1. Introduction
Inflammation is the body’s innate response to invading pathogens or tissue
trauma. Tissue macrophages, one of the immune system’s most prevalent cytokine
producers detect stress signals or pathogen-associated molecular patterns (PAMPs)
such as lipopolysaccharides (LPS) during the early onset of infection and release
a variety of pro-inflammatory mediators that include tumor-necrosis factor
(TNF)-, interleukin (IL)-1, IL-6, and nitric oxide (NO) [1].
This further promotes the recruitment and activation of leukocytes via signaling
pathways including the nuclear factor-kappa B (NF-B) pathway and the
mitogen-activated protein kinase (MAPK) pathway [2, 3, 4]. Macrophages also play a
key role in resolution of inflammation and restoration of tissue homeostasis [5, 6]. However, when such regulatory mechanisms fail, unresolved chronic
inflammation becomes harmful to the host and can lead to diseases such as
colitis, sepsis, and cancer [7, 8].
The lactoferrin-derived antimicrobial peptide (AMP), lactoferricin, is of
particular interest in the development of immunotherapeutic agents to help combat
inflammatory diseases and related conditions. Lactoferrin, which yields the
pepsin-cleavage product lactoferricin [9, 10, 11, 12], is found in exocrine secretions
including bile, saliva, and tears, as well as in the secondary granules of
neutrophils, which can be released during an inflammatory response [13].
Lactoferrin and lactoferricin peptides can act as immunomodulatory agents with
the ability to suppress in vitro and in vivo pro-inflammatory
responses. Bovine lactoferrin attenuates a pro-inflammatory response induced by
microorganisms [14, 15]. One study that examined an inflammatory response
mediated by THP-1 monocytes shows that bovine lactoferrin downregulates
LPS-stimulated TNF- cytokine production in a NF-B-dependent
manner [16]. Studies of human lactoferrin show the significance of this peptide
as an anti-inflammatory agent [15, 17].
Bovine lactoferricin is an effective anti-inflammatory and anti-catabolic agent,
mitigating the production of the pro-inflammatory mediators IL-6 and inducible
nitric oxide synthase (iNOS) in human chondrocytes [18]. In vitro and
ex vivo evidence shows that lactoferricin is an important down-regulator
of LPS-induced inflammation in nucleus pulposus cells derived from the
intervertebral disc [19]. Bovine lactoferricin also inhibits the production of
IL-6 by THP-1 monocytes more strongly than its parent peptide [20].
Interestingly, bovine lactoferricin also upregulates anti-inflammatory cytokines
IL-10, IL-11, and IL-4 [18]. There is evidence that lactoferricin exerts its
anti-inflammatory effects in an extracellular signal-regulated kinase (ERK)- and
p38 MAPK-dependent manner [18, 21].
Lactoferricin peptides demonstrate a wide variety of immunomodulatory and
anti-cancer properties; however, few studies have addressed their influence on a
macrophage-associated inflammatory response, and no investigations have addressed
the possible differential efficacies of lactoferricin derived from different
species. Macrophages are a primary source of cytokines at the site of
inflammation, thus making them an ideal cell model for studying inflammation and
possible therapeutic interventions. Given the accumulating evidence of bovine
lactoferricin as an immunomodulatory agent and the most potent antimicrobial
agent of different species-specific lactoferricin, it was hypothesized that this
peptide would have the greatest regulatory effect on macrophage-associated
inflammation when compared to mouse and human lactoferricin.
2. Materials and methods
2.1 Reagents
Bovine serum albumin (BSA), phosphate buffered saline (PBS),
phenylmethylsulfonyl fluoride (PMSF), Roswell Park Memorial Institute 1640 medium
(RPMI), Dulbecco’s Modified Eagle’s Medium (DMEM), phorbol 12-myristate
13-acetate (PMA), and Triton-X-100 were purchased from Sigma-Aldrich Canada
(Oakville, ON). Fetal bovine serum (FBS), 10,000 U/mL penicillin/10,000
g/mL streptomycin solution, and 1M
4-(2-hydroyethyl)-1piperazineethanesulfonic acid (HEPES) buffer solution were
obtained from Invitrogen Canada (Oakville, ON). Sodium dodecyl sulfate (SDS),
Tris base, and Tween-20 were acquired from Bio-Shop Canada Inc. (Burlington, ON).
Ethylene diamine tetraacetic acid (EDTA) was purchased from EM 46 Industries Inc.
(Hawthorne, NY). Luminata Forte Western HRP substrate were purchased from
EMD Millipore (Etobicoke, ON). Bio-Rad Protein Assay Dye Reagent was obtained
from Bio-Rad Laboratories Inc. (Mississauga, ON). Lactoferricin peptides (HCl
salt) (Table 1, Ref. [22]) in a linear configuration were synthesized and
purchased from American Peptide (Sunnyvale, CA).
Table 1.Amino acid sequences of bovine, mouse, and human
lactoferricin.
| Lactoferricin |
Amino acid sequence |
| Bovine |
NH-PHE-LYS-CYS-ARG-ARG-TRP-GLN-TRP-ARG-MET-LYS-LYS-LEU-GLY-ALA-PRO-SER-ILE-THR-CYS-VAL-ARG-ARG-ALA-PHE-COOH |
| Mouse |
NH-GLU-LYS-CYS-LEU-ARG-TRP-GLN-ASN-GLU-MET-ARG-LYS-VAL-GLY-GLY-PRO-PRO-LEU-SER-CYS-VAL-LYS-LYS-SER-SER-COOH |
| Human |
NH-THR-LYS-CYS-PHE-GLN-TRP-GLN-ARG-GLN-MET-ARG-LYS-VAL-ARG-GLY-PRO-PRO-VAL-SER-CYS-ILE-LYS-ARG-ASP-SER-COOH |
| The 25-amino acid residue sequence of lactoferricin from bovine, mouse, or human
origin. Highlighted residues (in bold) represent the presence of a disulfide bond
that exists between two cysteine residues in each peptide. Lactoferricin peptides
used in this study were synthesized in a linear configuration. Adapted from
Vorland et al. [22]. |
2.2 Antibodies
Rabbit (Rb) anti-p65, Rb anti-phospho p44/42 MAPK (ERK1/2) (pTpY202/204), Rb
anti-p44/42 MAPK (ERK1/2), Rb anti-phospho-IB (Ser32), Rb
anti-IB, Rb anti-phospho-c-Jun (Ser63), Rb anti-c-Jun and Rb
anti--actin (horse radish peroxidase (HRP) conjugate) antibodies were all purchased
from Cell Signaling Technology (Beverly, MA). HRP-conjugated donkey anti-rabbit
secondary antibody was purchased from Santa Cruz Biotechnology (Santa Cruz, CA).
Goat-anti-rabbit secondary antibody conjugated to Alexa Fluor ®
488 was obtained from Invitrogen. TNF-, and IL-6 capture antibodies and
biotin-conjugated detection antibodies for ELISA were purchased from eBioscience
(San Diego, CA).
2.3 Cell lines
RAW 264.7 mouse macrophage-like cells were purchased from ATTC ®
(Manassas, VA). THP-1 human monocytic-like cells were kindly provided by Dr.
Brent Johnston (Dalhousie University, Halifax, NS).
2.4 Culture medium and conditions
THP-1 and RAW 264.7 cell lines were maintained at 37 C in a humidified
5% CO incubator and cultured in complete RPMI (cRPMI) 1640 medium. Cells
were cultured in 75 mm tissue culture flasks (Fisher Scientific, Waltham,
MA) and passaged at 80–90% confluency. RAW 264.7 cells were passaged using a 25
cm long cell scraper (VWR, Mississauga, ON).
2.5 Isolation and differentiation of mouse Bone Marrow Derived
Macrophages (BMDMs)
C57BL/6 female mice, purchased from Charles River Laboratories (Wilmington, MA),
were euthanized at 8-10 weeks of age, and bone marrow was collected from the
tibias by flushing with PBS (pH 7.2) making a single cell suspension. Red blood
cells were lysed by hypo-osmotic shock. The remaining bone marrow cells were
differentiated over 7 d by culturing in cRPMI containing 15% (v/v)
L929-conditioned DMEM as a source of M-CSF. After 3 d of culture, the cells were
fed with fresh medium. At 6 d, culture medium and non-adherent cells were
removed, and the remaining cells fed with fresh medium. At 7 d the resulting
macrophages were used in experiments.
2.6 Cell seeding
Both cell lines and primary cells were seeded 1 d prior to treatment to allow
for adherence to plastic. For all cell types, cells were cultured in cRPMI. THP-1
monocytic like cells were treated with 200 ng/mL PMA in order to cause cells to
differentiate into macrophages and adhere to the plate overnight prior to any
additional treatment. For ELISA experiments THP-1 cells were seeded in 24-well
plates at 2.5 10 cells/well in 1 mL of medium. Cells were plated
at 6 10 cells/well in a 6-well plate for quantitative real-time
polymerase chain reaction (q-PCR) experiments. For immunofluorescent assays,
cells were seeded on 10 mm glass coverslips placed in a 12-well plate at 1.5
10 cells/well in 1 mL medium. Once adhered, cells were cultured
overnight in serum-free medium to allow for cell growth arrest. For western blot
experiments, cells were seeded at 1 10 cells in 75 mm
tissue culture flasks and left for 36–48 h until 90% confluency was reached,
then cells were treated with 200 ng/mL PMA overnight and serum-starved an
additional night prior to treatment. For ELISA experiments, RAW 264.7 cells were
seeded in 24-well plates at 1.5 10 cells/well
in 1 mL of medium. Cells were plated at 2.5 10 cells/well in a
6-well plate for q-PCR. For western blot experiments cells were seeded at 1
10 cells/well in 10 cm cell culture plates (Thermo Fisher)
and left for 36–48 h until 90% confluency was reached, then they were
serum-starved overnight prior to treatment. For immunofluorescent assays, cells
were seeded on 10 mm glass coverslips placed in a 12-well at 1 10 cells/well in 1 mL medium. BMDMs were seeded in 24-well plates at 2.5
10 cells/well in 1 mL of medium. Cells were plated at 6
10 cells/well in a 6-well plate for q-PCR experiments.
