1 Biotechnology Research and Innovation Council-National Institute of Biomedical Genomics, 741251 Kalyani, India
2 Department of Biotechnology, GLA University, 281406 Mathura, India
3 Department of Biotechnology, Brainware University, 700125 Barasat, India
4 Department of Biotechnology, The Neotia University, 743368 Parganas, India
5 Department of Medicine, Division of Hematology & Oncology, Medical College of Wisconsin, Milwaukee, WI 53226, USA
Abstract
Extracellular vesicles (EVs), the ubiquitous part of human biology, represent a small heterogenous, membrane-enclosed body that contains a diverse payload including genetic materials in the form of DNA, RNAs, small non-coding RNAs, etc. mostly mirroring their source of origin. Since, a vast majority of research has been conducted on how nucleic acids, proteins, lipids, and metabolites, associated with EVs can be effectively utilized to identify disease progression and therapeutic responses in cancer patients, EVs are increasingly being touted as valuable and reliable identifiers of cancer biomarkers in liquid biopsies. However, the lack of comprehensive clinical validation and effective standardization protocols severely limits its applications beyond the laboratories. The present review focuses on understanding the role of circulating EVs in different cancers and how they could potentially be treated as cancer biomarkers, typically due to the presence of bioactive molecules such as small non-coding RNAs, RNAs, DNA, proteins, etc., and their utilization for fine-tuning therapies. Here, we provide a brief general biology of EVs including their classification and subsequently discuss the source of circulatory EVs, the role of their associated payload as biomarkers, and how different cancers affect the level of circulatory EVs population.
Keywords
- biomarkers
- cancer biomarkers
- circulating extracellular vesicles
- clinical trials
- extracellular vesicles
- therapeutics
Intercellular communication, the conversation between two cells, is an essential biological process of multicellular organisms contributing to the growth, development, and normal functioning of the cells [1, 2]. However, ‘mis’ communication, the defect in intercellular messaging often culminates in the emergence of various disease conditions such as neurological disorders [1, 2], age-related diseases [3, 4], inflammation-associated diseases [5, 6, 7], cardiovascular anomalies [8, 9], and cancer [10, 11, 12, 13, 14], etc. Intercellular crosstalk is primarily mediated by three distinct mechanisms: (1) cell-cell interaction via cytoplasmic bridges, (2) membrane protein-dependent interaction between adjacent cells, and (3) the release of secretory molecules from the cells [15, 16, 17, 18]. However, emerging evidence identifies a fourth mechanism of intercellular communication which includes the intercellular transfer of extracellular vesicles (EVs) [19, 20, 21, 22, 23]. Earlier, EVs were recognized as ‘platelet dust’ by Peter Wolf in 1967 [24]. However, the advancement of EV research indicates that EVs are not just an inert ‘dust’ but play a pivotal role both in normal physiology and various pathological conditions [25, 26]. EVs are lipid bilayer-enclosed nanosized bodies [27, 28], termed so, as they are secreted by cells across all living kingdoms [19]. The release of EVs has been associated with almost all types of cells, including epithelial cells, endothelial cells, platelets, fibroblasts, stromal cells, immune cells, etc. [29, 30]. EVs contribute to the intercellular communication through the transfer of bioactive molecules in the form of proteins, lipids, DNA, mRNAs, microRNA (miRNAs), metabolites, etc. [31, 32, 33, 34, 35].
Depending on the biogenetic mechanism, formation, release, content, size, and functions EVs are broadly categorized into microvesicles, exosomes, and apoptotic bodies [36, 37, 38, 39, 40]. Microvesicles (MVs; also called microparticles, MPs, or ectosomes) are cell-secreted membranous structures, produced by outward budding followed by pinching off the plasma membrane [41, 42]. The interplay of cytoskeletal proteins, such as actin and microtubules as well as molecular motor proteins, such as kinesin and myosin contribute to the formation and simultaneous release of MVs from the cells [43, 44, 45]. The size distribution of MVs is shown to be ranging from 100 nm to 1 µm in diameter [23, 36, 37, 38]. Exosomes, the other category of EVs with a typical diameter of 30–150 nm [46], are of endocytic origin [23]. The biogenetic mechanism of exosomes appears to be completely different from MVs. It includes the inward invagination of the endosomal membrane to generate the multivesicular bodies (MVBs) [23, 37, 47] which eventually fuse with the plasma membrane, thereby releasing the content in the MVBs outside the cells in the form of exosomes [47, 48, 49, 50]. Exosome biogenesis involves endosomal sorting complexes required for the transport (ESCRT) pathway [51, 52]. Unlike MVs and exosomes, apoptotic bodies are considerably larger, ranging in size from 50 nm to 5 µm in diameter, and generated from cells undergoing programmed cell death [38]. The enhanced hydrostatic pressure, generated during the contraction of the cells, separates the plasma membrane from the cytoskeleton, resulting in the release of such apoptotic bodies [53]. However, although such terminologies are still being used in some of the literature, the minimal information for studies of extracellular vesicles (MISEV2023) strictly recommends using the generic term ‘EV’ to denote any types of extracellular vesicle omitting the biogenesis-based terms [54]. The reason behind this lies in the difficulties in accurately establishing the biogenetic mechanisms of MVs or exosomes and their overlapping size distribution [54]. Hence, as per the MISEV2023 guidelines, in the present review we have denoted all types of extracellular vesicles such as MVs or exosomes as ‘EVs’. The mechanisms of EVs’ uptake into the recipient cells include direct fusion of EV membrane with the plasma membrane of recipient cells or endocytosis [55, 56, 57, 58].
Recently, a free web-based compendium, ‘Vesiclepedia (http://www.microvesicles.org/)’ has come up which catalogs different EV cargoes from both published and unpublished data [59]. About 3533 EV studies which include 50,550 RNA entries, 566,911 protein entries, 3839 lipid entries, 192 metabolites, and 167 DNA entries are presently being included in ‘Vesiclepedia’ [59]. Another international consortium, the EV-TRACK (http://evtrack.org/) has also emerged which encourages uploading both published and unpublished EV-related experimental data [60]. It not only provides the relevant experimental parameters but also aids in interpreting the data with improved scientific rigor and documenting the evolution of EV research [60]. Both these knowledgebases not only facilitate interpretation of the data but also help in replicating the experiments.
The present review begins with the general classification of EVs, followed by a brief discussion of the different cellular sources of EVs in circulation. The next section deals with how various cargo molecules render the EVs being considered as biomarkers which is followed by a detailed understanding of the impact of EVs as biomarkers on different types of cancer. The final section of the review presents a brief discussion on how EVs could be used as cancer therapeutics and the ongoing EV-based clinical trials in different types of cancer.
However, the major challenges in the discovery of predictive biomarkers can be categorized into biological and clinical [61]. The biological complexity of different molecular pathways accounts for the biological challenges whereas the lack of favorable clinical outcomes in all the patients constitutes the clinical challenges. Emerging evidence identifies how EVs modulate various complex signaling pathways in the context of cancer pathogenesis which can overcome the biological challenges in biomarker discovery. In addition, it leads to the therapeutic application of EVs in clinical settings in the treatment of complex diseases like cancer.
A detectable number of EVs are found in human body fluids, such as blood, urine, saliva, synovial fluid, and interstitial spaces between the cells [23, 29, 37, 62]. However, blood is considered to be the richest source of EVs [63]. The most abundant EVs population in blood arises from platelets [64], although erythrocytes, mast cells, neutrophils, eosinophils, monocytes, macrophages, dendritic cells, natural killer cells (NK cells), lymphocytes, endothelial cells, fibroblasts, and mesenchymal stem cells contribute significantly to the population of blood-borne EVs [65]. Recent evidence indicates that 99.8% of the EV population in blood arises from hematopoietic cells, whereas the other 0.2% of EVs originate from the tissues [66]. The present section briefly delineates how circulating EVs from different cellular origins contribute to the pathogenesis of cancer.
