1 Translational Bioscience, Human Health Therapeutics Research Centre, National Research Council Canada, Ottawa, ON K1A 0R6, Canada
2 Department of Biochemistry, Microbiology and Immunology, University of Ottawa, Ottawa, ON K1H 8M5, Canada
3 Center for Mitochondrial Medicine and Free Radical Research, Changhua Christian Hospital, 50046 Changhua, Taiwan
4 Institute of Clinical Medicine, School of Medicine, National Yang Ming Chio Tung University, 11221 Taipei, Taiwan
5 Researcher Emeritus, Human Health Therapeutics Centre, National Research Council Canada, Ottawa, ON K1A 0R6, Canada
Academic Editor: Josef Jampilek
Abstract
Background: Mitochondrial biogenesis occurs in response to chronic
stresses as an adaptation to the increased energy demands and often renders cells
more refractive to subsequent injuries which is referred to as preconditioning.
This phenomenon is observed in several non-neuronal cell types, but it is not yet
fully established in neurons, although it is fundamentally important for
neuroprotection and could be exploited for therapeutic purposes.
Methods: This study was designed to examine whether the preconditioning
treatment with hypoxia or nitric oxide could trigger biogenesis in
undifferentiated and differentiated neuronal cells (rat PC12 and human NT2 cells)
as well as in primary mouse cortical neurons. Results: The results
showed that both preconditioning paradigms induced mitochondrial biogenesis in
undifferentiated cell lines, as indicated by an increase of mitochondrial mass
(measured by flow cytometry of NAO fluorescence) and increased expression of
genes required for mitochondrial biogenesis (Nrf1,
Nrf2, Tfam, Nf
Keywords
- oxidative stress
- rat PC12 cells
- human NT2 cells
- differentiation
- antioxidant
- coenzyme Q10
- neuroprotection
Cells regulate their energy production, which is mainly carried out by the mitochondrial oxidative phosphorylation system (OXPHOS), according to their needs. Depending on the challenge, mitochondria respond to the energy demands by either subtle change in the activities of OXPHOS enzymes, by changing the expression of enzyme subunits or by increasing the number and size of the organelle, i.e., by biogenesis. For example, mitochondrial proliferation occurs in skeletal muscles in response to the increased contractile activity [1], in adipose tissue in response to adaptive thermogenesis [2], in cardiac myocytes in response to electrical stimulation [3] or hypothermia [4]. Mitochondrial biogenesis is also observed in human endothelial cells in response to hypoxic stimulation via vascular endothelial growth factor-Akt-dependent signaling [5], and in human HeLa cells in response to oxidative stress [6]. In clinical settings, the proliferation of abnormal mitochondria was observed in skeletal muscles of patients carrying defective OXPHOS due to the abnormalities of the mitochondrial genome [7]. This implies that reactive oxygen species (ROS), generated either exogenously or endogenously by the respiratory enzymes, might play a role in mitochondrial biogenesis.
Numerous studies, carried out in both heart and brain, revealed remarkable
abilities of these tissues to adapt to stress, a phenomenon known as
preconditioning, whereby a sublethal dose of stress induces the tolerance to the
subsequent lethal insults [8]. A variety of preconditioning stimuli, including
brief periods of ischemia, anoxia, hypothermia and pharmacological agents known
to impair mitochondrial function, i.e., iodoacetate (inhibitor of
glyceraldehyde-3-dehydrogenase), 3-nitropropionic acid (inhibitor of succinic
dehydrogenase) and diazoxide (a selective opener of ATP-sensitive mitochondrial
K
We asked whether preconditioning treatments, such as hypoxia and nitric oxide (NO), could evoke the same response, i.e., trigger mitochondrial biogenesis and increase tolerance to injuries, in undifferentiated and differentiated post-mitotic neuronal cells. The answer to this question bears relevance to designing better neuroprotective strategies. We utilized two different immortalized neuronal cell lines (rat pheochromocytoma PC12 and human teratocarcinoma NT2 cells) and applied well-established differentiation protocols induced by nerve growth factor (NGF) and all-trans-retinoic acid (RA), respectively, to generate terminally differentiated post-mitotic neurons that were arrested in the G0 state. Rat PC12 cells were derived from adrenal medulla and contains neuroblastic cells originating from the neural crest [11]. Upon exposure to NGF, PC12 cells can be differentiated into neuron-like cells with long processes (neuritic varicosities) containing neurosecretory vesicles; hence, these cells have been frequently used to study neuronal differentiation [12]. Human NT2 cells were originally isolated from lung metastasis of primary embryonal carcinoma of the testis. The tumor was xenografted onto a mouse from which the NTERA-2 cells, characteristic of a committed neuronal progenitor cell type, were obtained. These cells can be readily differentiated by treatment with RA into postmitotic neuron-like cells (NT2-N) of the central nervous system [13, 14]. The NT2 cells have been utilized in many studies related to neurogenesis, neurotoxicity and neuroprotection [13, 14, 15]. Due to their irreversible commitment to a stable neuronal phenotype, the NT2-N cells have also been tested in transplantation studies aimed to repair brain damage [16].
In this study, we used undifferentiated and NGF-differentiated rat PC12 cells, RA-differentiated human NT2-N and primary mouse cortical neurons to study the effects of preconditioning, induced by hypoxia and NO, on mitochondrial biogenesis. The results showed that undifferentiated PC12 cells responded to the preconditioning treatments by increasing mitochondrial copy number per cell and better tolerance to the subsequent oxidative stress, but the differentiated post-mitotic neurons did not engage mitochondrial biogenesis during such preconditioning treatments. However, mitochondrial biogenesis was taking place in both cell lines during their differentiation and transition to the post-mitotic state. Consistently, post-mitotic neurons had higher mitochondrial copy numbers per cell, higher steady-state levels of ATP and better tolerance to oxidative stress than their undifferentiated counterparts. To our knowledge this is the first comprehensive study that compared the mitochondrial biogenesis response and tolerance to oxidative stress in undifferentiated and differentiated neuronal cells subjected to the same preconditioning treatments.
PC12 cells were purchased from American Type Culture Collection (ATCC). The
cells were plated at a density of 1
NT2/D1 progenitor cells (Stratagene, La Jolla, CA, USA) were cultured in high
glucose Dulbecco’s modified Eagle’s medium (HG/DMEM, cat# 11965-092)
supplemented with 10% FBS. Cells were seeded at a density of 2
Primary cultures of mouse cortical neurons were prepared from CD1 mice (Charles
River, St. Constant, QC, Canada) in accordance with Canadian Council on Animal Care and
the procedures approved by the Institutional Animal Care Committee. Briefly, CD1
mice (postnatal day 3) were sacrificed by CO
The cells were plated and grown in complete medium for 24 hr under normoxic
conditions. For hypoxic preconditioning (HP), cells were incubated for 24 hr at
37
For nitric oxide (NO) preconditioning (NP), cells were treated for 6 days with
50
In some experiments, the preconditioning was performed in the presence of
antioxidants: PTS (polyoxyethanyl
For the induction of oxygen and glucose deprivation (OGD), cells were washed with PBS and switched to DMEM without glucose and supplemented with 10% horse serum and 5% FBS and incubated in the GasPak chamber for 16 hr as described above. The cells were then cultured again in the growth medium for 24 hr before subsequent analysis.
