1 Department of Pathology, The First Affiliated Hospital of Soochow University, 215123 Suzhou, Jiangsu, China
2 Department of Dermatology and Venereology, The Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou Municipal Hospital, 215123 Suzhou, Jiangsu, China
Abstract
Psoriasis is a chronic and incurable skin inflammation driven by an abnormal immune response. Our study aims to investigate the potential of interferon-γ (IFN-γ) primed mesenchymal stem cells (IMSCs) in targeting T cells to attenuate psoriasis-like inflammation, and to elucidate the underlying molecular mechanism involved.
Mesenchymal stem cells (MSCs) were isolated from the umbilical cord and identified based on their surface markers. Psoriasis models were established and then treated with IMSCs. Flow cytometry analysis was used to examine cell surface markers and T cell percentages. Indoleamine-2,3-dioxygenase (IDO) was knocked down by small interfering RNA (siRNA) and examined with western blot assay. The proliferative capacity of T cells was assessed using water-soluble tetrazolium salt-1(WST-1). Additionally, an immunohistochemical assay was used to determine epidermal thickness. The psoriasis area and severity index (PASI) scores were also assessed.
We observed significant therapeutic efficacy of IMSCs against psoriasis-like inflammation in mice. Treatment with IMSCs resulted in a notable reduction in T cell infiltration within psoriatic lesions. Furthermore, we demonstrated that the therapeutic efficacy was mediated by the upregulation of IDO through IFN-γ stimulation. In vitro, IDO inhibited T cell proliferation, and in vivo, the therapeutic efficacy was eliminated when MSCs were transfected with IDO siRNA.
IMSCs can treat psoriasis by suppressing T cell infiltration and the suppression is mediated by IDO.
Keywords
- mesenchymal stem cells
- psoriasis
- T cell
- IDO
Psoriasis is a chronic and incurable skin inflammation that affects approximately 2% of the global population [1]. Its primary clinical manifestations include red and scaly plaques, commonly occurring on the knees, scalp and back [2]. Psoriasis is also associated with various comorbidities such as metabolic syndrome, cardiovascular disease and non-alcoholic fatty liver disease [3]. The predominant characteristic of psoriasis is the aberrant infiltration of inflammatory cells, which has acquired increasing attention in the field of immunology for elucidating its pathogenesis and potential therapeutic interventions.
Although the pathogenesis of psoriasis is not fully understood, it has been
demonstrated that the aberrant accumulation of T cells in the skin drives this
disease [4]. According to previous studies, T cells are the
primary cells involved in the adaptive immune response within psoriatic lesions[5].
They stimulate myeloid DCs (mDCs) by secretion of
interferon-
MSCs, also known as multipotent progenitor cells, have been
applied to treat various immune disease [8]. Generally, MSCs do
not inherently possess immunomodulatory properties [9], but they can acquire such
abilities through exposure to various cytokines, like IL-1
The specimens of skin (5 psoriasis patients and 5 trauma patients with healthy skin) and umbilical cord were obtained from The First Affiliated Hospital of Soochow University following institutional guideline and the ethical approval (20220238). The procedures were applied in accordance with the ethical standards of the Ethics Committee of The First Affiliated Hospital of Suzhou. Informed consent was achieved from patients or their families/legal guardians with psoriasis, who visited dermatology department without any concurrent systemic diseases and therapeutic interventions.
The procedures were conducted in accordance with the guidelines of the Ethics Committee of The First Affiliated Hospital of Suzhou. To isolate MSCs, we removed all blood vessels from the umbilical cord, which was stored in the Phosphate buffered saline (PBS). And the umbilical cord was then minced into small segments, which were subsequently transferred to culture plates containing the complete medium. Next, the cells were collected upon reaching 80% confluence. The MSCs were identified based on their cell surface markers, including cluster of differentiation 73 (CD73), CD29, CD90, CD105, CD45, CD31, CD34, CD11b, human leukocyte antigen (HLA) and HLA- D related isotype (HLA-DR). Antibodies (IgG) were diluted at 1:1000 and procured from BioLegend (San Diego, CA, USA). All samples were collected using a Cytoflex Flow Cytometer (Beckman Coulter, CA, USA), and the data were analyzed using FlowJo (FlowJo LLC, Ashland, OR, USA).