The first recognized EVs in the blood were derived from platelets, which were referred to as ‘platelet-dust’ [24]. However, a growing body of evidence indicates that platelet-derived EVs (pEVs) play a significant role in various pathophysiological conditions, including cancer. For example, EVs are shown to transfer integrin CD41 to lung cancer cells, leading to the induction of cell proliferation, metastasis, and angiogenesis [67]. Mechanistically, pEVs-mediated delivery of CD41 promotes phosphorylation of mitogen-activated protein kinase p42/44 and serine/threonine kinase or expression of membrane type 1-matrix metalloproteinase (MT1-MMP), matrix metalloproteinase 9 (MMP-9), vascular endothelial growth factor (VEGF), interleukin-8 (IL-8), and hepatocyte growth factor (HGF), thereby promoting cancer progression [67]. In epithelial ovarian cancer, pEVs transfer miR-939 to the cancer cells which promotes cell proliferation and migration via the induction of epithelial to mesenchymal transition (EMT) [68]. However, the target of miR-939 and the mechanistic details have not been specified in their study [68].
Erythrocyte-derived EVs (eEVs) constitute the second-highest population of circulating EVs. Very few studies have identified the role of EVs in cancer progression. Cheng et al. [69] have demonstrated that EVs from stored erythrocytes enhance migration and invasion of lung cancer cells via the accumulation of miR-1246 and miR-150-3p, probably leading to Ras-proximate-1 (Rap1)-mediated induction of integrin beta-1 (ITGB1) expression.
Mast cell-derived EVs (mcEVs), another EVs’ subpopulation in the circulation,
are often shown to influence cancer progression. For example, in lung cancer,
mcEVs-mediated transfer of transforming growth factor
Neutrophil-derived EVs (nEVs) are often shown to confer anti-tumor responses. For example, nEVs can deliver cytotoxic proteins to cancer cells, leading to tumor apoptosis via the activation of the caspase signaling pathway [71].
Tumor-associated macrophages (TAMs) account for ~50% of local
hematopoietic cells in the tumor microenvironment (TME) [72]. TAM-EVs also
contribute significantly to the progression of cancer by various unique
mechanisms. For example, TAM-derived EVs exhibit a higher expression of miR-95
which promotes prostate cancer progression via targeting JunB and subsequent
induction of cell proliferation [73]. Moreover, TAM-EVs are also shown to deliver
miR-501-3p to pancreatic ductal adenocarcinoma (PDAC) cells, leading to cell
invasion and migration via targeting transforming growth factor beta receptor
type 3 (TGFBR3) and activation of TGF-
Like nEVs, dendritic cell (DC)-secreted EVs (dcEVs) also exert anti-tumor activities. dcEVs often carry major histocompatibility complex (MHC) class I and class II molecules, thereby showing anti-tumor responses by activating cognate T-cells and thus can be effectively used in cancer treatment [79].
On several occasions, NK cell-derived EVs (nkEVs) have been shown to suppress tumor progression. nkEVs, due to the presence of various cytolytic proteins, show anti-tumor activity via the induction of apoptosis [80]. A study by Zhu et al. [81] also demonstrated that nkEVs suppress glioblastoma survival upon down-regulation of cell survival markers, p-ERK, and p-AKT as well as enhanced expression of apoptotic markers, cleaved-caspase 3, cytochrome-c and cleaved-poly (ADP-ribose) polymerase (PARP).
Lymphocyte-derived EVs (lEVs) also influence tumor progression by multiple
mechanisms. For example, in uterine corpus endometrial cancer,
CD45RO-CD8+ T-cells release miR-765-laden EVs which promotes
EMT, leading to cancer progression [82]. In an immunosuppressive environment of
nasopharyngeal carcinoma,
Emerging evidence indicates that endothelial cell-derived EVs (ecEVs) are actively involved in tumor progression. For example, in breast tumoral conditions, ecEVs show a downregulation of miR-503 expression which is known for targeting cyclin D (CCND) 2 and -3, and miR-503 under expression in the EVs promotes breast cancer cell proliferation and invasion [84]. ecEVs, positive for carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1), are shown to promote the proliferation of CD3 or CD3/CD28-induced T-cells, thereby modulating immune response and associated tumor progression [85].
EVs derived from mesenchymal stem cells often play a dual role in the
development and progression of different types of cancer. Human umbilical
cord-mesenchymal stem cell (hUC-MSC)-derived EVs are shown to induce
proliferation, migration, invasion, and epithelial-to-mesenchymal transition
(EMT) in human breast cancer cells, through the activation of extracellular
signal-regulated kinase (ERK) pathway [86]. Similarly, Mao et al. [87]
have demonstrated that p53 deficient mouse bone marrow mesenchymal stem cell
(mBMMSC)-derived EVs promote gastric cancer growth and metastasis via the
delivery of ubiquitin-protein ligase E3 component n-recognin 2 (UBR2) and
regulation of Wnt/
Cancer-associated fibroblasts (CAFs) in the tumor microenvironment (TME) release a significant number of EVs which are shown to influence cancer progression by different mechanisms. CAF-EVs are reported to carry miR-181d-5p which is taken up by the breast cancer cells thereby promoting proliferation, invasion, migration, and EMT while inhibiting apoptosis via targeting caudal-related homeobox 2 (CDX2) [93]. CAF-EVs are also shown to transfer signature miRNAs such as miR-22, let-7a, and miR-125b to prostate cancer cells, leading to the downregulation of mitochondrial oxidative phosphorylation and reprogram metabolic pathways [94]. In a study by Gao et al. [95], CD63+ CAF-EVs have been shown to confer tamoxifen resistance to breast cancer cells via transferring miR-22 and downregulating the expression of its targets, estrogen receptor 1 (ESR1) and phosphatase and tensin homolog (PTEN).
‘Biomarker’ can be defined as a biological component of a process, event, or condition [96]. It reflects the health status of an individual [96]. For example, high blood lead level is an indicator of nervous debility and cognitive disorders [97]. High levels of cholesterol in the blood can be considered as the biomarker for heart disease risk [98]. Similarly, a growing body of evidence indicates that circulating EVs could be used as biomarkers for different diseased conditions. For example, signature miRNA molecules in the plasma EVs of chronic obstructive pulmonary disease (COPD) patients render the EVs to become the biomarkers for COPD and could be used in the diagnostic, prognostic, and therapeutic purposes of COPD [99]. EVs in the blood of several neurodegenerative diseases often contribute to assessing the severity and progression of the diseases and, thereby can be defined as the biomarkers for neurodegenerative disorders [100]. Elevated levels of serum EVs in alcoholic hepatitis (AH) patients not only serve them as potential biomarkers but also facilitate the development of novel therapeutic targets against AH [101]. Furthermore, the level of fibronectin (FN) is well-elevated in the plasma EVs of breast cancer patients as compared to healthy controls, thereby being identified as a promising biomarker for breast cancer [102]. An increase in the concentration of plasma EVs which is accompanied by a significant decrease in EVs’ diameter is monitored in patients, undergoing cardiac allograft rejection, and hence the EVs can also be used as a biomarker for graft rejection in cardiac diseases [103]. The release of large PS+ EVs is one of the characteristic features of a cell undergoing apoptosis, thereby apoptotic EVs (ApoEVs) are often termed as circulating biomarkers of cell death [104]. In Alzheimer’s disease (AD), signature miRNA molecules, associated with the plasma EVs help measure the invasive potential of AD and hence are used as promising diagnostic biomarkers for AD [105]. Recently, circulating platelet-derived EVs have been designated as diagnostic biomarkers for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, which rapidly develops acute respiratory distress syndrome (ARDS), as the count of platelet-derived circulating EVs is increased significantly, post-SARS-CoV-2 infection [106]. The elevated levels of liver-specific proteins in the circulating EVs are often considered the biomarkers for alcohol- and drug-induced liver injury [107]. Moreover, recent reports demonstrate the active participation of circulating EVs in the progression of several renal diseases, which often contribute more than only a biomarker [108].