The CFDA (5-carboxyfluorescein diacetate) assay was used to measure cell
viability as described previously [14, 20]. CFDA is a cell-permeant dye that can
be taken up by live cells. Once inside the cells, hydrolysis by non-specific
esterases results in the formation of a highly charged and fluorescent compound
that leaks out of the cells slowly. Cells were incubated with 2.5
Cells were harvested by trypsinization, resuspended in PBS with 1% BSA and 50
ng/mL 10-nonyl acridine orange (NAO, Molecular Probes), and incubated at 37
Cells were harvested in Tris-acetate buffer (pH 7.75) and the ATP content was analyzed using the luciferase-luciferin solution (Thermo Labsystems, Helsinki, Finland) as described previously [20]. The intensity of the emitted light was measured using a Fluostar optima plate reader (BMG Lab Technologies, Germany). ATP concentration was determined from a standard curve ranging from 10–100 pmole ATP. Protein content was determined with the bicinchoninic acid assay (Pierce Chemical Co., TX, USA).
Total cellular RNA was extracted using TriZol (Molecular Research Center, Inc.,
Cincinnati, OH, USA) according to the manufacturer’s instructions. The extracted
RNA was treated with DNase I (Ambion DNA-free DNA removal kit, Invitrogen,
Waltham, MA, USA) for 20 min at 37
| Gene | Forward primer (5′ to 3′) | Reverse primer (5′ to 3′) |
| Tfam | TCATGACGAGTTCTGCCGTTT | AACAATTCACCACTGCATGCA |
| Nfκb1 | GAAATTCCTGATCCCGACAAGA | TGTTCAATATCCCCAGACCTAACTT |
| Hif1- |
ACACGAGCTGCCTCTTCGA | CAGCCGCTGGAGCTAGCA |
| Nrf1 | GCTCAGCTTCGGGCATTTAT | TCCCCCAGCCTGGTTTTC |
| Nrf2 | TCCGAGCCGGTGTAAGTAGAGAT | TACTGGCATGGCCCTCAGAA |
| Cox3 | ACGAGATATCATCCGTGAAGGAA | TTCCGTATCGGAGGCCTTTT |
| Hk1 | CTCGCCTGGACCCCTAATC | TTCACTCATGGGCAATGCAT |
| Abbreviations of genes: Tfam, mitochondrial transcription factor A;
Nf | ||
Cells were processed for transmission electron microscopy (TEM) as described
previously [20, 21]. Briefly, cells were fixed with 2.5% (v/v) glutaraldehyde in
100 mM sodium phosphate (pH 7.4), centrifuged and embedded in 22% (w/v) BSA.
After washing with 100 mM sodium cacodylate (pH 7.2), the cells were post-fixed
with 1% osmium tetroxide in 100 mM sodium cacodylate (pH 7.2) for 1 hr at 4
The number of mitochondria per cell was counted in at least 25–50 cells in each group of undifferentiated and differentiated PC12 or NT2 cells subjected to preconditioning paradigms (hypoxia and NO treatments). Each count was verified by at least 2 observers blinded to the experimental treatments.
All results are presented as the mean
Two experimental preconditioning paradigms (hypoxia and NO treatment) were
standardized on undifferentiated PC12 cells to make sure that the applied
conditions did not trigger cell death, and ensured cell survival. The hypoxic
preconditioning was achieved by placing cells in an anoxic chamber for 24 hr and
subsequently allowing them to recover for 72 hr under normal tissue culture
conditions. For NO preconditioning we treated cells with DETA/NO, a NO donor,
known to spontaneously dissociate and liberate 2 moles of NO per mole of the
parent compound [17]. This treatment required 50
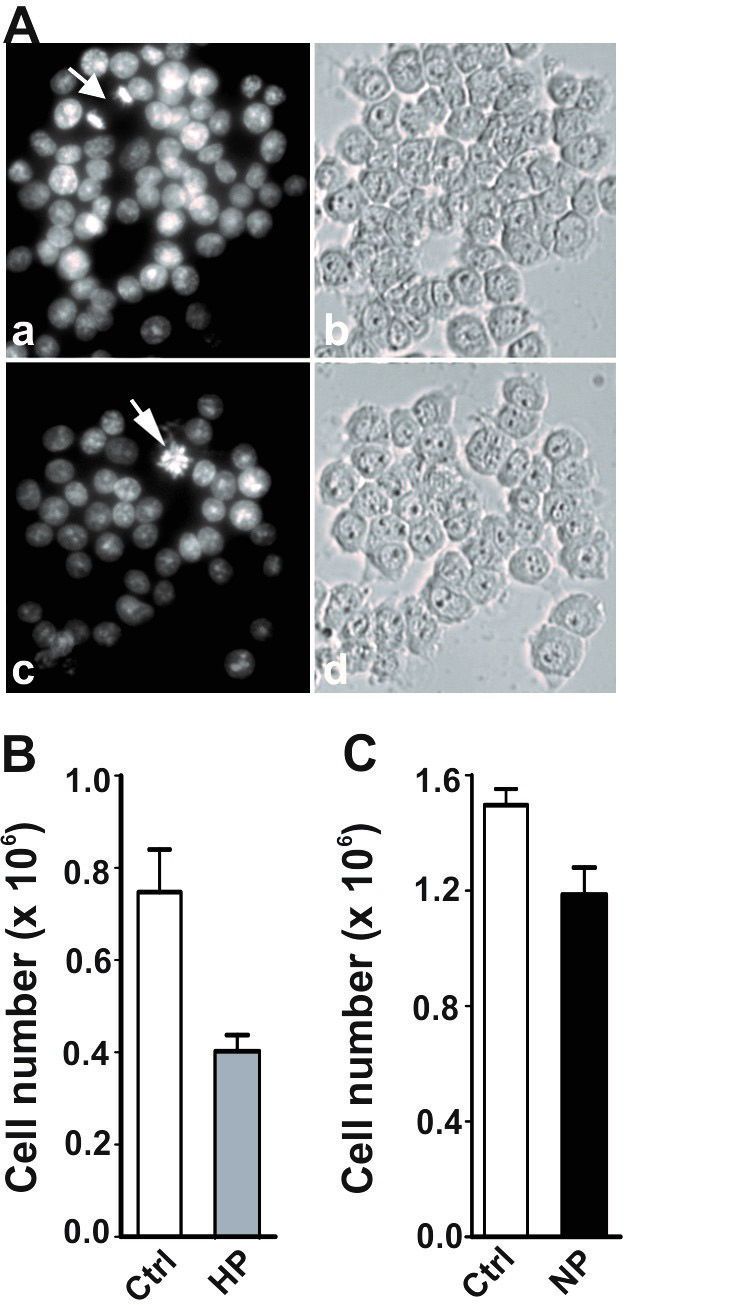 Fig. 1.
