1. Introduction
Alzheimer’s disease (AD), the most common cause of dementia, is a progressive
neurodegenerative disease characterized by continuous cognitive decline. The
extracellular neuritic plaques with -amyloid protein (A), as
the major component, is the hallmark and earliest neuropathology of AD [1]. Lower
A throughout life is associated with a longer life span without
cognitive decline [2].
A is produced by sequential - and -cleavage of the
amyloid precursor protein (APP). APP is a type I transmembrane protein containing
a large extracellular domain (or ectodomain), a hydrophobic transmembrane domain,
and a small intracellular domain. In the amyloidogenic pathway, APP is first
cleaved at the -site close to the end of the ectodomain by
-secretases to generate a secreted N-terminal fragment (sAPP)
and the C-terminal fragment C99, and C99 is further cleaved by
-secretase within the transmembrane domain to shed A. The
-cleavage of APP producing A1-x is considered to be the
rate-limiting step in this process, and beta-secretase 1 (BACE1), the major
-secretase cleaving APP at the -site is indispensable for
neuritic plaque formation in vivo [3]. The -secretase consists
of four major subunits: presenilin-1/2, anterior pharynx-defective 1 (APH-1),
presenilin enhancer 2 (PEN2), and nicastrin (NCT) [4]. In the
-secretase protein complex, presenilin-1/2 are the catalytic subunit,
and NCT is required for assembly of the -secretase and substrate
priming [5, 6].
In the non-amyloidogenic pathway, APP is cleaved by -secretases at the
-site to generate sAPP and a C-terminal fragment comprising
the last 83 residues of APP (C83), or by the -secretase BACE2 to
produce the C-terminal fragment C80 [7]. C83 and C80 shed truncated A
after -cleavage [8].
Normally, -secretase does not directly cleave its substrates before
truncation of the ectodomain of the substrates by other enzymes [9]. Such a rule
also applies to APP. The - and -secretases first remove the
large majority of the ectodomain of APP before -secretase can cleave
the transmembrane domain of the resulting C-terminal fragment. However, some
-secretase substrates such as cluster of differentiation 269 (CD269,
also known as B-cell maturation antigen or tumor necrosis factor receptor
superfamily, member 17) on the surface of plasma cells and amyloid-like protein 1
(APLP1), an APP-like protein, can be directly cleaved by -secretase.
CD269 contains an ectodomain of only 57 residues and APLP1 has a non-canonic
transmembrane to allow direct -cleavage [10, 11]. It is worth noting
that most, if not all studies demonstrating the requirement of removing the large
ectodomain of APP before -cleavage have been performed in cell-free
systems. Nonetheless, the intracellular processing of APP could be more
complicated and dependent on a number of factors such as the location and
post-translational modifications of APP and the secretases, the
microenvironments, binding proteins, changing pH, and ion strengths [12, 13]. None
of these factors could be explicitly re-established in cell-free experiments.
While - and -secretases are crucial for A generation
and AD pathogenesis, they are also required for many fundamental biological
functions [14, 15]. As an example, Notch1 is one of the best characterized
-secretase substrates, playing essential roles in cell proliferation,
cell fate, differentiation, cell death, immune defense, neurogenesis, and
neuronal activities [16, 17, 18, 19, 20]. Notch1 is a receptor at the cell surface. After
final cleavage by -secretase, the cytoplasmic domain of Notch1 is
released into the nucleus as a transcriptional regulator. Inhibiting
-secretase abolishes Notch1 signaling, leading to adverse effects [21].
All clinical trials targeting - and -secretases for AD therapy
have been halted, mainly because of the adverse side effects [22, 23].
Some interesting questions are as follows. (1) Direct cleavage of APP by the
-secretase has never been observed. If it can be achieved by artificial
means, can the resulting N-terminal fragment (dubbed sAPP) containing
the entire A sequence be further cleaved by -secretases and
shed A? (2) Is there a sequence in the ectodomain of APP that inhibits
the -cleavage of APP? If so, methods could be developed to target this
sequence and induce direct -cleavage of full-length APP, thereby
reducing A production without inhibiting any enzyme.
To address these questions, this study showed that sAPP is not a
favored substrate of -secretase and therefore, the strategy of enabling
-cleavage of full-length APP is valid for A reduction.
However, -cleavage of full-length APP is not inhibited by a specific
sequence in the ectodomain of APP; instead, the large size of the ectodomain
abrogates this cleavage. Even green fluorescent protein (GFP), a protein
irrelevant to APP, impedes the -cleavage of C99 when fused to the
N-terminus of C99. Mechanistically, GFP fused to the N-terminus of C99 prevents
the binding of C99 with NCT, a subunit of the -secretase, suggesting a
spatial effect impairing the accessibility of -secretase to C99.
2. Materials and Methods
2.1 Plasmid Construction
Plasmid pcDNA4-mic-hisA was used as the expression vector, cDNA coding for
APP was used as the template, and the fragments of interest were
amplified by polymerase chain reaction (PCR). The signal peptide of APP was added to the N-termini of the
truncated APP fragments by PCR. The coding sequence for the FLAG tag was inserted
behind the signal peptide of APP or at the end of the C-terminal of C99 to
generate plasmids expressing N- or C-terminal FLAG-tagged protein. For
endoplasmic reticulum (ER) retention, the last four residues Glutamine-Methionine-Glutamine-Asparagine (QMQN) of the APP
C-terminal fragments were replaced by Lysine-Lysine-Glutamine-Asparagine (KKQN).
2.2 Cell Culture and Transfection
HEK293 cells purchased from American Type Culture Collection (CRL-1573;
Manassas, VA, USA) were cultured in high-glucose Dulbecco’s Modified Eagle Medium
(Servicebio, Wuhan, China) containing 10% fetal bovine serum (ProCell Therapies,
New York, NY, USA), 1 mM sodium pyruvate, and 4 mM L-glutamine at 37 °C,
5% CO. The plasmids were transfected into cells using polyethylenimine
according to the manufacturer’s instructions. After culturing for 24 h, cells
were treated with or without the -secretase inhibitor L685458
(MedChemExpress, Monmouth Junction, NY, USA) for 3 h before cell lysis. The BACE1
inhibitor MK-8931 (MedChemExpress) was also introduced for 3 h before cell lysis. Authentication of cell lines was performed by the company (Procell, Wuhan, Hubei Province, China). We performed mycoplasma test every 20 days using the MycoBlue Mycoplasma Detector (Vazyme, Nanjing, Jiangsu Province, China).
