1 Department of Biochemistry and Molecular Biology, School of Basic Medical Sciences, Southern Medical University, 510515 Guangzhou, Guangdong, China
2 The 2016 Class, 8-Year Program, The First Clinical Medical School, Southern Medical University, 510515 Guangzhou, Guangdong, China
3 School of Science, The Hong Kong University of Science and Technology, 999077 Hong Kong, China
4 School of Basic Medical Sciences, Southern Medical University, 510515 Guangzhou, Guangdong, China
5 Guangdong Provincial Government Office, 510031 Guangzhou, Guangdong, China
†These authors contributed equally.
Academic Editor: Graham Pawelec
Abstract
Background: Gui Shao Tea (GST), a long-aged tea with a Chinese herbal
aroma, can treat many stubborn and malignant diseases, according to traditional
Chinese medicine. This research aimed to discover and define GST, study the
anti-gastric cancer effects of GST extracts and preliminarily elucidate the
mechanism of action in the PI3K/Akt signaling pathway and the gut microbiota.
Methods: GST was analyzed by GC/MS and HPLC. Cell proliferation, the
cell cycle and apoptosis were evaluated by a CCK8 assay and flow cytometry. The
effects of GST extracts on tumor inhibition and survival time were explored by a
gastric cancer xenograft model in nude mice. The PI3K/Akt signaling pathway was
assessed by western blotting and immunohistochemistry. Gut microbiota detection
and fecal microbiota transplantation were performed to examine whether the tumor
inhibition observed in mice was related to gut microbiota changes.
Results: The ingredients in GST, mostly terpenes and their derivatives,
were novel and more concentrated than those in tea made from the branches and
leaves of the same plant species, Camellia sinensis, picked and produced
the same year, while the levels of polyphenols and alkaloids were significantly
reduced. In BGC-823, MGC-803, and SGC-7901 gastric cancer cells, GST extracts
significantly inhibited proliferation (p = 0.037), induced G0/G1 arrest
(p
Keywords
- Gui Shao Tea
- gastric cancer
- antitumor
- terpenoids
Gastric cancer is one of the most common malignant tumors globally; it has the third highest mortality rate among all cancers [1], and China ranks second worldwide in both gastric cancer incidence and mortality [2]. The overall prognosis of terminal gastric cancer patients is poor due to the high heterogeneity of this disease [3, 4]. Gastric cancer is regulated by multiple signaling pathways. In particular, the PI3K/AKT signaling pathway is frequently activated [5, 6, 7, 8, 9], cyclinD1 is a known downstream target of the PI3K/AKT pathway [10], and cyclinD1 binds to CDK4/6 to regulate progression in the G1 phase of the cell cycle. Because tumorigenesis is largely dependent on uncontrolled progression of the cell cycle, the cyclinD1 pathway has emerged as an ideal target for gastric cancer therapy [11, 12, 13, 14]. In recent years, the gut microbiota has been found to be closely related to many cancers [15], including colorectal cancer [16] and liver cancer [17]. Changes in the composition and diversity of the gut microbiota have also been found in patients with gastric cancer [18]. Enterobacteriaceae abundance is associated with the occurrence and prognosis of gastric tumors [19], while some bacterial taxa are predictive markers of gastric cancer [20].
Camellia sinensis (L.) Kuntze (Theaceae) is an important natural plant. At present, anticancer research related to this plant has been mainly based on the polyphenols [21, 22, 23, 24, 25, 26, 27, 28, 29, 30] and alkaloids [31, 32] in its branches and leaves such as catechins, flavonoids, flavonoid glycosides, phenolic acids, and phenolics. According to traditional Chinese medicine texts, the branches and leaves of C. sinensis that have been stored for more than 20 years (called long-aged tea) and have a Chinese herbal aroma after being soaked or boiled can treat a variety of stubborn and malignant diseases [33, 34]. Current studies suggest that the specific aroma and elements of the herbs may be the result of the Maillard reaction occurring during the processing and storage of long-aged tea [35, 36]. Due to the long storage time, a large number of Maillard reaction products are produced after decades of oxidation, which may also furnish an antioxidant activity to this long-aged tea [37].
To clarify the changes in long-aged tea with a Chinese herbal aroma when boiled, we first formulated a test and found that this tea was completely different from tea made from the branches and leaves of the same species of plant, C. sinensis, which contains polyphenols, theanine, and alkaloids, so we named and defined the long-aged tea as Gui Shao Tea (GST). Then, to verify whether GST is effective against gastric cancer, we explored its anti-gastric cancer effect and investigated the molecular mechanism of water-soluble extracts of GST involving the PI3K/Akt signaling pathway and gut microbiota.
This is the first scientific research on GST, which is no longer limited to the general records in traditional Chinese medicine texts. The importance of these findings lies in the discovery of innovative anti-gastric cancer plant extracts and the hypothesis explaining how GST affects the development of gastric cancer.
