1 Department of Gynecology, Jincheng Hospital Affiliated to Changzhi Medical College, Jincheng People’s Hospital, 048026 Jincheng, Shanxi, China
2 Department of Gynecology, Liuzhou Maternity and Child Healthcare Hospital, 545001 Liuzhou, Guangxi, China
3 State Key Laboratory of Ultrasound in Medicine and Engineering, College of Biomedical Engineering, Chongqing Medical University, 400016 Chongqing, China
†These authors contributed equally.
Abstract
Endometriosis is a complicated and enigmatic disease that significantly diminishes the quality of life for women affected by this condition. Increased levels of human telomerase reverse transcriptase (TERT) mRNA and telomerase activity have been found in the endometrium of these patients. However, the precise function of TERT in endometriosis and the associated biological mechanisms remain poorly understood.
We analyzed TERT expression in ectopic endometrial (EC), eutopic endometrial (EU), and normal endometrial (NC) tissues. Human endometrial stromal cells (HESCs) were used to study the effects of TERT depletion and knockdown on cell behavior. We also assessed methyltransferase-like 3 (METTL3)-mediated N6-methyladenosine (m6A) modification in TERT transcripts and its impact on mRNA stability and cell functions.
The current results indicate that TERT expression is elevated in EC tissue compared to both EU and NC. Depletion of TERT suppressed the proliferation and migration of HESCs, while TERT overexpression had the opposite effect. We found high levels of METTL3-mediated m6A modification in TERT transcripts, particularly in the coding sequence region, resulting in increased translation. However, EC tissues had lower m6A levels due to the downregulation of METTL3. Mechanistically, m6A modification mediated by METTL3 negatively regulates the stability of TERT mRNA in a YTH N6-methyladenosine RNA binding protein 2 (YTHDF2)-dependent manner. Furthermore, METTL3 negatively regulated the proliferation and migration of HESCs.
Together, our study identified a new molecular mechanism that underlies the pathogenesis of endometriosis. Inhibition of m6A modification and of the METTL3/TERT axis may enhance cellular proliferation and migration, thereby contributing to the progression of endometriosis.
Keywords
- endometriosis
- TERT
- METTL3
- YTHDF2
Endometriosis is defined by the occurrence of tissue similar to the endometrium located outside the uterus [1], and it is linked to persistent pelvic discomfort and female infertility. This condition adversely impacts the patients’ quality of life, sexual function, and personal relationships [2, 3, 4, 5]. More than 200 million women worldwide are affected by this condition, representing over 10% of females of reproductive age [6]. Ovarian endometriosis is the most prevalent form of endometriosis [7]. Monthly, the ectopic endometrial tissue on the ovary undergoes continuous proliferation, shedding, and bleeding, which progressively accumulates and leads to the formation of an ovarian endometriotic cyst, commonly referred to as a chocolate cyst. Ovarian endometriosis is particularly challenging to manage due to its unique pathophysiological characteristics. The formation of endometriomas often leads to ovarian damage, including inflammation, fibrosis, and scarring, which can severely impact ovarian reserve and function [8]. Moreover, ovarian endometriosis is frequently associated with more severe disease presentations, including deep infiltrating endometriosis and extensive pelvic adhesions, which complicate both clinical management and surgical intervention [9]. Despite the high prevalence and significant impact on quality of life, the underlying molecular mechanisms driving ovarian endometriosis remain poorly understood. Despite being a non-cancerous condition, endometriosis exhibits features reminiscent of malignancy, including abnormal cell migration and invasion. These factors contribute to endometriosis and to its recurrence after surgery. Despite extensive research over the past few decades into the multifactorial causes of endometriosis [10, 11, 12, 13], the underlying mechanism remains unclear.
Telomerase plays a vital role in preserving telomere length and ensuring cellular immortality [14, 15, 16]. With the exception of hematopoietic stem cells and endometrial cells, telomerase is generally not expressed in normal somatic cells. Nonetheless, telomerase activity is present in various cancers and is linked to their capacity for limitless replication. Human telomerase reverse transcriptase (hTERT) is a catalytic protein subunit essential for telomerase function. A significant relationship has been noted between hTERT mRNA levels and telomerase activity across different tissues [17, 18], with hTERT mRNA serving as a key factor in regulating telomerase activity [19]. Telomerase activity and hTERT mRNA levels in endometrial cancer are significantly higher than in the normal cycling endometrium [20]. Interestingly, telomerase activity and hTERT mRNA expression in patients with endometriosis correlate with the proliferative potential of the endometrium [21]. These findings indicate that TERT plays an essential role in the adhesion of endometrial cells, and that abnormal TERT expression may contribute to the pathological processes involved in endometriosis. However, this connection has yet to be investigated, and the upstream regulatory mechanisms of TERT remain unknown.
N6-methyladenosine (m6A) is among the most common modifications found in RNA [22, 23]. Like DNA methylation, m6A exhibits dynamic and reversible regulatory properties. This modification is added by a methyltransferase complex comprising two primary subunits [24]. Demethylation of m6A requires the action of fat mass and obesity-associated protein (FTO) [25] and alkB homolog 5 (ALKBH5) [26]. The equilibrium between m6A methyltransferases and demethylases is essential for the dynamic regulation of m6A. Increasing evidence indicates that m6A plays a significant role in various disease processes, particularly in cancer metastasis [27]. Conversely, reduced methyltransferase-like 14 (METTL14) expression promotes hepatocellular carcinoma cell metastasis by influencing the processing of m6A-dependent primary microRNA (pri-miRNA) [28]. However, the precise role of m6A and its associated mechanisms in the development of endometriosis remains elusive and requires further investigation. This study was to explore the influence of m6A modification on the upregulation of TERT expression observed in ectopic endometrial tissue. A better understanding of the epigenetic regulation of TERT via m6A modification may shed light on the pathogenesis of endometriosis and identify potential therapeutic targets for this condition.
This study provides evidence that reduced m6A modification is a key driving factor for the aberrant expression of TERT in endometriosis. Our findings highlight the significance of methyltransferase-like 3 (METTL3)/TERT in the development of endometriosis and suggest that TERT could serve as a novel target in the diagnosis and therapy of this condition.
Individuals with ovarian endometriosis were chosen from the Obstetrics and Gynecology Department at Liuzhou Maternal and Child Health Hospital. Post-surgery, the endometrial tissues were categorized into eutopic endometrial (EU) and ectopic endometrial (EC). EU samples were sourced from the endometrial lining within the uterus, while EC samples were taken from the ovarian tissue. We included women aged 18–45 with confirmed ovarian endometriosis and excluded those with malignancies, autoimmune diseases, other gynecological conditions, recent hormone therapy (within six months), or systemic diseases. To control for age, we focused on women aged 18–45 and ensured all samples were collected during the proliferative phase of the menstrual cycle without recent hormone treatment to minimize hormonal and systemic effects. Normal endometrium (normal control; n = 8) was obtained during the surgical operation. Human tissue samples were obtained with patients’ or their families/legal guardians’ written informed consent, in accordance with the Declaration of Helsinki guidelines. The research received approval from the Ethics Committee of Liuzhou Maternal and Child Health Hospital (KS-KY-2020-073).