Fig. 1.Cytostatic effects of preconditioning paradigms. PC12 cells
were incubated for 24 hr in a chamber containing GasPak Plus envelopes followed
by a 72 hr recovery under normal culture conditions (A,B; HP) or were treated
for 6 days with 50
Highly aerobic tissues, such as the heart, are known to adapt to hypoxia by
increasing mitochondrial mass and therefore respiratory capacity [22]. We stained
the preconditioned PC12 cells with a fluorescent dye 10-nonyl acridine orange
(NAO), known to bind to mitochondria-specific cardiolipin, which is widely used
as a marker of mitochondrial mass in living cells. The cell samples were analyzed
for NAO staining by flow cytometry (Fig. 2). In the hypoxic preconditioning, a
time-dependent elevation of the NAO signal was observed during the 72 hr
post-anoxia recovery period (Fig. 2A–C, grey vs. black histograms). Similar
results were seen in cells exposed to NO preconditioning (Fig. 2D, grey vs. black
histogram). The NAO staining signal increased by ~15% in the
hypoxia-treated cells and by ~30% in response to the NO
preconditioning (Fig. 2E, p
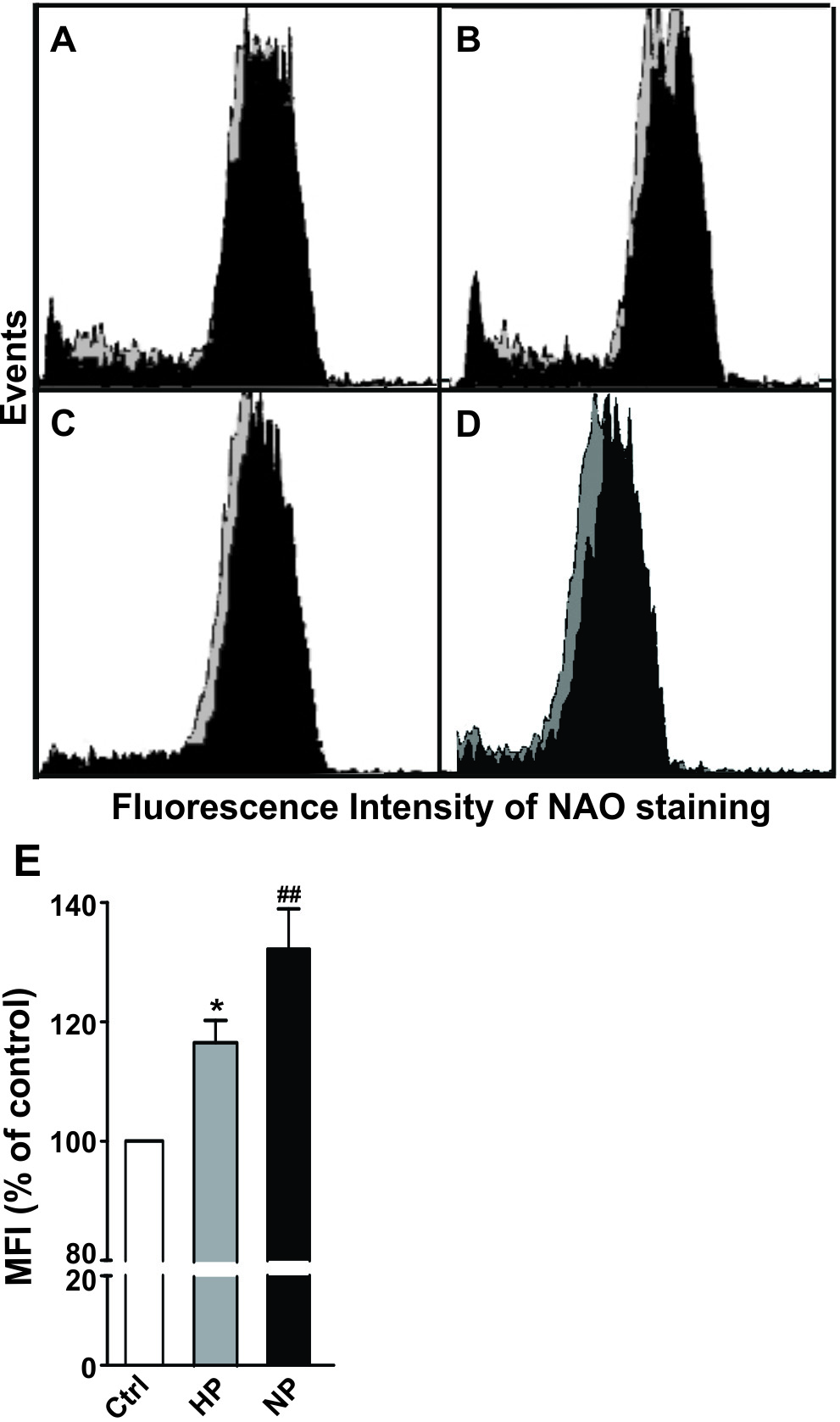 Fig. 2.
Fig. 2.Changes in mitochondrial mass in response to the preconditioning
treatments. PC12 cells were either subjected to hypoxia or DETA/NO
preconditioning as described in the Materials and Methods. The cells were stained
with NAO (50 ng/mL) and analyzed by flow cytometry using a Coulter Elite ESP flow
cytometer and EXPO32
Mitochondrial biogenesis is a highly regulated process requiring a coordinated
transcription of several genes in the nucleus as well as in the mitochondria
[23]. Here we were presented with a question whether the above observed changes
in the mitochondrial mass could be linked to the relevant changes in gene
expression. Accordingly, we evaluated the mRNA expression levels of key
transcription factors known to participate in the regulation of mitochondrial
biogenesis, such as nuclear respiratory factors NRF-1 and NRF-2, mitochondrial
transcription factor A (Tfam), oxidative stress response proteins
NF-
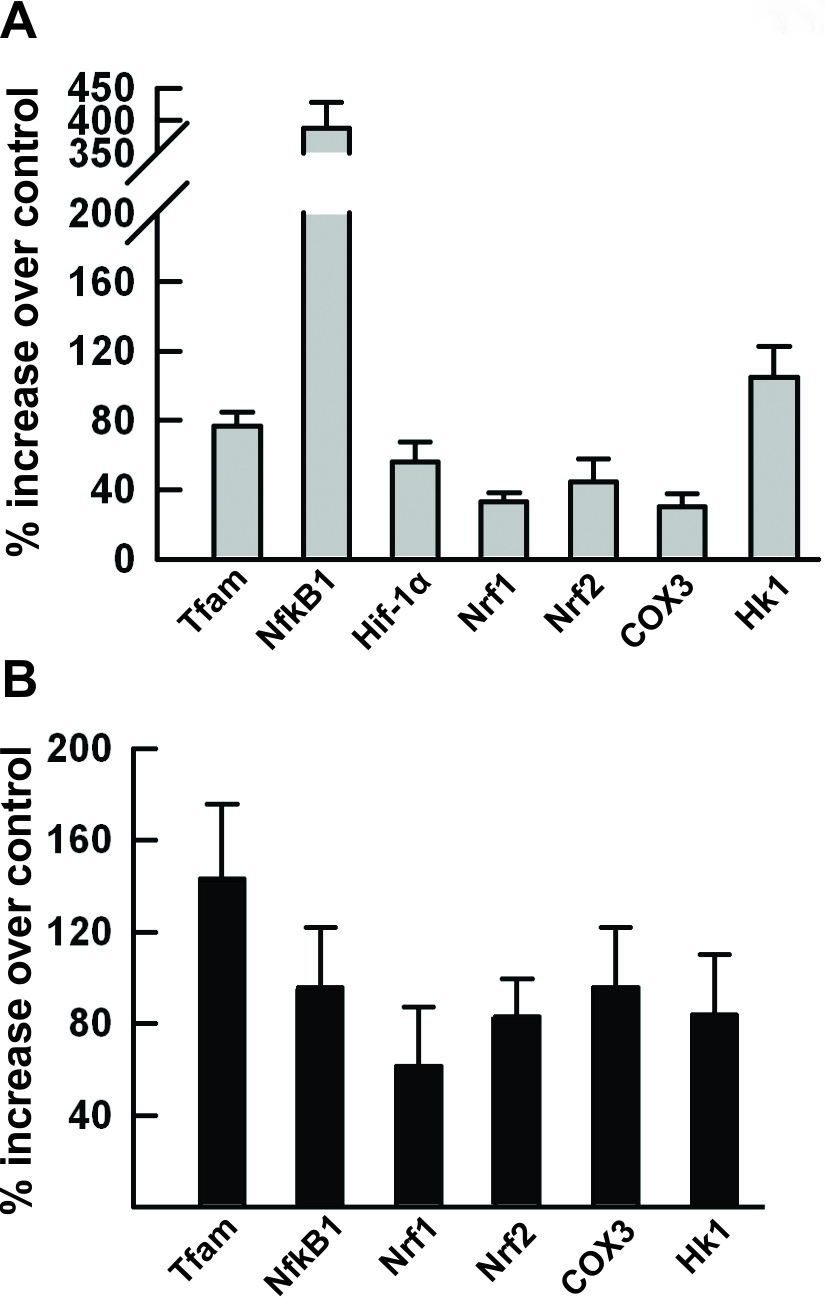 Fig. 3.