Four samples that met the standards for GST were collected in Peking (a is from
the Chinese Tea Company Guangxi Branch ‘Chinese Tea Brand’, China), Guangxi (b is
from the Guangxi Wuzhou Qianyuan Green Leaf Tea Company ‘Farmer’s Brand’, China),
Yunnan (d is from the Yunnan Menghai Chunfurun Tea Company ‘Chunfurun Brand’,
China), and Guangdong (e is from the Chinese Tea Company Yunnan Branch ‘Chinese
Tea Brand’, China). They were all kept in dark, relatively sealed, ventilated and
odor-free places under controlled temperature (
After being crushed and passed through a 20-mesh sieve, 50 g of experimental
sample was placed in a headspace-solid phase microextraction device at 80
°C for 20 min. The GC inlet mode was set in the split mode with a split
ratio of 5:1. The inlet temperature was 250 °C. The carrier gas (He) was
set in the constant flow mode with a flow velocity of 1.0 mL/min. The analytical
column was a TG-5MS capillary quartz column (30 m
After being crushed and passed through a 20-mesh sieve, 0.2 g of experimental
sample was extracted at 70 °C with 5 mL of preheated 70% methanol
solution for 20 min. The extraction process was repeated twice. Then, the extract
solutions were combined, cooled to room temperature, and centrifuged at 3500
r/min for 10 min. The supernatant was decanted, diluted to volume with a 70%
methanol solution in a 10-mL volumetric flask and then filtered through a
0.45-
The chromatographic conditions applied were the result of optimization of the
elution mode, pH of the mobile phase, column temperature and flow rate. They
included maintaining an Agilent-C18 column (250 mm
Among the four GST samples, the sample with the highest number of new components
and significantly concentrated components was selected (Fig. 1E), ground into
powder, and soaked in 5
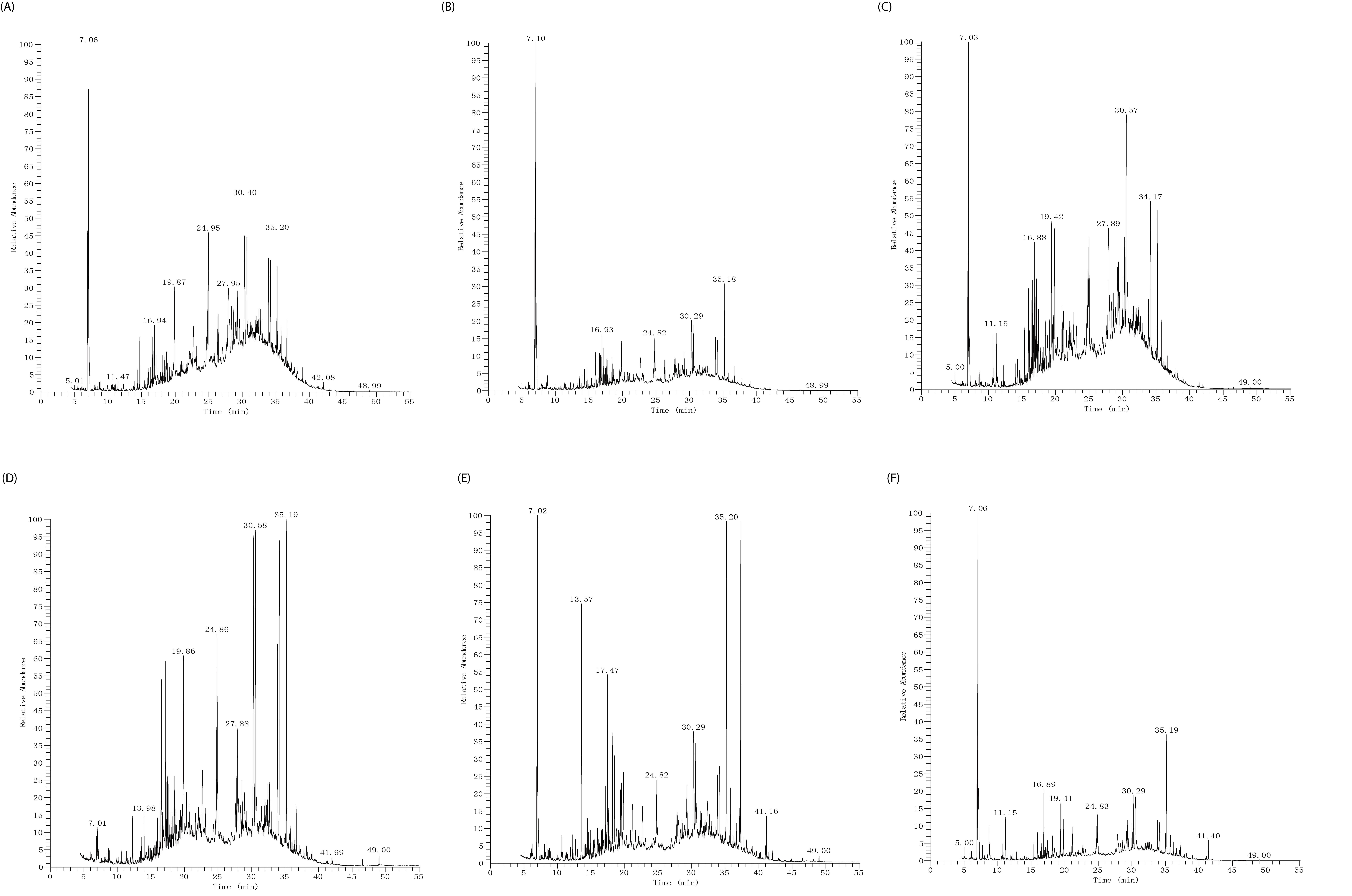 Fig. 1.
Fig. 1.Overview of the total ion chromatograms of the GST and NC samples generated by GC/MS. (A,B,D,E) GST; (C,F) NC.
The human poorly differentiated mucinous gastric adenocarcinoma cell line
MGC-803, human poorly differentiated gastric adenocarcinoma cell line BGC-823,
human moderately differentiated gastric adenocarcinoma cell line SGC-7901 and
human gastric mucosal epithelial cell line GES-1 were donated by the Department
of Gastroenterology, Nanfang Hospital, Southern Medical University (China); the
cell lines were recently authenticated by STR profiling and tested for mycoplasma
contamination. Cells were cultured in complete RPMI-1640 medium (Gibco, Grand
Island, NY, USA) containing 10% inactivated calf serum (Gibco, Grand Island, NY,
USA). Cell culture was performed at 37 °C in a 5% CO
BGC-823, MGC-803, SGC-7901 and GES-1 cells were seeded in 96-well plates at a
density of 10
BGC-823, MGC-803, SGC-7901 and GES-1 cells were seeded in 96-well plates at a
density of 10
BGC-823, MGC-803, SGC-7901 and GES-1 cells were seeded in six-well plates at a
density of 1.2
BGC-823, MGC-803, SGC-7901 and GES-1 cells were seeded in six-well plates at a
density of 1.2
Sixty 5-week-old BALB/c nude mice (half male and half female) (Quality Certificate of Laboratory Animals in Guangdong Province of China No. 44002100008649) were provided by the Animal Experimental Center of Southern Medical University (Guangzhou, China).
Nude mice were reared under specific pathogen-free conditions with free access to food and water, and gastric cancer models were established by subcutaneous injection of BGC-823 cells. When the tumor diameter reached more than 8 mm (approximately 2 weeks), the nude mice were randomized into two experimental groups: one group was used to observe tumor-suppressive effects, and the other group was used to observe the survival time of the tumor models; each group contained 24 nude mice, half male and half female. Each group was divided into four subgroups: the low-dose GST-fed group (LTF), middle-dose GST-fed group (MTF), high-dose GST-fed group (HTF), and negative control group (harbored tumors but treated with saline; NC). There was also a blank control group (subcutaneous injection of saline and treatment with saline; CK). Each group included 6 nude mice, half male and half female.
