1 State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, Department of Oral Implantology, West China Hospital of Stomatology, Sichuan University, 610041 Chengdu, Sichuan, China
Academic Editor: Tsuyoshi Sugiura
Abstract
Liquid biopsy has advantages over traditional biopsy, which cannot determine tumor dynamics. As a noninvasive and precise test, liquid biopsy detects biomarkers that carry information on tumor progression and has undergone tremendous development in recent years. Exosome detection is one of the methods of liquid biopsy. Radiotherapy affects the release of exosomes and intercellular communication. Based on the properties, extractability, and detectability of exosomes, key exosomal cargoes after tumor radiotherapy can be used as biomarkers for tumor prognosis. Exosomes after tumor radiotherapy can be used for liquid biopsy. The main applications include (1) predicting radiotherapy efficacy, (2) predicting tumor prognosis, and (3) optimizing the regimen of tumor treatment. This review provides further research directions for liquid biopsy after tumor radiotherapy.
Keywords
- exosomes
- liquid biopsy
- cancer
- radiotherapy
Radiotherapy is one of the major therapies for many types of tumors [1, 2, 3]. Although radiotherapy can locally control tumors and improve the quality of life of patients [4], some problems often occur after radiotherapy. These include poor efficacy [5], tumor recurrence [6, 7], poor prognosis [8], and resistance to radiotherapy [9]. As a noninvasive and precise detection tool, liquid biopsy has great potential to address these problems of tumor radiotherapy [10]. Liquid biopsy is performed on free DNA, circulating tumor cells, and exosomes [11]. Exosomes, extracellular vesicles secreted by most cells, are present in many body fluids [12]. Radiation affects the composition, secretion, and function of exosomes [13, 14]. Based on the extractability of exosomes [15], after tumor radiotherapy, detectable changes in exosomes in body fluids can be used for the prediction of radiotherapy efficacy and prognosis [16, 17, 18]. Moreover, because of the heterogeneity of exosomes, the separation of exosomes into distinct subtypes by specific biomarkers is beneficial for clinical translation. There is a phage display-based platform for exosome diversity characterization, which identifies, isolate, and molecularly characterizes the disease-related exosomes depending on the different antigenic reactivities [19].
Many researchers have reviewed the prospect of applying exosomes to the liquid biopsy of tumors [20, 21, 22, 23, 24], but the prospect of applying exosomal liquid biopsy to the prediction of the efficacy and prognostic testing of tumors after radiotherapy has yet to be strengthened. For application, we have summarized the methods for the extraction and detection of exosomes based on their properties in vivo; in terms of the mechanism, radiation affects exosome secretion and function in cellular communication. This review summarizes the specific exosomes produced after tumor radiotherapy in the context of the response of different tumors to radiotherapy, expecting to provide certain ideas for clinical and basic researchers from the perspective of the mechanism. On this basis, we summarize the application and potential of exosomes in liquid biopsy after tumor radiotherapy. Altered exosomal cargo after tumor radiotherapy is used (1) as an indicator of tumor radiotherapy and can be used for the continuous assessment of radiotherapy efficacy through dynamic monitoring; (2) to predict and detect tumor recurrence, overall survival, early progression, and metastasis, as well as to suggest cognitive impairment after radiotherapy; and (3) to help optimize treatment planning (guide tumor patients’ medications during radiotherapy, reduce radiotherapy resistance through targeted therapy, and perform a risk assessment to select treatment options). This review provides an innovative direction for liquid biopsy after tumor radiotherapy and for the monitoring of oncology radiotherapy.
Exosome biogenesis is associated with the double invagination of the plasma membrane [25]. The formation of early-sorting endosomes (ESEs) containing proteins from the cell surface and extracellular milieu is the first invagination of the plasma membrane [26]. Then, after the maturity of late-sorting endosomes (LSEs) from ESEs, the invagination of the endosomal limiting membrane (the second invagination) occurs to form multivesicular bodies (MVBs). Subsequently, the intraluminal vesicles (ILVs) in MVBs contain proteins, nucleic acids, lipids, and so on (the cargoes of exosomes), and exosomes are ILVs secreted extracellularly [27, 28]. Most MVBs will be degraded by fusing with lysosomes or autophagosomes; otherwise, MVBs will fuse with the cytomembrane and release ILVs [28]. The mechanism of how to choose cargoes for ILVs has not been well clarified. The widely used theory to explain cargo sorting is the endosomal sorting complex required for transport (ESCRT)-dependent and ESCRT-independent signals [29]. The process of endosomal sorting has been shown to be mediated by ceramide and proteins of the tetraspanin family [30, 31]. Moreover, ESCRT-associated components (Alix and TSG101), tetraspanins (CD9, CD63, and CD81), and heat shock proteins (HSP60 and HSP70) exist in exosomes specifically, which are different from the original cell [29, 32]. The cytoskeleton transports MVBs to the cell membrane and is regulated by multifarious Rab GTPases, which shift from GDP- to GTP-bound states to activate effectors [33]. As the last step, the fusion between MVBs and the plasma membrane is controlled by soluble N-ethylmaleimide-sensitive factor attachment protein receptor (SNARE) proteins and their regulators [34]. Three mechanisms may be involved in the recipient cell uptake of exosomes. (1) Exosome fusion with the cellular membrane leads to the exosomal cargo being directly released into the recipient cell. (2) Exosome ligands interact with the receptor in the recipient cell. (3) The recipient cell absorbs exosomes through endocytosis or phagocytosis [35].
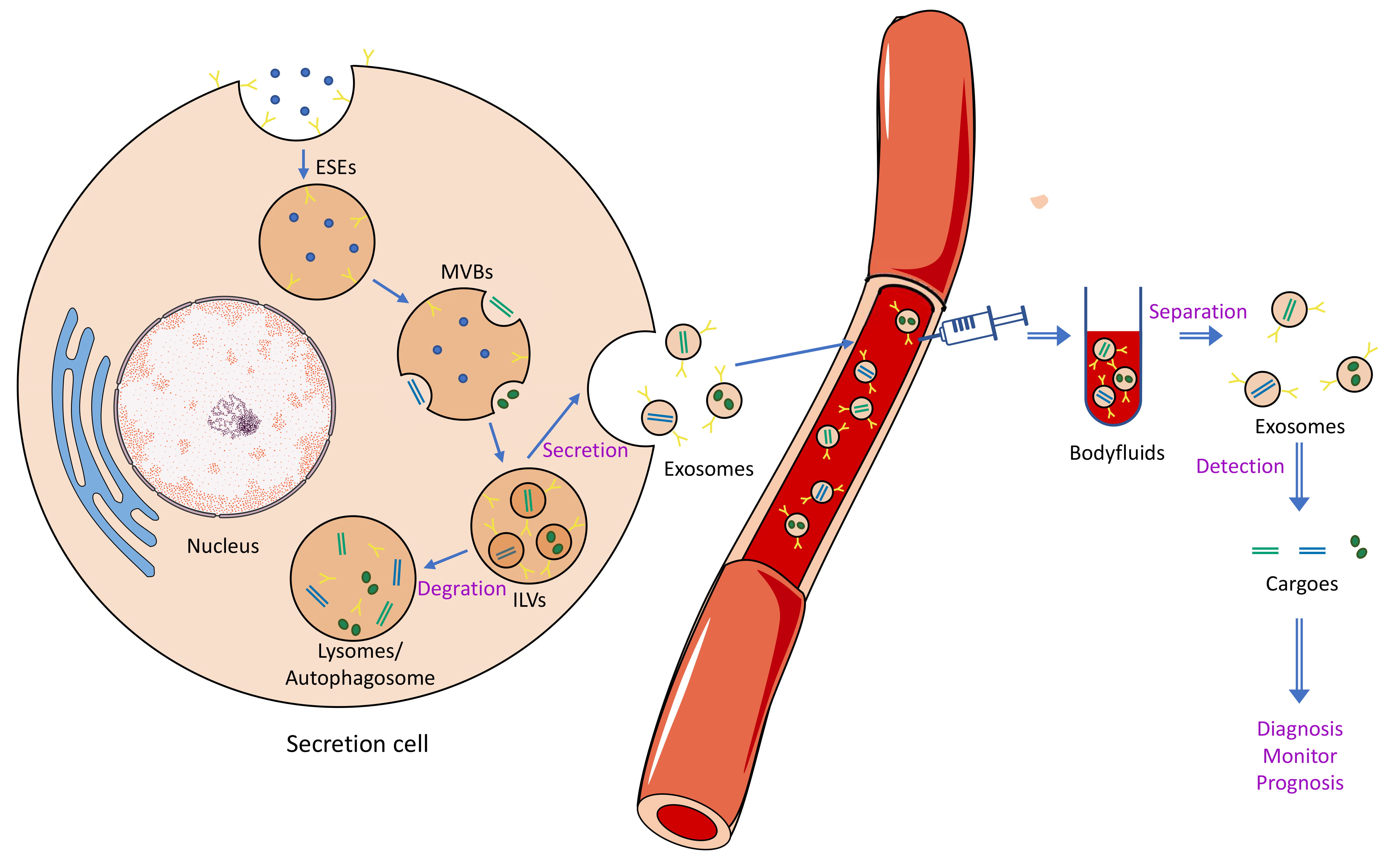
The formation, contents, and size of exosomes are heterogeneous. It is necessary to isolate and enrich exosomes from different body fluids. To date, several methods have been applied to isolate exosomes; however, the efficiency varies greatly (Table 1, Ref. [36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51]). The conventional common methods include centrifugation techniques, size-based techniques, capture-based techniques, and polymer-based techniques [35]. The ultracentrifugation technique, including differential ultracentrifugation and density gradient centrifugation, is the most common method used as the gold standard for isolation [52]. Precipitation techniques use a highly hydrophilic polymer to reduce solubility, and these techniques are the most commonly used to produce commercial kits. Some commercial kits have been developed to separate exosomes, such as the Total Exosome Isolation and ExoQuick™ Kit [53]. Now, a new exosome isolation method, microfluidic, has been developed to solve issues in traditional methods that can be sufficiently combined with size-based, immunoaffinity-based and dynamic separation. After isolation of the exosomes, the identification of exosomes should be analyzed by western blot detection of exosome-specific markers and transmission electron microscopy (TEM). Proteins and nucleic acids are meaningful for exosome content detection and specific biomarkers for diagnosis and prognosis prediction [54]. Enzyme-linked immunosorbent assay (ELISA) and western blot analysis are regularly and widely used to detect proteins [55]. The expression levels and kinds of exosomal nucleic acids can be detected and quantified by real-time quantitative reverse transcription PCR (RT–qPCR), microarray, and next-generation sequencing (NGS) [56]. Based on the extractability, exosomes may play an important role in liquid biopsy (Fig. 1).