Human endometrial stromal cells (HESCs) were sourced from Otwo Biotech (HTX2487-2, Shenzhen, China) and cultured with DMEM as previously [29]. The culture medium was enriched with 1.5 g/L sodium bicarbonate (144-55-8, Sigma-Aldrich, St. Louis, MO, USA), 1% recombinant Human Insulin (ITS)+ Premix (354352, Corning, Corning, NY, USA), 500 ng/mL puromycin, and 10% charcoal-stripped fetal bovine serum (F6765, Sigma-Aldrich), and kept in a humidified incubator at 37 °C in a 5% CO2 environment. All cell lines were validated by short tandem repeat (STR) profiling and tested negative for mycoplasma. 5-Azacytidine (5-aza-dC) (HY-10586) and Suberoylanilide Hydroxamic Acid (SAHA) (HY-10221) were obtained from MCE (Middlesex County, NJ, USA), while sodium butyrate (NaB) (S1999) was acquired from Selleck. The expression of TERT in HESC cells was assessed by real-time quantitative PCR (qPCR) after treatment for 12 hours with SAHA (5 µM) or NaB (5 mM), or with 5 µM 5-Aza-dC for 48 hours.
To overexpress TERT and METTL3 in HESCs, human TERT and METTL3 were synthesized and cloned into the pLVX plasmid vector (General Biosystems, Anhui, China). For the knockdown of TERT and METTL3, lentiviruses containing small interfering RNAs (siRNAs) targeting TERT (5′-AAAAATGTGGGGTTCTTCCAA-3′), METTL3 (5′-TCTAACTCAGGATCTGTAGCT-3′), or a nonsense siRNA (5′-CAGCCATCAACTCAGATTGTT-3′) were constructed by General Biosystems (Anhui, China). Recombinant lentiviral vectors or control vectors, along with two auxiliary packaging plasmids (pSPAX2 and pMD2G), were co-transfected into HEK293T cells to produce the lentiviruses, as provided by General Biosystems (Anhui, China). After 48 hours, the supernatant was collected and used to infect HESCs with an initial multiplicity of infection (MOI) of 50. Transduction efficiency was assessed 48 hours post-infection using Western blot analysis (Supplementary Fig. 1).
The retrieved tissue specimens were promptly preserved in 4% buffered formalin and subsequently prepared for immunohistochemical (IHC) analysis by Servicebio Biotechnology Co., Ltd. (Wuhan, China). Paraffin-embedded slices were subjected to staining with a primary antibody targeting METTL3 (1:200, Proteintech, 15073-1-AP, San Diego, CA, USA) according to the manufacturer’s instructions.
To assess m6A levels in total RNA samples, an EpiQuik m6A RNA Methylation Quantification Kit (Epigentek, P-9005-48, Farmingdale, NY, USA) was utilized. Initially, the RNA was applied to wells pre-coated with RNA high binding solution, then capture and detection antibody solutions were introduced. The m6A levels were determined by measuring absorbance at 450 nm and calculating relative quantification.
Cell proliferation was evaluated through the Cell Counting Kit-8 (CCK-8) assay (MCE, HY-K0301). Cells were seeded in a 96-well plate and treated as needed. After treatment, 10 µL of CCK-8 solution was added to each well and incubated at 37 ℃ for 1–4 hours. Absorbance at 450 nm was then measured using a microplate reader to assess cell proliferation and viability. For 5-ethynyl-2’ -deoxyuridine (EdU) staining, the culture medium was supplemented with 10 µM EdU reagent (Beyotime Biotechnology, C0078S, Shanghai, China) and incubated for 3 hours at 37 °C. Cells were then stained with Hoechst 33342 as per the manufacturer’s instructions and examined under a fluorescence microscope.
Cell invasion and migration were assessed using Transwell chambers (Corning, Corning, NY,
USA). For invasion assays, Matrigel (Corning, 354248) was mixed with fetal bovine
serum (FBS)-free DMEM/F12 medium at a 1:3 ratio and applied to the upper chamber.
For migration assays, Matrigel was not used. Cells (8
Once cells reached 90–100% confluence, a sterilized pipette tip (200 µL) was used to create scratches. Following PBS washes to remove detached cells and debris, wound healing was monitored and photographed at 0 and 24 hours post-scratching. The wound area at each time point was compared to the initial size at 0 hours, and the percentage of area closure was calculated.
The RIP assay was conducted with the Magna™ RIP RNA-Binding Protein Immunoprecipitation Kit (Millipore, 17-700, Burlington, MA, USA). Cells were lysed using a RIP lysis buffer, and magnetic beads were conjugated with human antibodies against YTH N6-methyladenosine RNA binding protein 2 (YTHDF2) (Proteintech, 24744-1-AP) or m6A (Synaptic Systems, 202-003, Gottingen, Germany). Normal mouse Immunoglobulin G (IgG) antibody (Millipore, 12-370) was used as a negative control. RNA obtained with the RIP assay was assessed by RT-qPCR and normalized to the input.
Total protein was extracted with RIPA buffer (Beyotime, P0013B). Following protein quantification, SDS-PAGE separation, and transfer onto polyvinylidene difluoride (PVDF) membranes were performed. The membranes were treated with 5% skim milk to prevent non-specific binding, and then incubated with the specified antibodies: TERT (1:1000, Thermo, MA5-16034, Waltham, MA, USA), METTL3 (1:2000, Cell Signaling Technology, #86132, Danvers, MA, USA.), YTHDF2 (1:2000, Abcam, ab246514, Cambridge, UK), YTHDF3 (1:2000, Abcam, ab220161), IGF2BP1 (1:2000, Abcam, ab290736), IGF2BP2 (1:2000, Abcam, ab124930), IGF2BP3 (1:2000, Abcam, ab177477), and GAPDH (1:6000, Proteintech, 60004-1-Ig) overnight at 4 °C. Subsequent incubation with secondary antibodies was followed by detection of protein bands using ECL chemiluminescent reagent (Beyotime, P0018S) and densitometry analysis using ImageJ (version 1.41, LOCI, University of Wisconsin, Madison, WI, USA).
The luciferase reporter assay was carried out utilizing the Dual-Luciferase Reporter Assay System. Cells were transfected with a pmiGLO-based luciferase vector carrying either wild-type or mutated TERT, or a control vector, employing Lipofectamine 3000 reagent (Invitrogen, Waltham, MA, USA). After 24 hours, the luciferase and Renilla activities were measured according to the manufacturer’s guidelines. For further analysis, pmirGLO-TERT-3′UTR-WT and pmirGLO-TERT-3′UTR-Mut constructs were introduced into control or shMETTL3 cells for 24 hours, and F-luc and R-luc activities were evaluated.
To measure RNA stability, cells were treated with 5 µg/mL actinomycin D (Act-D, Sigma-Aldric, #A9415). RNA samples were collected at various time intervals post-treatment, and real-time PCR was performed to analyze RNA levels. The TERT mRNA half-life (t1/2) was calculated with the formula ln2/slope, using GAPDH as the reference control.
RNA was extracted with Trizol reagent (Invitrogen, A33250). For cDNA synthesis, 500 ng of mRNA was reverse transcribed with the PrimeScript RT Master Kit (Takara, Dalian, China). qPCR was performed using TB Green Premix Ex Taq II (TaKaRa, RR820Q), and transcript levels were determined based on the threshold cycle number. GAPDH served as the normalization control, and relative expression was calculated using the 2-ΔΔCt method. The primer sequences are listed in Supplementary Table 1.
