1 Institute of Regenerative Medicine, and Department of Dermatology, Affiliated Hospital of Jiangsu University, Jiangsu University, 212001 Zhenjiang, Jiangsu, China
2 Guangdong Provincial Key Laboratory of Large Animal Models for Biomedicine, and South China Institute of Large Animal Models for Biomedicine, Wuyi University, 529020 Jiangmen, Guangdong, China
3 School of Pharmacy and Food Engineering, Wuyi University, 529020 Jiangmen, Guangdong, China
4 Department of Medical and Life Sciences, Faculty of Pharmaceutical Sciences, Tokyo University of Science, 278-8510 Chiba, Japan
5 Center for Stem Cell Biology and Regenerative Medicine, Institute of Medical Science, The University of Tokyo, 108-8639 Tokyo, Japan
Abstract
Circadian rhythms, the natural cycles of physical, mental, and behavioral changes that follow a roughly 24-hour cycle, are known to have a profound effect on the human body. Light plays an important role in the regulation of circadian rhythm in human body. When light from the outside enters the eyes, cones, rods, and specialized retinal ganglion cells receive the light signal and transmit it to the suprachiasmatic nucleus of the hypothalamus. The central rhythm oscillator of the suprachiasmatic nucleus regulates the rhythm oscillator of tissues all over the body. Circadian rhythms, the natural cycles of physical, mental, and behavioral changes that follow a roughly 24-hour cycle, are known to have a profound effect on the human body. As the largest organ in the human body, skin plays an important role in the peripheral circadian rhythm regulation system. Like photoreceptor cells in the retina, melanocytes express opsins. Studies show that melanocytes in the skin are also sensitive to light, allowing the skin to “see” light even without the eyes. Upon receiving light signals, melanocytes in the skin release hormones that maintain homeostasis. This process is called “photoneuroendocrinology”, which supports the health effects of light exposure. However, inappropriate light exposure, such as prolonged work in dark environments or exposure to artificial light at night, can disrupt circadian rhythms. Such disruptions are linked to a variety of health issues, emphasizing the need for proper light management in daily life. Conversely, harnessing light’s beneficial effects through phototherapy is gaining attention as an adjunctive treatment modality. Despite these advancements, the field of circadian rhythm research still faces several unresolved issues and emerging challenges. One of the most exciting prospects is the use of the skin’s photosensitivity to treat diseases. This approach could revolutionize how we think about and manage various health conditions, leveraging the skin’s unique ability to respond to light for therapeutic purposes. As research continues to unravel the complexities of circadian rhythms and their impact on health, the potential for innovative treatments and improved wellbeing is immense.
Keywords
- circadian rhythm
- retinal ganglion cells
- melanocytes
- photoneuroendocrinology
- genetic clock
- BMAL1
- liver fatty
- T2DM
- phototherapy
The rotation of the Earth causes a rhythm of sunrise and sunset which in turn affects the physiological rhythms of living organisms. Light exposure plays a modulatory role in adjusting circadian rhythms. The central clock in the suprachiasmatic nucleus responds to changes in light by regulating the peripheral clock, which is found throughout the body, such as the heart, liver, lungs, and intestines.
Numerous physiological processes in the human body are rhythmic, such as sleep and wake cycles, gastrointestinal motility, and blood pressure fluctuations. However, circadian rhythms are often disrupted by artificial lighting and shift work, increasing the risk of many diseases. Epidemiological surveys on rotating shift workers have shown increased risk of tumors [1, 2, 3, 4] and gastrointestinal symptoms compared with the general population. In addition, circadian disruptions increase the risk of cardiovascular diseases, such as heart attacks [5], strokes [6], and sudden death [7]. These events usually occur in the morning and are associated with increases in heart rate, blood pressure, plasma viscosity and other parameters. The correlation between diseases and the disturbance in circadian rhythm has been demonstrated by an increasing volume of academic research. However, studies can only elucidate the correlation between circadian gene expression and disease development, but the underlying molecular mechanism of circadian disruption affecting gene expression and the gene expression causing a disease remains the focus of on-going investigations. In addition, the peripheral clock is not only influenced by the master clock but can also adjust itself. This may be attributed to the widespread presence of melanocytes in the skin. Melanocytes are responsible for the ability of the skin to detect light [8, 9], allowing blind people to be affected by changes in light [10]. It remains important to understand how the central and peripheral clocks balance their relationships.
