1 Department of Experimental and Clinical Medicine, University of Florence, 50134 Florence, Italy
2 Department of Health Sciences, University of Florence, 50139 Florence, Italy
3 Department of Neuroscience, Psychology, Drug Research and Child Health NEUROFARBA, University of Florence, 50139 Florence, Italy
4 Careggi University Hospital, Clinical Toxicology and Poison Control Centre, 50134 Florence, Italy
5 Network of Immunity in Infection, Malignancy and Autoimmunity (NIIMA), Universal Scientific Education and Research Network (USERN), 50134 Florence, Italy
†These authors contributed equally.
Keywords
- cocaine
- gut microbiota
- substance use disorder
- cocaine use disorder
- gut-brain axis
Microbial dysbiosis is acknowledged as a contributing factor to cognitive and neuropsychiatric disorders, including substance use disorders (SUD) [1]. Specifically, there’s evidence linking SUD, particularly alcohol use disorder (AUD), to heightened permeability of the intestinal barrier (known as “leaky gut”) and disrupted gut-brain communication [2]. In this context, the effects of cocaine on gut microbiota (GM) have been scarcely investigated. However, the interplay between cocaine and microbial community alterations is arousing interest, leading to an increased number of preclinical studies on GM in both animal models and humans with cocaine addiction [3]. Cocaine is a psychoactive and addictive drug that affects GM and undermines nutritional status in addition to disrupting immunological and gastrointestinal systems and dysregulating neurotransmitter systems, particularly dopamine. Cocaine use disorder (CUD) represents a significant public health problem worldwide but no FDA-approved medications are currently available and all the proposed pharmacological therapies resulted in elusive and modest long-term success rates [4]. GM modulation may represent an innovative and promising diagnostic and therapeutic tool, aiding in the future advancement of supplementary strategies for managing complications linked to cocaine abuse.
The human gastrointestinal tract harbors trillions of microorganisms and the proper functioning of the GM is crucial for human health; in fact, its alteration (i.e., intestinal dysbiosis) is associated with various local and systemic diseases. Specifically, recent findings have revealed that individuals with substance addiction exhibit altered GM composition and diversity, which affect stress response, depression, immune modulation, and the synthesis and metabolism of neurotransmitters, particularly serotonin and dopamine. In this scenario, since many drugs of abuse are well known to alter gastrointestinal functionality through the alteration of GM structure [5], the relationship between GM dysbiosis and SUD has been widely documented in both rodents and humans. For instance, increased permeability of the intestinal barrier and altered gut-brain communication are noted in alcohol use disorder (AUD). In particular, several studies have documented that prolonged alcohol intake disturbs GM balance in rodent models of alcohol seeking, chronic consumption and withdrawal. Interestingly, the transfer of GM from AUD patients to germ-free animals induces learning and memory dysfunctions, depression and anxious behaviours, indicating the pivotal role of GM in AUD development [6]. Moreover, alterations in GM related to opiate dependence have been observed in both human and animal models, often manifesting as opioid-induced constipation, a prevalent adverse effect in chronic pain patients undergoing opioid therapy. Among drugs of abuse, also nicotine, amphetamine, and cannabinoids have been reported to cause significant GM dysbiosis. To date, despite cocaine being one of the most widespread recreational drugs worldwide, studies on the gut-brain axis and cocaine addiction are limited, primarily relying on animal models.
In the broad scope of research on the gut-brain axis and cocaine addiction, Kiraly and colleagues [7] reported that mice subjected torepeated cocaine administration showed enhanced sensitivity to cocaine reward, increased locomotor sensitization and modifications in the expression of synaptic proteins in the brain’s reward circuitry, along with a reduction in intestinal bacterial diversity. Other reports highlighted the critical association between CUD and gut-barrier alterations and significant reductions in alpha diversity in rats subjected to repeated exposure to volatilized cocaine [8, 9]. Importantly, oral administration of Lactobacillus rhamnosus was found to reduce cocaine-induced gut oxidative stress, inflammation, glial activation, and locomotion activity, suggesting a role for certain microbial species in modulating cocaine outcomes [10]. Moreover, Cuesta and colleagues [11] recently found that cocaine-exposed mice exhibited increased norepinephrine levels, promoting gut colonization by Gammaproteobacteria, including Escherichia coli. Interestingly, this over-representation was associated with heightened addiction-like behaviour, such as increased locomotion and exploration [11]. Finally, García-Cabrerizo and colleagues [12] documented that the GM depletion via antibiotiotic treatment reduced cocaine place preference in mice, along with improvements of social stimuli. Overall, these findings confirm that gut microbes may modulate sociability and its neurobiological underpinnings, suggesting the targeting of GM could be essential for programming social behaviors.