The doses of GST extracts used for intragastric administration were 0.5, 1.5,
and 2.5 g/kg of body weight (BW) for nude mice, which were equivalent to 4.5,
13.5, and 22.5 g/kg of BW GST for nude mice, respectively. The lowest dose for
nude mice was calculated based on the daily human consumption of GST. Mice were
orally administered 400
The observation time points set in the experimental protocol for animals used to
observe tumor-suppressive effects, including those detected by
immunohistochemistry (IHC) and gut microbiota evaluation, required euthanasia by
cervical dislocation at different times followed by tumor collection; the time
points included day 14 for the HTF group and day 28 for the MTF, LTF, and NC
groups. The humane endpoints for animals used to observe the survival time were
weight loss greater than or equal to 20% of BW, an inability to ambulate, an
inability to reach food or water, tumors greater than or equal to 10% of BW,
tumor ulceration, and a body condition score of 1 or less using the
IACUC-approved scoring system. At the end of a study, the animals were euthanized
by continuous CO
BGC-823 cells were treated with a 20
Tumor tissues were collected from the HTF group on the 14th day and from the MTF, LTF, and NC groups on the 28th day and fixed with 4% paraformaldehyde. Then, IHC analysis was performed using paraffin-embedded tissue sections that underwent conventional dehydration, clearing, waxing, embedding, and slicing. After the paraffin sections were baked for 60 min, the tissues were conventionally dewaxed in dimethyl benzene and water, washed with PBS and heated with trisodium citrate for antigen retrieval. Then, a 3% peroxidase blocking solution was added, and the sections were washed with PBS. The slides were incubated with primary antibodies specific for p-Akt, cyclinD1, p-Rb, and E2F1 overnight at 4 °C in a refrigerator and then rinsed with PBS. A corresponding secondary antibody was added, and the slides were incubated at room temperature for 45 min. Traditional methods were used for DAB color development, hematoxylin counterstaining, dehydration, sealing, and microscopy imaging. The images were analyzed with Image-Pro Plus (Media Cybernetics, United States); brown particles indicated positive expression, and positive staining in images was quantified as integrated optical density (IOD)/area, i.e., the average density = sum of densities/area sum [39]. Specifically, 5 positive fields were selected from each section, and the mean of the mean densities was calculated. The antibodies used were as follows: a rabbit anti-p-Akt polyclonal antibody (Abcam, Cambridge, UK, ab8933, 1:200), a rabbit anti-cyclinD1 monoclonal antibody (Abcam, Cambridge, UK, ab16663, 1:100), a rabbit anti-p-Rb polyclonal antibody (Abcam, Cambridge, UK, ab47763, 1:200), a rabbit anti-E2F1 monoclonal antibody (Abcam, Cambridge, UK, ab179445, 1:200), and an HRP-labeled goat anti-rabbit IgG secondary antibody (Abcam, Cambridge, UK, ab6721, 1:500).
Feces were collected from nude mice in each group every 7 days with an Eppendorf
(EP) tube and stored in a –80 °C freezer until the humane endpoint
occurred. In the experiment, 200 mg of each fecal sample was weighed, mixed with
1 ml of 70% ethanol, and centrifuged at 10,000 rpm for 3 min at room
temperature. Then, the supernatant was discarded, and 1
Gastric cancer models were established with nude mice by subcutaneous injection
of BGC-823 cells and divided into donor and transplantation groups, which
included 4 subgroups with 6 mice (3 males and 3 females) in each group. There was
also a blank control group (CK) containing 6 mice (3 males and 3 females). The
mice in the donor and transplantation groups were considered eligible FMT
recipients if their tumor diameter exceeded 8 mm. Donors were intragastrically
fed the low, middle, or high dose of GST extracts or fed normal saline (LTF, MTF,
HTF, and NC groups, respectively). The transplantation groups corresponding to
the donor groups for FMT were designated the low-dose GST-fed transplantation
group (LTT), middle-dose GST-fed transplantation group (MTT), high-dose GST-fed
transplantation group (HTT), and negative control transplantation group (NCT).
FMT was performed by oral administration using fresh graft material made from a
total of 200 mg of feces from the same donor group, which was resuspended in 2 mL
of sterile saline, vigorously mixed for 10 s, and centrifuged at 800 rpm for 3
min, with the pellet discarded. The preparation process was usually performed
within 10 min to prevent changes in the bacterial composition. The treatment was
administered to the mice at 100
We commissioned the Guangdong Provincial Center for Disease Control and Prevention, China, to conduct acute toxicity tests with GST extracts in accordance with the standard procedures prescribed by China.