2.3 Primary Mouse Cortical Neuron Cultures
E18 cortical neurons of the APP Swedish mutant transgenic mouse (Stock
No. C001076) were prepared as previously described. Briefly, the mouse cortices
were dissected out, and the meninges were completely removed under a dissection
microscope. The cortices were pooled in a 15 mL tube and digested with trypsin at
37 °C with gentle rotation for 20 min. Then the digestion solution was
removed and cells were dissociated by pipetting with inactivation solution
(Minimum Essential Medium containing 0.6% D-[+]-glucose, 1 mM pyruvate, 10%
horse serum, 2.5% bovine serum albumin (BSA), and 2.5% trypsin inhibitor).
Cells were cultured in neurobasal medium supplemented with B27 in
poly-D-lysine-coated plates. The culture medium was replaced every 3 days.
Plasmid transfection was performed using Lipofectamine 8000 (Beyotime, Beijing,
China) according to the manufacturer’s instructions.
2.4 Western Blot Analysis
The cell lysates were resolved by sodium dodecyl sulfate (SDS) polyacrylamide
gel electrophoresis, and the proteins were electrotransferred to nitrocellulose
membranes. The membranes were blocked in phosphate-buffered saline containing 3%
BSA for at least 1 h. The primary antibody used to detect APP and its C-terminal
fragments was C20 (a rabbit polyclonal antibody reacting with the last 20 amino
acids of APP, in-house generated, see [7]). Anti-FLAG antibody (Sigma, St. Louis,
MO, USA) was used to detect the FLAG tag, and anti-BACE antibody (Cell Signaling
Technology, Danvers, MA, USA) was used to detect BACE1. For A detection,
82E1 (Immune-Biological Laboratories, Inc., Minneapolis, MN, USA) and 6E10
(BioLegend, San Diego, CA, USA) primary antibodies specifically reacting with
A were used. The protein bands were quantified using ImageJ-2 (National
Institutes of Health, Bethesda, MD, USA) software.
2.5 Co-Immunoprecipitation
Cells were lysed in co-immunoprecipitation (co-IP) buffer (50 mM HEPES, pH 7.4,
250 mM NaCl, 0.5% Triton X-100) supplemented with protease inhibitors. The
lysates were cleared by centrifugation at 15,000 g for 20 min, and added to
anti-FLAG magnetic beads (Sigma). The reaction was incubated overnight at 4
°C with rotation. After three washes with co-IP buffer, the precipitated
proteins were eluted using 2 SDS sample buffer and boiled for 5 min.
2.6 Immunofluorescence
At 24 h after transfection, cells were fixed in 4% paraformaldehyde and
permeabilized in 0.2% Triton X-100. After blocking in 5% BSA, cells were
stained with C20 antibody for APP or the fragments and GM130 (BD Biosciences,
Franklin Lakes, NJ, USA) for the Golgi. The primary antibodies were detected with
Alexa 568 and Alexa 488 secondary antibodies (Abcam, Cambridge, MA, USA). DNA was
stained with 46-diamidino-2-phenylindole (DAPI). Images were taken using the
BZ-X810 fluorescence microscope (Keyence, Chicago, IL, USA).
2.7 Statistical Analyses
Statistical analyses were performed using GraphPad Prism 8 (GraphPad Software,
San Diego, CA, USA). Results are expressed as the mean standard
deviation. The two-tailed t-test was used to analyze the difference
between two groups. p 0.05 was considered statistically significant.
3. Results
3.1 -Secretase Inefficiently Cleaves sAPP
To determine if sAPP can be further cleaved by -secretase to
produce A, we overexpressed in HEK293 cells an APP fragment spanning
residues 1 to 636 (as in the APP isoform, 636 is the end of
A) to mimic sAPP (Fig. 1a). To enhance
-cleavage for clear A detection, residue substitutions by the
Swedish mutation (K594N/M595L, as in APP) were introduced into this
protein to enhance A production. For the detection of APP N-terminal
fragments in the conditioned medium, a FLAG tag was inserted after the signal
peptide of APP. Compared to full-length APP containing the Swedish mutations
(APPswe), the expression levels of sAPP in cells and in conditioned
media (detected using anti-FLAG antibody) were similar to those of APPswe (APPswe
is cleaved by - or -secretases, and the resulting N-terminal
fragments sAPP and sAPP are secreted), but A
production from sAPP was below the detectable level. By contrast,
A produced from APPswe by endogenous - and
-secretases was clearly detected (Fig. 1b,c). To further confirm that
sAPP is not a -secretase substrate, we co-expressed BACE1 with
APPswe or sAPP in HEK293 cells, and treated the cells with or without
the BACE inhibitor MK-8931. If sAPP can be weakly cleaved by
-secretase, the overexpression of BACE1 may enhance the cleavage. Upon
-secretase inhibition, the protein levels of full-length APPswe were
greatly increased (p = 0.0001) and the N-terminal fragment sAPP
produced by BACE1 cleavage in cells was abolished, both of which indicated that
-secretase was successfully inhibited. Because of the short duration of
-secretase inhibition, the extracellular sAPP in the
conditioned media was unaffected (Fig. 1d). By contrast, the overexpressed
sAPP displayed no difference with or without MK-8931 treatment in both
the cells and conditioned media (Fig. 1d). Hence, although having never been
observed, sAPP, if it exists, is not a favored substrate of endogenous
-secretase. Inducing direct -cleavage of full-length APP
before -cleavage is a valid strategy to reduce A production
without affecting these secretases. A method to counter the mechanism by which
full-length APP suppresses -cleavage would benefit AD prevention and
therapy.
