1 Department of Obstetrics and Gynecology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, 430022 Wuhan, Hubei, China
Abstract
Polycystic ovary syndrome (PCOS) is a prevalent reproductive, endocrine, and metabolic disease that affects 5–18% of women worldwide, with a rising incidence. Hyperandrogenemia and insulin resistance are two key pathophysiological factors that contribute to PCOS, both of which contribute to a variety of health issues such as menstrual irregularities, obesity, dysfunctional glucose and lipid homeostasis, infertility, mental disorders, and cardiovascular and cerebrovascular diseases. Despite ongoing studies, the origin and pathogenesis of PCOS remain elusive; there is also a clinical need for simpler, more effective, longer lasting, and more comprehensive treatments for women with PCOS. The gut–fat axis, a critical regulatory route for metabolism, endocrine function, and immune response, has received considerable interest in recent years in the research of the etiology and treatment of metabolic illnesses such as type 2 diabetes mellitus and non-alcoholic fatty liver disease. The latest research in PCOS has revealed significant alterations in the homogeneity and phylogenetic diversity of the gut microbiota. Animal research using fecal microbiota transplantation has confirmed the importance of gut microbiota in regulating insulin sensitivity and sex hormone balance in PCOS. Furthermore, studies have shown a decrease in the volume and/or activity of brown adipose tissue (BAT) in PCOS patients, a change that alters adipokine release, leading to insulin resistance and hyperandrogenemia, aggravating PCOS progression. Given the function of BAT in increasing energy expenditure and alleviating metabolic parameters, efforts to activate BAT or induce browning of white adipose tissue have emerged as possible treatments for PCOS. Recent research has suggested that the gut microbiota can influence BAT creation and activity via metabolites such as short-chain fatty acids and bile acids, as well as the gut–brain axis. Cold exposure, healthy dieting, metformin, bariatric surgery, glucagon-like peptide 1 receptor agonists and melatonin have all been shown in basic and clinical studies to modulate BAT activity by influencing the gut microbiota, demonstrating significant clinical potential. However, more studies into the regulation mechanisms of the gut–BAT axis are required to produce more effective, comfortable, and safe tailored therapeutics for PCOS.
Keywords
- polycystic ovary syndrome
- gut microbiota
- brown adipose tissue
- insulin resistance
- brown adipokine
Polycystic ovary syndrome (PCOS) is a gynecological disorder that has a significant impact on patients’ reproductive, metabolic, and psychological well-being throughout their lifetime [1, 2]. The prevalence of this illness in women is estimated to be about 5–18%, and it is consistently rising annually [3]. The cause of PCOS is complex is not completely understood to date [4]. Established risk factors for this condition encompass genetic and epigenetic vulnerability, dysfunction in the hypothalamus and ovaries, excessive androgen exposure, insulin resistance, and obesity, among other factors [5]. Recently, there has been increasing attention on the involvement of altered gut microbiota and adipose tissue function in the development of PCOS [6]. The main objective of this study was to offer a comprehensive review of the relationships among gut microbiota, brown adipose tissue (BAT), and their bidirectional interaction in the development of PCOS. It offers fresh insights for foundational research, as well as the diagnosis and treatment of PCOS.
The gut microbiota is an intricate ecosystem situated in the human gastrointestinal (GI) tract, comprising a multitude of microorganisms such as bacteria, fungi, viruses, archaea, and protists. The gut microbiota has a collective weight of approximately 1–2 kg and possesses more than 100 times the number of genes found in the human body it resides [7]. The gut microbiota is a symbiotic community consisting of numerous distinct communities that interact mutually beneficial relationships with the host [8]. The establishment of the gut microbiota commences immediately after birth and is influenced by genetic and environmental variables, particularly the amount and composition of the diet.
In 2012, Tremellen et al. [9] introduced a new model about the development of PCOS: an unhealthy diet can lead to dysbiosis of the gut microbiota, which can result in increased permeability of the GI mucosa. This, in turn, allows lipopolysaccharide (LPS) from Gram-negative bacteria in the GI tract to enter the bloodstream. As a result, the immune system is activated and the function of insulin receptors is disrupted. Elevated levels of insulin and androgens disrupt the growth of ovarian follicles, leading to impaired follicular development [9]. Subsequent studies have consistently verified that the gut microbiota of PCOS patients exhibit a notable decrease in both homogeneity and phylogenetic diversity compared to the control group [10, 11, 12]. Nevertheless, the findings of current studies have been inconsistent regarding the alteration of beta diversity in the gut microbiota of PCOS patients [13]. A meta-analysis of 19 observational human studies, which comprised 617 women with PCOS and 439 healthy individuals, revealed that the alpha diversity indexes (such as the Chao index, Shannon index, and observed operational taxon) were significantly lower in PCOS women compared to the control group. This also indicated that the diversity of gut microbiota in PCOS women had undergone alterations. The imbalance of gut microbiota is hypothesized to be a potential pathogenic component of PCOS [12]. Animal studies have revealed that the gut microbiota of mice with androgen-induced PCOS undergoes changes. Additionally, the transfer of fecal bacteria from PCOS mice to healthy mice can cause disruptions in glucose and lipid metabolism, as well as imbalances in hormone levels [14].
Women diagnosed with PCOS have a reduction in Lachnospira and Prevotella, alongside an enrichment in Bacteroides, Parabacteroides, Lactobacillus, Fusobacterium, and Escherichia/Shigella. These findings suggest an uneven distribution of gut microbiota in individuals with PCOS, characterized by a decrease in bacteria that generate short-chain fatty acids (SCFAs) and metabolize cholic acid. As a result, the gut microbiota has changed to favor bacteria that promote inflammation instead of bacteria that reduce inflammation [13]. Untreated PCOS patients with imbalanced hormone levels have a significantly higher presence of Bacteroides in their gut microbiota, compared to PCOS patients with normal hormone levels. The changes in Bacteroides abundance found in the gut microbiota can be reproduced in the letrozole-induced PCOS animal model. Administering antibiotics to deplete the microbiota can improve the phenotype of PCOS, reduce insulin resistance, and alleviate glucose intolerance [15]. PCOS individuals are more prone to mental disorders, and the gut microbiota of PCOS patients with mental disorders also show noticeable changes. A study found that the composition of gut microbiota in PCOS patients with depression showed unique patterns compared to healthy controls, patients with PCOS, and patients with depression only [16].
There have been limited studies on the potential alterations in gut microbiota between PCOS patients with different phenotypes. Suturina et al. [17] examined the gut microbiota of 83 reproductive-age PCOS patients and 121 healthy controls and discovered no differences in the alpha diversity of gut microbiota among PCOS subgroups with different phenotypes. However, the alpha diversity of gut microbiota in the classical PCOS subgroup showed a significant decrease compared to healthy controls [17]. The alterations in gut microbiota in PCOS patients have been succinctly outlined in several reviews [6, 12, 13]. Nevertheless, it remains crucial to further ascertain the key flora involved in the development of PCOS and the precise mechanism by which it affects patients with PCOS. Ongoing research has shown that the gut microbiota and its metabolites can contribute to the development of insulin resistance and the regulation of glucose and lipid metabolism in various metabolic disorders (e.g., diabetes, non-alcoholic fatty liver disease) through the gut–brain axis [18], gut–liver axis [19], and gut–fat axis [20]. Our current review examined the involvement of the gut–brown fat axis in the development of PCOS (Fig. 1).