Results are expressed as mean
To explore the involvement of TERT in endometriosis, we first evaluated its expression levels using Western blotting and qRT-PCR across normal endometrial (NC), EU, and EC. Our analysis revealed a notable elevation in TERT expression in EC relative to both EU and NC (Fig. 1A,B). No significant variation in expression levels was detected between EU and NC. To confirm these findings, we conducted immunohistochemistry (IHC) to assess TERT expression across NC, EU, and EC samples. The IHC results verified higher TERT expression in EC relative to NC and EU (Fig. 1C), supporting the results obtained from qRT-PCR and Western blot analyses. This indicates that increased TERT levels may play a role in the progression of endometriosis.
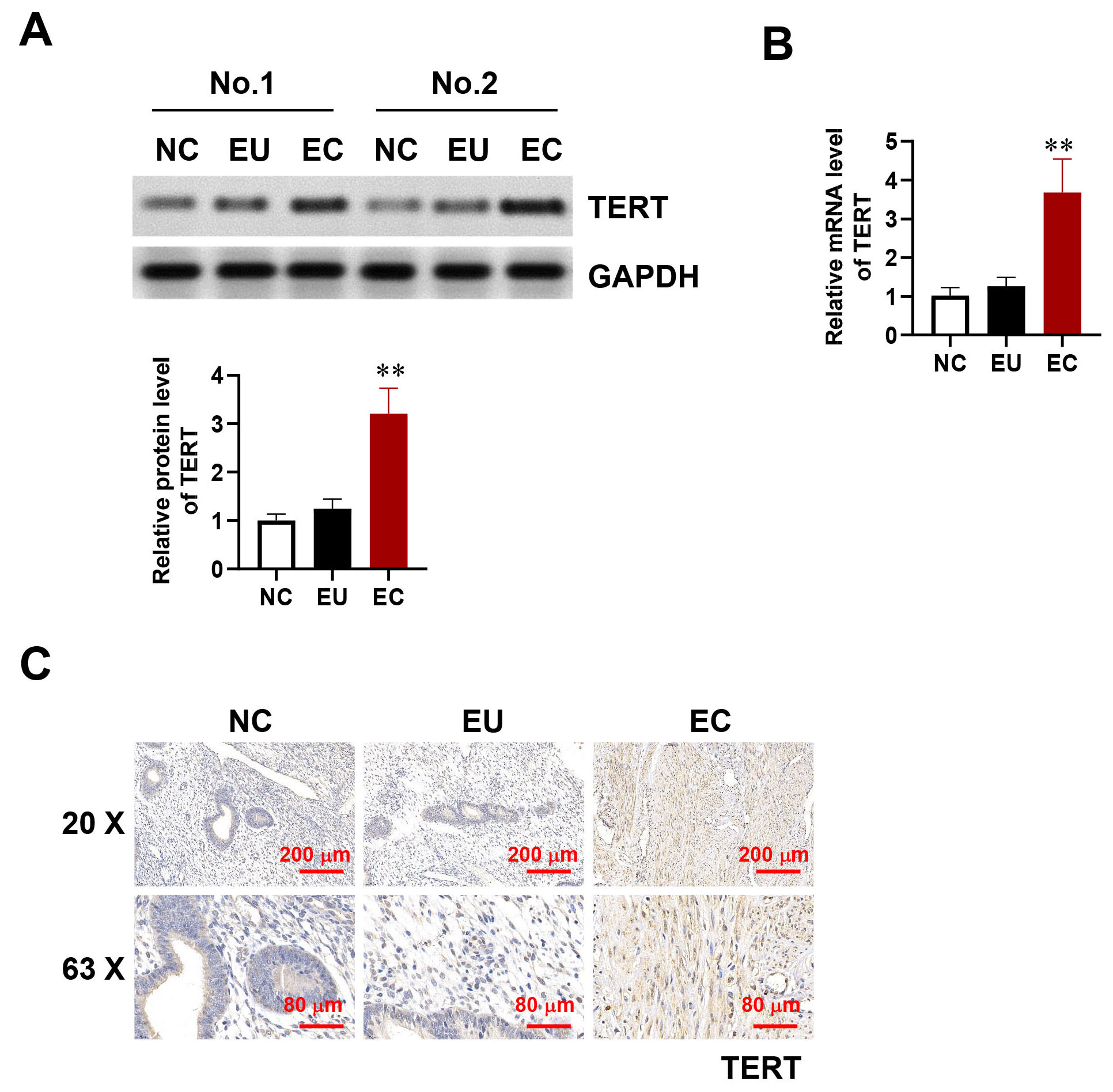 Fig. 1.
Fig. 1.
TERT overexpression in endometriosis. (A,B) Protein (A) and
mRNA (B) expression levels of TERT in NC, EU, and EC tissue. (C)
Immunohistochemistry of TERT expression in the endometriosis and control groups.
Scale bar: 200 µm or 80 µm. Error bars represent the mean
To explore the potential biological function of TERT in HESCs, lentiviruses were used to knockdown (shTERT) or overexpress TERT. EdU assay and CCK-8 assays were then performed to evaluate cell viability following TERT knockdown or overexpression. TERT knockdown significantly reduced the proliferation of HESCs, whereas TERT overexpression had the opposite effect (Fig. 2A,B).
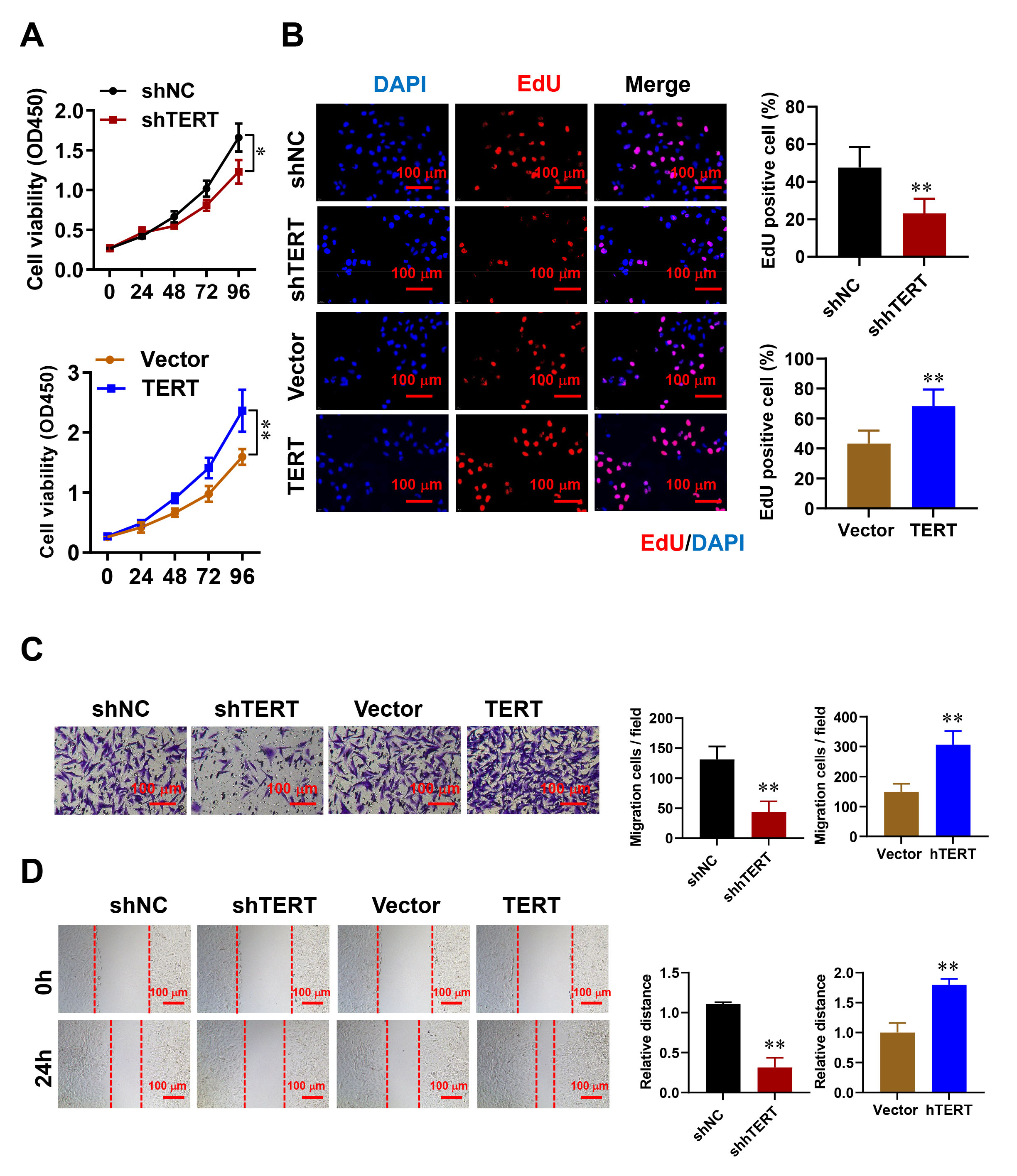 Fig. 2.