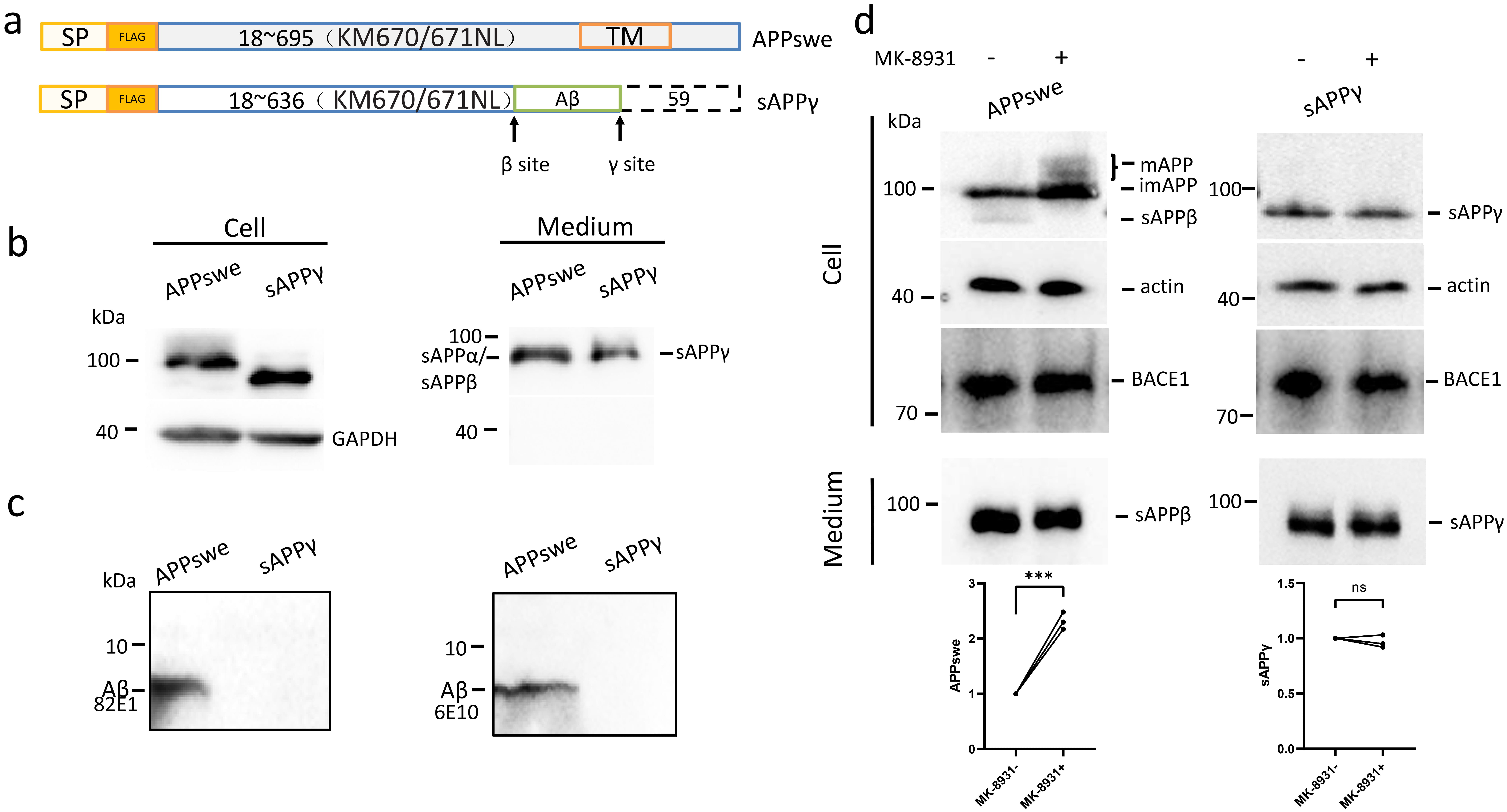 Fig. 1.
Fig. 1.
sAPP does not produce A. (a) Schematic
diagram comparing the difference of full-length APP (APPswe) and sAPP.
The box with dashed lines indicates that the last 59 residues in full-length APP
is absent in sAPP. The arrows indicated the cleavage sites that APP
been cleaved for A production. SP: signal peptide of APP to guide APP or
sAPP into the secretion pathway (residues 1–17); FLAG: FLAG tag
inserted after the signal peptide; KM670/671NL: substitution by Swedish
mutations; TM: transmembrane domain. (b) Expression of APPswe and sAPP
in HEK293 cells, and sAPP and sAPP derived from APPswe by
- and -cleavages, respectively and directly overexpressed
sAPP in conditioned media. (c) A in the conditioned media of
cells expressing APPswe and sAPP were blotted using both the 82E1
antibody (to detect the N-terminus of C99 and A with a primary amine)
and the 6E10 antibody (N-terminus of A as the epitope). (d) APPswe or
sAPP, was co-expressed with BACE1 in HEK293 cells, and full-length
APPswe, sAPP derived from APPswe by -cleavage, and
sAPP in cells and in conditioned media were detected by Western
blotting. The relative amounts of protein bands with or without MK-8931 treatment
were plotted. The numbers represent the mean standard deviation
(***p 0.001, ns: non-significant). n = 3 or more independent
experiments for all figures. sAPP, soluble amyloid precursor protein ; A, amyloid- peptide; APP, amyloid precursor protein.
3.2 -Secretase does not Cleave Full-Length APP in Primary
Neurons
Previous cell-free studies have suggested that -secretase does not
cleave APP because of the large ectodomain of APP. However, APP is a protein
destined to the secretion pathway, and its cleavage is closely regulated by the
changing intraorganellar environments that cannot be explicitly re-established in
cell-free systems. For example, we previously reported that nascent APP in the ER
is not efficiently cleaved by - or -secretases, and the
-cleavage of C99 in the ER is inefficient because the
-secretase in the ER is inactive. By contrast, in the Golgi apparatus
and later compartments along the secretion pathway, the cleavages of APP and C99
are activated [13, 24]. Moreover, APP is a type I transmembrane protein with the
ectodomain either inside the lumen of organelles (including ER, the Golgi, and
vesicles for transport) during anterograde transport to the plasma membrane or in
the extracellular space when APP is present on the plasma membrane. Like most
proteins in the canonic secretion pathway, APP is synthesized on ER surface and
translocates into the ER lumen via its signal peptide to enter the secretion
pathway. The transmembrane domain of APP stops the translocation, which leaves a
short intracellular cytoplasmic domain (APP intracellular domain [AICD]) in the
cytoplasm. After entry into the ER, APP is further transported through the Golgi
for further modification (APP maturation), and then to the plasma membrane, from
where APP is quickly internalized back through the endosomes to the Golgi or to
the lysosome for degradation [12]. Along the secretion pathway, the luminal pH of
the organelles gradually decreases, and it is unclear how the changing pH affects
APP processing. To test in cells if -secretase directly cleaves
full-length APP, we treated the primary cortical neurons of the APP
Swedish mutant transgenic mice (PN) with the -secretase
inhibitor L685458 for 3 h. Western blotting using the C20 antibody, which detects
APP and all APP C-terminal fragments containing the last 20 residues, revealed
that full-length APP was unaffected by -secretase inhibition (Fig. 2a).