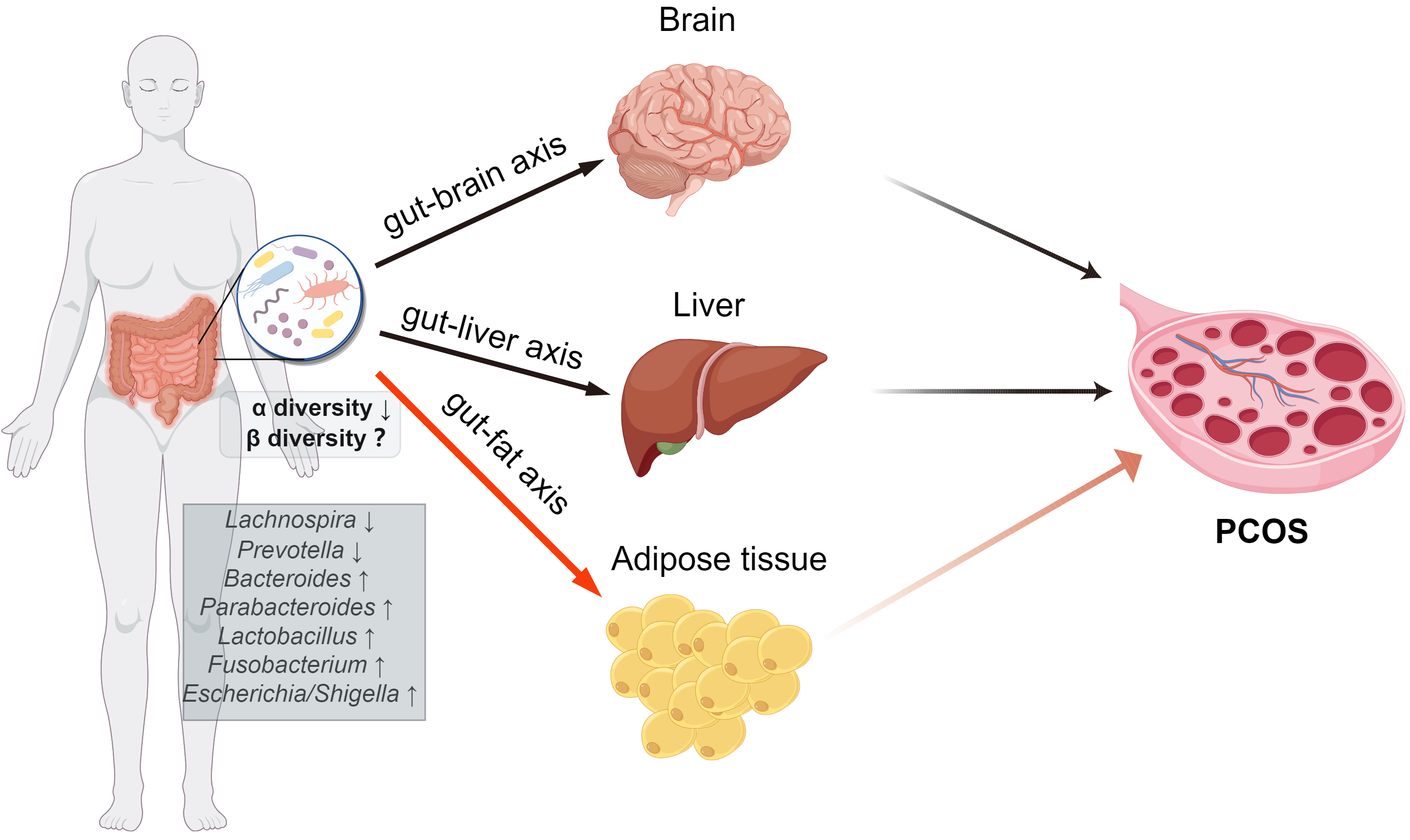 Fig. 1.
Fig. 1.The role of gut bacteria in the development of PCOS. The gut microbiota and its metabolites have the potential to contribute to the onset and progression of PCOS by influencing the gut–brain axis, gut–liver axis, and gut–fat axis. PCOS, Polycystic ovary syndrome. (The Fig. 1 was drawn by Figdraw, ID: ROYRYd448d).
Adipose tissue (AT) is essential for maintaining metabolic health and promoting healthy longevity. Human AT can be classified into two types, white AT (WAT) and brown AT (BAT), both of which have the capacity to secrete unique adipokines. WAT consists of many adipocytes that contain a single lipid droplet and a small number of mitochondria. WAT serves a vital role in storing energy and supplying the essential fuel to fulfill the body’s nutritional needs. By contrast, the lipid droplets present in adipocytes of BAT are small, despite the significant number of mitochondria. BAT can produce heat without shivering through uncoupling protein 1 (UCP-1)-dependent or UCP-1-independent pathways [21]. UCP-1 utilizes glucose and fatty acids as energy sources to convert the electrochemical gradients produced by cellular respiration into heat [22]. Following optimal stimulation, BAT generates 300 times more calories per unit mass than any other organ in the body, constituting about 10% of the overall daily caloric expenditure [23].
Pioneering embryological studies have demonstrated that BAT cells originated
from progenitor cells expressing myogenic factor 5 (MyF5), which were also
present in skeletal muscle. By contrast, WAT cells were formed from progenitor
cells lacking MyF5 expression. On the contrary, subsequent research has
demonstrated that the embryonic origin of adipose cells might be multifaceted and
intricate [24, 25, 26]. Human WAT originates mostly from the mesoderm and undergoes
development in the second trimester. By the time of birth, both visceral and
subcutaneous reservoirs of WAT are formed. BAT emerges throughout the second and
third trimesters of development, serving as a protective mechanism against cold
temperatures for newborns until they acquire the capacity to shiver [27, 28].
However, in adulthood, the reliance on BAT thermogenesis decreases, resulting in
its limited distribution in the human body, where it is only found in particular
anatomical regions such as the neck, supraclavicular region, posterior chest, and
belly [29]. Quantifying active BAT in humans remains a challenging task [30].
Currently, detection is primarily performed through the use of positron emission
tomography-computed tomography (PET-CT) or Z-spectral imaging [31, 32]. The
maximum mass of BAT detectable by humans is about 1 kg; in individuals aged 20 to
50 years, it ranges from 50 to 500 g, or 0.1% to 0.5% of adult total body
weight or 0.2% to 3.0% of adult total AT mass [29]. The overall amount of BAT
differs by sex [33] and is inversely related to age and body mass index (BMI)
[34, 35]. BAT is more active in young people, lean people, and women. In addition
to WAT and BAT, an induced form of brown-like adipocytes, namely beige/brite
adipocytes [36], can exist in rodents and humans as cellular clusters dispersed
within white fat depots, although their origin requires additional clarification
[24]. However, studies have confirmed that exercise, cold exposure, sympathetic
The anterior hypothalamus, a thermoregulatory area, activates BAT in response to a peripheral cold sensing signal, resulting in non-shivering thermogenesis. The hypothalamus stimulates sympathetic activation, which leads to the release of norepinephrine from the nerve terminal that innervates BAT. This norepinephrine activates a series of reactions within the cells, known as the intracellular lipolysis cascade, resulting in the release of fatty acids. These fatty acids are then used as fuel in the BAT mitochondrial electron transport chain [39]. Although human BAT primarily utilizes intracellular triglyceride storage during cold-induced activation, it is also capable of simultaneously uptaking fatty acids and glucose from the bloodstream [40]. This activation of BAT helps to counteract obesity and maintain the equilibrium of glucose and lipid metabolism. Accumulated evidence from the last four decades provides significant support for the notion that the stimulation of heat generation by BAT cells leads to a reduction in obesity in mice and rats [41]. BAT was previously believed to have no connection to adult physiology [42]. However, recent findings have “rediscovered” BAT and demonstrated that its activation offers numerous health advantages for various tissues including the GI tract, cardiovascular system, and musculoskeletal system [43].
