1 Department of Orthopaedics, The First Affiliated Hospital of Kunming Medical University, 650032 Kunming, Yunnan, China
†These authors contributed equally.
Academic Editor: Ioanna-Katerina Aggeli
Abstract
Background: Long non-coding RNA (lncRNA) hypoxia
inducible factor 1
Keywords
- HIF1A-AS1
- HUVECs
- miRNA
- mRNA
- cardiovascular diseases
Cardiovascular disease (CVD) encompasses a variety of conditions that affect the heart and blood vessels, such as cerebrovascular disease, peripheral artery, and irregular heartbeat [1]. It is a serious threat to human health, characterized by a high prevalence, high disability rate, and high mortality. The number of people who die of CVD and cerebrovascular disease annually is up to 15 million, ranking first among all causes of death. Vascular endothelial cells (VECs), which are located in the innermost part of blood vessels, are vulnerable to stimuli. The apoptosis of VECs is closely correlated with numerous cardiovascular diseases (CVDs) such as arteriosclerosis, thrombus formation, and plaque erosion [2, 3]. Due to the complex pathogenesis and serious complication, an increasing number of studies has focused on the etiology of VEC injury. However, the underlying mechanisms remains unclear, hindering the prevention and treatment of related diseases. Hence, it is necessary to identify novel apoptosis-related therapeutic targets in VECs for CVD.
In recent years, non-coding RNAs (ncRNAs), mainly circular RNAs, microRNAs (miRNAs), and long ncRNAs (lncRNAs), have been widely investigated [4]. LncRNA is widely expressed and plays an essential role in numerous life activities such as regulation of the cell cycle and cell differentiation. The abnormal expression or function of lncRNA is closely related to the occurrence of human diseases including cancer, immune responses, and other related diseases [5, 6]. The biogenesis of lncRNA is associated with its specific subcellular localization and function. LncRNA is a potential biomarker that can be applied to clinical targeting and has potential therapeutic effects [7].
A number of noncoding RNAs, including lncRNAs and miRNAs, play pivotal roles in
the progression of vascular diseases [8, 9, 10, 11, 12]. Some of them are emerging as
diagnostic biomarkers or therapeutic targets due to their specific role in some
CVDs [13, 14, 15, 16]. LncRNA hypoxia inducible factor 1
In the current study, we elucidated the role of HIF1A-AS1 in HUVECs and its underlying mechanisms. The results could provide insights into potential research directions for CVD treatment in the future. First, as previously reported [24], we simulated cardiovascular occlusion by treating HUVECs with PA. qPCR confirmed the successful transfection of plasmids containing HIF1A-AS1 or short hairpin RNA (shRNA). Using flow cytometry, Cell Counting Kit-8 (CCK-8), transwell and wound healing assays, we found that this lncRNA promoted apoptosis and reduced proliferation, migration, and invasion. Moreover, miRNA sequencing (miRNA-seq) results showed that HIF1A-AS1 globally mediated the expression of miRNAs. Bioinformatics analysis indicated that multiple target genes of differentially expressed miRNAs (DEmiRNAs) were involved in cell metabolism and apoptosis. Subsequently, RNA sequencing (RNA-seq) and bioinformatics analysis was applied to identify differentially expressed genes (DEGs). Interesting, quite a few DEGs overlapped with the target genes of DEmiRNAs. Finally, a co-expression network showed the strength of the correlation between expression levels of DEmiRNAs and some initial DEGs. These data suggest that HIF1A-AS1 regulates the function of HUVECs by not only directly regulating the expression of some genes but also by influencing some miRNAs to indirectly mediate the expression of mRNA. The experimental process and relevant mechanism are schematically illustrated in Scheme.
