1 The Key Laboratory for Gout Research in Heilongjiang Province, School of Basic Medicine, Jiamusi University, 154000 Jiamusi, Heilongjiang, China
Abstract
Background: Persistent hyperuricemia can lead to the generation and deposition of monosodium urate (MSU) crystals. This can trigger gouty arthritis (GA), which in turn induces inflammation. Activation of the Nod-like receptor pyrin domain containing 3 (NLRP3) inflammasome plays a critical role in the onset and progression of GA. Autophagy may have a dual effect on GA with regard to the NLRP3 inflammasome. Therefore, the present study aimed to gain a deeper comprehension of the interaction between autophagy and NLRP3 inflammasome activation is imperative for developing more efficacious treatments for GA. Methods: Peripheral blood monocytes (PBMCs) were first isolated from GA patients and healthy controls and underwent bulk RNA sequencing analysis. Overexpression and knockdown of dual specificity phosphatase 1 (DUSP1) was performed in THP-1 monocytes to investigate its role in the immune response and mitochondrial damage. The luciferase assay and Western blot analysis were used to study the interaction between autophagy and NLRP3 inflammasome activation. Results: Bulk RNA sequencing analysis showed significant upregulation of DUSP1 expression in PBMCs from GA patients compared to healthy controls. This result was subsequently verified by reverse transcription quantitative polymerase chain reaction (RT-qPCR). DUSP1 expression in human THP-1 monocytes was also shown to increase after MSU treatment. Downregulation of DUSP1 expression increased the secretion of inflammatory cytokines after MSU treatment, whereas the overexpression of DUSP1 decreased the secretion levels. Lipopolysaccharides (LPS) combined with adenosine-triphosphate (ATP) led to mitochondrial damage, which was rescued by overexpressing DUSP1. DUSP1 overexpression further increased the level of autophagy following MSU treatment, whereas downregulation of DUSP1 decreased autophagy. Treatment with the autophagy inhibitor 3-Methyladenine (3-MA) restored inflammatory cytokine secretion levels in the DUSP1 overexpression group. MSU caused pronounced pathological ankle swelling in vivo. However, DUSP1 overexpression significantly mitigated this phenotype, accompanied by significant downregulation of inflammatory cytokine secretion levels in the joint tissues. Conclusions: This study revealed a novel function and mechanism for DUSP1 in promoting autophagy to mitigate the MSU-induced immune response in GA. This finding suggests potential diagnostic biomarkers and anti-inflammatory targets for more effective GA therapy.
Keywords
- gouty arthritis
- monosodium urate
- autophagy
- dual specificity phosphatase 1
Gouty arthritis (GA) is a type of chronic inflammatory disease caused by the generation and deposition of monosodium urate (MSU) crystals in articular and periarticular spaces. This occurs in the context of persistent hyperuricemia and can subsequently activate the innate immune system [1]. The prevalence of GA in adults ranges from about 0.68% to 3.9% worldwide [2]. Current therapeutic approaches for GA consist primarily of reducing hyperuricemia and inflammation. However, these treatments are far from optimal due to various factors, including adverse reactions [1, 3, 4, 5]. Many studies have shown that activation of the Nod-like receptor family, pyrin domain containing 3 (NLRP3) inflammasome plays a crucial role in the initiation and progression of GA [6, 7, 8, 9, 10]. Elucidating the functions and mechanisms of the NLRP3 inflammasome in the pathogenesis of GA may therefore have important implications for the development of therapeutic drugs against GA.
Various pathogen-associated molecular patterns (PAMPs), or damage-associated
molecular patterns (DAMPs), have been identified as priming signals that
effectively promote the transcriptional activation of NLRP3, pro-IL-1
Autophagy is a programmed digestion process that is involved in cell development, metabolism, immune regulation and aging. It does this via the sequestration of damaged proteins and organelles, as well as the destruction of intracellular pathogens, within a double-membrane vesicle-autophagosome, and then fusing with lysosomes to cause degradation [16, 17, 18, 19]. Autophagy has been demonstrated to play a dual role in some inflammatory diseases associated with the NLRP3 inflammasome, including GA [18, 20, 21, 22, 23]. Autophagy can reduce NLRP3 inflammasome activation and the inflammatory response by removing activators such as damaged mitochondria that produce reactive oxygen species (ROS), as well as inflammasome components and cytokines [24, 25, 26, 27, 28]. Activation of the NLRP3 inflammasome regulates autophagy through several different pathways to promote the occurrence and development of GA [18, 21, 29, 30, 31]. Therefore, a better understanding of the inter-regulatory mechanisms between autophagy and activation of the NLRP3 inflammasome is required to develop more efficient treatments for GA.
Dual specificity phosphatase 1/mitogen-activated protein kinase phosphatase-1 (DUSP1/MKP-1) is a dual-specific phosphatase involved in the regulation of MAPK family isoforms. It plays an important anti-inflammatory role in acute and chronic diseases such as asthma, diabetes and cancers [32, 33, 34, 35]. A recent study showed that upregulation of DUSP1 can attenuate renal tubular mitochondrial dysfunction by promoting autophagy and restoring parkin-mediated mitophagy in diabetic nephropathy [36]. Fan et al. [37] demonstrated that RGD1564534 protected neurons in cerebral ischemia/reperfusion injury by competitively binding with miR-101a-3p to upregulate DUSP1, thereby promoting mitochondrial autophagy and blocking NLRP3 inflammasome activity. However, the expression and function of DUSP1 in GA is still unknown.
