1 Department of Chemistry, University of Swabi, 23561 Anbar, Khyber Pakhtunkhwa, Pakistan
2 Department of Chemistry, College of Science, Princess Nourah bint Abdulrahman University, 11671 Riyadh, Saudi Arabia
3 University Institute of Diet and Nutritional Sciences, Faculty of Allied Health Sciences, The University of Lahore, 54000 Lahore, Pakistan
4 Department of Nutrition and Dietetics, National University of Medical Sciences, 00666 Rawalpindi, Pakistan
5 Institute of Molecular Biology and Biotechnology, The University of Lahore, 54000 Lahore, Pakistan
6 Department of Pharmacy, Bacha Khan University, 24420 Charsadda, Khyber Pakhtunkhwa, Pakistan
7 Department of Botany, School of Basic and Applied Sciences, Shri Guru Ram Rai University, 248001 Dehradun, Uttarakhand, India
8 Department of Biomedical, Surgical and Dental Sciences, Università degli Studi di Milano, 20133 Milan, Italy
9 National Interuniversity Consortium of Materials Science and Technology [INSTM], 50121 Firenze, Italy
10 Department of Rasa Shastra and Bhaishajya Kalpana, Faculty of Ayurveda, Institute of Medical Sciences, Banaras Hindu University, 221005 Varanasi, Uttar Pradesh, India
Abstract
Background: The present study aimed to investigate the
in-vitro anti-diabetic, anti-cholinesterase, and anti-inflammatory
potential of extracts from different parts of Ficus benghalensis,
including leaves, stem, and roots, as well as isolated column fractions (F-B-1 C,
F-B-2 C, F-B-3 C, and F-B-4 C). Methods: The extracts and subsequent
fractions were evaluated for their inhibitory activity against key enzymes
involved in diabetes [
Keywords
- Ficus benghalensis
- α-amylase
- α-glucosidase
- AChE
- BChE
- COX-2
- 5-LOX
Nowadays, health professionals are seriously concerned regarding the escalating prevalence of diabetes mellitus in both developing and developed countries. World Health Organization (WHO) stats reveal that 30 million people were diagnosed with diabetes in the year 1985, which increased to 135 million by 1995. It has been further projected that nearly 300 million population will be suffering from diabetes by the year 2025 [1]. This rise in the prevalence of diabetes is causing substantial financial problems in healthcare settings of underdeveloped countries [2, 3]. There are two main categories of diabetes: type-I accounts for nearly 5 to 10% of cases, while type-II diabetes accounts for approximately 90% of cases [4]. Impaired secretion of insulin results in type-I diabetes, whereas insulin resistance causes type-II diabetes [5, 6]. Hyperglycemia is a state associated with uncontrolled diabetes that results in elevation of blood sugar levels. Prolonged hyperglycemic conditions may result in significant deterioration of different body organs, specifically blood vessels and the central nervous system. Presently, diversified synthetic drugs have been developed for the management of diabetes, such as meglitinides, biguanides, incretin mimetics, thiazolidinediones, sodium/glucose co-transporter 2 (SGLT2) inhibitors, and dipeptidyl peptidase-IV inhibitors [7]. Even though various developed anti-diabetic drugs have demonstrated significant results, they are associated with different side effects [8]. These adverse effects include visual complications, headache, diarrhea, impotency, and hypoglycemia [9]. Therefore, research and development of innovative anti-diabetic medications that have significant effects against diabetes along with the least adverse effects remains a vital opportunity for scientists [10, 11]. For this purpose, investigations on alternative natural medications have attained significant importance owing to their safe nature. Various folk medications, such as administration of different herbal plants and their resultant extracts, have been efficient alternatives to synthetic drugs in managing diabetes. Herbal plants have been a potential source of novel drug development, as nearly 90% of pharmaceuticals are directly or indirectly derived from them [12, 13].