A two-tailed Student’s t test was employed to analyze both the
in vitro data and in vivo data. One-way ANOVA was applied to
analyze tumor growth. The Mantel-Cox (log-rank) test was used for survival
analysis. All analyses were performed with SPSS 20.0 (IBM Corp., Chicago, IL,
USA). A threshold of p
GST and NC samples were analyzed by GC/MS and HPLC. The results showed that
compared with the NC group, new components appeared in the GST group, including
vitispirane, tridecane, 2,6,10-trimethyldodecane, 6,10-dimethyl-2-undecanone,
thujopsene, hydroxydihydroedulan,
2,6-di-tert-butyl-p-benzoquinone,
| No. | CAS | Compound name | Content, mg/g | |||||
| A | B | C | D | E | F | |||
| 1 | 83-67-0 | Theobromine | 0.52 | 0.19 | 1.03 | 0.07 | 0.19 | 1.52 |
| 2 | 58-55-9 | Theophylline | 0.45 | 0.27 | 0.98 | 0.01 | 0.04 | 0.31 |
| 3 | 58-08-2 | Caffeine | 6.30 | 3.79 | 24.16 | 1.49 | 1.71 | 29.07 |
| 4 | 989-51-5 | EGCG | — | — | 191.80 | 18.84 | — | 116.62 |
| 5 | 1257-08-5 | ECG | 35.23 | 26.51 | 105.14 | 17.06 | 14.75 | 98.98 |
| No. | CAS | Compound name | Sample abundance, % | |||||
| A | B | C | D | E | F | |||
| 1 | 20126-76-5 | 4-Carvomenthenol | 1.64 | — | — | — | 1.2 | — |
| 2 | 127-91-3 | 13.52 | — | 12.61 | — | 9.29 | 6.27 | |
| 3 | 555-10-2 | 6.76 | — | 4.59 | — | 2.04 | — | |
| 4 | 123-35-3 | 8.88 | — | 7.39 | — | 2.95 | 1.47 | |
| 5 | 13877-91-3 | 1.74 | — | 1.53 | — | 0.77 | 0.77 | |
| 6 | 110-93-0 | 6-Methyl-5-hepten-2-one | 1.06 | — | 0.45 | — | 0.21 | — |
| 7 | 138-86-3 | 11.16 | 27.36 | 6.31 | — | 7.58 | 26.41 | |
| 8 | 99-86-5 | 2.95 | — | 2.25 | — | 1.48 | — | |
| 9 | 99-83-2 | 5.5 | — | 4.05 | — | 1.58 | 0.58 | |
| 10 | 65416-59-3 | Vitispirane | — | 0.59 | — | — | 5.98 | — |
| 11 | 629-50-5 | Tridecane | — | 0.56 | — | 0.81 | — | — |
| 12 | 3891-98-3 | 2,6,10-Trimethyldodecane | 0.95 | 1.95 | — | 0.69 | — | — |
| 13 | 1604-34-8 | 6,10-Dimethyl-2-undecanone | — | 1.15 | — | 0.63 | — | — |
| 14 | 470-40-6 | Thujopsene | — | 1.21 | — | 1.32 | — | — |
| 15 | 1344702-48-2 | Hydroxydihydroedulan | — | 1.09 | — | 0.76 | — | — |
| 16 | 719-22-2 | 2,6-Di-tert-butyl-p-benzoquinone | 1.05 | 1.4 | — | 1.51 | — | — |
| 17 | 17066-67-0 | — | 0.78 | — | — | 2.13 | — | |
| 18 | 2883-02-5 | n-Nonyl cyclohexane | 1.1 | — | — | 1.17 | — | — |
| 19 | 137986-80-2 | Methyl 2-endo-Acetamidobicyclo[2.2.1]heptane-2-exo-carboxylate | — | 2.41 | — | 2.47 | 2.47 | — |
| 20 | 1560-93-6 | 2-Methyl-pentadecane | 2.68 | — | 1.62 | — | — | 1.52 |
| 21 | 65990-71-8 | 5,6-dihydro-5,6-dimethyl benzo[c]cinnoline | 1.92 | 1.35 | — | 1.31 | — | — |
| 22 | 6418-43-5 | 3-Methyl-hexadecane | 1.44 | 1.52 | — | 2.14 | — | — |
| 23 | 629-92-5 | Nonadecane | 1.21 | 0.78 | — | 0.83 | 0.7 | — |
| 24 | 78-70-6 | Linalool | 0.77 | — | 1.17 | — | 1.62 | 2.82 |
| 25 | 10482-56-1 | — | — | 1.17 | — | — | 1.94 | |
| 26 | 17092-92-1 | dihydroactinidiolide | — | — | 1.53 | — | 0.99 | 1.54 |
| 27 | 119-36-8 | Methyl salicylate | 0.39 | — | 1.17 | — | 0.77 | 17.8 |
| 28 | 79-77-6 | — | — | 3.61 | 0.67 | 1.81 | 3.19 | |
| 29 | 74367-33-2 | Propanoic acid 2-methyl-2,2-dimethyl -1-(2-hydroxy-1-methylethyl) propyl ester | — | — | 1.09 | — | — | 0.87 |
| 30 | 85-91-6 | Methyl N-methyl anthranilate | — | — | 4.13 | — | — | 3.74 |
| 31 | 60305-17-1 | — | — | 1.86 | — | — | 0.97 | |
| 32 | 781-33-9 | 1,1-Diphenyl-2-methylpropene | — | — | 2.17 | — | — | 2.17 |
| 33 | 6416-39-3 | 2,3-Dihydro-1-methyl-3-phenyl-1H-indene | — | — | 1.62 | — | — | 1.97 |
In the GST group, other components, particularly polyphenols and alkaloids, were
decreased, including EGCG, ECG, theobromine, theophylline, caffeine, linalool,
dihydroactinidiolide, methyl salicylate,
In terms of composition, GST was completely different from the tea made from the branches and leaves of the same plant species, C. sinensis, which contains polyphenols, theanine, and caffeine simultaneously. In view of the significant increase in terpenes and their derivatives in GST extracts, and given that the anti-tumor activity of terpenes is well known, we choose the sample (Fig. 1E) with the highest number of new components and significantly concentrated components among the four GST samples, extracted it and performed antitumor experiments with the GST extracts.
We obtained 79 g of extracts from 700 g of GST, and the extraction rate was
11.3%. The viability of BGC-823, MGC-803, and SGC-7901 cells with different
concentrations of GST extracts were detected using the CCK8 method at 48 h. The
results showed that GST extracts significantly inhibited cell viability in a
concentration-dependent manner (Fig. 2A). The IC50 values for BGC-823, MGC-803,
and SGC-7901 cells at 48 h were 19.31
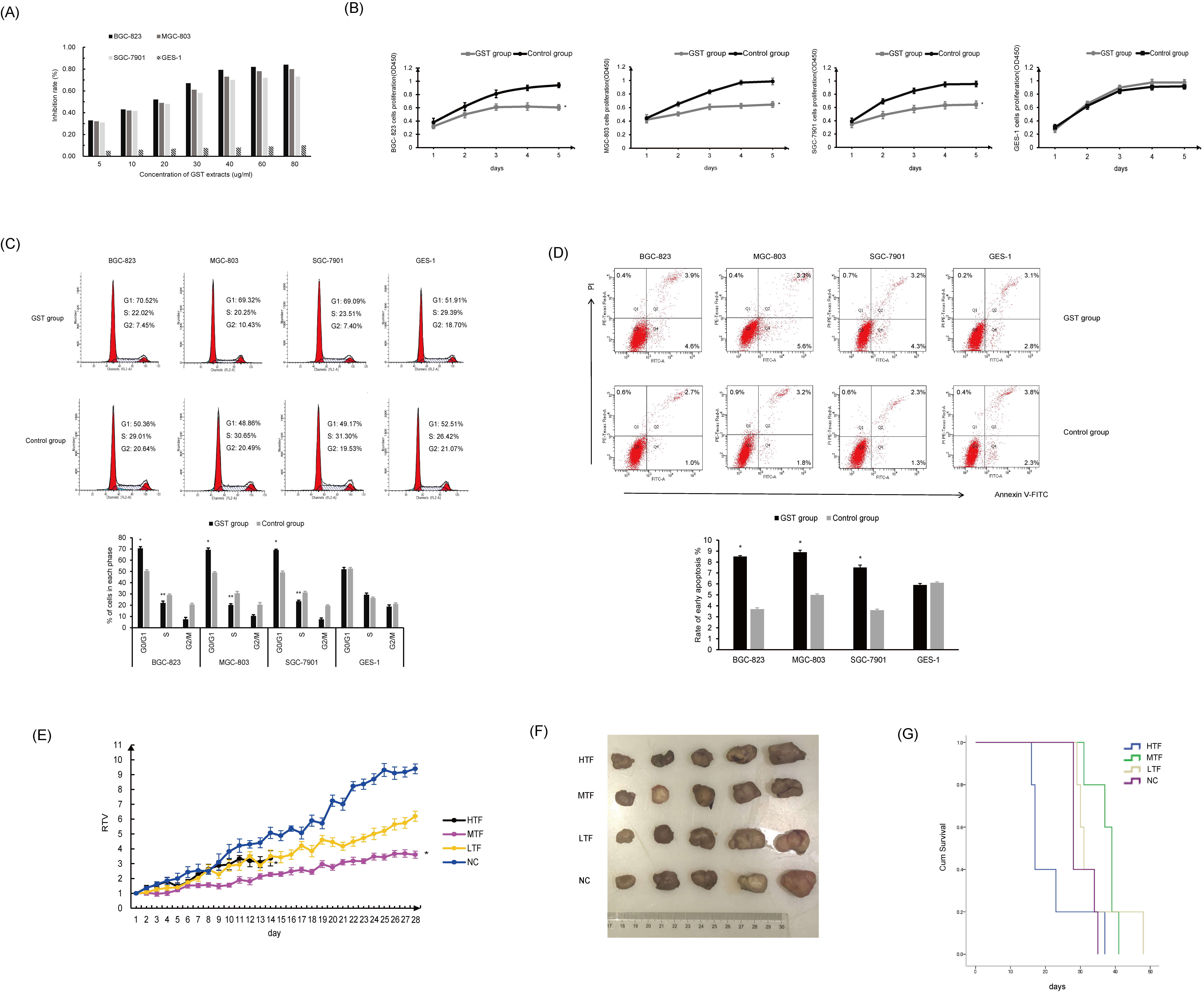 Fig. 2.