In the present study, peripheral blood monocytes (PBMCs) were isolated from GA patients and healthy controls and used for bulk RNA sequencing analysis. DUSP1 was found to be significantly upregulated in the GA group. Furthermore, DUSP1 was significantly upregulated in human THP-1 monocytes after MSU treatment. Downregulation of DUSP1 in THP-1 cells increased the secretion of inflammatory cytokines after MSU treatment, whereas DUSP1 overexpression reduced cytokine secretion. DUSP1 overexpression was also found to enhance autophagy, thereby reducing mitochondrial damage and efficiently rescuing MSU-induced immune response, both in vitro and in vivo. Taken together, this research has revealed a novel mechanism involving DUSP1 in GA diseases, opening the possibility of novel diagnostic biomarkers and anti-inflammation targets for GA patients.
Gout patients and healthy controls from the First Affiliated Hospital of Jiamusi University were enrolled in the study after signing an informed consent form. This study was approved by the Ethics Committee of Jiamusi University. Peripheral blood samples were collected from GA patients and healthy controls and the PMBCs were isolated and harvested for subsequent experiments according to the protocol described in the kit (11350D, ThermoFisher, Carlsbad, CA, USA).
The human THP-1 cell line, which were
purchased from the Cell Bank of Type Culture Collection of the Chinese Academy of
Sciences, Shanghai Institute of Cell Biology (Shanghai, China) was grown in
RPMI-1640 media containing 10% FBS, 0.05 mM
THP-1 cells were incubated with 1 µg/mL Lipopolysaccharide (LPS) + 20 µM adenosine triphosphate (ATP) or vehicle for 4 h following DUSP1 over-expression. To measure mitochondrial ROS, inner transmembrane potential, and mitochondrial mass, the cells were stained with 5 µM MitoSOX and 25 nM MitoTracker Deep Red + 25 nM MitoTracker Green for 15 min. The signals were then detected by flow cytometry (FACS). The degree of mitochondrial membrane polarization in THP-1 cells was measured by staining with 2 µM JC-1, followed by signal detection with confocal microscopy.
Male C57BL/6 mice (6-weeks old) were purchased from SPF (Beijing, China)
Biotechnology Co., Ltd. The mice were maintained in accordance with institutional
policies, and the study was approved by the Ethics Committee of the First
Affiliated Hospital, Jiamusi University (2022-500-177). The mice were
anesthetized with 50 mg/kg sodium pentobarbital via intraperitoneal injection
(i.p.) and then injected at the hind ankle joint with phosphate-buffered saline (PBS) (20 µL) or MSU (1
mg MSU dissolved in 20 µL PBS) after 1 week of lv-NC or MSU + lv-DUSP1 (1.2
THP-1 cells were infected with the tandem GFP-LC3 reporter adenovirus (Beyotime, Shanghai, China, C3006) according to the manufacturer’s instructions. To investigate the function of DUSP1 in regulating autophagy, vectors for DUSP1 overexpression, DUSP1 knockdown (short hairpin RNA, shRNA), or controls were added to the cell culture medium for 24 h. The THP-1 cells were then harvested and the cell nuclei stained with Hoechst 33342 (Beyotime, C1029) for 10 min to detect signals by confocal microscopy.
Total RNA from samples was extracted with Trizol reagent (Thermo Fisher
Scientific, 15596026, Waltham, MA, USA) and phenol-chloroform precipitation. After undergoing
removal of genomic DNA and reverse transcription, qRT-PCR was performed using the
ABI PRISM 7900 system (Applied Biosystems, Carlsbad, CA, USA) with the SYBR Green
Real time PCR Master Mix plus (TOYOBO, Osaka, Japan). GAPDH was used for internal
normalization. The primers used for RT-qPCR were: human GAPDH (Forward:
5
The RNA quality and purity of samples were assessed and only those showing RNA
Integrity Number
Following the different treatments, total protein was extracted from THP-1 cells
using Radioimmunoprecipitation (RIPA) lysis buffer (Beyotime) on ice. Protein concentrations were determined
using a bicinchoninic acid (BCA) kit (Beyotime). A total of 20 µg protein was separated by
electrophoresis and then transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were
blocked with 5% skimmed milk and incubated overnight at 4 °C with
primary antibodies (Abconal, Wuhan, China) against_DUSP1 (1:1000
dilution), IL-6 (1:1000), TNF-
Following different treatments for 24 h, the culture medium from THP-1 cells was collected and the level of inflammatory cytokines was assessed using ELISA kits as recommended by the manufacturer (Sangon Biotech, Shanghai, China).