It is of great significance to systematically understand the mechanisms by which circadian rhythm disorders lead to various diseases. This work attempts to delineate and summarize how changes in circadian rhythms affect physiological processes. On the basis of our findings, we suggest directions for future research.
As an old Chinese saying goes, “Work when the sun rises and rest when the sun sets”. The sleep-wake cycle is a classic expression of a circadian rhythm. The presence of a central biological clock keeps cells and tissues coordinated throughout the body. This rhythm is beneficial for regulating DNA repair and enhancing resistance to DNA damage [11, 12]. When this synchronism is broken, it can affect cardiovascular [6, 7], metabolic [13, 14, 15], cognitive [16, 17, 18], immune [19, 20] and carcinogenic functions as well as work efficiency and productivity. Circadian disruption has also been linked to bone fractures, car crashes, and other accidents [21, 22, 23]. This indicates the necessity of maintaining circadian rhythms.
The suprachiasmatic nucleus (SCN) in the anterior hypothalamus serves as the
human body “master clock” [24]. Neurons within the SCN regulate the body’s
molecular clock via neurohormonal modulations that enable all the cells to work
in concert. The molecular mechanism underlying circadian rhythms is thought to
involve a series of interlocking molecular rings. The circadian locomotor output
cycles kaput (CLOCK) and brain and muscle aryl hydrocarbon receptor
nuclear translocator-like 1 (BMAL1, ARNTL) are fundamental
helix-loop-helix transcription factors that make up the positive arm of the core
clock loop [25]. The clock’s restraining leg, represented by the cryptochrome
(CRY) 1-2 and period (PER) 1-3, is activated by the
CLOCK/BMAL1 heterodimer to activate clock control genes (CCGs). The
transcription and translation of PER and CRY are boosted by
CLOCK/BMAL1 heterodimer binding to conserved promoter regions enhance
elements (E-box) [26]. CRY and PER then enter the nucleus and
form a negative feedback loop through inhibiting CLOCK/BMAL1
transactivating activity and ending CRY and PER transcriptional
activation. The negative feedback loop stops when CLOCK/BMAL1
suppression is removed. The entire process forms a loop with a period of
approximately 24 h. A secondary loop comprises retinoid-related orphan receptor
alpha (ROR
Melanocytes may also perform sensory functions that sense external light sources [9, 27, 28]. Research has shown that light has an important effect on circadian rhythm. Retinal rod and cone photoreceptors, which contain visual pigments such as rhodopsin in rods and different photopigments in cones sensitive to various wavelengths of light, transmit light information to the suprachiasmatic nucleus (SCN) and other brain regions, along with specialized retinal ganglion cells (RGCs) [29]. Among these, a subset of RGCs known as intrinsically photosensitive retinal ganglion cells (ipRGCs) are particularly noteworthy. ipRGCs possess a unique light-sensitive pigment called melanopsin (OPN4), which is especially sensitive to certain wavelengths of light, particularly blue light. This sensitivity enables ipRGCs to directly detect environmental light changes. Furthermore, the presence of melanopsin makes these RGCs photosensitive, playing a crucial role in conveying light signals to the SCN and contributing to the regulation of circadian rhythms. In addition to their conventional functions, melanocytes have special functions, such as decoding and responding to light information [30]. Therefore, neurons in the SCN can operate autonomously and reset in response to light even in blind people [10]. In addition, the skin has also been reported to have light-sensing functions. There are several types of opsins (OPN1-SW, OPN2, OPN3, and OPN5) expressed in human epidermal cells, which suggests the existence of photoreceptors in the epidermis [30]. OPN5 can induce the expression of PER in mammals, thus the skin is involved in the regulation of circadian rhythms in the surrounding tissues [31]. Melanocytes have more sensory functions to be discovered [9]. The skin’s response to light exposure in relation to circadian rhythm regulation is a burgeoning area of research, with no definitive quantitative standards currently established to determine the specific characteristics of light exposure necessary to elicit meaningful circadian effects. Human response to light exposure is influenced by a multitude of factors, including the intensity and wavelength of light, duration of exposure, and individual physiological traits. Blue light, with wavelengths around 460–480 nanometers, is considered particularly effective in modulating human circadian rhythms [32]. The duration of light exposure also plays a critical role [33, 34, 35]. Even low-intensity light, if exposed for a sufficient duration, can impact circadian rhythms. Furthermore, the extent of skin surface area exposed to light is an aspect warranting exploration in understanding its impact on circadian regulation. Different individuals exhibit substantial variability in their sensitivity to light exposure. Phillips and colleagues [36] have demonstrated that the variability in individual sensitivity to nocturnal light can exceed a 50-fold difference. Such inter-individual differences may be a crucial determinant of the role of the biological clock in human health and disease. The variability in individual sensitivity to nocturnal light might be genetically determined. For instance, the polymorphism rs10830963 in the melatonin receptor MTNR1B gene is significantly associated with the sensitivity of melatonin to light exposure [37] (Fig. 1).
