1 Center of Excellence for Poultry Science, Division of Agriculture, University of Arkansas, Fayetteville, AR 72701, USA
Abstract
Water scarcity is a current, significant global concern that will only increase under the pressure of climate change. Improving water efficiency of poultry is a new and promising area to help temper agriculture's future impact on fresh water availability. Here, we explored the effects of acute heat stress (HS) on circulating stress and inflammatory markers in 2 lines of broilers divergently selected for water efficiency.
Male chicks from low (LWE) and high water efficient (HWE) lines were raised in 12 environmental chambers (2 pens/chamber, 6 chambers/line, 20 birds/pen) under normal conditions until day 28. On day 29, birds were subjected to thermoneutral (TN, 25 °C) or HS (36 °C) conditions, resulting in four treatments (2 lines × 2 environmental conditions). After 3 h of HS, whole blood was collected (8 birds per line × environment) and analyzed for target gene expression and plasma cytokine levels. Data were analyzed by 2-way ANOVA, with line, environment, and their interaction as main factors, and means were compared using Tukey's multiple range test.
Gene expression of heat shock protein (HSP) 27, HSP70, interleukin (IL)-6, IL-18, c-reactive protein (CRP), tumor necrosis factor-α (TNFα), C-C motif chemokine ligand 4 (CCL4), CCL20, nucleotide-binding domain, leucine-rich repeat (NLR) family pyrin domain containing 3 (NLRP3), NLR family CARD domain containing 5 (NLRC5), and NLR family member X1 (NLRX1) were increased by HS, with no differences between the lines. HSP70, IL-10, and NLRC3 were lower in the HWE as compared to the LWE lines. Additionally, there were interactive effects between line and environment for HSP90, IL-4, and CCL4, where HS induced HSP90 expression in the LWE only, and IL-4 and CCL4 in HWE only. Arginine vasopressin (AVP) gene expression was significantly lower in the whole blood of the HWE line; however, plasma protein levels were not different.
Overall, most of the effects seen on cyto (chemokines) and inflammatory markers were due to acute HS, with only a few genes differentially regulated between the lines. This likely indicates that the divergent selection for water efficiency for four generations did not elicit changes in inflammation and stress molecular signatures.
Keywords
- broilers
- water efficiency
- heat stress
- cytokine
- chemokine
- inflammasome
- arginine vasopressin
Water scarcity is a real and pressing concern worldwide. Only approximately 3% of the world’s water is freshwater, and only 0.5% is usable [1]. Importantly, agriculture uses over 70% of freshwater resources [2]. As climate change and global population expansion will further tax an already strained resource, it is estimated that two thirds of the global population, or 4 billion people, will experience severe water shortages for at least one month a year [3]. Indeed, rising temperatures coupled with fluctuation in distribution and amounts of rainfall will dramatically change water resources and create uncertain availability. Although tackling water scarcity necessitates action at every level of use, agriculture is uniquely situated in that any reductions in water use must also maintain animal production and welfare standards to meet the needs of the growing population.
It is estimated that broiler chickens consume 1.6–2.0 grams of water for each gram of feed [4]. Considering an average market age of 47 days for US broilers in 2023, and an average feed intake [5], this equals 8–10 kg (2.2–2.7 gallons) of water consumed per bird during production. The US alone produced 9.16 billion broilers in 2023, which equates to over 20 billion gallons of drinking water consumed by these birds. Additionally, during times of elevated environmental temperatures, productivity, in terms of growth, feed efficiency, meat yields, and livability, is decreased and the volume of water consumed can increase significantly [6], further impacting water usage. Even a small improvement in water efficiency in broilers could save billions of gallons of water annually, allowing that water to be diverted for other uses. Recently, our research group has published on 2 lines of chickens divergently selected for water efficiency (ratio of water consumed to body weight gain). We have shown an 18% reduction in water intake in the high water efficient (HWE) as compared to their low water efficient (LWE) counterparts, while maintaining body weight gain (BWG) and improving in feed conversion ratio (FCR) [7]. Additionally, the HWE line performed better under chronic cyclic heat stress (HS) conditions, despite consuming less water and having a higher core body temperature [7]. In that study, however, the cumulative, and possibly adaptive, effects of 20 days of HS were examined. As older and heavier broilers are more susceptible to the negative effects of HS [8], we undertook this study to determine the effect of acute (3 h) HS on 49 d-old LWE and HWE chickens. In particular, increased heat load has been shown to induce oxidative stress and the inflammatory response through alteration of cytokines, chemokines, and other inflammatory markers [9, 10, 11, 12], likely contributing to growth declines and HS-productivity losses seen in poultry [13]. What is unknown, however, is how selection for water efficiency may impact these parameters. Determining the profile of these factors would be of significant interest to the poultry industry, as robustness (defined as maintaining a high level of production while being adaptable to a wide variety of conditions [14]) is a key goal of breeding and selection programs. Here, in order to further characterize these divergently selected lines under HS conditions, we report the gene expression and relative levels of circulating chemokines, cytokines, and other inflammatory markers.
All work was conducted in accordance with the National Institutes of Health recommendations guide for laboratory animal use and care. University of Arkansas Animal Care and Use Committee approved all procedures under protocol #23015. The genetic selection program for water efficiency was started in 2019, and has been previously described [7, 15]. HWE and LWE broiler chickens from the 4th generation were used in this study.
HWE and LWE chicks were hatched at the University of Arkansas hatchery.
On the day of hatch, male chicks (240 chicks/line) were
individually wing-banded for line identification and weighed. Chicks were
assigned by body weight-matched groups and placed into 12 environmental chambers
in the Poultry Environmental Research Laboratory at the University of Arkansas (2
floor pens/chamber, 6 chambers/line, 20 birds/pen, density of
0.096 m2/bird). Each pen was equipped with separate hanging
feeders and water lines covered, and with clean pine wood shavings. Three-phase
(starter, grower, and finisher) industry standard diets and water were provided
ad libitum. The ambient temperature was gradually decreased from 32
°C to 25 °C by day 21. The lighting program provided was
24 h light for the first 3 days,
23 h light:1 h dark from Day 4–7, and
18 h light:6 h dark for the remainder of the study.