Statistical analysis was performed using GraphPad Prism software version 9.0
(GraphPad Software, San Diego, CA, USA). Measurement data was presented as the mean
Monocytes and macrophages have been shown to play a vital role in MSU-driven,
pro-inflammatory cytokine production, thereby initiating an acute inflammatory
response [30, 38, 39]. PBMCs were isolated from GA patients and healthy controls
and analyzed by bulk RNA sequencing to screen for differential gene expression
between the two groups. Altered genes reaching a
significance level of p
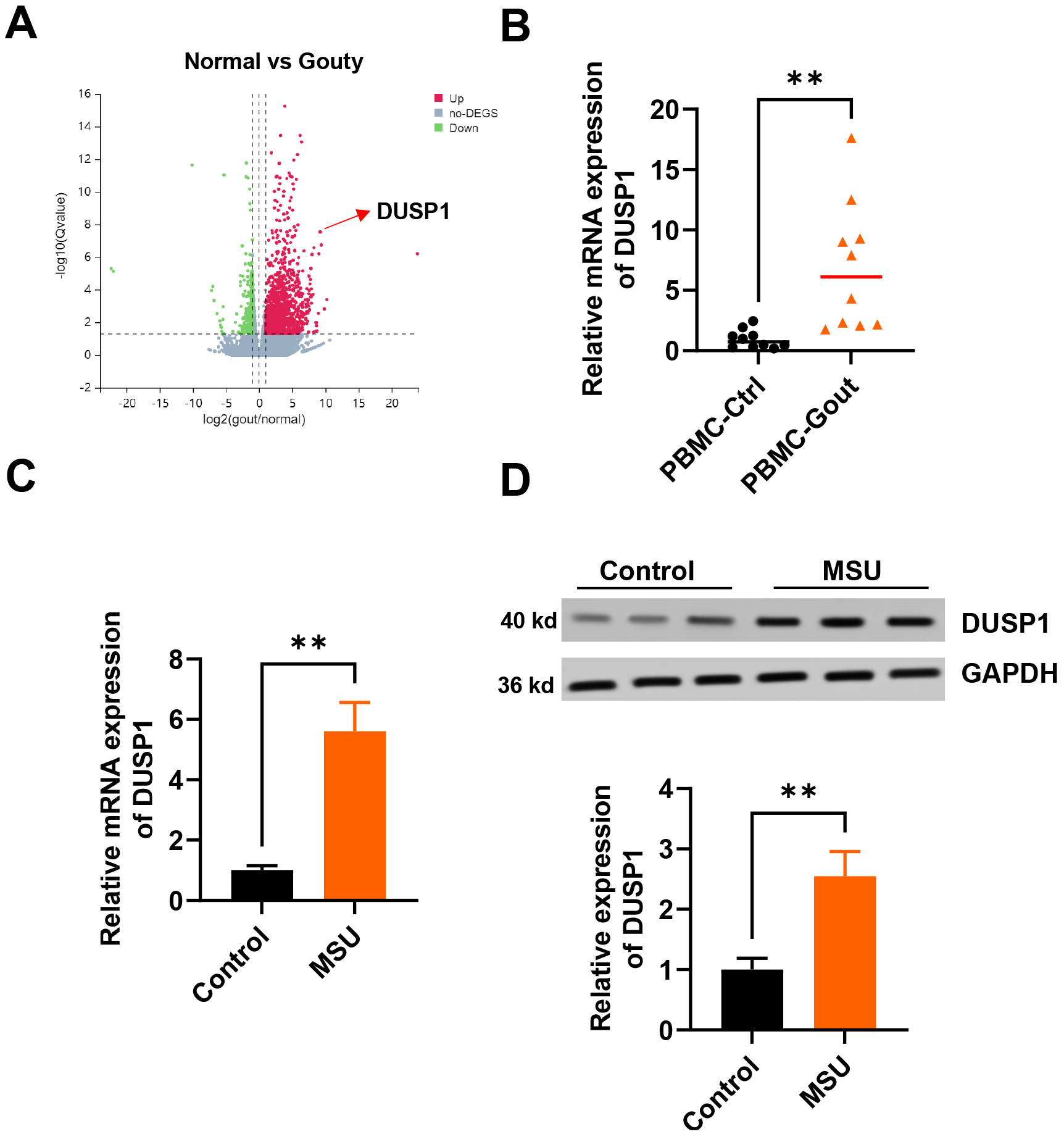 Fig. 1.
Fig. 1.DUSP1 expression is upregulated in peripheral blood monocytes
(PBMCs) from gouty arthritis (GA) patients, and in monosodium urate (MSU)-treated
THP-1 monocytes. (A) Volcano plot of differential mRNA expression in PBMCs from
GA patients and healthy controls. (B) Reverse transcription quantitative
polymerase chain reaction (RT-qPCR) analysis of dual specificity phosphatase 1
(DUSP1) mRNA expression in PBMCs from GA patients and healthy controls (n = 10).