In stark contrast, C99 (p = 0.0005) by -secretase and C83
(p = 0.0002) by -secretase were greatly increased by the
-secretase inhibitor, and the increase of C83 in terms of fold was
significantly higher than that of C99 (Fig. 2a). As -secretase activity
is also linked to autophagy–lysosome functions that may affect the protein
levels of APP and APP C-terminal fragments, we also inhibited the lysosome with
chloroquine (CHL) in PN. Distinct from -secretase inhibition,
CHL treatment significantly increased mature APP (p = 0.0002; Fig. 2b).
As such, it is unlikely that -secretase inhibition caused overall
lysosome inhibition, because at least for full-length APP, its degradation in
lysosome was unaffected by the -secretase inhibitor. These results are
in line with cell-free studies in that full-length APP is not a substrate of the
-secretase, but given the complexity of the intracellular environments
and regulatory machineries in cells, the mechanism underlying the inability of
the -secretase to cleave full-length APP might be different.
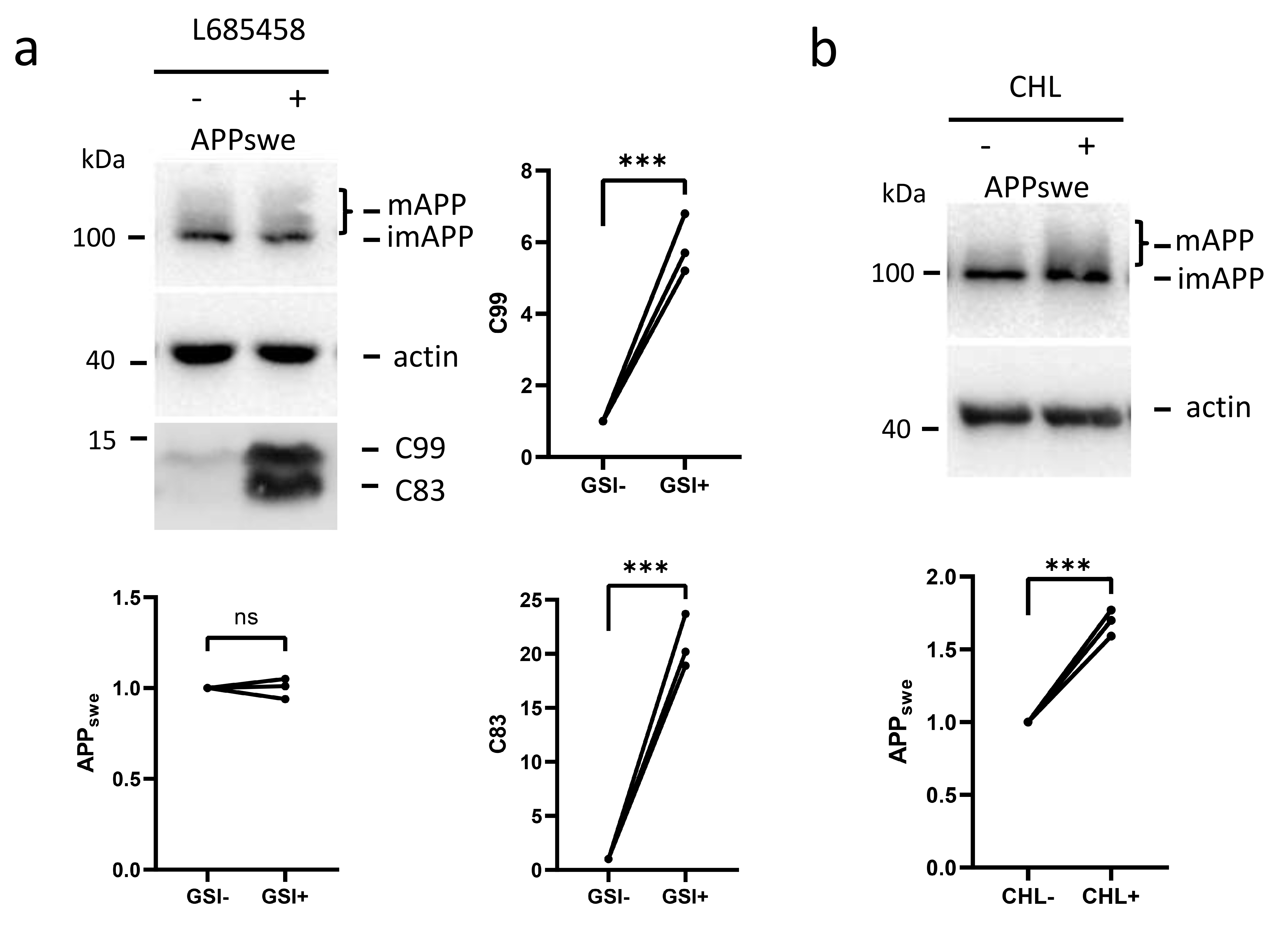 Fig. 2.
Fig. 2.
-secretase does not cleave full-length APP in primary
neurons. (a) PN (primary neurons from APP Swedish mutant
transgenic mice) were treated with or without the -secretase inhibitor
L685458 (GSI) for 3 h. The cell lysates were analyzed by Western blotting for
APPswe using the C20 antibody targeting the last 20 amino acids of APP. The
relative amounts of protein bands with or without -secretase inhibition
were plotted. (b) PN were treated with or without CHL overnight, and the
cell lysates were also analyzed by Western blotting for APPswe. The relative
amounts of protein bands with or without CHL treatment were plotted. The numbers
represent the mean standard deviation (***p 0.001, ns:
non-significant). n = 3 or more independent experiments for all figures. CHL, Chloroquine.
3.3 The -Secretase-Inhibiting Juxtamembrane Helix of APP
is not a Factor Preventing -Cleavage of Full-Length APP
To determine if there is a signal in the ectodomain of APP that inhibits the
-cleavage of APP, we first assessed the juxtamembrane helix (JH) domain
in APP. The JH is an -helix structure within the C99/A region.