As EVs are known to carry bioactive molecules such as proteins, lipids, mRNAs, miRNAs, DNA, metabolites, etc., more often the EVs’ components are identified as potential biomarkers for various diseases [109]. Table 1 (Ref. [110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120, 121, 122, 123, 124, 125, 126, 127, 128, 129]) provides a brief description of how different EVs’ constituents serve as biomarkers in different pathophysiological conditions. Fig. 1 also illustrates how different EV-associated molecules could be considered biomarkers in different diseased conditions.
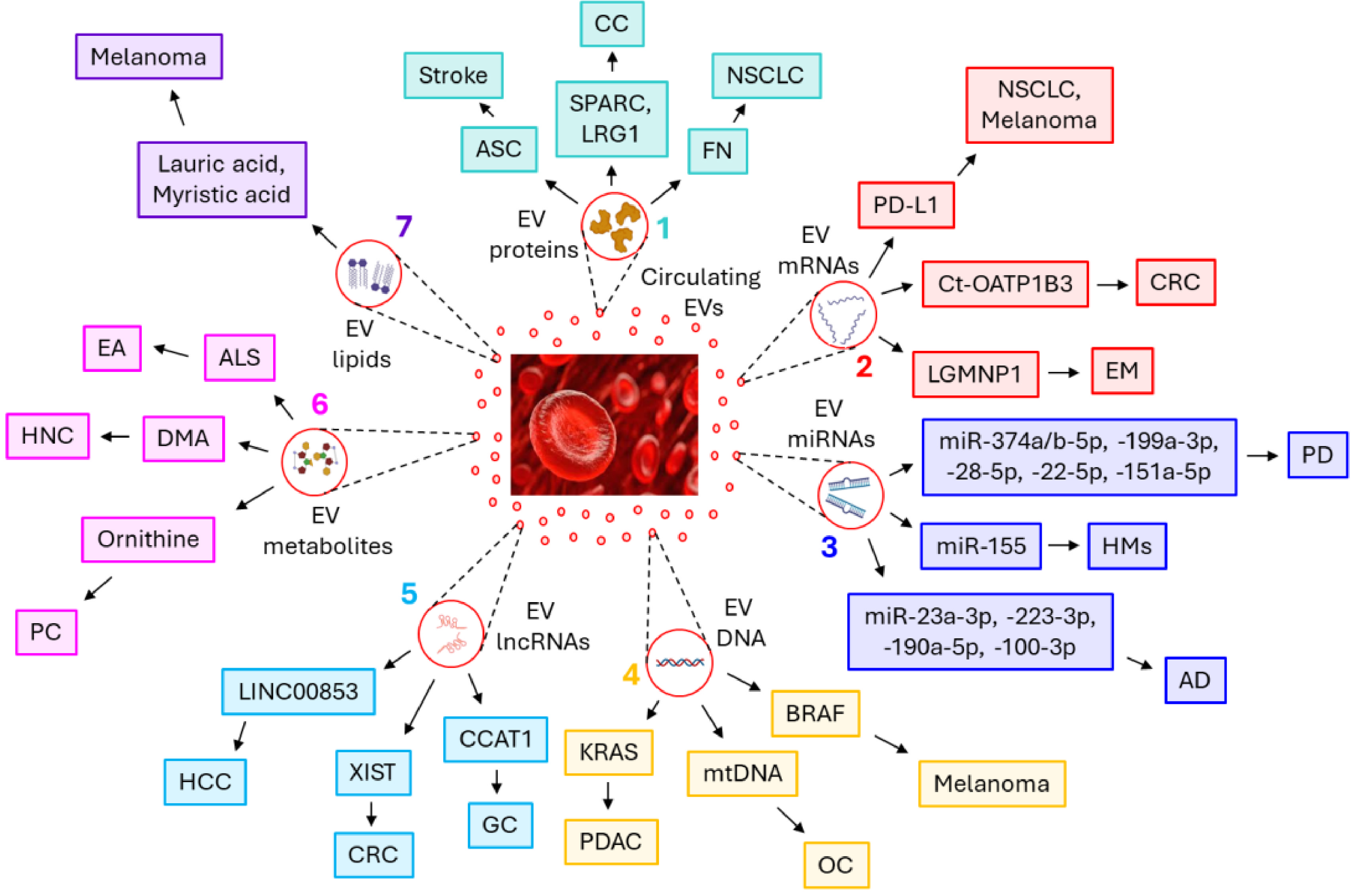 Fig. 1.
Fig. 1.
Circulating extracellular vesicles’ (EVs’) carried bioactive
molecules as biomarkers of different pathophysiological conditions. Circulating
EVs’ carried
| EVs’ component | Name | Diseased condition | Up/Down regulation | Reference/s |
| Protein | ASC | Ischemic stroke | Up |
[110] |
| SPARC, LRG1 | Colon cancer | Up |
[111] | |
| Fibronectin | NSCLC | Up |
[112] | |
| mRNA | PD-L1 | Melanoma, NSCLC | Up |
[113] |
| Ct-OATP1B3 | CRC | Up |
[114] | |
| LGMNP1 | EM | Up |
[115] | |
| miRNA | miR-374a-5p, | PD | Up |
[116] |
| miR-374b-5p, | Up |
|||
| miR-199a-3p, | Up |
|||
| miR-28-5p, | Up |
|||
| miR-22-5p, | Up |
|||
| miR-151a-5p | Up |
|||
| miR-155 | HMs | Up |
[117] | |
| miR-23a-3p, | AD | Up |
[118] | |
| miR-223-3p, | Up |
|||
| miR-190a-5p | Up |
|||
| miR-100-3p | Down |
|||
| DNA | KRAS mutant | PDAC | Up |
[119, 120] |
| mtDNA | Ovarian cancer | Down- Early stages |
[121] | |
| Up- Advanced stages |
||||
| BRAF mutant | Melanoma | Up |
[122] | |
| lncRNA | CCAT1 | Gastric cancer | Up |
[123] |
| XIST | CRC | Up |
[124] | |
| LINC00853 | HCC | Up |
[125] | |
| Metabolite | Ornithine | Prostate cancer | Up |
[126] |
| N,N-Dimethylaniline | HNC | Up |
[127] | |
| N-arachidonyl L-serine | EA | Up |
[128] | |
| Lipid | lauric (C12:0) acid | Melanoma | Up |
[129] |
| myristic (C14:0) acid |
Abbreviations: ASC, apoptosis-associated speck-like protein containing
a caspase-recruitment domain; SPARC, secreted protein acidic and cysteine rich;
LGR1, leucine-rich
Inflammasome protein, an apoptosis-associated speck-like protein containing a
caspase-recruitment domain (ASC) is found to be significantly elevated in the
serum EVs of ischemic stroke patients as compared to control subjects and hence,
considered as the biomarker for stroke [110]. Higher levels of extracellular
matrix (ECM)-related proteins such as secreted protein acidic and cysteine-rich
(SPARC) and leucine-rich
Plasma EVs’ programmed death-ligand 1 (PD-L1) mRNA level can be considered as the biomarker for melanoma and NSCLC progression, as its expression increases with the progression of the disease, and a significant down-regulation is monitored during the successful treatment with anti-PD-1 antibodies [113]. Cancer-type organic anion transporting polypeptide 1B3 (Ct-OATP1B3) mRNA is abundant in the serum EVs of colorectal cancer (CRC) and serves as a prognostic biomarker for CRC [114]. Serum EVs, in patients with ovarian endometriosis (EMs), are enriched with Legumain pseudogene 1 (LGMNP1) which is considered to be a promising biomarker for predicting ovarian EMs recurrence [115].
Serum EVs, of patients with Parkinson’s disease (PD), are enriched with several miRNAs which include miR-374a-5p, miR-374b-5p, miR-199a-3p, miR-28-5p, miR-22-5p, and miR-151a-5p, thereby serve as biomarkers for PD progression and early diagnosis [116]. miR-155 is abundant in the circulating EVs of patients with different types of hematological malignancies (HMs) and is considered a potential biomarker for the HMs [117]. Signature miRNA molecules, miR-23a-3p, miR-223-3p, and miR-190a-5p are shown to be up-regulated whereas, miR-100-3p is down-regulated in the plasma EVs of Alzheimer’s disease (AD) patients, thereby could be recognized as AD biomarkers [118].