2.7 Enzyme-linked immunosorbent assay
Supernatants were collected from THP-1, RAW 264.7, and BMDM cell cultures for
detection of TNF-, and IL-6 using a sandwich enzyme-linked
immunosorbent assay (ELISA) Ready-SET-Go!® ELISA kit from
eBioscience (San Diego, CA) according to the manufacturer’s instructions. Cells
were treated with medium alone, 100 ng/mL LPS alone, or with LPS in combination
with 5 M lactoferricin peptides (bovine, mouse, human). A subset of
experiments used cadmium chloride (CdCl), purchased from Sigma Aldrich. For
these experiments, cells were treated with medium alone, 10 M
CdCl alone, or with CdCl in combination with 5 M
lactoferricin peptides. Supernatants being analyzed for TNF- were
collected after 6 h, and all other supernatants were collected after 24 h of
treatment. Previous experiments indicated peak cytokine expression at these
time-points after LPS-stimulation of macrophages. The absorbance values at 450 nm
were determined using a ELx800 UV universal microplate reader (BioTek
Instruments, Winooski, VT), Digiread software, and SOFTmax® PRO
software (version 4.3; Molecular Devices Corp., Sunnyvale, CA).
2.8 Griess assay
Supernatants were collected from RAW 264.7 cells and BMDMs for detection of
nitrite in solution as an indicator of NO production using Griess reagent
purchased from Sigma Aldrich, as per the manufacturer’s instructions. Cells were
seeded in a 24-well plate and left overnight. Cells were then treated with 500
ng/mL of LPS alone or with LPS in combination with 5 M
lactoferricin (bovine, mouse, or human) for 24 h. Supernatants were then
collected and added to an equal volume of Griess reagent in a 96-well plate.
Sodium nitrite purchased from Sigma Aldrich was used to make a standard curve.
Plates were then incubated at room temperature in the absence of light.
Absorbance values at 570 nm were determined using a ELx800 UV universal
microplate reader (BioTek Instruments, Winooski, VT), Digiread software, an d
SOFTmax® PRO software (version 4.3; Molecular Devices Corp.,
Sunnyvale, CA).
2.9 cDNA synthesis
Previous experiments established that peak cytokine mRNA expression occurred 4 h
after LPS-stimulation of macrophages. RNA was harvested using a RNeasy Mini Kit
purchased from Qiagen (Valencia, CA) and stored at –80 C for the
purpose of cDNA synthesis. Approximately 500 ng RNA was then reverse transcribed
using an iScript cDNA synthesis kit (Bio-Rad Laboratories; Hercules, CA)
according to the manufacturer’s instructions. The iScript reaction mix (2
L) and iScript reverse transcriptase (0.5 L) was
added to RNA template and nuclease-free water to a final volume of 10
L and final concentration of RNA template 50 ng/L.
The reaction was incubated in a Bio-Rad T100 Thermocycler using the
following reaction protocol: 5 min at 25 C, 30 min at 42 C,
and 5 min at 85 C. Once synthesized, the cDNA was stored at –20
C for future use.
2.10 Quantitative real-time Polymerase Chain Reaction
SsoFast EvaGreen Supermix® (Bio-Rad Laboratories) was
used for the q-PCR. The cDNA samples were diluted 1:4 in pyrogen-free water.
Primers were optimized (data not shown) before primer mixes of 100 nM for
glyceraldehyde-3phosphate dehydrogenase (GAPDH), TNF-, and iNOS (Table 2) (Integrated DNA Technologies; Skokie, IL) were made from 10 L
of both the forward and reverse primers added to 80 L of water. A 1
L sample of diluted cDNA was then added to a master mix solution
containing 5 L EvaGreen Supermix, 3 L pyrogen-free
water, and 1 L primer mix in a final volume of 10 L.
Negative controls did not contain any cDNA. Reactions were conducted in
triplicate using a Stratagene Mx3005p q-pcr system (Agilent Technologies, Santa
Clara, CA) and a Rotor-Gene 6000 q-PCR machine (Qiagen, Valencia, CA). Cycling
conditions consisted of a 30 s activation step at 95 C, followed by 40
amplification cycles for 5 s at 95 C and 30 s at an annealing
temperature specific to each primer set used (refer to Table 2). To confirm that
the PCR reaction had produced the specific and intended products, a melt curve
analysis was conducted using MxPro q-PCR Software (Agilent Technologies, Santa
Clara, CA) cycle threshold (CT) values, indicating the number of cycles it takes
for the fluorescent signal to surpass the background fluorescence. The relative
amounts of amplicons were determined by normalizing the CT values of the target
gene to the endogenous control, GAPDH. These values were then normalized to the
untreated control values, giving the expression fold values.
Table 2.Human and mouse primer sequences used for q-PCR experiments.
| Human primers |
Primer sequence |
| GAPDH |
F-5′CAACGGATTTGGTCGTATTGG-3′ |
| R-5′GGCAACAATATCCACTTTACCAGAGT-3′ |
| TNF- |
F-5′CCAGGCAGTCAGATCATCTTCTC-3′ |
| R-5′AGCTGGTTATCTCTCAGCTCCAC-3′ |
| Mouse primers |
|
| GAPDH |
F-5′CCACTTCAACAGCAACTCCCACTCTTC-3′ |
| R-5′TGGGTGGTCCAGGGTTTCTTACTCCTT-3′ |
| TNF- |
F-5′CATCTTCTCAAAATTCGAGTGACAA-3′ |
| R-5′GCACCTCAGGGAAGAGTCTG-3′ |
| iNOS |
F-5′CAGCTGGGCTGTACAAACCTT-3′ |
| R-5′TGAATGTGATGTTTGCTTCGG-3′ |
2.11 Western blotting
THP-1 cells, RAW 264.7 cells, and BMDMs treated with medium only, bovine, mouse,
or human lactoferricin alone [5 M], LPS [100 ng/mL], or a
combination of lactoferricin peptides and LPS were lifted from tissue culture
flasks using 5 mL of 10 mM EDTA and a cell scraper and collected in tubes that
were centrifuged at 500 g for 5 min. Cells were lysed in ice-cold RIPA
buffer (50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 50 mM NaHPO, 0.25%
sodium deoxycholate [w/v], 0.1% Nonidet P-40 [v/v], 5 mM
ethylenediaminetetraacetic acid, and 5 mM ethyleneglycoltetraacetic acid)
supplemented with protease and phosphatase inhibitors (1 mM NaVO, 1
mM NaF, 1 mM phenylmethylsulfonyl fluoride, 1 g/mL aprotinin, 1
g/mL leupeptin, and 1 g/mL pepstatin) on ice for 15 min. Samples
were centrifuged at 14,000 g for 10 min to remove cell
debris. Supernatants containing protein were then collected and stored at –80
C for future use. A Bradford assay (Bio-Rad, Hercules, CA, United
States) was used to quantify total protein concentrations from each sample. The
proteins in each sample were then denatured by the addition of sample buffer (200
mM Tris-HCl [pH 6.8], 30% glycerol [v/v], 6% sodium dodecyl sulfate [w/v], 15%
-mercaptoethanol [v/v], and 0.001% bromophenol [w/v]) and placed in a
heating block at 95 C for 5 min. If not used immediately, samples were
stored at –80 C until future use. Equal amounts of protein sample (10
g) were loaded onto Tris-HCl acrylamide resolving gels and
separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Proteins
were transferred to a nitrocellulose membrane using the iBlot®
dry blotting system (Life Technologies, Burlington, ON). Membranes were washed
with Tris-buffered saline (TBS)-Tween-20 (TBST; 20 mM Tris-HCl [pH 7.6], 200 mM
NaCl, 0.05% Tween-20 [v/v]) and blocked in TBST containing 5% fat-free milk
powder [w/v] for 1 h at room temperature or overnight at 4 C with
gentle rocking.
Membranes were washed and exposed to the primary antibody for 1 h at room
temperature or overnight at 4 C with gentle rocking. Membranes were
washed and then exposed to the HRP-conjugated secondary antibody (1:1000 in TBST
blocking solution) for 1 h at room temperature with gentle rocking. Membranes
were washed and reacted with Luminata Forte Western HRP Substrate
(Millipore; Taunton, MA), exposed to X-ray film (Sci-Med Inc.; Truro, NS), and
then developed using a Kodak X-OMAT 1000A automated X-ray developer. In order to
account for any variation of loading between protein samples, membranes were
re-probed for -actin. Image Studio Software
(LI-COR®; Guelph, ON) was used to determine the relative
intensity of each band through densitometry. The ratio of actin normalized to
phospho-protein was compared to that of total protein normalized to
phospho-protein and subsequently normalized to the medium control.
2.12 Immunofluorescence
Imaging of cells stained with fluorescent antibodies was used to determine
nuclear localization of the inflammation-associated transcription factor
NF-B. Cells were grown on coverslips that were placed in a 12-well
plate, left for 24 h, serum-starved overnight, and treated with peptide and LPS
for 4 h. Medium was removed and cells were fixed with 4% paraformaldehyde.