In a retrospective study that included 52,487 patients with 134,529 18F-fluorodeoxyglucose PET-CT results, the researchers employed propensity score matching to construct the study cohort based on the presence or absence of BAT. According to their findings, those who had BAT had a decreased incidence of cardiometabolic disorders. The presence of BAT was independently associated with a lower incidence of type 2 diabetes, dyslipidemia, coronary artery disease, cerebrovascular disease, congestive heart failure, and hypertension. Significantly, the advantageous impacts of BAT were particularly evident in individuals who were overweight or obese, indicating that BAT might have a role in alleviating the detrimental consequences of obesity [43]. Emerging research indicates that BAT is negatively correlated with the basal metabolic rate and activates several parameters that enhance metabolic well-being, decrease the occurrence of metabolic disorders, and impede disease advancement [44, 45, 46]. Consequently, there is considerable enthusiasm surrounding the prospect of activating this tissue to combat diseases associated with metabolism.
Studies in rodents have shown that long-term activation of BAT improves liver cholesterol homeostasis, bile acid metabolism, and microbiome composition [47]. The prevention of chronic metabolic disorders by BAT has conventionally been ascribed to their capacity to utilize glucose and lipids for thermogenesis. However, BAT can also secrete more than 100 different adipokines (the so-called brown adipokines or batokines), including fibroblast growth factor-21 (FGF21), neuregulin 4 (NRG-4), phospholipid transfer protein, nerve growth factor, S100 calcium-binding protein B, and C-X-C motif chemokine 14 [48]. These adipokines demonstrate autocrine, paracrine, and endocrine activities, impacting a range of tissues including WAT, liver, pancreas, heart, bone, and the central nervous system (CNS). Consequently, they also play a pivotal role in exerting systemic regulatory functions [49, 50]. Their involvement spans various processes such as promoting brown adipocyte proliferation, hypertrophy, and activity, modulating glucose-lipid metabolism, and stimulating the browning of WAT [51, 52, 53]. Exosomes, as a special form of adipokine secreted by BAT, have also received attention in recent years for their role in the physiological pathology of BAT [54]. Exosomes contain a variety of bioactive substances such as transmembrane proteins, soluble proteins, lipids, and non-coding RNA derived from donor cells. Although the vast majority of body cells can release exosomes in response to stimulation signals, about 80% of the exosomes in the human blood circulation come from AT [55]. In addition, the relative content of exosomes in blood circulation is significantly increased in obese patients, and the cargos are also significantly altered [56, 57]. Studies have shown that BAT-derived exosomes can deliver bioactive molecules through paracrine/autocrine and endocrine pathways, and participate in local and systemic immunity, metabolism, and cell tissue regeneration [58, 59, 60]. Our previous studies also found that BAT-derived exosomes can directly regulate the metabolism of female ovarian tissue and oocyte quality [61].
The intricate interplay of diverse genetic, epigenetic, and environmental factors in the genesis of PCOS has been a focal point of research and debate [62]. Fresh insights have recently emerged, shedding light on the potential role of BAT in the pathogenesis of this complex endocrine disorder.
Abnormal alterations in AT function in PCOS patients are linked to insulin resistance and chronic inflammation, and similar changes can be seen in the AT of non-obese PCOS patients [63, 64, 65]. In animal models, dehydroepiandrosterone (DHEA)-induced PCOS rats have considerably lower BAT activity than normal control rats. Insulin resistance, irregular estrous cycle, and low birth rates are significantly improved in PCOS rats after BAT transplantation [66]. In human research, 18F-FDG PET-CT scan results have revealed that BAT activity in PCOS patients is considerably lower than that in healthy controls, and that BAT activity is inversely linked to the patient’s BMI and waist circumference [67]. The temperature of the supraclavicular skin is a reliable qualitative indication of BAT activity [68]. In a study including 44 women with PCOS and 11 control women, it was discovered that women with PCOS had a reduction of –0.6 °C to –0.7 °C, and that in general, the more severe the hyperandrogenism, the lower the supracclavian skin temperature was likely to be [69]. In addition to decreased BAT activity, PCOS patients had lower BAT mass. Li et al. [70] used Z-spectral imaging technology to analyze 13 PCOS patients, 19 healthy female controls, and 17 healthy male controls, and discovered that PCOS patients had a lower BAT score and larger adipose water fraction, indicating a reduction in BAT quality and function (Table 1, Ref [66, 67, 69, 70, 71]). BAT may contribute to the pathophysiology of PCOS in the following ways.
| Author, year (ref) | Country | Study design | Subjects | Sample size | Methods for evaluating BAT activity | Results |
| Yuan et al. [66], 2016 | China | In vivo animal experiment | PCOS mice (induced by the injection of dehydroepiandrosterone) | 8–10 mice in each group | Micro PET-CT | In DHEA-induced PCOS mice, there was a substantial decrease in BAT activity compared to the control mice. |
| Zheng et al. [71], 2022 | China | In vivo animal study | PCOS mice (induced by the injection of dehydroepiandrosterone) | 10 mice in each group | Micro PET-CT | BAT activity of mice in the PCOS group was significantly lower than that in the control group. |
| Oliveira et al. [67], 2019 | Brazil | Cross-sectional study | PCOS and healthy control women | 45 PCOS women; 25 healthy women | 18F-FDG PET-CT | Total BAT activity was significantly decreased in PCOS women compared to healthy controls. Furthermore, BAT activity correlated negatively with BMI and waist circumference. |
| Li et al. [70], 2020 | USA | Case-control study | PCOS women; Healthy control women and man | 13 PCOS women; 19 healthy control women; 17 healthy control men | Z-spectral imaging | PCOS women had a lower BAT score and a larger adipose water fraction, indicating a reduction in BAT quality and function. |
| Shorakae et al. [69], 2019 | Australia | Cross-sectional substudy, nested within a randomized control trial | PCOS women and non-PCOS control | 44 PCOS women and 11 non-PCOS control | Supraclavicular skin temperature (BAT activity index) was recorded with dataloggers | Compared with the control group, the supraclavicular skin temperature in PCOS women was lower overall (33.9 |
Abbreviations: 18F-FDG, 18-Fluoro-deoxyglucose; BAT, Brown adipose tissue; BMI, body mass index; DHEA, Dehydroepiandrosterone; PCOS, Polycystic ovary syndrome; PET-CT, Positron emission tomography–computed tomography.