Although several studies have investigated cocaine-related modifications in the GM composition in animal models, research on cocaine effects on microbial communities in humans is limited.
For instance, Volpe and colleagues [13] assessed the GM structure in CUD patients with HIV infection, identifying differences in GM phyla (i.e., Bacteroidetes, Firmicutes, Proteobacteria, and Actinobacteria), as well as in markers of inflammation and microbial translocation. Moreover, Fu and colleagues [14], although not evaluating the intestinal microbiota structure, noted decreased saliva microbial diversity in CUD patients compared to healthy subjects. Interestingly, this study revealed that chronic cocaine use-associated inflammation in the blood may result from increased oral Streptococcus spp. and its effects on myeloid cell activation, but does not result from cocaine directly [14]. Finally, a recent study conducted on fifty-eight CUD patients has documented the presence of a profound compositional and functional dysbiosis of both oral and intestinal microbiota. Specifically, in comparison to healthy people, cocaine-addicted patients reported a significant decrease in alpha diversity, modification of the abundances of several taxa in both intestinal and oral microbiota, dysregulation of many predicted metabolic pathways, as well as reduced levels of fecal butyric acid (a microbial-derived metabolite with anti-inflammatory effects) and increased abundances of pro-inflammatory medium chain fatty acids [15]. Interestingly, repetitive transcranial magnetic stimulation (rTMS) which has positive effects on the reduction of both cocaine intake and craving in CUD patients, determined a beneficial reduction of periodontal disease-associated bacteria and a significant increase in butyric acid [15]. Altogether, these findings underscore the critical role of microbes in the pathogenesis of CUD and neuropsychiatric pathologies.
Since various preclinical and clinical evidence have highlighted the role of GM in SUD, therapeutic interventions targeting the GM, such as prebiotics, probiotics, and fecal transplantation, have been recently proposed to counteract gut disorders related to drug consumption and restore healthy gut-brain communication. More precisely, the interplay between these two organs mainly occurs through the diffusion of microbial-derived metabolites (especially short chain fatty acids) and neuromodulators (e.g., dopamine, serotonin, γ-aminobutyric acid), which can directly influence the central nervous system via the vagus nerve or by passing the blood-brain barrier [16]. In general, research in this area is still in its early stages, particularly concerning CUD patients. However, although no clinical trials have yet examined the effects of pre- or probiotics on drug addiction, a recent review by Leclercq and colleagues [17] evaluating clinical trials on probiotics and prebiotics in alcohol abuse reported promising results for GM restoration. Additionally, targeting depression and anxiety during withdrawal from cocaine or methamphetamine through probiotic supplementation may reduce the risk of relapse.
Another promising strategy for GM modulation is fecal microbiota transplantation (FMT), which consists in the transfer of whole GM communities from healthy donors to diseased receivers. Studies in mice have shown that FMT from patients with AUD to healthy individuals alters their social and anxiety behaviors, while FMT from healthy donors reduces anxiety and depressive behaviors in mice exposed to alcohol [18, 19]. Also, in a recent randomized controlled trial conducted by Bajaj and colleagues [20] on overdrinking patients with AUD-related cirrhosis, those who underwent FMT reported reduced alcohol craving and consumption, accompanied by beneficial microbial changes. In general, additional research is needed to validate the effects of GM modulation on SUDs and to understand whether dysbiosis contributes to the chronic nature of addiction (Fig. 1).