C57BL/6 male mice, aged 10–12 weeks, were procured from Vital River Laboratory (Beijing, China). They were kept under specific pathogen-free conditions. The experiments received approval from the Institutional Animal Care and Use Committee of Soochow University (SUDA20210916A02). The mice received the daily application of 62.5 mg Imiquimod (IMQ) cream (Aldara, 3M Pharmaceuticals, St. Paul, MN, USA) on their shaved backs for a continuous seven-day period. Mice were euthanized by cervical dislocation on day 8. The skin was taken for histological staining and flow cytometry analysis. We evaluated the symptoms of the psoriatic mice by psoriasis area and severity index (PASI), which utilizes a scoring system ranging from 0 to 4 for scaling, erythema, and thickness: minimal (0), mild (1), moderate (2), severe (3), and very severe (4).
Cell suspensions of skin were prepared using collagenase I (1.5 mg/mL, 17100017, Gibco, Waltham, MA, USA) and incubated for 1.5 h at 37 °C, followed by immunolabeling with targeted antibodies, such as CD45 (10 µg/mL, 982322, Biolegend, San Diego, CA, USA) and CD3 (10 µg/mL, 981002, Biolegend). Isotype control antibodies (10 µg/mL, Biolegend) were also used. All samples were then examined by using a Cytoflex Flow Cytometer (Brea, CA, USA).
106 IMSCs and IDO-Knockdown-IMSCs (IDO-KD-IMSCs) were dissolved in 100 µL PBS, and subcutaneously administered on days 1 and 4 post-IMQ application. The control mice received an equivalent volume of PBS and scramble-KD-IMSCs (Scr-KD-IMSCs).
For siRNA transfection, cells were seeded in a 6-well plate and transfected with IDO-siRNA (sense sequence 5′-CCCUGACUUAUGAGAACAUTT-3′, antisense sequence 5′- AUGUUCUCAUAAGUCAGGGTT-3′) and Scramble-siRNA (sense sequence 5′-UUCUCCGAACGUGUCACGUTT-3′, antisense sequence 5′- ACGUGACACGUUCGGAGAATT-3′) complexes formed by mixing siRNA with a transfection reagent in optimized-minimum essential medium (Opti-MEM). After incubating for 20 minutes to allow complex formation, the mixture was added to the cells and incubated for 6–8 h. Following this, the transfection medium was replaced with fresh complete medium, and cells were cultured for 24–72 h before assessing knockdown efficiency.
Protein was extracted and quantified from the cells. Then, it was separated by
sodium dodecyl sulfate - polyacrylamide gel electrophoresis (SDS-PAGE) and
transferred to a polyvinylidene fluoride (PVDF) membrane, which was incubated
overnight with IDO (13268-1-AP, Proteintech, Rosemont, Illinois, USA) and
The samples were fixed with 4% paraformaldehyde for 48 h. They were then dehydrated sequentially in 75%, 85%, 90%, 95%, and 100% ethanol solutions. The dehydrated samples were cleared with xylene and then embedded in paraffin. Finally. All samples were sectioned into 3–4 µm slices, and stained with hematoxylin (Beyotime, Nantong, Jiangsu, China) for 6 min and eosin (Beyotime) for 1 min.
Skin sections were then stained with CD3 antibody (10 µg/mL, ab16669, Abcam, Cambridge, UK), and incubated with secondary antibody (10 µg/mL, Abcam) for 1 h at room temperature. Subsequently, they were next stained with hematoxylin, and the images were captured using a microscope (Leica, Wetzlar, Hesse, Germany). Additionally, the results are quantified by enumerating the positive cells in each field, and the scorers were blinded to the experiment.
The experiment consisted of three groups: a positive control group stimulated by CD3 and CD28 antibody, a scramble-KD-IMSC group with the stimulation, and an IDO-KD-IMSC group also stimulated in the same manner. Peripheral Blood Mononuclear Cells (PBMCs) are isolated from whole blood using density gradient centrifugation. Both MSCs and PBMCs were seeded in 96-cell plates, and their proliferative capacity was evaluated using the water-soluble tetrazolium salt-1 (WST-1) cell proliferation and cytotoxicity assay kit (05015944001, Roche, Basel, Switzerland).
Statistical analysis was conducted using Graphpad prism 5 (San Diego, CA, USA).