Alzheimer’s disease (AD) is characterized as a neurodegenerative ailment known for its deteriorating effect on the central nervous system [14]. AD is related to different factors such as neuroinflammation, oxidative stress, aggregation of amyloids, and cholinergic deficiency [15, 16, 17, 18]. To date, the underlying origin and optimum treatment of AD is still unclear and unidentified, while managing AD might include targeting its associated conditions. Effective management of the progression of AD can be possible by significantly dealing with oxidative stress, cholinergic deficiency, aggregation of amyloids, and neuroinflammation [19]. Mostly, the treatments of AD focus on easing the symptoms associated with its progression rather than providing an ultimate cure. These therapies help in improving cognitive functionality, managing behavioral symptoms, and improving overall quality of life [19]. Even though these treatments are of significant benefits, continual research to hunt for a definitive cure is needed [20]. As a result, recent investigations focus on exploring improved and alternative therapies for effective treatment of neurodegenerative ailments. Acetylcholine-a, neurotransmitter that is consumed by cholinergic neurons via the central nervous system, plays a vital role in normal functionality [21]. However, acetylcholinesterase (AChE), an enzyme, is considered to be responsible for the reduction of acetylcholine (ACh), hence causing problems in neurotransmission. Recently, AChE inhibitors have been used as an effective alternative approach for the management of AD as they elevate the levels of acetylcholine in the brain [22]. Various investigations have shown the anti-cholinesterase potential of different plant extracts. In spite of recent advancements in the field of modern medication, plant constituents play a significant part in the health and well-being of the community. Pharmaceutical companies are increasingly interested in exploring higher plants as potential sources for discovering new lead compounds and developing effective drugs. This renewed interest highlights the potential therapeutic value of plant-based medicines in addressing various health conditions [23].
Inflammation is a complex biological process that plays a significant role in
the development and progression of various disorders, including arthritis and
cardiovascular disease [24]. The current approach for symptomatic treatment of
inflammation primarily relies on nonsteroidal anti-inflammatory drugs (NSAIDs),
which exert their anti-inflammatory effects by inhibiting cyclooxygenase (COX).
COX exists in two isoforms: cyclooxygenase-1 (COX-1), which is constitutively
expressed in most cells under normal conditions, and cyclooxygenase-2 (COX-2),
which is induced by pro-inflammatory agents like tumor necrosis factor-
Ficus benghalensis, a member of the Moraceae family, is widely
recognized as the Banyan tree in English. It is native to a vast region of Asia.
This plant is grown in several botanical gardens throughout different tropical
areas across the world [29]. Studies have been performed to assess the
phytochemistry and health-promoting benefits of F. benghalensis.
Nevertheless, very few investigations have been conducted on leaves of F.
benghalensis, which is of great importance owing to its medicinal applications
[30]. F. benghalensis was reported to own different health-promoting
benefits such as antibacterial [31], anti-diabetic, anticancer, and
anti-inflammatory [32]. Moreover, it has demonstrated beneficial effects against
skin ailments, gastric ulcers, and other gastrointestinal problems [33].
F. benghalensis methanolic extract was found to be effective against
inhibiting acetylcholinesterase activity [21]. The composition of the leaves of
F. benghalensis reveals the presence of crude protein (9.63%), Crude
fiber (2.53%), phosphorous (0.4%), and calcium oxalate (2.53%). Phytochemical
screening of leaves shows the presence of tannins, sterols, phenolic acids,
saponins, and flavonoids. On the other hand, compounds like triterpenoids,
volatile oils, and aromatic acids were not present in this plant [30]. In
Ayurveda medications, this plant has been consumed for treating piles, dysentery,
and diarrhea. Additionally, it is also employed as a hypoglycemic agent,
indicating its potential in managing blood sugar levels [34]. Different extracts
of Ficus bengalensis were assessed for their potential anti-allergic and
anti-stress effects in asthma using milk-induced leucocytosis and milk-induced
eosinophilia as indicators [35]. Literature reveals the potential of different
Ficus species as anti-diabetic, antimicrobial, anticancer, and anti-inflammatory
activities [36, 37]. The current study is unique as we have compared the
in-vitro anti-diabetic, anti-cholinesterase, and anti-inflammatory
potential of extracts and sub-fractions of different parts of F.
bengalensis. In light of this, current study was designed to evaluate the
in-vitro anti-diabetic [
In June 2022, plant samples of F. benghalensis were gathered from Anbar Swabi. The collected specimen was taken to the Department of Botany, where it was examined and identified by Dr. Muhammad Ilyas, a member of the Botany Department at the University of Swabi. The voucher specimen number UOS-BOT/103 was then placed in the herbarium of the aforementioned department.
