1 Department of Genetics, Faculty of Natural Sciences, Tabriz University, 5166616471 Tabriz, Iran
2 Department of Biology, Faculty of Biological Sciences, North Tehran Branch, Islamic Azad University, 1651153311 Tehran, Iran
3 Department of Chemical Engineering, Faculty of Chemical Engineering, Shahreza Branch, Islamic Azad University, 8648146411 Shahreza, Iran
4 Department of Medical Biology, Faculty of Medicine, Ankara Yildirim Beyazit University (AYBU), 06800 Ankara, Turkey
5 Department of Biology, College of Basic Sciences, Kermanshah Branch, Islamic Azad University, 6718997551 Kermanshah, Iran
6 Now with Department of Biotechnology, College of Environmental and Bioresource Sciences, Jeonbuk National University, 54896 Jeonbuk, Republic of Korea
7 Department of Biomedical Engineering, College of Basic Sciences, Qom Branch, Islamic Azad University, 3716146611 Qom, Iran
8 Department of Biology, Faculty of Basic Sciences, University of Maragheh, 5518779842 Maragheh, Iran
9 Department of Herbal Medicine, University of Poona, 411007 Poona, India
10 Clinical Diagnosis Laboratory, Shahid Beheshti University-affiliated Hospital, Zanjan University of Medical Sciences (ZUMS), 4513956111 Zanjan, Iran
11 Department of Medical Laboratory Science, Faculty of Paramedicine, Zanjan University of Medical Sciences (ZUMS), 4513956111 Zanjan, Iran
12 Department of Obstetrics and Gynecology, Faculty of Medicine, Iran University of Medical Sciences (IUMS), 1449614535 Tehran, Iran
13 Division of Pediatric Neurosurgery, Department of Neurosurgery, Ankara Bilkent City Hospital, 06800 Ankara, Turkey
14 Department of Brain and Nerve Surgery, Faculty of Medicine, Ankara Yildirim Beyazit University (AYBU), 06800 Ankara, Turkey
Abstract
As one of the most common solid pediatric cancers, Neuroblastoma (NBL) accounts for 15% of all of the cancer-related mortalities in infants with increasing incidence all around the world. Despite current therapeutic approaches for NBL (radiotherapies, surgeries, and chemotherapies), these approaches could not be beneficial for all of patients with NBL due to their low effectiveness, and some severe side effects. These challenges lead basic medical scientists and clinical specialists toward an optimal medical interventions for clinical management of NBL. Regardingly, taking molecular and cellular immunopathophysiology involved in the hypoxic microenvironment of NBL into account, it can practically be a contributing approach in the development of “molecular medicine” for treatment of NBL. Interestingly, pivotal roles of “herbal medicine” in the hypoxic microenvironment of NBL have been extensively interrogated for treating a NBL, functionally being served as an anti-cancer agent via inducing a wide range of molecular and cellular signaling, like apoptosis, cell cycle arrest, and inhibiting angiogenesis. Hence, in this review study, the authors aim to summarize the anti-tumor effects of some medicinal plants and their phytoconstituents through molecular immunopathophysiological mechanisms involved in the hypoxic microenvironment of NBL. In addition, they try to open promising windows to immune gene-based therapies for NBL “precision medicine” through clinical advantages of herbal and molecular medicine. An interdisciplinary collaboration among translation and molecular medicine specialists, immunobiologists, herbal medicine specialists, and pediatric neuro-oncologists is highly recommended.
Keywords
- curcumin
- garlic
- green tea
- hypoxia
- medicinal herbs
- molecular medicine
- neuroblastoma
Neuroblastoma (NBL) is one of the most prevalent solid, embryonal, and extracranial cancers among the infants (five-year old children and younger). Though it may rarely occur in the older children, there are a few cases of asymptomatic primary thymic intracranial NBL (with an anterior mediastinal or thoracic mass) in the elderlies, while a narrow range of those rare patients with NBL have been clinically described in the available literature, up to now. Accordingly, due to the possible presence of the abnormal epithelial cells within the thymus as a rare malignancy disorder, NBL is literally considered as a thymoma [1, 2, 3].
From cellular aspects, NBL majorly involves a neoplasm in the progenitor cells of abdominal Sympathetic Nervous System (SNS), which are considered as the embryonic sympathoadrenal (sympathoadrenergic) lineage of the neural crests. With a less possibility, primary NBL sometimes occurs in paraspinal sympathetic ganglia (ganglionic lineage precursors of the SNS) [4, 5, 6, 7, 8, 9]. Indeed, NBL is derived from the adrenergic neuroblasts (immature nerve cells) presented in the neuroectoderm, showing a high frequency for developing metastasis in the procedure of NBL carcinogenesis [10, 11]. Bio-statistically, NBL approximately accounts for 13%–15% of cancer-related deaths worldwide among children with an increasing incidence rate worldwide [5, 9, 12, 13]. According to the International Neuroblastoma Staging System (INSS), NBL is classified into five subgroups (1, 2A, 2B, 3, 4, 4S). More interestingly, according to the International Neuroblastoma Pathology Classification (INPC), morphology-based classification of neuroblastic tumors is categorized into: NBL (Schwannian stroma-poor); ganglioneuroblastoma-intermixed (GNBi, Schwannian stroma-rich); ganglioneuroma (GN, Schwannian stroma-dominant); and ganglioneuroblastoma, nodular (GNBn, composite Schwannian stroma-rich/stroma-dominant and stroma-poor) [14].
Taking clinical and biologic indices of tumors (age, histopathological properties, cancer-related predisposition disorders, genetics, and tumor grading or staging) into account, clinical manifestations of patients with NBL are pathologically divided into three categories, including low, moderate, and high-risk groups [15, 16]. Patients with NBL may indicate various clinical manifestations, varying from spontaneous regression or even therapy-induced regression into benign ganglioneuroma, to aggressive progression, as well as from symptomless abdominal mass, to hypertension, emesis, anorexia, constipation, diarrhea, fatigue, and severe abdominal or bone pain [17, 18].