The data were presented as means
We first isolated MSCs from umbilical cord tissues obtained from the First Affiliated Hospital of Soochow University. After removing the arteries and veins, we dissected the umbilical cords and cultured them onto plates (Fig. 1A). Through adherent purification, we obtained a population of homogeneous fibroblast-like spindle cells with robust proliferative capacity. When MSCs were expanded to the second generation, the cells were identified based on their common characteristic, and their surface markers were examined by flow cytometry. We observed positive expression of CD73, CD29, CD90 and CD105 in almost all cells, while CD45, CD31, CD34, CD11b, HLA and HLA-DR were negatively expressed (Fig. 1B). Thus, MSCs were successfully isolated.
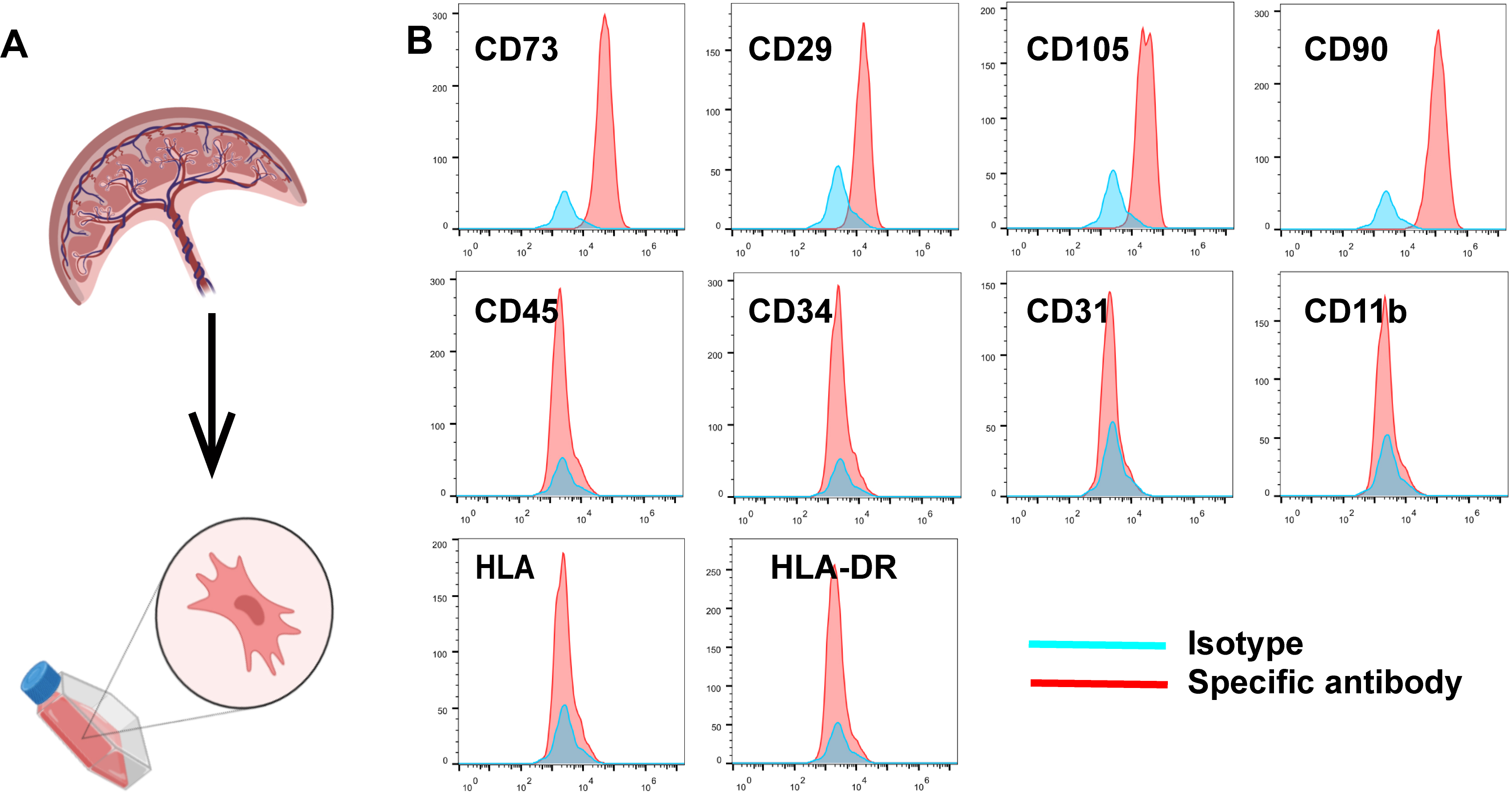 Fig. 1.
Fig. 1.