All information about software/equipment/drugs/reagents can be found in Supplementary Material and article. After collecting the plant material, consisting of stems, leaves, and roots, each weighing 2.2 kg, they were dried in the shade. Subsequently, the dried plant material was subjected to extraction using methanol. The obtained extracts were concentrated using a rotary evaporator (Rotavapor® R-300, BUCHI Corporation, New Castle, DE, USA), resulting in crude extracts of 18.00 g for the stems, 14.76 g for the leaves, and 16.1 g for the roots. Thin Layer Chromatography (TLC) profiling revealed that stem extract possessed the highest number of constituents, which were subjected to chromatographic analysis yielding different sub-fractions like F-B-1 C: CC chloroform: methanol (7:3); F-B-2 C: (chloroform: methanol (7.5:3.5); F-B-3 C: chloroform: methanol (6:4) and F-B-4 C: chloroform: methanol (6.5:3.5). The extracts and sub-fractions obtained were carefully stored in a freezer to maintain their integrity and preserve their biological activity. Later, these stored samples were utilized for various biological studies and investigations.
An investigation was conducted to examine the impact of extracts and column
semi-purified fractions of F. benghalensis on
The
Here: OD (control) = absorbance of control, while OD (sample) = absorbance of extracts/fractions (AbsC: Absorption of control; AbsS: Absorption of sample).
The anti-cholinesterase activity of both the extracts and column semi-purified
fractions of F. benghalensis was assessed using the Ellman method [40].
In summary, the cholinesterase enzyme breaks down the acetyl thiocholine
substrate, resulting in the formation of thiocholine. Thiocholine then reacts
with Ellman’s reagent (DTNB), generating 2-nitrobenzoic-5-mercaptothiocholine
(thiocholine-thionitrobenzoate disulfide) and 5-thio-2-nitrobenzoic acid
(thionitrobenzoate). These reaction products can be detected at a wavelength of
405 nm. For this assay, the reaction mixture had a total volume of 1 mL. Within a
1 mL cuvette, 50 µL of 3.5 mmol L
In this study, an in-vitro assay was performed to assess the anti-inflammatory effectiveness of extracts and column semi-purified fractions derived from F. benghalensis. In this study, the evaluation of COX-2 inhibitory potential was conducted using the methods outlined by Jan et al. [41] in 2020. Initially, 300 U/mL of COX-2 enzyme was prepared, from which 10 mL of COX-2 solution was ice-cooled for activation of enzymatic activity. To activate the enzymatic activity, the whole procedure was carried out for 5 to 10 minutes. Moreover, a co-factor solution (hematin: 1 mM, N,N,N,N-tetramethyl-p-phenylenediamine (TMPD): 0.24 mM, and glutathione: 0.9 mM) containing 50 mL that was prepared in Tris-HCl buffer (pH: 8, 0.1M) was mixed with this activated enzymatic solution. Afterwards the plant extracts and fractions were mixed with enzyme solution at varied concentrations (62.5–1000 µg/mL). Later, this mixture was incubated at 25 °C for 5 minutes. To initiate the reaction, 20 mL of a 30 mM arachidonic acid solution was introduced to the mixture. The resulting solution was then incubated for a duration of 4–5 minutes. In this assay, celecoxib was employed as a reference drug to enable comparison with the tested samples. Finally, absorbance was noticed at 540 nm wavelength using a UV-vis spectrophotometer.
Here: AbsC = absorbance of control, while AbsS = absorbance of sample [extract/fractions].
Additionally, this study included a 5-LOX (5-lipoxygenase) assay to assess the
in-vitro anti-inflammatory activity of extracts and column semi-purified
fractions obtained from F. benghalensis [42]. For the 5-LOX assay,
different doses (ranging from 62.5 to 1000 µg/mL) of extracts and
fractions derived from F. benghalensis were prepared. Subsequently, a
solution of the 5-LOX enzyme was prepared with a concentration of 10,000 units
per milliliter. To initiate the enzymatic reaction, an 80 mM linoleic acid
(L1376-5G, Sigma-Aldrich, St. Louis, MO, USA) substrate was added to the
solution. To create the desired reaction mixture, 250 µL of crude
extracts were combined with a phosphate buffer (50 mM, pH 6.3). Next, an enzyme
solution of 250 mL was introduced to the mixture, and the resulting mixture was
incubated for a duration of 5 minutes. Following the incubation, the substrate
solution was added to the enzyme mixture and thoroughly mixed together. The
absorbance (Abs) of both the control and test samples was measured at a
wavelength of 234 nm using a UV-Vis spectrophotometer. A graph was constructed to
examine the correlation between different extract concentrations and the extent
of enzyme inhibition. This analysis allowed for the calculation of the IC
Here: AbsC = absorbance of control, while AbsS = absorbance of sample (extract/fractions).