The chambers’ temperature and humidity were continuously recorded using HOBO pro
V2 data loggers (ONSET, Bourne, MA, USA), and relative humidity was maintained at
~30%–40%. At Day 49, birds were exposed to either
thermoneutral (TN, 25 °C) or HS (36 °C for 3 h)
environmental conditions, which resulted in four treatments (2
lines
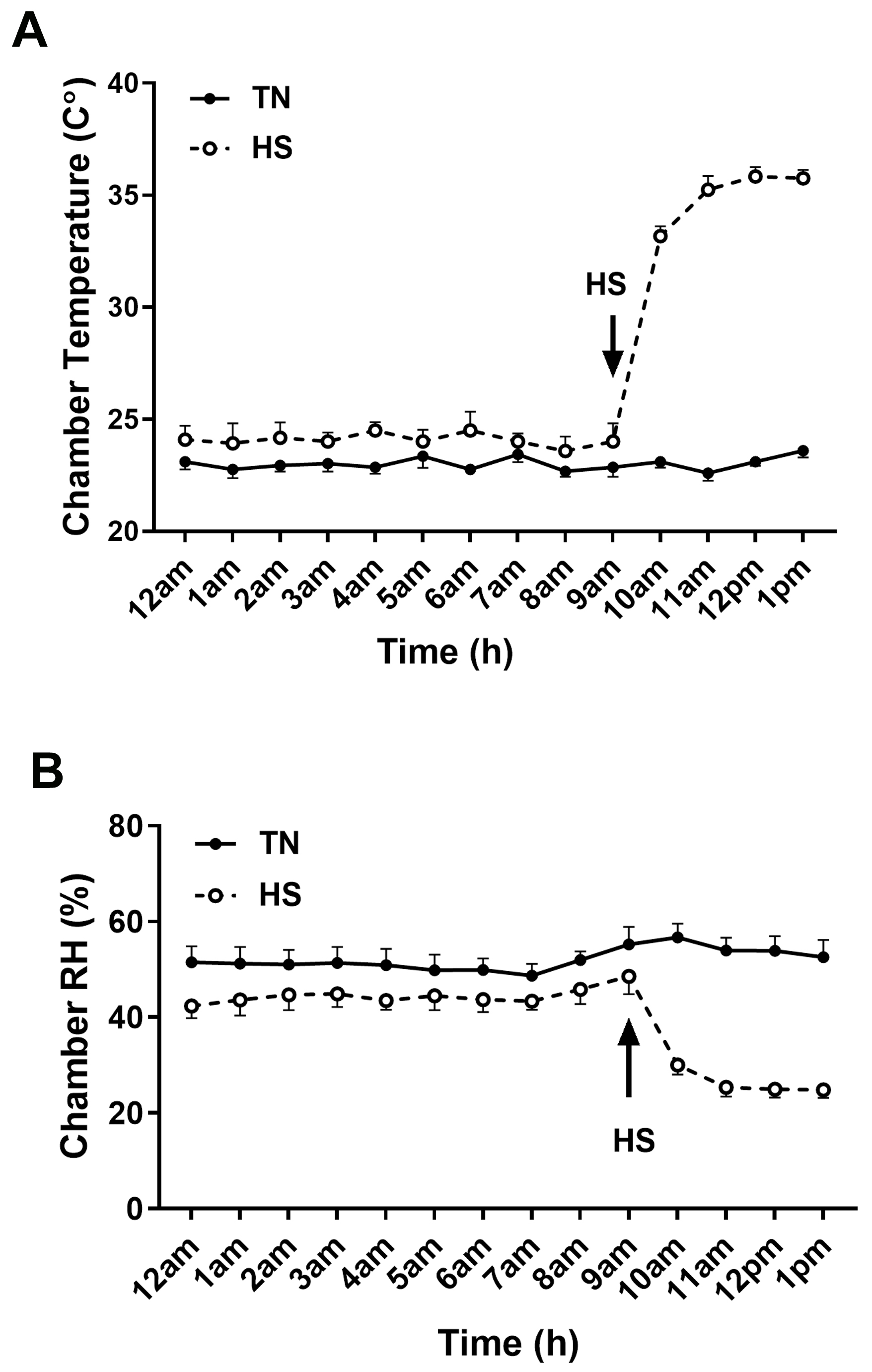 Fig. 1.
Fig. 1.
Chamber temperature and relative humidity. Chamber temperature (A) and relative humidity (B) on the day of blood sampling. Arrow indicates time of onset of HS. HS, heat stress; RH, relative humidity; TN, thermoneutral.
Whole blood was collected in Trizol LS reagent (Life Technologies, Carlsbad, CA, USA) and total RNAs were extracted based on the manufacturer’s guidelines. Total RNA concentration and quality were determined with use of the Take3 microvolume plate and the Synergy HTX multimode microplate reader (BioTek, Winooski, VT, USA). Reverse transcription was performed using qScript cDNA Synthesis Supermix (Quanta Biosciences, Gaithersburg, MD, USA), and cDNAs were amplified by real-time quantitative PCR (Applied Biosystems 7500 Real Time System, ThermoFisher Scientific, Waltham, MA, USA) with PowerUp SYBR green master mix (Life Technologies, Carlsbad, CA, USA) as previously described [16, 17]. Relative expression of the target genes was determined using the 2-ΔΔCT method [18]. Ribosomal 18S (18S) gene expression was used for normalization. Oligonucleotide primer sequences specific for chicken are listed in Table 1.
| Gene | Accession numbera | Primer Sequence | Orientation | Product size, bp |
| IL-4 | NM_001007079.2 | GCTCTCAGTGCCGCTGATG | F | 60 |
| GAAACCTCTCCCTGGATGTCAT | R | |||
| IL-6 | NM_204628.1 | GCTTCGACGAGGAGAAATGC | F | 63 |
| GGTAGGTCTGAAAGGCGAACAG | R | |||
| IL8-L1 | NM_205018.2 | CAGAACCAAACCCAGGTGACA | F | 61 |
| ACAGCCTTGCCCATCATCTT | R | |||
| IL-10 | NM_001004414.4 | CGCTGTCACCGCTTCTTCA | F | 63 |
| CGTCTCCTTGATCTGCTTGATG | R | |||
| IL-18 | NM_204608.1 | TGCAGCTCCAAGGCTTTTAAG | F | 63 |
| CTCAAAGGCCAAGAACATTCCT | R | |||
| TNF |
NM_204267.1 | CGTTTGGGAGTGGGCTTTAA | F | 61 |
| GCTGATGGCAGAGGCAGAA | R | |||
| CRP | NM_001039564.1 | AAGCTCAGGACAACGAGATCCT | F | 71 |
| TTTCCCCCCCACGTAGAAG | R | |||
| IFN |
NM_205149.2 | AAAGCCGCACATCAAACACA | F | 64 |
| GCCATCAGGAAGGTTGTTTTTC | R | |||
| NLRP3 | NM_001348947.1 | GTTGGGCAGTTTCACAGGAATAG | F | 63 |
| GCCGCCTGGTCATACAGTGT | R | |||
| NLRC5 | NM_001318435.2 | CTCGAAGTAGCCCAGCACATT | F | 81 |
| CATGTCCAGAGGTGTCAGTCTGA | R | |||
| NLRC3 | XM_015294675.2 | CTCCAACGCCTCACAAACCT | F | 93 |
| GCCTTTGGTCATTTCCATCTG | R | |||
| NLRX1 | XM_004948038.3 | GGCTGAAACGTGGCACAAA | F | 59 |
| GAGTCCAAGCCCAGAAGACAAG | R | |||
| CCL4 | NM_204720.1 | CCTGCTGCACCACTTACATAACA | F | 63 |
| TGCTGTAGTGCCTCTGGATGA | R | |||
| CCL20 | NM_204438.2 | TGCTGCTTGGAGTGAAAATGC | F | 62 |
| CAGCAGAGAAGCCAAAATCAAA | R | |||
| CCLL4 | NM_001045831.2 | CTTGCTGTCGGGTCCAATG | F | 60 |
| CGAGGGAAGTGCTCTGTTTAAGA | R | |||
| CXCL14 | NM_204712.2 | CCGGCTCGCCATGAAG | F | 54 |
| ATCGCGATGACCAGCAGAA | R | |||