Coverslips were washed in PBS and allowed to dry overnight. Slides were blocked
and incubated with anti-p65 antibody diluted 1:100 in antibody dilution buffer.
From this point on, all incubations were performed in the absence of light. Cells
were washed in PBS, incubated in goat-anti-rabbit secondary antibody conjugated
to Alexa Fluor ® 488 diluted 1:500 in antibody dilution buffer,
washed again, and incubated in 30 M 4’,6-diamidino-2-phenylindole
(DAPI) diluted 1:100 in 1xPBS. Coverslips were lifted from the plate. Dako
Fluorescent Mounting Medium was applied, and each coverslip was mounted to
individual Fisherbrand Superfrost® Plus slides. Visualization of
slides was done using a Zeiss Axioplan II Motorized Microscope (Zeiss Canada,
North York, ON) and AxioVision 4.8 Microscopy Software.
2.13 Statistical analysis
A one-way analysis of variance (ANOVA) with a Dunnett’s post-test was conducted
using GraphPad Prism Software (GraphPad Software Inc.; La Jolla, CA). Data were
considered significantly different when the p value was less than 0.05
(indicated by *); when the p value was greater than 0.05, data were
considered to not be statistically significant.
3. Results
A sub-lethal concentration of lactoferricin peptides was used to ensure
regulation of inflammatory cytokine synthesis was not due to cell stress or
death. Previous studies established that a 5 M concentration of
either bovine, mouse, or human lactoferricin did not affect the viability of RAW
264.7, THP-1 or BMDM cells (data not shown). This is consistent with other
studies of AMPs and their immunomodulatory effects on a variety of different
cells that employed peptide concentrations ranging from 1-30 M
[23, 24, 25, 26, 27, 28].
3.1 Lactoferricin peptides decrease pro-inflammatory cytokine
synthesis by LPS-stimulated macrophages
The ability of bovine, mouse and human lactoferricin peptides to downregulate
pro-inflammatory cytokine mRNA expression by LPS-stimulated macrophages was
investigated in a series of q-PCR experiments. A 5 M concentration
of each lactoferricin peptide dampened LPS-induced TNF- mRNA expression
in RAW 264.7 macrophages (Fig. 1A; p 0.01) and THP-1 macrophages
(Fig. 1B; p 0.05). However, in BMDMs, only bovine lactoferricin
downregulated LPS-induced TNF- mRNA expression (Fig. 1C; p 0.05).
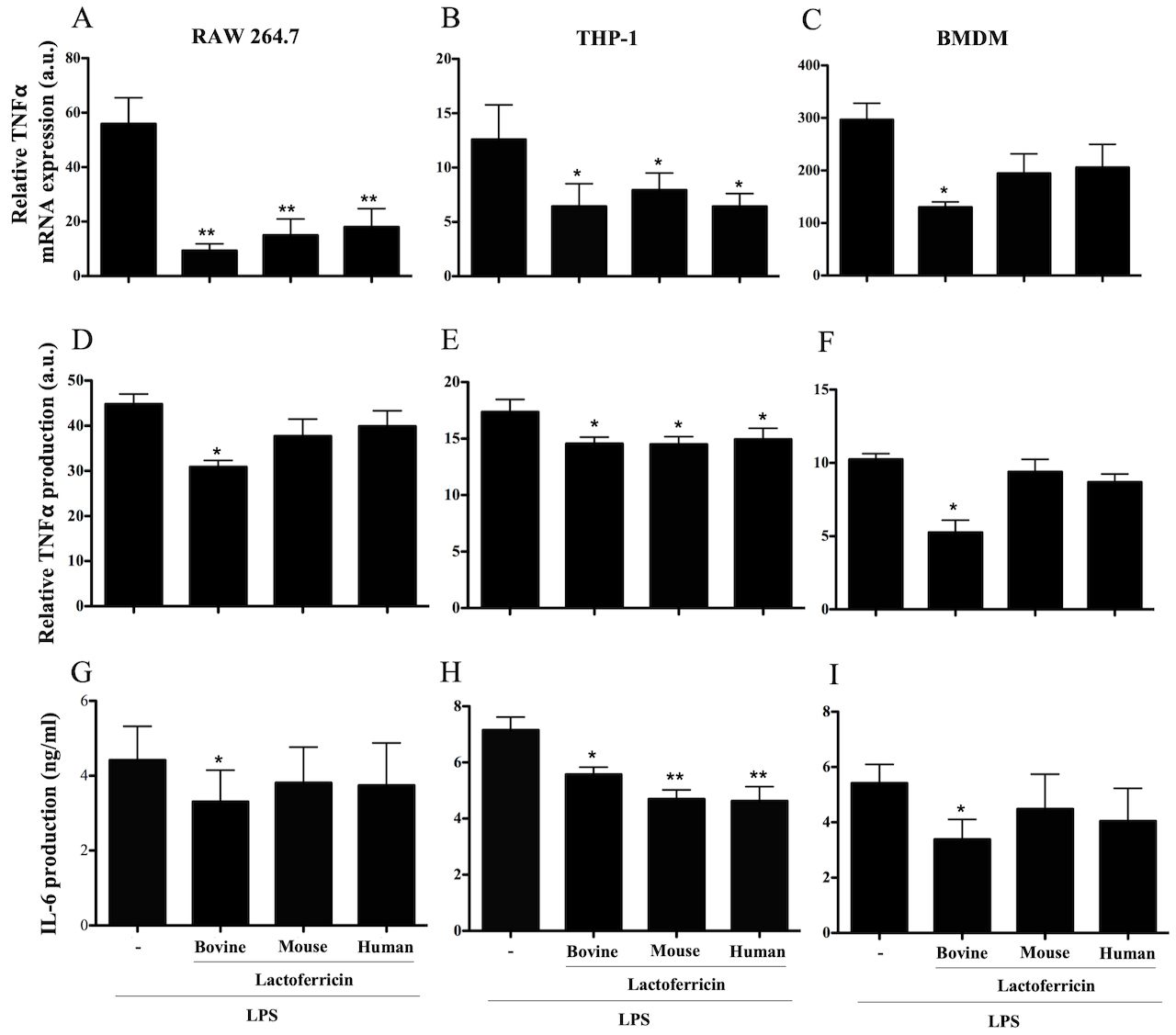 Fig. 1.
Fig. 1.
Species-specific lactoferricin peptides decrease LPS-induced
TNF- and IL-6 cytokine production in macrophages. (A, D, and G) RAW
264.7 cells, (B, E, and H) THP-1 cells, and (C, F and I) BMDMs were treated with
100 ng/mL LPS alone or in combination with 5 M bovine, mouse, or
human lactoferricin. RNA was isolated for cDNA synthesis and then qPCR was
performed to determine expression levels of TNF-, 4 h after treatment,
relative to untreated control treatments (A–C) (calculated by dividing the
expression value for the lactoferricin treated cultures by the untreated control
and multiplied by 100). Supernatants were collected and used in ELISA assays to
detect TNF- (D–F) 6 h after treatment and IL-6 (G–I) 24 h after
treatment. Levels of TNF- production are relative to untreated control
treatments (calculated by dividing the expression value for the lactoferricin
treated cultures by the untreated control and multiplied by 100). Data are the
mean of 3–4 independent experiments SEM. * denotes p 0.05
compared to LPS positive control as determined by ANOVA with Dunnett’s multiple
comparisons post-test. a.u. denotes arbitrary units.
In line with the mRNA expression findings, ELISA analysis of 24 h culture
supernatants demonstrated that 5 M bovine lactoferricin
significantly reduced the production of TNF- following LPS stimulation
of RAW 264.7 cells, THP-1 cells, and BMDMs comparison to LPS treatment alone
(Fig. 1D–F). Mouse and human lactoferricin also decreased LPS-induced
TNF- production by THP-1 cells (Fig. 1E), but did not affect
TNF- production by LPS-stimulated RAW 264.7 cells or BMDMs (Fig. 1D–F). Representative TNF- concentrations from RAW 264.7 cells are as
follows: no treatment, 63 pg/mL; LPS alone, 3120 pg/mL; bovine lactorferricin
plus LPS, 1853 pg/mL.
We next asked whether lactoferricin peptides affected LPS-stimulated IL-6
production by RAW 264.7 cells, THP-1 cells, and BMDMs. ELISA analysis of 24 h
culture supernatants from LPS-stimulated RAW 264.7 cells, THP-1 cells, and BMDMs
that were treated with bovine lactoferricin showed a significant decrease in IL-6
production in comparison to the untreated control (Fig. 1G–I, p
0.05). Neither human nor mouse lactoferricin affected IL-6 production by
LPS-stimulated RAW 264.7 cells or BMDMs. All three lactoferricin peptides
decreased LPS-induced IL-6 production by THP-1 cells (Fig. 1H, p
0.05). Lactoferricin peptides had a similar effect on IL-6 mRNA expression by
LPS-stimulated macrophages (data not shown). Lactoferricin treatment without LPS
stimulation did not influence the production of TNF- or IL-6 by RAW
264.7 cells, THP-1 cells, or BMDMs (data not shown).
3.2 Lactoferricin peptides decrease NO production in LPS-stimulated
macrophages
To determine if lactoferricin peptides inhibited the LPS-stimulated production
of NO, RAW 264.7 cells and BMDMs were stimulated with 500 ng/mL LPS in the
absence or presence of different lactoferricin peptides for 24 h. Culture
supernatants were tested for the presence of NO using Griess reagent. A
significant decrease in LPS-induced NO production was observed in RAW 264.7 cells
treated with either bovine or human lactoferricin (Fig. 2A, p 0.01).