From etiological aspects, the etiopathology of NBL has not been completely understood, yet. However, it has been demonstrated that some genetic predispositions, age of the patient at diagnosis, chromosomal aberrations, changes in the patterns of DNA, amplification of the N-myc proto-oncogene (MYCN), Anaplastic Lymphoma tyrosine Kinase (ALK) activating mutations, and environmental factors depict indisputable roles in the risk assessment and susceptibility to NBL [9, 19, 20].
From cancer genomics aspects, it has been interrogated that several genetic variations (like alterations in the chromosomal copy number, and intratumoral/intertumoral heterogeneity (such as spontaneous regression, and resistance to treatment)) make a poor prognostic responses against conventional therapies, frequent relapses, as well as a poor survival rate in patients with NBL. Among them, those patients with MYCN-amplified NBL (more than 10 copies like v-myc myelocytomatosis viral related oncogene, and neuroblastoma derived (avian) or MYCN), 1p36 deletion or 17q gain, as well as ALK activating mutations have been recognized as the most typical genetic features for clarifying the advanced stages of NBL (or malignant NBL), angiogenesis, resistance to therapy, genomic instability, and a very poor survival rate [9, 20, 21].
From diagnostic aspects, there are not a wide array of diagnostic procedures for NBL. According to the recently-conducted clinical and basic medical sciences-based studies, some chromosomal abnormalities and urinary catecholamine (as tumor markers) have been extensively used for the proper paraclinical diagnosis of NBL [11, 22, 23, 24, 25, 26].
From therapeutic aspects, chemo-radiotherapy regimens (including cycled administration of Cisplatin, Vincristine, Etoposide, Cyclophosphamide, and Carboplatin (COJEC)), Procarbazine, Irinotecan, and surgery (if possible followed by administration of alkylating agents like Temozolomide), high-dose chemotherapy regimens, and myeloablative chemotherapy regimens with the transplantation of autologous stem cells have been considered as commonly-used therapeutic strategies for patients with NBL [25, 26, 27].
In some clinical reports, cytotoxicity, conjunctivitis, dysgeusia, and cognitive impairments have been considered as the most frequently recorded adverse effects among patients with NBL due to the low efficacy of those aforesaid diagnostic and therapeutic procedures. Especially, transplantation-associated thrombotic microangiopathy has been considered as a severe complication for a wide array of patients with cancer receiving hematopoietic stem cell transplantation , and patients with NBL were not an exception. Therefore, it seems that there is an imperative need for warranting tailored therapeutics aimed at re-educating Tumor Microenvironment (TME) or drug repurposing, as more efficient therapeutic alternatives for all types of patients with NBL [9, 10, 23, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37].
In case of targeted therapies for clinical management of NBL, results acquired from some those basic medical sciences-based studies have depicted the therapeutic efficacies of several novel immunotherapeutics, including antibody-based, immune-cell-based, and anti-angiogenic-based therapies, being under investigation in clinical trials for patients with NBL [35, 36]. Among those antibody-based immunotherapeutics, anti-disialoganglioside monoclonal Antibody (mAb) ch14.18 against GD2, which is a ganglioside presented in human NBL cells, can be clinically utilized either alone or in combination with IL- 2, and Granulocyte Macrophage-Colony Stimulating Factor (GM-CSF). Retinoic acid, as well as Bevacizumab are considered as another prime instances of immunotherapeutics used against NBL. Immune-cell based therapeutics for patients with NBL literally include genetically-non-engineered immune-cell-based therapies (like activated donor Natural Killer cells (NKs) with Hu14.18-IL-2 especially for patients with relapsed or refractory NBL), as well as genetically-engineered immune-cellbased therapies (like Anti-GD2 Chimeric Antigen Receptor (CAR) T cells [35, 36].
Totally, these aforementioned challenges in the current therapeutic approaches against NBL make basic medical scientists, neurologists, and pediatricians looking for more optimal, highly-sensitive, efficient, and time-preserving medical interventions based on the fundamental cellular and molecular aspects of tumor biology, tumor immunopathophysiology, as well as immune genes involved in the hypoxic TME, contributing to the initiation, glucose metabolism, progression, and metastasis of NBL, opening promising windows to alternative medicine and developing cellular and molecular medicine for treatment of NBL. Totally, according to the substantial developments in complementary medicine (including herbal medicine, alternative medicine, translation medicine, phytomedicine, naturopathic medicine, and traditional medicine) over multiple decades, herbal remedies have been broadly utilized for the treatment of different disorders, developing “herbal medicine”, and playing indispensable roles in human health [38, 39, 40, 41, 42, 43]. Accumulative evidence indicated that medicinal plants, essential oils, phytocomponents, bio-compounds, phytochemicals, and their hydroalcoholic extracts or natural components had shown anti-malaria, anti-inflammatory, anti-viral, and anti-cancer properties through possible different molecular or cellular mechanisms. As prime instances, several cellular and molecular procedures like induction of cancerous cell apoptosis, cell cycle arrest, immune regulation, and inhibition of angiogenesis can be enumerated as the most major signalings that determine immunopathophysiology involved in the TME [44, 45, 46, 47]. In case of NBL, it has been demonstrated that phytochemicals, as plant-derived pharmacologically-active bio-compounds and secondary metabolites, may indicate chemo-preventive properties with fewer side effects, targeting molecular signalings that suppress the cellular growth of NBL cells with less toxicity [48, 49, 50, 51, 52]. According to the in vitro cellular and molecular-based reports, there is a relationship between herbal products and hypoxic microenvironment of NBL, being served as an anti-cancer agent via different functional signaling, like induction of apoptosis, cell cycle arrest, inhibition of inflammation, altering the chemosensitivity/radiosensitivity to the chemotherapeutic/radiotherapeutic agents, and angiogenesis [53, 54, 55, 56]. Accordingly, it seems that achievements in the novel fields of medicine like “molecular medicine” can accelerate the development of “molecular herbal medicine” aimed at the treatment of cancers and NBL is not an exception. Moreover, it seems that clinical administration of medicinal plants is attributed with a wide array of molecular signalings and immune genes involved in the microenvironment of NBL, deciphering the secrets for NBL “precision medicine” through immunotherapeutic-based approaches via in vitro and in vivo genetic manipulation in the microenvironment of NBL, and making a bright future for an interdisciplinary collaboration between a wide range of clinical specialists, complementary medicine specialists, and basic medical sciences, simplifying a more efficacious treatment of NBL with less side effects [7, 11, 57, 58, 59, 60, 61].