A previous study showed that the JH can partially inhibit the -cleavage
of C99, and the peptide composed of the residues of JH could be used as an
efficient -secretase inhibitor [25]. We recently also reported that
releasing JH-mediated -secretase inhibition by the binding of clusterin
to the JH strongly enhances -cleavage of C99 [26]. To the best of our
knowledge, the JH is the best, if not the only characterized -secretase
inhibiting motif in APP/C99. To determine if the JH in APP also prevents
-cleavage of full-length APP, the APP mutant was
overexpressed in HEK293, and the cells were treated with a -secretase
inhibitor for 3 h. The F615P substitution is sufficient to disrupt the
-helix structure of the JH [7]. While C83 (p = 0.0004) derived
from this APP mutant was apparently increased by -secretase inhibition,
similar to wild-type APP (Fig. 3a), the full-length APPF protein
remained unchanged (Fig. 3b). Because of the inhibition of -cleavage by
the F615P substitution [27], C99 (p = 0.0027) produced from
APP relative to C83 (p = 0.0001) appeared to be much higher
than that from wild-type APP. Therefore, the JH is not necessary for the
inhibition of -cleavage of full-length APP.
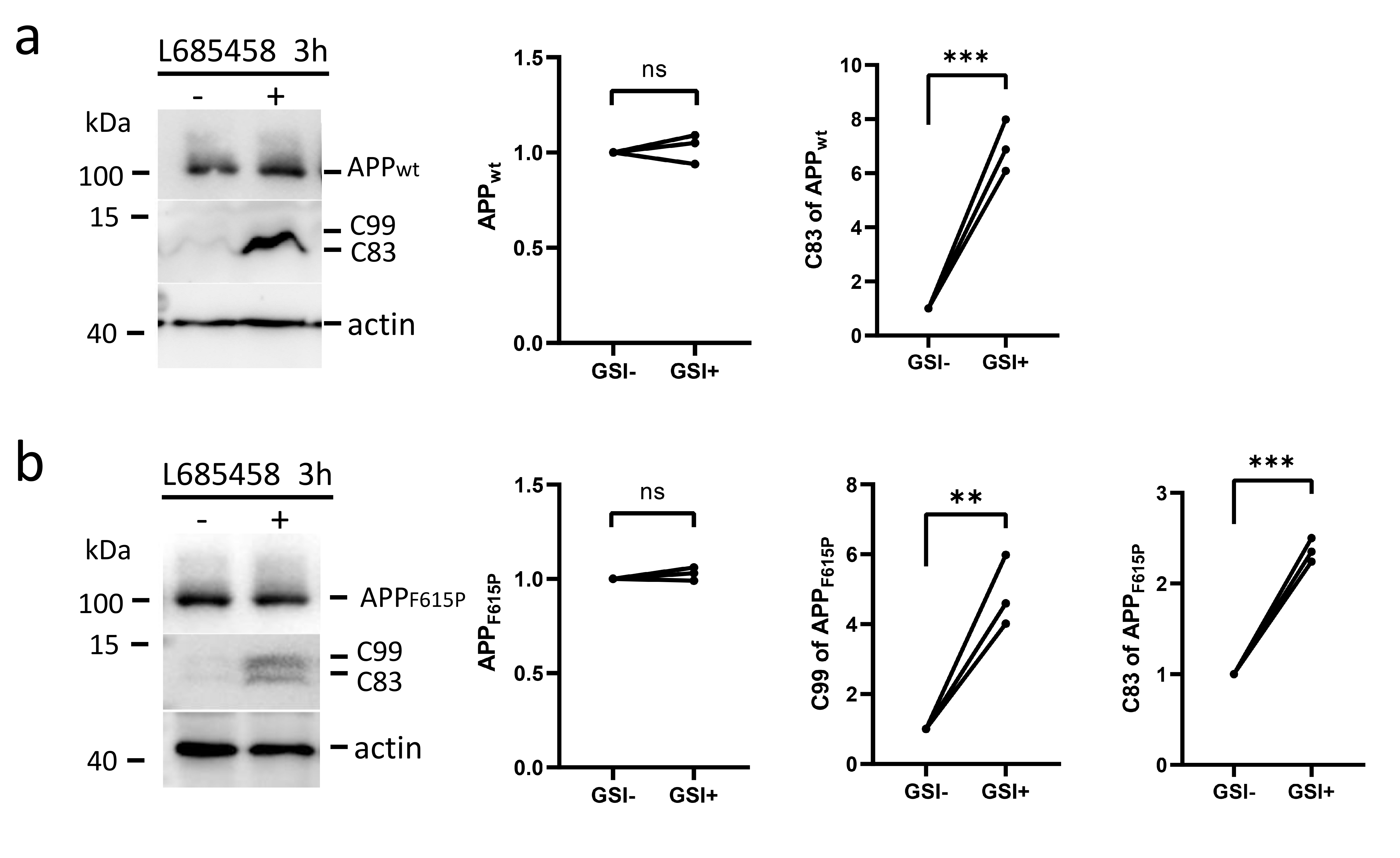 Fig. 3.
Fig. 3.
The JH is not necessary for the inhibition of
-cleavage of full-length APP. APP (a) and the APP
mutant (b) were overexpressed in HEK293 cells and the cells were treated with or
without the -secretase inhibitor L685458 for 3 h. The cell lysates were
analyzed by Western blotting for APP and C-terminal fragments using the C20
antibody targeting the last 20 amino acids of APP. The relative amounts of
protein bands with or without -secretase inhibition were plotted. The
numbers represent the mean standard deviation (**p 0.01,
***p 0.001, ns: non-significant). n = 3 or more independent
experiments for all figures. JH, juxtamembrane helix.
3.4 There is no Single Specific Sequence in the Ectodomain of APP
that Inhibits -Cleavage of Full-Length APP
To further explore whether there is an inhibitory domain in the ectodomain of
APP, we generated several plasmids to express the C-terminal fragments C200,
C139, C116 and C99 that contain the last 200 (starting from residue 496), 139
(starting from residue 564), 116 (starting from residue 579), and 99 residues of
APP, respectively. Based on the predicted structures, these sites are not within
helical or -sheet structures. To ensure the correct topology, the signal
peptide of APP was fused to the N-termini of these fragments (Fig. 4a). When
overexpressed in HEK293 cells, all of these fragments could be cleaved by
endogenous -secretases into C83, with C116 and C99 as the least
efficient substrates for -secretases, possibly because of the absence
of the O-glycosylation sites Thr in these two fragments [28] (Fig. 4b).
C200 and C139 showed strong upper bands above the expected size (Fig. 4b). These
upper bands probably due to post-translational modifications suggested that these
fragments, similar to full-length APP, were exported out of the ER to later
organelles where -, - and -cleavages take place.