(C) RT-qPCR analysis of DUSP1 mRNA expression in MSU-treated THP-1 and in THP-1
controls (n = 3). (D) Representative Western blot analysis of DUSP1 protein
expression in MSU-treated THP-1 and in THP-1 controls (n = 3). **p
The NLRP3 inflammasome requires initiation and activation signals before it can
trigger IL-1
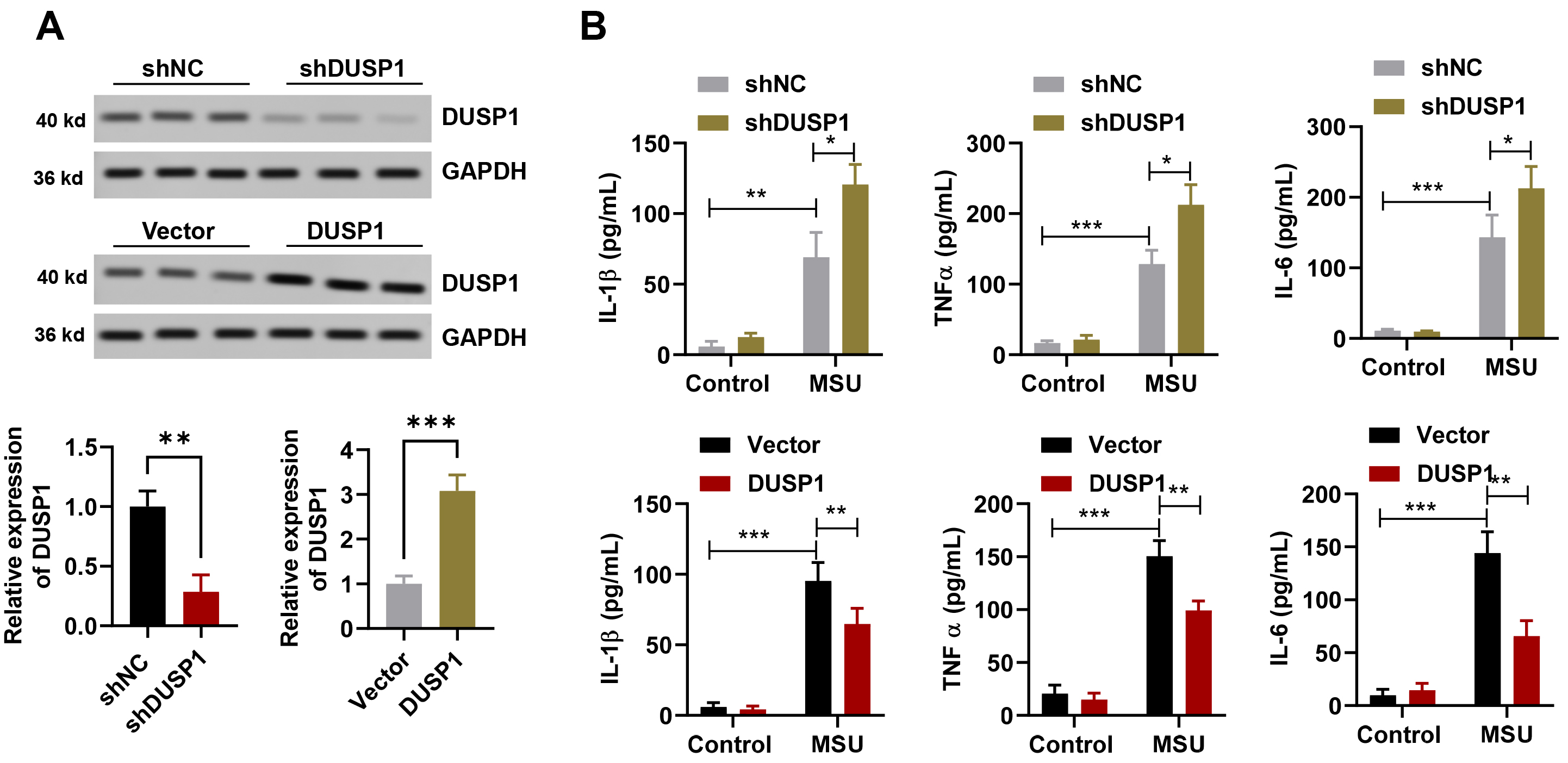 Fig. 2.
Fig. 2.DUSP1 inhibits the inflammatory response in MSU-treated THP-1
monocytes. (A) Representative Western blot analysis of the DUSP1 protein level
in THP-1 monocytes following infection with shRNA (downregulation), or with
adeno-associated virus (overexpression) (n = 3). (B) Enzyme-linked immunosorbent
assay (ELISA) analysis of inflammatory cytokine levels in THP-1 with DUSP1
overexpression (upper panel) or knockdown (lower panel) in the MSU treatment or
control groups (n = 3). *p
Mitochondrial damage is an essential trigger for inflammasome formation and is increased following MSU treatment [9, 31, 45]. To evaluate the level of cellular oxidative stress/ROS, LPS combined with ATP was added to the medium of THP-1 monocytes. The number of positive cells for mitochondrial superoxide increased, but overexpression of DUSP1 caused this ratio to decrease (Fig. 3A). In addition, the overexpression of DUSP1 rescued LPS + ATP caused membrane potential of mitochondria down-regulation (Fig. 3B,C). Therefore, DUSP1 appears to be an inhibitor of NLRP3 inflammasome activation after MSU treatment in vitro.