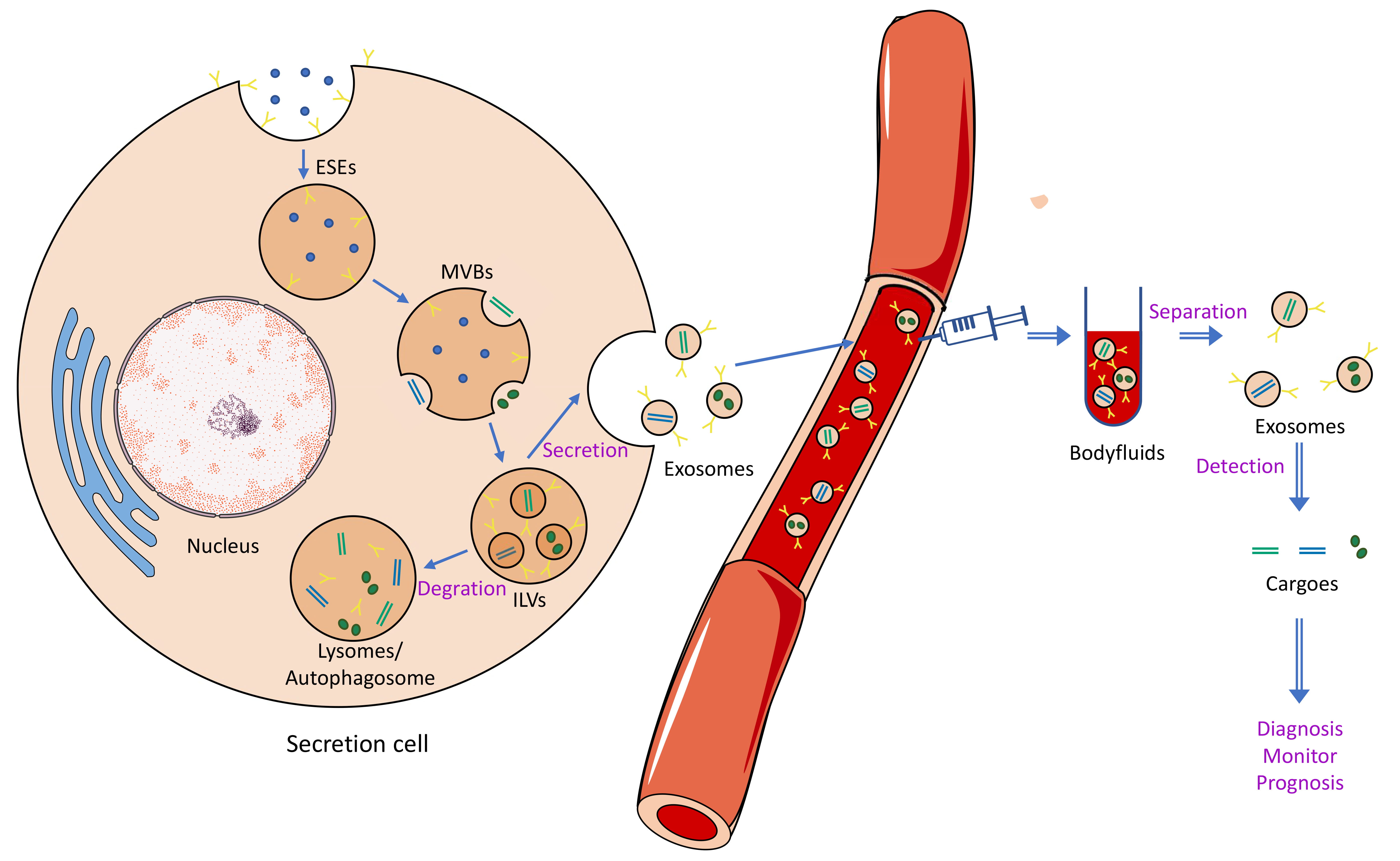 Fig. 1.
Fig. 1.Biological generation, secretion, and isolation of exosomes. Detection of exosomes in body fluids and analysis of their cargo can be used for liquid biopsy (diagnosis, monitoring, and prognosis).
| Separation technology | Principium | Advantages | Disadvantages | Commercial products | Ref | |
| Centrifugation techniques | Differential Ultracentrifugation | Density, shape, and size, larger and more dense particles sedimented out first | Widely used and well developed | Low purity, costly instruments, long time and large starting volumes of sample | - | [36, 37] |
| Density Gradient Centrifugation | Size and density and a preconstructed density gradient | High practicability and efficiency in separating exosomes from bodily fluids | Long time and low exosome recovery | - | [38, 39] | |
| Size-based techniques | Ultrafiltration | Size, molecular weight | Less time, size uniformity of yield and no special instrumentation | Loss of exosomes and low isolation efficiency | - | [40, 41] |
| Sequential Filtration | A series of filtration steps | Automation | - | ExoMir Kit (Bioo Scientific; Austin, TX, USA) | [42] | |
| Size Exclusion Chromatography (SEC) | Column packed with a porous stationary phase | Economical and non-destructive | Complicated | qEVoriginal (IZON), Exo-spin™ (Cell Guidance Systems) | [43, 44] | |
| Flow Field-Flow Fractionation (FFFF) | Sample subjected to parabolic flow and a crossflow (a flow perpendicular to the parabolic flow) making the separation | - | - | - | [45] | |
| Capture-Based techniques | Enzyme-Linked Immunosorbent Assay (ELISA) | Immunoaffinity, the antibodies for a specific antigen of interest attached to plate | High purity and Isolation of exosomes from a specific source | Sample pretreatment by other methods | - | [38] |
| Magnetic beads and immunoaffinity | Immunoaffinity, the antibodies for a specific antigen of interest attached to magnetic beads | low capacity | ExoRNeasy Serum/Plasma Kit (Qiagen, Germany), MagCapture™ Exosome Isolation Kit PS (Wako) | [46] | ||