Fig. 2.
The impact of TERT on HESC proliferation, migration, and
invasion. (A,B) The influence of TERT silencing or overexpression on HESC
proliferation was determined by CCK-8 assay (A) and 5-ethynyl-2’ -deoxyuridine (EdU) incorporation (B) Scale
bar: 100 µm. (C) Transwell assays were conducted to evaluate migration
(without matrigel) and invasion (with matrigel) 24 hours post-TERT manipulation.
Representative images and quantification are displayed. Scale bar: 100 µm.
(D) The wound healing assay was employed to measure cell motility at 0 and 24
hours, with representative images and quantitative data provided. Scale bar:
100 µm. Error bars denote the mean
Next, the migratory and invasive abilities of HESCs with altered TERT expression were investigated using wound healing and transwell assays. TERT overexpression was found to promote cell migration and invasion, whereas TERT knockdown inhibited these processes (Fig. 2C,D). These findings indicate that TERT plays a crucial role in promoting cell proliferation and migration during endometriosis.
Recent studies demonstrated the involvement of epigenetic mechanisms in gene dysregulation. We therefore investigated whether epigenetic factors contribute to the upregulation of TERT in endometriosis. TERT expression remained unchanged following the treatment of HESCs with a DNA methyltransferase inhibitor (5-aza-dC), suggesting that DNA methylation does not play a role in regulating TERT. Furthermore, the application of broad-spectrum histone deacetylase (HDAC) inhibitors SAHA and NaB did not affect TERT expression, indicating that histone acetylation is not involved in TERT regulation in HESCs (Supplementary Fig. 2).
m6A modification in endometriosis was investigated by measuring m6A levels in NC, EU, and EC using a colorimetric method. The results showed that m6A levels were lower in EC compared to NC and EU. No significant difference in the m6A level was observed between EU and NC (Fig. 3A). The expression of m6A-associated genes in NC, EU, and EC was subsequently evaluated in order to identify the key molecules responsible for reduced m6A in endometriosis. Both the mRNA and protein levels of METTL3 were found to be downregulated in EC relative to NC and EU (Fig. 3B–D). These findings were consistent with the lower m6A levels, suggesting a potential role for METTL3 in m6A modification in endometriosis. Moreover, overexpression of METTL3 was found to significantly reduce TERT mRNA and protein expression, while reducing METTL3 levels led to an increase in TERT expression relative to normal controls (Fig. 3E,F).
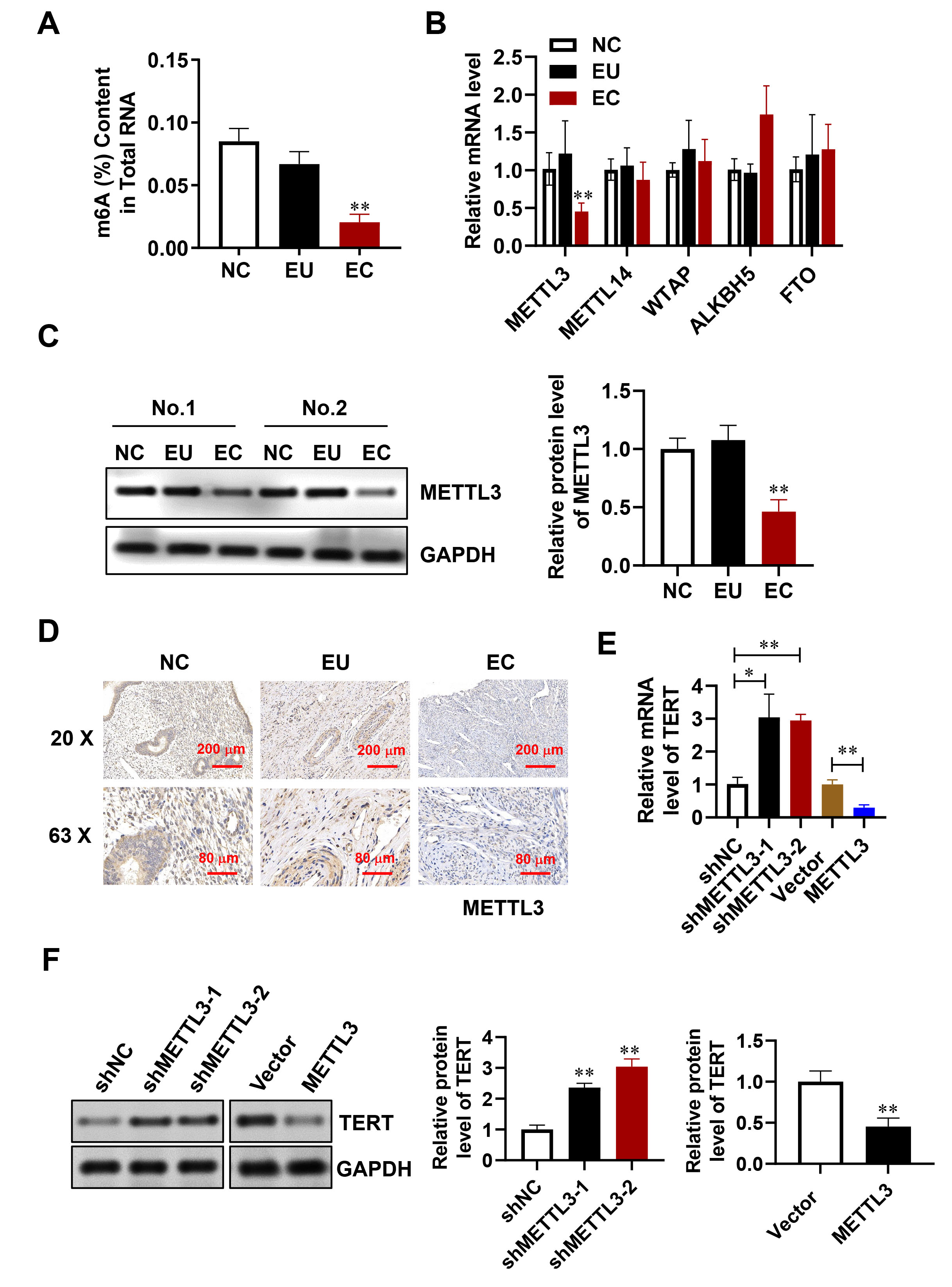 Fig. 3.