Mesenchymal stem cells (MSCs) derived from umbilical cord were successfully isolated and identified. (A) The way to isolate MSCs. (B) The surface markers of MSCs were identified by flow cytometry: cluster of differentiation 105+ (CD105+), CD29+, CD73+, CD90+, CD11b–, CD45–, CD31–, CD3–, human leukocyte antigen– (HLA–) and HLA- D related isotype– (HLA-DR–).
The previous study has demonstrated that MSCs primed by IFN-
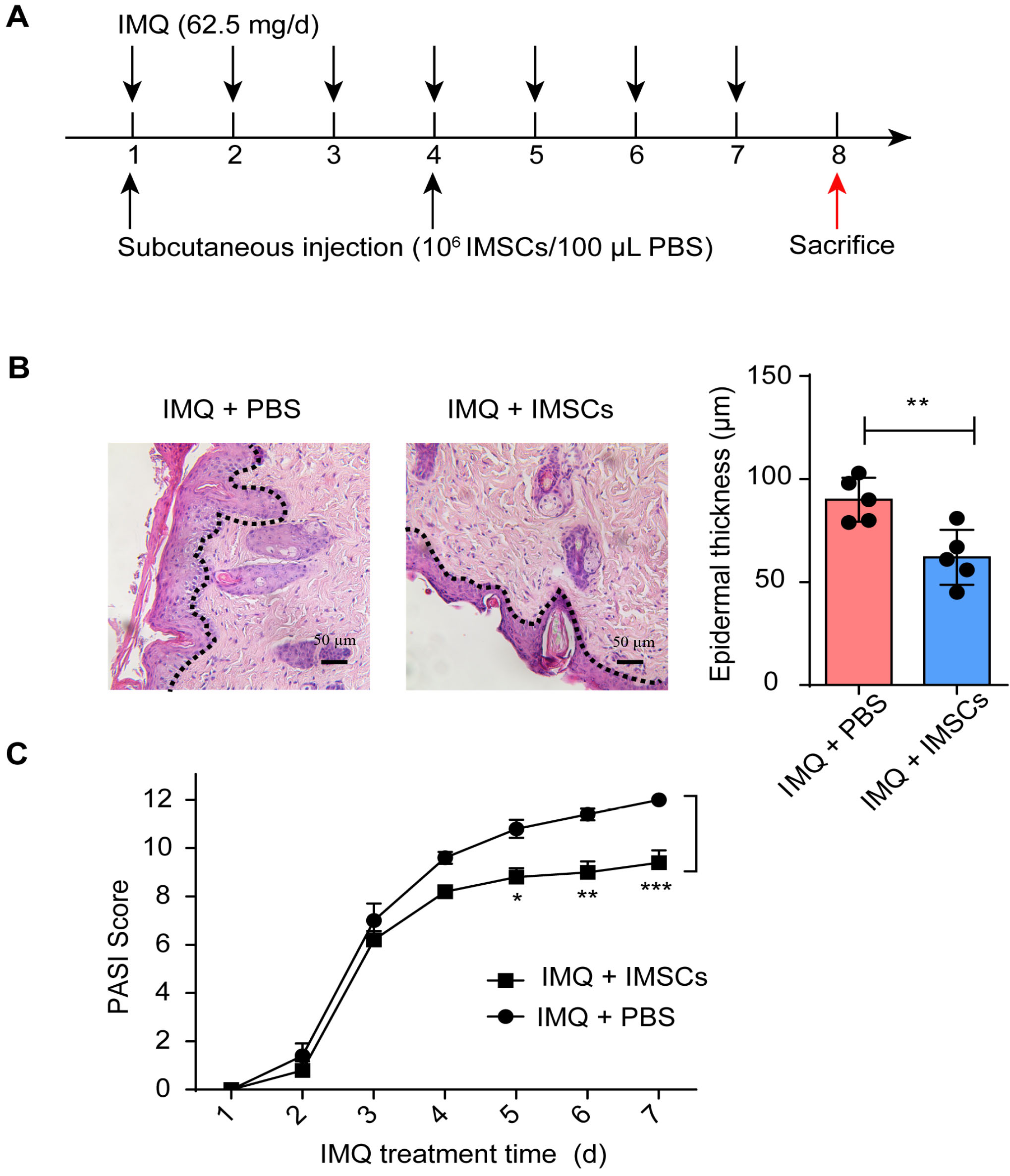 Fig. 2.
Fig. 2.