Fig. 2.Inhibition of gastric cancer by GST extracts in vitro
and in vivo. (A) GST extracts inhibited BGC-823, MGC-803, and SGC-7901
cell viability in a concentration-dependent manner. (B) The CCK8 assay results
showed that the GST extracts significantly inhibited BGC-823, MGC-803, and
SGC-7901 gastric cancer cell proliferation without clear inhibitory effects on
human gastric mucosal epithelial GES-1 cells. *p = 0.037, compared with
the control group. (C) The cell cycle of BGC-823, MGC-803, and SGC-7901 gastric
cancer cells and GES-1 cells was analyzed using flow cytometry. The results
showed that the GST extracts inhibited the cell cycle in gastric cancer cells via
G0/G1 arrest. *p
The GST extracts were administered to model mice with gastric cancer, and on the 14th and 28th days, at least 5 nude mice from each group had survived. The results showed that gastric tumor growth was significantly inhibited in the HTF and MTF groups. The inhibition rate of the HTF group on the 14th day was 33.77% (p = 0.042), and the inhibition rate of the MTF group was 55.21% on the 14th day (p = 0.002) and 61.6% on the 28th day (p = 0.008) (Fig. 2E,F, Table 3). The survival time of the MTF group was significantly prolonged by 22.2% compared with that of the NC group (p = 0.013) (Fig. 2G, Table 4). Although the survival time of the HTF group was shorter than that of the NC group, there was no significant difference (p = 0.434) (Fig. 2G, Table 4). There were no significant differences in tumor growth (p = 0.060) (Fig. 2E,F, Table 3) or survival time (p = 0.469) (Fig. 2G, Table 4) between the LTF group and the NC group. Therefore, we concluded that treatment with GST extracts at a suitable dose (MTF) not only had a strong tumor growth inhibitory effect but also prolonged mouse survival time. These results suggest that GST extracts have the potential to treat gastric cancer.
| Group | n | 14th day RTV | 14th day TGI | 28th day RTV | 28th day TGI |
| HTF | 5 | 3.361 |
33.77% | ||
| MTF | 5 | 2.273 |
55.21% | 3.607 |
61.60% |
| LTF | 5 | 3.502 |
31.00% | 6.192 |
34.09% |
| NC | 5 | 5.075 |
9.394 |
||
| F | 4.422 | 5.169 | |||
| p | 0.019 | 0.024 | |||
| In vivo, GST extracts significantly inhibited the growth of gastric
cancer. 1 vs. NC, p = 0.042; 2 vs. NC, p = 0.002; 3 vs. NC, p = 0.060; 4 vs. NC, p = 0.008; 5 vs. NC, p = 0.101. | |||||
| Survival time | n | Donor group | Transplantation group | ||
| mean | median | mean | median | ||
| High-dose | 5 | 22 |
17 |
44.8 |
45 |
| Middle-dose | 5 | 37.4 |
39 |
44.4 |
34 |
| Low-dose | 5 | 34.4 |
31 |
46.8 |
44 |
| NC | 5 | 30.6 |
28 |
34.6 |
38 |
| Chi-Square Log Rank | 8.549 | 0.785 | |||
| p | 0.036 | 0.853 | |||
| The survival time of the MTF group was significantly longer than that of the NC
group, and the survival time of the HTT group was significantly prolonged
compared with that of the HTF group. 1 vs. NC, p = 0.434; 2 vs. NC, p = 0.013; 3 vs. NC, p = 0.469; high-dose transplantation group vs. high-dose donor group, p = 0.002. | |||||
Given that our results suggest that GST extracts play a role in the transition of gastric cancer cells from G1 to the S phase, we used western blotting to analyze the expression of key regulators of the cell cycle in the PI3K/Akt signaling pathway of BGC-823 cells after GST extract treatment. The results showed that the expression levels of p-Akt, cyclinD1, p-Rb, and E2F1 were significantly lower than those in BGC-823 cells not treated with GST extracts (control group) (p = 0.016). When BGC-823 cells were treated with the PI3K/Akt signaling pathway inhibitor LY294002, the pathway changes were similar to those observed in the GST extracts treatment group (p = 0.023) (Fig. 3A), indicating that GST extracts can inhibit the PI3K/Akt signaling pathway in gastric cancer.