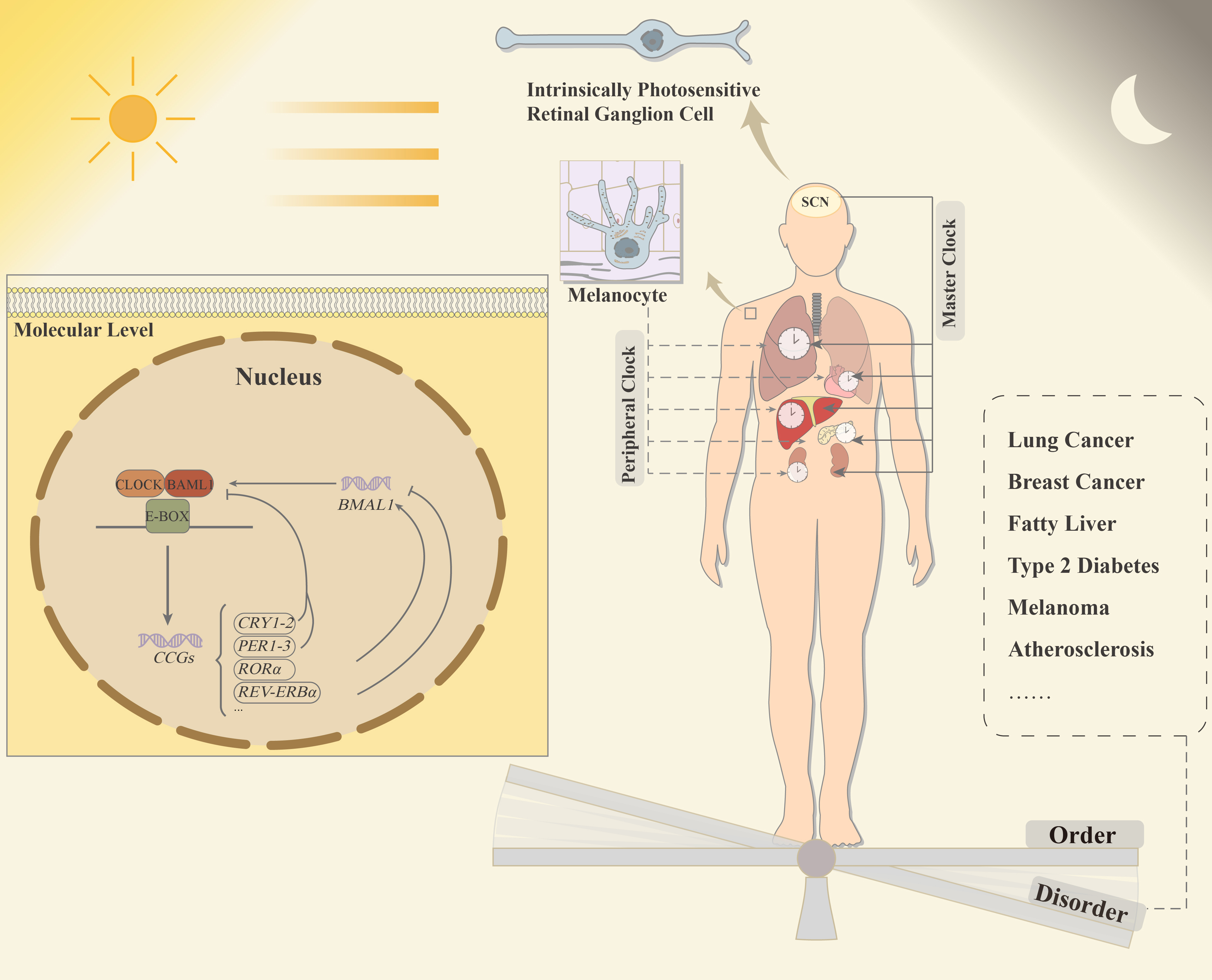 Fig. 1.