The results in this study were expressed as mean
Supplementary Table 1 shows the results of the anti-diabetic activity of
extracts from different parts and isolated column fractions of F. benghalensis. At 1000 µg/mL, the highest
In this study, extracts of F. benghalensis roots, leaves, and stem,
along with derived fractions (F-B-1 C, F-B-2 C, F-B-3 C, and F-B-4 C) were
analyzed for their potential anti-cholinesterase activity. Glutamine was used in
this study as a reference standard drug. Results revealed a significant reduction
in activity of AChE and BChE in a concentration-dependent manner (Supplementary
Table 2). It is evident from Supplementary Table 2 that extracts of roots,
leaves, and stems demonstrated potential inhibition of AChE and BChE with
IC
The results of the anti-inflammatory activity of extracts from different parts
and isolated column fractions F. benghalensis against COX-2. At 1000
µg/mL, the highest COX-2 inhibitory potential was demonstrated by F-B-3 C
(85.72%), followed by F-B-1 C (83.13%), F. benghalensis stem extract
(80.85%), F. benghalensis leaves extract [79.00%], F-B-2 C (73.08%),
and F. benghalensis roots extract (69.47%). In the case of 5-LOX
inhibitory activity, F-B-1 C showed maximum (87.63%) potential, while F. benghalensis root extract had minimum inhibitory activity (73.39%). The
standard (Montelukast and Celecoxib) used in this study showed a percent
reduction of 95.20 and 93.55% in the activity of COX-2 and 5-LOX, respectively
as shown in Figs. 1,2. The IC
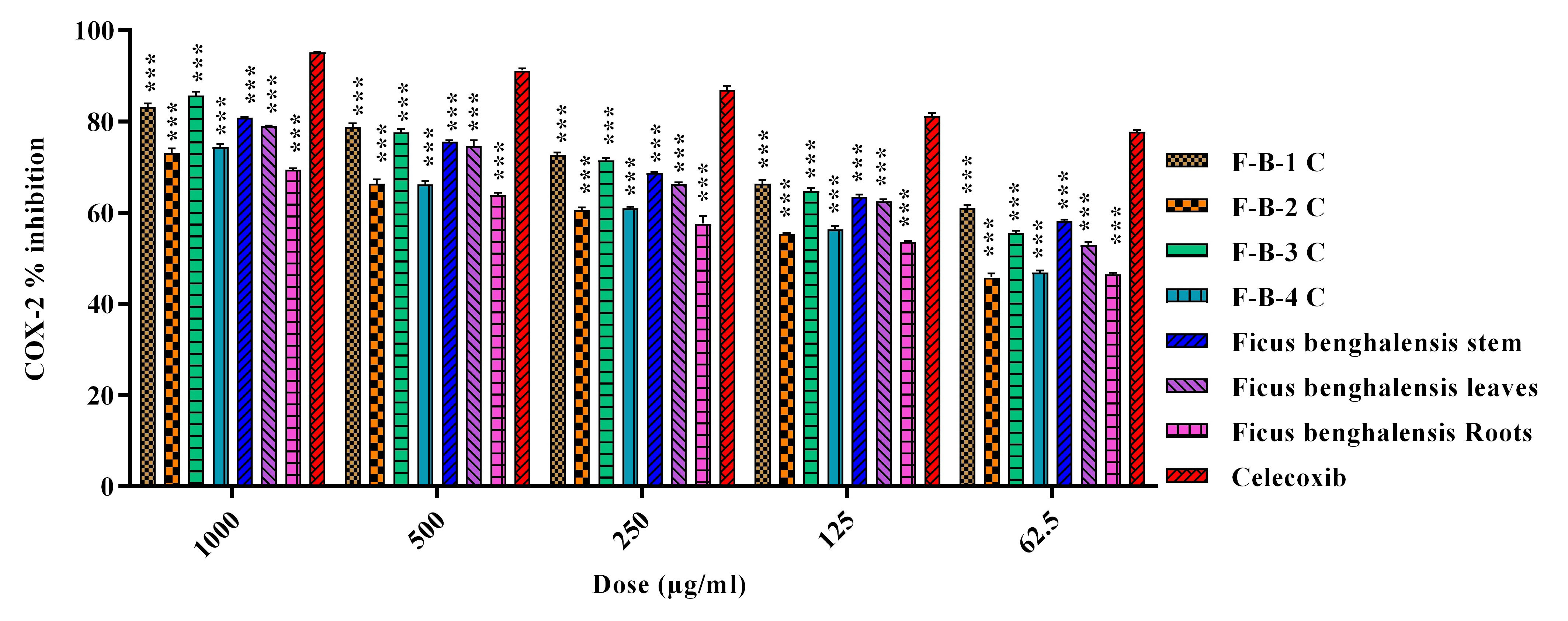 Fig. 1.