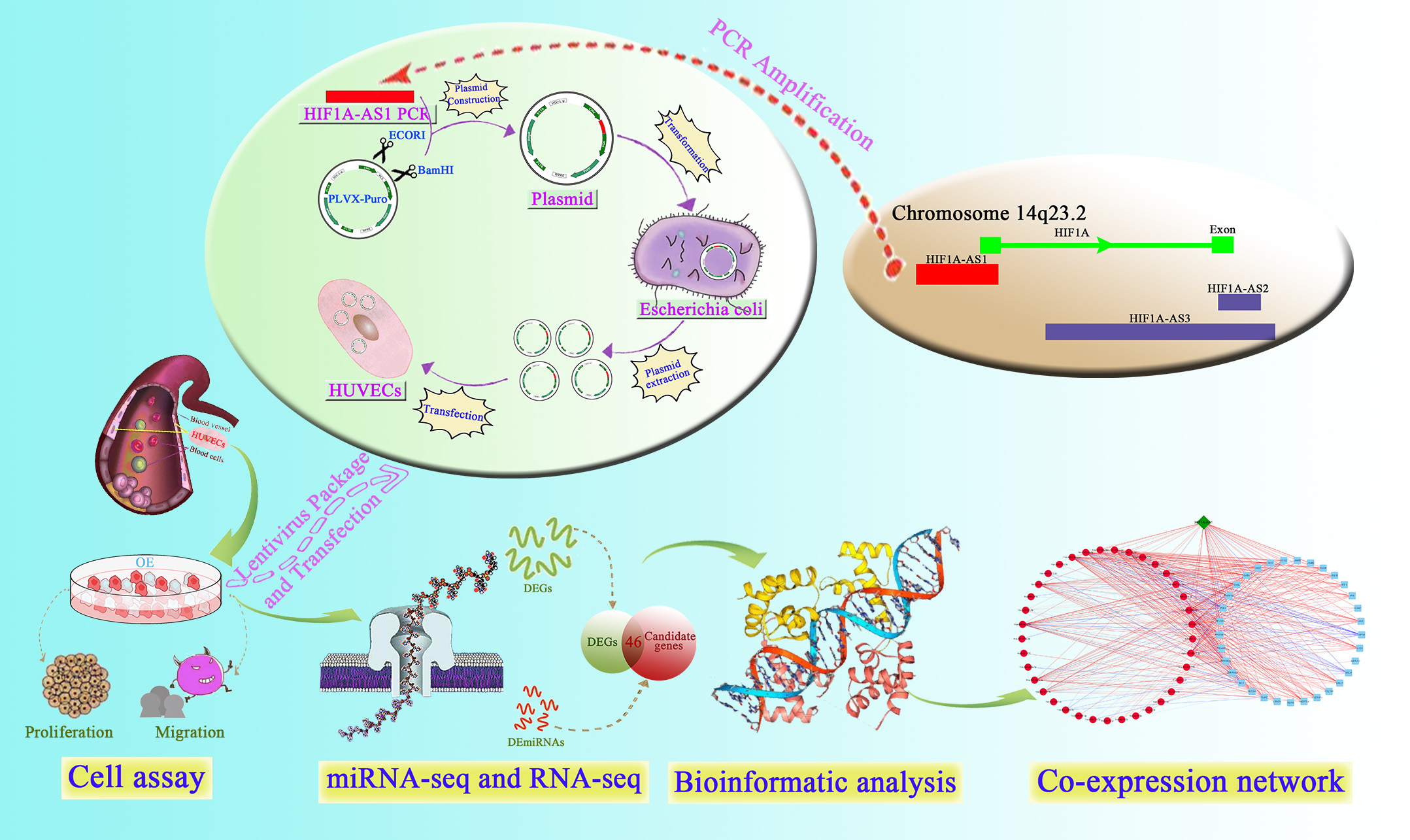 Scheme.
Scheme.Schematic of the experimental process and related mechanism.
HUVECs were obtained from the Shanghai Cell Bank of the Chinese Academy of
Sciences (Shanghai, China). The cells were taken from a male, and cells from
passages 3 to 8 were used for the experiments. The cells were cultured in
Dulbecco’s modified Eagle medium (Catalog Number: 30030, Gibco, Waltham, MA, USA)
containing 10% fetal bovine serum, 100
HIF1-AS1 was amplified and subcloned into the EcoRI and BamHI
restriction sites of pLVX-Puro 1.0 empty plasmid (Thermo Fisher Scientific).
