1 Laboratory of Influenza Research, College of Veterinary Medicine, Chungnam National University, 34134 Yuseong Gu, Daejeon, Republic of Korea
2 Institute of Influenza Virus, Chungnam National University, Yuseong Gu, 34134 Daejeon, Republic of Korea
†These authors contributed equally.
Academic Editor: Graham Pawelec
Abstract
Background: Over the last 20 years, circulating highly pathogenic (HP) Asian H5 subtype avian influenza viruses have caused global pandemics in poultry and sporadic infections in humans. Vaccines are a desirable solution to prevent viral infections in poultry and reduce transmission to humans. Herein, we investigated the efficacy of an oil-adjuvanted inactivated H5N6 vaccine against highly pathogenic H5N6 and H5N1 influenza virus infections in chickens. Methods: The polybasic amino acid cleavage site depleted HA gene and NA gene of A/Waterfowl/Korea/S57/2016 (clade 2.3.4.4) (H5N6) was assembled with the rest of the A/PR/8/34 (H1N1) genes to construct the vaccine virus. The vaccine virus was propagated in fertilized eggs, partially purified using a tangential flow filtration (TFF) system, and inactivated using formalin. The chickens were intramuscularly immunized with 384 HA, 192HA, and 96HA units of oil-adjuvanted inactivated H5N6 vaccine. Antibody titer, survival rate, and lung pathology were evaluated against the homologous H5N6: A/waterfowl/Korea/S57/2016 (clade 2.3.4.4) and heterologous H5N1: A/Hong Kong/213/2003 (clade 1) viruses 12 and 4 weeks post-vaccination (p.v.), respectively. Data were statistically analyzed using the Mann-Whitney U test. Results: The 384HA (n = 10) and 192HA (n = 5) antigen-immunized chickens showed 100% survival after lethal infections with homologous H5N6, and no virus shedding was observed from tracheal and cloacal routes. All chickens that received the 384HA vaccine survived the challenge of heterologous H5N1 after 4 weeks of immunization. The chickens that received the 384HA vaccine showed mean HI titers of 60 and 240 after 12 and 4 weeks of vaccination, respectively, against HP H5N6, whereas a mean HI titer of 80 was observed in sera collected 4 weeks after vaccination against HP H5N1. Conclusions: Our findings indicate that one dose of 384HA oil-adjuvanted inactivated H5N6 vaccine can induce a long-lasting immune response against both homologous H5N6 and heterologous H5N1 infections in chickens.
Keywords
- inactivated vaccine
- highly pathogenic influenza virus
- H5N6
- cross-clade immunity
Avian influenza viruses (AIVs) are major viral pathogens that cause sporadic outbreaks in poultry. Aquatic birds, including wild waterfowl, have been identified as the primary reservoirs of influenza A viruses. However, these virus infections are also transmitted to other avian hosts and mammals, including humans [1]. All AIVs are categorized as influenza type A viruses and belong to the family Orthomyxoviridae. The avian influenza virion has eight segments of negative-sense, single-stranded RNA [2, 3]. The cleavage site of hemagglutinin (HA) is a critical determinant of influenza virus virulence. AIV Subtypes H5 and H7 underwent intermittent mutations in HA, acquiring a polybasic amino acid moiety containing arginine (R) and lysine (K) in the HA cleavage site. As a result, low-pathogenic AIVs were converted into highly pathogenic AIVs in poultry [4, 5, 6].
In 1996, H5N1 HP infection was detected in a domestic goose in Guangdong China (Gs/Gd lineage), and it later developed into a panzootic disease, causing drastic death rates among wild birds, poultry, and humans, and invading four continents [7, 8]. Since 2008, the evolution of the HA backbone of the H5N1 Gs/Gd lineage and the neuraminidase (NA) gene of low-pathogenic AIVs has led to the development of novel H5 HP avian influenza viruses. In early 2014, widespread infection by the novel HP avian influenza H5N6 subclade 2.3.4.4 was seen in China, Laos PDR, and Vietnam [9] and caused high death rates in poultry. Moreover, it had higher virulence than the previously reported H5N8 subtype, and the H5N6 virus crossed from poultry to humans, causing 17 human infections and seven deaths in China in mid-2017 [10]. In late 2016, HP H5N6 was first reported in migratory avian and subsequently spread to the domestic poultry population, causing a devastating outbreak in South Korea [11, 12].
Highly pathogenic H5N1 avian influenza viruses are salient viral pathogens that cause recurrent epidemics in poultry and mammals, including humans. Since 2003, there have been several reports of HP H5N1 infections in humans with a mortality rate of over 50% [13], which increases the risk of recurring pandemics. The continuous presence of HP H5N6 and H5N1 viruses in bird reservoirs poses a serious threat to the public. Therefore, the establishment of effective protective measures is imperative for a rapid response to the emergence of pandemic strains.