| Polymer precipitation | Polyethylene Glycol (PEG) Precipitation | The water molecules absorbed by PEG | Speediness, simplicity, and little technical expertise or expensive equipment | Lack of selectivity | ExoQuick (System Biosciences, USA), Total Exosome Isolation Kit (Thermo Fisher Scientific, USA), Total Exosome Isolation Reagent (Invitrogen, USA), ExoPrep (HansaBioMed, Estonia), miRCURY Exosome Isolation Kit (Exiqon, Denmark), Exosome Purification Kit (Norgen Biotek, Canada) | [38, 41] |
| Lectin Induced Agglutination | Lectins binding to carbohydrates on the surface of exosomes | Simpleness, and little technical expertise | - | - | [47] | |
| Microfluidics-based techniques | Size-based Microfluidics | Size | Fast separation | Complicated equipment | ExoTIC device, ExoChip, ExoSearch Chip | [48, 49, 50] |
| Immunoaffinity-based | Immunoaffinity | |||||
| Acoustic Nanofilter | Ultrasound waves | Simplicity, quickness, tunability, and low starting volume | - | - | [51] | |
| -: Not applicable. | ||||||
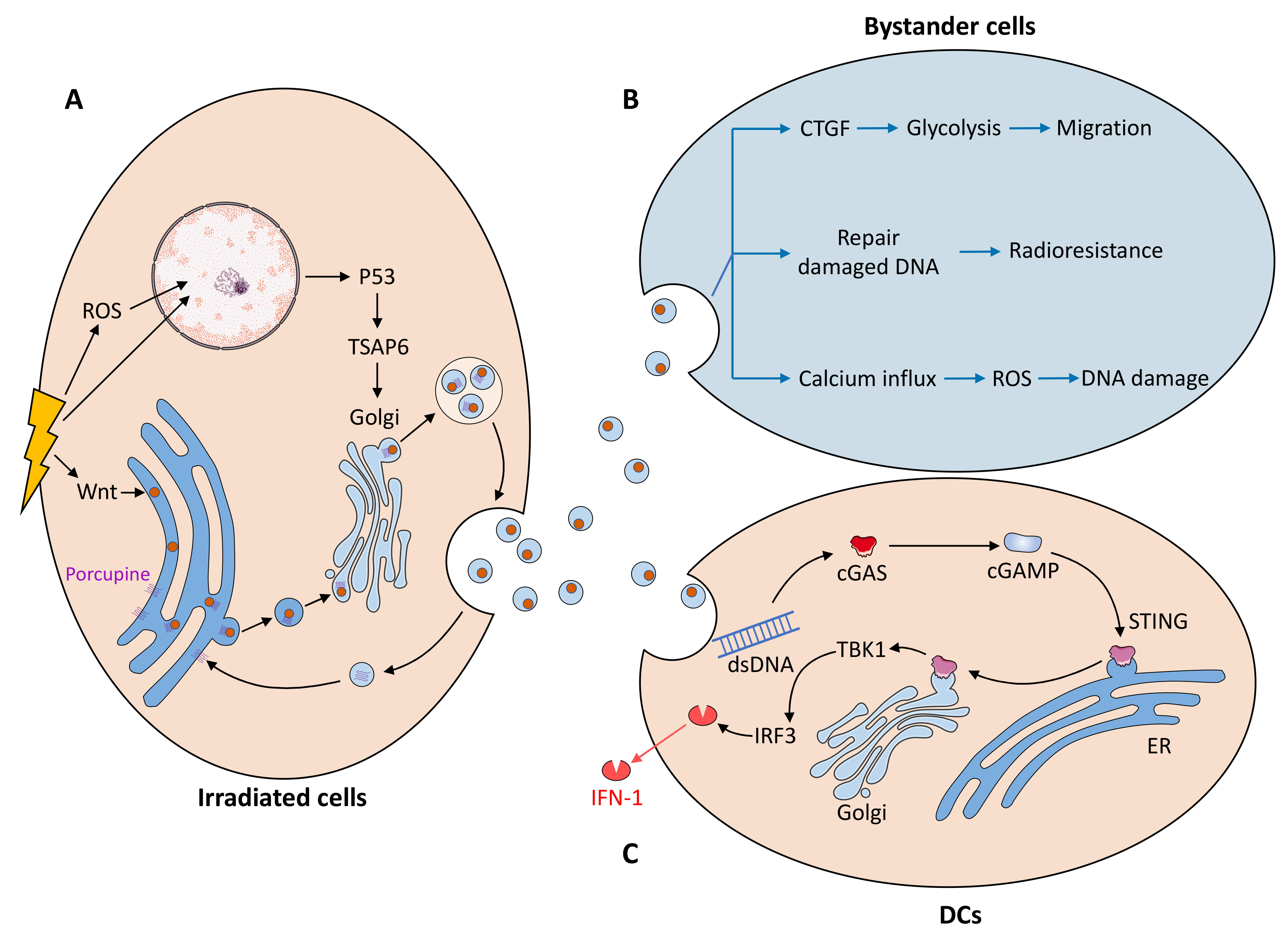
Radiation damages DNA directly in the nucleus [57, 58] and indirectly by promoting intracellular reactive oxygen species (ROS) production and altering aerobic metabolism (Fig. 2) [58]. Radiation-induced DNA damage activates the p53/TSAP6 axis and increases exosome biogenesis [59, 60]. TSAP6 is a p53-induced product of transmembrane proteins [61] that is closely biologically linked to the trans-Golgi network (TGN) compartment responsible for exosome biogenesis [60]. Certain exosome cargoes bud on the TGN-associated membrane and release exosomes after fusion with the plasma membrane [62, 63]. TSAP6 may facilitate this process and increase exosome secretion.
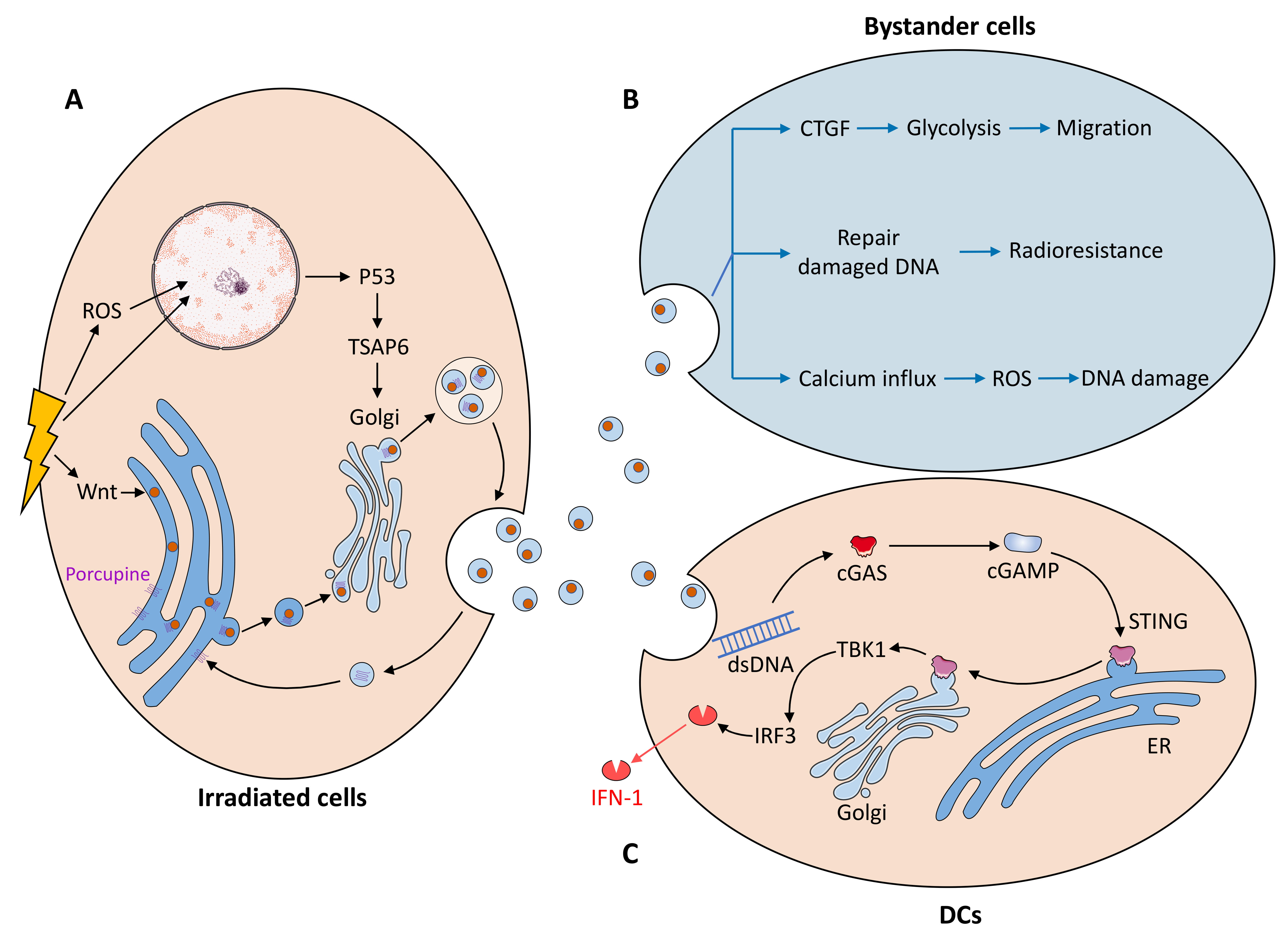 Fig. 2.
Fig. 2.The mechanism by which radiation affects exosomes. (A) The mechanism of increased exosome secretion by radiation cells is mainly the activation of the P53/TSAP6 axis and Wnt pathway after DNA damage. Radiated cells affect nonradiated cells through exosome exchange mainly by (B) promoting migration, radioresistance and DNA damage in recipient cells through bystander effects and (C) promoting IFN-I secretion by dendritic cells through the cGAS/STING axis in the tumor microenvironment.