Fig. 3.
METTL3 decreases m6A levels in endometriosis. (A) m6A content
in NC, EU, and EC tissue. (B) mRNA expression of m6A-related genes measured by
quantitative Reverse Transcription PCR (qRT-PCR). (C) METTL3 protein levels assessed through Western blotting. (D) METTL3
localization in tissues identified via Immunohistochemistry (IHC). Scale bar: 200 µm or 80 µm. (E,F) TERT
mRNA levels evaluated by qRT-PCR (E) and Western blotting (F). Error bars
indicate the mean
We investigated how TERT influences METTL3-driven cell proliferation, migration, and invasion in HESCs by introducing a METTL3-overexpressing lentivirus into cells with TERT overexpression. It was observed that elevated METTL3 levels markedly reduced cell proliferation, migration, and invasion. In contrast, TERT overexpression counteracted these suppressive effects (Fig. 4A–D). These findings indicate that TERT plays a role in modulating the METTL3-mediated inhibition of HESC proliferation, migration, and invasion.
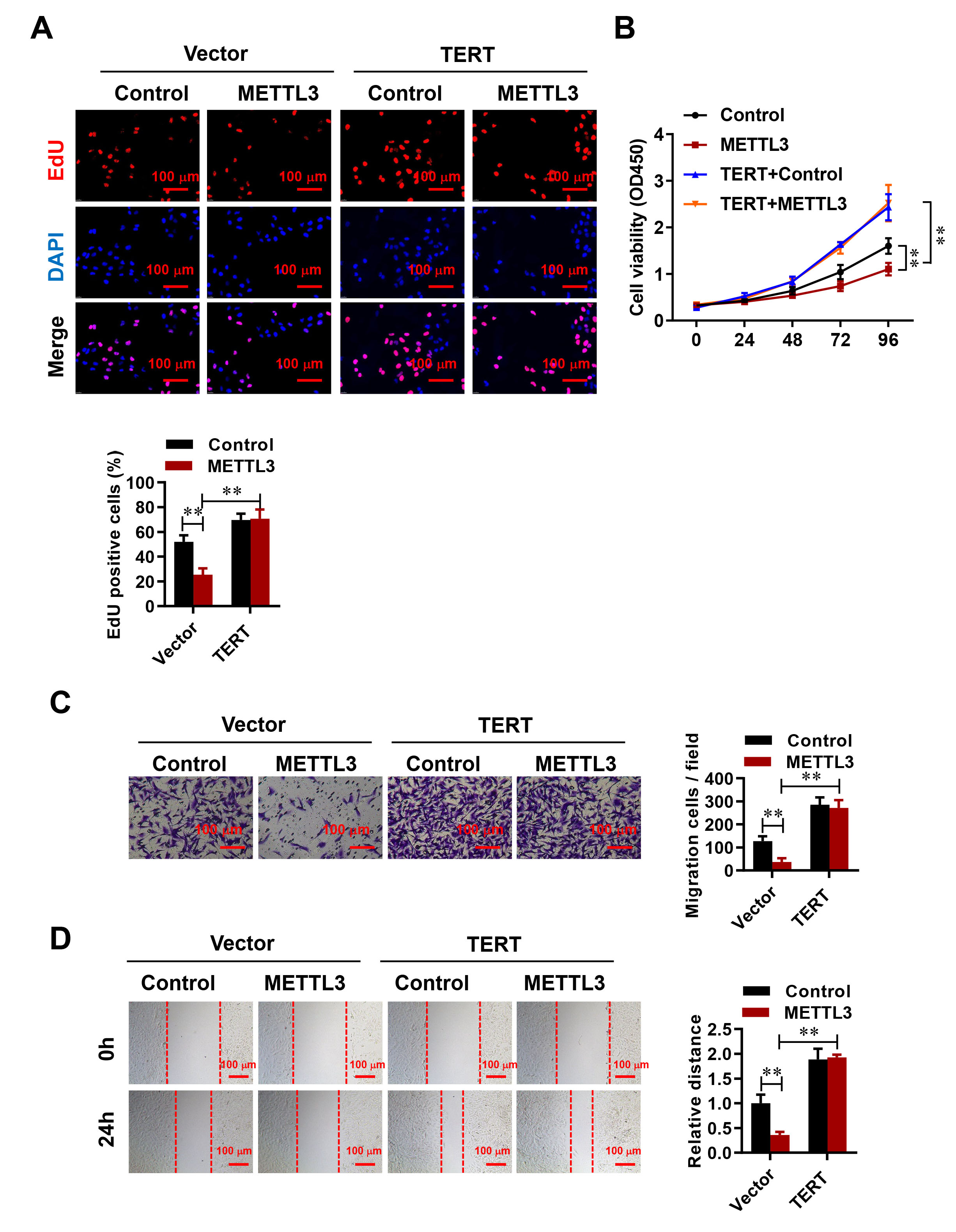 Fig. 4.
Fig. 4.
METTL3 regulates HESC proliferation, migration, and invasion via
TERT. (A,B) The EdU (A) and CCK-8 assay (B) were used to evaluate the effect of
negative control and METTL3-overexpressing vector on HESC proliferation, both in
the absence or presence of TERT overexpression. Scale bar: 100 µm. (C)
Transwell assays were performed 24 h after transfection. Scale bar: 100 µm.
(D) Changes in cell motility were assessed through wound healing assays at 0 and
24 h. Representative images and quantification are presented. Scale bar:
100 µm. Error bars represent the mean
Emerging evidence suggests that m6A influences various aspects of RNA fate [30, 31, 32]. We next investigated the role of m6A modification in TERT upregulation. Data from the online RNA modification resource (RMBase, http://rna.sysu.edu.cn/rmbase/) indicate that multiple m6A sites are present within the TERT transcript (Supplementary Fig. 3). In order to validate whether TERT is a target of METTL3-mediated m6A modification, we performed MeRIP-qPCR in HESCs. A remarkable decrease in m6A modification of TERT was observed when METTL3 was knocked down (Fig. 5A). Furthermore, the distribution of m6A methylation in TERT mRNA was investigated using fragmented RNA isolated from HESCs. The highest level of m6A methylation was observed in the 3′ untranslated region (UTR) of TERT (Fig. 5B). Two m6A modification sites were identified within the 3′UTR of TERT (Fig. 5C). To investigate the potential role of m6A in the 3′UTR, luciferase reporters containing the wild-type or mutant TERT m6A methylation sites were generated (Fig. 5D). A firefly luciferase reporter assay demonstrated that, in HESCs with METTL3 knockdown, the luciferase activity of the reporter with the wild-type site was markedly elevated compared to that observed in control cells. However, mutations in either one or both of the TERT m6A motifs abolished the increased luciferase activity observed in cells with METTL3 knockdown (Fig. 5E). These findings suggest that m6A methylation in the TERT 3′UTR is responsible for mRNA stability. Additionally, TERT mRNA expression was higher in cells co-transfected with METTL3 and mutant TERT-3′UTR sites (TERT-3′UTR-MUTs) compared to cells with the wild-type site (TERT-3′UTR-WT) (Fig. 5F). Furthermore, the mRNA stability of TERT-3′UTR-WT was greater in control cells than in cells with METTL3 overexpression, and the mutation in TERT-3′UTR abolished this difference (Fig. 5G,H). These results indicate that m6A in the TERT 3′UTR is crucial for mRNA stability, possibly due to its impact on the secondary structure of TERT mRNA.