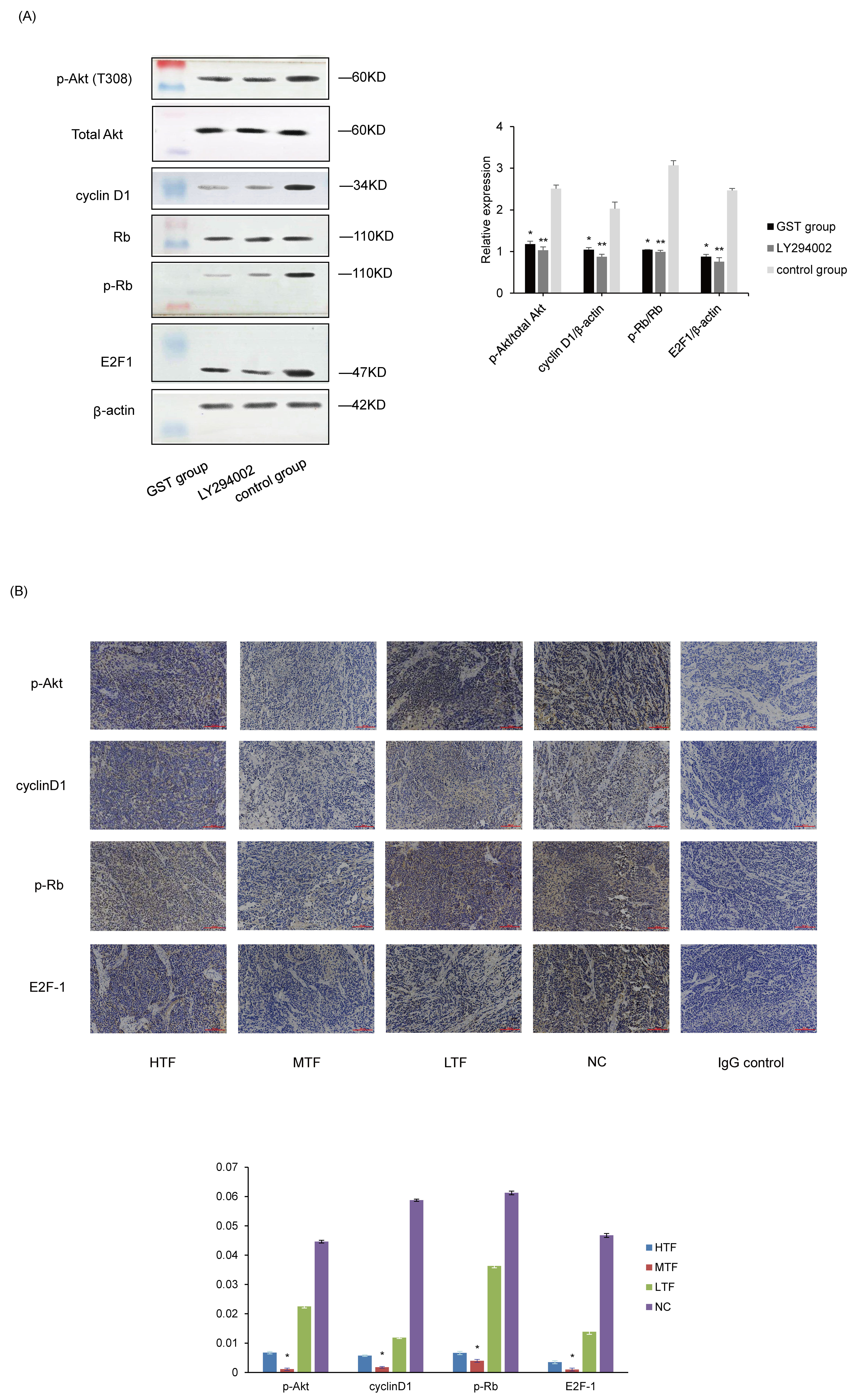 Fig. 3.
Fig. 3.Inhibition of the PI3K/Akt signaling pathway in gastric cancer
by GST extracts. (A) Western blot results for BGC-823 cells showed that the
expression levels of p-Akt, cyclinD1, p-Rb, and E2F1 in the GST extracts
treatment group were significantly lower than those in the control group
(*p = 0.016). When the PI3K/Akt signaling pathway inhibitor LY294002 was
administered, the pathway changes were similar to those observed in the GST
extracts treatment group. **p = 0.023, compared with the control group.
(B) The immunohistochemistry results showed that the expression levels of p-Akt,
cyclinD1, p-Rb, and E2F1 in the MTF group were significantly suppressed, which
was consistent with the western blot results. *p = 0.029, compared with
the NC group (200
Next, we used IHC to detect changes in the PI3K/Akt signaling pathway in tumor tissues from the GST extracts treatment group. The results showed that the expression levels of p-Akt, cyclinD1, p-Rb, and E2F1 in the MTF group were significantly suppressed (p = 0.029) (Fig. 3B), which was consistent with the western blot results.
The effect of GST extracts on the gut microbiota of nude mice with gastric
cancer was detected by 16S DNA sequencing. UniFrac-based principal coordinate
analysis (PCoA) revealed distinct clustering of the microbiota composition in
each group (Fig. 4A). The gut microbiota of the MTF group on the 28th day and
that of the HTF group on the 14th day were similar to that of the normal group
(CK) (Fig. 4B). Taxonomic profiling demonstrated that the ratio of Firmicutes to
Bacteroidetes decreased over time in the NC group and increased over time in each
GST-fed group. In particular, this ratio was significantly higher in the MTF
group on the 28th day and in the HTF group on the 14th day than those in the NC
and blank control groups. Additionally, Proteobacteria abundance was notably
increased in all GST-fed groups (Fig. 4B). Differences in bacterial function
between the NC group and the MTF group on the 28th day or the HTF group on the
14th day were identified by Kyoto Encyclopedia of Genes and Genomes (KEGG)
analysis, which revealed that the altered microbiota functions were mainly
concentrated in metabolism, growth, death, enzymes, replication, and repair
(p
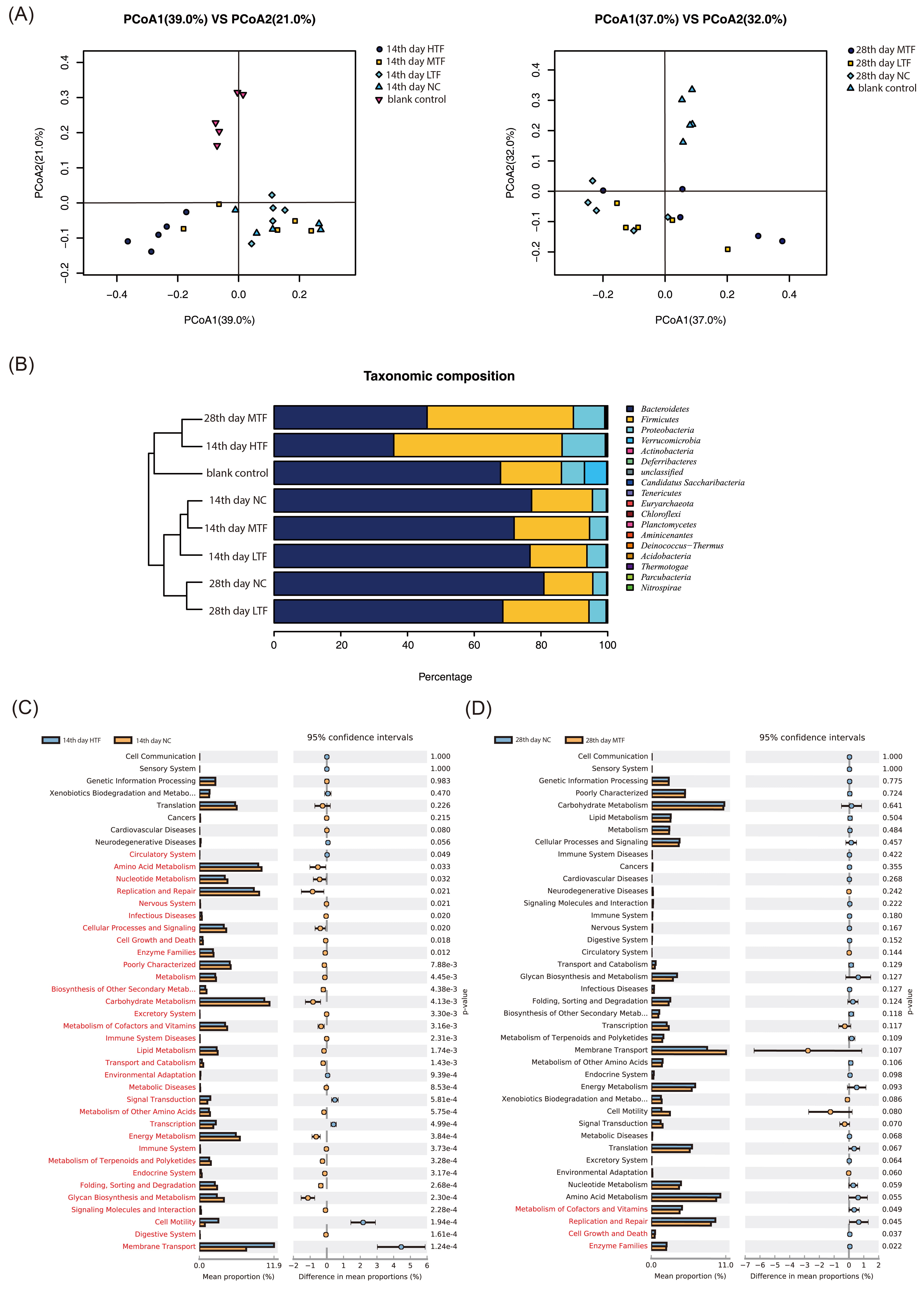 Fig. 4.
Fig. 4.GST extracts altered the gut microbiota composition in a nude
mouse model of gastric cancer. The gut microbiota composition in the feces of
mice harboring gastric cancer xenografts treated with or without GST extracts was
analyzed using 16S DNA sequencing (n = 5 for each group). (A) Plots were
generated using the weighted version of UniFrac-based PCoA. (B) Taxonomic
profiling of the gut microbiota in different groups at the phylum level.
(C,D) Analysis of the functional differences in the gut microbiota
based on KEGG analysis showed that the changes were mainly focused on metabolism,
growth and death, enzymes, replication and repair (red indicates p
Because GST extracts altered the gut microbiota of nude mice with gastric cancer and inhibited tumor growth, we speculated that the alteration of gut microbiota most likely lead to tumor suppression. To test this hypothesis, we performed FMT experiments and found that tumor growth was not inhibited in any of the transplant groups on the 14th day (p = 0.742) or the 28th day (p = 0.572) (Table 5). Although the survival time of the HTT group was significantly longer than that of the HTF group (p = 0.002), there was no significant extension of survival in the other transplant groups (Table 4).