Irradiation may increase exosome secretion from human endothelial cells by inducing Wnt signaling [64]. Most genes involved in the typical Wnt pathway, such as Wnt1, 2b, 3, 8a, 11, and 16, are induced in irradiated cells [64]. After Wnt enters the endoplasmic reticulum (ER), it is processed by palmitoylation of Porcupine protein [65] and transported from the ER to the Golgi apparatus after binding to Wntless (Wls) [66]. The Wnt-Wls complex from the Golgi apparatus is included in the generation of MVBs [67], which fuse with the plasma membrane, leading to the release of Wnt-containing exosomes [68]. WLS is reverse transported to the Golgi apparatus and ER for another Wnt secretion [66].
Radiation may affect exosome secretion by regulating autophagy [64]. Increased LC3 levels and decreased P62 levels in irradiated human umbilical vein endothelial cells (HUVECs) induce autophagic responses while increasing exosome secretion [64]. However, it has been shown that the overexpression of LC3 and induction of autophagy direct MVBs to the autophagic pathway and reduce exosome release [69]. Therefore, the relationship among radiation, autophagy, and exosome release remains to be further investigated.
Irradiated cells transmit signals to nonirradiated cells through nontargeting
effects (NTEs). Irradiated NTEs include three major effects: abscopal, cohort,
and bystander effects [70, 71]. The most prominent effect of radiation on exosome
function is the radiation-induced bystander effect (RIBE). Irradiated
glioblastoma-derived exosomes enhanced the migration of unirradiated receptor
cells via RIBEs [72]. In bystander cells, CTGF protein expression was enhanced in
unirradiated cells [72]. CTGF protein may promote the migratory phenotype of
receptor cells by binding integrin
Radiotherapy-mediated immune activation is associated with the paracrine secretion of exosomes. Irradiated cancer cells produce exosomes containing cytoplasmic double-stranded DNA that is released into the tumor microenvironment [79]. Upon entry into dendritic cells, exosomal dsDNA activates the dendritic cell production of IFN-I via the cyclic GMP-AMP synthase (cGAS)/interferon gene (STING) pathway, thereby activating immune cells and immune responses [79, 80]. cGAS senses cytoplasmic dsDNA and catalyzes the production of the second messenger cGAMP [81]. cGAMP interacts with STING for processing and transport from the ER to the Golgi apparatus [82, 83] and then activates the downstream protein kinase TBK1, which phosphorylates the transcription factor interferon regulatory factor 3 (IRF3) and induces the expression of IFN-I [84]. Cancer-associated fibroblast (CAF)-derived exosomes play an important role in the regulation of cancer cells [85]; however, it has been shown that radiation does not cause substantial changes in CAF-EV secretion rates or EV protein content [86].
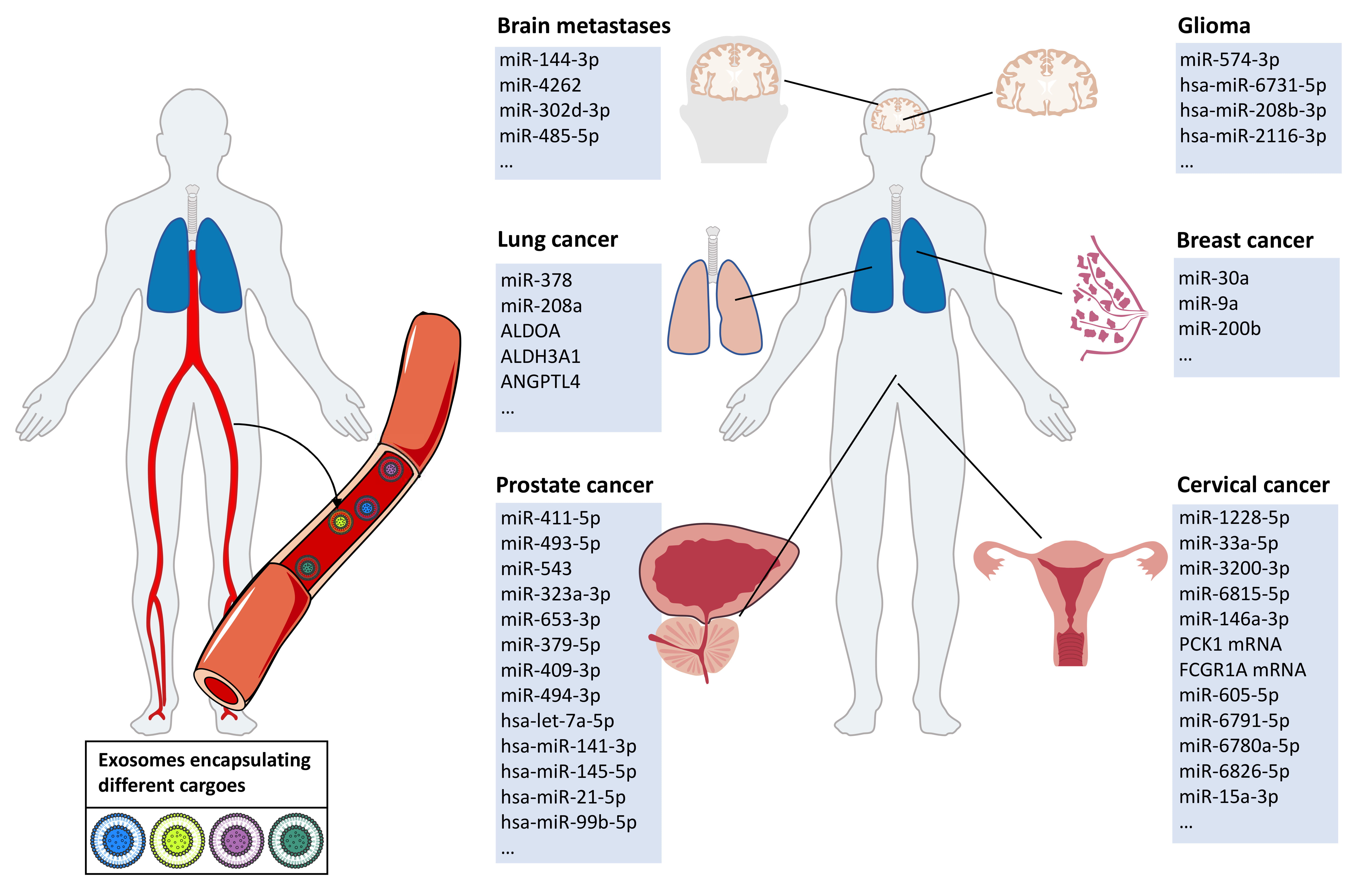
Exosomal cargoes in body fluids are significantly altered after tumor radiotherapy. The exosomal cargoes altered after tumor radiotherapy mainly include exosomal miRNAs, proteins, and other substances, such as circRNAs and lipids (Fig. 3). These cargoes are involved in pathways in the cancer microenvironment that regulate the aggressive phenotype of cancer (Table 2, Ref. [16, 17, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100]). On the one hand, exosomes after radiotherapy suppress aggressive phenotypes of tumor cells, such as proliferation and migration; on the other hand, exosome-mediated signaling after irradiation promotes the aggressive phenotype of cancer cells, such as drug resistance and radiotherapy resistance, by establishing communication between tumor cells and their microenvironment.