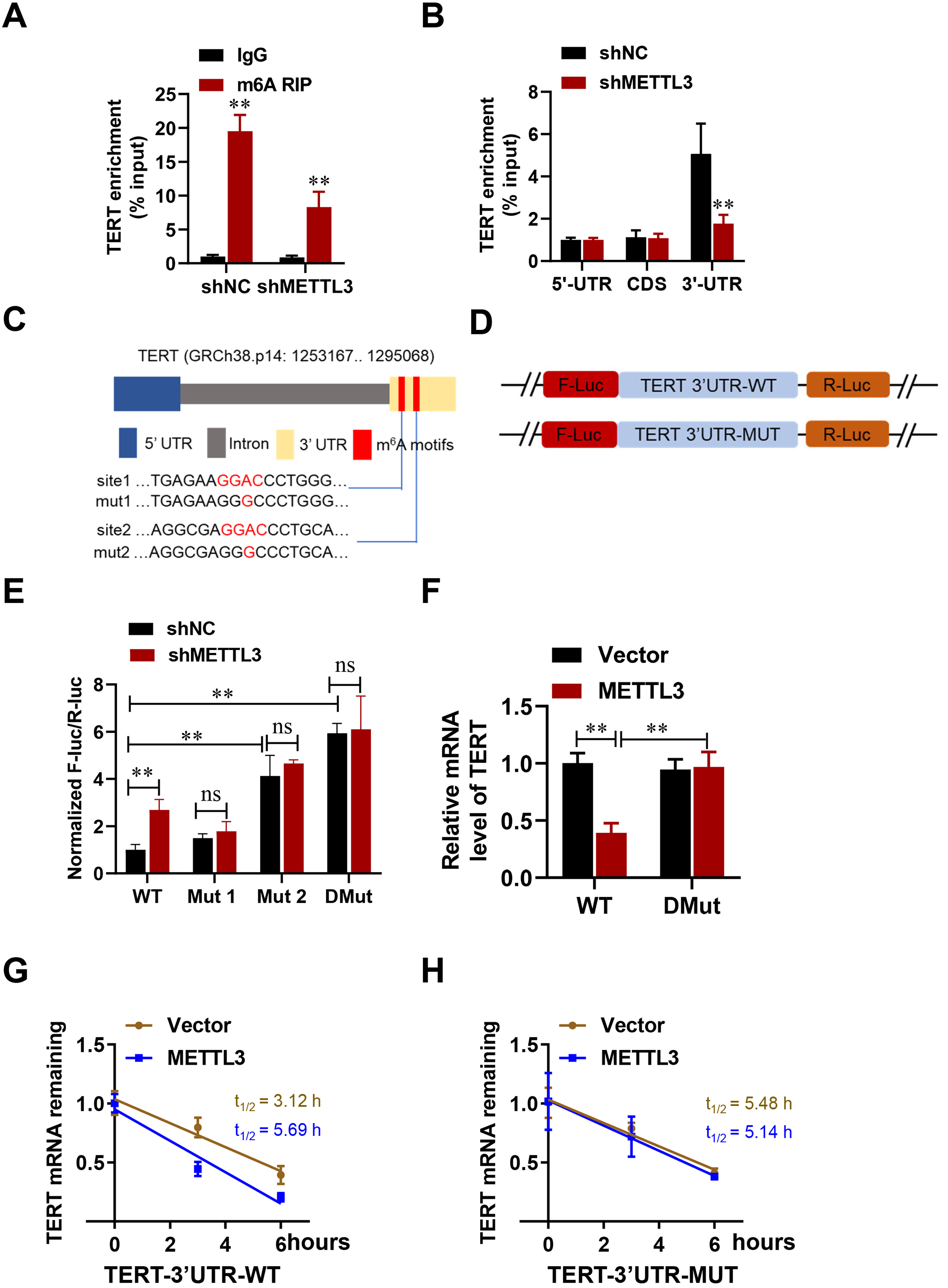 Fig. 5.
Fig. 5.
METTL3-driven m6A modification regulates TERT expression. (A,B)
Methylated RNA immunoprecipitation (MeRIP)-qPCR analysis of relative enrichment of m6A on TERT. (C) A diagram
illustrates the locations and mutations of m6A sites within the TERT 3′UTR,
with “mut” denoting mutant and “chr” referring to chromosome. (D) A schematic
shows the mutations introduced in the 3′UTR to examine the impact of m6A on
TERT expression. (E) The relative luciferase activity was measured for in control
or shMETTL3 HESCs. (F) TERT-3′UTR-WT or TERT-3′UTR-DMut was transfected
into control or shMETTL3 HESCs for 24 hours, followed by qPCR analysis to
determine TERT mRNA expression levels. (G,H) Following 24-hour
transfections of TERT-3′UTR-WT (G) or TERT-3′UTR-DMut (H) into control or
shMETTL3 cells, the cells were treated with actinomycin D (5 µg/mL)
for various durations. TERT mRNA levels were subsequently measured by
qPCR. Error bars indicate the mean
To regulate gene expression, m6A modification is performed by m6A writers and requires recognition by m6A readers [33, 34]. m6A readers, such as YTHDF2, YTHDF3, and IGF2BP1~3, can modulate mRNA stability [35, 36]. The present study found that YTHDF2, but not YTHDF1, YTHDF3, or IGF2BP1/2/3, showed significant binding to TERT mRNA in HESCs (Fig. 6A). Furthermore, the interaction between YTHDF2 and TERT was reduced in HESCs with METTL3 knockdown relative to control cells (Fig. 6B). Stability assays of mRNA indicated that TERT mRNA had an extended half-life in cells with YTHDF2 knockdown, while it was notably shorter in cells with YTHDF2 overexpression (Fig. 6C,D). Consistent with these findings, YTHDF2 negatively regulated TERT expression in HESCs (Fig. 6E,F). Additionally, the expression level of TERT mRNA in cells co-transfected with YTHDF2 and mutant TERT-3′UTR sites (TERT-3′UTR-MUTs) was higher than in cells co-transfected with YTHDF2 and the wild-type site (TERT-3′UTR-WT) (Fig. 6G). These findings demonstrate that YTHDF2 recognizes METTL3-methylated TERT mRNA and facilitates its decay (Fig. 7).