Fig. 1.Percent COX-2 inhibition activity of crude
extracts/fractions and isolated column fractions of Ficus benghalensis.
Data is represented as mean
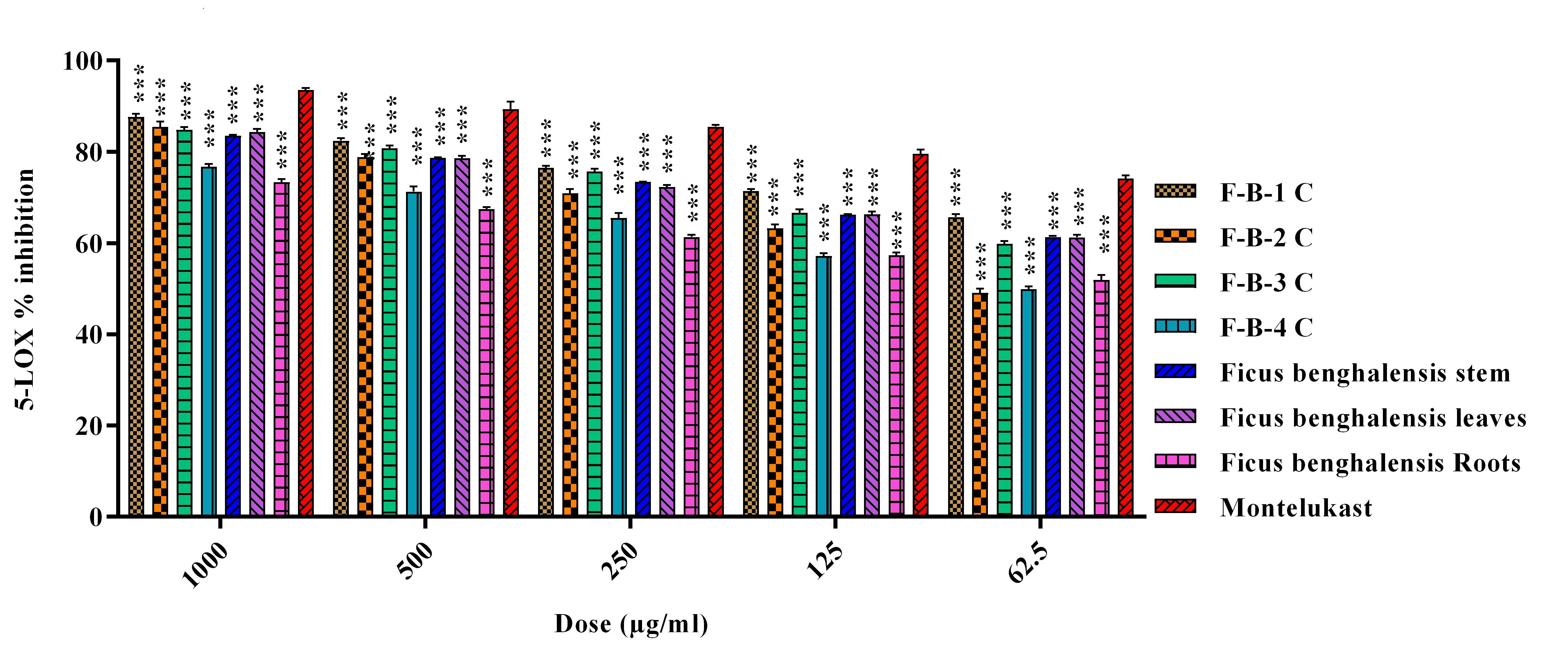 Fig. 2.