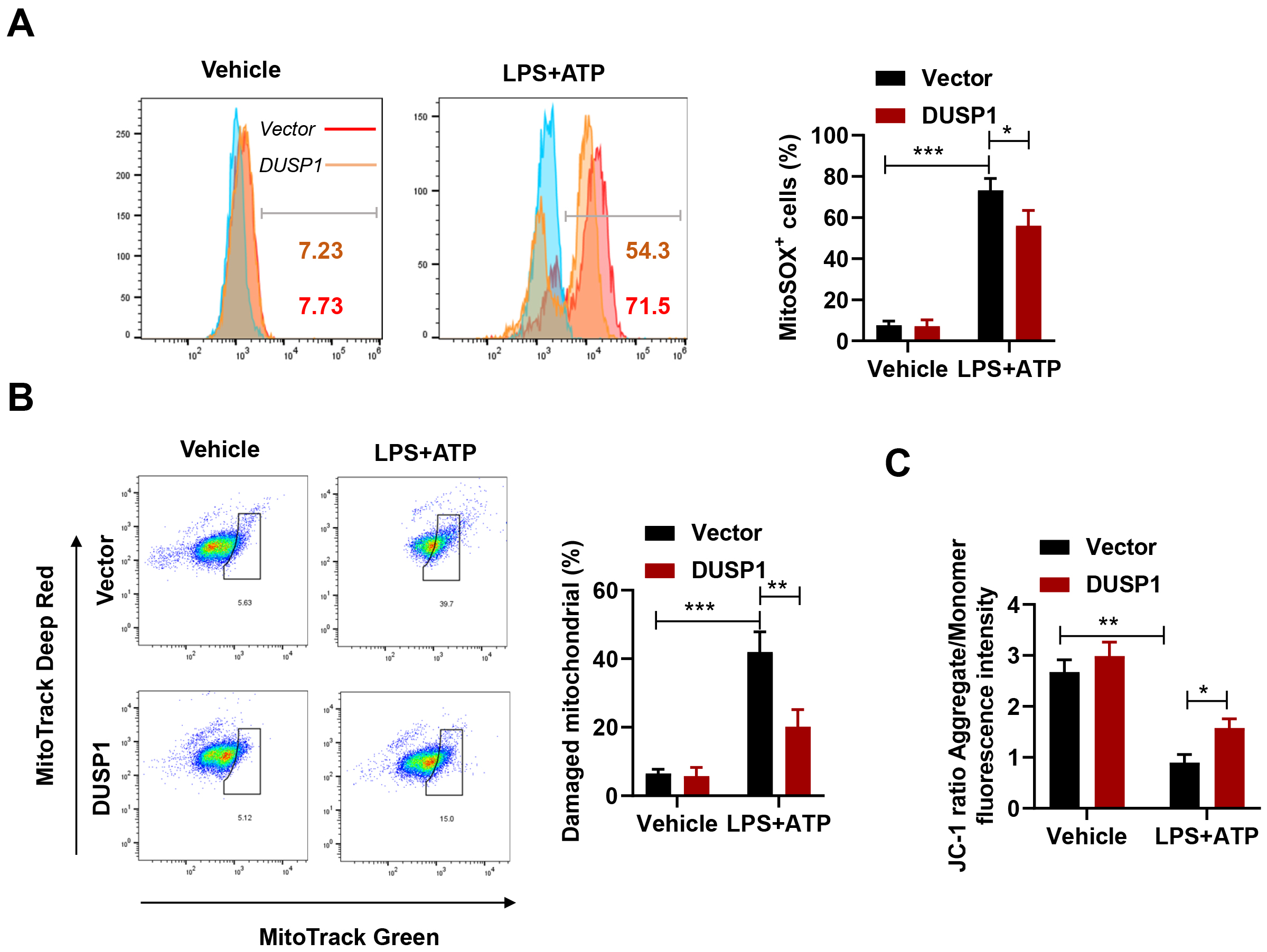 Fig. 3.
Fig. 3.DUSP1 decreases mitochondrial damage in Lipopolysaccharide (LPS)
+ adenosine triphosphate (ATP)-treated THP-1 monocytes. (A) Flow cytometry
(FACS) analysis of mitoSOX positive cells in THP-1 with DUSP1 overexpression and
in THP-1 controls after LPS + ATP treatment (n = 3). (B) FACS analysis of damaged
mitochondria in THP-1 with DUSP1 overexpression and in THP-1 controls after LPS +
ATP treatment (n = 3). (C) Analysis of the JC-1 ratio in THP-1 with DUSP1
overexpression and in THP-1 controls after LPS + ATP treatment (n = 3).
*p
The mechanisms involved in regulating NLRP3 inflammasome activation have only been partially elucidated. The manipulation of autophagy via suppression of the NLRP3 inflammasome may be a therapeutic approach for GA [18, 25, 46, 47]. The autophagy level in THP-1 monocytes was evaluated here by estimating the density of LC3 puntca after MSU treatment. MSU was found to activate autophagy, but more importantly, DUSP1 overexpression further increased the level of autophagy (Fig. 4A). Moreover, Western blot analysis showed that LC3II protein after MSU treatment was increased in the presence of DUSP1 