Hence, in this literature review, we will firstly discuss the pathologic mechanisms of the immunopathophysiology involved in the immune microenvironment of NBL. Then, we will focus on the anti-tumor potentials in some of the considerable herbal products, their phytoconstituents, hydroalcoholic extracts, and phytochemicals (in the forms of chemical effective agents and nanoparticles) according to the triggered molecular signaling, and highlighting the role of molecular herbal medicine and precision medicine in the treatment of NBL.
This present comprehensive review study was performed according to the academically acceptable statement guideline named “Preferred Reporting Items for Systematic review and Meta-Analysis Protocols (PRISMA-P)” on January 2015 (https://www.equator-network.org/reporting-guidelines/prisma-p/) and (http://www.prisma-statement.org/) (Fig. 1). In addition to the aforesaid guideline, one of our recently-conducted studies (as a comprehensive systematic review) was literally used for designing the main basis of our search protocol [62].
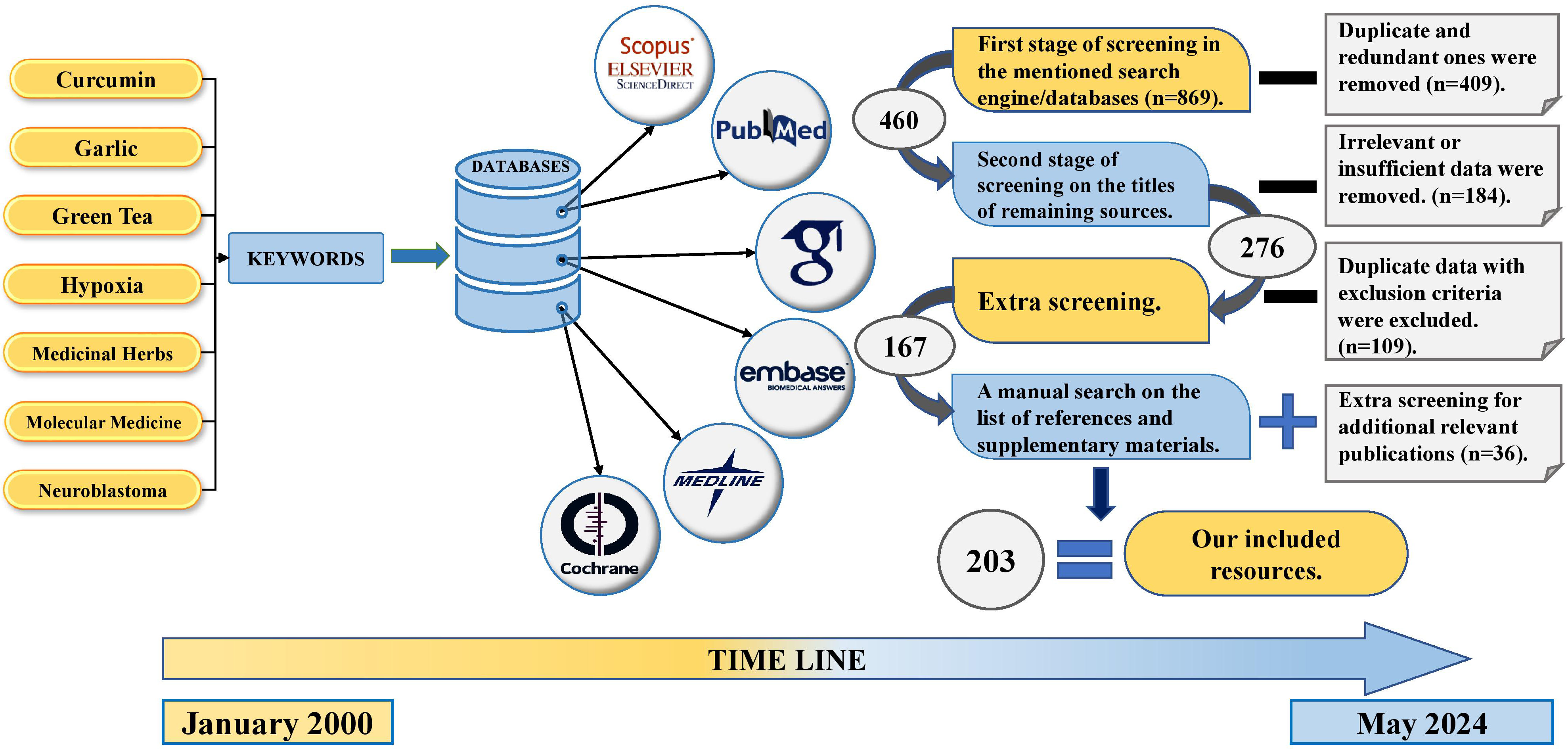 Fig. 1.
Fig. 1.
Search strategy according to the PRISMA guideline (PRISMA-S extension 2021 statement). PRISMA, Preferred Reporting Items for Systematic review and Meta-Analysis. Created with BioRender.com.
To conduct an electronic comprehensive literature review, a time interval
commencing from January 2000 to May 2024, seven main keywords (including:
Curcumin, AND Garlic, AND Green Tea, AND Hypoxia, AND Medicinal Herbs, AND
Molecular Medicine, AND Neuroblastoma), and 58 complementary keywords (including:
Alternative Medicine, AND Anti-cancer, AND Anti-proliferation, AND Aromatic
Compounds, AND Bio-active Compounds, AND Complementary Medicine, AND Experimental
Medicine, AND Gene Therapy, AND Herbal Diet, AND Herbal Medicine, AND
Hydroalcoholic Extracts, AND Hypoxia-Inducible Factor-1
To find the potentially-eligible resources, eight author independently conducted main screening process in several steps (including three main and one non-electronic backward steps on the references and bibliographies of included articles) (Fig. 1). Any uncommon points or disagreements were referred to the corresponding author for the final consultations.