A majority of epizootic diseases caused by influenza viruses have been restrained through stamping-out programs. However, vaccines are currently considered a surrogate solution for the protection of poultry from HP influenza virus infections and the prevention of transmission of the virus to humans. Numerous attempts have been made to develop vaccines against the HP H5 subtype viruses for use in poultry, and their cross-reactivity has also been evaluated against the H5N6 influenza virus [14, 15].
The effectiveness of the vaccine against H5 HP influenza virus infections seems to depend on the high homology between HA and NA antigenic factors. Therefore, recombinant vaccines have been developed through engineering HA and/or NA antigens from HP influenza viruses with the primary influenza virus PR8–H1N1. Depletion of the polybasic amino acid motifs of HA in highly pathogenic influenza viruses is one of the strategies practiced on a laboratory scale and has shown a remarkable immune response in the short-term [14, 16]. However, a vaccine that is effective against both HP H5N6 and HP H5N1 is lacking.
Therefore, we developed an inactivated vaccine with an oil adjuvant by depleting the polybasic amino acid cleavage site of the HP H5N6 influenza virus. We evaluated the efficacy and long-term immune response triggered by the vaccine against homologous HP H5N6 virus infection and the effectiveness of the vaccine against heterologous HP H5N1 virus infection.
Healthy fertilized White Leghorn eggs were purchased from a local farm in Korea and hatched in our laboratory. Chickens aged 4 weeks were used in the immunization experiments. Immunized chickens (384HA, 192HA and 96HA) were housed separately in cages arranged in the animal center of Chungnam National University. The chickens were moved to a Biosafety Level-3 (BSL-3) poultry isolator to perform the challenge study with HP H5N6 and HP H5N1 viruses, and each group of infected chickens was housed separately in a poultry isolator during the study period.
HP H5N6; A/Waterfowl/Korea/S57/2016 (clade 2.3.4.4) was isolated in our laboratory from the feces of wild birds and was used as a seed strain for vaccine development and homologous virus challenge studies. Dr. Malik Peiris of the University of Hong Kong kindly provided the HP H5N1 A/Hong Kong/213/2003 (clade 1) virus. Virus strains were propagated in the allantoic fluid of 10-day-old embryonated chicken eggs at 37 °C for 72 h.
All experiments related to highly pathogenic influenza viruses were conducted in BSL-3 facility certified by the Korean government.
A reassorted virus was generated using a plasmid-based reverse genetic
system, as described by Hwang et al. [16]. Briefly, RNA was extracted
from A/Waterfowl/Korea/s57/2016 (H5N6) cells using an RNeasy mini kit (Qiagen,
Germany). The gene sequence of HA, which encodes a polybasic amino acid cleavage
site, was deleted using overlapping primer PCR. The polybasic amino acid-depleted
HA of H5N6 and NA of H5N6 were cloned into the pHW 2000 vector. PB2, PB1, PA, NP,
M, and NS plasmids of A/Puerto Rico/8/34 (PR8) (H1N1) were kindly provided by Dr.
Robert Webster of St. Jude Children’s Research Hospital, Memphis, TN, USA. One
microgram of each plasmid was introduced into Vero cells at 80% confluency in
6-well cell culture plates, and then the Vero cells were transfected for 48 h.
The cell supernatant (300
The H5N6 vaccine virus was propagated in 10-day-old embryonated eggs (37 °C). The
allantoic fluid was harvested 72 h post-inoculation and centrifuged at 14,981
Complete inactivation of the vaccine virus was confirmed by inoculating the vaccine virus into the allantoic fluid of 10-day-old embryonated eggs, followed by the hemagglutination (HA) assay using 0.5% Turkish erythrocytes.
The inactivated vaccine virus (antigen) titer was determined using an HA assay
with 0.5% turkey blood. Antigen doses were selected based on the HA units. The
384HA, 192HA, and 96HA units of antigen-containing solutions were prepared using
PBS (pH = 7.40). The oil-adjuvanted inactivated H5N6 vaccine was formulated by
mixing 30% antigens diluted in PBS and 70% oil adjuvant (Montanide ISA 70 VG,
SEPPIC Co., Puteaux, France). Three groups of 4-week-old chickens (n = 10) were immunized
intramuscularly (i.m.) using 500
To determine the long-term efficacy of an immune response elicited by the
vaccine against homologous virus infections, each group of immunized chickens
(384HA (n = 10), 192HA (n = 5), and 96HA (n = 5)) was intranasally (i.n.)