Upon -secretase inhibition, C83 generated from these fragments was
remarkably increased, indicating that -secretase was successfully
inhibited in these cells. However, among these fragments, only C99 (p =
0.0002) and C116 (p = 0.0069) were upregulated by -secretase
inhibition, and the upregulation of C99 was more robust than that of C116
(p = 0.0041) (Fig. 4b). Similar to full-length APP, other fragments
longer than C116 and their modified forms did not respond to -secretase
inhibition (Fig. 4b). With the exception of the overexpressed C99 itself, other
fragments longer than C99 produced little C99 because without the Swedish
mutation, -cleavage by endogenous -secretases was extremely
weak. We did not use the AICD to indicate -cleavage because the AICD
could also be produced from C83 derived from these fragments, which may confound
the results. Since the -secretase is active in post-ER organelles, we
questioned if C200 and C139 were successfully exported out of ER, even though
they were cleaved into C83 by -secretase. Immunofluorescence for
overexpressed C200 and C139 revealed a mixed localization of these two fragments
in the ER, the Golgi, and some vesicle-like structures. Staining in the
perinuclear Golgi-like structure was the most prominent. By contrast, when the
dibasic motif was introduced into the C-terminal tails of these two fragments to
retain them in the ER through the COPI vesicle-dependent retrograde transport
mechanism, these fragments displayed a typical polygonal network and perinuclear
pattern of the ER (Fig. 4c). Hence, these two fragments efficiently entered the
secretion pathway, and not being cleaved by -secretase was not a
consequence of ER retention.
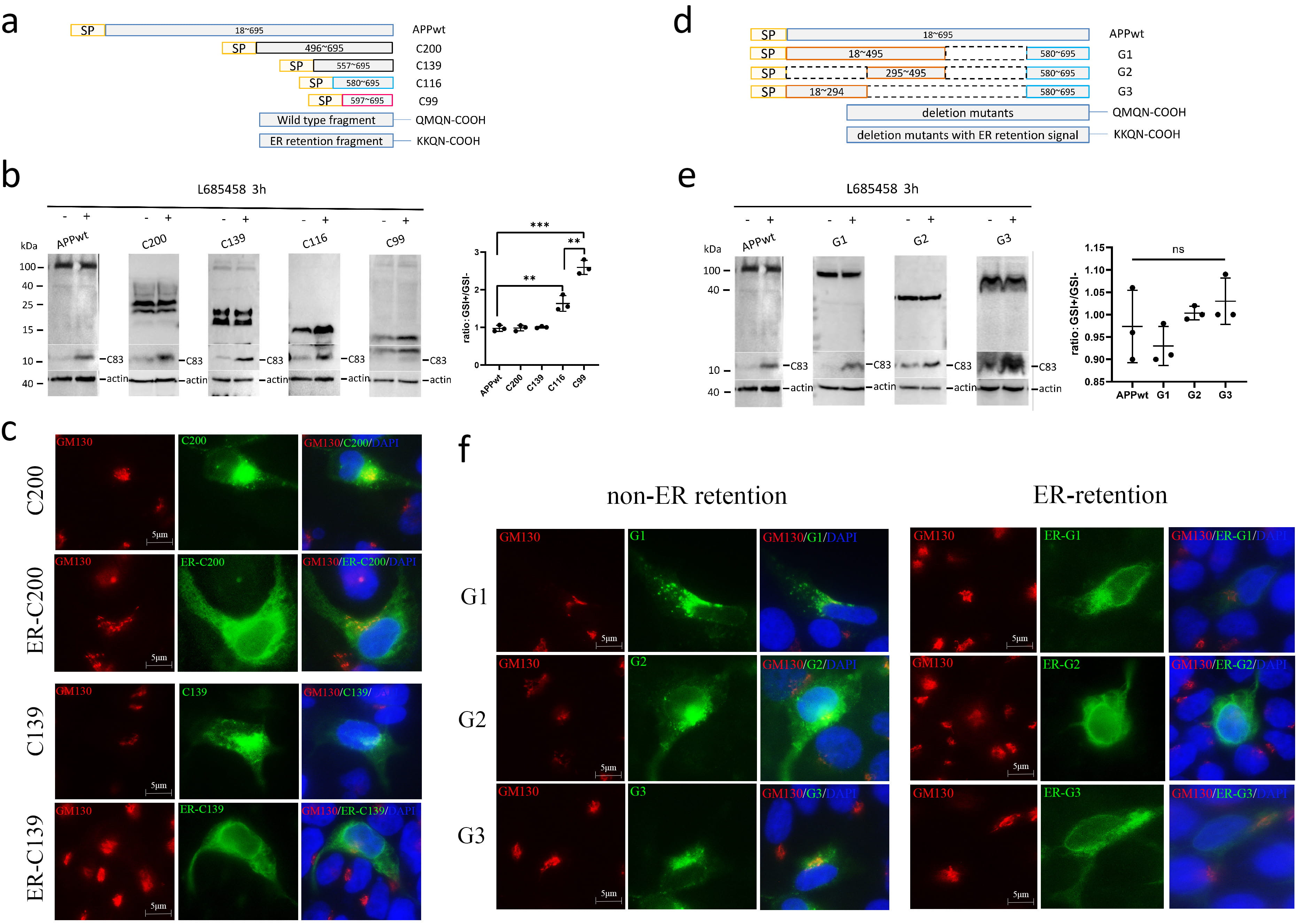 Fig. 4.
Fig. 4.
The -secretase does not cleave APP C-terminal
fragments longer than C116. (a) Schematic diagram showing the truncation. For ER
retention, the last four residues QMQN of the fragments were replaced by KKQN.
SP: signal peptide of APP. (b) APP and the truncation mutants were overexpressed
in HEK293 cells, and the cells were treated with or without the
-secretase inhibitor L685458 for 3 h. The cell lysates were analyzed by
Western blotting using the C20 antibody for APP, the mutants, and the
carboxy-terminal fragments (CTFs) generated from these APP variants by endogenous
- and -secretases. The ratios of protein bands with
-secretase inhibition (GSI+) to those without -secretase
inhibition (GSI-) were plotted. (c) HEK293 cells were transfected with the
plasmids expressing the truncation fragments with or without the ER retention
signal. The cell after fixation were co-stained using C20 antibody for the
fragments and GM130 antibody for the Golgi. (d) Schematic diagram showing the
deletion mutants of APP. Dashed lines indicate the deleted regions. (e) APP and
the deletion mutants were overexpressed in HEK293 cells, and the cells were
treated with or without the -secretase inhibitor L685458 for 3 h. The
cell lysates were analyzed by Western blotting using the C20 antibody for APP,
the mutants, and the CTFs generated from these APP variants by endogenous
- and -secretases. The ratios of protein bands with
-secretase inhibition to those without -secretase inhibition
were plotted. (f) HEK293 cells were transfected with the plasmids expressing the
deletion mutants with or without the ER retention signal. The cell after fixation
were co-stained using C20 antibody for the fragments and GM130 antibody for the
Golgi. The numbers represent the mean standard deviation (***p
0.001, **p 0.01, ns: non-significant). n = 3 or more
independent experiments for all figures. ER, endoplasmic reticulum; QMQN, Glutamin-Methionine-Glutamine-Asparagine; KKQN, Lysine-Lysine-Glutamine-Asparagine.
