local overexpression of CTRP9 was hypothesized to upregulate the plasma FFAs and enable the storage of excess energy in the white adipose tissue depot, leading to weight gain.
Cells are not passive bystanders during the hormone signaling process but actively customize the role of hormones [35]. Thyroid hormones contribute to heat production in BAT and affect the energy balance of both rodents and humans [36]. During the cold exposure-induced activation of BAT, the induction of Dio2 enhances local thyroid hormone signal transduction and increases energy consumption [37]. Previous studies have demonstrated that Dio2 knockout mice exhibit intolerance to cold stimulation, suggesting a crucial role for thyroid hormones in adaptive BAT thermogenesis. This study hypothesized an alternative mechanism through which CTRP9 overexpression protects mammals by upregulating the expression levels of Dio2 and consequently amplifying the local thyroid hormone signal and promoting thermogenesis.
Non-shivering thermogenesis is an energy-consuming process that primarily occurs in the BAT. In addition to their involvement in maintaining body temperature, these thermogenic adipocytes maintain glucose and lipid homeostasis in the bloodstream [38]. Thermogenesis in brown adipocytes depends on the import of lipids from the bloodstream. After entering the brown adipocytes, these lipids are degraded to serve as energy sources or signaling molecules. In this study, CTRP9 overexpression inhibited the differentiation of brown preadipocytes into mature adipocytes. Brown adipocytes differentiate into several multilocular lipid droplets, facilitating the absorption of extracellular FFAs and TGs. Therefore, the results of this study are consistent with those of previous animal studies. The decreased differentiation level of brown preadipocytes effectively explains the significant increase in body weight of mice in the Ad-CTRP9 group relative to the Ad-GFP group. Consistently, Haemmerle et al. [39] reported that compared with those in the Ad-GFP group, lipolysis was upregulated, and the protein expression was downregulated in the Ad-CTRP9 group.
Two or more members of the perilipin family, which comprises a group of lipid droplet surface proteins, decorate the lipid droplets in mammals [40]. The perilipins sequester lipids by protecting lipid droplets from lipase action [15]. In specific cells, perilipins adjust their protective properties and relative expression to maintain a balance between lipid storage and utilization [15]. Perilipin 1, which belongs to the perilipin family, is involved in suppressing lipolysis in adipose tissues [41]. In perilipin 1-deficient mice adipose tissue, triacylglycerol turnover was accelerated, leading to a reduction of approximately 70% adipose tissue mass [17]. Hence, perilipin 1 facilitates triacylglycerol storage under physiological conditions, at least partially by limiting the access of cytosolic lipase to triacylglycerol substrate stored in lipid droplets [17]. This research demonstrated that CTRP9 overexpression partly promotes lipolysis in BAT of mice by downregulating perilipin 1.
In vivo and/or in vitro studies have demonstrated that systemic CTRP9 overexpression enhances lipid metabolism [21], reduces systemic inflammation to alleviate insulin resistance [8], and decreases liver lipid accumulation and apoptosis [38]. These characteristics suggest that CTRP9 is a potential therapeutic target for diabetes, liver steatosis, and obesity. This study revealed that local CTRP9 overexpression in BAT of the scapula resulted in enhanced lipolysis of BAT and weight gain. Hence, additional evidence is required to establish the relationship between lipid-related diseases and CTRP9. A subsequent study will elucidate the role of CTRP9 in human lipid metabolism with a focus on BAT.
The local injection of CTRP9-encoding lentiviral particles into the scapula promoted weight gain in wild-type mice fed a HFD. CTRP9 overexpression increased the lipolysis of BAT, upregulated ATGL expression, and downregulated perilipin 1 expression. Although the molecular pathways were not elucidated, this study proposes that the dynamic balance of lipid droplet formation markedly contributes to CTRP9-mediated BAT lipolysis.
ATGL, adipose triglyceride lipase; Ad, adenovirus; AR
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
HG, BWD, ZYG, YG, and ZYL performed the experiments. TS, LW, and XLZ analyzed the data. HG and FWG designed the study and prepared the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Laboratory Animal Administration Committee of Xi’an Jiaotong University approved all animal experiments (Institutional Animal Care and Use Committee; Permit No. 2022-1323).
We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
This work was supported by grants from the Natural Science Foundation Project of Shaanxi Province (2024JC-YBMS-649, 22JS031, 2022SF-472 and 2022SF-019).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