IFN-
As we known, T cells are the main pathogenic factor contributing to psoriasis. To investigate the accumulation of T cells within psoriatic lesions, we conducted an IHC experiment to examine T cell infiltration in samples from both psoriatic patients and mice. As predicted, the lesions were heavily infiltrated by T cells (Fig. 3A,B). To elucidate the underlying mechanisms through which IMSCs exert their therapeutic efficacy, we detected T cell accumulation by flow cytometry after IMSCs treatment (Fig. 3C). Our results indicated that T cells were significantly reduced after IMSCs treatment. Thus, IMSCs can treat psoriasis by inhibiting T cell infiltration.
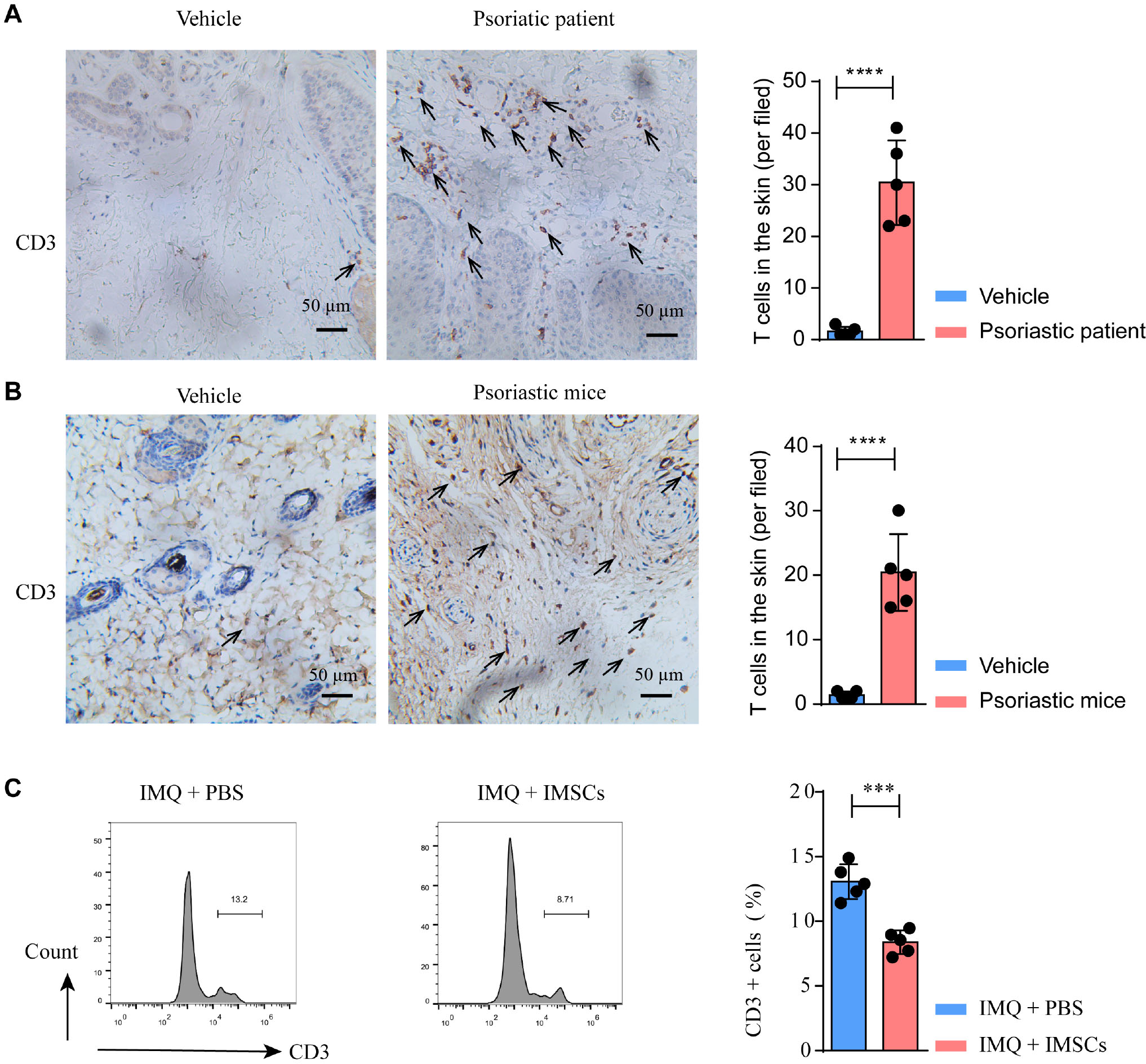 Fig. 3.