challenged using 1 mL of 100 TCID
To further examine the effectiveness of the 96HA vaccine, we infected five
chickens that had received two doses of the 96HA H5N6 vaccine with 1 mL of 100
TCID50/mL of HP H5N6 virus after 12 weeks of vaccination. A separate group of
unimmunized chickens (n = 3) i.n. infected with1 mL of TCID
Blood was collected from chickens in each vaccinated group at 2-week intervals
two weeks after immunization. Sera were extracted from each blood sample and
heat-inactivated at 56 °C for 30 min. An HI assay was performed to determine the
antibody titer in chickens against homologous HP H5N6 and heterologous HP H5N1
virus infections. Briefly, sera were diluted 10-fold before performing 2-fold
serial dilutions in PBS (pH 7.40) in V-bottom 96-well plates. Eight
hemagglutination (HA) units (25
The trachea and cloacae of surviving chickens were swabbed at 1, 3, 5, and 7
days post infection (p.i.). Tracheal and cloacal swab samples were collected in
250
Viral RNA was extracted from tracheal and cloacal swab samples of immunized and
unimmunized chickens challenged with HP H5N6 or HP H5N1, which were collected on
day 3 p.i. using a HiGene
To detect the virus, real-time quantitative fluorescent PCR was performed using
the TOPreal
To determine tissue histopathology, on day 6 p.i., lung and tracheal tissues
were collected from immunized chickens challenged with HP H5N6 or HP H5N1
viruses. Tissues from HP H5N6 infected unimmunized chickens and HP H5N1 infected
unimmunized chickens were obtained on days 1 and 3 p.i., respectively. The
tissues were immersed in phosphate-buffered formalin (10%) (Triangle Biomedical
Sciences, Durham, USA) and fixed for 6 h while shaking. The tissues were then
washed under running tap water and embedded in paraffin. Tissues were prepared
into 5-
Experimental data were analyzed using the Wilcoxon signed-rank test and multiple
t-tests in GraphPad Prism software (Version 8.0.1) (GraphPad software.
Inc., San Diego, CA, USA). Statistical p-values less than 0.05 (p
Chickens immunized with a single dose of the 384HA, 192HA, and 96HA vaccines and two doses of the 96HA vaccine were challenged with homologous HP H5N6 at 12 weeks p.v. Survival of chickens was observed up to 14 days p.i. The 384HA- or 192HA-immunized chickens exhibited 100% survival rate, while survival of the 96HA vaccinated chickens dropped to 20% on day 6 p.i. and remained constant until day 14 p.i. Chickens that received two doses of the 96HA vaccine also showed 100% survival up to 14 days p.i. When unimmunized chickens were infected with the homologous HP H5N6 virus, all chickens (n = 5) died within two days of infection (Fig. 1A).
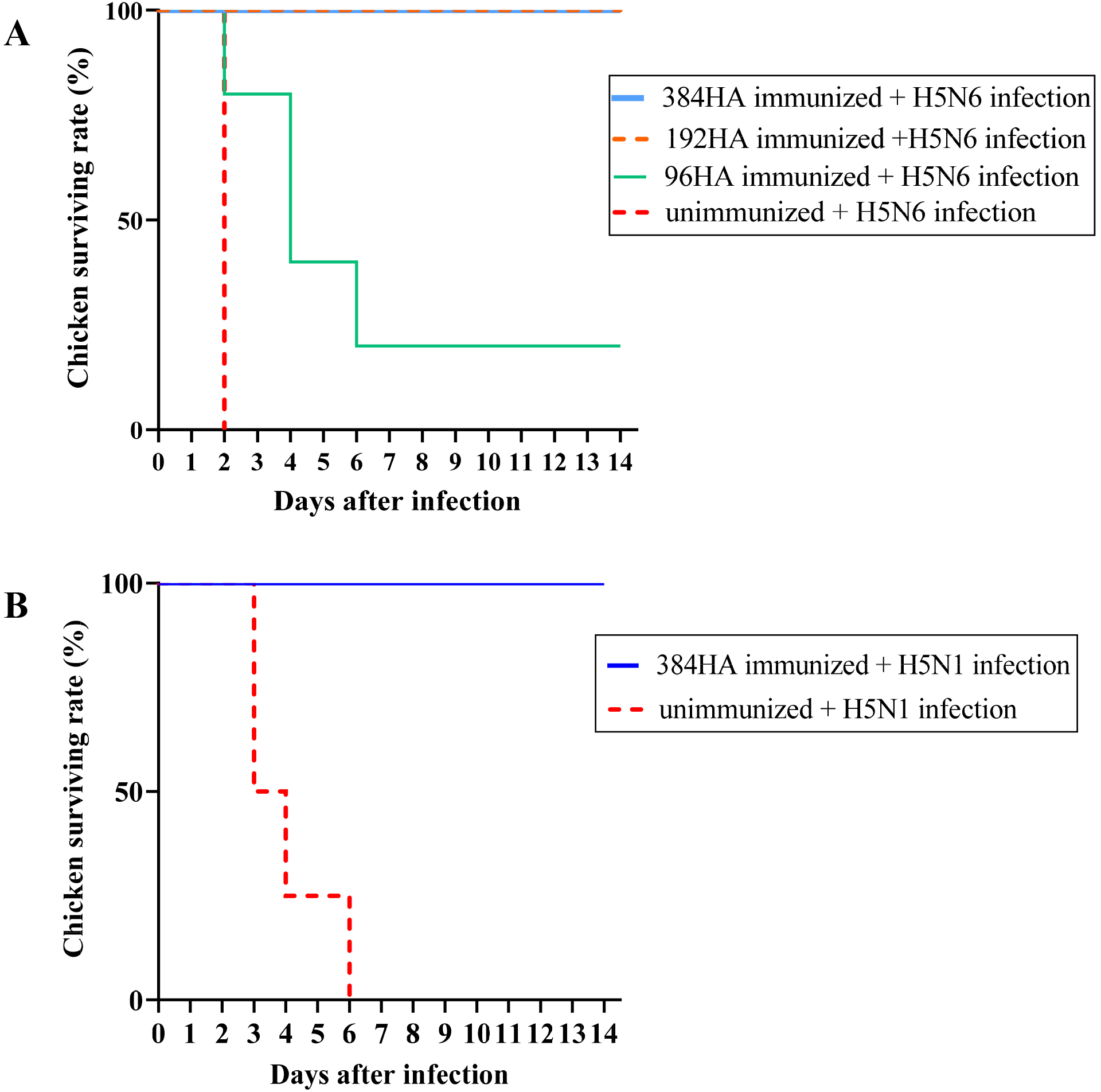 Fig. 1.