The two main pathogenic mechanisms of PCOS are insulin resistance and
hyperandrogenemia. Insulin is the primary nutrient absorption and storage hormone
in the human body. AT accounts for 5% of insulin-mediated glucose absorption in
the lean human body. The rate is 20% in obese people [72]. BAT produces
bioactive lipids such as 12,13-Dihydroxy9z-octadecenoic acid and
12-hydroxyeicosapentaenoic acid, which increase glucose and fatty acid absorption
by BAT and muscle to sustain the body’s continued thermogenesis [73]. BAT also
produces exosomal microRNA, which can control the expression of genes involved in
glucose and lipid metabolism in other organs, such as the liver [74]. As a
result, changing the volume and function of BAT in PCOS patients can impair the
sensitivity of various organs in the body to insulin, including AT, liver, and
muscle, leading to insulin resistance and hyperinsulinemia. Insulin, as a
gonadotropin, can promote cytochrome P450 17A1 expression and activity in
follicular cells while also stimulating androgen production in the ovaries [75].
It is also worth noting that follicular theca cells in PCOS women are more
sensitive to insulin’s androgen-stimulating actions than in healthy women [76].
High insulin levels can also lower circulating sex hormone-binding globulin
levels and stimulate the pulse secretion of gonadotropin-releasing hormone
neurons in the anterior pituitary. The occurrence of hyperandrogenemia in PCOS
patients is consistently related to the synergistic effects mentioned above.
Under the influence of hyperandrogenemia, the expression of UCP1, peroxisome
proliferator-activated receptor coactivator 1, and cell death-inducing DNA
fragmentation factor-like effector A is decreased in BAT [77, 78]. The
sympathetic nervous system regulates the activity of brown fat cells through
adrenergic receptors. Meanwhile, sex hormones, including testosterone,
17
BAT has the ability to release a variety of adipokines. FGF21, interleukin 6 (IL-6), NRG4, vascular endothelial growth factor A, and bone morphogenetic protein 8b were the initial adipokines identified as being generated from BAT. Other adipokines, including chemokines and adiponectin, which were initially recognized as adipokines produced from WAT, have recently been confirmed to also be secreted from BAT [79]. BAT-derived adipokines, also referred to as batokines, exert impacts on metabolic and endocrine signaling in PCOS women. Batokine abnormalities have been described in PCOS women, including reduced serum adiponectin levels [80], increased leptin levels [81], and increased FGF21 levels [82]. Adiponectin is a very potent insulin sensitizer. Transplantation of BAT into DHEA-induced PCOS rat models has been shown to activate BAT activity. After a 3-week period, this transplantation leads to an increase in circulating adiponectin levels and a partial reversal of the PCOS phenotype. The observed improvements include the normalization of ovarian steroid hormone synthesis, reversal of glucose intolerance and insulin resistance, and an increase in live birth rates [66]. It is worth noting that a similar effect of BAT transplantation described above can be partially replicated in PCOS mouse models treated with recombinant adiponectin for 20 days. Furthermore, FGF21 is a cold-induced activator of BAT activity and an inducer of WAT browning in humans. FGF21 improves glucose consumption and increases energy expenditure in AT by improving insulin sensitivity and BAT thermogenesis [79]. As a result, when FGF21 production is aberrant, it contributes to the pathophysiology of PCOS by influencing insulin sensitivity and glucose and lipid metabolism. In the future, advancement of proteomics technology and its utilization in analyzing BAT secretome may lead to the discovery of more batokines that are associated with the development of PCOS. Nevertheless, it remains imperative to further characterize the batokines that have the potential for clinical translation.
The gut microbiota plays an active role in regulating the production of BAT. A recent study revealed the presence of brown fat cells in the WAT of neonatal mice, but their presence is diminished after weaning. The browning of WAT before weaning can be impeded by depleting the microbiota through antibiotic therapy or altering the microbiota via a high-fat diet. The inhibitory effect was also observed through the transplantation of cecal microbiota from neonatal mice fed a high-fat diet. Additionally, these interventions led to a reduction in the expression of liver genes associated with bile acid synthesis and a decrease in serum bile acid levels. The abundance of Porphyromonadaceae and Ruminococcaceae in the microbiota exhibited positive and negative correlations, respectively, with the induction of beige adipocytes [83].
Moreover, the gut microbiota and its metabolites play a crucial role in
influencing BAT activity. Specifically, butyric acid, a metabolite produced by
the gut microbiota, not only impacts appetite through the gut-brain axis but also
activates BAT via sympathetic nerve output, leading to an increase in the
oxidative metabolism of triglycerides [84]. In Mammalian 5
Recent research has indicated that the gut microbiome may influence the
progression of PCOS by impacting the function of BAT. The work conducted by Qi
et al. [11] involved collecting fecal bacteria from mice with PCOS
induced by DHEA. Then these bacteria were transplanted into recipient mice,
resulting in a decrease in the expression of Ucp1 and Pgc1a
mRNA in the BAT and WAT of the recipient animals. Nevertheless, the
administration of glycodeoxycholic acid and IL-22 can counteract the decrease in
Ucp1 and Pgc1a mRNA expression in the BAT and WAT of PCOS mice.
Gut microbiota dysbiosis in PCOS mice is believed to impede BAT function and
suppress WAT beiging [11]. In addition, Zhu et al. [86] discovered in a
PCOS mouse model induced by DHEA that enoxacin treatment could alleviate insulin
resistance and glucose intolerance, and stimulate the browning of subcutaneous
AT. This was evidenced by an increase in the expression of UCP1, as well as
mitochondrial regulatory factors PGC-1
In summary, the gut microbiota engages in active bidirectional communication with BAT through chemical messengers and metabolites. The equilibrium of BAT within the host’s body plays a pivotal role in the metabolic health of women. When the bidirectional communication between BAT and gut microbiota becomes imbalanced, it not only disrupts this crucial equilibrium but also contributes to and accelerates the development of PCOS (Fig. 2).