| Group | n | 14th day RTV | 28th day RTV |
| HTT | 5 | 3.490 |
6.021 |
| MTT | 5 | 3.258 |
5.624 |
| LTT | 5 | 2.977 |
4.309 |
| NCT | 5 | 2.598 |
4.369 |
| F | 0.418 | 0.691 | |
| p | 0.742 | 0.572 | |
| Gastric cancer growth was not affected by fecal microbiota transplantation. | |||
The acute oral toxicity of GST extracts was LD50 = 10.80 g/kg of BW (GST was LD50 = 95.6 g/kg of BW) in female Kunming mice and LD50 = 12.60 g/kg of BW (GST was LD50 = 111.5 g/kg of BW) in male Kunming mice; both values indicated that GST extracts were nontoxic.
Current studies on C. sinensis have mainly focused on polyphenols and alkaloids; however, our results show that these polyphenols and alkaloids are greatly reduced or absent in GST, so the active ingredients in GST are clearly not the polyphenols and alkaloids that have always been of concern. In contrast, terpenes and their derivatives, especially monoterpenes and sesquiterpenes, are increased significantly in GST, and even some new terpenes, such as limonene, which has been shown to have significant anticancer effects, have been found [40, 41]. Terpenoids, the largest category of natural products, include more than 30,000 different compounds, most of which are derived from plants and are primary or secondary metabolites of plants. In animals and humans, terpenoids have anticancer activity [42], and these molecules include paclitaxel [43], artemisinin [44], ganoderic acid [45], limonene [40, 41], etc., so terpenoids are important candidates for cancer therapy. The terpenoids in GST were increased significantly, and even new terpenoids were identified, which is likely to be the molecular basis for the treatment of stubborn and malignant diseases recorded in traditional Chinese medicine texts. It is worth mentioning that one of the emerging components is the norisoprenes (degradants of sesquiterpenes) vitispirane, on which few research articles have been published, and it may have some special pharmacological effects that deserve further study.
As a new focus, we aimed to explore whether GST has an anticancer effect in a gastric cancer model. The in vitro results showed that GST extracts could inhibit the growth of gastric cancer cells; in view of the relatively good inhibitory effect of GST extracts on BGC-823 cells, we chose to use BGC-823 cells to establish a gastric cancer model in nude mice, and found that GST extracts could inhibit the growth of gastric cancer in vivo and prolong the survival of nude mice. All these results suggest that GST extracts have the potential to treat gastric cancer. In addition, the results showed that the HTF group exhibited significantly inhibited tumor growth on the 14th day, but the survival time was not significantly different from that of the NC group, although it appeared that the HTF group had a shorter survival time than the NC group. To explain this result, we performed an acute oral toxicity test, and the results showed that GST extracts are nontoxic, suggesting that a high dose of GST extracts may have other side effects that require further research. In short, although we conducted a preliminary exploration in a gastric cancer model, GST extracts showed very surprising antigastric cancer effects, which suggests that GST has important potential for the development of therapeutic drugs for gastric cancer.