A decrease in NO production upon LPS-stimulation was observed with the addition
of bovine, mouse, or human lactoferricin in BMDMs (Fig. 2B, p 0.05).
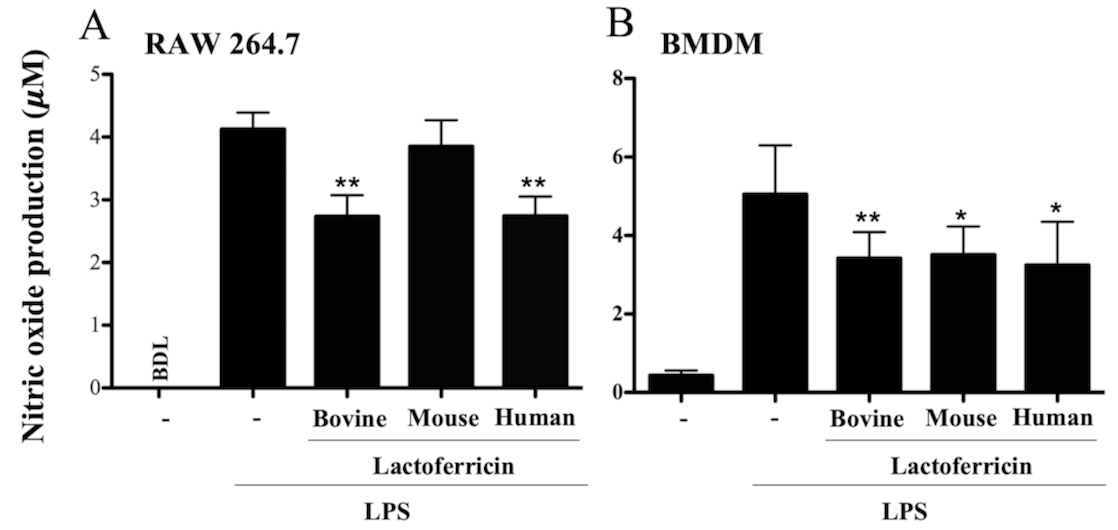 Fig. 2.
Fig. 2.
Lactoferricin peptides decrease nitric oxide production in
LPS-stimulated macrophages. (A) RAW 264.7 cells and (B) BMDMs were treated with
500 ng/mL of LPS alone or in combination with 5 M bovine, mouse, or
human lactoferricin for 24 h. Supernatants were collected and used in a Griess
assay to measure nitric oxide production. Data are the mean of 4 independent
experiments SEM; * denotes p 0.05 and ** p 0.01
compared to LPS treatment as determined by ANOVA with Dunnett’s multiple
comparisons post-test. BDL, below detectable levels.
3.3 Lactoferricin peptides decrease inducible NO synthase expression
in LPS-stimulated macrophages
To determine whether lactoferricin-mediated inhibition of NO was
associated with decreased iNOS expression, RAW 264.7 and BMDM cells were treated
with 500 ng/mL LPS alone or in combination with 5 M bovine, mouse,
or human lactoferricin for 4 h prior to RNA isolation. Levels of iNOS expression
were normalized to the LPS control since iNOS mRNA was below the level of
detection in untreated cells. A significant decrease in LPS-induced iNOS mRNA
expression was seen in RAW 264.7 cells treated with either bovine or human
lactoferricin (Fig. 3A, p 0.05). When LPS-stimulated BMDMs were
treated with either mouse or human lactoferricin there was a significant
reduction in iNOS expression in comparison to LPS treatment alone (Fig. 3B,
p 0.05).
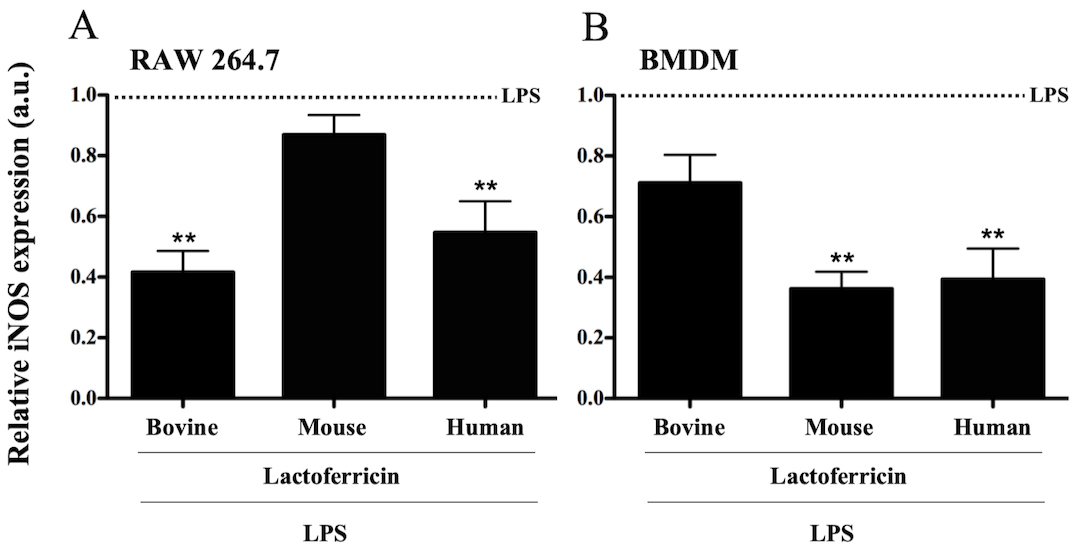 Fig. 3.
Fig. 3.
Lactoferricin peptides decrease inducible nitric oxide synthase
expression in LPS-stimulated macrophages. (A) RAW 264.7 cells and (B) BMDMs were
treated with 500 ng/mL of LPS in combination with 5 M bovine,
mouse, or human lactoferricin for 4 h. RNA was isolated for cDNA synthesis, and
qPCR was performed to determine levels of iNOS expression. Data are expressed as
a relative value of LPS induced iNOS expression (iNOS in lactoferricin-treated
cells divided by iNOS in cells treated with LPS alone). Data are the mean of 3
independent experiments SEM; * denotes p 0.05 and **
p 0.01 compared to LPS positive control as determined by ANOVA with
Dunnett’s multiple comparisons post-test. a.u. denotes arbitrary units.
3.4 Lactoferricin peptides reduce expression of phosphorylated
IB in LPS-stimulated macrophages
To determine whether lactoferricin peptides influenced LPS-induced
pro-inflammatory signaling cascades, RAW 264.7 and THP-1 cells were treated with
LPS in the absence or presence of bovine, mouse, or human lactoferricin, and
western blot analysis of cell lysates for phospho-IB
expression was conducted. Bovine lactoferricin reduced LPS-induced
phospho-IB expression in RAW 264.7 cells at 1 h but not at 30
min or 4 h (Fig. 4A). Subsequent western blot analysis was conducted after 1 h
incubation with peptide, LPS, or LPS plus peptide. Treatment of RAW 264.7 cells
with the combination of LPS and lactoferricin peptides led to a decrease in
phospho-IB expression in comparison to LPS treatment alone,
whereas treatment with peptide alone had no significant effect on
phospho-IB expression (Fig. 4B–C p 0.01). In a
similar manner to RAW 264.7 cells, there was a significant decrease in THP-1 cell
expression of LPS-induced phospho-IB following treatment with
bovine, mouse, or human lactoferricin (Fig. 4D–E, p 0.01).