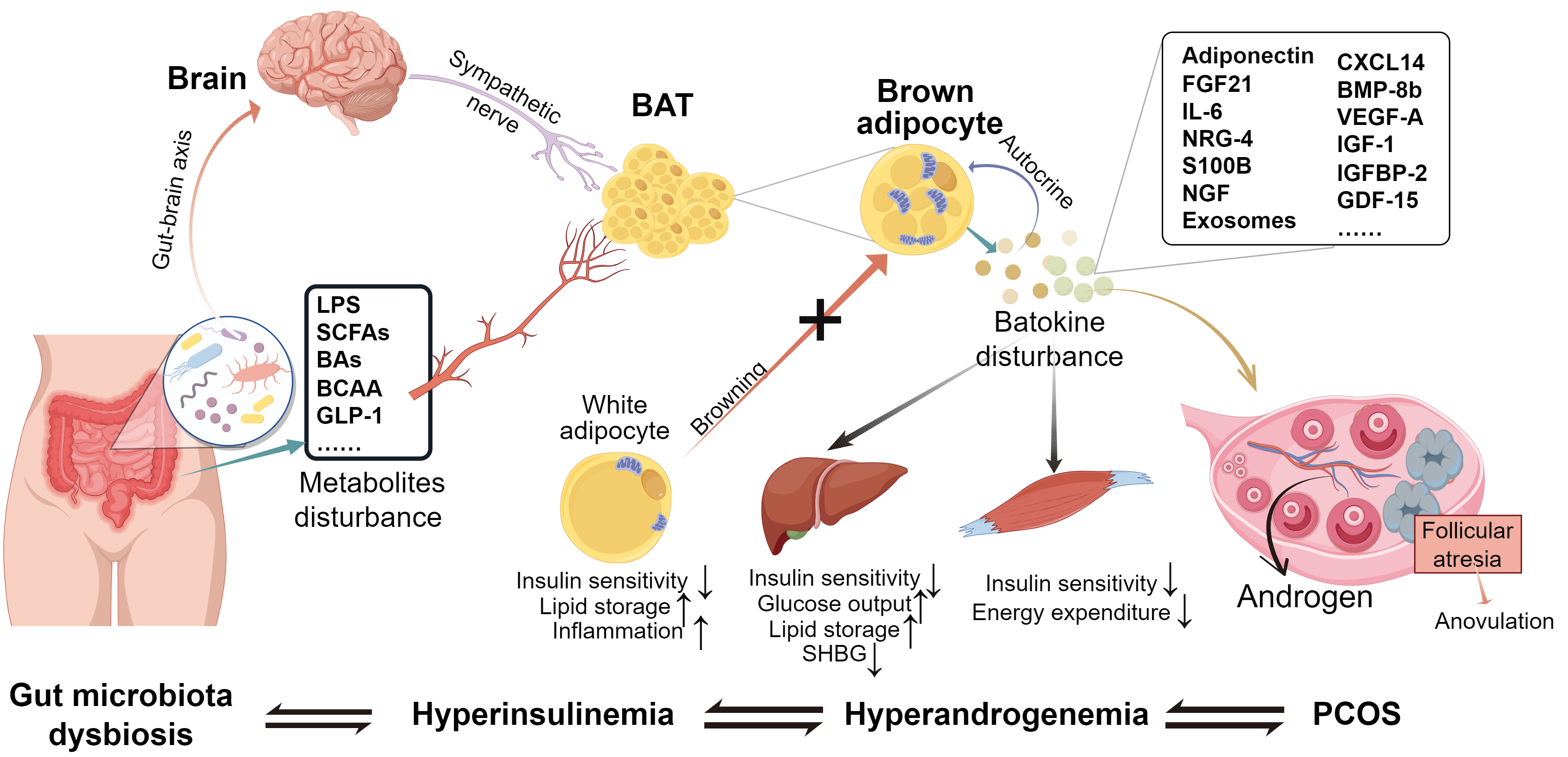 Fig. 2.
Fig. 2.The gut microbiota–BAT axis in PCOS pathogenesis. The dysbiosis of gut microbiota and its metabolites, which is affected by external as well as internal factors, can have a direct or indirect effect on the formation and function of BAT through the gut–brain axis. PCOS patients exhibit a decline in both the functionality and abundance of BAT, resulting in reduced energy expenditure and disturbance of the secretion of batokines. Consequently, this leads to the progression of obesity, insulin resistance, and hyperandrogenism. Essentially, the disruption of gut microbiota, abnormalities in BAT function, and their mutual interaction together contribute to the development of PCOS. LPS, Lipopolysaccharides; SCFAs, Short-chain fatty acids; BCAAs, Branched-chain amino acids; GLP-1, Glucagon-like peptide 1; BAT, Brown adipose tissue; FGF21, Fibroblast growth factor 21; NRG-4, Neuregulin-4; IL-6, Interleukin-6; S100B, S100 calcium-binding protein B; NGF, Nerve growth factor; CXCL14, C-X-C motif chemokine ligand 14; BMP-8b, Bone morphogenetic protein 8b; VEGF-A, Vascular endothelial growth factor A; IGF-1, Insulin-like growth factor 1; IGFBP-2, Insulin-like growth factor-binding protein 2; GDF-15, Growth differentiation factor 15; SHBG, Sex hormone-binding globulin. (The Fig. 2 was drawn by Figdraw, ID: TRIST8c31c).
The gut microbiota is now recognized as a bridge between the external environment and host homeostasis. In the regulation of BAT, the gut microbiota may influence through the following pathways: (1) enhancing the quality of BAT by inducing BAT progenitor cells; (2) promoting the WAT browning by stimulating the formation of beige adipocytes; and (3) increasing BAT function by upregulating the regulatory pathway of BAT [88]. Emerging evidence suggests that the gut microbiota can influence BAT function via a variety of molecular mechanisms, with microbial metabolites, including SCFAs and bile acids, as well as the gut–brain axis being proposed as avenues for the gut microbiota to influence BAT.
Many studies have been conducted to investigate the role of gut microbiota, membrane components (LPS), and metabolites (SCFAs, endocannabinoids, bile acids, aryl hydrocarbon receptor ligands, and tryptophan derivatives) in WAT browning and/or beiging, as well as changes in BAT activity [89]. There has, however, been limited studies in this area in PCOS, with the main focus being on SCFAs. As a result, our review is primarily about SCFAs.
SCFAs are fatty acids characterized by carbon chains of fewer than six carbon atoms. These acids originate from dietary fiber in foods, such as fructooligosaccharides and resistant starch, and are produced through the fermentation of gut microbiota in the cecum, colon, and feces. Notably, the human body lacks corresponding enzymes for digesting dietary fiber. SCFAs, including propionate, acetate, and butyrate, exert their biological effects primarily through two classes of G-protein-coupled receptors, GPR41 and GPR43. Both human and animal studies have demonstrated that acetate has beneficial effects on host metabolism. It improves insulin resistance, suppresses appetite, reduces lipolysis, and decreases systemic pro-inflammatory cytokine levels. These effects are partly attributed to the stimulation of the intestinal hormone GLP-1 secreted by colon L cells [90, 91]. Furthermore, the metabolites propionate and butyrate generated by gut microbiota activate intestinal gluconeogenesis via a complementary mechanism. Under cold exposure, UCP-1-dependent thermogenesis is decreased and WAT browning is repressed in germ-free animals or mice treated with antibiotics to deplete gut microbiota; The foregoing defects can be reversed by recolonizing the gut microbiota or using butyrate gavage [92]. Butyrate has also been shown in studies to decrease the activity of appetite neurons expressing neuropeptide Y in the hypothalamus and reduce food intake in experimental mice when administered orally rather than intravenously. Long-term administration may aid in the prevention of obesity and the reversal of insulin resistance. Butyrate strongly stimulates BAT due to an increase in sympathetic nerve output, which innervates BAT. The benefits of butyrate on food intake and stimulation of BAT metabolic activity are removed by subphrenic vagotomy, indicating that the gut–brain axis partly mediates the metabolism improvement effects of butyrate [84].
Indoles are a distinct group of metabolites found in the gut microbiota. This category includes substances such as indoxyl sulfate, indole-3-acetic acid, indole-3-propionate, indole, and 3-methylindole (3-MI). A previous study indicated a correlation between these factors and alterations in insulin sensitivity and glucose metabolism in PCOS patients. A significant inverse relationship was identified between 3-MI and GLP-1 levels [93].