Kirsten rat sarcoma virus (KRAS) mutations in the DNA of serum EVs are frequently observed in patients with pancreatic ductal adenocarcinoma (PDAC), which could serve as a predictive biomarker for PDAC [119, 120]. Mitochondrial DNA (mtDNA) in the serum EVs often defines different stages of ovarian cancer, a down-regulation is monitored in the early stage of ovarian cancer whereas, a significant up-regulation is associated with the advancement of tumor development [121]. B-rapidly accelerated fibrosarcoma (BRAF) mutations in the DNA of plasma EVs of melanoma patients are considered to be the biomarkers for melanoma [122].
The lncRNA, colon cancer-associated transcript 1 (CCAT1), is highly expressed in the serum EVs of gastric cancer patients as compared to healthy individuals, thereby serving as a biomarker for gastric cancer [123]. X inactive-specific transcript (XIST) in the serum EVs serves as a diagnostic biomarker for colorectal cancer as its expression is significantly up-regulated in colorectal cancer EVs [124]. Furthermore, LINC00853+ EVs in the serum of hepatocellular carcinoma (HCC) are significantly up-regulated which marks them as diagnostic biomarkers for HCC [125].
EV metabolites have recently been identified as diagnostic and prognostic biomarkers for various diseases. For example, a high concentration of ornithine in the plasma EVs of prostate cancer patients renders it to be considered a prostate cancer biomarker [126]. The higher amount of N, N-Dimethylaniline is found in the serum EVs of head and neck cancer (HNC) patients as compared to healthy individuals [127]. Moreover, the presence of N-arachidonyl L-serine in the plasma EVs of patients with endometrioid adenocarcinoma (EA) enable it to be used as a predictive biomarker for EA [128].
Little is known about the lipid components of the circulating EVs, associated with the diseased conditions. Higher levels of lauric (C12:0) and myristic (C14:0) acids are found in the plasma EVs of stage II and III–IV melanoma patients which signify the accelerated metabolism during these advanced stages of cancer [129].
EVs are important mediators of cell-cell communications [31] and hence, are taken as biomarkers for different types of cancer. Circulating EVs, via the delivery of their cargoes, mostly proteins and miRNAs, often serve as predictive biomarkers in various cancers. Table 2 (Ref. [130, 131, 132, 133, 134, 135, 136, 137, 138, 139, 140, 141, 142, 143, 144, 145, 146, 147, 148, 149, 150, 151, 152, 153, 154, 155, 156, 157, 158, 159, 160, 161, 162]) and Table 3 (Ref. [31, 163, 164, 165, 166, 167, 168, 169, 170, 171, 172, 173, 174, 175, 176, 177, 178, 179, 180, 181, 182, 183, 184, 185, 186, 187, 188, 189, 190, 191, 192, 193, 194, 195]) briefly describe how specific proteins and miRNAs respectively, of circulating EVs are recognized as biomarkers in different types of cancer. Fig. 2 also summarizes the role of circulating EVs’ carried proteins and miRNAs as the biomarkers of various cancers.
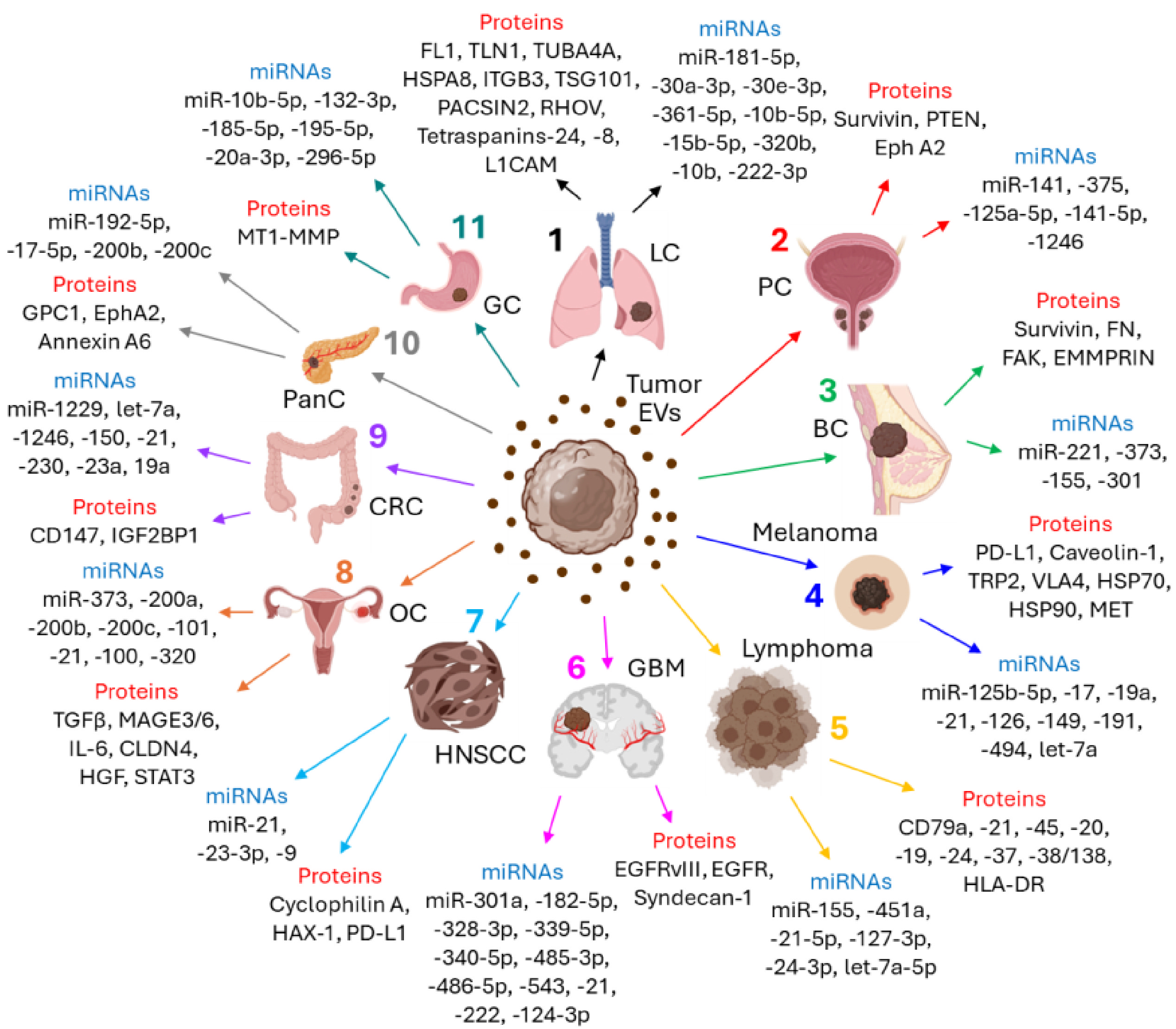 Fig. 2.
Fig. 2.