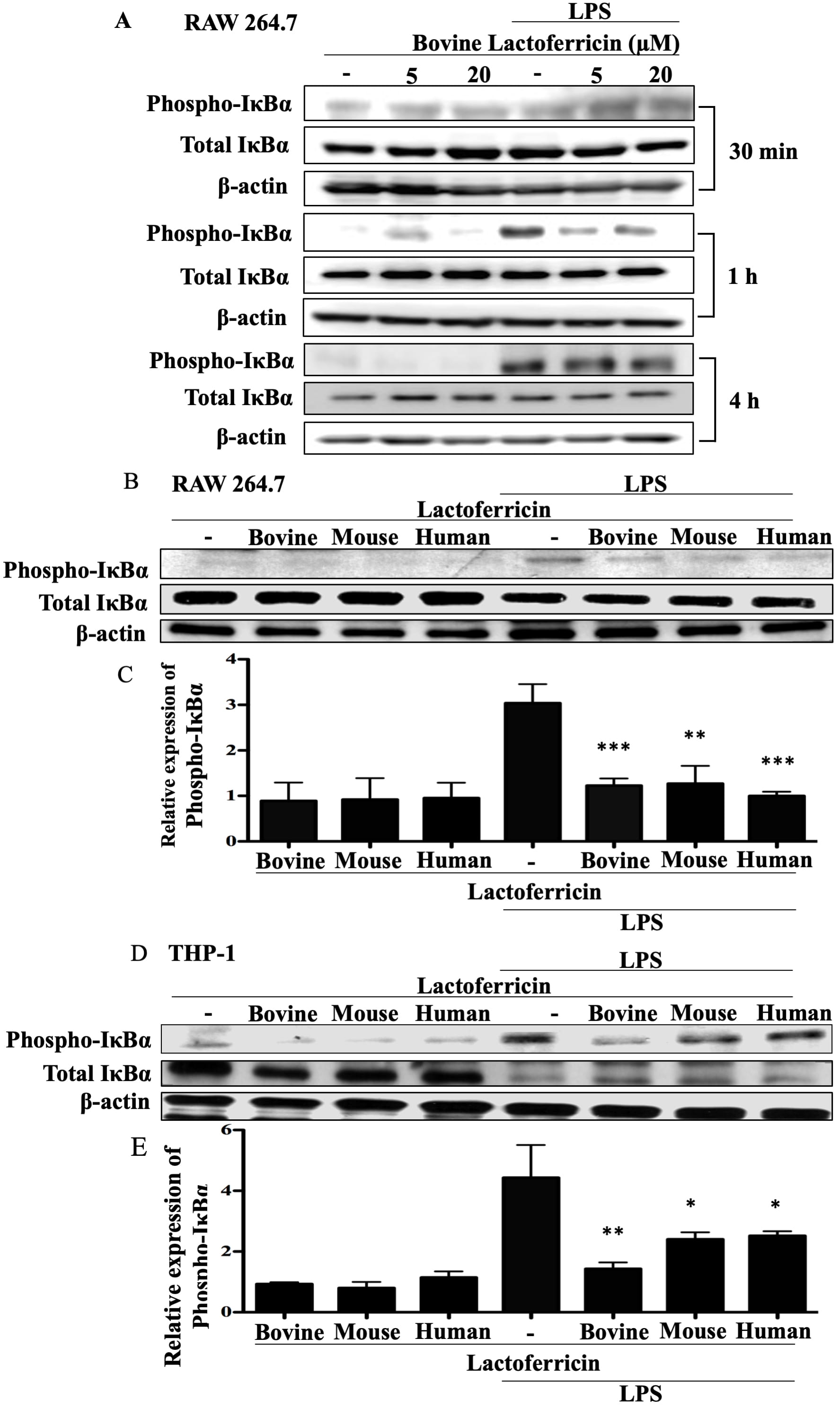 Fig. 4.
Fig. 4.
Lactoferricin peptides reduce expression of phosphorylated
IB in LPS-stimulated macrophages. (A) RAW 264.7 cells were
treated with the indicated concentrations of bovine lactoferricin and 100 ng/mL
LPS for the indicated times. Cell lysates were collected and protein expression
was determined by western blotting. Nitrocellulose membranes were probed with the
indicated antibodies and corresponding secondary antibodies. Data shown are
representative of two independent time-course experiments. (B), (C) RAW 264.7 and
(D), (E) THP-1 cells were incubated with 100 ng/mL LPS, 5 M bovine,
mouse, or human lactoferricin, or a combination of LPS and one species-specific
lactoferricin peptide as indicated. Cell lysates were collected after 1 h and
used in western blotting as described in (A). Data shown are one representative
blot. (B), (D) One representative western blot and (C), (E) the mean density of
phosphorylated IB normalized to untreated controls and to
total IB and -actin from 4 (RAW 264.7 cells) or 3
(THP-1 cells) independent experiments SEM; ** denotes p 0.01
and *** denotes p 0.001 compared to LPS alone treatments as
determined by ANOVA with Dunnett’s multiple comparisons post-test.
3.5 Lactoferricin peptides inhibit the nuclear translocation of
NF-B in LPS-stimulated macrophages
To determine whether lactoferricin peptides inhibited the nuclear translocation
of NF-B, RAW 264.7 and THP-1 cells were treated with LPS in the absence
or presence of bovine, mouse, or human lactoferricin for 1 h and stained with
anti-p65 (a subunit of NF-B) antibody and the nuclear stain DAPI. Cells
were visualized using fluorescent microscopy. LPS stimulation of RAW 264.7 and
THP-1 macrophages resulted in a prominent nuclear translocation of p65 (Fig. 5A–B). RAW 264.7 cells stimulated with LPS in the presence of bovine, mouse, or
human lactoferricin showed reduced nuclear-localized p65 in comparison to the
LPS-only control (Fig. 5A). Nuclear translocation of p65 in THP-1 cells was also
inhibited by lactoferricin treatment (Fig. 5B). Species-specific lactoferricin
treatments therefore inhibited the LPS-induced translocation of NF-B
into the nucleus of mouse and human macrophages. Western blot analysis showed
that lactoferricin peptides alone did not affect cytosolic p65 levels in
LPS-stimulated RAW 264.7 and THP-1 macrophages in comparison to cells treated
with LPS alone (data not shown).
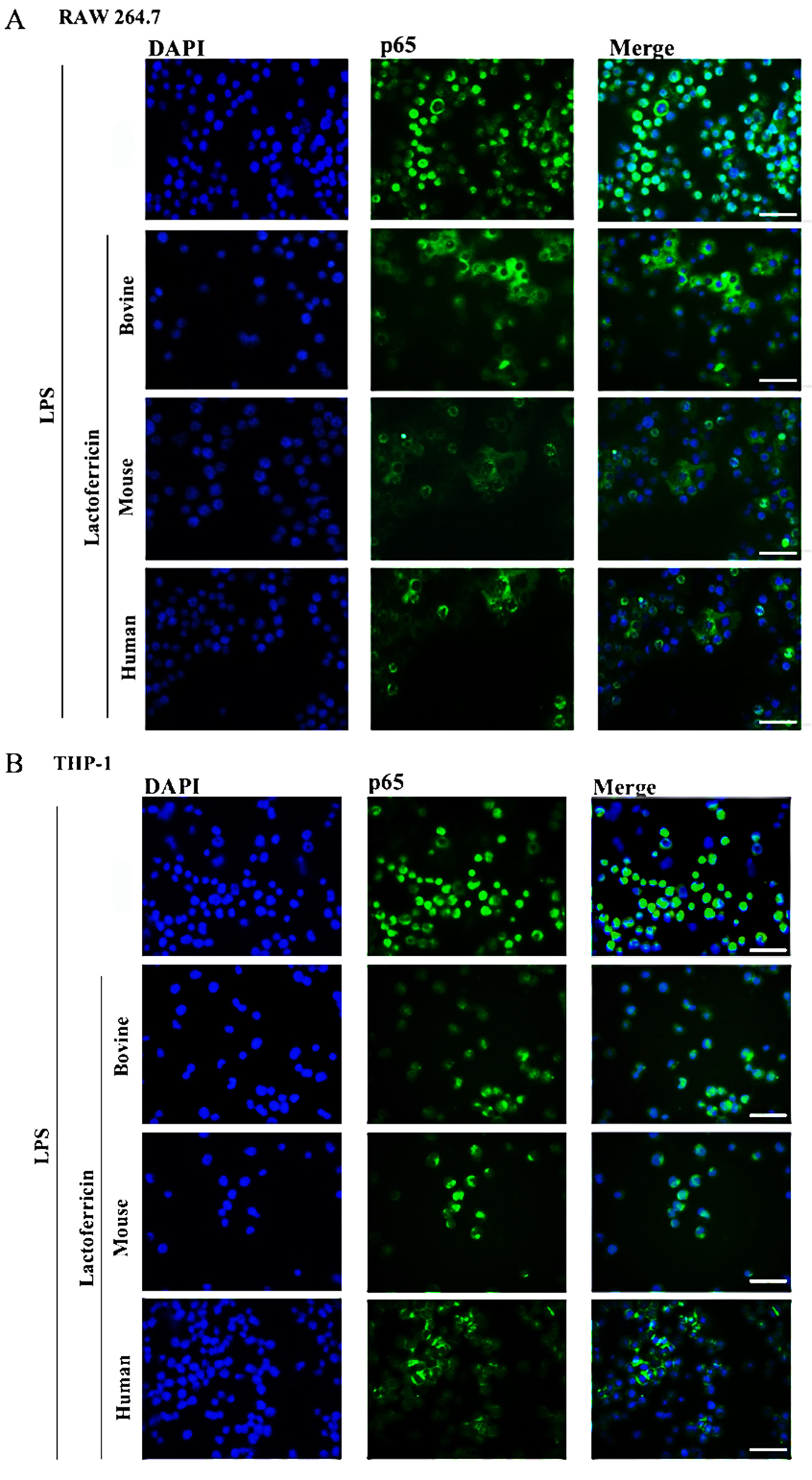 Fig. 5.
Fig. 5.
Lactoferricin peptides inhibit the nuclear translocation of
NF-B in LPS-stimulated macrophages. (A) RAW 264.7 cells and (B) THP-1
cells were treated with 100 ng/mL LPS alone or in combination with 5
M bovine, mouse, or human lactoferricin for 1 h. Cells were fixed
and incubated with nuclear stain, DAPI (30 M), and rabbit anti-p65
antibody with secondary goat-anti-rabbit Alexa Fluor 488 ®
conjugate, then imaged using fluorescent microscopy. Images are representative of
two independent experiments. Scale bar represents 40 m.
3.6 Lactoferricin peptides reduce ERK phosphorylation in
LPS-stimulated macrophages
To determine if lactoferricin peptides target MAPK signaling pathways, western
blot analysis was conducted using lysates from RAW 264.7 and THP-1 cells treated
with lactoferricin alone, LPS alone, or LPS plus lactoferricin. RAW 264.7 cells
treated with either bovine or mouse lactoferricin showed a significant reduction
in LPS-induced phospho-ERK 1/2 expression in comparison to LPS treatment alone
(Fig. 6A–B, p 0.05). THP-1 cells showed a similar trend of reduced
phospho-ERK 1/2, demonstrating a decrease in LPS-induced ERK 1/2 expression with
peptide treatment (Fig. 6C–D, p 0.05).