Fig. 3.Upregulation of mitochondrial biogenesis relevant genes
in preconditioned cells. Total cellular RNA was extracted from 24 hr hypoxia (A)
or 72 hr DETA/NO (B) preconditioned PC12 cells. cDNA was synthesized and real
time quantitative RT-PCR analysis was performed by using rat gene specific
primers (Table 1) as described in the Materials and Methods. The mRNA expression
levels of Nrf1, Nrf2, Tfam, Nf
Thus, both preconditioning paradigms transiently activated the gene expression
of transcription factors NRF1, NRF2 and Tfam as well as the NF-
Subsequently, we assessed the mitochondrial morphology of the cells by transmission electron microscopy (Fig. 4). The electron micrographs of cells treated with hypoxia (Fig. 4B) as well as DETA/NO (Fig. 4C) revealed dynamic intracellular tubular networks of numerous mitochondria with heterogeneous morphology and clear evidence of fission and fusion (Fig. 4B,C and enlarged insets) of mitochondria that are scattered throughout the cytoplasm. These images were strikingly different from those observed in the untreated control cells (Fig. 4A) in which a smaller number of mitochondria with normal rounded morphology was observed (Fig. 4A–C insets).
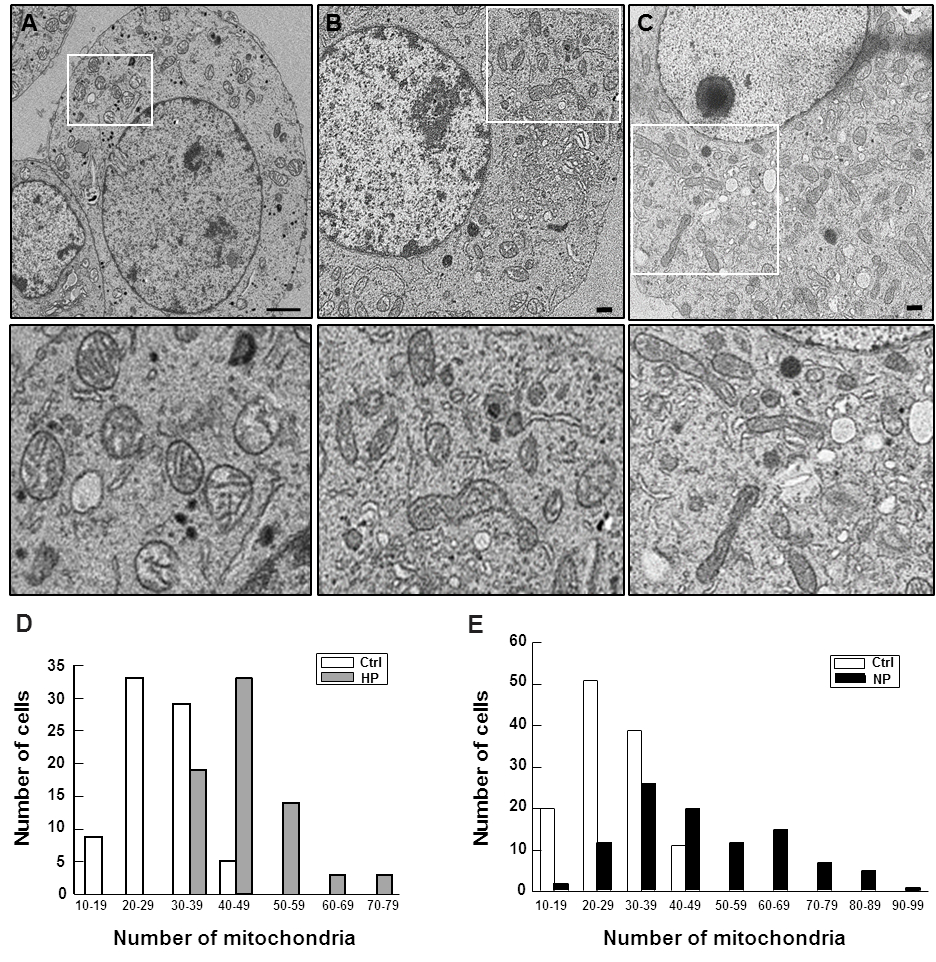 Fig. 4.