About 2
| Gene/shRNA | Forward primer (5′-3′) | Reverse primer (5′-3′) |
|---|---|---|
| HIF1A-AS1 | GCCTATGGTTGTTCATCTCGTCTC | TGCGTAAAGTACCGAAGAATGGAG |
| BAX | GATGATTGCCGCCGTGGAC | CCCAGTTGAAGTTGCCGTCAG |
| MMP1 | GGGAGCAAACACATCTGACCTAC | AGTTGGAAGGCTTTCTCAATGGC |
| hsa-miR-1298-5p | ACACTCCAGCTGGGTTCATTCGGCTGTCCA | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGTACATCTG |
| hsa-miR-30c-5p | ACACTCCAGCTGGGTGTAAACATCCTACACT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGCTGAGAG |
| hsa-miR-27b-5p | ACACTCCAGCTGGGAGAGCTTAGCTGATTG | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGTTCACCA |
| hsa-let-7a-5p | ACACTCCAGCTGGGTGAGGTAGTAGGTTGT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGAACTATAC |
| hsa-miR-4664-3p | ACACTCCAGCTGGGCTTCCGGTCTGTGAGC | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGACGGGGC |
| hsa-miR-769-5p | ACACTCCAGCTGGGTGAGACCTCTGGGTTC | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGAGCTCAGA |
| hsa-miR-106b-5p | ACACTCCAGCTGGGATCTGCACTGTCAGC | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGATCTGCAC |
| hsa-miR-548o-3p | ACACTCCAGCTGGGCCAAAACTGCAGTTA | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGCAAAAGT |
| h-actin | TGGACTTCGAGCAAGAGATG | GAAGGAAGGCTGGAAGAGTG |
| U6 | CTCGCTTCGGCAGCACA | AACGCTTCACGAATTGTGCGT |
| shRNA-1 | ACCGGTGGGCATCCATAGACATATTCTTTCAAGAGAAGAATAATTGTCTATGGATGCCCTTTTTgaattc | |
| shRNA-2 | ACCGGTGGAAACCTGAAACTTGAATGTTTCAAGAGAACATTCATAAGTTTCAGGTTTCCTTTTTgaattc | |
The proliferation of HUVECs was measured with the CCK-8 assay (Catalog Number: C0037, Beyotime,
Shanghai, China). Briefly, HUVECs were cultured in 96-well
plates for 24 h. The cells were divided into normal control
(NC, untreated HUVECs), PA-treated HUVECs (PAT, 0.8 mmol/L), PAT + overexpression
(OE) (HIF1A-AS1 overexpressed in PAT), PAT + sham-OE (PAT transfected
with empty plasmid), PAT + sh (HIF1A-AS1 knockdown in PAT), and PAT +
sham-sh (PAT transfected with empty plasmid) groups. At 0, 24, 48, and 72 h, 10
The apoptosis of HUVECs was analyzed by flow cytometry using an Annexin V-Conjugated FITC Apoptosis Detection Kit (Catalog Number: BMS500FI-20, BD Biosciences, Franklin Lakes, NJ, USA). The cells were divided into NC, PAT, OE, sham-OE, sh, and sham-sh groups. Briefly, prepared cells were harvested after cultivating for 48 h, washed twice with phosphate-buffered saline (PBS) and incubated in the dark with Annexin V-FITC and propidium iodide (PI) for 30 min. Subsequently, the stained cells were detected with the MoFlo XDP flow cytometer (Catalog Number: V145577, Beckman, Brea, CA, USA) and Cell Quest 3.3 software (BD Biosciences).
To perform the invasion assay, chambers were assembled in 24-well plates with 8
HUVECs (5
Total RNAs were extracted from HUVECs with TRIzol reagent (Catalog Number: 15596026, Invitrogen, Thermo Fisher Scientific). The absorbance of purified RNA at 260 and 280 nm and the A260:A280 ratio were measured using the NanoDrop ND-1000 spectrophotometer (Bio-Rad, Guangzhou, China). Six groups (NC, PAT, sham-OE, OE, sham-sh, and sh) were generated and two biological replicates were made.
Total RNAs (3
The FASTX‑Toolkit (version 0.0.13, http://hannonlab.cshl.edu/fastx_toolkit/) was applied to process raw
reads to obtain reliable clean reads. RNAs
To validate the miRNA-seq data, qPCR was conducted. B-cell lymphoma 2
(Bcl-2)-associated X protein (BAX) and matrix metalloproteinase 1
(MMP1), key factors of HUVEC apoptosis and proliferation, were also
validated by qPCR. The primers used are presented in Table 1. The PCR experiments
were performed with the following conditions: pre-denaturation at 95 °C
for 1 min, 40 cycles of denaturing at 95 °C for 15 s, annealing at 60
°C for 30 s, and elongation at 72 °C for 40 s. The results were
calculated with the 2
Western blotting (WB) was performed according to standard methods. Total cell
lysates were made in 1
To detect the miRNA expression profile of the miRNAs identified, the frequencies
of miRNA counts were normalized to transcripts per million (TPM) with the following
formula: normalized expression = actual read count/total read count
To predict the gene function of candidate genes or DEGs, Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Ontology (GO) analyses were conducted using the DAVID bioinformatics database [28]. Co-expression networks were generated by calculating the Pearson’s correlation coefficient (PCC) for the expression levels of candidate genes or DEGs. To display the co-expression networks, Cytoscape (version 3.0.2) was employed.