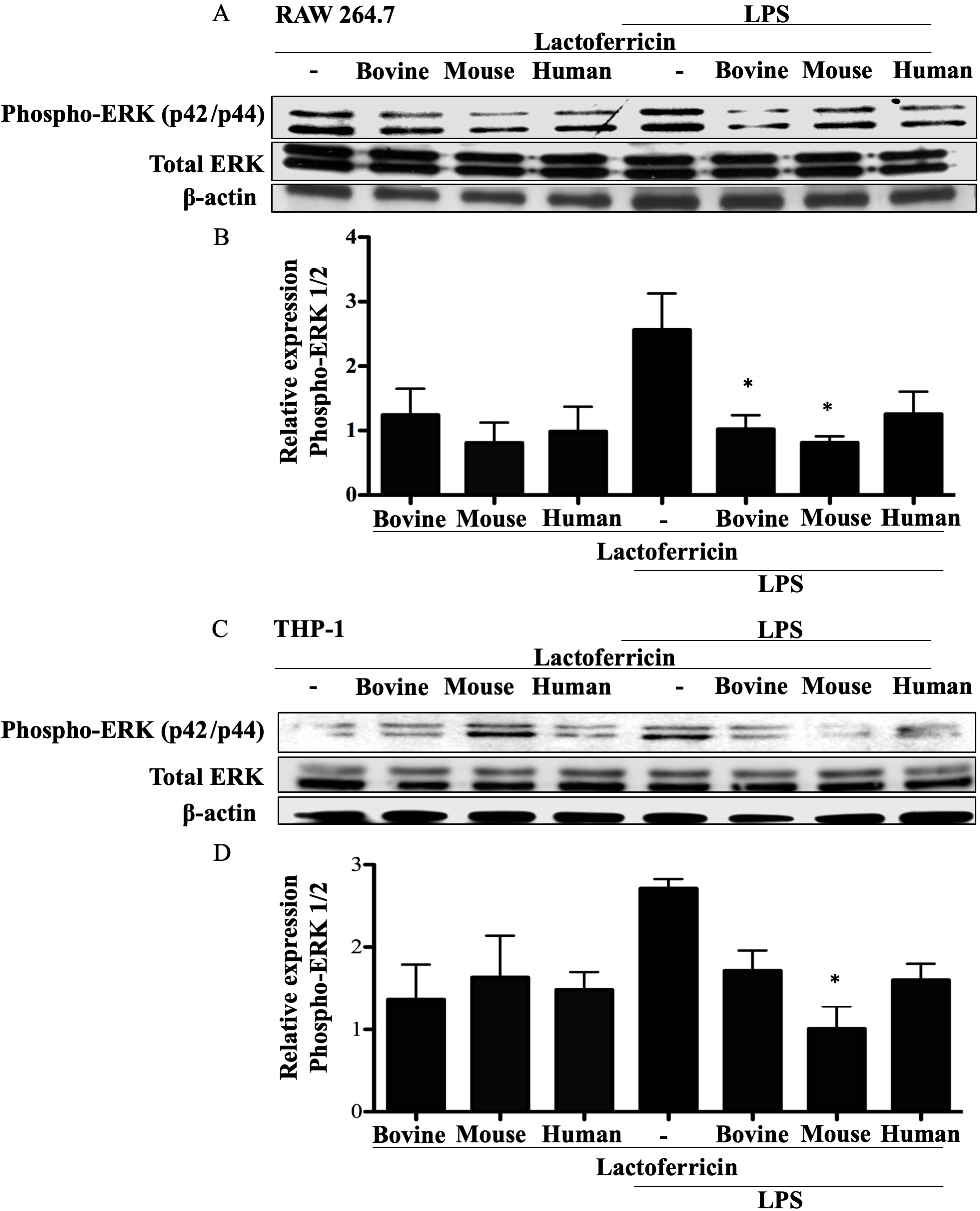 Fig. 6.
Fig. 6.
Lactoferricin peptides inhibit ERK phosphorylation in
LPS-stimulated macrophages. (A), (B) RAW 264.7 and (C), (D) THP-1 cells were
incubated with 100 ng/mL LPS, 5 M bovine, mouse, or human
lactoferricin, or a combination of LPS and one species-specific lactoferricin as
indicated. Cell lysates were collected and protein expression was determined
using western blotting. Nitrocellulose membranes were probed with the indicated
antibodies and the appropriate secondary antibodies. (A), (C) Data shown are from
one representative western blot. (B), (D) The mean density of phosphorylated ERK
1/2 normalized to untreated controls and to total ERK 1/2 and -actin
from 4 (RAW 264.7 cells) or 3 (THP-1 cells) independent experiments SEM.
3.7 Lactoferricin peptides decrease c-Jun phosphorylation in
LPS-stimulated mouse macrophages
Western blot analysis of phospho-c-Jun expression in lactoferricin-treated,
LPS-stimulated RAW 264.7 cells demonstrated a significant reduction in
phospho-c-Jun in comparison to cells treated with LPS alone (Fig. 7A–B,
p 0.05). However, LPS-stimulated THP-1 macrophages treated in a
similar manner did not show a significant decrease in phospho-c-Jun expression
following lactoferricin treatment (Fig. 7C–D).
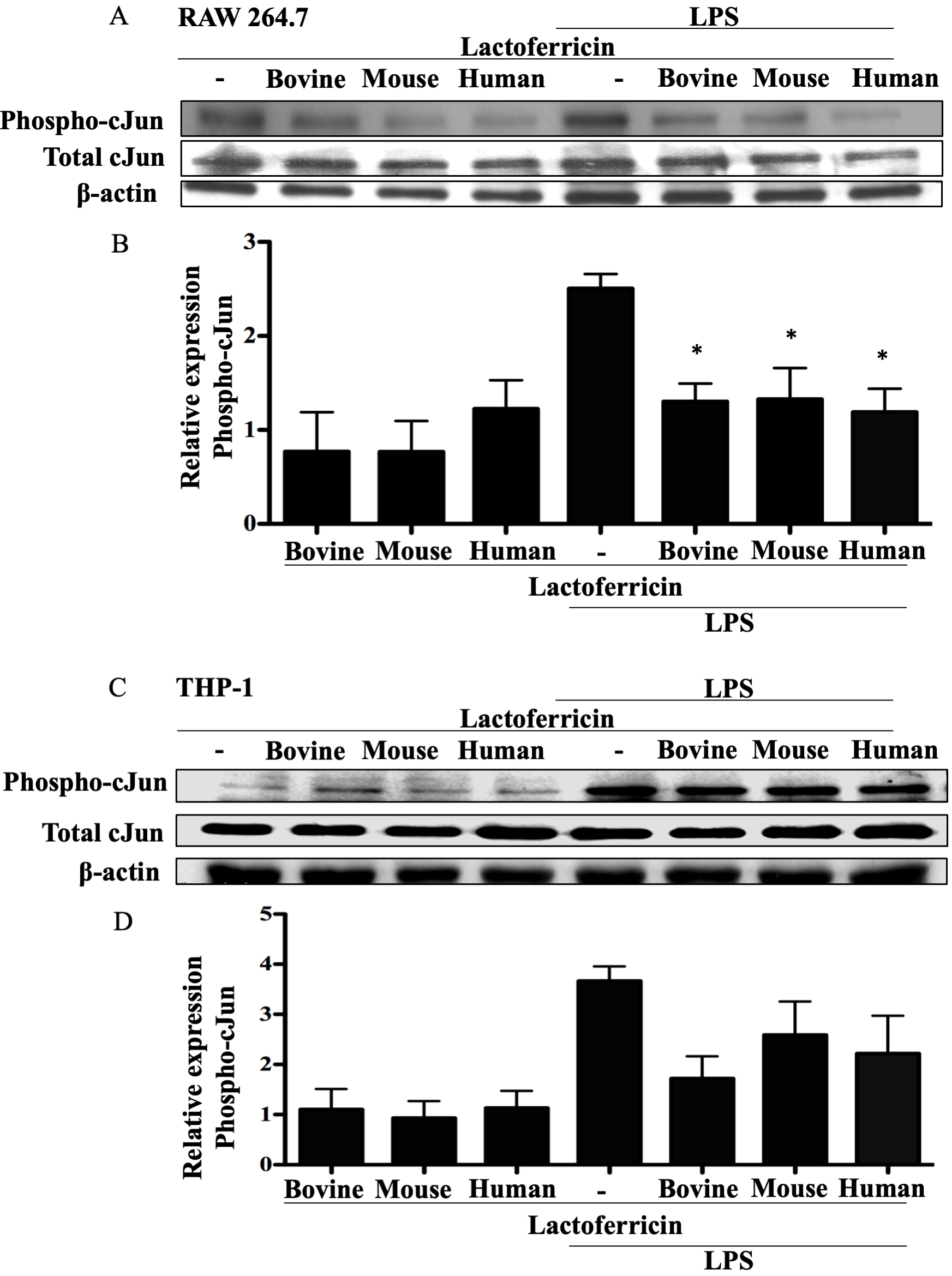 Fig. 7.
Fig. 7.
Lactoferricin peptides inhibit c-Jun phosphorylation in
LPS-stimulated mouse macrophages. (A), (B) RAW 264.7 and (C), (D) THP-1 cells
were incubated with 100 ng/mL LPS, 5 M bovine, mouse, or human
lactoferricin, or a combination of LPS and one species-specific lactoferricin
peptide, as indicated. Cell lysates were collected and protein expression was
determined using western blotting. Nitrocellulose membranes were probed with the
indicated antibodies and the appropriate secondary antibodies. (A), (C) Data
shown are from one representative western blot. (B), (D) The mean density of
phosphorylated c-Jun normalized to untreated controls and to total c-Jun and
-actin from 4 (RAW 264.7 cells) or 3 (THP-1 cells) independent
experiments SEM.