Fig. 3.
IMSCs inhibited the proliferation of T cell. (A,B)
Immunohistochemistry (IHC) staining of the lesion in the psoriatic patients and
mice compared with healthy skin. The images were captured at 20
It is suggested that the immunomodulatory properties of MSCs depend on the key mediators they produce [10]. Additionally, IDO is the key effector regulating T cell proliferation [12]. However, it still needs to be determined whether IDO is implicated in T cell inhibition in IMSCs-treated psoriasis. To investigate this issue, we examined the expression of IDO by western blot analysis. As predicted, IDO was overexpressed in IMSCs (Fig. 4A). To confirm the immunomodulatory effect of IDO on T cells, we knocked down IDO in IMSCs (Fig. 4B). Moreover, we cocultured PBMCs with Scr-KD-IMSCs and IDO-KD-IMSCs. We observed that the proliferation was inhibited when PBMCs were cocultured with Scr-KD-IMSCs, but this inhibitory effect was eliminated upon IDO knockdown (Fig. 4C). Therefore, it can be concluded that IDO is the key factor in regulating T cell infiltration.
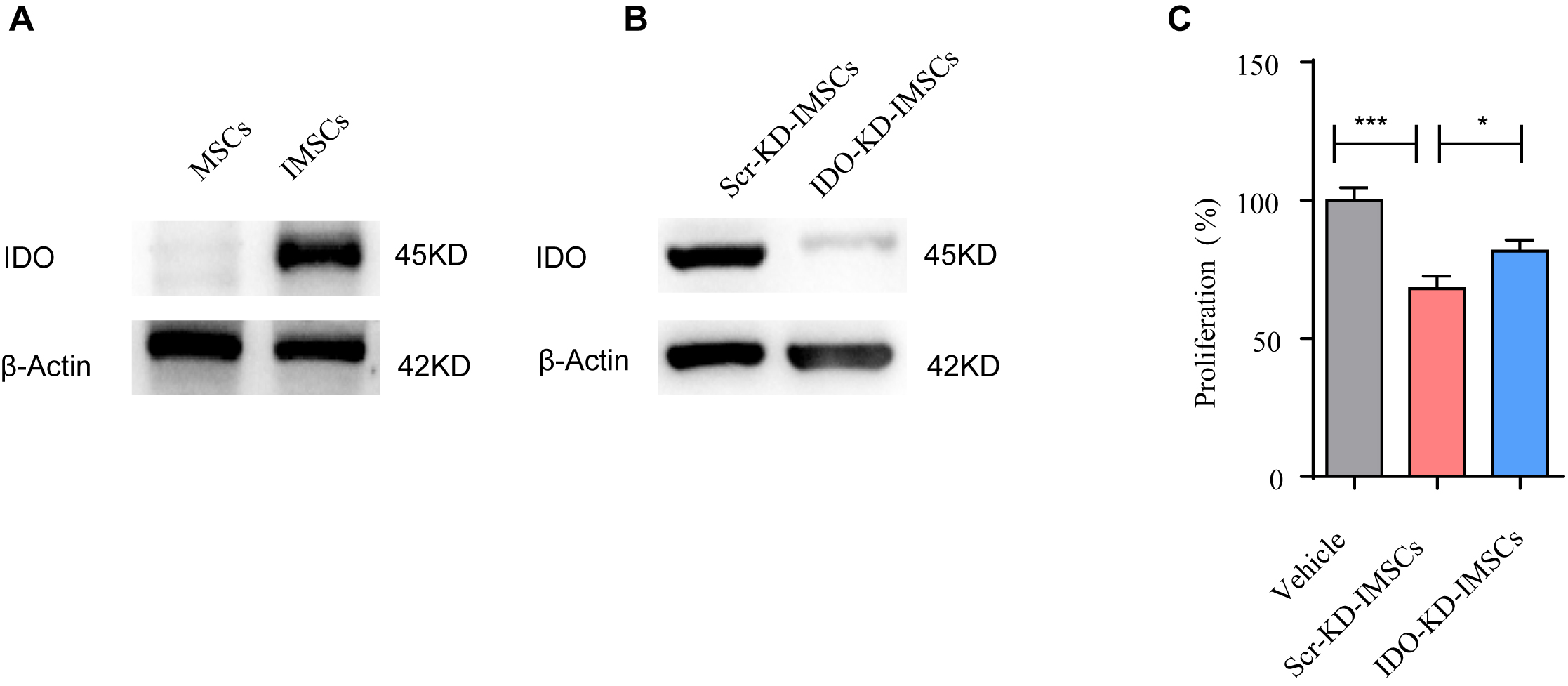 Fig. 4.