Fig. 2.Percent 5-LOX inhibition of crude extracts/fractions and
isolated column fractions of Ficus benghalensis. Data is represented as
mean
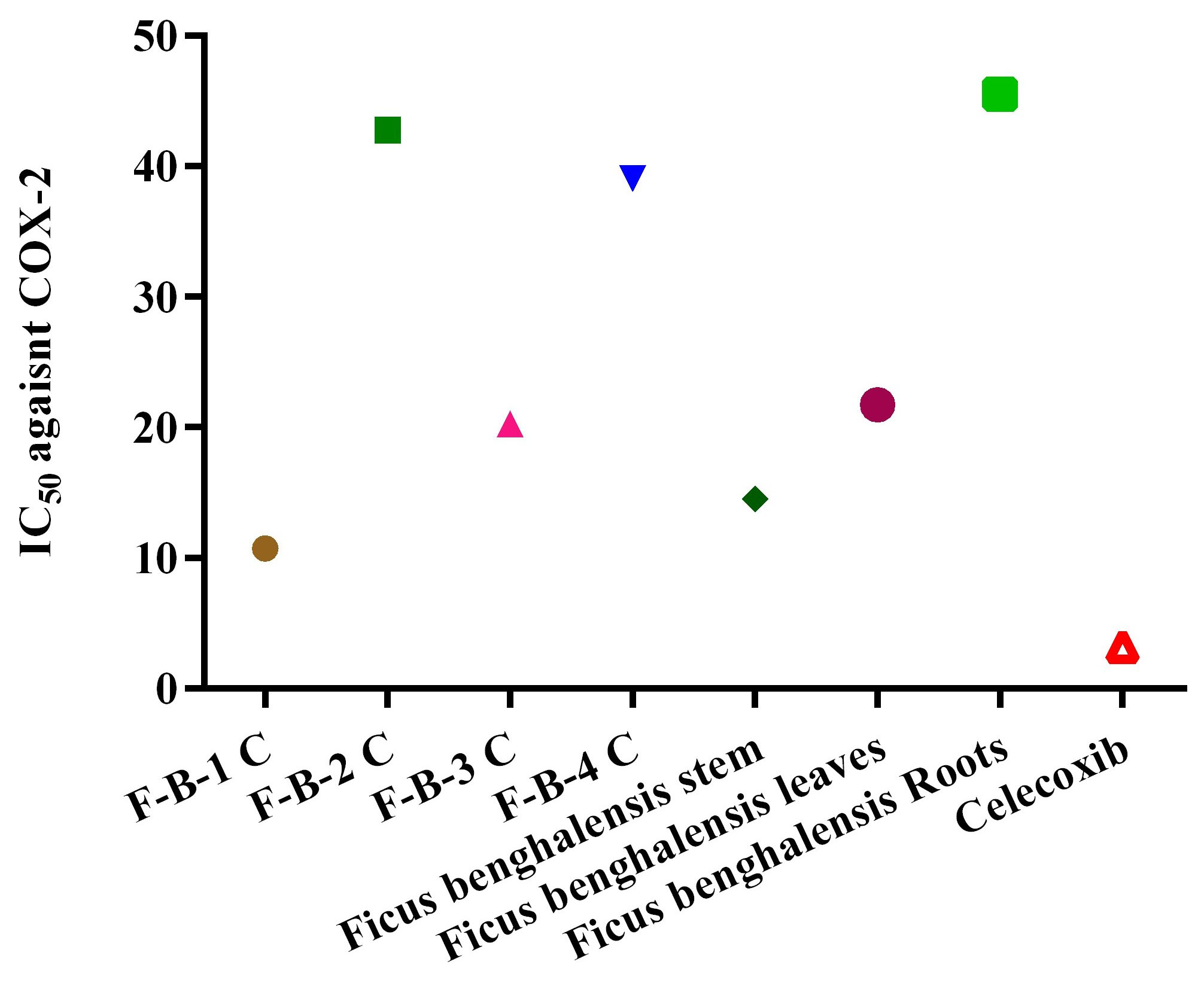 Fig. 3.