Fig. 4.Mitochondrial distribution and counts in preconditioned
cells. PC12 cells (control – A; preconditioned B and C) were processed for
electron microscopy as described in the Materials and Methods. Ultrathin sections
were cut and stained with lead citrate and images were digitally captured on a
JEOL 1230 transmission electron microscope. (A–C) Electron micrographs of a
typical control PC12 cell (A), a hypoxia preconditioned PC12 cell (B) and a
DETA/NO preconditioned PC12 cell (C). TEM images of PC12 cells (scale bar = 2
Preconditioned PC12 cells were challenged with oxygen-glucose deprivation (OGD),
a well-established in vitro model of ischemia-reperfusion injury of the
brain [24]. Morphological examination of Hoechst-stained cultures indicated that
cell viability was higher in the preconditioned cells challenged with OGD (Fig. 5A,b, hypoxia; Fig. 5A,d, DETA/NO) than control cells without preconditioning
(Fig. 5A,a,c). Whereas cell death was evident in control cultures after 24 hr of
OGD (Fig. 5A,a,c, numerous pyknotic nuclei), the preconditioned cell cultures
appeared far less affected by OGD (Fig. 5A,b,d, normal diffuse chromatin
distribution in the majority of nuclei). CFDA live cell assay confirmed the above
observations (Fig. 5B). Approximately 50% of OGD-treated control cells lost
viability (p
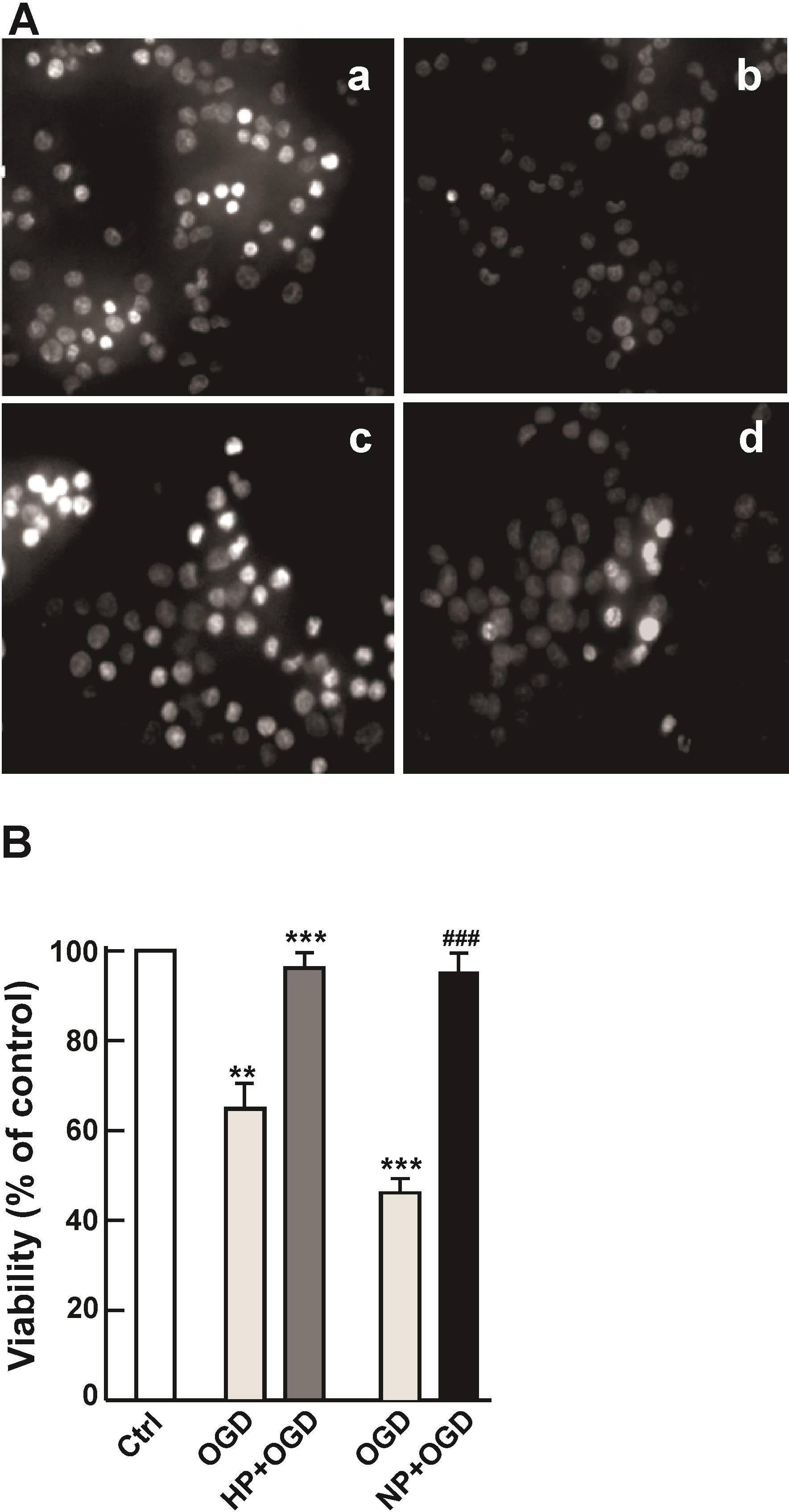 Fig. 5.
Fig. 5.Increased tolerance of preconditioned cells to
subsequent OGD stress. PC12 cells, untreated and preconditioned by HP or NP,
were subsequently challenged with 16 hr OGD followed by 24 hr of recovery. (A)
Microscopic evaluation of PC12 cells challenged with OGD. Cells were stained with
Hoechst 33258 and imaged on a Carl Zeiss Axiovert microscope. Shown are untreated
control cells (a) and (c), hypoxia preconditioned cells (b) and NO preconditioned
cells (d). (B) Cell viability was analyzed by CFDA assay as described in the
Materials and Methods. Bars show the mean
Low levels of ROS act as signaling molecules and stimulate mitochondrial
biogenesis in diverse cell types [25]. We then investigated whether
intracellularly generated ROS played any role in the observed mitochondrial
biogenesis during the preconditioning treatments (Fig. 6). Accordingly, the
preconditioning was performed in the presence of antioxidants, CoQ
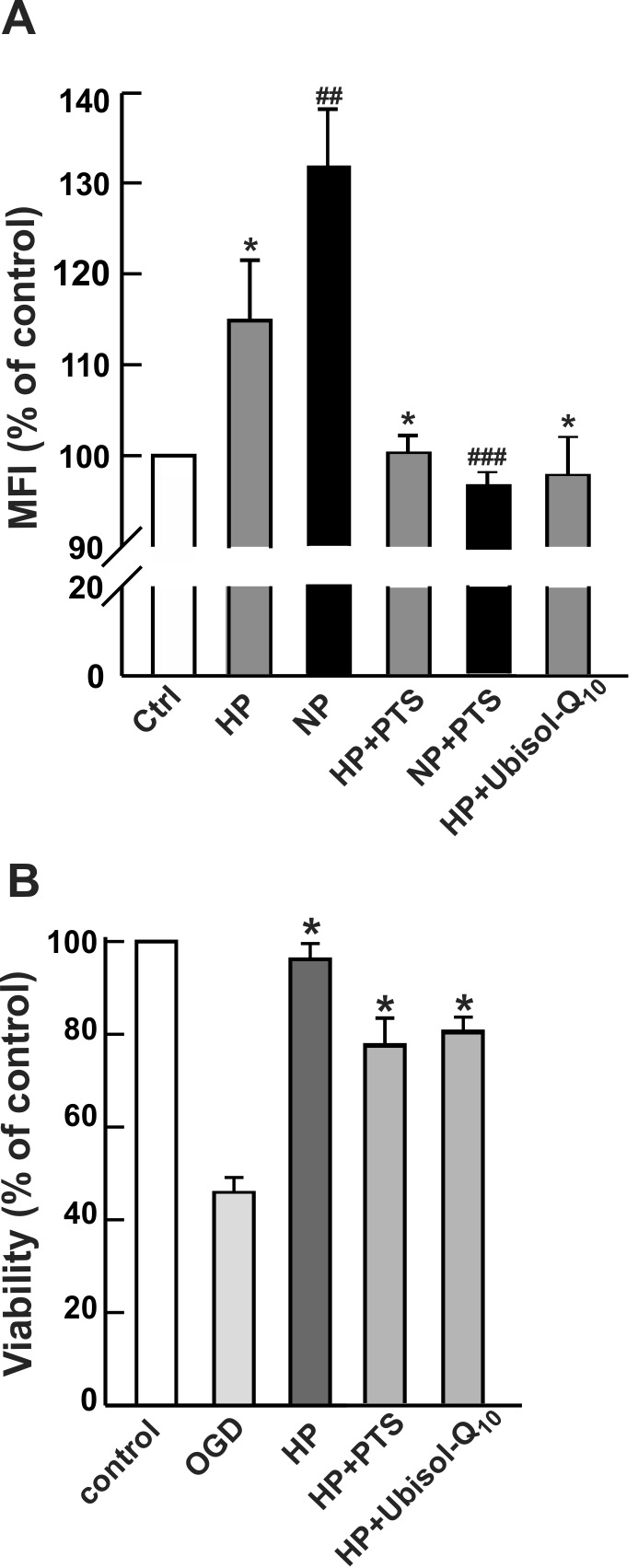 Fig. 6.