According to the aim of this comprehensive review study, published contents in the format of the original (including experimental (research/full-length), and non-experimental (hypothesis) ones), review (including mini-review, best evidence, narrative (traditional) review, critical reviews, systematic review, systematic review and meta-analysis), comparative, cross-sectional, cohort, retrospective, prospective, viewpoint, observational, commentary, letter to the editor, editorial, opinion, short/rapid/brief communication, Randomized Clinical Trial (RCT), case report, and case series articles were accepted for the consideration in the forms of full-text/full-length, abstract, section of book, chapter, and conference papers/presentation, and in English language (or only abstract in English language).
Those studies that clinically or experimentally had investigated the diagnostic/prognostic/therapeutic advantages of molecular signaling induced by the phytoconstituents, phytocompounds, aromatic compounds, pharmacologically-active bio-components, or pharmacologically-active bio-compounds of the medicinal herbs (in the forms of chemical effective agents or nanoparticles) in patients with NBL (on the human subjects) were included. Those clinical or experimental studies that comparatively had investigated the clinical advantages of molecular signaling induced by aforementioned compounds of medicinal herbs in comparison with other compounds or other types of techniques in patients with NBL (on the human subjects) were included, as well. Those clinical or experimental studies that comparatively had investigated the clinical advantages of molecular signaling induced by aforementioned compounds of medicinal herbs between patients with NBL (on the human subjects) and control groups were included, as well.
In order to refrain from any bias, those clinical and experimental studies involving human subjects after the usage of any immunomodulators (immunostimulants/immunosuppressive/immunoinhibitor), adjuvants, neoadjuvants, vaccines, other external stimulators/inhibitors, any previous treatment with anti-cancer therapeutic regimens, antibiotic regimens, chemotherapeutic regimens, immune cell/gene-based immunotherapeutic regimens, monoclonal antibody-based therapeutic regimens, and self-treatment regimens all were excluded. In addition, those clinical or experimental studies that investigated the clinical advantages of molecular signaling induced by aforementioned compounds of medicinal herbs in patients with NBL who had been previously patients with other types of cancer, autoimmunities, immunodeficiencies, chronic inflammatory disorders, previous acute or chronic (viral/bacterial/fungal/parasitological) infectious/co-infectious diseases, and other types of predisposition disorders were excluded, too. Those studies that clinically or experimentally had been conducted on non-human samples were excluded, too. Additionally, those studies with irrelevant/insufficient/ambiguous data, lack of data, undefined therapeutic values, and undescribed molecular signaling all were excluded, as well.
In this study, eight independent author majorly performed the data extraction and made forms to collect study characteristics (including author name, publication date, name of the journal, type (format) of the article, language of the article, form of the content, study design, used samples, used cell lines, type of the used chemical compounds/bio-active compounds/hydroalcoholic extracts/phytoconstituents/phytocompounds/aromatic compounds/pharmacologically-active bio-components of the medicinal herbs, types/subtypes of herbs, molecular and chemical signaling, molecular genes, type of the prognosis/therapeutic advantages that have been fully mentioned in the aims of this study). In case of overlapping data or several published reports from the same studies in the same/different search engines or databases, the authors tried their best to cover the details and present the most complete and necessary data according to the aim and inclusion/exclusion criteria of this study.
According to the structure and type of this study (comprehensive literature review), no bio-statistical approaches were done.
According to the structure and type of this study (comprehensive literature review), there is no need to register for Research Ethical Committee (REC). It is worth-mentioning that all of the data that supports the findings of this study are openly available in the context of this manuscript.
Despite a wide array of clinical studies, a comprehensive understanding on the complex immunopathogenesis of NBL has been poorly recognized [63, 64]. However, basic medical scientists and geneticists believe that some genomic characteristics such as somatic genetic abnormalities, germline mutations, transcriptomics, epigenetics, and copy number of chromosome can play contributing roles in the etiopathogenesis of NBL [65].
From histopathological aspects, in the context of NBL as a developmental malignancy occurring the neural ganglia, it has been postulated that the neural crest can be considered as a temporary (migratory) and multipotent embryologic tissue, being originated from neuroectoderm [66]. It is worth-mentioning that superenhancer properties of NBL cell lines classifies it into two main groups, including noradrenergic (early type originating from sympathetic neurons), and mesenchymal (late type originating from schwan precursor cells) ones [66, 67]. In vertebrate species, a notable cellular maturation in the neural crest happens during the development of neural tube, reacting to an intricate transcription factor pattern [68, 69]. This structural signaling makes neural crest precursors acquire multipotent differentiation capacities [7, 67]. It seems that a defective sympathetic neuronal differentiation, as well as disruption of neural crest maturation can probably lead to the onset and immunopathogenesis of NBL. Biologically, it seems that cancerous cells modulate gene expression in the target immune cells via some exosomes-based signaling pathways aimed at attenuating the molecular signalings through non-coding RNAs in patients with NBL (especially in high risk cases, or the ones coping with relapse, or recurrent tumors) [7, 32].
Accordingly, a critical procedure in the maturation of neural cell is Epithelial-to-Mesenchymal Transition (EMT) [7, 70]. The expression of the Zinc finger E-box Binding homeobox 2 (ZEB2) and SRY-related HMG box (SOXE) family can stimulate mesenchymal transformation via activation of Matrix Metalloproteinase (MMPs) and lack of E-Cadherin and cell-cell contacts. Then, Fibroblast Growth Factor (FGF), Wnt, and Bone Morphogenic Protein (BMP) signaling induce the differentiation of neural crest cells [71].
During the procedure of embryogenesis, some types of transcription factors, like Paired Box gene 3 (PAX3) (also known as WS1, WS3, CDHS and HUP2), Zink finger protein of the Cerebellum (ZIC1), Transcription Factor AP-2 alpha (TPAP2α), PR Domain zinc finger protein 1 (PRDM1A) (also known as B lymphocyte-Induced Maturation Protein-1 (BLIMP-1)), and Notch start the development of neural crest cells following the formation of neural tube. So, a failure in these signaling pathways and aforementioned triggered transcription factors may concurrently induce the etiopathogenesis of NBL [72, 73] (Fig. 2).