Fig. 1.Schematic diagram of circadian rhythms and organismal
homeostasis and disorders. When light from outside enters the eyes, the
intrinsically photosensitive retinal ganglion cells (ipRGC) capture the light
signal and convey it to the suprachiasmatic nucleus (SCN) in the hypothalamus.
SCN acts as the master clock to synchronize circadian rhythms in peripheral
tissues. From the molecular perspective, circadian locomotor output cycles kaput
(CLOCK) and brain and muscle aryl hydrocarbon receptor nuclear
translocator-like 1 (BMAL1, ARNTL) make up the positive arm of
the core clock loop. The clock’s restraining leg, represented by the proteins
cryptochrome (CRY) 1-2 and period (PER) 1-3, is activated by CLOCK/BMAL1
heterodimer to activate clock control genes (CCGs). The transcription and
translation of PER and CRY are boosted by the CLOCK/BMAL1
heterodimer’s binding to conserved promoter regions enhance elements (E-box).
Then CRY and PER proteins enter the nucleus and exert a negative feedback effect,
inhibiting CLOCK/BMAL1 transactivating activity and ending CRY
and PER transcriptional activation. A secondary loop comprises
retinoid-related orphan receptor alpha (ROR
Multiple studies have shown that circadian rhythm regulates metabolism [13, 38]. Disruption of the molecular clock disrupts central and peripheral circadian rhythms and appears to be associated with metabolic syndrome. Disruption of the central circadian rhythms can cause melatonin levels to drop [31]. Melatonin, a hormone primarily produced by the pineal gland, is a vital regulator of the sleep-wake cycle and has significant influences on metabolic processes. By monitoring melatonin production, researchers found that central circadian disorders are closely related to obesity and insulin resistance [39, 40, 41]. These findings correlate circadian disruption with metabolic syndrome.
Disturbed circadian rhythms can also lead to unusual cell division, which is typical in cancer. Core clock genes appear to regulate MYC expression. c-MYC plays an important role in regulating cell proliferation. Huber et al. [8] found that CRY2 contains an E3 ligase component of FBXL3 that targets c-MYC for ubiquitination. CRY and FBXL3 synergistically bind to and degrade c-MYC [8]. CRY2 reduction results in increased c-MYC expression, which increases the probability of lymphoproliferative disorders. The International Agency for Research on Cancer (IARC) categorized shift employment as a class 2A carcinogen [42].
The parameters of the immune mediators in the blood are rhythmically consistent with the activity of the immune system [19]. BMAL1 increases monocyte recruitment and lowers pro-inflammatory cytokine levels [20]. The immune system is important in defying microbes and mediating hypersensitivity reactions. Understanding the daily rhythms of the immune system is of great significance for vaccination and prevention of allergic diseases.