| HSP27 | XM_046936397.1 | TTGAAGGCTGGCTCCTGATC | F | 58 |
| AAGCCATGCTCATCCATCCT | R | |||
| HSP60 | NM_001012916.3 | CGCAGACATGCTCCGTTTG | F | 55 |
| TCTGGACACCGGCCTGAT | R | |||
| HSP70 | J02579 | GGGAGAGGGTTGGGCTAGAG | F | 55 |
| TTGCCTCCTGCCCAATCA | R | |||
| HSP90 | NM_001109785.2 | TGACCTTGTCAACAATCTTGGTACTAT | F | 68 |
| CCTGCAGTGCTTCCATGAAA | R | |||
| AVP | NM_205185.3 | TCCGGGCACACTCAGCAT | F | 81 |
| ATGTAGCAGGCGGAGGACAA | R | |||
| 18S | AF173612 | TCCCCTCCCGTTACTTGGAT | F | 60 |
| GCGCTCGTCGGCATGTA | R |
aAccession number refers to GenBank (NCBI) and primers were produced by
Integrated DNA Technologies (IDT, Coralville, IA, USA); AVP, arginine
vasopressin; CCL4, C-C motif chemokine ligand 4; CCL20, C-C motif chemokine
ligand 20; CCLL4, chemokine-like ligand 4; CRP, c-reactive protein; CXCL14, C-X-C
motif chemokine ligand 14; HSP27, heat shock protein 27; HSP60, heat shock
protein 60; HSP70, heat shock protein 70; HSP90, heat shock protein 90;
IFN
Relative levels of circulating cytokines: caronte, interferon gamma
(IFN-
Total plasma proteins were quantified by the Bradford assay and subjected to western blot as previously described [19]. Primary antibodies utilized were rabbit anti-arginine vasopressin (AVP, 1:1000 dilution, #20069, ImmunoStar, Hudson, WI, USA) with rabbit anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH, 1:1000 dilution, #NB300-327, Novus Biologicals, Centennial, CO, USA) as a loading control. Membranes were incubated for 1 h at room temperature with secondary anti-rabbit IgG-HRP-linked antibody (1:5000, #7074S, Cell Signaling, Technology, Danvers, MA, USA). The protein signals were visualized by enhanced chemiluminescence (Super ECL, ABP Biosciences, Beltsville, MD, USA) and collected using the FluorChem M bioimager (Proteinsimple, Santa Clara, CA, USA). Image acquirement and analysis were achieved with AlphaView software (Version 3.4.0, 1993–2011, Proteinsimple, Santa Clara, CA, USA).
Data were analyzed by 2-way ANOVA, with chicken line (LWE, HWE) and
environmental temperature (TN, HS) as main factors. Means were compared using
Tukey’s multiple range test when significant main effects were detected. When the
line by environment interaction was not significant, the significant main effects
were analyzed separately with Student’s t-test. Significance was
considered at p
Body weight of the 4 groups is presented in Table 2. There were no significant differences in the body weights of the sampled birds in the 4 groups.
| LWE | HWE | p-value | ||||
| TN | HS | TN | HS | L | E | LxE |
| 3433 |
3476 |
3354 |
3227 |
0.1228 | 0.6883 | 0.4129 |
Data represent means
Gene expression of heat shock proteins (HSP) were differentially regulated in
the blood of heat-stressed LWE and HWE chickens. HSP27 (Fig. 2A,B) and HSP70
(Fig. 2C,D) were significantly increased by HS (p
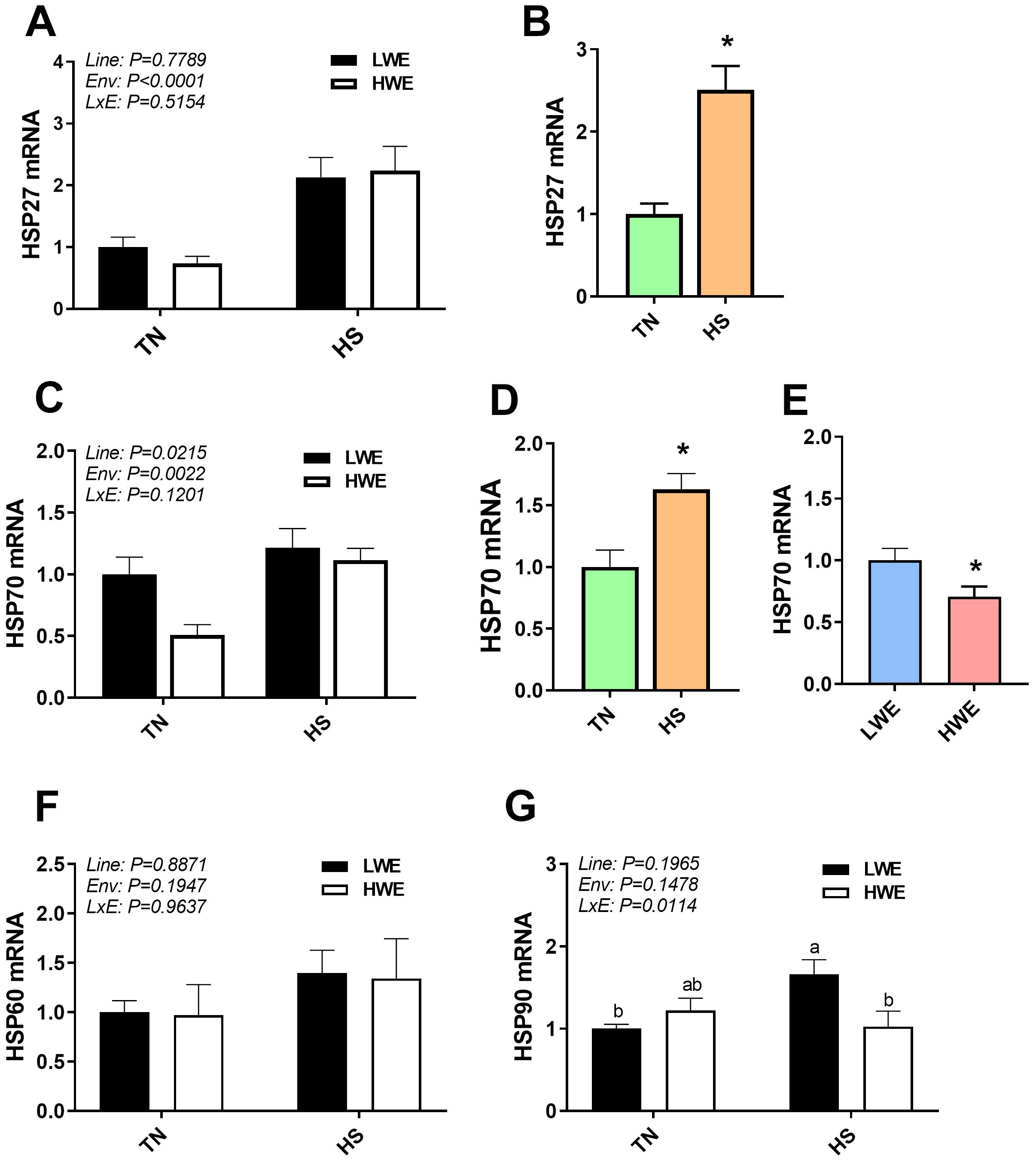 Fig. 2.
Fig. 2.
Blood heat shock protein gene expression in LWE and HWE Chicken
Lines during Acute HS. HSP27 (A,B), HSP70 (C,D,E), HSP60 (F), and HSP90 (G).