Fig. 4.
IMSCs inhibited the proliferation of T cells by
indoleamine-2,3-dioxygenase (IDO). (A) IDO expression in MSCs and IMSCs were
examined by western blot. (B) IDO expression in MSCs knocked down by scramble small Interfering RNA (Scr-KD-MSCs) and IDO small Interfering RNA (IDO-KD-MSCs) were examined by western blot. (C) IMSCs
inhibited the proliferation of T cells through IDO. Vehicle group was the
positive group, which was stimulated by CD3 and CD28 antibody. For three group
comparisons, one-way ANOVA was employed. (n = 3 for each group), * for p
To determine the therapeutic efficacy of IDO in the psoriatic model, we treated the psoriatic mice with Scr-KD-IMSCs and IDO-KD-IMSCs (Fig. 5A). We found that the therapeutic efficacy of IMSCs were impaired in the IDO-KD-IMSCs group (Fig. 5B). Furthermore, T cell infiltration in the skin lesions was recovered, and PASI scores were increased after IDO knockdown (Fig. 5C–E). These findings suggested that the therapeutic efficacy of IMSCs was diminished by IDO knockdown. Together, our results indicated that IMSCs could treat psoriasis through restraining T cell infiltration by IDO.
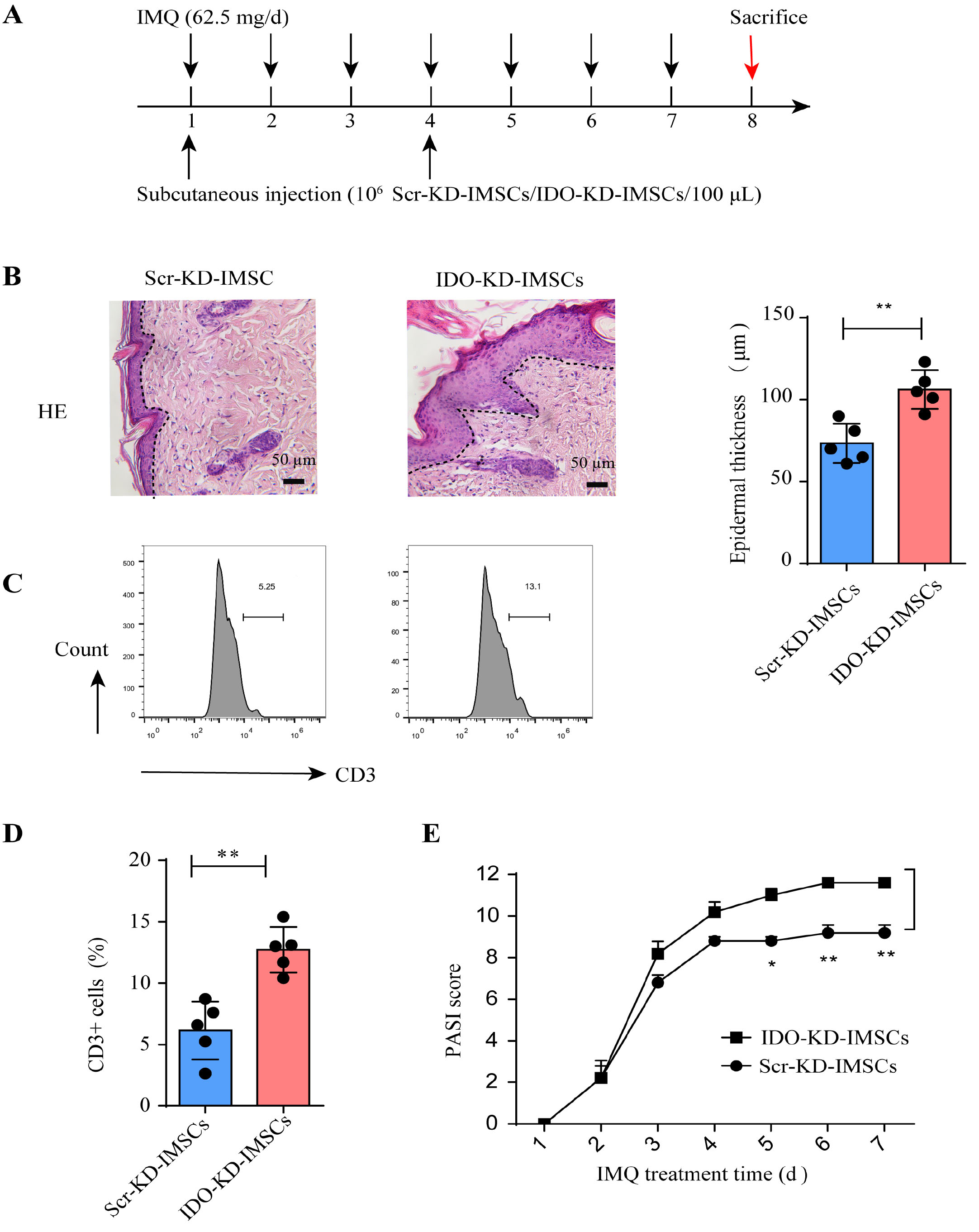 Fig. 5.