The development of melanoma may be facilitated by the disruption of the
circadian rhythm [43]. The BMAL1 gene is closely associated with immune
response signaling pathways [44]. Alexander et al. [43] injected B16-F10
melanoma cells into Bmal1-knockout mice (M-BKO) and wild-type mice and
compared tumor growth rates. In Bmal1-knockout mice, the proportion of
CD8
The development of type 2 diabetes mellitus (T2DM) is associated with circadian
rhythm disruption [48]. In skeletal muscles, a peripheral circadian oscillator
significantly correlated with the development of T2DM exists [49, 50]. The
protein TBC1D1 is critical for the translocation of glucose transporter 4 (GLUT4)
from the cytoplasm to the plasma membrane [51]. Studies have shown that in
Bmal1-KO mice, a reduction in TBC1D1 expression is associated
with decreased Glut4 expression and translocation, as well as reduced
glucose tolerance. Similarly, the functions of the liver, an important organ in
maintaining euglycemia [52], are circadian. The hepatic metabolic genes are
important for the regulation of hepatic glucose uptake, storage, and production.
Rey et al. [53] found more than two thousand Bmal1-binding
sites in the liver. Jacobi et al. [54] demonstrated that knock-out of
Bmal1 in liver of mice caused oxidative stress, leading to an increase
in blood glucose. Liver-specific loss of Bmal1 in mice leads to the loss
of the circadian rhythm associated with the regulation of hepatic glucose
metabolism. Additionally, islet cells have an internal clock that coordinates
circadian oscillations in the transcriptome. CLOCK/BMAL1 heterodimer
binds to the key islet transcription factor, PDX-1, and recruits islet
cell-specific enhancers [55]. This process regulates the secretion of insulin.
The disruption of the
The clock also regulates various aspects of lipid and bile acid metabolism [56].
Dysregulation of de novo adipogenesis is one of the main causes of lipid
accumulation in the liver cells of patients with non-alcoholic fatty liver
disease (NAFLD). Knebel et al. [57] suggested that sterol regulatory
element binding protein-1c (SREBP1c) is a key transcription factor in de novo
lipogenesis (DNL). They constructed a mouse model of ALB-SrebP-1C by inserting
the transcriptionally active N-terminal domain of SREBP-1c into a vector
construct containing a mouse albumin promoter, a liver-specific enhancer element,
and a polyadenylation site. Compared with wild-type mice, ALB-SrebP-1C mice
showed a 50% increase in the level of fatty acids in their livers, and serum
analysis showed a two-fold increase in triglycerides and free fatty acids. In
addition, the expression of SREBP-1C and its target genes in the liver was shown
to increase the risk of fatty liver compared with healthy individuals. This
suggests that SREBP1c may induce mild fatty liver and obesity. Recent studies
have shown that SREBP1c and its target genes are regulated by the biological
clock mechanisms [58, 59, 60]. BMAL1, REV-ERB
Papagiannakopoulos et al. [63] exposed a genetically engineered mouse model (GEMM) infected with lung cancer cells to mice with either normal or jet-lag circadian rhythms. After eight weeks, the jet-lag mice had significantly larger tumors. This result suggests that circadian disturbances may promote the growth of lung adenomas and adenocarcinomas. In further studies, they found increased tumor load and higher cancer grade in animals with Per2 deficiency. To further investigate the effect of this gene dysfunction on tumor progression, Papagiannakopoulos et al. [63] used an alternative native Kras-driven GEMM and induced tumors to evaluate the effects of germline Per2 and Bmal1 mutations on tumor growth. They found an increased number of lung tumors in Kras-driven GEMM. These results suggest that the circadian genes Per2 and Bmal1 can inhibit the growth of lung tumors and that the deletion of circadian genes disrupts the circadian rhythm, melanoma thus increasing the incidence of lung tumors. In addition, they suggested that the loss of Per2 or Bmal1 independently leads to increased transcription of c-Myc, a protein that promotes tumor cell proliferation. They found that tumors with low Per2 levels were enriched in myc markers, suggesting that the deletion of Per2 increases c-Myc activity. Therefore, it is speculated that the core circadian genes Per2 and Bmal1 can inhibit lung tumor growth by inhibiting c-Myc transcription during lung tumor development.