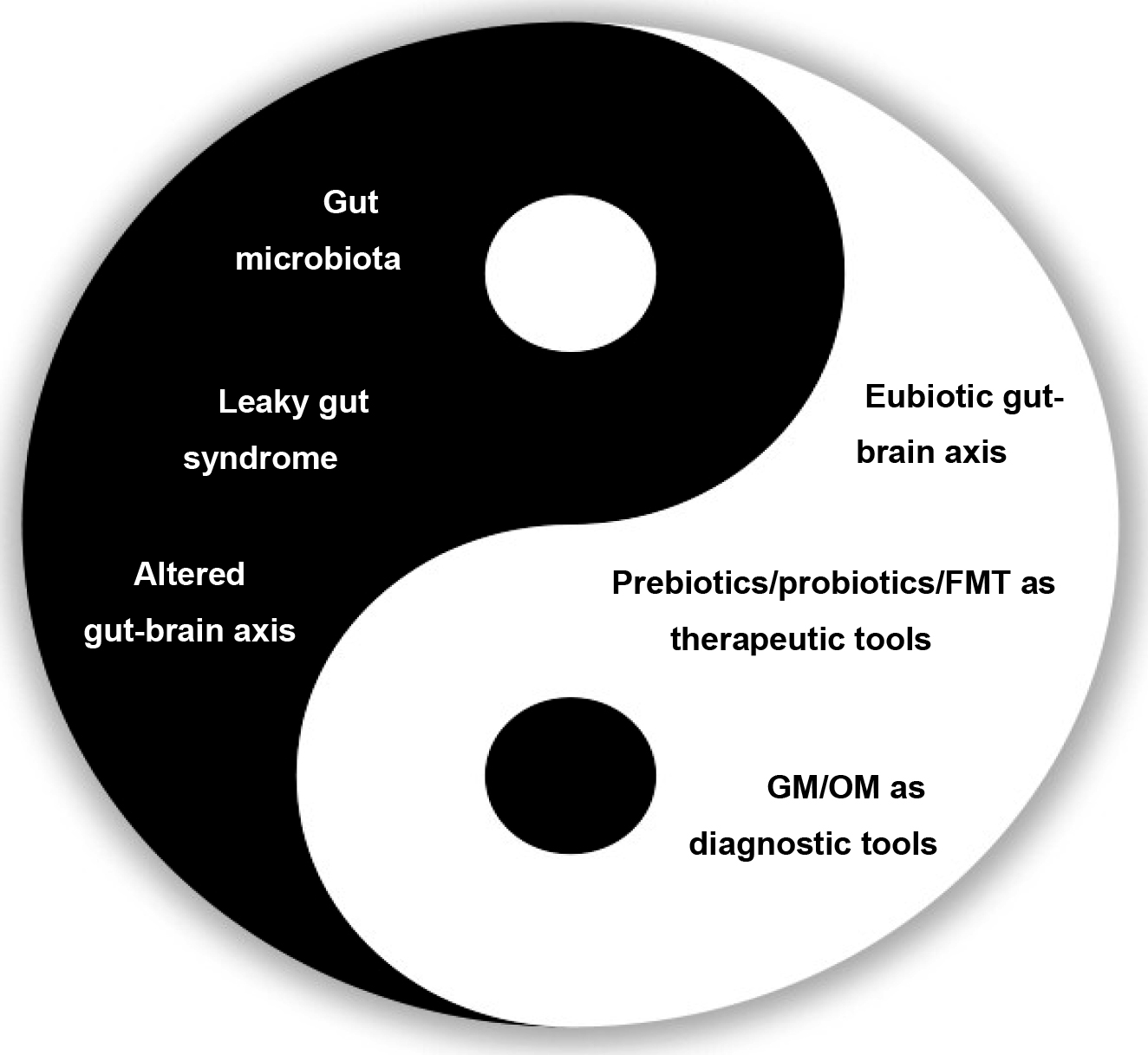 Fig. 1.
Fig. 1.The “yin and yang” of gut microbiota in patients with cocaine use disorder (CUD). FMT, fecal microbiota transplantation; GM, gut microbiota; OM, oral microbiota.
Psychostimulants abuse, particularly cocaine, has a significant global impact, profoundly affecting the microbial, metabolic and immunological profiles of the host that, in turn, may alter cocaine-taking behaviour. Hence, exploring the microbiome holds great potential to uncover novel drug targets and therapeutics associated with the GM and cocaine-related pathways. To date, studies assessing the effects of cocaine use on microbiota–gut–brain interplay are scarce and mainly conducted on animal models; therefore, more research is needed to refine existing strategies used to modulate the GM and restore intestinal eubiosis. Additionally, longitudinal studies investigating the connection between the microbiota and SUD are lacking, leaving the temporal effects of drug use on the GM unclear.
In conclusion, as the the principle of yin and yang from Chinese philosophy describing an opposite but interconnected, self-perpetuating cycle, a better understanding of the relationship between cocaine and GM may provide new mechanistic insight influencing behavioural changes, that could be helpful to develop innovative non-pharmacological therapeutic approaches novel treatments for CUD and mitigate cocaine-induced adverse outcomes.
AUD, Alcohol use disorder; CNS, Central nervous system; CUD, Cocaine use disorder; GM, Gut microbiota; OM, Oral microbiota; rTMS, Repetitive transcranial magnetic stimulation; SUD, substance use disorder.
SB and EG designed the study. SB and EG wrote the first draft of the manuscript. GM and AA supervised the study and critically revised the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by the MNESYS project (PE0000006) – A Multiscale integrated approach to the study of the nervous system in health and disease (DN. 1553 11.10.2022), supported by #NEXTGENERATIONEU (NGEU) and funded by the Ministry of University and Research (MUR), National Recovery and Resilience Plan (NRRP).
The authors declare no conflict of interest. Given his role as Guest Editor and Editorial Board member, Amedeo Amedei had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Graham Pawelec.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