overexpression, whereas P62 protein was decreased (Fig. 4B). In contrast, downregulation of DUSP1 markedly suppressed the level of autophagy, decreased the expression of LCII, and increased the expression of p62 (Fig. 4C,D).
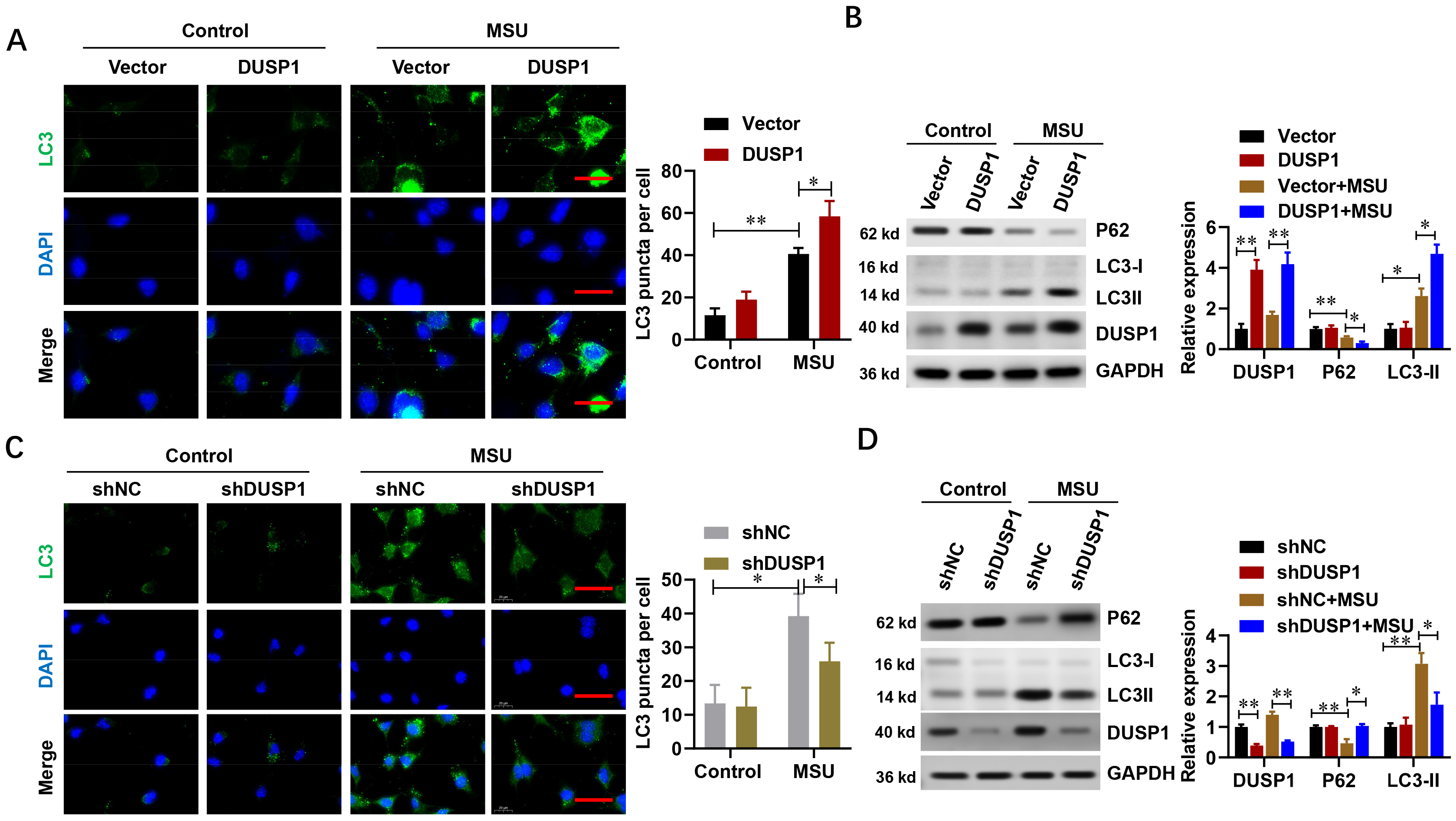 Fig. 4.
Fig. 4.DUSP1 promotes autophagy in THP-1 monocytes following MSU
treatment. (A) Representative immunofluorescence (IF) staining and analysis of
autophagy in THP-1 cells with DUSP1 overexpression and in THP-1 controls after
MSU treatment (n = 3). Scale bar, 200 µm. (B) Representative Western blot
results for autophagy-related proteins in THP-1 cells with DUSP1 overexpression
and in THP-1 controls after MSU treatment (n = 3). (C) Representative IF staining
and analysis of autophagy in THP-1 cells with DUSP1 knockdown and in THP-1
controls after MSU treatment (n = 3). Scale bar, 200 µm. (D) Representative
Western blot analysis of autophagy-related proteins in THP-1 cells with DUSP1
knockdown and in THP-1 controls after MSU treatment (n = 3). *p
Next, we used a well-established drug for autophagy inhibition, 3-Methyladenine
(3-MA), to investigate whether DUSP1-mediated inhibition of
MSU-driven inflammation was via the promotion