Fig. 1.Survival Rate of immunized chickens after the challenge.
Immunized chickens were intranasally infected with 100 TCID
Interestingly, all chickens immunized with the 384HA oil-adjuvanted inactivated vaccine and infected with heterologous HP H5N1 virus (n = 4) four weeks p.v. survived throughout the 14 days of observation. All unimmunized chickens infected with heterologous HP H5N1 (n = 4) survived until day 6 p.i. (Fig. 1B).
Sera were extracted from blood collected from the immunized chickens starting at 2 weeks p.v. up to 12 weeks p.v. The HI assay was performed to determine antibody titers in immunized chickens. The mean hemagglutination inhibition (HI) titer was 240 against homologous HP H5N6 and 80 against heterologous HP H5N1 influenza virus (clade 1) 4 weeks p.v. in 384HA-vaccinated chickens (Fig. 2A,C).
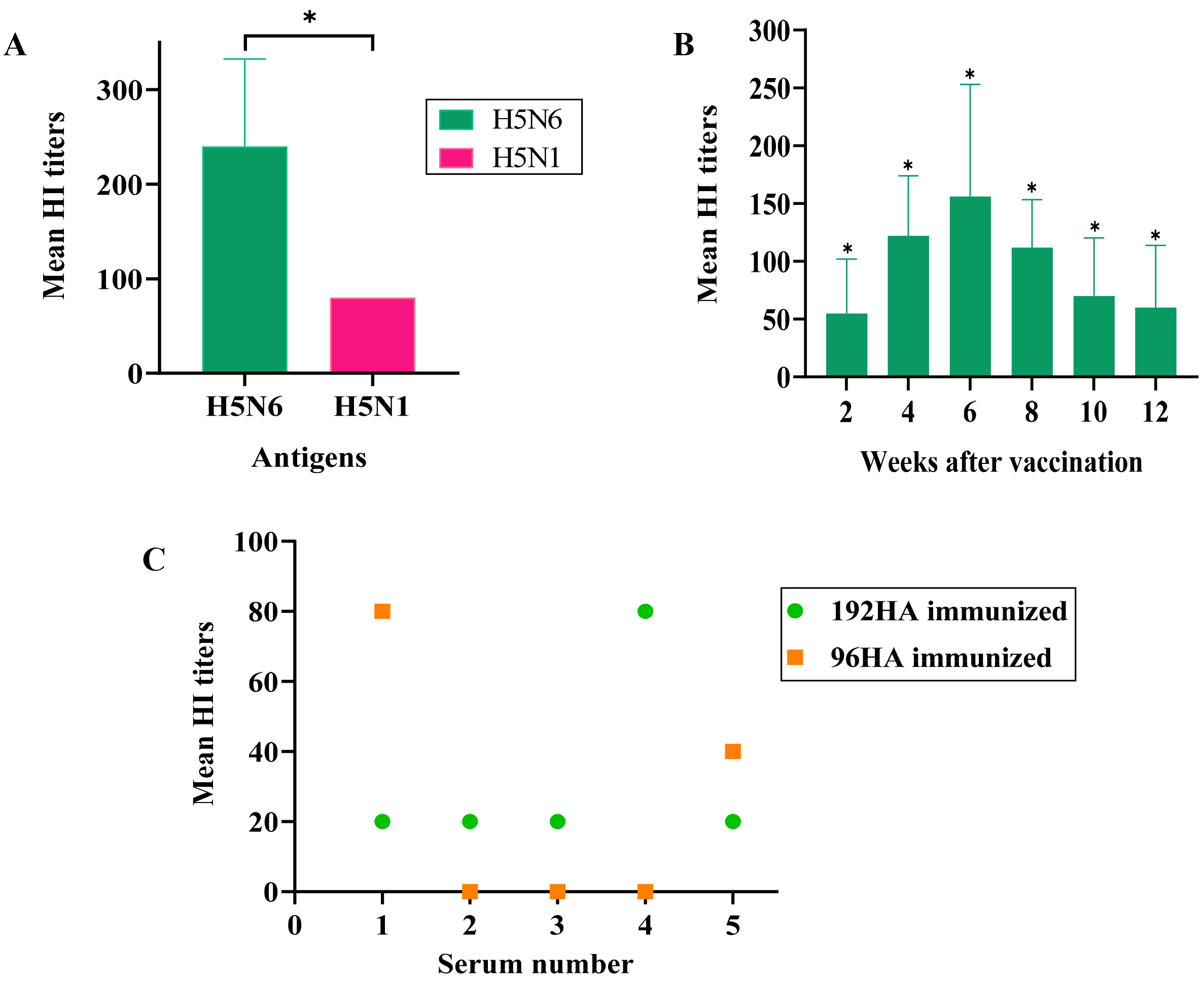 Fig. 2.