When the Line by Environment interaction was not significant, main effects were
analyzed individually by Student’s t-test. Data represent means
Overall, there was a significant effect of HS on pro-inflammatory cytokines gene
expression, where IL-6, IL-18, c-reactive protein (CRP), and tumor necrosis
factor-
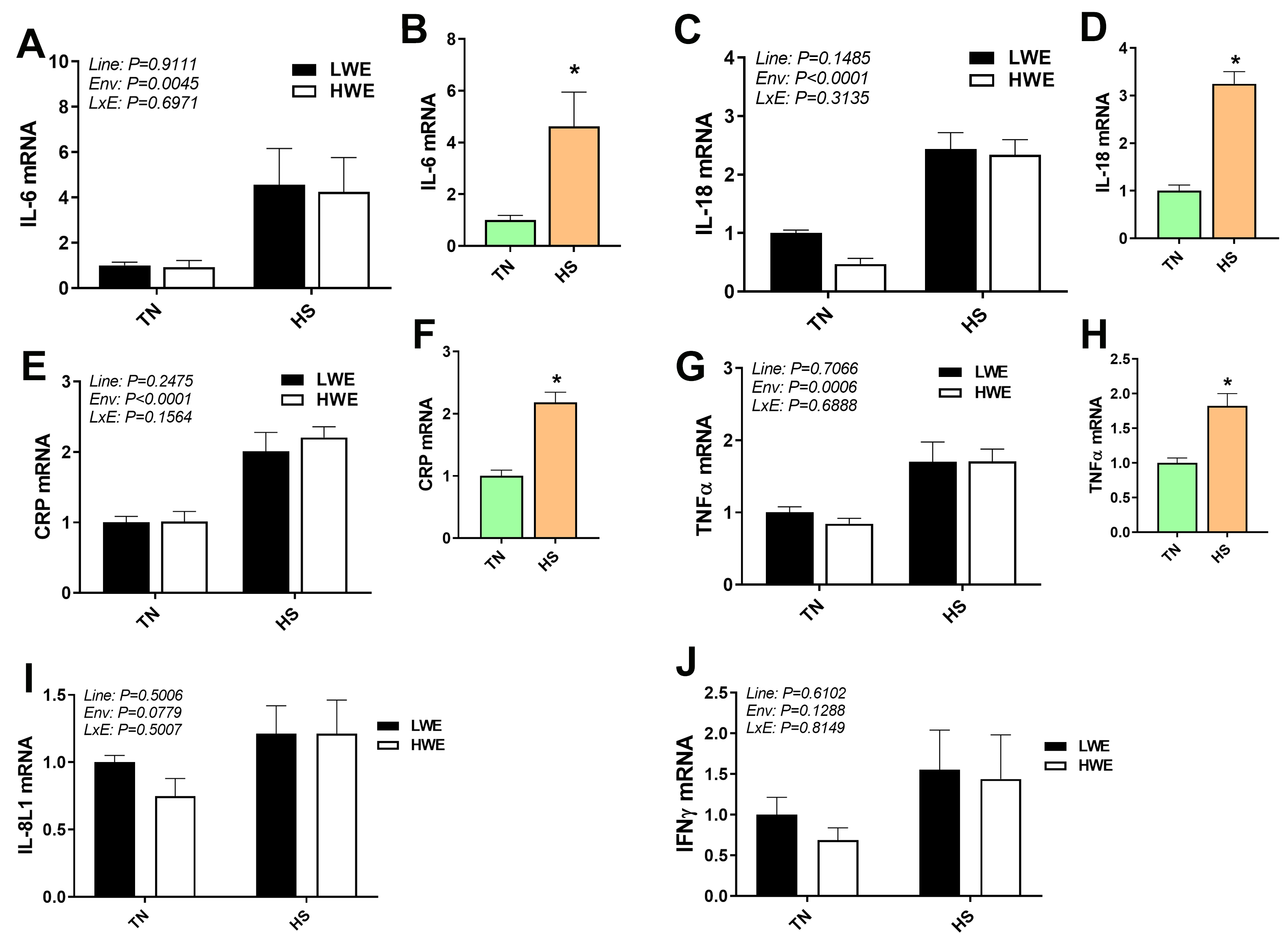 Fig. 3.
Fig. 3.
Pro-inflammatory marker gene expression in blood of LWE and HWE
chicken lines during acute HS. The expression of IL-6 (A, B), IL-18 (C, D), CRP (E, F), TNFa (G, H), IL-8L1 (I), and INFg (J) was determined by qPCR. When the Line by Environment interaction was not
significant, main effects were analyzed individually by Student’s
t-test. Data represent means
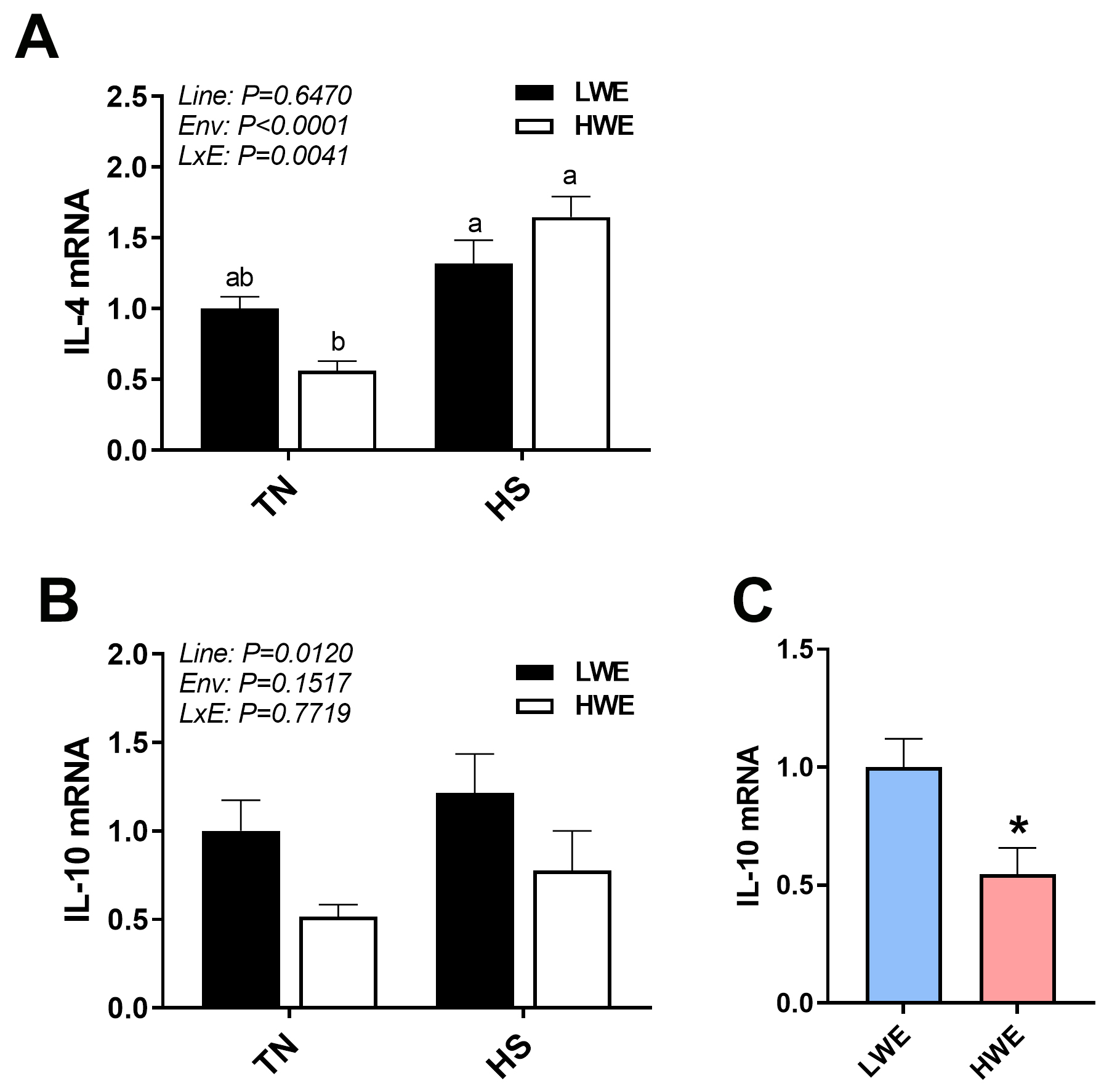 Fig. 4.