Fig. 3.The IC
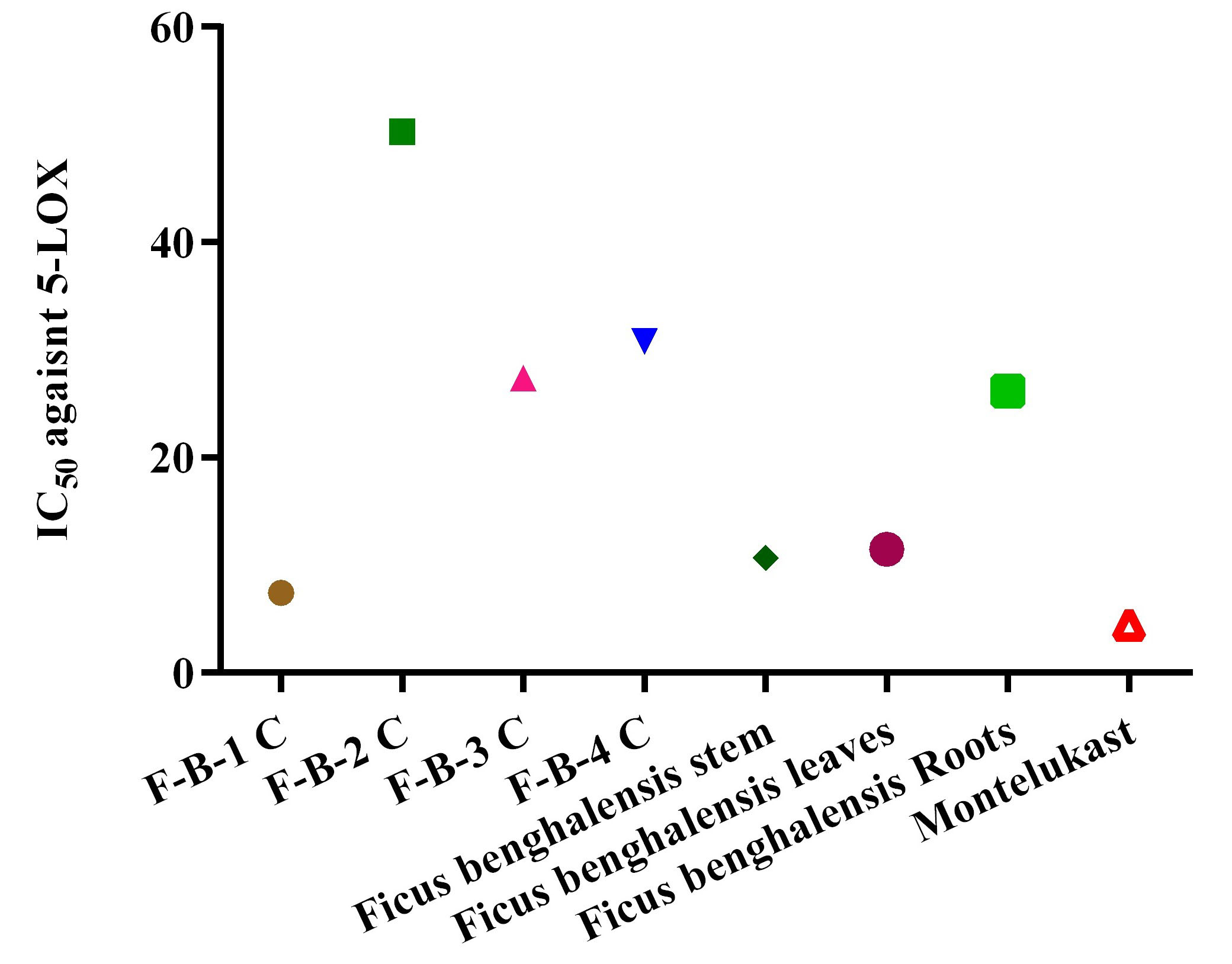 Fig. 4.
Fig. 4.The IC
The global population’s reliance on plant-based remedies is on the rise,
primarily attributed to their easy accessibility and affordability [43, 44].