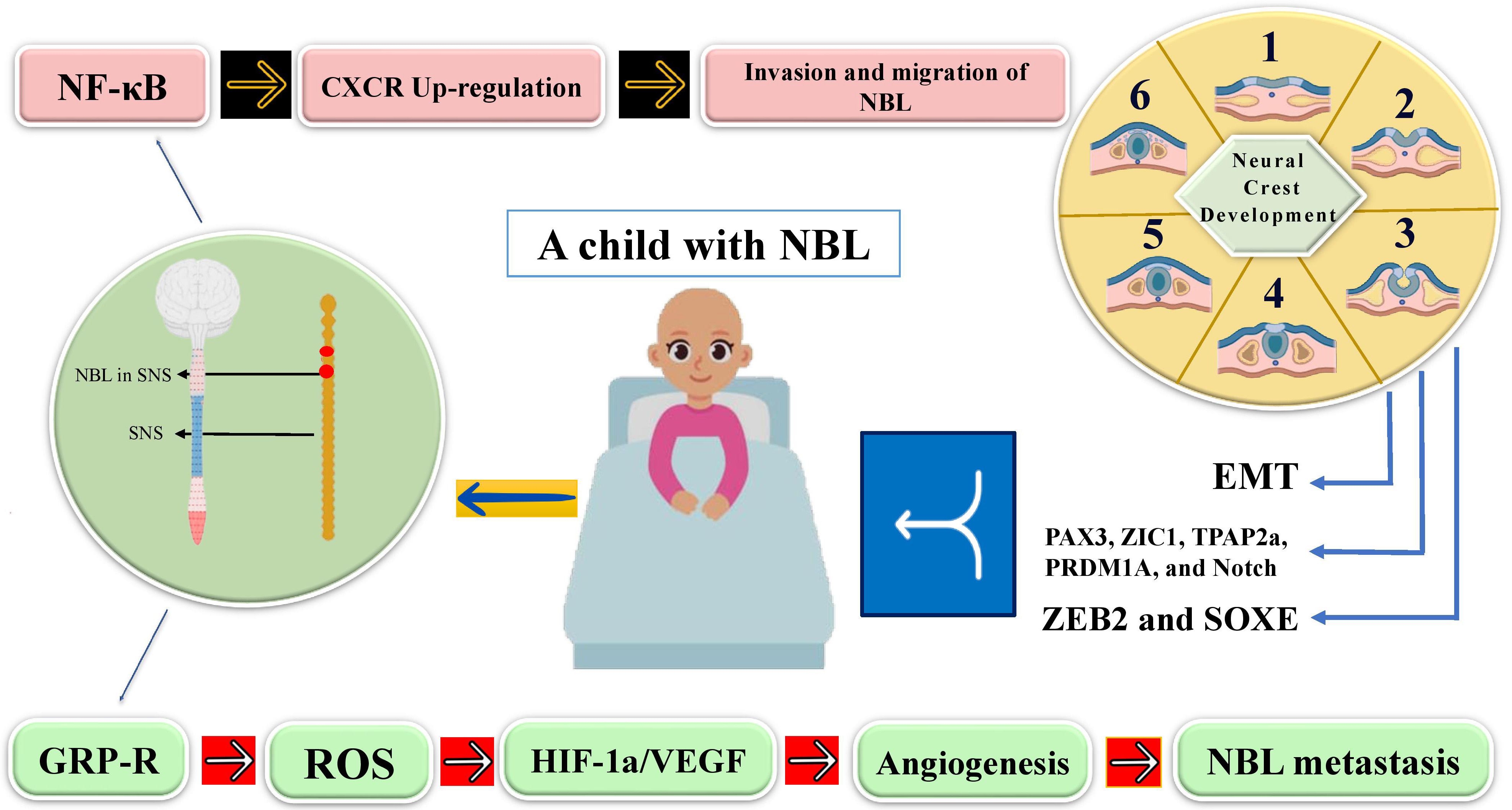 Fig. 2.
Fig. 2.
A schematic presentation of immunopathogenesis of NBL.
NF-
Moreover, it has been demonstrated that MYCN (a member of myc family) amplification has a pivotal role in the etiopathogenesis of NBL [74]. Demodulation of this transcription factor can exacerbate the procedure of tumorigenesis through affecting genetic material, such as long non-coding RNAs (lncRNAs), microRNAs (miRNAs), and messenger RNAs (mRNAs), via DNA binding and protein to protein interplay mechanisms [7].
Another molecular factor involved in the tumorigenesis of NBL is related to the biological functions of Gastrin-Releasing Peptide Receptor (GRP-R), exerting pro-tumorigenic effects in the tumorigenesis of NBL through alterations in the levels of Reactive Oxygen Species (ROS) in the microenvironment of NBL [75]. Fig. 2, depicts some of immune markers involved in the immunopathogenesis of NBL during neural crest and neural tube development.
Previous investigations in adult solid cancers have reported that the increment
in the levels of ROS formed by cancerous cells may promote the expression of
HIF-1
In case of oxygen levels in a TME, there are two distinct phases, including normoxic and hypoxic conditions. While normoxia implies to the physiological level of oxygenation in a tissue or a cell that leads to ATP production and oxidative phosphorylation, hypoxia is attributed to an inadequate level of oxygenation and anaerobic metabolism [81, 82]. Hypoxia is particularly studied as the low level of oxygen especially in a TME with an irregular kinetic in the growth of tumor cells, which has been considered as a signature for poor clinical outcomes in patients with cancer, affecting the tumor cell residing either in hypoxic or well-vascularized areas, linking hypoxia to a lower Overall Survival (OS) and Disease-Free Survival (DFS) rates, angiogenesis, immune tolerance, tumor cell survival, dysregulation of neural crest cell migration and differentiation, EMT, Cancer Stem Cells (CSCs) proliferation (like NBL stem cell proliferation), metastasis, as well as more primary and acquired resistance to the conventional therapies [82, 83, 84].