Many epidemiological studies have shown that shift work is highly associated with breast cancer [1, 2, 3]. Nurses with long-term history of night shifts reportedly have a higher risk of breast cancer. These findings could be attributed to structural variations in PER [64, 65, 66, 67]. Variations in other clock control genes such as CRY and CLOCK are also associated with breast cancer [68]. Melatonin, secreted by the pineal gland, is an important cancer inhibitor. Tamarkin et al. [65] excised the pineal gland of rats and induced cancer using 9, 10-dimethyl-1, 2-benzothiophene. Melatonin treatment significantly decreased the incidence of breast cancer in those rats. Melatonin can also down-regulate the P38 pathway to inhibit the expression of Mmp-2 and Mmp-9, which inhibit the invasion of breast cancer cells [66]. These findings suggest that melatonin can be used to treat breast cancer.
In the absence of BMAL1, CLOCK increases and promotes the
phosphorylation of P65. P65 further binds to the NFKB2 promoter to boost
NFKB2 transcription. Therefore, NFKB2 is upregulated when
BMAL1 is absent. NFKB2 can upregulate P100 and P52 of
NF-
Above, we have summarized diseases that have been proved to be significantly
linked with circadian rhythms. In addition, many diseases are thought to be
related to circadian rhythms, but they are still lacking in-depth research.
Long-term follow-up has shown that people with disrupted circadian rhythms have
an increased risk of all-cause dementia [16, 17]. People with Parkinson’s disease
(PD) and Alzheimer’s disease (AD) frequently experience inconsistent sleep
patterns [69]. Postmortem neuropathological studies in humans have demonstrated
that amyloid-
The development of hypersensitivity diseases such as asthma is also affected by circadian rhythms [72, 73]. Histamine plays a key role in asthmatic bronchoconstriction. Cyclic epinephrine and cyclic adenosine monophosphate (cAMP) levels are positively correlated with each other throughout the circadian rhythm variations whereas plasma histamine levels are inversely correlated. The lowest level of circulating epinephrine occurs at 4 AM when asthma is most likely to develop. Clinical trials have demonstrated a significant decrease in histamine levels measured at 4 AM upon low-dose levoepinephrine injections.
Epidemiologic studies have found that shift work increases the risk of fracture [74], possibly by upsetting the balance between bone resorption and formation. Clock genes have been identified in osteoclasts, osteoblasts, and bone cells [75, 76]. Both bone resorption and production exhibit circadian cycles. However, the reason for this remains unclear. Recent studies on circadian rhythms at the molecular level have focused on cancer and metabolic syndromes. In this study, metabolic syndromes including glucose and lipid metabolism, which can lead to disorders such as diabetes, fatty liver, and atherosclerosis, were discussed along with cancers such as melanoma, lung cancer, and breast cancer. However, these studies were limited to correlations between circadian gene abnormalities and diseases, without establishing any causation. In addition, these studies tend to focus on the relationship between a single disease and circadian disruption and do not consider the inter-relatedness between diseases.
The fluctuation of disease severity in autoimmune disorders is thought to be
closely associated with circadian rhythms. Circadian rhythm regulatory factors
influence the expression of numerous immune-related genes, such as cytokines,
chemokines, receptors, and signaling molecules [77]. When the expression or
function of these circadian rhythm regulatory factors is disrupted, immune cell
dysfunction may occur. Consequently, this can lead to the onset and exacerbation
of autoimmune diseases. For example, rheumatoid arthritis (RA) and systemic lupus
erythematosus (SLE) are autoimmune diseases with symptoms known to be influenced
by circadian rhythms. In RA patients, joint pain, stiffness, and swelling
typically worsen in the morning and improve as the day progresses [78, 79]. SLE
patients often experience increased fatigue, joint pain, and other symptoms in
the morning [80]. Researches have shown that pro-inflammatory cytokines, such as
tumor necrosis factor-alpha (TNF-
| Category | Disease | Pathogenicity and Associated Clock Genes & Proteins | Study subjects | Year |
| Cancer | Melanoma | Bmal1 & Rora | Mice | 2020 [43] |
| Breast Cancer | PER | Mice & Human | 2017 [68] | |
| Lung Cancer | BMAL1, PER2 | Mice & Human | 2016 [63] | |
| Metabolic Syndrome | Type 2 Diabetes | BMAL1, CLOCK (muscle) | Mice & Human | 2016, 2014 [49, 50]; |
| BMAL1 (liver) | Mice & Human | 2011, 2015 [53, 54]; | ||
| Clock/Bmal1 heterodimer (islet) | Mice | 2015 [55] | ||
| Fatty Liver | BMAL1, Rora, Rorg & REV-ERB |
Mice & Human | 2020 [56] | |
| Atherosclerosis | BMAL1 | Human | 2020 [83] |
Some diseases can be treated and prevented by artificially altering the circadian rhythm. In addition to actively changing sleep patterns, circadian rhythms can also be altered via phototherapy and timed feeding.