Bile acids have long been recognized as vital in dietary lipid absorption and
cholesterol catabolism; however, in recent years, the importance of bile acids as
signaling molecules has been recognized. The liver synthesizes bile acids via two
distinct pathways including several enzyme activities. The rate-limiting enzyme,
cholesterol 7-alpha-hydroxylase, generally dominates the conventional bile acid
production route. Bile acids can also be produced via an alternate bile acid
synthesis pathway that begins with the activity of sterol 27-hydroxylase and
25-hydroxycholesterol 7-alpha hydroxylase. These two synthetic pathways produce
the identical types of bile acids, which are then combined with either glycine or
taurine [94]. The gut microbiota metabolizes the bile acids released into the gut
to generate a range of bile acid species, including oxalonic acid and deoxycholic
acid [95]. These bile acids can regulate thermogenesis by activating
mitogen-activated protein kinase pathways (which are also ligands for G
protein-coupled receptor, TGR5) and nuclear hormone receptors (such as farnesoid
X receptor alpha [FXR-
The gut–brain–BAT axis is a bidirectional, dynamic network. On the one hand, the gut microbiota influences the abundance of metabolites such as fatty acids, neurotransmitters, and secondary bile acids, as well as directly or indirectly regulates CNS activity via the gut–brain axis signaling pathway. On the other hand, signals from the complex neuronal network of the CNS can influence intestinal motility, gut microbiota composition, and BAT activity. Many aspects of the gut–brain–BAT axis, a complex regulatory network, still need to be elucidated (particularly the CNS output signaling pathway). Here, we primarily explored the potential role of the gut–brain–BAT axis in the pathogenesis of PCOS, focusing on input stimulus signals.
The gut microbiota exerts influence on the secretion of GI hormones through various metabolites and immune regulation. The GI system plays a role in food intake and metabolism through a variety of orexigenic and anorexigenic GI hormones. Leptin stands as the sole circulating orexigenic hormone, while anorexigenic hormones comprise GLP-1, gastric inhibitory peptide, peptide YY (PYY), and cholecystokinin (CCK). In PCOS patients, postprandial leptin inhibition was either decreased or unchanged compared to healthy women. GLP-1, PYY, and CCK levels remained unchanged or decreased during both fasting and postprandial states [101]. GLP-1 is produced peripherally by intestinal endocrine L cells and neurons in the nucleus tractus solitarii. Intestinal GLP-1 enhances glucose-stimulated insulin secretion and reduces food intake. Central administration of GLP-1 or GLP-1 receptor (GLP-1R) agonists significantly increases whole-body energy expenditure [102], potentially through hypothalamic GLP-1R and subsequent activation of the thermoregulatory network [103] that regulates BAT thermogenesis. However, it is unlikely for intestinal GLP-1 to reach hypothalamic GLP-1R due to degradation by dipeptidyl peptidase-4 in circulation. Therefore, intestinal GLP-1 is more likely to regulate BAT activity through the gut–brain axis pathway. Nonetheless, studies have indeed shown that intestinal GLP-1 can control satiety through vagoafferent neurons (VANs) and regulate BAT thermogenesis through the synaptic connection between VANs and BAT [104]. Notably, the activation of BAT thermogenesis in obese PCOS patients after bariatric surgery is also associated with increased GLP-1 production in the gut [105].
By burning calories, BAT activation can have a positive influence on blood sugar, lipids, and fat mass from a metabolic perspective. Research has demonstrated that the activation of a single sugar cube of BAT over the course of 1 year can result in the burning of 3–4 kg WAT [106]. Utilizing the characteristics of BAT to enhance energy expenditure and address metabolic issues [36, 107], such as PCOS, can be achieved by inducing WAT browning or activating BAT through the management of gut microbiota [108]. Several treatment options have been proposed to potentially address PCOS and its related symptoms by modulating AT (BAT) and/or gut microbiota. These approaches have demonstrated encouraging therapeutic efficacy (Fig. 3).
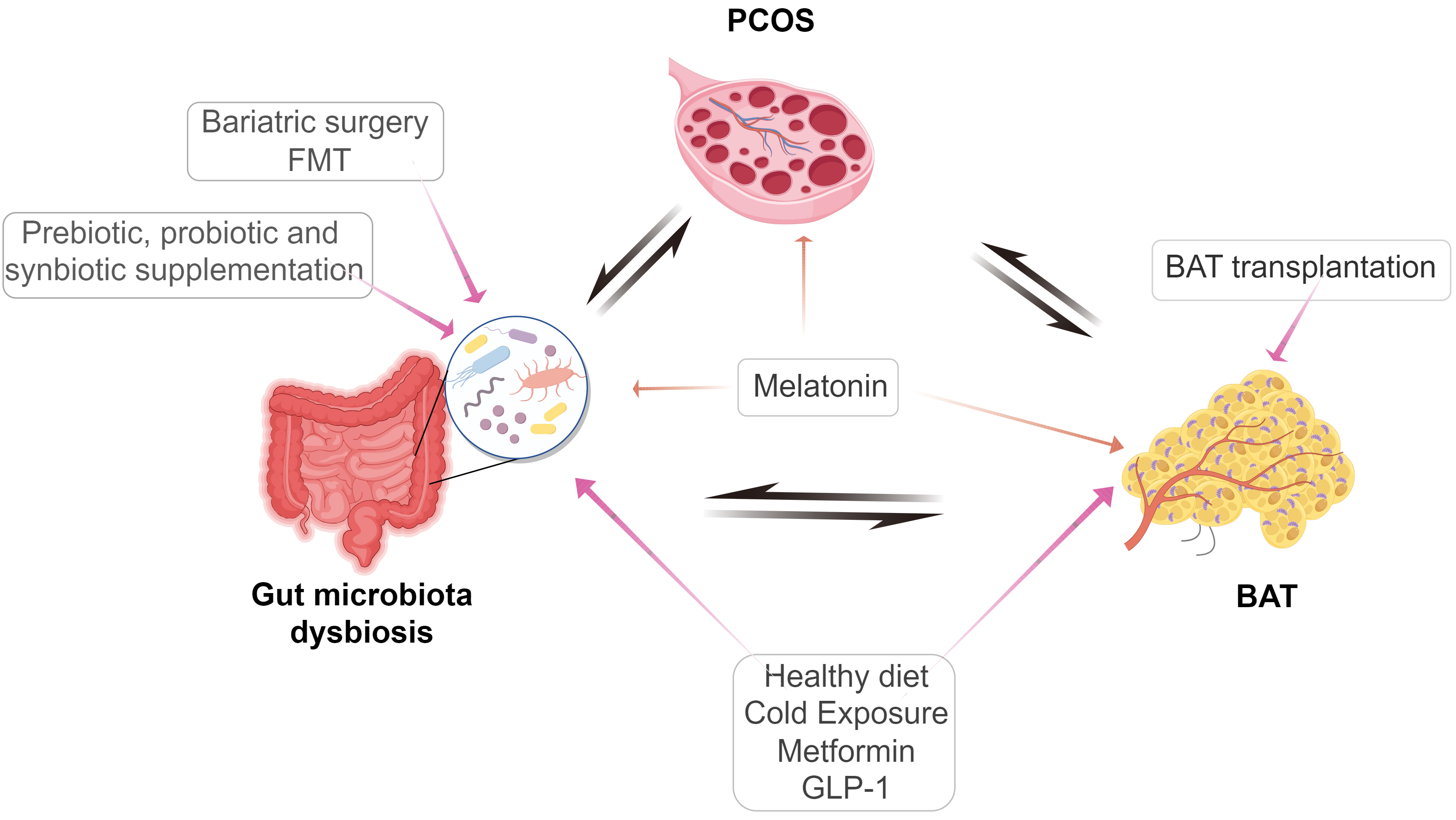 Fig. 3.
Fig. 3.Possible therapeutic targets for these PCOS treatment strategies that are being applied or attempted. FMT, Fecal microbiota transplantation; BAT, Brown adipose tissue; GLP-1, Glucagon-like peptide 1. (The Fig. 3 was drawn by Figdraw, ID: RTWRI10971).