All values are presented from independent experiments done in triplicate as the
mean
The datasets generated in the current study were deposited in the Gene Expression Omnibus database of the National Center for Biotechnology Information under the accession numbers GSE107409 and GSE85610.
We constructed a model of cardiovascular occlusion by treating HUVECs with PA [23]. To investigate how HIF1A-AS1 regulates HUVEC function, we overexpressed or knocked down this lncRNA in PA-treated HUVECs (Fig. 1A), and analyzed its expression level by qPCR (Supplementary Figs. 1,2). Compared with the NC group, the overexpression and knockdown of HIF1A-AS1 were successful. Then, we obtained six groups of cells with different treatment strategies, namely NC, PAT, PAT + sham-OE, PAT + OE, PAT + sham-sh, and PAT + sh groups.
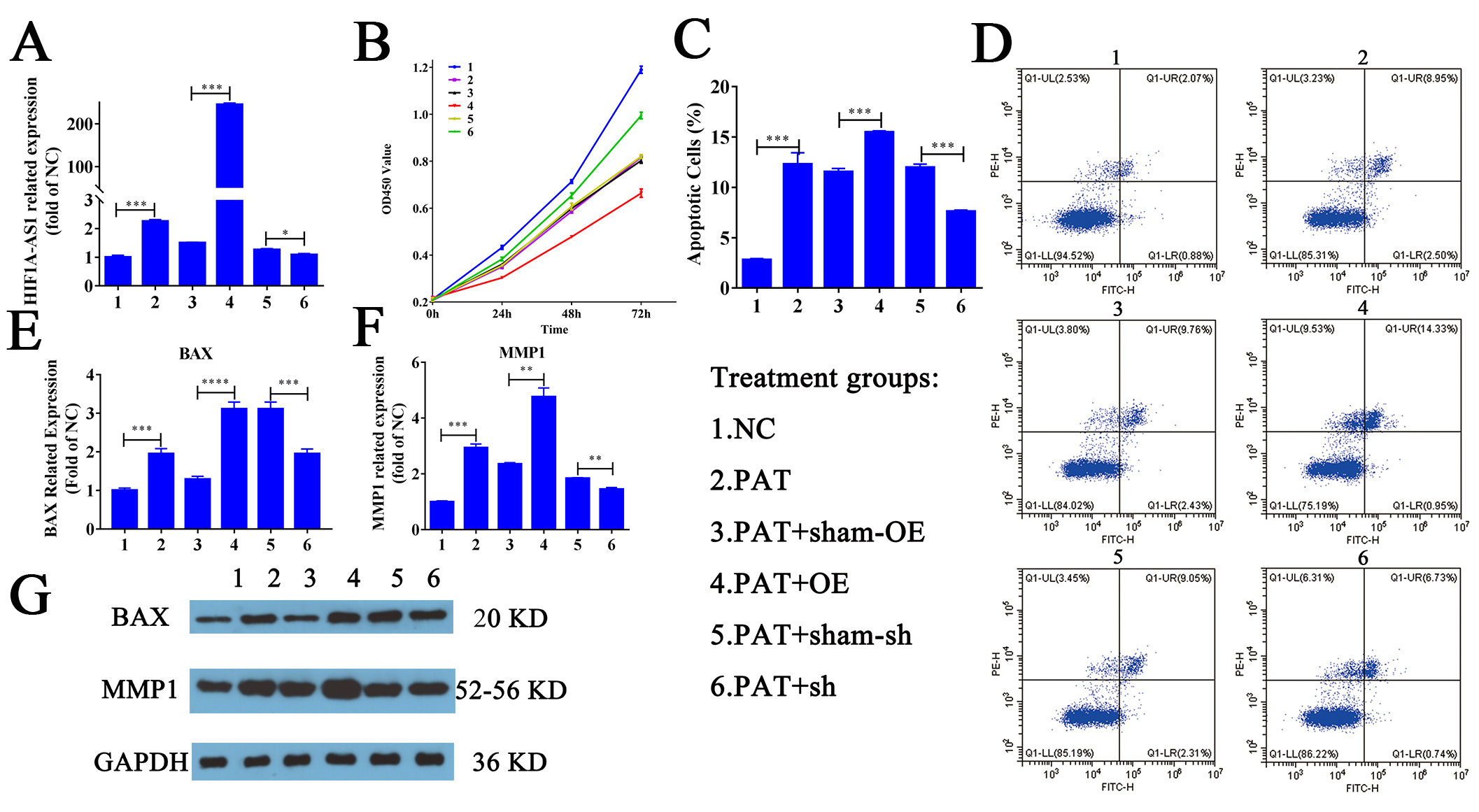 Fig. 1.