Circulating EVs, via the transfer of bioactive proteins and
miRNAs, serve as biomarkers for different types of cancer. (1) Lung cancer (LC):
Circulating EVs’ carried proteins as biomarkers- FN1, TLN1, TUBA4A, HSPA8, ITGB3,
TSG101, PACSIN2, RHOV, Tetraspanins-24, -8, L1CAM; miRNAs as biomarkers-
miR-181-5p, -30a-3p, -30e-3p, -361-5p, -10b-5p, -15b-5p, -320b, -10b, -222-3p;
| Cancer type | Protein/s name | Found in | Up/down regulation | Reference |
| Lung cancer | FN1, TLN1, TUBA4A, HSPA8, ITGB3, TSG101, PACSIN2 | Plasma | Present | [130] |
| (LUAD) | RHOV | Plasma | Up |
[131] |
| CD151/tetraspanin-24, tetraspanins-8, D171/L1CAM | Plasma | Present | [132] | |
| Prostate cancer | Survivin | Plasma | Up |
[133] |
| PTEN | Blood | Up |
[134] | |
| Eph A2 | Serum | Present | [135] | |
| Breast cancer | FN, FAK | Plasma | Up |
[136] |
| Survivin, spliced variants | Serum | Up |
[137] | |
| EMMPRIN | Peripheral blood | Up |
[138] | |
| Melanoma | PD-L1 | Serum | Present | [139] |
| Caveolin-1 | Plasma | Present | [140] | |
| TRP-2, VLA-4, HSP70, HSP90, MET | Plasma | Present | [141] | |
| Lymphoma | ||||
| (DLBCL) | CD79a, CD21, CD45, CD20 | Plasma | Present | [142] |
| (BCL) | CD19, CD20, CD24, CD37, HLA-DR | Serum | Present | [143] |
| (MM) | CD38/CD138 | Serum | Present | [144] |
| (AIDS-NHL) | PD-L1, CD40, CD40L or TNF-RII | Plasma | Up |
[145] |
| Leukemia | ||||
| (BCP-ALL) | CD317, CD38, IGF2BP1, PCNA, CSDE1, and GPR116 | Plasma | Present | [146] |
| (B-ALL) | CD29, CD146 | Plasma | Up |
[147] |
| (AML) | CD44, CD105, CD133, and SSEA-4 | Plasma | Up |
[147] |
| GBM | EGFRvIII | Serum | Present | [148] |
| EGFR and EGFRvIII | Serum | Present | [149] | |
| Syndecan-1 | Plasma | Present | [150] | |
| HNSCC | ||||
| (NPC) | Cyclophilin-A | Serum | Present | [151] |
| (NPC) | HAX-1 | Serum | Up |
[152] |
| PD-L1 | Plasma | Down |
[153] | |
| Ovarian cancer | TGF- |
Plasma | Up |
[154] |
| Claudin 4 | Plasma | Present | [155] | |
| HGF, STAT3, IL-6 | Serum | Present | [156] | |
| Colorectal cancer | CD147 | Serum | Up |
[157] |
| IGF2BP1 | Plasma | Up |
[158] | |
| Pancreatic cancer | GPC1 | Plasma | Present | [159] |
| Annexin A6 | Serum | Present | [160] | |
| EphA2 | Plasma | Up |
[161] | |
| Gastric cancer | MT1-MMP | Serum | Up |
[162] |
Abbreviations: TLN1, Talin 1; TUBA4A, tubulin alpha-4A chain; HSPA8,
heat shock protein member 8; ITGB3, integrin beta 3; TSG101, tumor susceptibility
gene 101; PACSIN2, protein kinase C and casein kinase substrate in neurons 2;
LUAD, lung adenocarcinoma; RHOV, Ras homolog family member V; L1CAM, L1 cell
adhesion molecule; PTEN, phosphatase and tensin homolog; FAK, focal adhesion
kinase; EMMPRIN, extracellular matrix metalloproteinase inducer; PD-L1,
programmed cell death ligand 1; TRP-2, Tyrosinase-related protein-2; VLA-4, very
late antigen 4; MET, mesenchymal-epithelial transition factor; DLBCL, diffuse
large B-cell lymphoma; BCL, B-cell lymphoma; HLA-DR, human leukocyte antigen-DR
isotype; MM, multiple myeloma; AIDS-NHL, acquired immunodeficiency syndrome
(AIDS)-related non-Hodgkin lymphoma; BCP-ALL, B-cell precursor acute myeloblastic
leukemia; CD, cluster of differentiation; IGF2BP1, insulin like growth factor
2 mRNA binding protein 1; PCNA, proliferating cell nuclear antigen; CSDE1, cold
shock domain-containing protein E1; GPR116, G-protein coupled receptor 116;
SSEA4, stage-specific embryonic antigen 4;
AML, acute myeloid leukemia; GBM, glioblastoma; EGFRvIII, epidermal
growth factor receptor variant III; HNSCC, head and neck squamous cell carcinoma;
NPC, nasopharyngeal carcinoma; HAX-1, HS1-associated protein X-1; TGF-
| Cancer type | miRNA/s name/s | Found in | Up/down-regulation | Reference/s |
| Lung cancer | ||||
| (NSCLC) | miR-181-5p, miR-30a-3p, miR-30e-3p, miR-361-5p | Plasma | Present | [163] |
| (LSCC) | miR-10b-5p, miR-15b-5p, miR-320b | Plasma | Present | [163] |
| (NSCLC) | miR-10b | Plasma | Present | [164] |
| (NSCLC) | miR-222-3p | Serum | Up |
[165] |
| Prostate cancer | miR-141, miR-375 | Serum, plasma | Up |
[166] |
| miR-125a-5p/miR-141-5p | Plasma | Present | [167] | |
| miR-1246 | Serum | Present | [168] | |
| Breast cancer | miR-221 | Plasma | Up |
[31] |
| miR-373 | Serum | Up |
[169] | |
| miR-155, miR-301 | Plasma | Down |
[170] | |
| Melanoma | miR-125b-5p | Serum | Up |
[171] |
| miR-17, miR-19a, miR-21, miR-126, miR-149 | Plasma | Up |
[172] | |
| miR-191, let-7a and miR-494 | Serum | Present | [173, 174] | |
| Lymphoma | ||||
| (MM) | miR-125b | Plasma | Up |
[175] |
| (DLBCL) | miR-155 | Plasma | Up |
[176] |
| (DLBCL) | miR-451a | Serum | Up |
[177] |
| (HL) | miR-21-5p, miR-127-3p, let-7a-5p, miR-155-5p, miR-24-3p | Plasma | Down |
[195] |
| (ALCL) | miR-146a-5p | Liquid biopsy | Up |
[178] |
| GBM | miR-301a | Serum | Down |
[179, 180] |
| miR-182-5p, miR-328-3p, miR-339-5p, miR-340-5p, miR-485-3p, miR-486-5p, miR-543 | Serum | Present | [181] | |
| miR-21, miR-222, miR-124-3p | Serum | Up |
[182] | |
| HNSCC | ||||
| (LSCC) | miR-21 | Serum | Up |
[183] |
| miR-24-3p | Serum | Up |
[184] | |
| miR-9 | Serum | Up |
[185] | |
| Ovarian cancer | miR-373, miR-200a, miR-200b, miR-200c | Serum | Up |
[186] |
| miR-101 | Serum | Present | [187] | |
| miR-21, miR-100, miR-200b, miR-320 | Plasma | Present | [188] | |
| Colorectal cancer | let-7a, miR-1229, miR-1246, miR-150, miR-21, miR-223, miR-23a | Serum | Up |
[189] |
| miR-19a | Serum | Up |
[190] | |
| Pancreatic cancer | miR-192-5p | Plasma | Up |
[191] |
| miR-200b, miR-200c | Serum | Up |
[192] | |
| miR-17-5p | Serum | Up |
[193] | |
| Gastric cancer | miR-10b-5p, miR-132-3p, miR-185-5p, miR-195-5p, miR-20a-3p, miR-296-5p | Serum | Present | [194] |
Abbreviations: NSCLC, non-small cell lung cancer; LSCC, lung squamous
cell carcinoma; miR, microRNA; DLBCL, diffuse large B-cell lymphoma; HL,
Hodgkin’s lymphoma; GBM, glioblastoma; HNSCC, head and neck squamous cell
carcinoma; ALCL, anaplastic large cell lymphoma;
Lung cancer is considered to be one of the most common causes of cancer-related death worldwide [196]. EVs often serve as biomarkers for lung cancer. For example, signature protein molecules such as Fibronectin 1 (FN1), Talin 1 (TLN1), tubulin alpha-4A chain (TUBA4A), heat shock protein member 8 (HSPA8), Integrin beta 3 (ITGB3), tumor susceptibility gene 101 (TSG101), and protein kinase C and casein kinase substrate in neurons 2 (PACSIN2) are readily detected in the plasma EVs of lung cancer patients, thereby serving as lung cancer biomarkers [130]. Ras homolog family member V (RHOV) is elevated in the plasma EVs of lung adenocarcinoma (LUAD) [131]. CD151/tetraspanin-24, CD171/L1CAM, and tetraspanin-8 are exclusively present in the plasma EVs of patients with histological lung cancer and can be used as predictive biomarkers for lung cancer [132]. Four signature miRNAs, namely miR-181-5p, miR-30a-3p, miR-30e-3p, and miR-361-5p as well as three other miRNAs, such as miR-10b-5p, miR-15b-5p, miR-320b are found in NSCLC and lung squamous cell carcinoma (LSCC), respectively, and hence, demonstrated as predictive biomarkers for early diagnosis [163]. A report from Zhong et al. [164] indicates that miR-10b is an excellent plasma EVs biomarker for NSCLC. Gemcitabine-resistant A549 cells trigger the sorting of miR-222-3p into the EVs which are readily taken up by parental-sensitive recipient cells, leading to increased metastasis and gemcitabine resistance [165].