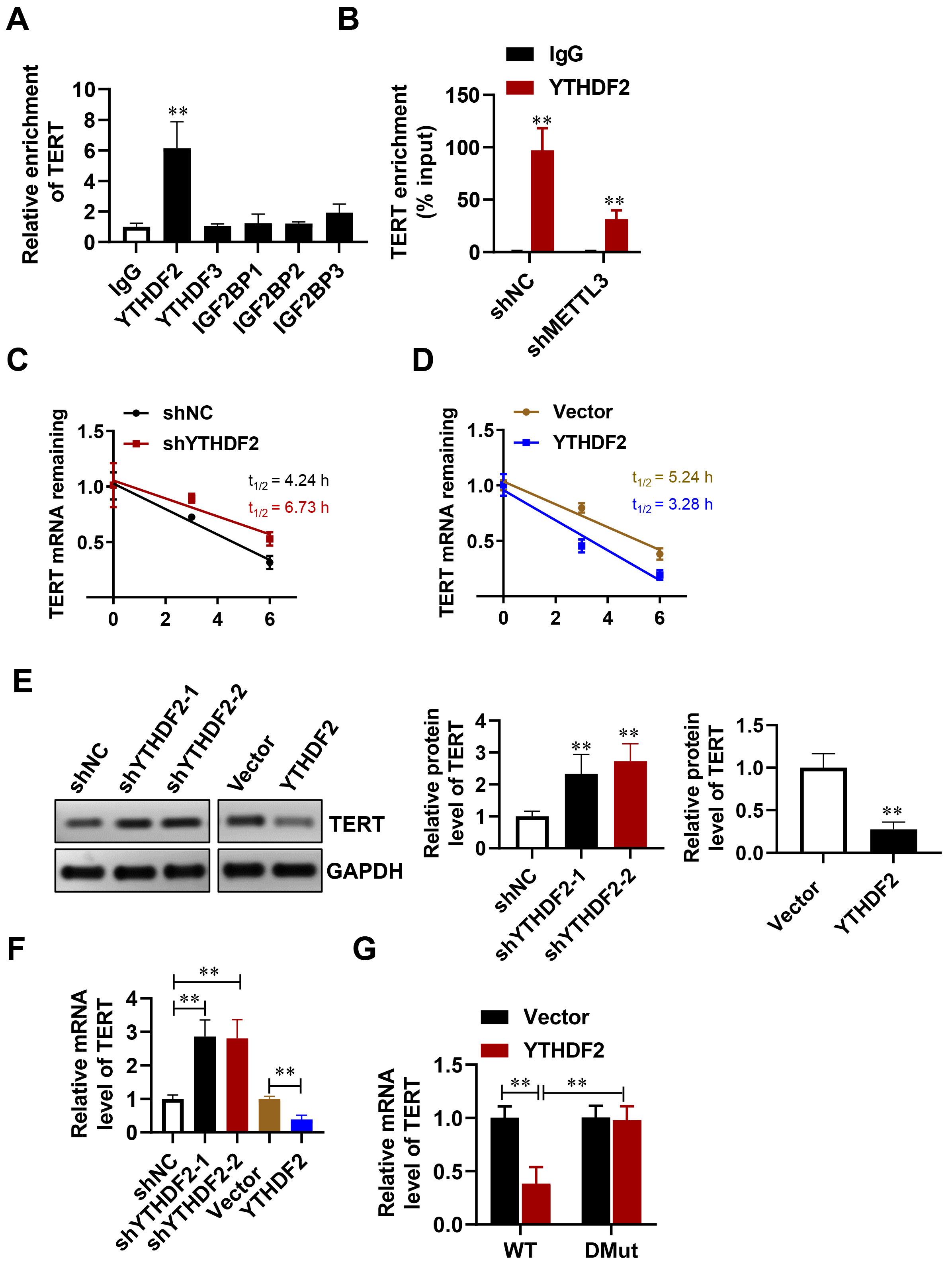 Fig. 6.
Fig. 6.
YTHDF2 regulates the decay of TERT mRNA in an
m6A-dependent fashion. (A) TERT mRNA was examined using RIP-qPCR with
antibodies specific to YTHDF2, YTHDF3, and IGF2BP1~3. (B) The
binding of YTHDF2 to TERT mRNA was assessed in control and shMETTL3 cells via
RIP-qPCR. (C,D) The degradation rate of TERT mRNA was analyzed at
various time points after treating with actinomycin D (5 µg/mL).
(E,F) The levels of TERT protein (E) and TERT mRNA (F) were measured
following the suppression or overexpression of YTHDF2 in HESCs. (G)
TERT-3′UTR-WT or TERT-3′UTR-DMut was transfected into control or
YTHDF2-overexpressing HESCs for 24 hours, after which TERT mRNA
expression was quantified using qPCR. Error bars denote the mean
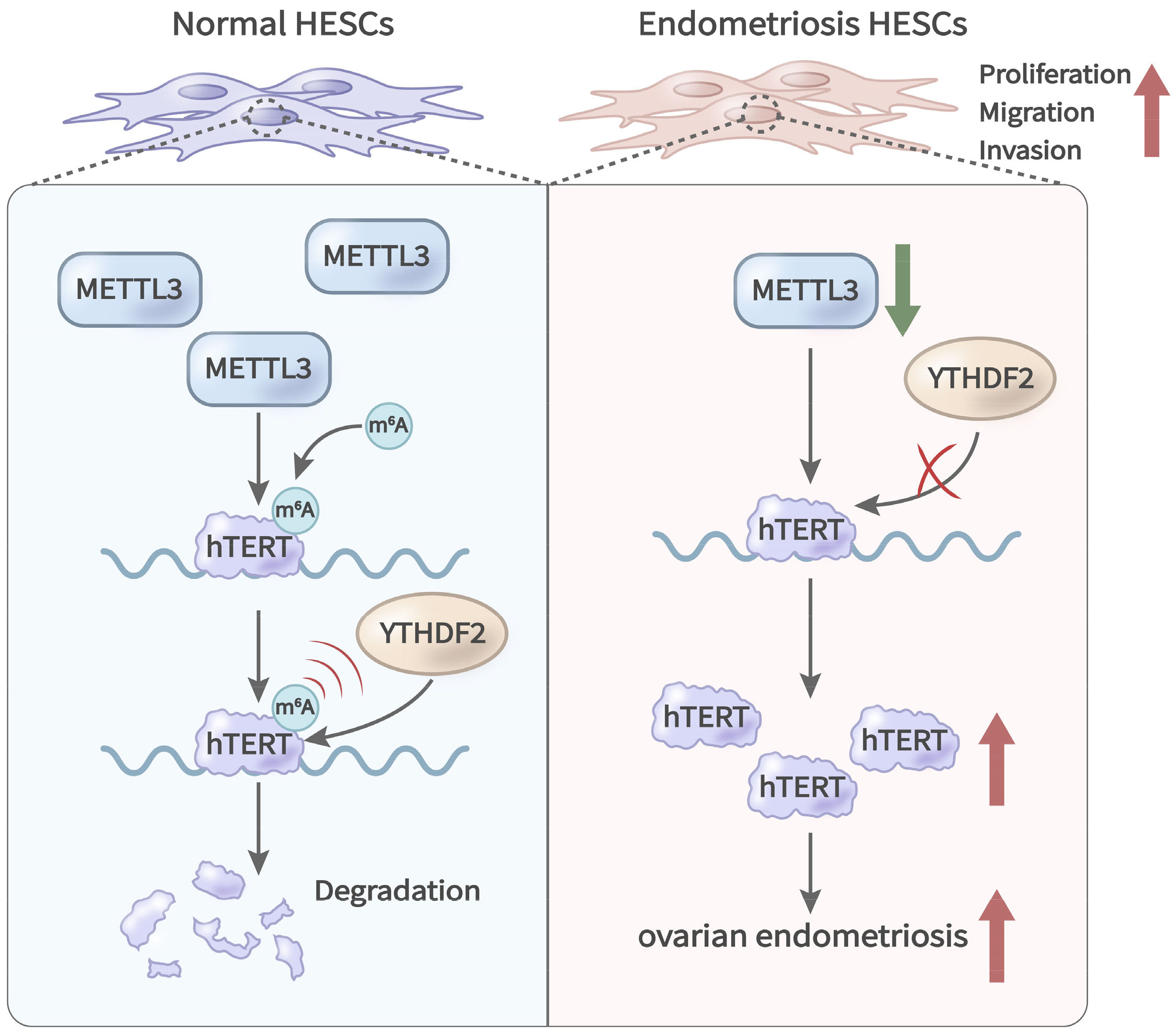 Fig. 7.