A series of clinical studies have demonstrated that regulating gut microbiota through prebiotic, probiotic, and synbiotic supplements has a range of positive effects on PCOS patients, including improved metabolism, reduced inflammation, and improved reproductive health [109]. However, more research is needed to further validate these effects and determine the optimal supplement and dosage.
Cold exposure occurs when mammals are exposed to temperatures below their thermoneutral threshold (~30 °C in mice and ~24 °C in humans), which activates BAT and stimulates the emergence of beige fat cells in WAT, boosting energy expenditure by non-shivering thermogenesis [110]. Cold exposure induces active thermogenic fat by promoting the release of neurotransmitters such as catecholamines from nerve terminals or M2 macrophages. Recent research has found that cold-induced thermogenic activation of beige and brown fat cells coincides with changes in gut microbiota. Cold exposure alters the composition of the gut microbiome, often known as the cold microbiome. Transplanting cold microbiota into germ-free mice improves insulin sensitivity and cold tolerance, in part through promoting browning of white fat, resulting in higher energy expenditure and fat loss [111]. In different antibiotic cocktails (ABX) therapy to deplete gut microbiota or in germ-free animals exposed to cold, the expression of UCP1 in BAT was reduced and the browning process of WAT was inhibited. Butyrate was administered intragastrically to ABX-treated mice to increase their thermogenesis capacity. This study further supported the significance of gut bacteria in cold-induced BAT activation [92].
By activating endogenous BAT in a DHEA-induced PCOS rat model, low temperature exposure for 20 days can reverse estrous cycle irregularity, decrease circulating testosterone and luteinizing hormone, and significantly reduce the expression of steroidase and inflammatory factors in the ovaries. Cold exposure significantly decreases ovarian follicular and increased corpus luteum, indicating that ovulation returns to normal levels. Cold exposure significantly reduces diet-induced obesity and is linked to improved thermogenesis of iBAT in the spine region and a plasma bile acid profile comparable to that of germ-free mice, according to Ziętak et al. [112]. The gut microbiota composition of mice is dramatically altered at the phylum and genus levels on the first day after acute cold exposure and after 4 weeks at 12 °C. Changes in gut microbiome composition include increased levels of Adlercreutzia, Mogibacteriaceae, Ruminococcaceae, and Desulfovibrio, as well as lower levels of Bacilli, Erysipelotrichaceae, and the genus rc4-4. These genera are linked to leanness and obesity, respectively. In comparison to germ-free mice reared at room temperature on a high-fat diet, the transplantation of cecal microbiota from mice housed in a 12 °C environment resulted in enhanced glucose tolerance and decreased fat accumulation in germ-free mice. However, no such effects were observed in FMT from mice housed in a 29 °C environment. Thus, at lower temperatures, the gut–liver–BAT axis may mediate a protective effect against obesity. Worthmann et al. [47] also documented a metabolic program that is activated in mice when exposed to cold. This program coordinates the processing of lipoproteins in BAT with the conversion of cholesterol to bile acids in the liver, using alternate synthetic pathway. The process depends on the liver to stimulate the production of cytochrome P450 family 7 subfamily B polypeptide 1, which leads to higher amounts of bile acids in circulation and feces. This is followed by notable alterations in the gut microbiota and an increase in thermogenesis. Genetic and pharmacological therapies aimed at inhibiting bile acid synthesis and bile excretion effectively prevents the excessive excretion of bile acids in the stool. These interventions also resulted in changes to the composition of microbiota and influenced the body’s thermogenic responses. This study additionally recognized bile acids as crucial metabolic agents for the activation of BAT, and also suggested the impact of the host’s cholesterol metabolism on the alterations in gut microbiota and energy metabolism [47].
Nevertheless, there are limited studies available on the prolonged induction of “beiging” through cold exposure. Studies involving rodents have typically lasted between 4 h [113] and 6–7 days [114]. Cold exposure, which is the most extensively studied method for inducing browning, is challenging to sustain for extended periods in humans due to its discomforting nature. In addition, there have been no reported clinical studies on the application of cold exposure in PCOS patients. Further research is anticipated to investigate the optimal duration of cold exposure and its impact on human PCOS.
The diet significantly influences the composition of the gut microbiota. Alterations in dietary patterns can induce rapid and significant modifications in gut microbiota in just 1 day [115]. Currently, numerous studies have demonstrated that the imbalance of gut microbiota, resulting from diets high in fat, sugar, and calories due to urbanization, are associated with the incidence and phenotype of PCOS [116]. Nevertheless, adhering to a diet rich in fiber, with a low glycemic index and low glycemic load, might effectively enhance insulin sensitivity and alleviate irregularities in the menstrual cycle among individuals with PCOS [116, 117]. For all PCOS sufferers, starting with a healthy lifestyle is the first step towards therapy [118]. A healthy diet involves consuming sufficient, yet not excessive, quantities of nutrients and health-enhancing compounds from nourishing foods, while abstaining from the consumption of substances that are detrimental to one’s well-being. Health-related diets commonly share several commonalities, including a high content of fiber, unsaturated fatty acids, and polyphenols, while being low in saturated fat, sodium, and refined carbs [119]. In a recent randomized controlled trial (RCT), the combination of a high-fiber diet with acarbose had a more pronounced effect on the composition of the gut microbiota compared to a high-fiber diet alone. Furthermore, it resulted in a reduction of circulating hyperandrogen, homeostasis model assessment-insulin resistance, and an increase in adiponectin levels [120]. Adiponectin, being a potent enhancer of insulin sensitivity, plays a significant role in stimulating the browning of WAT and activating the function of BAT [121].
The commonly utilized insulin sensitizers include biguanides, thiazolidinediones, alpha-glucosidase inhibitors, and GLP-1RAs. In this discussion, our main focus will be on metformin, a commonly used medication in PCOS patients, and GLP-1RA, a comparatively recent therapeutic option for PCOS.
The mechanism of action of metformin is complex, mainly by reducing
glycogenolysis in the liver, while the gut also plays a key role. Metformin’s
therapeutic effects are determined by factors such as medication dosage and
duration. At the molecular level, it involves both AMP-activated protein kinase
(AMPK)-dependent and AMPK-independent pathways, mitochondrial respiration
regulation, and the lysosome pathway [122]. The study shows that the positive
benefits of metformin on glucose tolerance and weight loss are diminished when
the intestine AMPK
GLP-1 is an incretin hormone secreted by L cells in the ileum and distal colon. Enterogenic GLP-1 exerts its effects on the islets and brain, leading to decreased blood glucose levels and reduced appetite and body weight, respectively. GLP-1RAs represent a novel class of medications that help lower blood glucose levels by stimulating insulin release [129]. GLP-1RA has been found to indirectly influence the composition of gut microbiota in individuals with metabolic disorders, both in animals and humans, by improving glycemic control, reducing body weight, modulating intestinal motility, and affecting immunological function and interactions between microbiota and epithelial lymphocytes [130]. Clinical trials have revealed that GLP-1RA enhances menstrual regularity in PCOS patients [131, 132]. Animal studies have demonstrated that peripheral administration of GLP-1RA in obese mouse models effectively alleviates obesity induced by a high-fat diet, primarily through the stimulation of WAT browning [133]. Another study reported that GLP-1RA administration in a PCOS mouse model significantly reduced hyperinsulinism and hyperandrogenemia by inhibiting the local inflammatory response of ovarian tissue and increasing WAT browning [134].