EVs often contribute to the development and progression of prostate cancers and are easily recognized as biomarkers for prostate cancer. Survivin expression in the plasma EVs of prostate cancer patients is shown to be significantly higher than the healthy controls [133]. PTEN, a tumor suppressor protein, is selectively packaged into the blood-borne EVs of prostate cancer patients but not in healthy subjects and is considered a diagnostic biomarker for prostate cancer [134]. The ‘area under the receiver operating characteristic (ROC) curve’ (AUC) value of Ephrin A2 in the serum of aggressive prostate cancer patients is 0.906 as compared to benign prostate hyperplasia (BPH) [135]. Furthermore, metastatic prostate cancer has high levels of miR-141 and miR-375 in the plasma and serum EVs [166]. The AUC value of miR-125a-5p/miR-141-5p ratio in plasma EVs of distinguishing prostate cancer patients is 0.793, thereby allowing them to be used as diagnostic biomarkers for prostate cancer [167]. miR-1246 has been identified by NanoString nCounter technology as one of the predominant miRNAs in the serum EVs of aggressive prostate cancer patients [168].
FN and focal adhesion kinase (FAK) level in the plasma EVs of breast cancer patients is well-elevated as compared to healthy controls, contributing to the diagnostic accuracy of breast cancer [136]. The level of Survivin and its spliced variants is significantly higher in the serum EVs of early breast cancer patients which serve as prognostic and diagnostic biomarkers for early breast cancer [137]. The glycosylated form of the extracellular matrix metalloproteinase inducer (EMMPRIN) is significantly increased in the peripheral blood-derived EVs of highly metastatic breast cancer patients, considering a predictive biomarker for metastatic breast tumors [138]. Coagulation protease activated factor VII (FVIIa) promotes the aggressiveness of human breast cancer [197, 198] which is already shown to be mediated by the release of EVs. The level of miR-221 is found to be significantly higher in the plasma EVs of highly aggressive invasive ductal carcinoma patients as compared to healthy individuals [31] which could be used as a predictive biomarker for this type of cancer. An elevated level of miR-373 is reported in the serum EVs of highly aggressive, triple-negative breast cancer (TNBC) patients, rendering it a promising biomarker for TNBC [169]. The down-regulation of miR-155 and miR-301 in the plasma EVs of breast cancer patients is frequently observed in post-neoadjuvant therapy through uni- and multivariate models, predicting pathological complete response [170].
PD-L1, which is exclusively found in the serum EVs of melanoma patients, is proven to be the diagnostic biomarker for melanoma [139]. Moreover, caveolin-1, usually associated with metastasis and progression of different types of cancer [199], is also detected in the plasma EVs of melanoma patients [140]. Peinado et al. [141] have documented signature EVs-associated proteins in the plasma of melanoma patients which include tyrosinase-related protein 2 (TRP-2), VLA-4, HSP70, HSP90, and mesenchymal-epithelial transition factor (MET). miR-125b-5p, recognized as one of the most abundant miRNAs in the melanoma cell lines, is often found in the serum EVs of melanoma patients and hence, act as a predictive biomarker [171]. miR-17, miR-19a, miR-21, miR-126, and miR-149 are shown to be enriched in the plasma EVs of metastatic sporadic melanoma patients as compared to healthy controls [172]. EVs-associated miR-191 [173], let-7a [173] and miR-494 [174] are also detected in the circulation of melanoma patients in clinical settings.
Elevated expression of plasma EVs is observed in canine diffuse large B-cell lymphomas (DLBCL) as compared to healthy controls, which are positive for pan B-cell markers, CD79a, CD21, CD45, CD20 [142]. The presence of CD19, CD20, CD24, CD37, and human leukocyte antigen - DR isotype (HLA-DR) on the serum EVs of B-cell lymphomas (BCL) is also reported [143]. CD38/CD138+ EVs in the serum of multiple myeloma (MM) patients often turn out to be excellent diagnostic biomarkers for MM [144]. The level of miR-125b in the plasma EVs of MM and lymphoma patients is shown to be up-regulated in post-autologous hematopoietic stem cell transplantation (AHSCT) conditions, hence EVs’ miR-125b can be considered as a predictive biomarker for the outcome after AHSCT [175]. A higher population of EVs and EVs-associated miR-155 are detected in the plasma of refractory/relapsed DLBCL patients. Similarly, a significant level of miR-451a in the serum EVs of DLBCL patients was observed post-chemo- and radiation therapy with an AUC of 0.8038 [177]. A study by Matthiesen et al. [200] delineates that EVs proteins from the plasma of DLBCL patients often represent the disease onset and can be used as prognostic biomarkers for DLBCL. Furthermore, a study by Rutherford et al. [201] delineates that mutation in certain EV RNAs often represents a mutation in the originating cells, which could be explored in understanding the pathogenesis of DLBCL. A significant down-regulation of miR-21-5p, miR-127-3p, let-7a-5p, miR-155-5p, and miR-24-3p in the plasma EVs of Hodgkin’s lymphoma (HL) is monitored after successful treatment, which further gets up-regulated when the disease is relapsed. Repetto et al. [202] further identified 89 unique EV cargo proteins in the plasma of pediatric HL patients with a relapsed occurrence which were undetectable in healthy plasma, thereby can be considered as a potential source of biomarkers for relapsed occurrence of HL. Interestingly, acquired immunodeficiency syndrome (AIDS)-related non-Hodgkin lymphoma (AIDS-NHL) patients express significant level of PD-L1, CD40, CD40L or TNF-RII in the plasma EVs which are reduced post-cancer treatment, thereby rendering these signature molecules as biomarkers for AIDS-NHL [145]. The expression of miR-146a-5p is shown to be significantly up-regulated in the EVs of liquid biopsies of relapsed pediatric anaplastic large cell lymphoma (ALCL) patients which promote macrophage infiltration and differentiation, contributing to ALCL aggressiveness [178].
A growing body of evidence indicates that EVs play a major role in the pathogenesis of different types of leukemia and EVs have been purposefully used in recent times in leukemia diagnosis and treatment [203, 204]. The next-generation sequencing (NGS) technique determines the concentration and mutations in specific double-stranded DNA (dsDNA) in the EVs isolated from the liquid biopsy of acute myeloid leukemia (AML) patients which can be used as biomarkers for AML detection [205]. In a contemporary study, Kontopoulou et al. [206] also identified certain leukemic-specific mutations in the plasma EVs’ dsDNA of AML patients which were undetectable following treatment, thus playing a critical role in AML diagnosis. Recently, Saidu et al. [146] have demonstrated the presence of six signature proteins, CD317, CD38, insulin-like growth factor 2 mRNA-binding protein 1 (IGF2BP1), PCNA, CSDE1, and GPR116 in the plasma EVs of B-cell precursor acute lymphoblastic leukemia (BCP-ALL) patients as compared to healthy controls which can be used as biomarkers for BCP-ALL. Moreover, the expression of CD29 and CD146 is shown to be significantly higher in the plasma EVs of B-ALL patients whereas a higher expression of CD44, CD105, CD133, and SSEA-4 is observed in AML patients’ plasma EVs, aiding them to be considered as prognostic biomarkers for leukemia [147]. A study by Ghetti et al. [207] indicates that a higher expression of circular RNA PVT1 (circPVT1) in the plasma EVs of AML patients correlates with AML pathogenesis. In addition to the above findings, Li et al. [208] have delineated signature miRNA patterns in the serum EVs of AML patients which have the potential to become diagnostic biomarkers for AML.