Fig. 2.Antibody titers in immunized chickens. 4-week-old chickens were
injected with 500
The mean HI titer peaked 6 weeks after vaccination and was reported to be 156 against homologous HP H5N6 infection. The vaccinated chickens showed a mean HI titer of 60 against A/waterfowl/Korea/s57/2016 (H5N6) at 12 weeks p.v., which was significantly higher than the HI titers in unimmunized chickens (Fig. 2B).
To determine the viral titer, the trachea and cloacae of surviving chickens were
swabbed on day 3 p.i. No virus was detected in the tracheal and cloacal samples
of 384HA-,192HA-, and 96HA-vaccinated chickens that were challenged with the
homologous H5N6 virus (Fig. 3A). Cloacal and tracheal swabs from
chickens vaccinated with two doses of the 96HA vaccine and challenged with HP
H5N6 virus did not show any viral titers. However, unimmunized chickens exposed
to HP H5N6 showed a significantly high viral titer of (411.9 PFU/mL) in the
trachea on day 1 p.i. and no viruses were shed through the cloacae 1 day p.i.
(Supplementary Fig. 1). We could not measure the viral titer of
unimmunized chickens in subsequent days since they died 2 days p.i.
Interestingly, no virus was detected in tracheal and cloacal swabs when the 384HA
vaccine was administered to chickens infected with HP H5N1, while HP H5N1
infected unimmunized chickens showed significantly higher viral titers in the
cloacal (Log
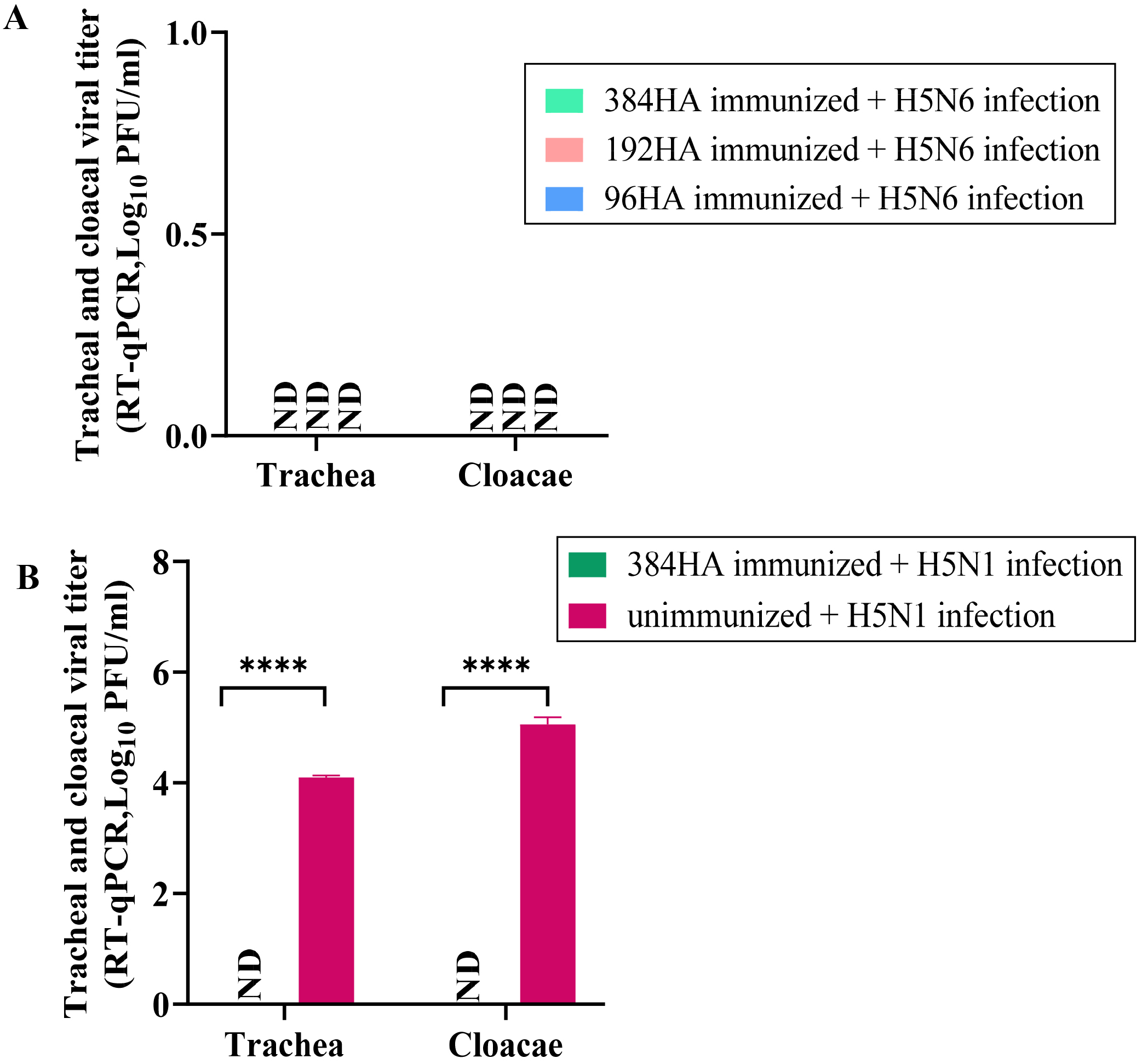 Fig. 3.