Fig. 5.
The therapeutic effects were abolished after IDO knockdown in
IMSCs. (A) The establishment of the experiment. (B) H&E staining of the lesion.
The images were captured at 20
Psoriasis is an immune-mediated recurrent skin inflammation that needs lifelong therapy [13]. T cells are the main contributors to the promotion of psoriasis through various cytokines that they secrete, and they can also interact with other cells, including mDCs, neutrophils and macrophages, leading to the progression of psoriasis [14]. Therefore, targeting T cells may be a promising therapeutic strategy for achieving complete remission of psoriasis. Our study demonstrated that IMSCs possessed strong potential in inhibiting T cell infiltration in psoriatic mice, and the inhibitory effect was mediated by the high expression of IDO. Thus, our study provides a new strategy for targeting T cells in the treatment of psoriasis.
IFN-
It was shown that psoriatic plaque evolution is characterized by T cell accumulation [18]. Additionally, the activation of T cells promotes the recruitment of inflammatory cells and triggers epidermal alterations [19]. Our results confirmed this notion, as we observed extensive infiltration of T cells in psoriasis lesions. It was identified IDO as a key mediator in regulating T cells through modulation of tryptophan concentration [20]. Consistent with these earlier studies, we observed an overexpression of IDO in IMSCs which led to a significant inhibition of T cell infiltration in the psoriatic lesions. Importantly, this inhibitory effect was eliminated when IDO was knocked down in IMSCs. Thus, we conclude that IDO mediates the suppression of T cells induced by IMSCs. Although IMSCs may secrete other effectors with therapeutic potential, this study confirms the crucial involvement of IDO in the treatment of psoriasis.
In summary, our study presents a novel application of IMSCs in the treatment of psoriasis as it exhibits a notable inhibitory effect on T cell accumulation, which holds great potential for clinical translation. Furthermore, we demonstrated that the IDO produced by IMSCs mediated the immunosuppression of T cells in psoriasis, highlighting the crucial role of IDO as a key mediator in the treatment of psoriasis. While the inhibitory effect of IDO on T cell infiltration was investigated in psoriasis, these findings may also be applicable to other inflammatory diseases involving T cells.
During the preparation of this work, the authors used ChatGPT to check spelling and grammar. After using this tool, the authors reviewed and edited the content as needed and took full responsibility for the content of the publication.
The data and materials that support the study are available from the corresponding author on reasonable request.
WZ: conception and design, collection and/or assembly of data and manuscript writing. CQ, CS and HY: data analysis and interpretation and manuscript writing; FS: conception and design, data analysis and interpretation; YD: conception and design, financial support and final approval of the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors agree to take responsibility for all aspects of the research.
The specimens of skin and umbilical cord were obtained from The First Affiliated Hospital of Soochow University following institutional guideline and the ethical approval (20220238). Informed consent was achieved from patients or their families/legal guardians with psoriasis, who visited dermatology department without any concurrent systemic diseases and therapeutic interventions. The research followed the declaration of Helsinki. The experiments received approval from the Institutional Animal Care and Use Committee of Soochow University (SUDA20210916A02).
Not applicable.
The study was supported by Jiangsu Provincial Natural Science Foundation: BK20240370; Suzhou Basic Research Pilot Project: SSD2024075; Bo Xi Foundation: BXQN202231; Science and Technology Development Foundation of Nanjing Medical University: NMUB20220189, Suzhou Science and Technology Bureau Project: SKY2023199.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