EVs-associated epidermal growth factor receptor variant III (EGFRvIII) is readily detectable in the serum of glioblastoma (GBM) patients, whereas control groups show no evidence of EGFRvIII in the EVs [148]. Another group also reported the presence of EGFR and EGFRvIII in the serum EVs of GBM patients, thereby denoting these EVs-associated molecules as promising biomarkers for GBM [149]. Moreover, syndecan-1 is often detected in the plasma EVs of GBM patients which efficiently distinguish between low-grade and high-grade GBM tumors [150]. miR-301a could be used as a predictive biomarker for GBM as its expression in the serum EVs is shown to be down-regulated post-surgical resection of the tumor, however, further increased during GBM recurrence [179, 180]. A panel of seven serum EVs-associated miRNAs, miR-182-5p, miR-328-3p, miR-339-5p, miR-340-5p, miR-485-3p, miR-486-5p, and miR-543 are used to differentiate between GBM patients and normal healthy individuals [181]. Three miRNAs, miR-21, miR-222, and miR-124-3p are reported to be significantly elevated in the serum EVs of glioma patients and are often used as predictive biomarkers for glioma [182].
Cyclophilin A (CYPA), which is derived from immunophilin, is exclusively present in the serum EVs of nasopharyngeal carcinoma (NPC) and is considered a diagnostic biomarker for NPCs [151]. The serum EVs of NPC patients are enriched with HS1-associated protein X-1 (HAX-1), which serves as a biomarker for lymph node metastasis (LNM) of the NPC [152]. PD-L1 expression on circulating EVs is correlated with the LNM status of HNSCC, a higher PD-L1 expression exhibits longer disease-free survival and low EVs-PD-L1 expression signifies a high risk of recurrence or relapse [153]. Moreover, a higher serum EVs’ miR-21 expression corresponds to an LNM potential of laryngeal squamous cell carcinoma (LSCC), and hence EVs’ miR-21 is considered to be the diagnostic and prognostic biomarker for LSCC [183]. Again, miR-24-3p is found to be enriched in the serum EVs of nasopharyngeal carcinoma patients which correlates with worse disease-free survival [184]. miR-9 in the serum EVs of nasopharyngeal carcinoma patients positively correlates with overall survival of patients [185].
Transforming growth factor
The level of CD147+ EVs in the serum of colorectal cancer (CRC) patients is shown to be significantly higher as compared to healthy donors, however, their level significantly drops after surgery which allows these EVs to be considered as potential biomarkers for CRC. A recent study indicates that insulin-like growth factor 2 RNA binding protein 1 (IGF2BP1) is highly enriched in the plasma EVs of CRC xenograft mice which could serve as a diagnostic or prognostic biomarker for CRC [169]. Moreover, seven signature miRNA molecules, let-7a, miR-1229, miR-1246, miR-150, miR-21, miR-223, and miR-23a, are identified to be encapsulated within the serum EVs of CRC patients whose level is significantly higher in primary or early stages of CRC than healthy control and surgical resection of tumor down-regulates their abundance [189]. miR-19a is also shown to be up-regulated in the serum EVs of stage I and -II CRC patients concerning healthy controls and thus can be used as a prognostic biomarker for CRC [190].
More often, GPC1 (glypican 1) is shown to be associated with the plasma EVs of pancreatic cancer patients with an AUC value of 0.59 [159]. Annexin A6 is also found in the serum EVs of pancreatic cancer patients, thereby rendering it a predictive biomarker for pancreatic cancer. Moreover, ephrin type-A receptor 2 (EphA2) can also be referred to as a promising prognostic biomarker for pancreatic cancer as their level is significantly elevated in the plasma EVs of pancreatic cancer. Enriched miR-192-5p level in the plasma EVs of pancreatic cancer renders it to become a diagnostic biomarker for pancreatic cancer [191]. On the other hand, miR-200b and miR-200c are over-expressed in the serum EVs of PDAC, thereby often used as promising biomarkers for PDAC [192]. A higher level of miR-17-5p expression is observed in the serum EVs of PDAC patients as compared to chronic pancreatitis and healthy controls, therefore, can be correlated with metastatic and advanced stages of PDAC [193].
A high expression of membrane type 1 matrix metalloproteinase (MT1-MMP) in the serum EVs of gastric cancer patients is associated with the lymphatic metastasis of the tumor and hence can predict the risk of gastric cancer metastasis [162]. Moreover, six signature miRNA molecules, miR10b-5p, miR132-3p, miR185-5p, miR195-5p, miR-20a3p, and miR296-5p, associated with the serum EVs, are frequently used for diagnosing gastric cancer [194].
Heterogenous composition and diverse functions have rendered EVs an attractive vehicle for delivering drugs to target cells. The advancement of EV research from the past two decades indicates that EVs can be used as an important therapeutic nanomaterial. Due to biocompatibility, reduced toxicity, low immunogenicity, and ability to cross physiological barriers, e.g., blood-brain barrier, EVs are considered excellent therapeutic nanosized bodies when compared to traditional pre-existing carriers. In addition, EVs are biodegradable, and their ability to escape the host immune responses makes them an attractive option against various diseased conditions, most importantly cancer [209, 210]. The first EVs-based successful therapeutic application was the delivery of EVs-encapsulated siRNA into the murine brain which showed a significant down-regulation of the target gene expression [211]. The administration of chemotherapeutic drugs, entrapped within the tumor cell-derived EVs, into the recipient cells showed an enhanced level of drug-induced cytotoxicity [212]. However, EVs exert their therapeutic effects via three basic mechanisms- (1) elimination of the EVs in the circulation, (2) inhibition of EVs release, and (3) the disruption of EVs’ absorption.
One of the best EVs-related cancer therapeutic strategies has been thought to focus on removing EVs that cancer cells secrete. The current review thus far illustrates the various mechanisms by which EVs derived from cancer cells contribute to the progression and development of cancer. In the first intriguing study, Marleau et al. [213] used a hemofiltration system to target the human epidermal growth factor receptor 2 (HER-2) on the surface of EVs, which led to the elimination of HER-2+ EVs and may have been a useful approach for the treatment of cancer. The second strategy, which involves EV release perturbation, has received a lot of consideration in studies. It has been demonstrated that the sphingomyelinase inhibitor GW4869 blocks the biogenesis of EVs by concentrating on both the production of intraluminal vesicles and the subsequent fusion of MVBs with the plasma membrane. Additionally, inhibition of neutral sphingomyelinase 2 (nSMase 2), which is crucial for tumor metastasis and angiogenesis through the transfer of oncogenic miR-210-3p, results in a reduction in the release of EVs from cancer cells. In contrast, prostate cancer-related nSMase 2 perturbations do not prevent EV release. Additionally, it has been found that nSMases are present in healthy neural cells, which is consistent with the idea that they are involved in fundamental processes rather than EV release. Additionally, Datta et al. [214] have recently shown that several substances that do not affect the function of nSMase 2 either promote or inhibit the release of EVs from prostate cancer cells. These show that the biogenetic mechanism of EVs differs in various cancer types, which is important for the creation of anti-cancer drugs. EVs, which are released from the other cells, frequently help to eradicate cancer. For instance, EVs secreted by NK cells have anti-melanoma activity, which is crucial for creating treatments for the disease. Additionally, it has been demonstrated that EVs derived from dendritic cells carry class I and class II MHC molecules on their surface, which activates the immune system and aids in the antitumor immune responses. However, it can be difficult to use this strategy. The main drawbacks of this approach are the unclear mechanism of EV biogenesis, low product yield that is accompanied by reduced immunogenicity, and higher likelihood of EV content mutations. However, it has been discovered that the use of these endogenous EVs for drug delivery against cancer types is quite effective. The parental cells are frequently engineered to carry therapeutic cargoes, and when these cells release EVs, they are loaded with these therapeutics so they can be efficiently delivered to the cancer cells. RBCEVs, a group of red blood cell (RBC)-derived EVs, are used successfully in breast cancer and leukemia cell studies because they improve miRNA inhibition and CRISPR-Cas9 genome editing while also exhibiting low toxicity. Additionally, miR-379 abundant EVs are released when mesenchymal stem cells overexpress the miRNA, and these EVs inhibit the spread of metastatic breast cancer. Moreover, emerging evidence indicates that chimeric antigen receptor (CAR)-T cells are genetically engineered to kill tumor cells, thereby showing promising outcomes in cancer immunotherapy. However, CAR-T cell therapy also has significant adverse effects. EVs released from CAR-T cells not only show limited adverse effects but also the ability of the EVs to reach certain places that are inaccessible to the CAR-T cells often adds to the therapeutic value of CAR-T cells in the treatment of cancer [215, 216].