Fig. 4.
Anti-inflammatory marker gene expression in blood of LWE and HWE
chicken lines during acute HS. IL4 (A) and IL-10 (B,C). When the Line by
Environment interaction was not significant, the main effects were analyzed
individually by Student’s t-test. Data represent means
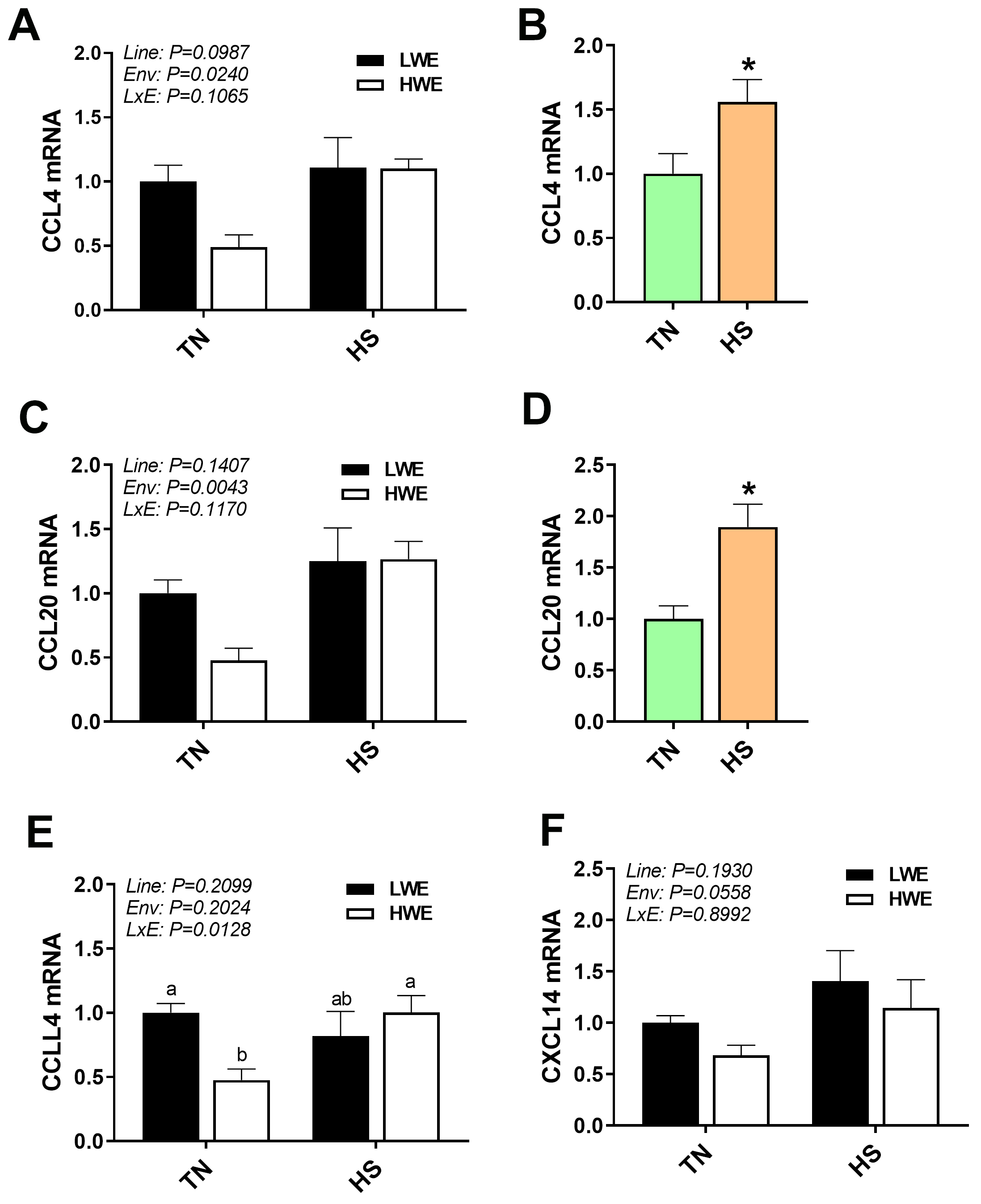 Fig. 5.
Fig. 5.