Phototherapy, one of the potential treatment options, is a non-invasive, simple,
and well-tolerated intervention that can be used to treat a variety of
neurodegenerative diseases and mental disorders. Exposure to bright light rich in
blue wavelengths during the morning can stabilize circadian rhythms, eliciting a
robust alerting effect and facilitating quicker reaction time [84]. Additionally,
a study showed that the exposure to blue light in the evening may promote bipolar
disorders. Wearing blue-blocking glasses in the evening may aid in the treatment
of bipolar disorders [85]. However, this study involved only forty-three
participants, and larger clinical trials are needed to prove the reliability of
light therapy for mood disorders. Additionally, different patients had different
medication profiles, which may have affected the observed results. Bright or dark
red light during sleep relieves daytime sleepiness and tremors in patients with
Parkinson’s [86]. Additionally, increased daytime light exposure may improve
rhythm and help consolidate sleep [87]. Lack of bright light has been associated
with severe depression, bipolar disorder, seasonal affective disorder, and other
mood disorders [88, 89]. Phototherapy was helpful to solve the depressive
symptoms of the patients [90]. Ultraviolet (UV) exposure also causes the brain to produce
endorphins, which contributes to the feelings of pleasure [91]. This is
considered a treatment using photo-neuroendocrinology. By changing other light
settings, additional problems can be alleviated. Thus, the parameter selection
for phototherapy requires further research. Disruption of the master clock alters
the rhythm of A
The circadian system controls the metabolism of glucose, lipids, and energy by up- and down-regulating each one at various times of the day. The regular fasting period between supper and breakfast the following morning is extended by time-restricted feeding (TRF), a kind of intermittent fasting (IF) [92]. Unlike most forms of IF, TRF can be performed with or without reduction in calorie intake and weight loss [93]. Men with prediabetes showed improved insulin sensitivity, blood pressure, and reactive oxygen species levels without losing weight after a form of intermittent fasting called early time-restricted feeding (eTRF). In addition, exogenous melatonin supplementation can affect the SCN, and peripheral ciETRF may also help reduce lipids in atherosclerosis [94]. However, whether this can affect the expression of clock genes remains controversial [95, 96]. This can effectively reduce obesity, improve insulin resistance, and promote insulin secretion. However, there are still insufficient clinical trials on the prevention and treatment of T2DM with melatonin.
The use of circadian rhythms to select the appropriate timing of drug administration has attracted the attention of researchers in recent years. Current studies are limited to their empirical use in clinical trials. Chrono chemotherapy is used to select the appropriate time of administration according to the circadian rhythm of metabolism to achieve the best efficacy. Pharmacokinetics is used to determine the efficacy and toxicity of drugs at different times of the day. Currently, this method is used in the clinical treatment of hypertension, cancer, diabetes, and other diseases. However, treatment relies largely on the accuracy of choosing the right time to administer the drug. Monitoring the rate at which drugs are metabolized in the body might help us choose the right time to administer them, but this remains practically difficult. There are insufficient data to establish an effect. Immunotherapy is a new type of cancer therapy that uses drugs to stimulate the immune response of the body to kill tumor cells. The timing of immunotherapy using circadian rhythms is also an emerging area of cancer treatment that requires further research [97].
Currently, circadian rhythms are often used as an adjunct to disease treatment, and there are not enough clinical trials to prove their efficacy. As each patient’s condition is different, they are often treated with different drugs and dosages. It is difficult to control for these variables when monitoring efficacy. This makes it difficult to demonstrate the effects of circadian rhythm. In addition, the use of circadian rhythm adjuvant therapy is a long-term and difficult process that requires the joint efforts of doctors and patients. Patients may be reluctant to cooperate, and doctors may be more inclined to administer drugs in the traditional way. This makes it difficult to observe efficacy.