On several occasions, it has been found that the data obtained in preclinical conditions could not be replicated in human subjects. A well-effective drug in preclinical settings is often shown to lose its effectiveness while applied on humans. Therefore, before releasing the drug into the market, its effectiveness should be thoroughly analyzed in clinical trials. Emerging evidence indicates that EVs actively participate in the progression of different types of human cancer. Recently, several EV-based cancer therapeutics are also available in clinical trials. Table 4 briefly summarizes the role of EVs as cancer biomarkers and therapeutics.
| Cancer type | NCT number | EV type | EV source | EV isolation and characterization technique |
| Breast Cancer | NCT05798338 | EVs | Plasma | Single Molecule Array (SiMoA)-ELISA |
| NCT05831397 | EVs | Plasma | SiMoA-ELISA | |
| NCT05417048 | EVs | Serum | Gly-Exo Capture technology | |
| Meningioma | NCT06104930 | EVs | Plasma | Nanopore sequencing-based DNA methylation profiling of EV DNA |
| Brain tumor | NCT06501521 | EVs | Plasma | Transcriptomics |
| NSCLC | NCT05424029 | Exosomes | Bronchial fluid | DNA profiling |
| NCT05469022 | EVs | BALF | Determination of EV EGFR mutation status | |
| NCT05732974 | EVs | Plasma, tissue | Analysis of EV miRNA | |
| NCT04564079 | EVs | Plasma | RNA sequencing to characterize EV small RNA | |
| NCT05724004 | EVs | Plasma | Protein profiling and RNA expression | |
| SCLC | NCT05191849 | EVs | Blood | Identifying EV long RNA profiles |
| Lung cancer | NCT05587114 | Exosomes | Peripheral and pulmonary blood | Exosome characterization by NTA, TEM and western blotting |
| NCT05854563 | EVs | Cough, BALF, Mouthwash | EV DNA mutation analysis will be done by using Single Molecule Mutation sequencing (SMM-seq), EV protein analysis by proteomics | |
| Prostate cancer | NCT06326216 | EVs | Blood, urine | EVs level will be evaluated and prostate specific antigen (PSA) in the EVs will be analyzed |
| NCT06046131 | EVs | Urine | EVs characterization as a measure of disease progression | |
| Upper GI cancer | NCT06278064 | Exosomes | Plasma | Exosomal proteins will be analyzed by Advanced Data-Independent Acquisition (DIA) proteomics and single-vesicle membrane protein detection techniques |
| Rectal cancer | NCT04852653 | Exosomes | Blood | Determining the presence of oncoexosomes and exosomal DNA (ExoDNA) |
| Pancreatic cancer | NCT05625529 | EVs | Blood | Using alternating current electric (ACE) field-based ExoVerita™ assay to isolate EVs for multiomics study |
| NCT05929885 | Exosomes | Plasma | Identification of exosomal proteins by mass spectrometry | |
| Hematopoietic and lymphoid neoplasm | NCT06192875 | EVs | Urine | Assessing methylated DNA markers (MDMs), RNA, or protein in EVs to measure disease progression |
| DLBCL | NCT06439173 | EVs | MSC | Determining the role of MSC-EVs in post-CAR-T therapy repair of bone marrow microenvironment |
| Renal cancer | NCT04053855 | Exosomes | Urine | Using electron microscopy, flow cytometry, and RT-qPCR in analyzing the exosomes for measuring disease progression |
| Liver cancer | NCT06236568 | EVs | Liquid biopsy | Identification of miRNAs by transcriptomics |
Abbreviations: EV, extracellular vesicle; BALF, bronchoalveolar lavage fluid; NTA, nanoparticle tracking analysis; TEM, transmission electron microscope; NSCLC, non-small cell lung cancer; SCLC, small cell lung cancer; GI, gastrointestinal; DLBCL, diffuse large B cell lymphoma; RT-qPCR, reverse transcription-quantitative polymerase chain reaction; NCT, national clinical trial; MSC, mesenchymal stem cells. (https://clinicaltrials.gov/; accessed on 24th August 2024).
Biomarkers are indispensable tools in clinical applications, they not only help delineate disease phenotypes and prognosis but are also vastly used to measure various treatment responses. Unlike some invasive techniques, the innocuous procedures used to obtain these biomarkers enable continuous measurement. It is well-established that early disease detection is highly correlated with optimum treatment outcomes in all forms of ailments, and this especially holds for the most aggressive forms of cancer.
The heterogeneous nature of EVs, along with their diverse cargoes and surface markers makes them a compelling choice for use as biomarkers. Their diverse payload not only contains lots of biological information but also provides significant details about the status of their potential site of origin. Moreover, the inherent stability makes them amenable to be utilized in different protocols and minimally invasive procedures with which they can be isolated opening possibilities for remote monitoring. The present review highlights how the differential expression of EVs’ cargoes in the form of proteins, miRNAs, etc. contributes to the pathogenesis of different types of cancer by various unique mechanisms. Identification and characterization of such EV biomarkers not only aid in identifying the disease stages and severity which help in the disease diagnosis, but also open a new therapeutic window in the treatment of cancer besides conventional therapeutic means.
Despite the huge potential, there remain several challenges associated with using EVs as biomarkers, which is primarily due to several challenges associated with the identification and separation of the target EVs from the background population as well as lack of standardization regarding EVs’ isolation techniques. Unsurprisingly, research has been strong in these areas and commercial kits have shown promising outcomes, for example, ExosomeDx® and ExoVita® for assessing prostate cancer and pancreatic cancer risk, respectively [217, 218]. Burgeoning interest in EVs along with the increasing pace of research will only accelerate its potential applications and thus it is only a matter of time before we see EVs being used for diverse bedside clinical applications ranging from therapy to biomarkers.
EVs, Extracellular vesicles; MVs, Microvesicles; MVBs, Multivesicular bodies; ESCRT, Endosomal sorting complexes required for transport; TME, Tumor microenvironment; miRNA, MicroRNA; KRAS, Kirsten rat sarcoma virus; EGFR, Extracellular growth factor receptor; MMP, Matrix metalloproteinase; ECM, Extracellular matrix; CAF, Cancer associated fibroblasts; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; TNF, Tumor necrosis factor; NK cells, Natural killer cells; PS, Phosphatidylserine; IL, Interleukin; RBC, Red blood cell; TGF
MC contributed to the literature search, data collection, and preparation of the manuscript. SG and SN contributed to the literature search. IR contributed to the draw charts and preparation of the revised manuscript. DP contributed to the draw charts and critical review and preparation of the final version of the manuscript. AM contributed to the study design, critical review, and preparation of the manuscript. KD contributed to the conceptualization, literature search, study design, critical review, and preparation of the final version of the manuscript. All authors have read and agreed to the published version of the manuscript. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
All the images in the manuscript were prepared with BioRender.com. We acknowledge that BioRender provided us with the platform for the preparation of images. We also acknowledge all the peer reviewers for their opinions and suggestions.
Kaushik Das received the Ramalingaswami Re-entry Fellowship (Ref: BT/HRD/35/02/2006) from the Department of Biotechnology, Government of India.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