Chemokine gene expression in blood of LWE and HWE chicken lines
during acute HS. CCL4 (A,B), CCL20 (C,D), CCLL4 (E), and CXCL14 (F). When the
Line by Environment interaction was not significant, main effects were analyzed
individually by Student’s t-test. Data represent means
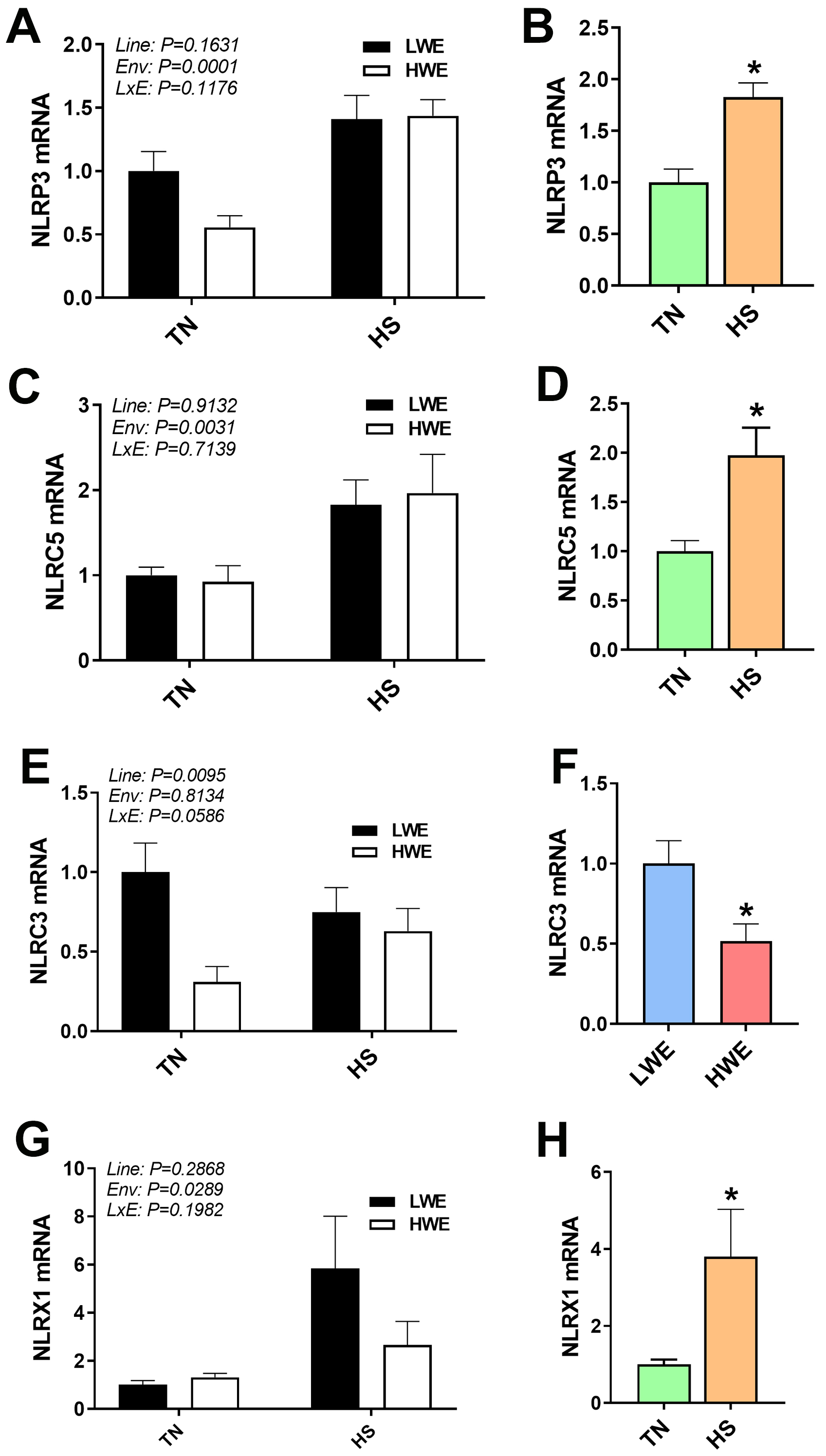 Fig. 6.
Fig. 6.
Inflammasome gene expression in blood of LWE and HWE chicken
lines during acute HS. NLRP3 (A,B), NLRC (C,D), NLRC3 (E,F), and NLRX1 (G,H).
When the Line by Environment interaction was not significant, main effects were
analyzed individually by Student’s t-test. Data represent means
In plasma, a significant interaction between line and environmental temperature was evident for circulating levels of caronte (p = 0.0013, Fig. 7A), pentraxin-3 (PTX3, p = 0.0039, Fig. 7B), and C-C motif ligand 5 (CCL5, p = 0.0490, Fig. 7C), but not for plasma netrin levels (Fig. 7D,E). Plasma caronte and PTX3 showed a similar pattern, where relative levels were lower in HWE birds under HS as compared to TN conditions, though no effects were seen in the LWE line. The chemokine CCL5, however, was increased by HS in the LWE line, with no effects in the HWE birds (Fig. 7C).
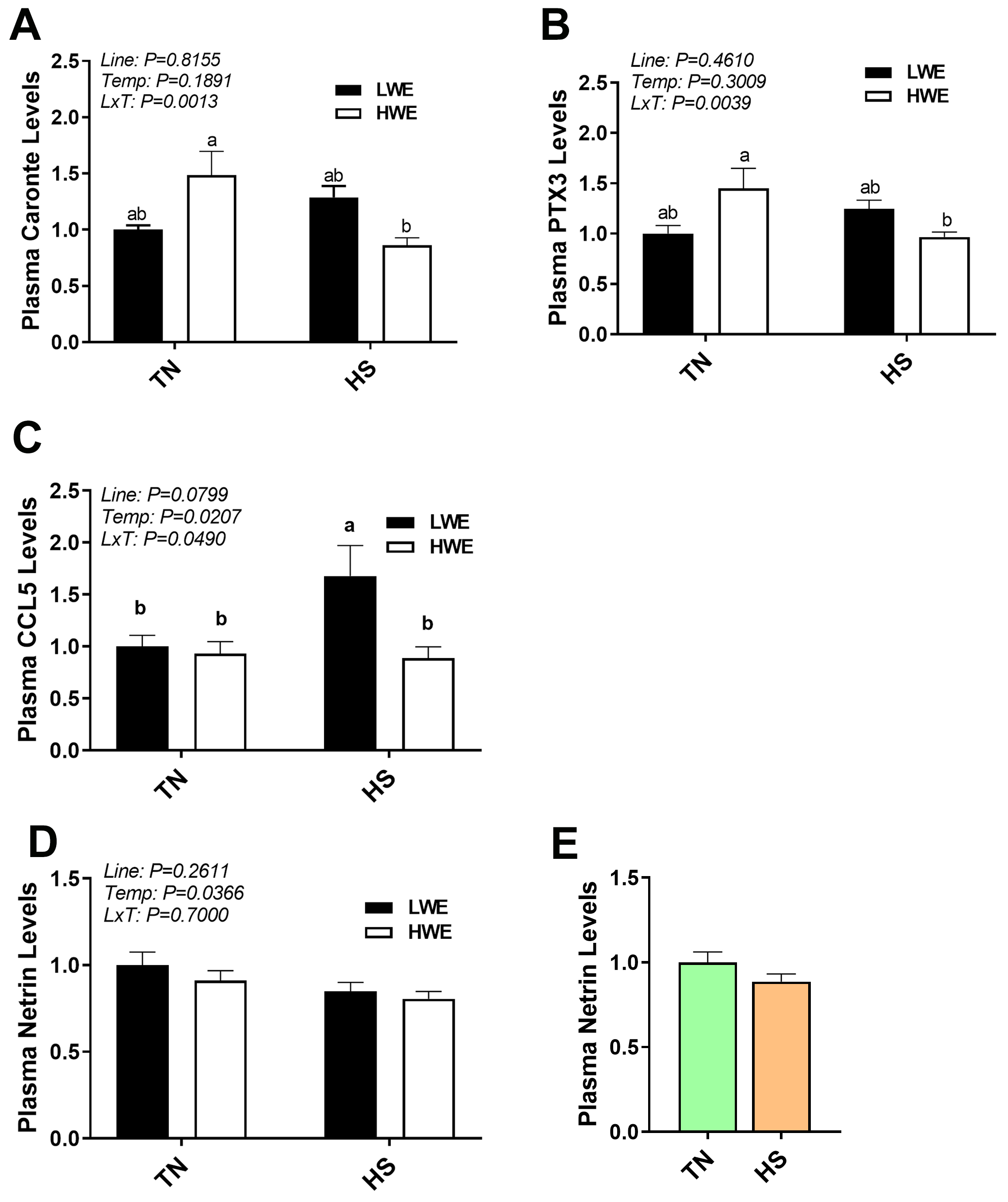 Fig. 7.
Fig. 7.