Recently, it has been demonstrated that the hypoxic events and their dual roles on the immunopathophysiology involved in a TME should not be underestimated. Functioning as a two-edge sword, one the one hand, it has been demonstrated that hypoxia can concurrently reveal immunostimulatory roles by induction of a pro-inflammatory TME (mainly through Tumor-Associated Macrophages (TAMs), as well as increased expression of C-C motif chemokine ligand 24 (CCL24)) [84, 85, 86, 87]. On the other hand, hypoxia can act as a prominent contributor to deteriorating tumor heterogenecity in solid tumors, as well as a crucial stressor driving adaptations allowing tumors to evade immune surveillance [88, 89]. To elucidate that, a comprehensive understanding on the tumor immunobiology and altered expression of immune checkpoint molecules such as Cluster of Differentiation 47 (CD47), Programmed Death Ligand 1 (PD-L1), and Human Leukocyte Antigen G (HLA G), telomerase activation, metabolic shifts (from oxidative phosphorylation to a glycolytic phenotype), metabolite alterations (hypoxia-induced glycolysis and lactate/adenosine accumulation, hypoxia-induced alterations in lipid metabolism), extracellular acidosis, cellular shifts to immature neural crest like cells (rather than neuroendocrine state), and pH regulation is of significance for hindering infiltration of immune cells [87, 89, 90]. Hence, it can be postulated that targeting hypoxia-related pathways has shown promise in enhancing T-cell-mediated tumor cell killing, suggesting a potential combinational therapeutic interventions to improve immunotherapy outcomes in solid tumors [90, 91]. To compare the hypoxic situation among different morphological and pathological statuses of NBL, it has been depicted that a less well-oxygenated condition adjacent to the necrotic zones are more common in patients with NBL than non-transformed sympathetic ganglia [84, 88, 92, 93, 94, 95, 96].
Biologically, hypoxia signaling is primarily mediated by Hypoxia-Inducible Factors (HIFs), a family of
transcription factors comprising alpha subunits (HIF-1
In case of hypoxia and cell cycle arrest, hypoxic condition can accelerate the
development of aggressive phenotypes of NBL through HIF-1
In the complex realm of immune escape, under hypoxic circumstances, there will
be an orchestrated contribution of HIF-1
In solid tumors, according to the various roles of HIF in exacerbating
angiogenesis, tumor cell proliferation, and resistance to treatment, it seems
there is an imperative need to targeting HIF and its associated pathways for
designation of targeted therapies and NBL is not an exception [21, 83, 92].
Accordingly, reduced synthesis of HIF-1
Although there is a substantial progress in the development of
anti-HIF-1
To sum up, it seems that clinical applications of those currently-used medications against NBL inhibiting hypoxia seems controversial. Hence, results of some studies imply to the neuroprotective effects of some medicinal plants via regulating cerebral hypoxia, and exerting inhibitory effects on ER stress, and inhibited apoptosis as a direct consequence during hypoxia [110, 111, 112].
Curcumin is known as a natural polyphenolic agent obtained from
turmeric plant (Curcuma longa) [113], with a wide array of
pharmacological advantages encompassing anti-cancer, anti-oxidative,
anti-protein-aggregate, and anti-inflammatory effects [47, 114, 115, 116]. Results of
several studies proved that polyphenol compounds of Curcumin has shown
potent abilities to refrain from the development of the cellular and molecular
mechanisms of carcinogenesis [47, 117, 118, 119]. Recently, great achievements in
nanotechnology and clinical advantages of drug delivery recommend us several
carrier-based systems like nanoparticles, exosomes, phytosomes, micelles,
dendrimers, liposomes, and microspheres/microbubbles systems aimed at increasing
the solubility, absorption, and bioavailability of curcumin to be used as a more
efficacious cancer treatment [120, 121, 122, 123, 124]. Also, from molecular immunological
viewpoints, Curcumin can down-regulate those immune genes involved in
NF-
In case of NBL, it has been demonstrated that Curcumin stimulates the expression
of apoptosis-related genes and diminishes p53 levels in N2a cell lines (as a
progressive NBL cell line), via a dose-dependent manner [126]. According to the
accumulative evidence, Curcumin exerts apoptotic effects by inhibition
of NF-
Green tea is a type of herbal product that is originated from
Camellia sinensis and can be produced as a beverage [142]. It
is worthy to mention that Green tea has significant therapeutic
applications, such as anti-cancer, anti-inflammatory, anti-angiogenic,
neuroprotective, anti-viral, anti-oxidative, and anti-bacterial effects
[143, 144, 145, 146, 147, 148, 149]. Green tea comprises multiple polyphenols, like flavonoids
[150]. Catechins of green tea, a flavonoid-based and polyphenolic formulations
with anti-oxidant and anti-cancer activities, are the main phytoconstituents of
Green tea. Among those catechins, Epigallocatechin Gallate
(EGCG) and Epigallocatechin (EGC) are the most important molecules [151, 152].
Catechins biologically affect WNT/
In case of NBL, it has been demonstrated that Green tea may be an effective herb on the number of neurite in NBL cell lines [151]. Results from an experimental study investigating the role of Polyphenon E in the inhibition of NBL indicated a reduction in the number of tumor-infiltrating myeloid cells in TH-MYCN transgenic mice, as well as impairing MDSCs and CD8 T cells in vitro in A/J mice (but not in immunodeficient NOD/SCID mice), leading to the development of neutrophilic forms through the 67 kDa laminin receptor signaling and induction of Granulocyte Colony-Stimulating Factor (GC-SF). In order not to interfere with the anti-tumor immune responses in patients with NBL, findings of this study proved an in vivo pharmacological manipulation of neuroblastoma-promoting activities of MDSCs through clinical advantages of catechins of green tea [154].