of autophagy. 3-MA was found to efficiently abolish the promotion of autophagy by
DUSP1 overexpression (Fig. 5A). Furthermore, the level of inflammatory
cytokines
(IL-1
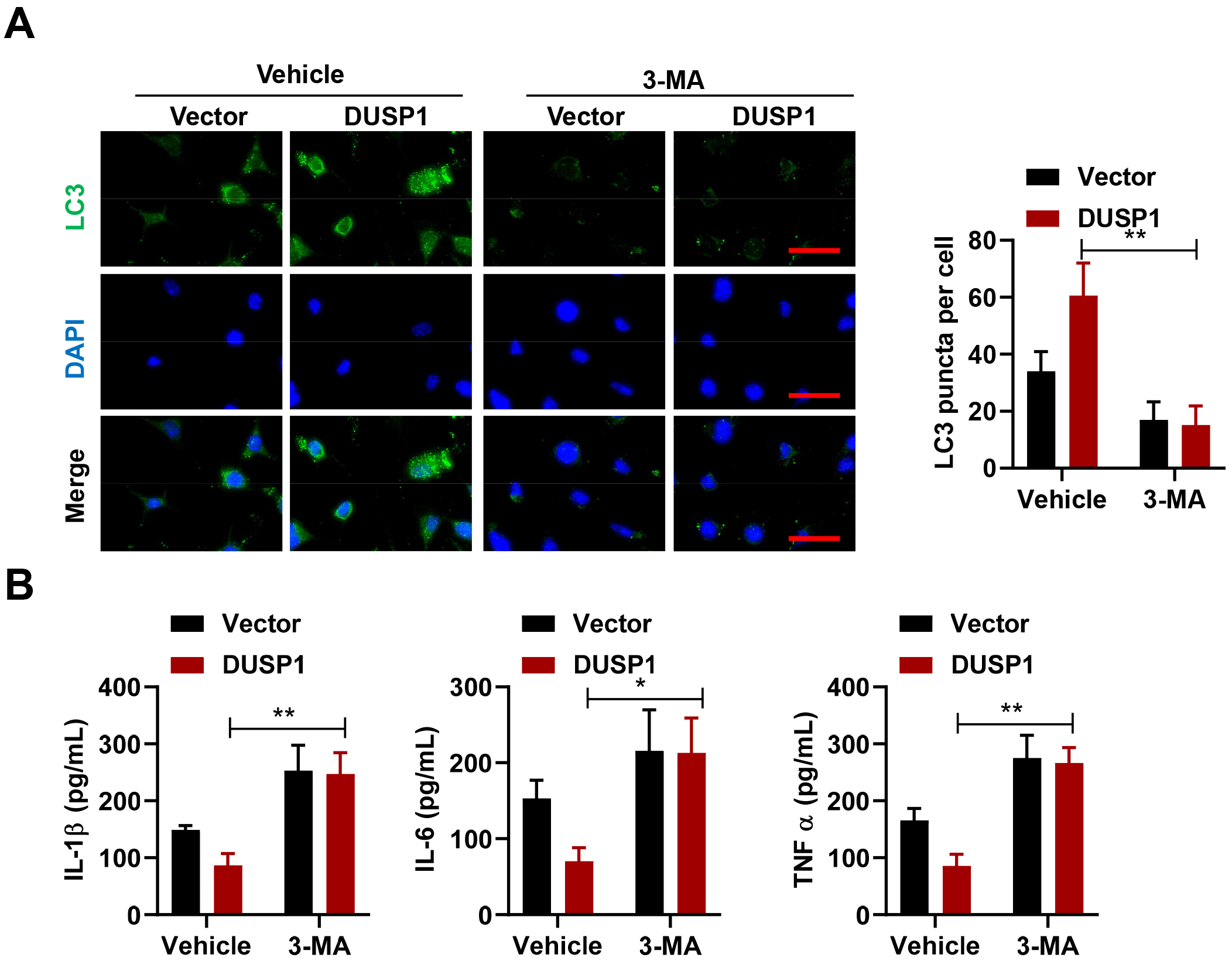 Fig. 5.
Fig. 5.DUSP1 inhibits MSU-driven inflammation by promoting autophagy.
(A) Representative IF staining and analysis of autophagy in DUSP1-overexpressing
THP-1 and in control THP-1 after 3-Methyladenine (3-MA) treatment (n = 3). Scale
bar, 200 µm. (B) ELISA analysis of inflammatory cytokine levels in
DUSP1-overexpressing THP-1 and in control THP-1 after 3-MA treatment (n = 3).
*p
To assess whether DUSP1 overexpression alleviates GA, lv-DUSP1 or lv-NC were
injected into the ankle joint of C57BL/6 mice, with subsequent injection of MSU
(Fig. 6A). MSU induced ankle swelling with distinct pathological morphology, and
also significantly increased foot thickness (Fig. 6B). These changes were
significantly reduced in the lv-DUSP1 group compared to the lv-NC group. MSU also
activated an inflammatory response, as reported previously [42, 48, 49].
However, lv-DUSP1 treatment significantly downregulated the protein levels for
IL-1
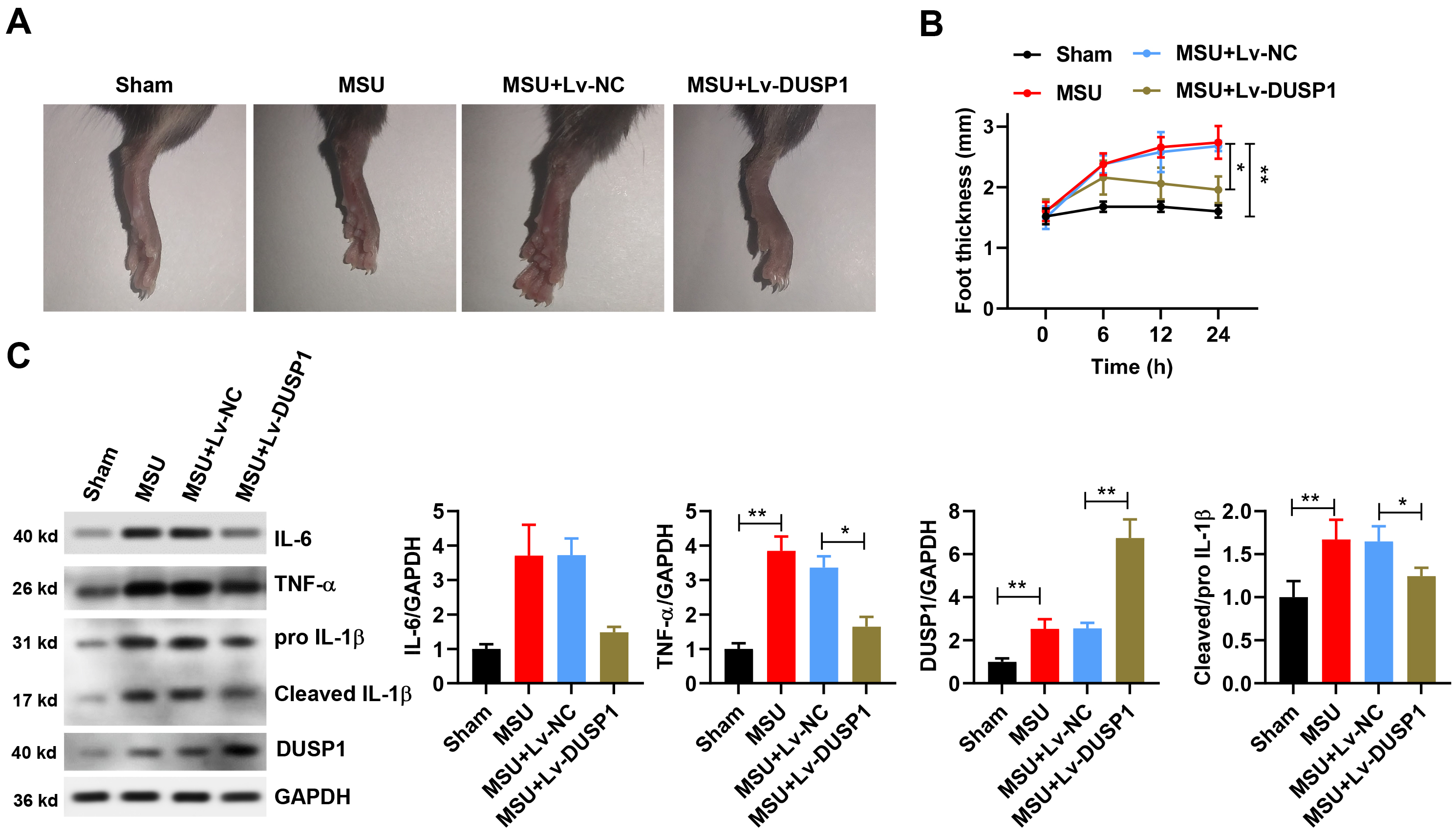 Fig. 6.