Consequently, researchers are persistently engaged in the pursuit of significant
natural-based remedies that can be employed in the treatment, diagnosis,
mitigation, or prevention of various disorders [45]. Validating the therapeutic
potential of these readily accessible natural products holds the promise of
offering more cost-effective treatment alternatives, particularly in light of the
prevailing high inflation rates globally. It is worth noting that the
pharmacodynamics of many drugs are associated with the inhibition of enzymes
present in various biological compartments. Local physicians have recognized the
therapeutic properties of the Ficus plant. The local physicians have long
recognized the therapeutic properties of various parts of the F.
benghalensis plant. The milky fluid obtained from the plant is known for its
external application in alleviating pain, rheumatism, bruises, backaches, and
swollen soles of the feet. In India, the roots of F. benghalensis are
traditionally used to treat conditions such as dysentery, biliousness, gonorrhea,
and liver swelling. Additionally, the aerial roots and tips of the plant are
employed for their medicinal benefits in the treatment of dysentery and vomiting
[46]. The infusion of small branches is consumed to alleviate hemoptysis, while
the bark is believed to possess potent tonic properties and is used as a cure for
diabetes [47]. Different components of F. benghalensis are employed in
the treatment of diarrhea, leucorrhea, wound healing, and skin diseases [48].
According to folkloric practices, the aerial parts of F. benghalensis
are employed to alleviate persistent vomiting and as an anti-asthmatic remedy
[49]. Additionally, F. benghalensis leaves and stems have been
documented for their therapeutic applications in various ailments [50]. However,
there is a limited exploration of the phytochemical composition of this plant.
Some compounds identified in its leaves include
The present study was performed to assess the anti-cholinesterase,
anti-inflammatory, and anti-diabetic properties of extracts/fractions of
F. benghalensis. In-vitro anti-diabetic assay revealed a
significant [p
In this study, the extracts and fractions of F. benghalensis were also
examined for their potent anti-inflammatory properties. These two assays, namely
COX-2 and 5-LOX inhibitory assays, were performed in this study. Results of both
assays demonstrated a significant [p
Phytochemical profiling of F. benghalensis extracts have indicated the
presence of diverse phytomolecules like steroids, tannins, flavonoids, alkaloids,
anthraquinones, and glycosides [63]. Roots extract of F. benghalensis
inhibited the activity of acetylcholinesterase in an in-vitro model. The
ethyl acetate extract of roots showed inhibitory action with an IC
The findings of this study revealed the in-vitro potential of Ficus benghalensis extracts and fractions in various pharmacological activities. The leaf extract exhibited the highest alpha-glucosidase and alpha-amylase inhibitory activities among all the tested samples, indicating its potential as an anti-diabetic agent. Furthermore, the roots, leaves, and stem extracts, as well as the derived fractions, exhibited significant anti-cholinesterase activity, highlighting their potential for the management of neurodegenerative diseases. Additionally, the F-B-3 C fraction showed the highest COX-2 inhibitory activity, suggesting its potential as an anti-inflammatory agent. Overall, this study provides valuable insights into the medicinal potential of F. benghalensis and supports its traditional use in the treatment of diabetes, neurodegenerative diseases, and inflammatory conditions. Further studies are warranted to identify and characterize the active compounds responsible for these observed bioactivities and to evaluate their in-vivo efficacy and safety profiles.
The data generated in the present study are included in the figures and/or tables of this article.
AR was responsible for conceptualization, visualization, supervision, project administration and writing-original draft. MIbr and MIri contributed by conducting formal analysis, interpreting data, investigation and writing-original draft. AAK, NA, TSA, TK and MUK contributed by writing-original draft, analyzing, data curation, reviewing and editing of manuscript. KB, MSJ, and RS made significant revisions, visualization, and editorial changes. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Plant samples of F. benghalensis were gathered from Anbar Swabi. The collected specimen was taken to the Department of Botany, where it was examined and identified by Dr. Muhammad Ilyas, a member of the Botany Department at the University of Swabi. The voucher specimen number UOS-BOT/103 was then placed in the herbarium of the aforementioned department.
Authors would like to acknowledge Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia for supporting this study.
Princess Nourah bint Abdulrahman University Researchers Supporting Project number [PNURSP2024R18], Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
The authors declare no conflict of interest. Given his role as Guest Editor and Editorial Board Member of Frontiers in Bioscience-Landmark, Marcello Iriti had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Jen-Tsung Chen.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