Accordingly, EGCG can stimulate apoptosis and growth arrest in NBL cells [155]. It seems that overexpression of miR-7-1 (Fig. 3) can mediate the induction of apoptosis by polyphenols in human NBL cells [156]. MiR-7-1 belongs to the family of Tumor Suppressor microRNAs (TSmiRs), suppressing the growth and proliferation of NBL cells amplified with N-Myc [156, 157]. Furthermore, it is interrogated that EGCG is capable of arriving at the brain parenchyma, and brain plasticity can be improved even by the low concentrations of EGCG in the brain tissues [151]. In addition, EGCG regulates mitochondrial membrane potentials, ATP levels, and mitochondrial respiration in animal NBL N2a cells induced with amyloid [158].
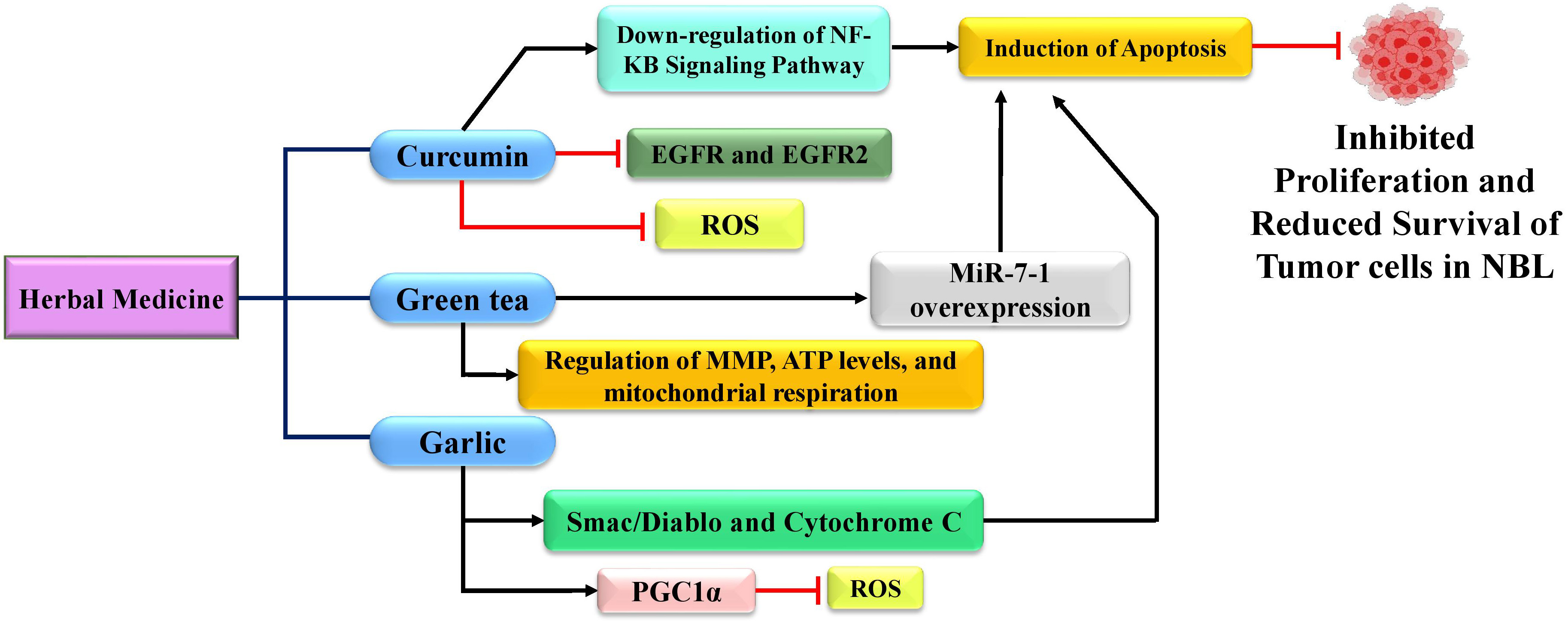 Fig. 3.
Fig. 3.
Herbal remedies (curcumin, green tea, and garlic) used for the
treatment of NBL by changing several processes in the etiopathogenesis of NBL. PGC1
Garlic (Allium sativum) has been defined as a native plant from Asia and a functional food, which is presently cultivated all around the world [159]. Even from cultural aspects, Garlic has been broadly utilized for treatment of cancers, cardiovascular diseases, and diabetes [160]. Additionally, anti-inflammatory, anti-bacterial, anti-hypertensive, anti-thrombotic and anti-oxidative properties of Garlic are of much clinical importance [161, 162, 163]. Results acquired from several clinical studies have mentioned the substantial roles of phytocomponents of Garlic in cancer prevention [164, 165, 166]. There is a positive relationship between oral consumption of Garlic and a decreased risk of stomach, colon, pancreas, breast, and esophagus cancers [167, 168, 169, 170]. Regardingly, it has been interrogated that S-allyl-L-Cysteine (SAC), a component derived from the extract of aged Garlic, can suppress the cellular growth of human NBL cells in vitro [171].
Interestingly, clinical advantages of Diallyl Disulfide (DADS) has been under
investigation in a wide array of experimental studies. From chemical aspects,
DADS includes an organosulfur including two sulfur atoms with two allyl groups.
As one of pharmacologically-active bio-component of garlic, DADS has revealed
anti-bacterial, anti-fungal, anti-viral, detoxifier, anti-inflammatory,
neuroprotective, anti-oxidant, and anti-cancer properties. Literally, anti-cancer
properties of DADS majorly involves inhibiting the expression of Matrix metalloproteinase-9 (MMP-9), MMP-2,
and MMP-7, reversing EMT by inhibiting the
In addition, DADS can stimulate the expression of Peroxisome
proliferator-activated receptor Gamma Co-activator 1 alpha (PGC1
According to the considerable mortality and morbidity rate of NBL, and inefficacies of current diagnostic and therapeutic approaches for all of the patients with NBL, it seems that there is an imperative need for more highly-sensitive and efficient medical interventions for a better clinical management and targeted therapy of NBL. Accordingly, alterations in the molecular immunopathophysiology involved in the microenvironment and etiopathogenesis of NBL by phytoconstituents of medicinal herbs can be a contributing approach in the development of “herbal molecular medicine” and “precision medicine” for treatment of NBL. There are a wide range of studies demonstrating the clinical roles of medicinal plants (or their phytoconstituents/pharmacologically-active bio-compounds/aromatic plants/hydroalcoholic extracts/phenolic compounds) aimed at inhibiting the development of cancers through a lower expression of VEGF and Terminal deoxynucletidyl transferase dUTP Nick End Labeling (TUNEL) staining, as well as higher endothelial Nitric Oxide Synthase (eNOS) expression, and NBL is not an exception [33, 182, 183, 184, 185, 186, 187, 188, 189, 190, 191, 192, 193, 194, 195, 196].