A previous study revealed that BAT transplantation successfully reverses anovulation, hyperandrogenism, and polycystic ovaries in rats with PCOS caused by DHEA. Simultaneously, BAT transplantation into rats with PCOS rats can effectively enhance the regularity of the estrous cycle and insulin sensitivity, as well as enhance the fertility. The therapeutic effects observed following BAT transplantation are attributed to the stimulation of the endogenous BAT and the elevation of adiponectin levels in the circulation [66, 135]. Nevertheless, due to the difficulty of acquiring BAT from healthy individuals and the inherent limits of xenotransplantation, the implementation of BAT transplantation remains arduous in PCOS patients. BAT-derived exosomes are utilized as an alternative therapeutic strategy. In a high-fat diet-induced obese mouse model, exosomes derived from BAT were administered intravenously through the tail vein. This intervention resulted in a decrease in body weight, blood glucose levels, and fat accumulation. These findings indicate that exosomes derived from BAT possess metabolic advantages similar to those of BAT itself in obese mice [136]. Furthermore, exosomes produced from BAT possess notable benefits such as potent biological efficacy, exceptional stability, straightforward manipulation, robust immunological tolerance, and are thus highly suitable for clinical translation [137, 138].
FMT is a procedure that involves transferring the functional microbiota of a
healthy individual into the GI tract of a patient to restore a healthy community
of gut microbiota and treat intestinal and parenteral disorders [139]. Studies in
animal models have shown that when fecal bacteria from a mouse model of
DHEA-induced PCOS are transplanted into healthy mice, they can suppress the
production of UCP-1 and PGC-1
Between 50% and 80% of women diagnosed with PCOS exhibit obesity. Bariatric
surgery can facilitate weight loss in PCOS patients, leading to improvements in
blood pressure, blood glucose levels, menstrual irregularities, and hirsutism, as
well as enhancing ovulation and birth rates. The 2023
international evidence-based guideline proposed that PCOS should be regarded as a
metabolic disorder, which may warrant considering bariatric surgery at a lower
BMI threshold, similar to other metabolic disorders [118]. The current guidelines
set forth by the American Society for Metabolic and Bariatric Surgery and the
International Federation for the Surgery of Obesity and Metabolic Disorders state
that individuals with a BMI of
Melatonin is an indoleamine hormone primarily produced by the pineal gland in the absence of light stimulation, playing a crucial role in regulating the circadian rhythm of peripheral organs [144]. Additionally, peripheral germ cells, including granulosa cells, cumulus cells and oocytes, also possess the capability to synthesize melatonin. The local synthesis of melatonin within ovarian tissue is considered a key factor contributing to the significantly higher concentration of melatonin observed in follicular fluid compared to blood circulation. Apart from circadian rhythm regulation, melatonin exhibits antioxidant, anti-inflammatory, and anti-obesity effects [145]. Notably, recent studies have suggested that melatonin may act as a thermogenic agent, promoting the formation of beige AT or browning of WAT, enhancing the quality and activity of BAT [146, 147], and influencing the production of adipokines such as leptin and adiponectin. Furthermore, melatonin has been implicated in the modulation of gut microbiota. In mice lacking the arylalkylamine N-acetyltransferase gene, decreased endogenous melatonin levels induce gut microbiota dysbiosis (characterized by reduced Bacteroidetes abundance and increased Lactobacillus abundance), increased gut permeability, gut and systemic inflammation, and exacerbation of diet-induced obesity [148]. The effects of melatonin on BAT activity and gut microbiota homeostasis suggest its potential therapeutic value in treating or ameliorating symptoms associated with PCOS. Both animal and human studies have demonstrated that maintaining regular circadian rhythms and adequate melatonin levels contribute to successful reproduction [149]. However, clinical studies have identified lower concentrations of melatonin in the blood circulation and follicular fluid of women with PCOS, which may be associated with increased oxidative stress within the ovarian microenvironment of PCOS and compromised oocyte and embryo quality [150]. Presently, several RCTs have shown that daily supplementation with 3 mg melatonin in women with PCOS can improve menstrual irregularity and biochemical hyperandrogenemia [151], as well as increase the rates of mature egg retrieval and embryo quality [152, 153], and enhance clinical pregnancy rates [153] during in vitro fertilization treatment. Nevertheless, studies investigating whether melatonin can enhance the endocrine status and reproductive capacity of PCOS patients through direct modulation of gut microbiota and BAT activity remain scarce.
The etiology of PCOS, a rapidly increasing and highly enigmatic condition posing a significant threat to women’s reproductive well-being globally, remains unclear. Recent findings have underscored the pivotal role of gut microbiota dysbiosis and BAT dysfunction in the development of this condition. The exploration of gut microbiota and AT has garnered increased attention in PCOS research, mirroring studies on metabolic disorders like type 2 diabetes and non-alcoholic fatty liver disease. Given the prevalent metabolic issues in individuals with PCOS, it is plausible to hypothesize that the interaction between gut microbiota and BAT contributes to the development and phenotype of PCOS. As highlighted in this review, recent advancements in animal and clinical research have underscored the close relationship between disturbances in gut microbiota and alterations in BAT activity and mass in the PCOS pathogenesis. Notably, there exists a profound bidirectional interaction among gut microbiota, BAT, and PCOS, collectively contributing to the development of insulin resistance, chronic inflammation, and hyperandrogenemia in PCOS pathophysiology. Various therapeutic approaches targeting the restoration of gut microbiota homeostasis and modulation of BAT activity or mass, such as cold exposure, metformin, GLP-1 agonists, weight loss surgery, and melatonin supplementation, have demonstrated efficacy in reversing the pathological processes underlying PCOS.
This review delves into recent scientific advancements concerning the interplay between gut microbiota and BAT, as well as their correlation with the development of PCOS. Furthermore, we explore the underlying mechanisms that drive these interactions. The article offers a comprehensive overview of therapeutic approaches for PCOS that target the modulation of the relationship between gut microbiota and BAT. Recent research findings highlight the gut microbiota’s capacity to influence BAT function through various pathways, including SCFAs, metabolites, and the gut–brain axis. A decline in BAT activity or quantity contributes to insulin resistance, hormonal imbalances, chronic inflammation, and associated conditions. This is attributed to the creation of a positive energy balance, thereby promoting the onset and diverse phenotypes of PCOS.
Further research is essential to increase the sample size and encompass a more representative population in order to utilize standardized sampling, sequencing, and analysis procedures. This comprehensive approach aims to reveal species-specific changes in the gut microbiota of PCOS patients and identify significant microbial groups.
It is imperative to clearly elucidate the involvement of BAT dysfunctions in the pathogenesis of PCOS induced by gut microbiota, especially in humans. A deeper exploration into the specific mechanisms of their interaction is crucial for uncovering new intervention targets and strategies.
Conducting more prospective randomized controlled trials is necessary to assess the efficacy of diverse therapies targeting gut microbiota, BAT, and their interactions in PCOS. The ultimate goal is to enhance the short- and long-term health outcomes for individuals affected by PCOS.
YL, YF and HZ contributed to the formal analysis, methodology, investigation, and writing the original draft. YL and HZ participated in conceptualization, and writing review & editing. HW designed the work, reviewed it critically, contributed to supervision and funding acquisition. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was funded by the National Key Research and Development Program of China (2023YFC2705400).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