Most previous studies have focused on the dominance of the master clock over the peripheral clock. The optical signal is transmitted via the ipRGC to the SCN, which regulates the rhythm of the whole-body tissues. Few reports have addressed the role of melanocytes in circadian rhythms. We believe that melanocytes also have photoreceptor functions and play a role in regulating the peripheral rhythm. We have outlined the potential disorders caused by circadian rhythm disturbances and their underlying processes. Based on these findings, we presented the budding challenges that need to be addressed and proposed a vision for the future of phototherapy.
Hormone secretion and regulation play a critical role in maintaining the body’s normal functioning, and disruptions in circadian rhythms can lead to imbalances in hormone production, which in turn may contribute to the development of various diseases. In this article, we have summarized diseases related to circadian rhythms that have been extensively studied. Current research focuses on malignant and metabolic diseases related to circadian rhythms, such as melanoma, lung cancer, fatty liver disease, and diabetes. The cardiovascular and immune systems have been poorly studied in correlation with circadian rhythms; these require further research. In contrast to humans, mice are nocturnal animals. These results might be more accurate if other diurnal animals, such as dogs, were used for these studies. Using the influence of light on biorhythms could prevent and treat many other diseases in the future. However, there are insufficient clinical data so far to demonstrate its effectiveness. Melatonin is known to play a significant role in ameliorating the effects of circadian disruptions and can be used in the treatment of certain diseases. All these issues deserve further research in the future.
Researchers found that bright light affected the circadian rhythms of blind patients, suggesting that the peripheral body clock may be autonomously regulated. Melanocytes are widely distributed in the basal layer of the skin. As the body’s largest organ, the skin receives a large amount of light during the day. Melatonin secretion is intricately regulated by light, with exposure to light inhibiting its release, aligning the sleep-wake cycle with environmental light-dark cycles [98]. Sleep is a primary trigger for the release of hormones such as GH and PRL, which may have a variety of effects on the nervous and immune systems [99, 100, 101]. This opens up the whole new field called “photoneuroendocrinology”. Melanocytes can sense this phenomenon. If the ability of the skin to detect light is proven, phototherapy may even be used to treat some diseases of the blind.
The current study mainly focuses on two aspects. Firstly, it discusses the mechanisms of circadian rhythm regulation including macro and molecular mechanisms. Second, it elucidates the link between changes in the expression of rhythmic genes and the development of diseases. In conclusion, the connection between circadian rhythms and endocrinology is a crucial aspect of understanding the development and progression of various diseases. By further investigating the role of light exposure, hormone regulation, and circadian rhythms in endocrine function, researchers may uncover novel therapeutic approaches and preventative measures for a wide range of health issues. In the studies that we reviewed, this was done mainly by knocking out relevant genes in mice or by examining changes in gene expression in people with pre-existing diseases. However, when circadian rhythms are disordered, how does the body become ill? Are these diseases related to one another? There are no systematic answers to these questions. If we determine these mechanisms, gene therapy may be used to address the changes caused by circadian disruption, leading to innovative ideas for the treatment of many diseases.
Conceptualization, YWZ, MD, HZ and YML; acquisition, analysis and interpretation of data, MD; writing—original draft preparation, MD; writing—review, revision and discussing, MD, YWZ, HZ and YML; supervision and resources supply, YWZ and YML; funding acquisition, YWZ and YML. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We thank Dr. Li-Ping Liu for their supervision and resources availability, and Drs. Yan-Yan Chen, and Chen Tan for critical discussion on this article. Additionally, we would like to express sincere gratitude to Mrs. Ji-Yue Yan, Xiao-Qing Yu and Dr. Fan-Kai Liu for their invaluable support and contributions to this work.
This research was funded partly by the National Natural Science Foundation of China (82070638), Jiangsu Provincial Medical Key Discipline Cultivation Unit (JSDW202229), Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX23_3764), and the Science and Technology Planning Project of Guangdong Province of China (2021B1212040016).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