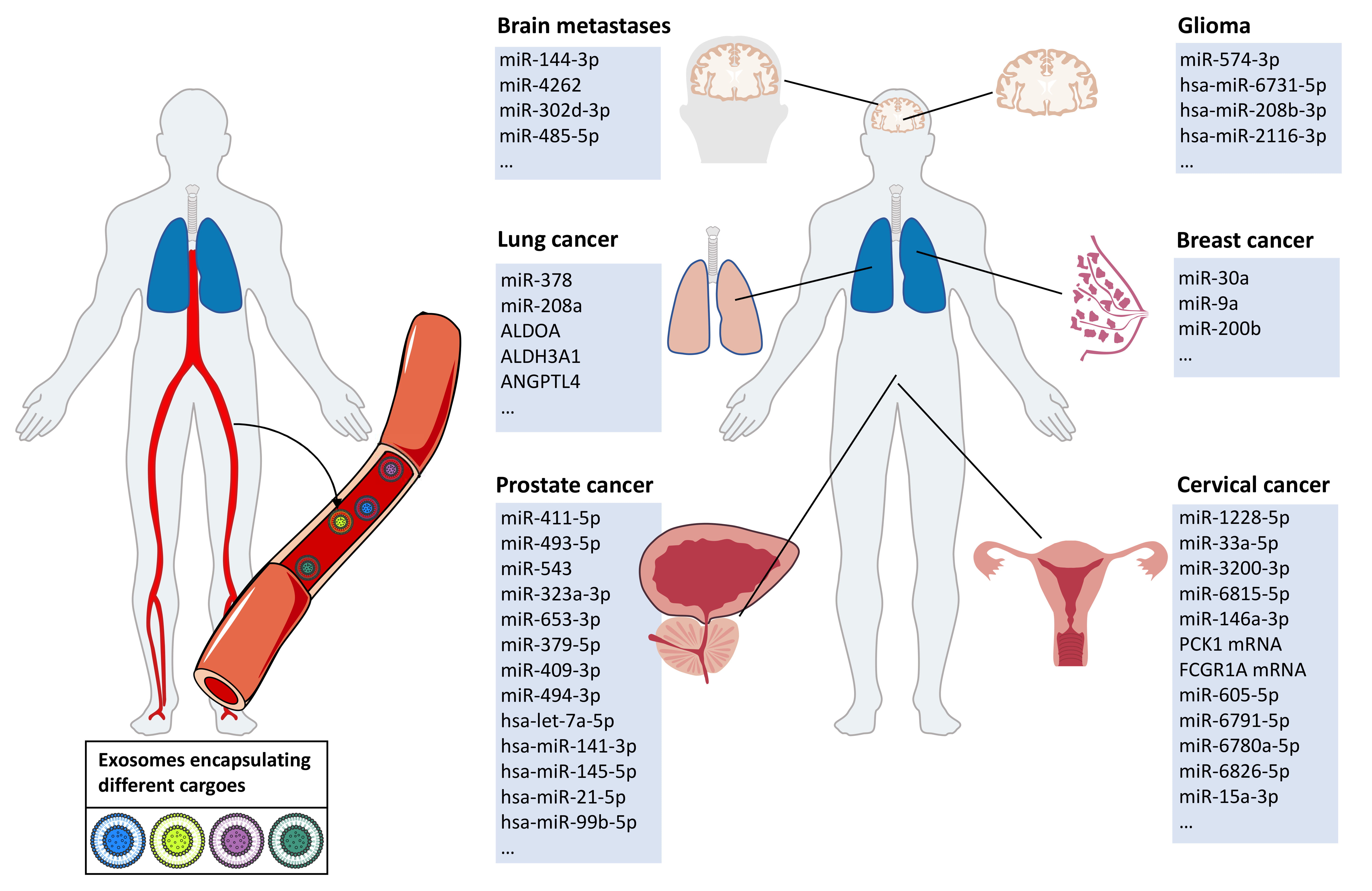 Fig. 3.
Fig. 3.After tumor radiotherapy, exosomal cargoes in the body fluids of tumor patients are altered. Tumor patients undergoing radiation therapy experience specific changes in the exocrine body fluids. We summarized the specific exosomal cargoes that appear in body fluids after radiotherapy in patients with brain metastases, gliomas, lung cancer, breast cancer, prostate cancer, and cervical cancer.
| Tumor | Body fluids | Type of cargoes | Cargoes | Signaling pathways | Effect on tumor aggressive phenotype | Ref |
| Brain metastases | Plasma | miRNA | miR-144-3p, miR-4262, miR-302d-3p, miR-485-5p, etc | - | Inhibits cancer cell proliferation, invasion, migration, and drug resistance | [87] |
| Glioma | Serum | miRNA | miR-574-3p, hsa-miR-6731-5p, hsa-miR-208b-3p, hsa-miR-2116-3p, etc | p53, TGF- |
Regulates cell proliferation, angiogenesis, invasiveness, radioresistance, and apoptosis | [88] |
| NSCLC | Serum | miRNA | miR-378 | Inhibits FOXG1 or interacts with heme oxygenase-1 | Promotes cell proliferation, migration, angiogenic capacity and radioresistance | [17] |
| Breast cancer | - | miRNA | miR-9a | Inhibits E-calmodulin | Promotes EMT and metastasis of breast cancer cells | [89] |
| - | miRNA | miR-200b | Targeting c-MYB | Induces EMT and tamoxifen resistance in breast cancer cells | [89] | |
| - | miRNA | miR-30a | Targeting Notch1 or Eya2 | Inhibit breast cancer cell viability, migration and invasion, and induce apoptosis | [89] | |
| Prostate cancer | Serum | miRNA | miR-411-5p, miR-493-5p, miR-543, miR-411-5p, miR-323a-3p, miR-543, miR-653-3p, miR-379-5p, miR-409-3p, miR-494-3p, etc | Inhibits RAS, PI3K/AKT and MAPK pathways | Promotes proliferation, migration, invasion and drug resistance of prostate cancer cells | [16] |
| Serum | miRNA | hsa-let-7a-5p, hsa-miR-141-3p, hsa-miR-145-5p, hsa-miR-21-5p, hsa-miR-99b-5p | - | Increases platinum drug chemoresistance in metastatic prostate cancer cells | [90] | |
| Serum | miRNA | miR-208a | Targeting p21; down-regulating the expression of apoptotic proteins PARP1 and Bax and up-regulating anti-apoptotic proteins Bcl-2 | Promotes cell proliferation and induces radioresistance and inhibits apoptosis of cancer cells | [91] | |
| Cervical cancer | Plasma | miRNA | miR-1228-5p, miR-33a-5p, miR-3200-3p, miR-6815-5p, miR-146a-3p, etc | PI3K/AKT/mTORsignaling pathway | Slows down the development of cervical cancer cells | [92] |
| Plasma | miRNA | miR-605-5p, miR-6791-5p, miR-6780a-5p, miR-6826-5p | Targeting bcl-xL, tumor protein D52 | Induces apoptosis and enhances radiosensitivity in cervical cancer | [92] | |
| Plasma | mRNA | PCK1, FCGR1A | miR-146a-3p/CCNO/PCK1 axis, miR-1228-5p/SLAMF1/PCK1 axis, PDE3A/PCK1 axis, miR-1228-5p, miR-3200-3p/FCGR1A axis | - | [92] | |
| HNSCC | - | protein | EIFs, PSMs, RPLs, RPSs, etc | Involved in transcription and translation, regulation of cell cycle/division, cell signaling | Inhibit cell proliferation | [93] |
| Brain metastases | Plasma | protein | Integrin |
- | - | [94] |
| Lung cancer | - | protein | ALDOA, ALDH3A1 | Promotes glycolysis | Promote the growth and motility of lung cancer cells | [95] |
| - | protein | ANGPTL4 | - | Promotes migration of cancer cells, angiogenesis of HUVEC | [96] | |
| Colorectal cancer | Serum | circRNA | circ_0067835 | circ_0067835/miR-296-5p/IGF1R axis | Regulates cell proliferation, tumor growth and resistance to radiation therapy | [97] |
| Pancreatic cancer | Plasma | circRNA | hsa_circ_0000284, etc | Lysine degradation | Promotes liver metastasis and drug resistance | [98] |
| HNSCC | Serum | Lipids and metabolites | Glycerol and cholesterol, 1-hexadecanol, citric acid, 4-hydroxybenzoic acid and propylene glycol | Fatty acid oxidation and ketone body metabolism | - | [99] |
| - | Plasma | Lipids and metabolites | Triglycerides, platelet activating factor, carnitine and C-16 sphingomyelin; palmitamide | - | - | [100] |
| -: Not applicable. Abbreviations: TGF- | ||||||
Systematic and personalized radiotherapy is the main treatment modality for brain metastases [101]. Clinical studies have shown that patients with brain metastases develop radiation necrosis leading to treatment failure after radiotherapy [102]. The optimal radiation dose should ensure effective tumor control without resulting in an adverse prognosis [103]. The changes that occur in exosomes after radiotherapy in patients with brain metastases may provide information related to the efficacy of tumor radiotherapy. Thirty-five differentially expressed miRNAs were identified in the plasma exosomes of brain metastasis patients before and after radiotherapy, of which 16 were downregulated and 19 were upregulated [87]. Analysis of their target genes and the KEGG pathways showed that miR-144-3p, miR-4262, miR-302d-3p, and miR-485-5p had research implications. miR-144-3p expression was upregulated after radiotherapy. miR-144-3p is associated with the progression of several cancers, inhibiting cervical cancer [104], gastric cancer [105], and non-small-cell lung cancer (NSCLC) [106] in terms of proliferation, invasion, and migration. The other three miRNAs are associated with promoting tumor proliferation [107], migration [108], and drug resistance [109]. The downregulation of their expression after radiotherapy may play a role in blocking tumor invasion.