To determine if the sequence N-terminal to C116 in APP contains a
-cleavage-inhibiting sequence, we further generated three APP deletion
mutants: one with the sequences 496–579 deleted (G1), one containing signal
peptides 295–495 and 580–695 (C116) (G2), and one with sequences 296–579
deleted (G3) (Fig. 4d). All of these deletion mutants were cleaved by the
endogenous -secretases, and the resulting C83 was upregulated by
-secretase inhibition, but still, the overexpressed mutants stayed the
same with or without -secretase inhibition (Fig. 4e). Since the deleted
regions combined cover the entire sequence N-terminal to C99, there is unlikely a
single specific domain to inhibit the -cleavage of APP in the
N-terminus of APP. Similar to the C200 and C139 fragments, all of these deletion
mutants of APP enter the secretion pathway without being retained in the ER (Fig. 4f).
3.5 The Large Ectodomain N-Terminal to C99 Inhibits the Binding with
the Secretase Subunit NCT
Previous studies on the -cleavage of Notch protein demonstrated that
the longer extracellular structure of Notch results in lower cleavage efficiency
by -secretase [29], which explains why the first truncation of Notch is
required for -cleavage. Since we failed to identify a specific sequence
in the ectodomain of full-length APP that inhibits -cleavage of APP, we
considered that the -cleavage of APP could follow the same principle as
that for Notch cleavage. To unambiguously test the hypothesis that the size, but
not the sequence of the ectodomain is the inhibitory factor, the GFP was inserted
between the signal peptide of APP and C99 (Fig. 5a). GFP is a protein irrelevant
to APP or APP processing, and it is simply to increase the size of the
ectodomain. When overexpressed into HEK293 cells, the GFP-C99 chimera was
robustly expressed but showed no change upon -secretase inhibition. By
contrast, C99 without fused GFP was significantly increased by the
-secretase inhibitor (p = 0.0022; Fig. 5b). Hence, the long
ectodomains, instead of a specific sequence, suppress the -cleavage of
APP.
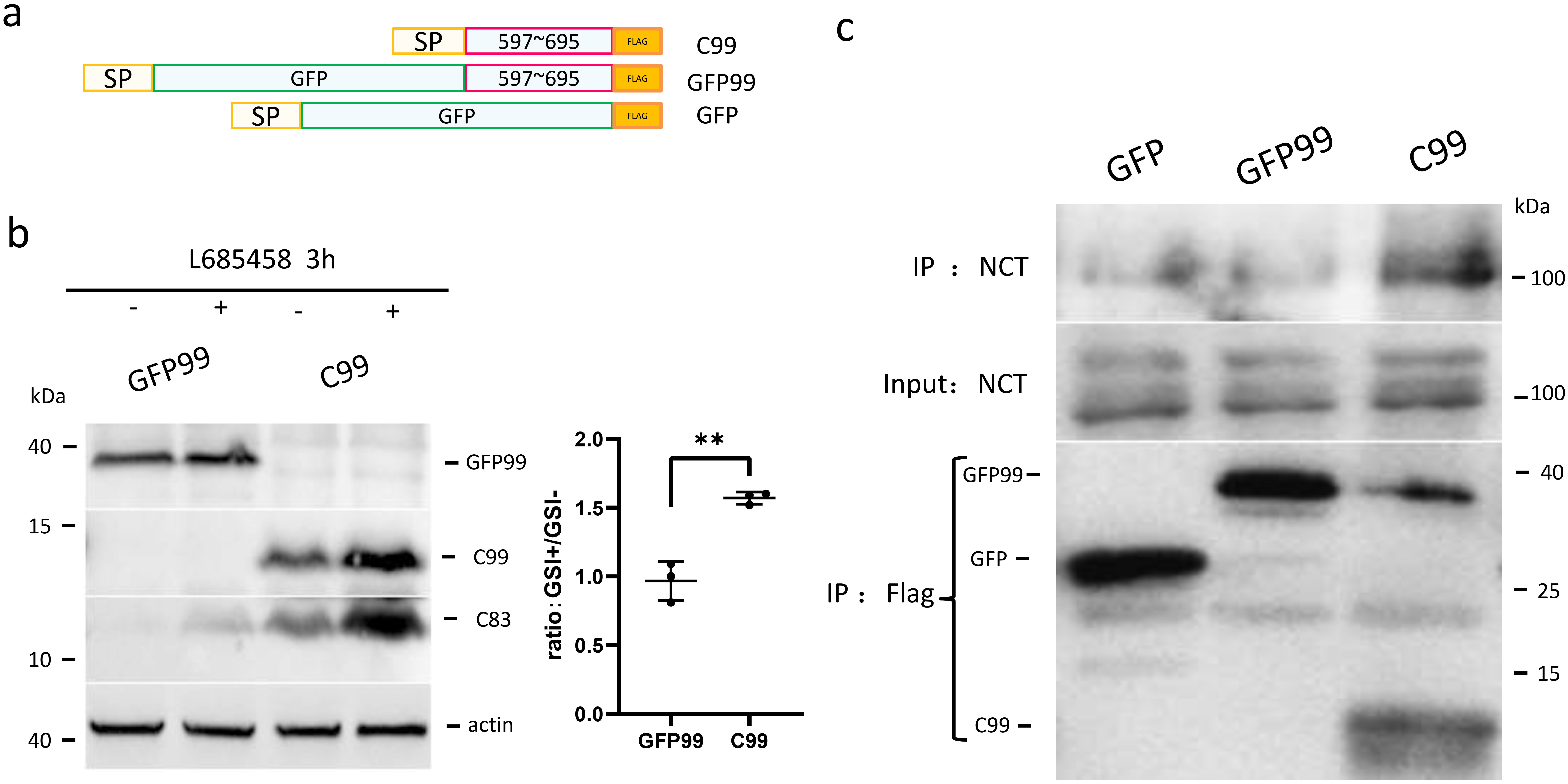 Fig. 5.