Fig. 3.Viral titers in tracheal and cloacal swabs of vaccinated
chickens after challenge with HP H5N6 or HP H5N1 viruses. Tracheal and cloacal
swabs were collected in PBS on day 3 p.i. from chickens that received the vaccine
and were challenged with HP H5N6 A/Waterfowl/Korea/S57/2016 (clade 2.3.4.4) and
HP H5N1 A/Hong Kong/213/2003 (clade 1) viruses. RNA was extracted from the swab
samples, and RT qPCR was performed using H5 influenza primers and probes. (A)
Viral titers of immunized chickens challenged with HP H5N6 (clade 2.3.4.4)
(384HA; n = 10, 192HA; n = 5, and 96HA; n = 4). (B) Viral titers in immunized
chickens challenged with HP H5N1 (clade 1) (n = 4). Unimmunized chickens infected
with HP H5N6 homologous virus (n = 5) and unimmunized chickens infected with HP
H5N1 heterologous virus (n = 4) were used as controls. Data are represented as
Log10 plaque forming units (PFU)/mL of viral titers in vaccinated chickens
None of the 384HA-immunized chickens that were infected with homologous HP H5N6 virus exhibited signs of pneumonia in the lung tissue (Fig. 4A). However, lung tissues of 192HA- and 96HA-vaccinated chickens that were challenged with homologous HP H5N6 virus showed symptoms of mild pneumonia (Fig. 4B,C) when compared with the lung tissues of only HP H5N6 infected chickens, which showed signs of severe pneumonia (Fig. 4D). When 384HA-immunized chickens were infected with the HP H5N1virus, their lung tissues showed mild pneumonia (Fig. 5A) compared to the lung tissues of HP H5N1-infected, unimmunized chickens, which showed signs of severe pneumonia (Fig. 5B). The lung tissues of unimmunized and non-infected chickens did not show any signs of pneumonia (Figs. 4E,5C).
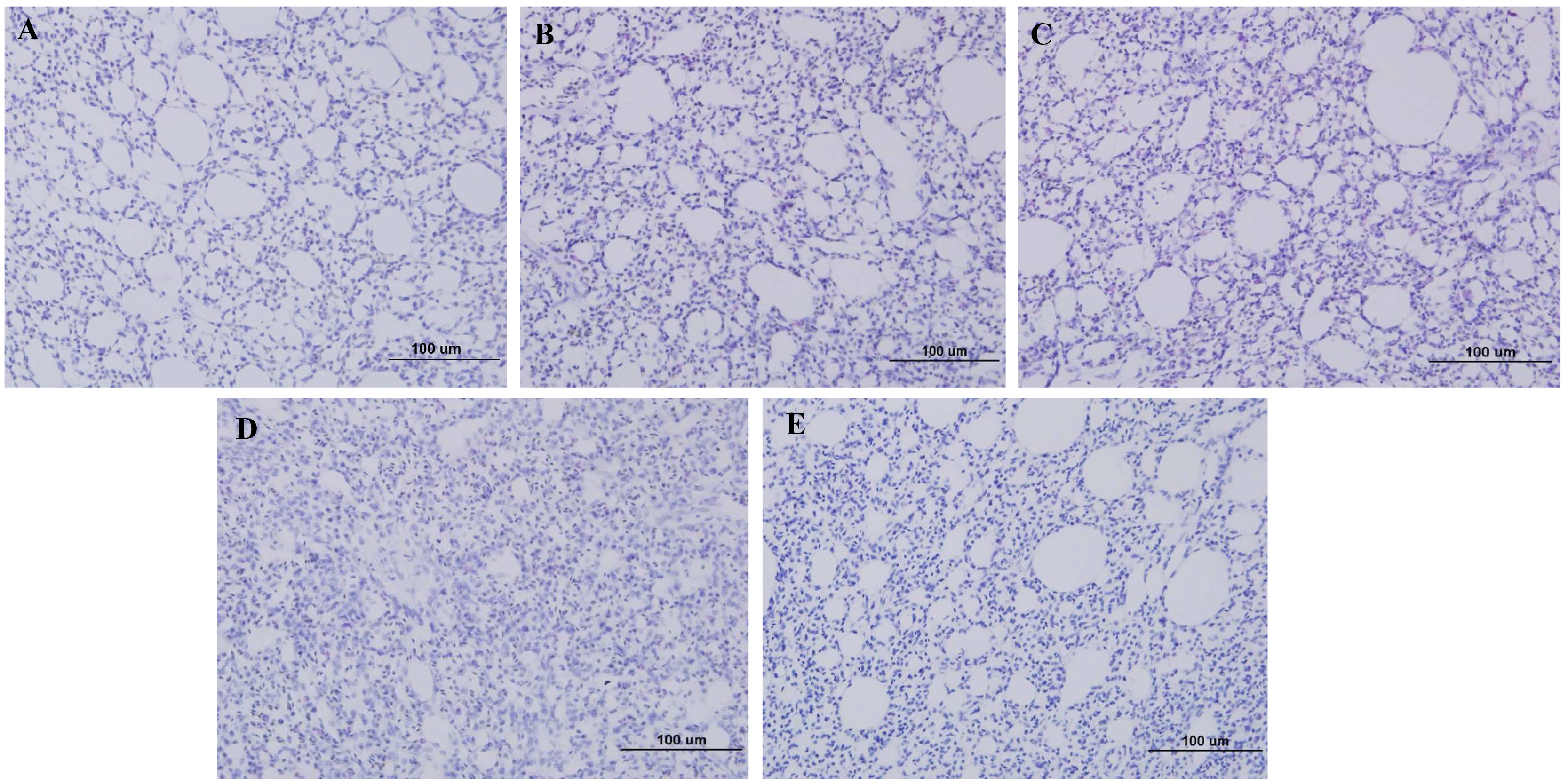 Fig. 4.
Fig. 4.Pathology of lung tissues of immunized chickens challenged with HP H5N6: A/Waterfowl/Korea/S57/2016 (clade2.3.4.4). (A) Lung tissue from 384HA-immunized chicken. (B) Lung tissue from 192HA-immunized chicken. (C) Lung tissue from 96HA-immunized chicken. (D) Lung tissue from HP H5N6-challenged, unimmunized chicken. (E) Uninfected chicken lung tissue.
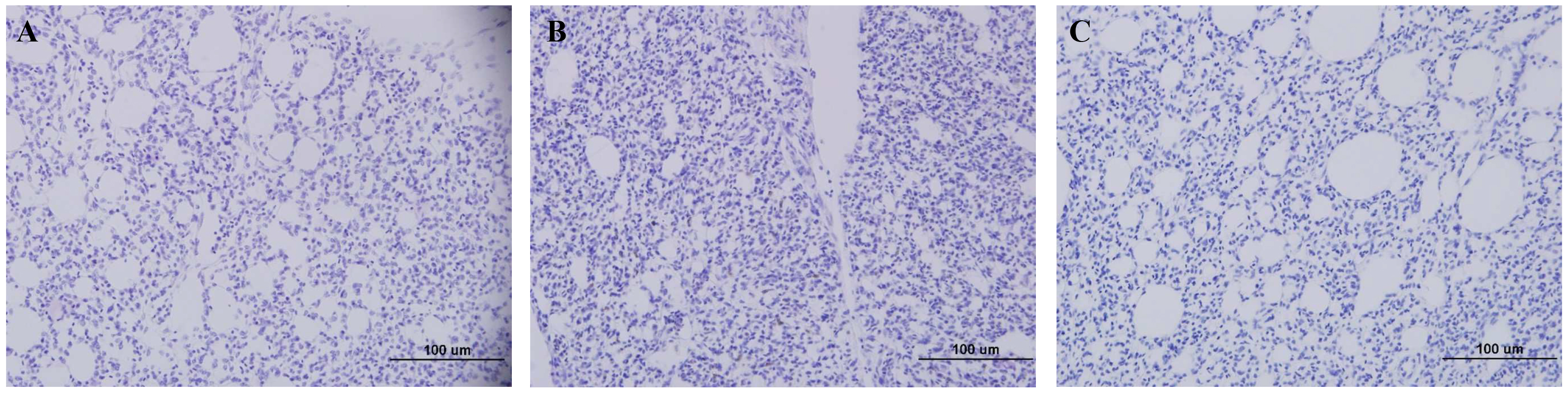 Fig. 5.