Fig. 6.Effects of antioxidants on mitochondrial biogenesis and
the tolerance of PC12 cells to OGD. PC12 cells were preconditioned either by
hypoxia (HP) or NO (NP) in the absence or presence of antioxidant PTS
(water-soluble derivatized vitamin E) or Ubisol-Q
NGF-differentiated PC12 cells [11] and human NT2-derived neurons [13, 26] were
used in this set of experiments. The differentiated cells expressed neuronal
markers,
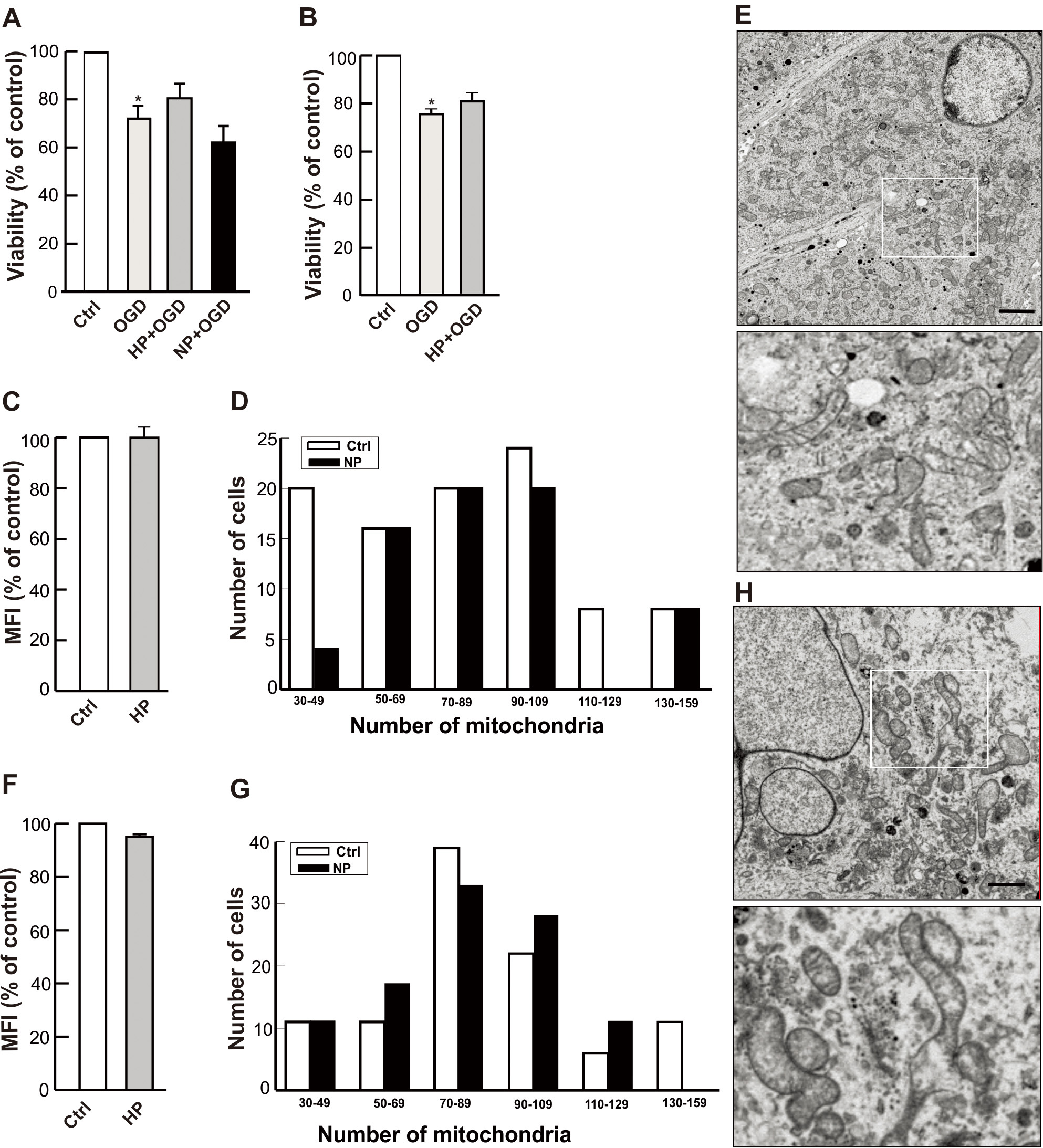 Fig. 7.
Fig. 7.Effects of preconditioning on differentiated neuronal cells.
NT2 cells were differentiated by retinoic acid (A,F–H) and PC12 cells by NGF
(B,C–E) as described in the Materials and Methods section. (A,B) The
differentiated neurons (NT2/N -A and PC12 neurons -B) were subjected to either
hypoxia (HP) or NO (NP) preconditioning and were subsequently challenged with 16
hr OGD. Cell viability was analyzed by the CFDA assay as described in the
Materials and Methods. Bars show the mean
The PC12- and NT2-derived neurons were also analyzed by NAO staining to establish whether any changes in the neuronal mitochondrial mass occurred after preconditioning. The results showed no increase in NAO fluorescence with respect to controls, neither in hypoxia preconditioned PC12 neurons (Fig. 7C) nor in hypoxia preconditioned NT2 neurons (Fig. 7F), nor in DETA/NO pre-treated neurons (data not shown). These findings indicate that mitochondrial mass was not increased in neurons by these preconditioning treatments.
Analysis of the transcriptional machinery involved in mitochondrial biogenesis
by RT-PCR also failed to show any changes in the mRNA levels of the relevant
transcription factors (NRF-1, NRF-2, Tfam, NF-
Mitochondrial density is regulated by a continuous cycle of fusion and fission [27]. These events allow the organelle to exchange content such as mtDNA, membranes and various metabolites to maintain a healthy and functional mitochondrion. These events were clearly revealed in the TEM micrographs of cell cross sections (Figs. 4,7). Comparison of electron microscopy images (Fig. 4A–C,7E–H) and quantitative mitochondrial counts (Fig. 4D,E,7D–G) revealed that differentiated neurons contained an increased number of mitochondria per cell than their undifferentiated counterparts, suggesting that mitochondrial biogenesis must have occurred during the neuronal differentiation processes. The data that support this conclusion are presented in Fig. 8. Whereas the great majority of undifferentiated cells (both PC12 and NT2 cells; Fig. 4D,E) contained between 20 and 40 mitochondria, the number of mitochondria in the great majority of neurons reached 70–130 per cell (Fig. 8A). Accordingly, the NAO staining in PC12 neurons, for example, was nearly 2-times higher (Fig. 8B) and a steady-state content of ATP was 50% higher than those of undifferentiated PC12 cells (Fig. 8C). Overall, the differentiated cultures responded much less severely to OGD, with only about 20% cell death 24 hr after the OGD insult (Fig. 7B) as compared to 50% death after OGD for undifferentiated cultures (Fig. 5B). These observations confirmed once again the significant role of mitochondria in maintaining cell viability, especially under oxidative stress.