Fig. 5.
GFP fused to the N-terminus of C99 prevents the binding and the
cleavage of C99 by -secretase. (a) Schematic diagram showing the
structures of artificial C99 variants. SP, signal peptide; GFP, green fluorescent
protein; FLAG, FLAG tag fused to the C-terminus of C99 or GFP. (b) Effects of
-secretase inhibition on C99 and GFP-C99. C99 and GFP-C99 were
expressed in HEK293 cells and the cells were treated with or without
-secretase inhibitor for 3 h. The cell lysates were blotted using C20
antibody for C99, GFP-C99, and C83 produced from C99 and GFP-C99. The ratios of
protein bands with -secretase inhibition (GSI+) to those without
-secretase inhibition (GSI-) were plotted. (c) C99, GFP-C99, and
GFP-FLAG as a negative control were overexpressed in HEK293 cells. These proteins
were IP’d with anti-FLAG magnetic beads from cell lysates, and the co-IP’d
endogenous NCT was detected using an NCT-specific antibody. The relative amount
of protein bands with or without MK-8931 treatment were plotted. The numbers
represent the mean standard deviation (**p 0.01). n = 3 or
more independent experiments for all figures.
To investigate the mechanism by which the large ectodomain inhibits
-cleavage, we performed co-IP of C99 and GFP-C99 with endogenous NCT.
NCT is a subunit in the -secretase complex that is crucial for
-secretase activity and substrate priming [30]. While C99 apparently
interacted with NCT, the interaction of GFP-C99 chimera with NCT was no stronger
than the background signal (Fig. 5c). Therefore, the large ectodomain impairs the
accessibility of -secretase to APP or its C-terminal fragments, which
precludes the -cleavage within the transmembrane domain of APP.
4. Discussion
AD is a devastating disease without a cure. Since the discovery of the
amyloidogenic pathway as the contributing factor to neritic plaques and AD
pathogenesis, great attempts have been made to target molecules in these pathways
for AD therapy. Inhibiting -and -secretases for AD therapy
used to be considered a promising strategy. However, almost all clinical trials
using inhibitors of these enzymes were halted before the scheduled trial was
finished, mostly because of the strong side effects. A number of studies have
indicated that both BACE1, the major -secretase, and the
-secretase may cleave a variety of substrates for essential biological
functions, and directly inhibiting them would inevitably affect these functions,
which leads to the side effects. Specific inhibition of the cleavages of APP/C99
by these two enzymes remains unrealistic at this time. Therefore, we determined
if there is a method to circumvent this technical difficulty by altering the
sequential order of - and -secretase cleavages. Such a
strategy could be designed very specific for APP without affecting other
functions of - and -secretases.
Our results showed that sAPP, if exists, can be efficiently released
out of cells despite the presence of part of the transmembrane domain. The
-cleavage of sAPP appeared to be very weak because A
production from sAPP or the -cleavage of sAPP was
much lower than from APP. One of the possible reasons is that sAPP can
be directly secreted into the extracellular space, which reduces the amount of
sAPP available for -cleavage. Another explanation is that the
-cleavage of APP requires APP being anchored onto the membrane to allow
the access of -secretases to the priming and cleavage sites in APP.
Without being anchored on the membrane, sAPP could be diffusive in the
lumen of organelles, and the chance to encounter -secretases would be
lowered. Moreover, the conformation of sAPP could be different from
that of full-length APP being anchored on the membrane.
We further determined if there is a certain sequence in APP that inhibits
-cleavage of APP. If true, antibodies or other small molecules could be
designed to bind this domain or motif, which releases -secretase
inhibition and allows the -cleavage of full-length APP to generate
sAPP. However, our results indicated that there appeared to be no such
an inhibitory domain in APP. Instead, a large ectodomain, regardless of the
sequence, would abrogate -cleavage. C139, a fragment with only 40
residues N-terminally flanking C99, is no longer cleavable by
-cleavage. This conclusion is consistent with the results in previous
studies using cell-free systems [21]. Mechanistically, the large ectodomain may
serve as a spatial hindrance to keep -secretase away from APP or the
longer C-terminal fragments of APP, as the binding of C99 with NCT is abolished
by GFP fused to the N-terminus of C99.
Although we failed to identify a sequence in APP that can be targeted to induce
the -cleavage of full-length APP, the strategy of enabling such a
cleavage that normally does not happen remains to be valid for the prevention of
A generation without affecting other functions of -secretase.
Methods other than searching for inhibitory domains in APP may help the
achievement of the strategy.
5. Conclusions
Our results suggest that if -secretase could directly cleave
full-length APP and generate the secreted fragment sAPP, it would
abolish the production of A. APP does not contain a specific inhibitory
sequence for -secretase; instead, the large ectodomain of APP prevents
-cleavage of APP, probably through a spatial effect. Hence, methods
other than targeting a sequence of APP to release this inhibition may benefit AD
prevention and therapy without causing the side effects of direct - and
-secretase inhibition.
Abbreviations
AD, Alzheimer’s disease; APP, amyloid precursor protein; BACE, beta-site amyloid
precursor protein cleaving enzyme; sAPP//, soluble
amyloid precursor proteins-//; NCT, nicastrin.
Availability of Data and Materials
Data generated and analyzed in this study are available from the corresponding
author on reasonable request.
Author Contributions
ZW conceived and designed the experiments. YuanL, HL, WL, YuL performed the
experiments. YuanL and ZW analyzed data. ZW contributed reagents, materials, and
analytical tools. YuanL and ZW wrote the manuscript. All authors reviewed the
manuscript. All authors read and approved the final manuscript. All authors have
participated sufficiently in the work and agreed to be accountable for all
aspects of the work.
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of Xuanwu Hospital of Capital Medical University.
Acknowledgment
Not applicable.
Funding
This work was supported by National Natural Science Foundation of China (no.
81870832), and Beijing Committees of Education-Science Foundation of Beijing
joint fund (no. KZ202010025040) to ZW.
Conflict of Interest
The authors declare no conflict of interest.
 Fig. 1.
Fig. 1. Fig. 2.
Fig. 2. Fig. 3.
Fig. 3. Fig. 4.
Fig. 4. Fig. 5.
Fig. 5.