Furthermore, our results showed that GST extracts reduced the expression of the
key cell cycle regulatory proteins p-Akt, cyclinD1, p-Rb and E2F1. Once cyclinD1
binds to CDK4/CDK6, Rb is phosphorylated. p-Rb is then released from E2F1, which
in turn acts as a transcription factor engaged in cell cycle progression and cell
proliferation. Our results indicated that GST extracts inhibit the PI3K/Akt
signaling pathway and cyclinD1, accounting for G0/G1 arrest through the
PI3K-Akt-cyclinD1-Rb-E2F1 pathway and ultimately inhibiting gastric cancer cell
proliferation. We only investigated cell cycle-related changes in the PI3K/Akt
signaling pathway; there are also PI3K/AKT/mTOR [46], PTEN/PI3K/AKT [47],
Wnt/
Then, we investigated whether the inhibitory effect of GST extracts on gastric cancer is related to the gut microbiota. The results showed that GST extracts could significantly change the composition of the gut microbiota in the HTF and MTF groups; in particular, the ratio of Firmicutes to Bacteroidetes as well as the abundance of Proteobacteria were greatly increased, whereas these taxa decreased in the NC group over time. Bacteria residing in the human gastrointestinal tract are typically classified into four phyla: Firmicutes, Bacteroidetes, Proteobacteria, and Actinobacteria [52]. The effects of microbiota on cancer development may be contradictory as some bacteria have been reported to facilitate carcinogenesis, whereas others appear to inhibit carcinogenesis in the gastrointestinal tract [53, 54]. Baxter et al. [55] found in mice models that several members of the Bacteroidales (Bacteroides, Parabacteroides, Alistipes, and Porphyromonodaceae) were positively correlated with tumor development, whereas members of the Clostridiales, particularly Clostridium Group XIVa, were associated with a decreased cancer risk. Bacteroides is one of the genera most strongly correlated with higher rates of tumorigenesis and is known as a mucin degrader, and several genes linked to mucin degradation are positively correlated with tumor incidence. Studies have linked mucin degradation by Bacteroides with intestinal inflammation, and it is possible that an overabundance of these or other mucin degraders could undermine the integrity of the mucosal barrier, leading to increased inflammation [56], and inflammation is a well-established risk factor for many cancers [57]. Our results showed that GST extracts reduced Bacteroidetes abundance in the HTF and MTF groups, and tumors were also suppressed. Lopetuso et al. [58] also found that the only identified precursor of esophageal adenocarcinoma (EAC) was Barrett’s esophagus (BE), and during the transition from BE to EAC, the ratio of Firmicutes to Bacteroidetes gradually decreased. Our results showed the same trend in the NC group, but GST extracts reversed this trend in the HTF and MTF groups. Zhou et al. [59] obtained bacteria with potent antimalignancy activities, which mostly belonged to Actinobacteria but also included lineages of other phyla such as Proteobacteria and Firmicutes. Our results also found that GST extracts increased Proteobacteria and Firmicutes in the HTF and MTF groups, which suggested that GST extracts could inhibit gastric cancer in nude mice by increasing antimalignancy bacteria. However, when H. pylori is added as a factor, the results are reversed; this has been confirmed in previous studies, and H. pylori-positive and H. pylori-negative individuals show significantly different bacterial communities [60, 61].
Because GST extracts altered the gut microbiota of nude mice with gastric cancer and inhibited tumor growth, we speculated that the alteration of gut microbiota most likely lead to tumor suppression. To determine that GST extracts inhibited gastric cancer in nude mice by modulating gut microbiota, we performed an FMT experiment; however, the FMT experiment found no effect on the growth of gastric cancer. This result showed that although the GST extracts could change the gut microbiota of nude mice, they could not inhibit the growth of gastric cancer by changing only the gut microbiota. Therefore, whether the inhibitory effect of GST extracts on gastric cancer depends on the components themselves or whether the components and gut microbiota play a role together in suppressing cancer needs further study. In addition, future studies may address whether changes in the gut microbiota have other effects on nude mice.
Limitations should be considered for this study. First, this study aimed to discover a new product, GST, and preliminarily confirm that GST extracts have anti-gastric cancer effects, so the sample size was small and the treatment course was relatively short. Second, we found that tumor growth was inhibited in the MTF group, while the survival time was significantly prolonged, but whether the middle dose of GST extracts is the best dose for gastric cancer therapy needs to be further studied. Finally, GST extracts are a complex mixture of multiple plant components. Existing studies have found that the antitumor effects of some plant extracts are derived from specific components [62], while the effects of other plant extracts are derived from the synergistic effect of multiple components [63]. We have only initially studied the anti-gastric cancer effect of GST extracts, and the active components are still unclear, so whether there is a synergistic effect among the components has not been studied.
For the first time, this study characterized long-aged tea with a Chinese herbal aroma when boiled, designated it as GST, and conducted an antigastric cancer study. As part of the research results, we have applied for and obtained two Chinese invention patents [64, 65].
GST is completely different from tea made from the branches and leaves of the same plant species, C. sinensis, which is picked and produced in the same year; specifically, the ingredients in GST, which are largely terpenes and their derivatives, were found to be novel and more concentrated, whereas the polyphenols and alkaloids were significantly reduced. GST extracts could significantly inhibit the growth of gastric cancer in vitro and in vivo, prolong survival in nude mice, inhibit the PI3K/Akt signaling pathway, and change the gut microbiota of nude mice. This study confirms that GST extracts have the potential to inhibit gastric cancer and provides new approaches and methods for the research and development of drugs for gastric cancer.
YL and ZX designed the research study. YL, ZJ, HY, JY, JZ, JW and ZX performed the research. ZX provided help and advice on conception, acquisition of data and supervision. YL, ZJ, HY, JY, JZ, JW and ZX analyzed the data. YL, ZX, ZJ and HY wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
All procedures performed in studies involving plants complied with the IUCN Policy Statement on Research Involving Species at Risk of Extinction and the Convention on the Trade in Endangered Species of Wild Fauna and Flora. Animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of Southern Medical University (Permission No.2017037) and were carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Thanks to Quanhong Zhu for GST extracting. Thanks to Feilong Chen for detection of GC-MS and HPLC. Thanks to Xuhui Tan for assisting in statistical analysis.
The main source of funding for this research was from YL and ZX. In addition, this research was funded by Undergraduate Innovation and Entrepreneurship Training Program of Guangdong [grant numbers S202112121124, 202112121348]; Science and Technology Planning Project of Guangdong Province [grant number 2015A010107005]; and Natural Science Foundation of Guangdong Province [grant number 2017A030310022].
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