Currently, a combination of chemotherapy combined with radiotherapy is mainly
applied to glioma patients [110, 111, 112]. Although radiotherapy has adverse effects,
it is still an indispensable treatment when the risk of glioma progression is
higher than the risk of possible radiotherapy toxicity [113]. It is crucial to
detect the effects of radiotherapy on glioma. In serum exosomes of glioma
patients, miRNAs were differentially expressed before and after radiotherapy,
including 18 upregulated and 16 downregulated miRNAs [88]. Statistical analysis
showed that the predicted miRNA target genes were involved in the p53,
TGF-
Radiotherapy is a safe and efficient therapy for patients without or with few metastatic sites [122]. Radiotherapy for NSCLC patients has a lower perioperative mortality rate than surgical treatment [123]. However, radioresistance is the biggest problem of radiotherapy failure and poor prognosis [124, 125]. Liquid biopsy of exosomes before and after radiotherapy not only provides information on the efficacy of radiotherapy but also provides a research direction on the mechanism of radiation resistance in NSCLC. Serum levels of exosomal miR-378 in patients with NSCLC were higher than those in healthy people and significantly decreased after radiotherapy [17]. miR-378 promotes cell proliferation, migration, and angiogenic capacity in NSCLC, possibly by inhibiting FOXG1 [126] or by interacting with heme oxygenase-1 [127]. miR-208a, an upregulated serum exosome in NSCLC patients after radiotherapy, is transferred to lung cancer cells, targets p21 to promote cell proliferation, and induces radioresistance. Moreover, in vitro experiments showed that miR-208a promoted the growth of cancer cells by downregulating apoptotic proteins and upregulating antiapoptotic proteins. The expression of PARP1 and Bax was downregulated, and the expression of Bcl-2 was upregulated. The expression of apoptosis-related proteins was significantly increased after transfection with miR-208a inhibitors [91].
Clinical research on breast cancer (BC) radiotherapy has focused on two aspects: first, the development of BC radiotherapy [128] to improve the efficacy of radiotherapy [129]; and second, improving the prognosis of BC after radiotherapy to address the problems of recurrence [130] and secondary malignancies [131]. Basic research on BC radiotherapy, especially with the help of exosomes as a detectable tool, would be an interesting research direction. The transfer of exosomal miRNAs from irradiated BC cells to unirradiated cells changes BC invasiveness [89]. After radiotherapy, miR-30a and miR-9a were upregulated, and miR-200b was downregulated. miR-9 inhibits E-calmodulin in BC cells, promoting epithelial–mesenchymal transition (EMT). Its silencing inhibits the metastasis of BC [132]. Reduced expression of miR-200b leads to increased c-MYB, which induces EMT as well as tamoxifen resistance in BC cells [133]. miR-9a upregulation and miR-200b downregulation both promote EMT in BC cells. miR-30a upregulation inhibits BC cell viability, migration, and invasion and induces apoptosis, possibly by targeting Notch1 [134] or Eya2 [135]. This shows that the regulation of the aggressive cancer phenotype by exosomes after radiotherapy is a complex network with synergistic and negative effects of exosomal cargo rather than a single regulatory pathway.
Radiotherapy for prostate cancer patients has demonstrated good biochemical control rates and acceptable toxicity [136, 137], but several studies have shown that patients suffer from radiation resistance and recurrence [138]. The study of the underlying mechanisms of radiotherapy for prostate cancer will help to reduce radiation resistance and explore better therapeutic approaches [139, 140]. The use of exosomes and miRNAs in prostate cancer radiotherapy will help to develop personalized radiotherapy for prostate cancer patients [141], showing the potential of research on liquid biopsy before and after patient radiotherapy. Radiotherapy induces the differential expression of serum exosomal miRNAs in prostate cancer patients. In the serum of prostate cancer patients, 57 exosomal miRNAs were remarkably changed after carbon ion radiotherapy (CIRT) [16]. These exosomes inhibit the RAS, PI3K/AKT, and MAPK pathways in recipient cells. Among them, miR-411-5p, miR-493-5p, and miR-543 inhibit the RAS signaling pathway. Activation of the Ras gene promotes the migration, invasion, and drug resistance of prostate cancer cells [142, 143], and inhibition of Ras activation suppresses proliferation and invasion [144]. miR-411-5p, miR-323a-3p, miR-543 and miR-653-3p block the MAPK pathway. miR-379-5p, miR-409-3p, miR-493-5p and miR-494-3p centrally regulate the PI3K-AKT axis. Activation of the MAPK signaling pathway and the PI3K-AKT axis facilitates prostate cancer growth, migration, and invasion [145, 146], while inhibition of this pathway and axis showed the opposite effects [147, 148]. Activation of the PI3K-AKT axis is involved in the chemoresistance of prostate cancer [149, 150]. Another study identified five differentially expressed miRNAs in the serum exosomes of prostate cancer patients, including hsa-let-7a-5p, hsa-miR-141-3p, hsa-miR-145-5p, hsa-miR-21-5p and hsa-miR-99b-5p [90]. Among them, hsa-let-7a-5p expression is upregulated after radiation and may increase platinum drug chemoresistance in metastatic prostate cancer cells [151].
Recurrence and distant metastases after radiotherapy in patients with cervical cancer are the main causes of poor prognosis and reduced survival [152, 153]. Exosomal liquid biopsy has the potential to provide information on the prognosis of radiotherapy in cervical cancer patients. Plasma exosomes were monitored in patients with cervical cancer before and after concurrent chemoradiotherapy (CCRT). miRNAs in patients with early progression (EP) suggested that inflammation was not resolved. The levels of miR-1228-5p, miR-33a-5p, miR-3200-3p and miR-6815-5p decreased, and the level of miR-146a-3p increased [92]. These miRNAs are involved in the EP of cervical cancer by regulating mRNAs. According to the analysis, PCK1 mRNA was upregulated by miR-1228-5p, miR-33a-5p, and miR-146a-3p, and FCGR1A mRNA was downregulated by miR-1228-5p and miR-3200-3p. miR-605-5p, miR-6791-5p, miR-6780a-5p and miR-6826-5p levels were increased, and miR-15a-3p levels were decreased in association with the extent/stage of cervical cancer metastasis [92]. miR-15a-3p targets Bcl-xL to induce the apoptosis of cervical cancer cells [154] and targets tumor protein D52 to reduce the radioresistance of cervical cancer [155]. Its reduction may increase tumor aggressiveness.
Exosomal proteins are altered after tumor radiation therapy, and their role is mainly to regulate the proliferation of cancer cells. Head and neck squamous cell carcinoma (HNSCC) cells secrete exosomes that are severely altered in terms of the exosome cargo after exposure to ionizing radiation [93]. Compared to controls, 236 new proteins and 69 missing proteins were found in the irradiated cell-derived exosomes. Upregulated exosomal proteins participate in transcription and translation, the regulation of the cell cycle, and cell signal transduction [93]. This implies that cell cycle arrest and the resulting blockage of transcription, translation, and cell division would be the primary cellular response to radiation. Proteins may also modulate cancer cell proliferation by altering cell adhesion. Actin L-plastin is strongly positive in the exosomes of nonirradiated prostate adenocarcinoma cells, whereas its expression is decreased after radiation [156]. In prostate cancer cells, L-plastin expression and phosphorylation contribute to proliferation and metastasis [157, 158]. The mechanism may be as follows: first, L-plastin is phosphorylated to become biologically active and then is packed into exosomes and released into the extracellular matrix; this process promotes the cross-linking of filamentous actin, leading to the formation of cell bundles; this cytoskeletal biological response is beneficial for cell adhesion, increasing cell proliferation [156].
Exosomal proteins may contribute to the distant metastasis and relapse of cancer. Irradiated lung cancer cell–derived exosomes promote tumor aggressiveness in nonirradiated cells [95, 96]. In analyzing the altered proteins in exosomes, it was found that elevated expression of the metabolic enzymes ALDOA and ALDH3A1 promotes glycolytic activity in recipient cancer cells, which in turn promotes the growth and motility of other lung cancer cells [95]; moreover, exosomal angiopoietin-like 4 (ANGPTL4)-derived proteins contribute to the migration of recipient cancer cells and the angiogenesis of HUVECs, increasing tumor aggressiveness [96].
circRNA is also an important exosomal cargo that is altered after radiation. Serum exosomal circ_0067835 is upregulated after radiotherapy in colorectal cancer (CRC) patients, enhancing radiation resistance through the circ_0067835/miR-296-5p/IGF1R axis [97]. Upregulation of circ_0067835 significantly downregulated miR-296-5p and upregulated IGF1R mRNA. With knockdown of circ_0067835, cell proliferation was inhibited, tumor growth was attenuated in vivo, and cell radiosensitivity was enhanced [97]. After irradiation of pancreatic cancer cells, 196 differentially expressed circRNAs were identified, consisting of 182 upregulated and 14 downregulated circRNAs, and KEGG analysis indicated that lysine degradation is an important pathway involved in these circRNAs [98]. Lysine catabolism produces acetyl coenzyme A, which activates Wnt signaling to promote the self-renewal of tumor-initiating cells; it also produces glutamate, which regulates the redox status of TICs and promotes liver metastasis and drug resistance in colon cancer [159]. This suggests that alterations in circRNA in exosomes after irradiation may be involved in biological processes that regulate tumor aggressiveness.