3.8 Lactoferricin peptides decrease cadmium-induced TNF-
production in macrophages
To determine whether species-specific lactoferricin peptides were
able to downregulate a pro-inflammatory response induced by a stimulus other than
LPS, RAW 264.7 and THP-1 cells were treated with CdCl alone or in the
presence of bovine, mouse, or human lactoferricin. After 24 h, culture
supernatants were collected, and ELISA was used to measure TNF-. As
with LPS stimulation, there was a significant decrease in CdCl-induced
TNF- production by THP-1 (Fig. 8A, p 0.05) and RAW 264.7
(Fig. 8B–C, p 0.05) cells treated with bovine, mouse, or human
lactoferricin. Lactoferricin peptides therefore downregulated pro-inflammatory
TNF- induced by nonmicrobial activation with CdCl.
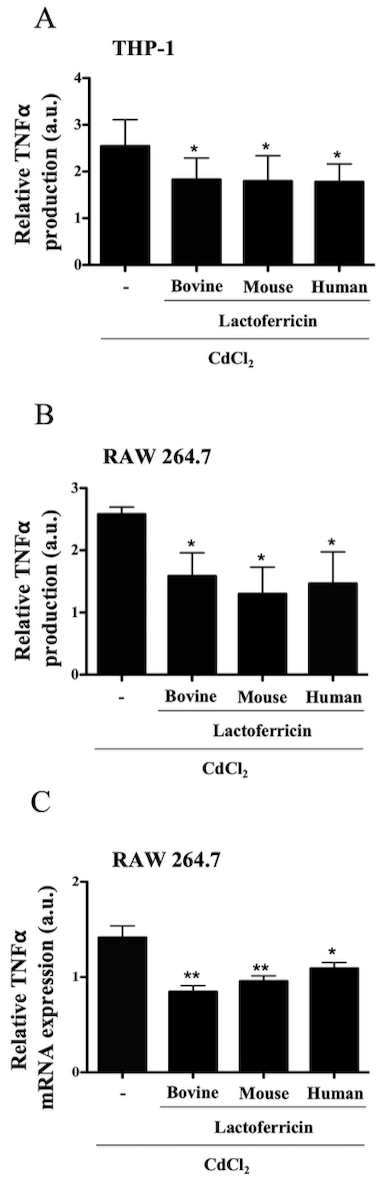 Fig. 8.
Fig. 8.
Lactoferricin peptides decrease cadmium-induced TNF-
production in macrophages. (A) THP-1 cells and (B) RAW 264.7 cells were treated
with 10 M CdCl alone or in combination with 5 M
bovine, mouse, or human lactoferricin, for 24 h. Supernatants were collected and
ELISA was used to determine relative levels of TNF- production. (C) RNA
was isolated from RAW 264.7 cells treated with 10 M CdCl
alone or in combination with 5 M bovine, mouse, or human
lactoferricin, for 4 h, cDNA was synthesized and q-PCR was performed to determine
relative levels of TNF- expression normalized to untreated control.
Data show are the mean of 3-5 independent experiments SEM. ELISA data was
normalized to untreated controls: 108 pg/mL and 118 pg/mL, respectively; *
denotes p 0.05 compared to LPS control as determined by ANOVA with
the Dunnett’s multiple comparisons post test; a.u. denotes arbitrary units.
4. Discussion
Previous studies of AMPs have often focused on the direct antimicrobial
properties of these cationic peptides; however, more recently the focus has
shifted towards their direct immunomodulatory features. To our knowledge, this is
the first study to demonstrate the differential immunomodulatory efficacy of
species-specific lactoferricin peptides and uncover several potential mechanisms
to account for inhibition of inflammatory responses by LPS-stimulated
macrophages. Lactoferrin released by neutrophils at sites of inflammation can be
as high as 2.5 M concentration [29]. Although lactoferricin
concentrations at sites of inflammation have not yet been reported in the
literature, proteolytic hydrolysis of lactoferrin is predicted to generate
lactoferricin at sites of inflammation, albeit at concentrations well below 2.5
M.
Given that TNF- is a prototypical pro-inflammatory cytokine released
at the onset of inflammation, the production of this cytokine was evaluated in
lactoferricin-treated macrophages. Our findings suggest that mouse and human
macrophages are susceptible to the anti-inflammatory effects of lactoferricin
peptides, albeit to varying degrees. Bovine lactoferricin decreased
TNF- production by mouse BMDMs and RAW 264.7 cells; however, mouse and
human lactoferricin had no significant effect on TNF- production by
these cells. In contrast, there was decreased LPS-induced TNF-production by human THP-1 cells treated with all three peptides. A similar effect
was seen on the production of IL-6 by LPS-stimulated macrophages. Bovine
lactoferricin consistently downregulated pro-inflammatory cytokine production in
mouse and human macrophages.
Supporting the observations with pro-inflammatory cytokines, lactoferricin was
also able to downregulate expression of TNF- mRNA in macrophages. As
before, bovine lactoferricin exhibited the most potent anti-inflammatory effect,
suppressing LPS-induced mRNA expression in mouse BMDMs and RAW 264.7 macrophages
as well as human THP-1 macrophages. The TNF- mRNA expression profile
mirrored that of cytokine production in THP-1 cells; however, this was not the
case for RAW 264.7 cells. Possible reasons for this discrepancy include
regulation of LPS-induced TNF- on multiple levels (post-transcriptional
and/or translational modifications) or a kinetics issue with lactoferricin
administration (use of a different time point for cytokine production).
NO is another important pro-inflammatory mediator produced by LPS-stimulated
macrophages [30, 31]. As with pro-inflammatory cytokine production, lactoferricin
peptides decreased LPS-induced NO production in RAW 264.7 cells and BMDMs, with
bovine and human lactoferricin having the greatest inhibitory effect. iNOS
expression experiments demonstrated the capacity for lactoferricin peptides to
downregulate another prominent pro-inflammatory marker in macrophages.
Lactoferricin peptides affected the expression of iNOS, the enzyme that
synthesizes NO via the conversion of arginine to citrulline in the cytoplasm
[30], in a manner that mirrored the effect on NO production in RAW 264.7 cells
and BMDMs. NO production in THP-1 cells was not investigated as previous studies
have shown that these cells do not produce detectable levels of NO when
stimulated with LPS or other microbial products [32, 33].
There are several possible reasons for the differential effects of
lactoferricin peptides from different species on pro-inflammatory mediator
production by macrophages [34, 35, 36, 37]. As seen in Table 1, the amino acid
composition between lactoferricin peptides from bovine, mouse, and human sources
contain subtle differences. The functional diversity that exists between each of
these peptides could lie in their different amino acid compositions. Although no
studies have directly looked at the structure-function relationship between these
three particular peptides in an immunomodulatory context, several previous
investigations have highlighted the importance of peptide structure in host
defense and antimicrobial effects of AMPs [38, 39, 40]. The secondary structure of
bovine lactoferricin and human lactoferricin in aqueous solution is an
antiparallel beta sheet [35, 36, 37]. It is reasonable to assume that mouse
lactoferricin also assumes an antiparallel beta sheet in an aqueous environment.
Among the three lactoferricin peptides investigated in the current study, bovine
lactoferricin is known to have the greatest antimicrobial activity [22]. Since
bovine lactoferricin and human lactoferricin have the same secondary structure,
increased antibacterial activity of bovine lactoferricin over human lactoferricin
is believed to be due to different distribution of charge surrounding the
hydrophobic core [34]. For example, bovine lactoferricin contains two Trp
residues whereas mouse and human lactoferricincontain one Trp residue. Like other
antimicrobial peptides [41], bovine lactoferricin binds to negatively charged
cell surface structures [42]; however, at the present time the specific
structures that lactoferricin binds are not known, nor is it known for certain
how lactoferricin peptides are internalized by macrophages, although it is likely
via endocytosis of lactoferricin-bound and crosslinked structures.
The most prominent of the inflammatory signaling pathways induced by LPS is the
IB-NF-B pathway [4]. In this study, bovine
lactoferricin transiently inhibited IB phosphorylation in
LPS-stimulated macrophages, with the greatest suppressive effect seen 1 h
post-treatment. All three lactoferricin peptides significantly decreased
phospho-IB expression in LPS-stimulated RAW 264.7 and THP-1
cells. Peptide treatment in the absence of LPS stimulation did not affect
IB signaling. Previous studies of other AMPs have revealed
similar downregulation of phospho-IB in macrophage cell lines
[43, 44, 45]. As phospho-IB is indicative of a pro-inflammatory
response due to the movement of NF-B into the nucleus, NF-B
nuclear translocation was then monitored in LPS-stimulated macrophages. The
inhibitory effect of lactoferricin peptides on nuclear translocation of p65 is
consistent with the inhibition of IB phosphorylation by these
peptides, which was approximately the same for bovine, human and mouse
lactoferricin peptides. These observations are consistent with other studies
demonstrating AMP regulation of inflammatory responses via inhibition of
NF-B nuclear translocation [46, 47, 48].