Fig. 7.
Graphical representation of the roles of METTL3 and TERT in endometriosis. METTL3 modifies TERT mRNA through m6A, while YTHDF2 mediates the decay of TERT mRNA in an m6A-dependent manner.
Endometriosis is a multifaceted and poorly understood condition that profoundly affects those who suffer from it. Although its origins are thought to be multifactorial, the precise functional and biological processes driving its onset remain largely elusive. Various studies have examined genes with increased expression in the endometrium of affected individuals. In this study, we explored the involvement of human TERT and m6A modification in endometriosis to uncover a potential new molecular mechanism underlying its development.
Human TERT is the catalytic subunit of telomerase and plays a crucial role in its activity [37]. The classical functions of TERT are to activate telomerase activity, synthesize telomere DNA, maintain telomere stability, and confer cells with the potential for unlimited proliferation. These functions are closely related to tumorigenesis and development, with TERT being highly expressed in over 90% of human malignancies [38]. Besides the telomerase pathway, recent studies have also shown that TERT participates in the regulation of gene expression and affects tumor cell invasion and gene regulation through telomerase-independent mechanisms. Overexpression of TERT remarkably increased the in vitro invasive ability of cells, and that hTERT expression in gastric cancer tissue was closely associated with tumor progression. High expression of TERT was also shown to enhance the invasive ability of tumor cells in cervical cancer, osteosarcoma and breast cancer [39]. This was accompanied by upregulation of metalloproteinase. Although endometriosis is not a malignant disease, many of its clinical and biological manifestations resemble the characteristics of malignant tumors, including abnormal cell proliferation, invasion, diffusion, and even metastasis. Similar to its role in the invasion and metastasis of tumors, hTERT may therefore also have an important role in the invasion of ectopic endometrial cells. Our findings revealed that TERT expression was upregulated in ectopic endometrial (EC) tissue compared to eutopic endometrial (EU) and normal endometrial (NC) tissue. This observation suggests the potential involvement of TERT in the aberrant cellular behaviors associated with endometriosis. To further elucidate the function of TERT, we conducted experiments to investigate the impact of its depletion in HESCs. We found that suppression of TERT expression led to a significant decrease in the proliferation, migration, and invasion of HESCs, whereas TERT overexpression had the opposite effect. This result suggests that TERT plays a crucial role in promoting the aggressive behavior of endometrial cells in endometriosis.
Epigenetic processes have been linked to gene expression dysregulation across various diseases, including cancers and developmental disorders [40, 41]. This study explored whether epigenetic mechanisms contribute to the elevated expression of TERT seen in endometriosis. We first examined whether DNA methylation and histone acetylation were involved in TERT expression, but found no significant changes in expression after treatment with inhibitors targeting these epigenetic modifications. This led us to investigate the potential role of m6A modification, the most frequent RNA modification, in the regulation of TERT expression. Our results showed lower m6A levels in endometriotic tissue compared to NC and EU. Previous research has indicated that decreased expression of the m6A methyltransferase METTL3 facilitates endometriosis development. METTL3 was found to be downregulated in endometriotic stromal cells, which enhances their migration and invasion through the METTL3/m6A/miR126 pathway [42]. Our study further confirmed the reduced expression of METTL3 in endometriotic cells. Additionally, we observed that overexpression of METTL3 led to decreased TERT expression, while METTL3 knockdown resulted in increased TERT levels. These results imply that METTL3-mediated m6A modification plays a role in regulating TERT expression in endometriosis. Moreover, the impact of METTL3-mediated m6A modification on TERT mRNA stability appears to be YTHDF2-dependent, suggesting this regulatory axis is involved in the pathogenesis of endometriosis.
Our study highlights a novel molecular mechanism involving METTL3-mediated m6A modification of TERT, which could have significant clinical implications. By elucidating the role of TERT and METTL3 in endometriosis, our findings provide a foundation for developing targeted therapies aimed at modulating this pathway. Inhibiting METTL3 or altering m6A modification could potentially offer new treatment options for managing endometriosis, improving patient outcomes, and enhancing quality of life. However, translating these insights into clinical practice will require additional research and validation in clinical settings. Despite the valuable insights provided by our study, several limitations must be acknowledged. Our findings are primarily based on in vitro experiments using HESCs and tissue samples. The absence of in vivo animal model studies limits our ability to fully understand how TERT and METTL3-mediated m6A modification impact endometriosis in a living organism. Further research using animal models is needed to validate these findings and assess their relevance to human disease. While our study identifies a potential role of METTL3-mediated m6A modification in regulating TERT mRNA stability and HESC cell behavior, the precise mechanisms underlying these interactions remain to be fully elucidated. Additional research is needed to explore how METTL3 influences TERT function and to identify other potential regulatory factors involved in this pathway.
Our study reveals a new molecular mechanism in endometriosis, suggesting that dysregulation of m6A modification through the METTL3/TERT axis enhances the migration and invasion of endometriotic cells. This pathway may offer potential therapeutic targets for novel treatments. However, the study’s limitations include its reliance on in vitro cell lines, necessitating further validation in animal models. Additionally, the interaction between TERT and m6A modification in endometriosis needs further investigation. In summary, our findings highlight the role of the METTL3/TERT axis in endometriosis and pave the way for future research and targeted therapies.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Conceptualization: JJC, YZ and FL; data acquisition: HT, YNW, RM, YSL, LFN and FL; data analysis: JJC, YZ, HT, YNW and FL; original draft preparation: JJC, YZ, HT, YNW and FL; review and editing: JJC, YZ, HT, YNW, RM, YSL, LFN and FL; all authors revised and agreed the final version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Human tissue samples were obtained with patients’ or their families/legal guardians’ written informed consent, in accordance with the Declaration of Helsinki guidelines. The research received approval from the Ethics Committee of Liuzhou Maternal and Child Health Hospital (KS-KY-2020-073).
We would like to express our sincere gratitude to all individuals and organizations who have contributed to this research. Special thanks to Yanjun Zheng for their invaluable guidance and support throughout this study.
This work was funded by the Guangxi Science and Technology Plan Project (Guangxi Clinical Research Center for Obstetrics and Gynecology), grant #GuiKe AD22035223, and Liuzhou Science and Technology Plan Project, grant #2020NBAB0825. Also was supported by Foundation of State Key Laboratory of Ultrasound in Medicine and Engineering [Grant No.2021KFKT020].
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.fbl2912421.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