Surprisingly, according to the role of plaque/amyloid-
Berberine is one of the well-known herbal alkaloid agents with
experimentally- and clinically-anti-cancer properties. It is documented that
Berberine can biologically curb the growth of human NBL cells by
stimulating p53-mediated apoptosis. Totally, it has been interrogated that
clinical advantages of Berberine on inducing neuronal differentiation, inhibiting
cancer-stemness and EMT and underlying signaling have been clarified by
laboratory techniques like immunofluorescence, real-time polymerase chain reaction (RT-PCR), and western blotting
[201, 202, 203]. Increased neuronal differentiation by the usage of Berberine was
verified through increased biomarkers of neuronal differentiation like MAP2,
Interestingly, in an experimental study, phytocompounds of Azadirachta indica were investigated whether they had inhibitory effects on Glycogen Synthase Kinase-3 (GSK-3) as an active serine/threonine kinase responsible for the cell proliferation. The authors of this study reported that in a dose-dependent manner, some phytoconstituents like gedunin extracted from the seeds of Azadirachta indica (with a high gastrointestinal absorption, acceptable aqueous solubility or membrane permeability, and fewer adverse effects) had depicted inhibitory effects on NBL through diminishing the cell viability in human NBL (SH-SY5Y) cells, as well as stimulatory effects on apoptosis, ROS, and cell cycle arrest [210].
In another experimental study on NBL, the researchers tried to clarify the clinical advantages and anti-tumor activities of Triptolide, which is a diterpene triepoxide extracted from the Chinese herb Tripterygium wilfordii Hook F. They reported inhibitory effects of Triptolide on the cell growth and tumor development in NBL in vivo (especially the ones resistant to chemotherapeutics) through increasing the expression levels of the apoptosisassociated proteins, caspase3 and caspase9, together with cell cycle arrest in the S phase [211]. Results of another experimental and comparative study on the neuroprotective effects of Pien Tze Huang, a Chinese traditional herbal extract, on neuroblastoma cells (SH-SY5Y) proved a decreased survival of cancerous cells in a dose-dependent manner [212]. It seems that clinical advantages of Triptolide in Chinese herb and Pien Tze Huang can be a good candidate for the treatment of NBL even in cases resistant with other therapeutics.
In another experimental study, the researchers tried to investigate the clinical
advantages of Thymoquinone (TQ), which is a bio-active compound derived from
Nigella sativa. They reported inhibitory effects of TQ on the adhesion
and cellular migration of Neuro-2a cells, functioning as a potent cytotoxic agent
for inducing apoptosis through down-regulating the expression of
NF-
In case of NBL, there are a swift incidence in the mortality rate, an increasing consideration due to the inefficacies of recently-used medications, and existed biological limitations of those newly-authorized medications. These points confirm clinical applications of more efficacious procedures like targeted molecular herbal medicine for treatment of NBL.
Regardingly, administrating pharmacologically-active bio-compounds of medicinal plants is triggering for induction of immunoregulatory genes in the microenvironment of NBL, reminding us of the clinical proficiency of immune gene manipulation-based immunotherapeutics by phytochemicals, and providing a platform for NBL molecular herbal medicine. Additionally, according to the dose-dependent inhibitory functionalities of phytocompounds against NBL, they can be alternatively proposed as a neoadjuvant in the structure of the cell-based vaccines, providing a promising prognostic and therapeutic strategy in NBL precision medicine.
Accordingly, further investigations and an interdisciplinary collaboration among pediatric neuro-oncologists, cellular and molecular cancer immunotherapists, medicinal nanochemists, phytochemists, molecular pharmacognosists, personalized medicine specialists, phytomedicine specialists, molecular medicine specialists, translation medicine specialists, traditional medicine specialists, complementary medicine specialists, alternative medicine specialists, naturopathic medicine specialists, experimental medicine specialists, vaccine researchers, biomolecule scientists, and health system coordinators are highly recommended.
Supervision, OOG; Conceptualization and Study Design, OOG, SR, and NB; Methodology, SR, and NB; Search Strategy, SR, and NB; Investigation, SR, KB, SMSM, YB, KRD, FS, MM, HZ, EN, MA, MB, AK, and NB; Literature Review, SR, KB, SMSM, YB, KRD, FS, MM, HZ, AK, EN, MA, MB, and NB; Visualization, Image, and Table Designation, SR, KB, SMSM, YB, KRD, FS, MM, HZ, and AK; Data Acquisition, SR, and NB; Academic, Scientific; Definition of Intellectual Content, OOG, ES, and PÖ, MB, and NB; Investigation of the Clinical and Experimental Studies, SR, KB, SMSM, YB, KRD, FS, MM, HZ, EN, MA, MB, AK, and NB; Formal Analysis, SR, and NB; Preparation of the first draft of the manuscript, SR, KB, SMSM, YB, KRD, MM, FS, HZ, and NB; Grammatical Revision, NB, OOG, ES, and PÖ; Preparation of the last draft of the manuscript, AK, EN, MA, MB, and NB. All of the authors listed on the title page attest to the fact that they have read and approved the final version of the manuscript, and they have received an electronic copy of the manuscript. All of the authors participated fully in this work, take public responsibility for appropriate parts of the content, and agree to be accountable for all aspects of the work and to ensure that questions related to the accuracy or completeness of the content are addressed.
Not applicable.
All of the authors would like to sincerely dedicate this article to all of the oncologists, neurologists, cancer researchers, plant biologists, basic medical scientists, and healthcare personnel all around the world.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