Plasma caronte, PTX3, CCL5, and netrin levels in LWE and HWE
chicken lines during acute HS. Caronte (A), PTX3 (B), CCL5 (C), and Netrin
(D,E). Data represent means
For the (anti)pro-inflammatory cytokines, a significant interaction between line
and environmental temperature was observed for circulating levels of
INF
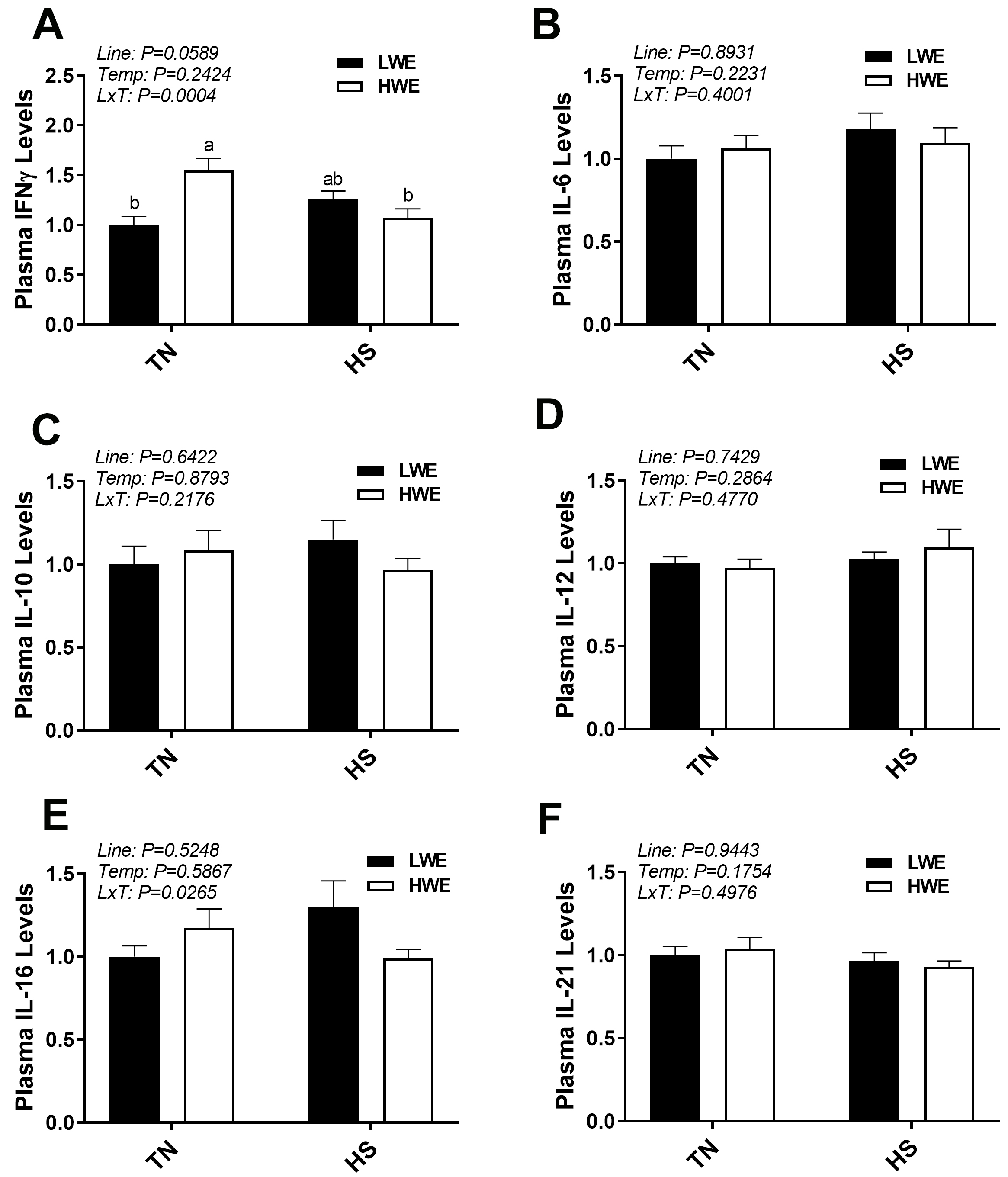 Fig. 8.
Fig. 8.
Plasma cytokine levels in LWE and HWE chicken lines during acute
HS. INF
Gene expression of AVP in blood was significantly different between the lines,
with lower expression in the HWE birds (p = 0.0044, Fig. 9A,B).
There was no effect of temperature on AVP mRNA. However, AVP protein in the
plasma was not significantly affected (p
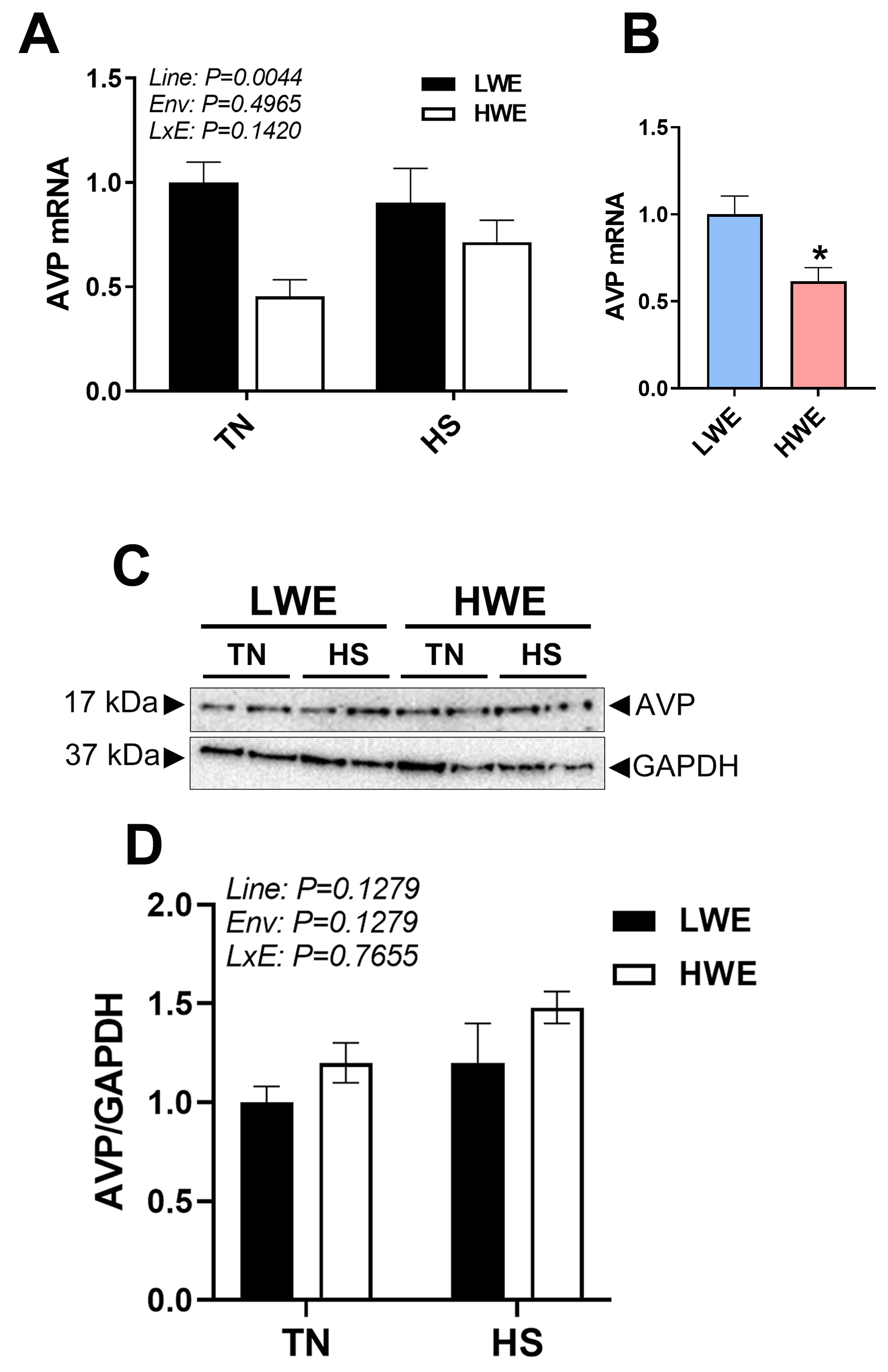 Fig. 9.