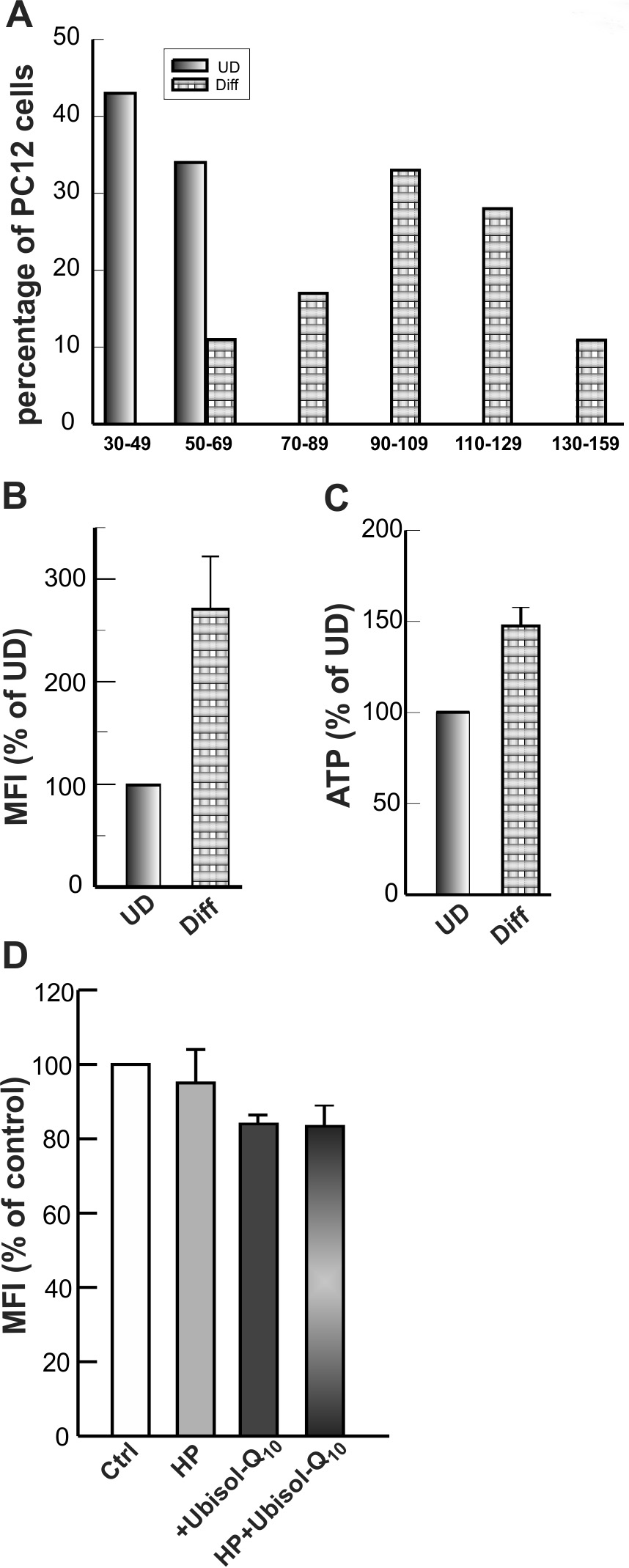 Fig. 8.
Fig. 8.Mitochondrial content, energy status during differentiation of PC12 cells. (A) Undifferentiated and differentiated PC12 cells were fixed, stained, embedded, and sectioned for electron microscopy as described in Materials and Methods. TEM images of cells were digitally captured on a JEOL 1230 electron microscope, and the number of mitochondria per cell slice was determined for cells with similar cytoplasmic volumes. (B) The fluorescence intensity of NAO was analyzed by flow cytometry for undifferentiated (UD) and differentiated (Diff) PC12 cells. (C) Basal ATP levels were measured by the luciferase assay for UD and Diff-PC12 cells. (D) Differentiated PC12 cells were hypoxia preconditioned in the presence or absence of antioxidants and mitochondrial mass was determined by staining with NAO and analysis by flow cytometry. Bars show the percent increase in the mean fluorescence intensity (MFI) values over control cells from three separate experiments.
We also asked whether intracellular ROS signaling was critical for mitochondrial
biogenesis during neuronal differentiation. To address this issue, PC12 cells
were differentiated by treatment with NGF for 7 days in the absence or presence
of Ubisol-Q
The effects of the preconditioning treatments with hypoxia and NO were also tested in the primary cultures of mouse cortical neurons and the results showed no difference in the mitochondrial mass and neuroprotection against OGD between preconditioned and control cell cultures (Fig. 9). Thus, the above results obtained with differentiated and cultured rat and human neuronal cells in vitro, are consistent with the phenomenon observed in the primary culture of mouse cortical neurons in vivo.
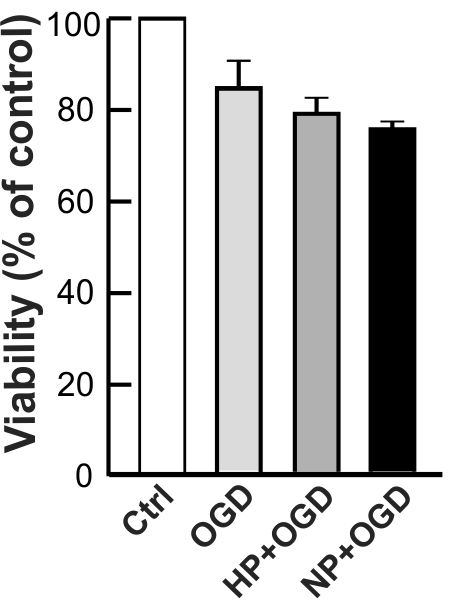 Fig. 9.
Fig. 9.Effect of preconditioning on the viability of primary mouse
cortical neurons. Primary mouse cortical neurons were prepared as described in
Materials and Methods. Cells were treated with hypoxia for 24 hr followed by 72
hr recovery or with DETA/NO for 6 days. Cell viability was determined using the
CFDA assay after a 16 hr OGD followed by 24 hr of recovery. The control group
represents cells without preconditioning and OGD challenge. Bars show the mean
Mitochondrial biogenesis, which has been observed in various paradigms of experimental preconditioning, is regarded by many as a practical way of enhancing cellular tolerance to injuries; hence, its activation is especially significant in the CNS. However, it is still unclear whether this phenomenon could be easily induced in post-mitotic neurons by methods shown to work in proliferating cells. With that in mind, we compared the mitochondrial biogenesis responses to non-lethal preconditioning in undifferentiated neuronal cells and in post-mitotic neurons. To our knowledge, no such direct comparison was previously reported and the data presented here bears relevance to the mechanisms controlling mitochondrial biogenesis and its role in neuroprotection. The preconditioning paradigms, hypoxia and NO treatments, have been extensively tested both in vitro and in vivo studies and induced tolerance in a variety of cells and tissues [28, 29, 30, 31, 32]. In this study, we applied the preconditioning methods that were suitable for in vitro treatments of all neuronal cells.
Mitochondria are dynamic organelles that are not synthesized de novo, but are formed from pre-existing organelles through fusion and fission. This results in the formation of constantly changing intracellular tubular networks observed in Figs. 4,7. Through mitochondrial fission, smaller mitochondria are created to which new mitochondrial contents created during mitochondrial biogenesis can be added. Mitochondrial biogenesis, in turn, requires the synthesis of new mtDNA, new proteins and membrane components. The observed activation of transcriptional regulatory networks (Fig. 3) clearly indicate that these processes indeed occur during the preconditioning treatments.