Lipids and lipid metabolism are also involved in the alteration of exosomal cargo after radiotherapy, which affects tumor growth, proliferation, and differentiation [160]. In the circulating exosomes of HNSCC patients treated with radiotherapy, 1-hexadecanol was increased, citric acid, 4-hydroxybenzoic acid, and propylene glycol were significantly decreased, and glycerol and cholesterol showed a moderate reduction. These metabolite-related pathways include fatty acid oxidation and ketone body metabolism [99]. Twenty-four significantly altered metabolites appeared in the plasma EVs of mice receiving cranial irradiation. Markers of the systemic inflammatory response were significantly enriched, including triglycerides, platelet-activating factor, carnitine, and C-16 sphingomyelin; palmitamide was significantly downregulated [100]. There was no such change in these metabolites during whole-plasma analysis, which means that studies of specific exosomes are necessary.
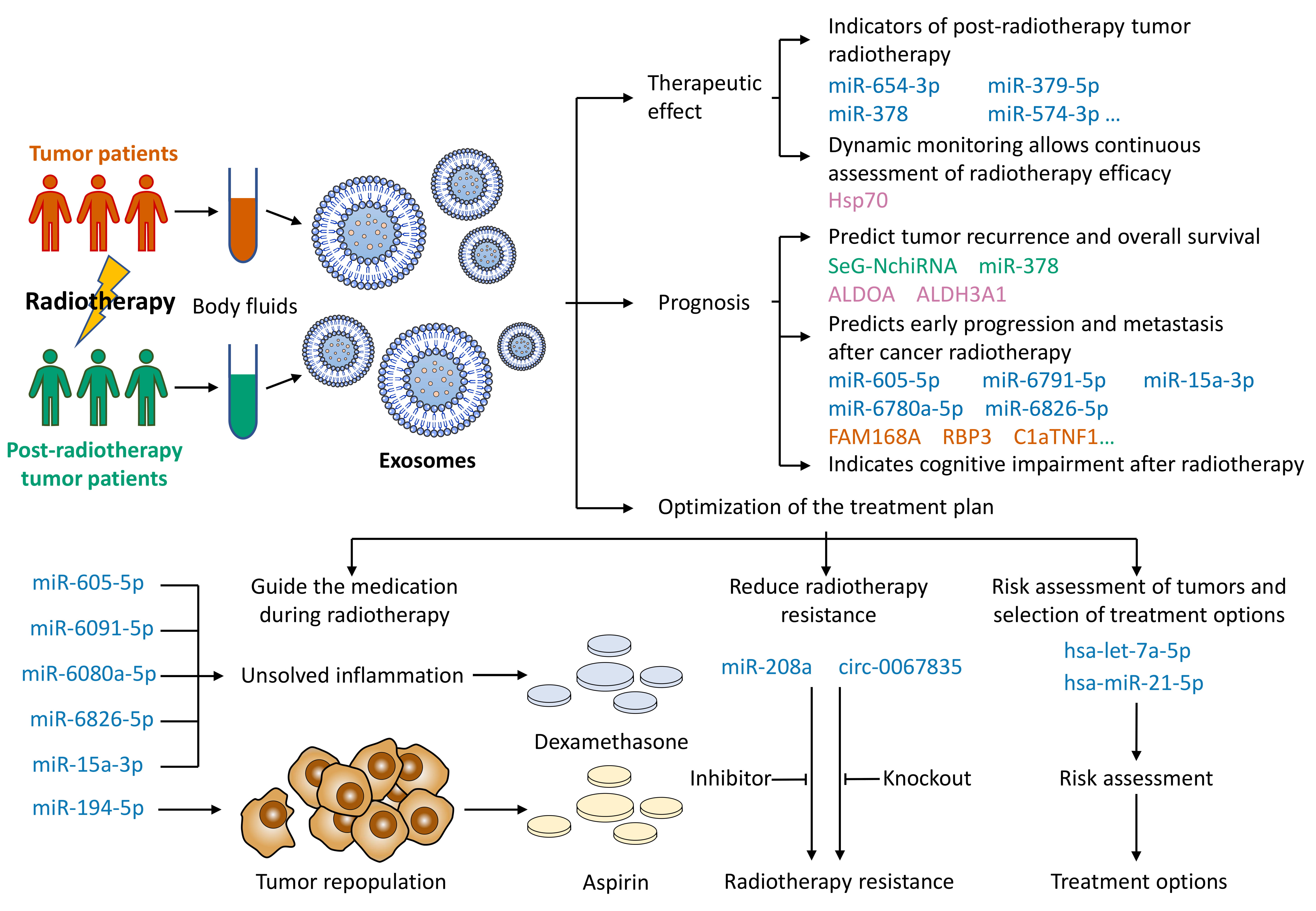
Based on the properties of exosomes, key exosomal cargoes after tumor radiotherapy can be used as diagnostic and prognostic biomarkers of cancer. Exosomes after tumor radiotherapy can be used for liquid biopsy, and the main applications include (1) predicting tumor radiotherapy efficacy, (2) predicting tumor prognosis, and (3) optimizing tumor treatment plans (Fig. 4).
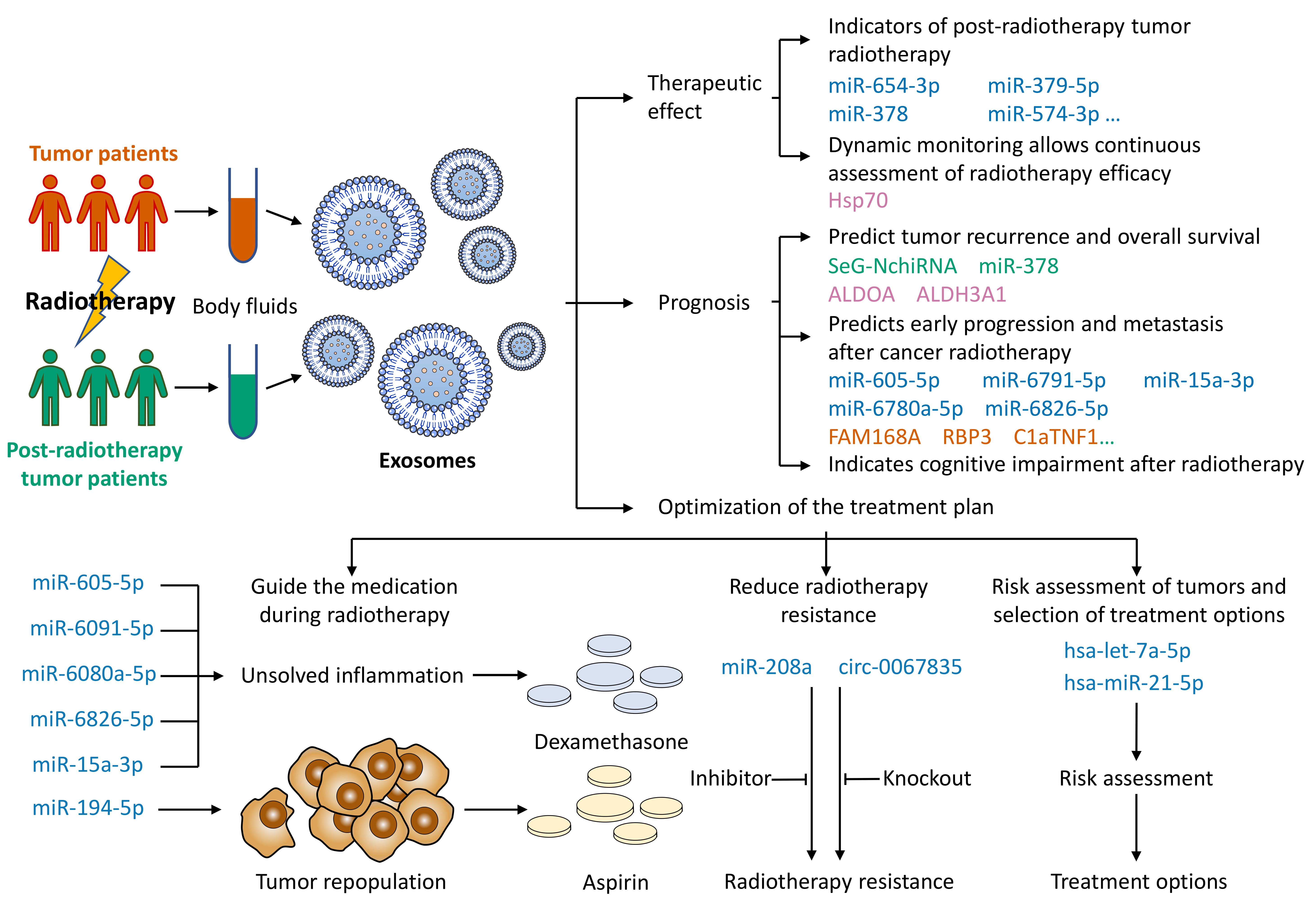 Fig. 4.