Past studies implicate MAPK signaling in AMP-mediated immunomodulation and the
selective manner in which these peptides can induce a response [49, 50, 51, 52]. The
present study shows that LPS-induced activation of ERK-1/2 signaling is decreased
in lactoferricin-treated RAW 264.7 and THP-1 cells. ERK-1/2 induces further
downstream signaling components such as c-Jun and c-Fos subunits of the AP-1
transcription factor family in macrophages [53, 54, 55]. Lactoferricin peptides also
decreased expression of phosphorylated c-Jun in macrophages. This is consistent
with the anti-inflammatory role of lactoferricin peptides as AP-1, like
NF-B, regulates the transcription of pro-inflammatory mediators when
macrophages are stimulated by LPS [56, 57].
CdCl, a heavy metal carcinogen, induces
mitochondrial-generated ROS in macrophages, which stimulates synthesis of
pro-inflammatory cytokines such as TNF-. AMPs such as human and
mouse-derived cathelicidins show antioxidant properties by reducing harmful
oxidative burden, similar to the effect of glutathione [47, 58]. Given that
CdCl leads to the production of ROS, which induces TNF-, and all
three peptides were able to downregulate CdCl-induced TNF-
production in mouse and human macrophages, it is possible that the lactoferricin
peptides act as antioxidants. One study demonstrates that the number of disulfide
bridges and cysteine residues correlate to the overall antioxidant effect of some
AMPs [59]. Although each lactoferricin peptide decreased CdCl-induced
TNF- production in macrophages, it is unlikely to involve disulfide
bridges since the lactoferricins used in this study had a linear rather than a
cyclic configuration.
A contributing factor to the decrease in pro-inflammatory cytokine production by
macrophages in the presence of lactoferricin peptides is the ability of
positively charged lactoferricin to bind negatively charged LPS, thereby
inhibiting endotoxin binding to TLR-4. Other studies have highlighted the
importance of endotoxin binding of AMPs to inhibit inflammation [43, 60];
however, given the selective manner in which lactoferricin targets cell signaling
and its ability to decrease CdCl-induced TNF- production, it is
likely that lactoferricin exerts its anti-inflammatory effect by a mechanism
other than simply binding to LPS and preventing TLR-4 stimulation.
In future studies it will be important to determine whether these
lactoferricin peptides have an anti-inflammatory effect in animal models. The
carrageenan-air pouch model would be appropriate to gain insight into the
potential effects of lactoferricin peptides in an acute in vivo
inflammatory response [61, 62]. A mouse model of colitis should also be studied
to determine the effects of lactoferricin in a chronic inflammatory setting
[63, 64, 65]. Enhancement of the current lactoferricin peptides, specifically bovine
lactoferricin, through synthetic alteration of their amino acid sequence may lead
to a peptide that is more potent as an immune regulator.
5. Conclusions
Inflammation is a complicated process consisting of pro-inflammatory and
anti-inflammatory components that allow for an appropriate response to microbial
pathogens and other insults without causing excessive tissue damage. Proper
inflammation results in eradication of the source of stimulation, the cleanup of
pathogenic and cellular debris, and repair of tissue damage in order to return to
homeostasis. When these tightly regulated mechanisms fail, inflammation persists
and can become destructive to the host. To circumvent the development of chronic
inflammation or address an already existing inflammatory condition, immune
regulators are required. Regulators that target a plethora of pro-inflammatory
mediators benefit the host and may offer a protective effect in a wide variety of
inflammatory diseases such as sepsis and colitis. Such regulators can include
certain AMPs, which offer great potential as therapeutic agents owing to their
relatively non-toxic nature and non-mutagenic properties in eukaryotic cells. Of
the three lactoferricin peptides studied, bovine lactoferricin consistently had
the greatest anti-inflammatory effects in macrophages, mediated via
downregulation of LPS-induced TNF-, IL-6, iNOS, and NO (Fig. 9).
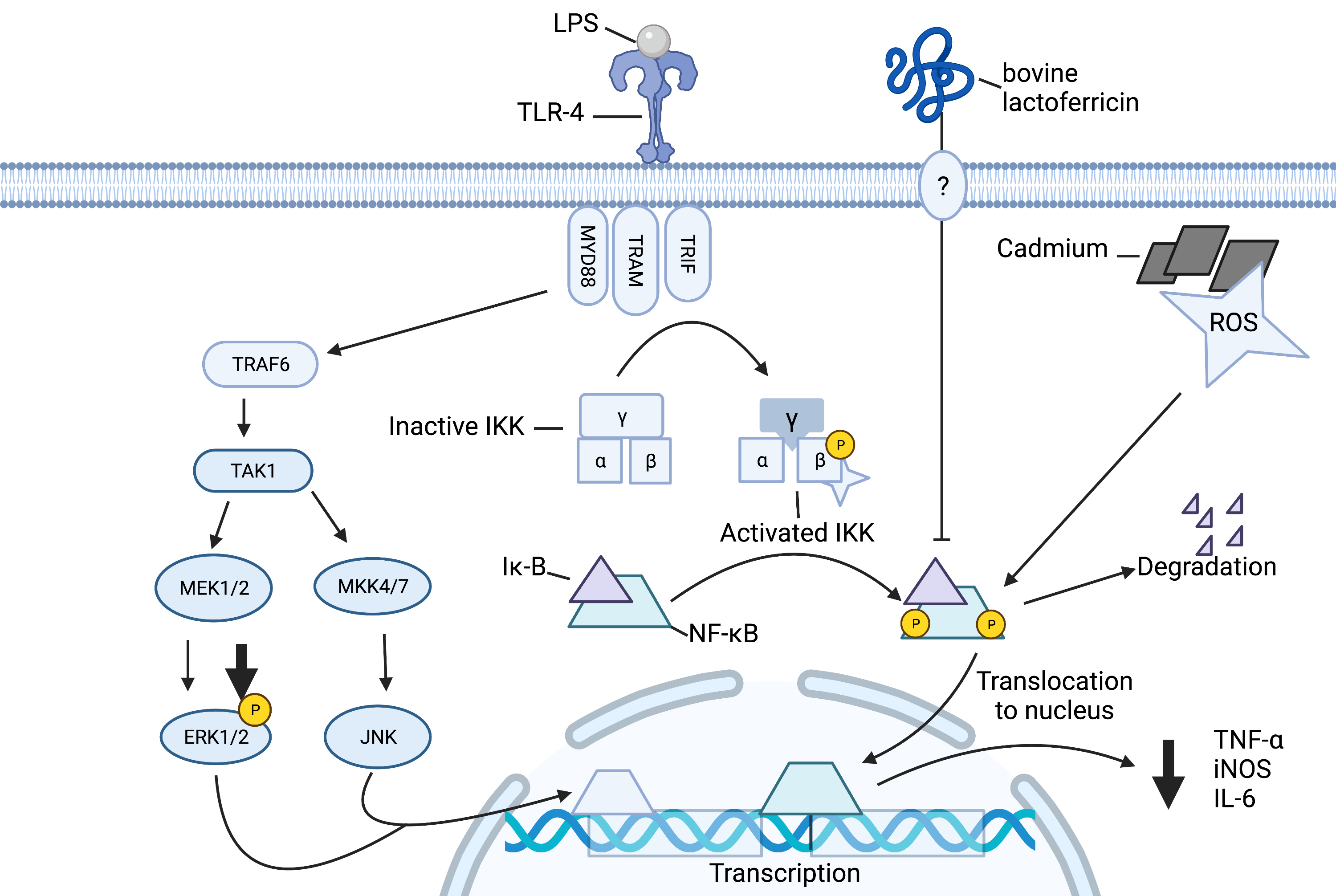 Fig. 9.
Fig. 9.
Schematic of outlined anti-inflammatory effects of lactoferricin
in macrophages. Lactoferricin peptides inhibit LPS-induced phosphorylation of
IB and nuclear translocation of NF-B. This
subsequently inhibits the synthesis and release of proinflammatory mediators
TNF-, IL-6, and nitric oxide. Lactoferricin peptides prevent the
cadmium-induced production of TNF-. Lactoferricin decreases LPS-induced
phosphorylation of ERK and c-Jun. Image created with biorender.com.
Although there is still much to be explored, this study has shown the selective
influence of species-specific lactoferricin peptides on several important
macrophage-associated inflammatory processes. The lactoferricin peptides,
especially of bovine origin, may therefore have therapeutic potential in the
context of downregulating excessive or inappropriate inflammatory responses.
Author contributions
AM and DH designed the research study. AM performed the research. DH
and MC provided help and advice on protocols. AM, RC, DH, and MC analyzed the
data. AM and RC made the summary figure. AM, RC, and MC wrote the manuscript. All
authors contributed to editorial changes in the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to participate
Ethics approval for animal use to collect bone marrow derived macrophages
(approval number 16-113) was obtained from the Dalhousie University Committee on
Laboratory Animals.
Acknowledgment
THP-1 human monocytic-like cells were kindly provided by Brent Johnston
(Dalhousie University, Halifax, NS).
Funding
This research was funded by a NSERC Discovery Grant (D.H., RGPIN2017-05339)) and
the Acadia University Research Fund (M.C.).
Conflict of interest
The authors declare no conflict of interest. MC is serving as the guest editor
of this journal. We declare that MC had no involvement in the peer review of this
article and has no access to information regarding its peer review. Full
responsibility for the editorial process for this article was delegated to GP.
 Fig. 1.
Fig. 1. Fig. 2.
Fig. 2. Fig. 3.
Fig. 3. Fig. 4.
Fig. 4. Fig. 5.
Fig. 5. Fig. 6.
Fig. 6. Fig. 7.
Fig. 7. Fig. 8.
Fig. 8. Fig. 9.
Fig. 9.