Fig. 9.
AVP gene Expression and plasma protein levels in LWE and HWE
chicken lines during acute HS. Relative gene (A) and protein (B) expression of
AVP. Representative western blot (C) and quantification (D) of AVP and GAPDH.
Data represent means
Water availability is already a concern for roughly a quarter of the world’s cities, which experience perennial water shortages [20]. As climate change and global warming will likely lead to more frequent and intense heat waves [21], the need for water, which is already limited, will increase. The poultry industry is particularly susceptible to HS [22], as modern chickens have high metabolic activity, increased body heat production, and decreased thermotolerance [6]. Therefore, the combined impact of these environmental challenges must be addressed in order to not only maintain, but improve productivity of a critical, efficient, and affordable protein source for a growing world population.
It is imperative to understand potential associated changes, whether positive or negative, that can arise during the selection process in broilers. Indeed, intensive genetic selection for improved growth rate and feed efficiency for more than 80 years has given rise to unintended negative traits that are now concerns for the industry [23]. Here, overall, we show that the impact of 4 generations of selection for improved water efficiency had minimal impact on circulating inflammatory markers in acute heat-stressed broilers. As we measured gene expression in whole blood, it encompasses a heterogeneous mix of cell types, including white blood cells, the traditional players in the immune and inflammatory response, as well as red blood cells. This is of importance, as in chickens, red blood cells are nucleated, and therefore are transcriptionally active and have protein synthesis abilities, and likely substiantially impact the immune response, through both sending and receiving cellular signals [24]. Additionaly, obtaining a blood sample is minimally invasive, and can allow for monitoring of health status or response to specific stressors (such as HS) within a breeding program.
Some of the spotted effects of HS might be associated with dysregulation of protein homeostasis-induced by protein misfolding and aggregation. To counter these detrimental effects, the cell activates the survival pathways through systematization of highly conserved responses. Contingent on the stress type, severity, intensity, amplitude, and duration, cells can activate a rapid and efficient stress response and protein quality control systems (refolding, degradation, etc.) to ensure their survival or start off the cell-death pathway [25]. At the cellular and molecular levels, a well known standard rapid response to HS is induced synthesis of heat shock proteins (HSPs). Based on their known functions, elevated HSP expression due to acute HS was not unexpected, and has been previously shown in multiple tissues in poultry [11, 26, 27]. The differences seen in HSP70 gene expression between the lines, however, are more intriguing. Lower blood HSP70 expression in the HWE line, regardless of environmental temperature, may indicate an improved state of intracellular homeostasis in these birds as compared to the LWE line, as HSP70 is a highly inducible molecular chaperone that catalyzes proper folding of nascent proteins under normal conditions and assist in refolding and clearing damaged proteins under stress conditions [28]. Further studies are needed to define the role and mechanisms of HSP70 in protein homeostasis, cell survival, and thermotolerance in HWE birds.
Unsurprisingly, HS increased gene expression of the majority of cytokines and
chemokines as has been previously reported [9, 10, 29], and alteration of the
innate cytokine and chemokine response by stressors is well noted [29, 30]. The
fact that the induction of these factors was to a similar extent in both HWE and
LWE lines indicates that there were neither detriments nor improvements to this
response associated with selection for water efficiency. However, IL-10, IL-4,
and CCL4 were differentially expressed between the two lines. IL-10 is increased
in circulation in response to inflammatory challenges, such as viruses, bacteria,
fungi, protozoa and helminths [31]. Studies have shown it to exhibit
immunosuppressive properties, through inhibition of cytokines such as
IFN-
The inflammasomes are a group of intracellular multimeric protein complexes that
influence inflammation and the protective immune response [38]. Multiple members
of the NLR family exist, with differences in stimuli and activated pathways, and
both inflammatory and anti-inflammatory properties [39, 40]. Here, the mRNA
abundances of the NLRP3, NLRC5, and NLRX1 inflammasomes were all upregulated in
circulation by HS, without any differences between the water efficiency broiler
lines. NLRP3 is the most well studied inflammasome, and is generally considered
pro-inflammatory. Although NLRC5 and NLRX1 are less well understood, NLRC5 has
been shown to have inflammatory properties via association with NLRP3 [41] and
through IFN production, but may also be able to suppress IFN and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-
The brain is a critical regulator and protector of homeostasis via neuroendocrine responses which guard set-points of body water concentration and volume [45]. AVP is a key hormone in fluid balance, as it is released from the posterior pituitary in response to increased plasma osmolarity, and acts to increase water retention at the kidney [46]. There are several studies in human medicine examining the difference between individuals with low and high habitual fluid intake [47]. In both intake groups, plasma osmolarity and sodium levels remain within normal ranges; however, one of the hallmarks is higher levels of circulating AVP in those that consume less [47]. Here, we found no differences in circulating AVP protein, although gene expression was lower in the HWE as compared to the LWE line. Other research has shown AVP expression in peripheral tissues [48, 49], therefore it is plausible that AVP produced by circulating cells may have autocrine effects separate from, or in addition to, centrally produced AVP. Studying these cells in isolation could help define the role of this locally produced AVP. Overall, our data suggests that although HWE birds are drinking less water, their blood inflammasome and chemocytokine expression profile seemed to not be altered. It is plausible that the HWE birds have more efficient metabolic water production and water (re)absorption in the gut, allowing overall homeostasis to be maintained in the body. Further research is warranted to examine other physiological, cellular and molecular pathways in the HWE birds with more selection generations to make sure that there are no undesirable changes occur.
In conclusion, water scarcity is a global concern that must be addressed at all levels of water use, but modifications in agricultural systems must also maintain optimal production levels. Herein, to the best of our knowledge, we are the first to report that broilers selected for high or low water efficiency displayed an overall similar expression pattern of circulating inflammatory markers in response to acute HS. This likely indicates that selection for improved water efficiency has not negatively impacted inflammation status in these birds.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
SD designed the research study. ESG, TT, WGB, SO, and SD performed the research. ESG analyzed the data and wrote the manuscript. SD edited the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was conducted in accordance with the National Institutes of Health recommendations guide for laboratory animal use and care. All the procedures in this study were approved by the University of Arkansas Animal Care and Use Committee under protocol #23015.
Authors would like to thank the University of Arkansas Poultry Research farm and the University of Arkansas feed mill for their assistance.
This study was supported by a grant from USDA NIFA Sustainable Agriculture Systems (#2019 69012-29905) to S.D.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