We have shown that while undifferentiated PC12 cells responded to the preconditioning treatments (hypoxia and NO) by increasing the mitochondrial number per cell and better tolerance to the subsequent oxidative stress, but the differentiated post-mitotic neurons did not engage mitochondrial biogenesis machinery during such preconditioning treatments. However, mitochondrial biogenesis occurred in both cell lines during their differentiation and transition to the post-mitotic state. Consistently, post-mitotic neurons had higher number of mitochondria per cell, higher steady-state levels of ATP and better tolerance to oxidative stress than their undifferentiated counterparts. To our knowledge this is the first comprehensive study that compared mitochondrial biogenesis response and tolerance to oxidative stress in undifferentiated and differentiated neuronal cells subjected to preconditioning treatments. In a study using clonal cell lines of murine neuroblastoma C1300 cells, mitochondrial biogenesis and the consequent increase in mitochondrial mass were shown to correlate with different stages of neuronal differentiation [33].
It is well established that mitochondrial biogenesis is linked to increased
mitochondrial ROS production, which can act as important second messengers and
activate the retrograde signaling cascades [34]. Such retrograde signaling,
originated from mitochondria, is fundamental to coordinating the transcription of
nuclear and mitochondrial genes as required for mitochondrial biogenesis to take
place [35]. Although the exact chemical nature of ROS engaged in this phenomenon
is unknown, it is plausible that the majority of ROS include superoxide anions
produced when unpaired electrons escape from the electron transport chain
(primarily at sites of Complexes I and III) during respiration and react with
molecular oxygen. Superoxide anions, in turn, can spontaneously or enzymatically
be converted to hydrogen peroxide, which can also act as a second messenger [36].
Similar types of ROS are known to be produced during various preconditioning
paradigms and elimination of ROS can abolish the observed neuroprotective effects
[37, 38]. Accordingly, we found that hypoxic and NO preconditioning of PC12 cells
in the presence of antioxidants or scavengers of ROS (PTS and Ubisol-Q
Mitochondrial biogenesis is a complex process that is regulated by
a highly orchestrated crosstalk between the nuclear and mitochondrial genomes and
works in concert with the members of the peroxisome proliferator-activated
receptor
Although PGC-1
Mitochondrial organogenesis in post-mitotic neurons is not yet fully understood, hence the specific pathways involved in its regulation remain unknown. In our experiments, hypoxic and NO preconditioning of differentiated either PC12, NT2 neurons or primary mouse cortical neurons did not result in the activation of mitochondrial biogenesis processes and did not further protect post-mitotic neurons against cell death inducing signals (Figs. 7,8,9). Interestingly, increased number of elongated tubular mitochondria, suggestive of mitochondrial fission-fusion was observed in differentiated PC12 and NT2 cells (Fig. 7E–H). We observed similar change of morphological features of mitochondria in undifferentiated cells following preconditioning treatments (Fig. 4). Thus, mitochondrial biogenesis clearly occurred during the differentiation processes as evidenced by the number of mitochondria per neuron being higher than the undifferentiated counterparts (Figs. 4,7), which resulted in more beneficial metabolic outcomes, higher steady-state ATP levels, better resistance to cell death-inducing insults (Figs. 7,9).
Since mitochondria cannot be synthesized de novo, they are produced from pre-existing mitochondria that are used as a template for mitochondrial biogenesis during cell cycle [47]. Mitochondrial biogenesis is a complex process that is regulated by a highly orchestrated and integrated crosstalk between the nuclear and mitochondrial genomes. This process requires transcription, translation, modification and functional assembly over 1500 proteins, of these only 13 polypeptides are encoded by mtDNA. The vast majority of proteins are transcribed from the nuclear genome and subsequently imported from the cytoplasm into the mitochondria by a highly specialized nanomolecular machinery known as the TOM complex, which is a gateway for almost all mitochondrial proteins [48]. Hence both nuclear and mitochondrial genomes must be transcriptionally and translationally competent and cells must be able to engage all vital activities in the cycling cells. Our findings on undifferentiated neural cells showed clearly that prior to engaging mitochondrial biogenesis processes the proliferating cells entered a temporary cell cycle arrest state and became quiescent (Fig. 1). Typically, quiescent cells can re-enter cell cycle when activated as they did not reach the growth factor dependent restriction points of the G1 phase and their genome remains transcriptionally competent. CDKs (cyclin dependent kinases) in association with CKIs (cyclin regulatory subunits/inhibitors) govern cell cycle progression or arrest. Stimulation of mammalian cells with mitogens or growth factors induces the expression of cyclin D1 resulting in phosphorylation of Rb, weakening its interaction with E2F, leading to transcription of genes needed for cells to enter the S-phase. This phenomenon of reversible activation of nuclear gene transcription in cell cycle competent cells might be the reason for observed mitochondrial biogenesis in proliferating cells, but these mechanisms are inactivated in quiescent cells. Besides the classical role of E2F1 in regulating cell cycle from G1-S transition, increasing evidence also implicates E2F1 in the regulation of mitochondrial function through interactions with NRF1 and NRF2, key regulatory factors of mitochondrial biogenesis [49]. E2F1 regulates self-renewal of human induced pluripotent stem cells [50], but in post-mitotic neurons E2F1 acts as a cell cycle suppressor [51], mainly by the ability of E2F1 to bind Rb and block the cells to enter S-phase. Earlier work indicated that injured neurons attempt to re-renter cell cycle by aberrantly activating the expression of cyclin D1 [52, 53, 54], leading to a false hope that these events are neuroprotective. Regretfully, these attempts lead to neuronal cell death, not neuroprotection [54].
In summary, we have demonstrated that although mitochondrial biogenesis could be
induced in undifferentiated rat PC12 cells, this phenomenon could not be induced
in post-mitotic rat PC12 and human NT2 neuron-like cells as well as in primary
mouse cortical neurons using the two preconditioning paradigms employed in this
study. Pharmacological induction of mitochondrial biogenesis to restore
mitochondrial function and homeostasis remains an attractive therapeutic strategy
for the treatment of brain diseases. The US Food and Drug Administration (FDA)
has approved many pharmacological agents, such as pioglitazone
(thiazolidinedione-type), lasmiditan (5-hydroxytryptamine receptor 1F receptor
agonist), and formoterol (
JKS, Y-HW and MS wrote the grant proposal, designed and managed the research work. CS and MR-L performed the experiments, generated data and contributed to analyses and plotting of the data. JKS and MS composed and wrote the manuscript. Y-HW, Y-TW and Y-SM made constructive changes to the final text of the manuscript and contributed to significant editing of the manuscript. All authors have read and approved the final manuscript.
All animal work was performed in accordance with the guidelines provided by the Canadian Council on Animal Care and the procedures approved by the institutional Animal Care Committee at the National Research Council Canada (ACC protocol #2011.10).
The authors are thankful to Erin Twomey, Patricia Lanthier and Roger Tremblay for their excellent technical assistance in carrying out some experiments, as well as Peter Rippstein, University of Ottawa Heart Institute for his help with electron microscopy.
This work was supported by a joint collaborative grant from the National Research Council Canada and the National Science Council Taiwan and a grant supported by the Ministry of Science and Technology, Taiwan (MOST 110-2320-B-371-001).
The authors declare no conflict of interest. Y-HW is serving as the guest editor and editorial board memeber of this journal. We declare that Y-HW had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to JJ.