Fig. 1.Vector construction and HIF1A-AS1 affects proliferation
and apoptosis in vitro. (A) qPCR was used to verify the successful
construction of the vector. (B) The CCK-8 assay was conducted to evaluate the
cell proliferation of six treatment groups at 0, 24, and 48 h. The data are
presented as the percentage relative to control cells and presented as the mean
The effect of HIF1A-AS1 on the proliferation of HUVECs was determined with a CCK-8 kit. A significant time-dependent increase in proliferation was found among the six groups. CCK-8 assays showed that treatment of HUVECs with PA for 72 h resulted in a 45.8% reduction in HUVEC survival. Silencing of HIF1A-AS1 significantly increased the absorbance values at all time points (24, 48, 72 h). In addition, OE of HIF1A-AS1 led to a 17.1% reduction of its basal inactivation (45.8%) at 72 h (Fig. 1B). The results of the CCK-8 assay indicated that silencing of HIF1A-AS1 promoted the proliferation of HUVECs, and OE led to the reverse effect.
Flow cytometry was conducted to count the number of apoptotic cells. After treatment of HUVECs with PA, the number of apoptotic cells increased more than 3 times (Fig. 1C). Indeed, the apoptosis of HUVECs has been identified as a crucial factor in the pathogenesis of various CVD processes [29, 30]. Thus, preventing the apoptosis of HUVECs may be a novel strategy for the treatment of CVD. Fig. 1C shows that silencing HIF1A-AS1 led to about 35.5% recovery, and OE led to 28.5% promotion of basal apoptosis induced by PA. These results suggest that the lncRNA HIF1A-AS1 plays a critical role in regulating the apoptosis and proliferation of HUVECs.
The expression of BAX and MMP1, key regulators of HUVEC apoptosis and proliferation, was analyzed by qPCR (Fig. 1E,F) and WB (Fig. 1G and Supplementary Figs. 3,4,5). Quantitative analysis of the WB results was also performed (Supplementary Fig. 6), which was consistent with the corresponding qPCR results. Compared with the sham-OE group, the expression of BAX and MMP1 was significantly increased in the OE group. These data indicate that OE of this lncRNA promotes PA-induced apoptosis and reduces the proliferation of HUVECs.
The transwell migration assay was performed to determine whether OE of HIF1A-AS1 can mediate the migration and invasion of HUVECs. In this assay, the cell invasion ability of PA-treated HUVECs decreased about 49.8% compared with blank groups. A 27.8% recovery was observed in invading cells upon silencing of HIF1A-AS1 compared to sham-sh cells, suggesting that silencing this lncRNA can enhance the migration ability of HUVECs. Conversely, OE of HIF1A-AS1 resulted in another 35.3% reduction, clearly indicating the weakened migratory ability of PA-treated HUVECs (Fig. 2A,B).