Fig. 4.Use of exosomes for liquid biopsy after cancer radiotherapy. Body fluids of tumor patients are extracted before and after radiotherapy. The body fluids are analyzed for exosomal cargoes. The cargo-specific exosomes can help predict the patient’s radiotherapy efficacy and prognosis and optimize the regimen for oncology treatment.
The miRNAs in serum/plasma exosomes are altered after radiation therapy in tumor patients. The alteration of specific miRNAs can be used as an indicator of tumor radiotherapy to predict efficacy. In prostate cancer patients, 57 miRNAs in serum exosomes were obviously changed after the application of CIRT. The high expression of eight specific miRNAs indicates that CIRT is effective. Among the miRNAs, the expression of miR-654-3p and miR-379-5p in serum exosomes is positively correlated with the curative effect, suggesting that these two miRNAs are prospective noninvasive biomarkers for forecasting the effect of CIRT on prostate cancer [16]. The greater the change in expression is, the better the therapeutic effect [16]. The level of serum exosomal miR-378 in NSCLC patients decreased significantly after radiotherapy and may be an indicator of the radiation effect [17]. Changes in plasma exosomal miRNAs were observed in glioma patients before and after radiotherapy, in which miR-574-3p decreased significantly after radiotherapy [88]. miR-574-3p was found in prostate cancer [161], head and neck cancer [162], osteosarcoma [163] and hepatocellular carcinoma [164], and many other patients with significantly increased serum/plasma exosomes, and its high expression was related to a poorer prognosis [162]. This finding indicates that exosomal miR-574-3p may be an important candidate biomarker for monitoring the efficacy of tumor radiotherapy.
Dynamic monitoring of plasma exosomes allows the continuous assessment of radiotherapy efficacy. Plasma-derived exosomes from NSCLC patients have significantly higher levels of Hsp70 than exosomes from healthy subjects. In patients irradiated at approximately 20 Gy and after completing radiotherapy, Hsp70 concentrations decreased with the decrease in surviving tumor mass [165]. Therefore, the liquid biopsy of exosomal Hsp70 levels during radiotherapy in oncology patients may be used for the risk assessment estimation and monitoring of treatment outcomes in NSCLC.
Exosomes can be used to predict and detect tumor recurrence and overall
survival. The salivary exosome GOLM1-NAA35 chimeric RNA (seG-NchiRNA) shows great
potential in evaluating the response to chemotherapy-radiotherapy in esophageal
squamous cell carcinoma (ESCC) patients. Two months after the initiation of
treatment, seG-NchiRNA levels increased significantly and continued to rise. The
detection of disease progression is earlier than that with radiological
assessment. The seG-NchiRNA level can be a biomarker to evaluate the effect of
ESCC radiotherapy, constituting a noninvasive, low-cost screening platform for
ESCC. It can also be used for postoperative monitoring, evaluating the response
to radiotherapy treatment and detecting tumor recurrence [18]. In NSCLC patients
with high serum exosomal miR-378 expression, the overall survival was poor [17].
Clinical survival analysis showed that in lung cancer patients, the expression of
the exosomal protein ALDOA or ALDH3A1 after radiotherapy was closely connected
with a worse prognosis [95]. High levels of circulating exosomal integrin
Exosomes can be used to predict the early progression and metastasis of cancer after radiotherapy. The increase and decrease in plasma exosomal miRNA levels (such as miR-605-5p) and mRNA (FAM168A, RBP3, and C1QTNF1) were associated with the degree/staging of cervical cancer metastasis before and after CCRT and can be considered a prognostic marker for early progression and metastasis after CCRT in patients with cervical cancer [92]. Plasma exosomes were extracted from patients with brain metastases from lung adenocarcinoma or melanoma. The differential expression of miRNAs before and after radiotherapy may be a candidate biomarker for the early detection of brain metastases [87].
Exosomes may suggest cognitive impairment after radiotherapy. Glioma cell-derived exosomes injected into the hippocampus of mice after radiotherapy inhibited the proliferation of neural stem cells and neurosphere formation, causing more pronounced neurogenesis inhibition and cognitive impairment [167]. Since central nervous system exosomes can pass through the blood–brain barrier into the blood and be isolated from peripheral blood [168], specific metabolites may be present in plasma EVs from mice receiving cranial irradiation [100]. Plasma EVs may become a microinvasive biomarker for injury to the central nervous system from ionizing radiation and for the early identification of radiation-induced early-onset damage to cognitive impairment, thus facilitating early treatment.
Exosomal liquid biopsy is beneficial in guiding medication during radiotherapy for tumor patients. Decreased levels of miRNAs in cervical cancer patients with EP before and after simultaneous radiotherapy indicate unresolved inflammation, suggesting the need for more dexamethasone to control inflammation after the completion of radiotherapy [92]. Dying pancreatic cancer cell-derived exosomal miR-194-5p is significantly elevated after radiotherapy. miR-194-5p downregulates the transcription factor E2F3, which contributes to the repair of damaged tumor repopulating cells. Tumor repopulation is the main reason for the failure of radiotherapy. The use of aspirin can inhibit tumor repopulation after radiotherapy by suppressing exosome secretion [169].
Exosomes suggest radiotherapy resistance in tumor patients and can be used to reduce radiotherapy resistance and improve radiotherapy efficacy through targeted therapy. Lung cancer patients have increased exosomal miR-208a in lung cancer cells after radiotherapy, which increases radiotherapy resistance [91]; CRC patients have upregulated serum exosomal circ_0067835 after radiotherapy, which decreases radiotherapy sensitivity [97]. Serum exosomal miR-208a and circ_0067835, as potential therapeutic targets, can improve tumor treatment outcomes by transfecting miR-208a inhibitors to reduce radiotherapy resistance and knocking down circ_0067835 to enhance radiotherapy sensitivity.
Exosomes can be applied for the risk evaluation of tumors and selection of therapeutic options. In prostate cancer patients before and after radiotherapy, the expression of serum exosomes hsa-let-7a-5p and hsa-miR-21-5p was significantly different in the two risk groups (moderate and high), probably because the function and role of miRNAs in prostate cancer differ according to the risk category in response to radiation [90]. In prostate cancer patients, circulating exosomes after radiotherapy may be potential biomarkers for detecting radiotherapy assessment in prostate cancer patients and may help to optimize prostate cancer radiotherapy treatment planning.
The mechanism of the modulation of the aggressive tumor phenotype by altered exosomal cargo after tumor radiotherapy is more complex. As mentioned earlier, exosomes after radiotherapy both promote and inhibit aggressive tumor phenotypes. The exploration of the regulatory mechanisms will be a promising direction of research. Current studies on exosomal cargoes focus on exosomal miRNAs, proteins, and other substances, such as mRNA, circRNA, lipids and other exosomal cargoes. It will be an interesting research direction to investigate the circRNAs altered in exosomes after tumor radiotherapy and the regulatory relationships among circRNAs, miRNAs, and mRNAs.
Exosomal cargo after tumor radiotherapy may be a candidate biomarker for liquid biopsy, but it has some problems. (1) The current study only provided initial data, which is insufficient to draw definitive results, and the findings need to be studied and tested in a broader prospective cohort. The relatively small size of the patient cohort limits the number of variables in the statistical model. (2) There was no nonoperative control group in the operative group, and it would be unethical to randomize tumor patients into a treatment-free group. A large, multicenter, double-blind (i.e., confidentiality of exosome test results by clinical evaluators and confidentiality of clinical status by exosome testers) prospective study is needed to definitively validate the use of this biomarker for the clinical detection of tumors. (3) Many of the current studies of exosomal cargo are only on potential biomarkers. Further exploration of plasma exosomes and specific cargo molecules is needed to apply specific molecules as noninvasive biomarkers for tumor detection. (4) Further improvement and refinement of the detection techniques are needed. The volume and extraction of exosomal miRNA are relatively small; the quantitative and qualitative alterations of exosomes and their cargo molecules are associated with sampling intervals before and after radiotherapy. (5) Whether the liquid biopsy technique for one tumor type can be applied to other tumor types remains unknown. These questions will provide directions and ideas for subsequent research.
Recently, the potential of exosomes for liquid biopsy after tumor radiotherapy has become a hot topic. An increasing number of studies have focused on exosomal cargoes and their impact on tumor aggressiveness after tumor radiotherapy as well as their application in radiotherapy efficacy, prognosis, and optimal treatment planning. Since many potential biomarkers have not been identified and determined, there is an urgent need for more exosome-based biomarker studies. Whether they have high clinical use-value is also a direction that should be considered for future research.
YS and YY designed the research study. YS and BQ performed the research. LH and YL provided help and advice on figures and/or tables. YS and YZ analyzed the data. YS and BQ wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
This work was supported by the National Natural Science Foundation of China Youth Science Foundation Project (No. 81700941), the Key Research and Development projects in the Sichuan Province (No. 2020YFS0172), and the Strategic Cooperation Special Project Sichuan University & Luzhou City (No. 2021CDLZ-8).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.