Fig. 6.DUSP1 suppresses GA in vivo by downregulating
MSU-induced inflammation. (A) Representative macroscopic views of ankle swelling
in different treatment groups. (B) Foot thickness in mice overexpressing DUSP1
and in controls after MSU treatment (n = 6). (C) Western blot analysis and levels
of inflammatory cytokines in mice overexpressing DUSP1 and in controls after MSU
treatment (n = 3). *p
This study identified a novel role for DUSP1 as a suppressor of NLRP3 inflammasome activation via the enhancement of autophagy in MSU-induced GA. DUSP1 expression was significantly elevated in PBMCs from GA patients compared to healthy controls. Overexpression of DUSP1 also reduced the inflammation response and mitochondrial damage in THP-1 monocytes after MSU treatment. Furthermore, DUSP1 knockdown worsened the MSU-induced disease phenotype. DUSP1 appears to upregulate the level of autophagy, thereby suppressing NLRP3 inflammasome activation both in vitro and in vivo. These findings help to clarify the function and mechanism of DUSP1 in suppressing NLRP3 inflammasome activation during GA. Moreover, they suggest that downstream targets of DUSP1 may be useful in the development of new drugs for GA therapy.
Bulk RNA sequencing data from this study revealed considerable differential gene
expression between PBMCs from GA patients compared with healthy controls.
Importantly, the observed upregulation of DUSP1 expression in PBMCs from GA
patients was also observed in THP-1 monocytes following MSU treatment. MSU is
commonly used to mimic the human GA phenotype in cells and mice. It does this by
activating the NLRP3 inflammasome, thereby promoting the maturation of
interleukins and inducing immune cell migration, leading to an immune response
[42, 48, 49]. In the present study, increased levels of secreted inflammatory
cytokines (IL-1
A comprehensive mechanism for NLRP3 inflammasome-mediated GA disease is yet to be fully elucidated, but the dual role of autophagy in this process is intriguing [18, 25, 46, 47]. MSU was shown to induce autophagy by increasing the levels of LC3-II and the presence of LC3 puncta, thereby activating the NLRP3 inflammasome [38, 50, 51]. This effect could be attenuated in different cell types by the autophagy inhibitor 3-MA. In contrast, the upregulation of autophagy by AMPK activators and SIRT1 agonist was reported to prevent the accumulation of p62 by MSU crystals and to inhibit NLRP3 inflammasome activation [38, 46, 50]. The present study also found that MSU triggers autophagy after increasing the extent of mitochondrial damage. In addition, DUSP1 overexpression further enhanced the level of autophagy, thereby reducing mitochondrial damage and the MSU-induced immune response in vitro and in vivo. Therefore, the benefit of DUSP1 upregulation in GA is derived primarily by increasing the level of autophagy in monocytes. The origin of high DUSP1 expression observed in PBMCs from GA patients requires further study, since PBMCs are comprised of different cell populations characterized by anti- or pro-inflammatory activities [39]. Whether there are potential factors that antagonize DUSP1 to suppress the immune response in GA also needs to be investigated. Furthermore, in order to better understand the mechanism by which DUSP1 regulates autophagy, the downstream signaling pathway needs to be clarified using proteomic analysis.
In conclusion, the results of this study revealed that DUSP1 expression was significantly increased in the PBMCs of GA patients. Moreover, DUSP1 overexpression could mitigate the MSU-induced immune response by enhancing the level of autophagy. This finding suggests that DUSP1 may be an attractive candidate target for human GA therapy.
DUSP1, dual specificity phosphatase 1; GA, gouty arthritis; MSU, monosodium urate.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Conceptualization and investigation: JN and HQ; methodology and resources: JN; writing and Original Draft Preparation: JN and HQ; Critical review and Editing: JN and HQ. Both authors contributed to editorial changes in the manuscript. Both authors have read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Informed consent was obtained from all participants included in the study. The study was approved by the Ethics Committee of the First Affiliated Hospital, Jiamusi University (QKW2021015). All procedures related to the care and sacrifice of animals were reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Jiamusi University (2022-500-177) and performed in accordance with international regulations.
Not applicable.
This work was supported by grants from the key program of the Natural Science Foundation, Heilongjiang Province of China (ZD2022H006) and Jiamusi University youth innovative talent training program (JMSUQP2020015).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