Fig. 5.Pathology of lung tissues of immunized chickens challenged with HP H5N1: A/ Hong Kong/213/2003 (clade 1). (A) Lung tissue from 384HA-immunized chicken. (B) Lung tissue from H5N1 infected unimmunized chicken. (C) Uninfected chicken lung tissue.
Over the last 20 years, circulating highly pathogenic H5 subtype avian influenza viruses have caused global outbreaks in poultry populations and sporadic infections in humans. Hence, the development of a vaccine with long-term effectiveness against these virus infections is crucial not only to minimize economic losses in the poultry industry but also to decrease the magnitude of infection by reducing viral shedding into the environment. In this study, we found that an oil-adjuvanted inactivated H5N6 vaccine containing 384HA units could induce sterile immunity in chickens against homologous HP H5N6 A/Waterfowl/Korea/S57/2016 (clade 2.3.4.4) up to 12 weeks after vaccination and cross-clade immunity against A/Hong Kong/213/2003 (H5N1) (clade 1) infection.
Our results demonstrate that a prime boost with 384HA units of oil-adjuvanted
inactivated H5N6 vaccine could completely protect chickens from homologous H5N6
virus, A/Waterfowl/Korea/S57/2016 (clade 2.3.4.4) infection after 12 weeks of
vaccination. Even though inactivated vaccines require several boosters to
activate protective immunity, one dose of the vaccine developed herein could
elicit an immune response in chickens for as long as 12 weeks, with a mean HI
titer of 60. This demonstrates that our vaccine can effectively provide sterile
immunity in chickens, even with prime boosting. Moreover, the H5N6 vaccine
elicited humoral immunity, which was higher than the protective immunity level of
1:40 HI. A previous study on an inactivated vaccine prepared using HA and NA
antigens of H5N1 (A/Vietnam/1203/04 (H5N1) (clade 1) showed a mean HI titer of
147 for homologous viruses 12 weeks p.v. [16]. This difference in mean HI titers
could be due to the level of vaccine purity. The assorted H5N1 vaccine used in
the study by Hwang et al. [16] was completely purified and the HA
protein content was measured in
Our vaccine provided cross-clade immunity against A/Hong Kong/213/2003 (H5N1)
(clade 1) in immunized chickens 4 weeks after vaccination. A previous study also
showed that a vaccine containing H5N1 antigens (clade 1) is effective in
immunizing chickens against heterologous challenges (clade 2.3.4) when vaccinated
with 5.0
Infections by highly pathogenic H5Nx, including H5N6 and H5N1, have been reported to cause severe disease symptoms and 100% mortality within 2–3 days [19, 20, 21, 22]. However, 192HA vaccinated HP H5N6 homologous virus-infected and 384HA vaccinated HP H5N1 heterologous virus-infected chickens showed a 100% survival rate, even though chickens showed signs of mild pneumonia in lung tissues on day 6 p.i. Elevated levels of virus-induced pro-inflammatory cytokines in HP H5N6 and HP H5N1 infected chickens and other susceptible poultry could result in severe disease symptoms [21, 22, 23]. As reported previously, induced pro-inflammatory immune responses could be the reason for mild pneumonia in immunized chickens. Moreover, the 96HA vaccinated and HP H5N6 challenged chickens showed 80% mortality (Fig. 1A) and mild pneumonia in the lung tissues (Fig. 4C). However, no viral shedding was observed from either the trachea or cloacae on day 3 p.i. Disease severity and associated deaths could be due to the overexpression of inflammatory cytokines in the lung tissues and not due to the complications driven by increased viral replication in host organs.
In summary, a single dose of oil-adjuvanted inactivated H5N6 vaccine may be a potential solution for protecting poultry from recurring highly pathogenic H5 influenza virus infections.
In conclusion, our findings show that a prime boost of the 384HA H5N6 vaccine can completely protect chickens from lethal infection by HP H5N6, A/waterfowl/Korea/S57/2016 (clade 2.3.4.4) for long-term and is also effective against HP H5N1, A/Hong Kong/213/2003 (H5N1) (clade 1) influenza infections.
KAPPK and JYY performed the experiments, analyzed the data, and drafted the manuscript. SHS contributed to the conceptualization, designing of the experimental work and provided help and advice on the experiment work and drafting of the manuscript. All authors read and approved the final manuscript.
All animal experiments were approved by the Internal Animal Use Committee at Chungnam National University (CNU), Daejeon, Republic of Korea (Approval: 202012A-CNU-171). The experiments were performed according to the guidelines and regulations of CNU, Republic of Korea.
Veterinary students; Nam Chan Woo and Kim Yong Joo assisted in the housing and maintenance of the chickens used in this study.
This work was supported by a National Research Foundation of Korea (NRF) Grant funded by the Korean government (MSIT) (2019R1A2C2002166812).
The authors declare no conflict of interest. Sang Heui Seo was serving as Guest Editor of this journal. We declare that Sang Heui Seo had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Graham Pawelec.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.fbl2709268.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