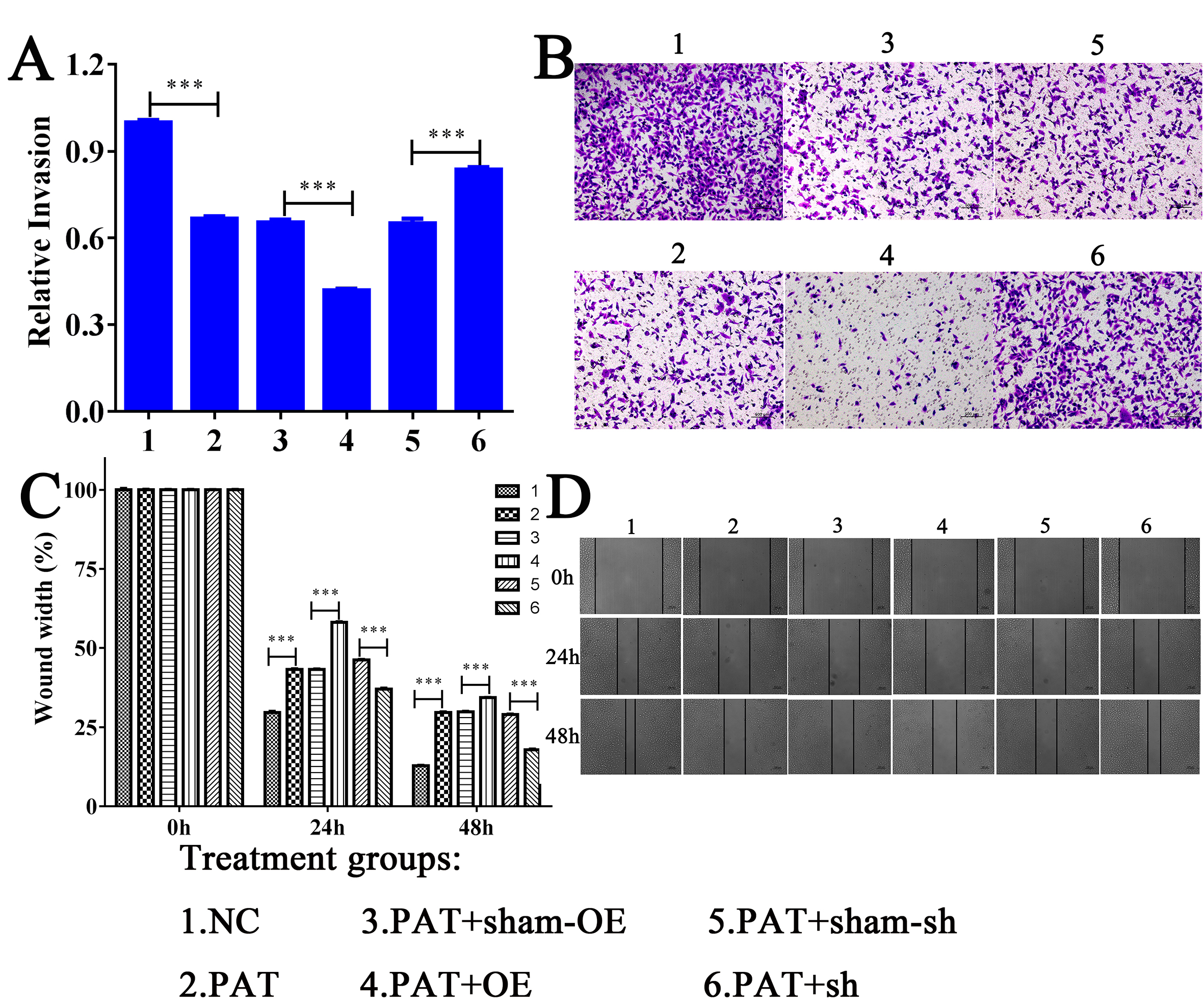 Fig. 2.
Fig. 2.HIF1A-AS1 affects migration and
invasion in vitro. (A) & (B) The wound healing assay was used to
detect the relative cell migration in six groups of cells, scale bar = 200
To further estimate the migration effect of HIF1A-AS1 lncRNA on HUVECs, we conducted a wound healing assay. As shown in Fig. 2C,D, the migration ability was weakened after OE of HIF1A-AS1 compared with the sham-OE group. By contrast, HUVECs migrated for a longer distance in the pore plate extracted from HIF1A-AS1 transgenic cells than from the sham-sh group. These results suggested that this lncRNA successfully inhibited HUVEC migration.
To investigate how HIF1A-AS1 regulates the expression of miRNAs in HUVECs, 12 small RNA libraries (NC-1, NC-2, NC-3, PAT-1; PAT-2, PAT-3, PAT-sham-OE-1, PAT-sham-OE-2; PAT-sham-OE-3, PAT-OE-1, PAT-OE-2 and PAT-OE-3) were constructed for miRNA-seq. More than 141.2 million reads were generated, with approximately 11.8 million sequence reads per sample. Comparing all reads to the human reference genome, it was found that 91.2% of the reads were successfully mapped to the reference genome (Table 2). More than 852 conserved miRNAs and about 29 novel miRNAs were identified from the boxplots of the 12 samples, no obvious differences were found in these groups (Fig. 3A and Supplementary Table 1). The above results confirmed the reliability and stability of miRNA-seq.
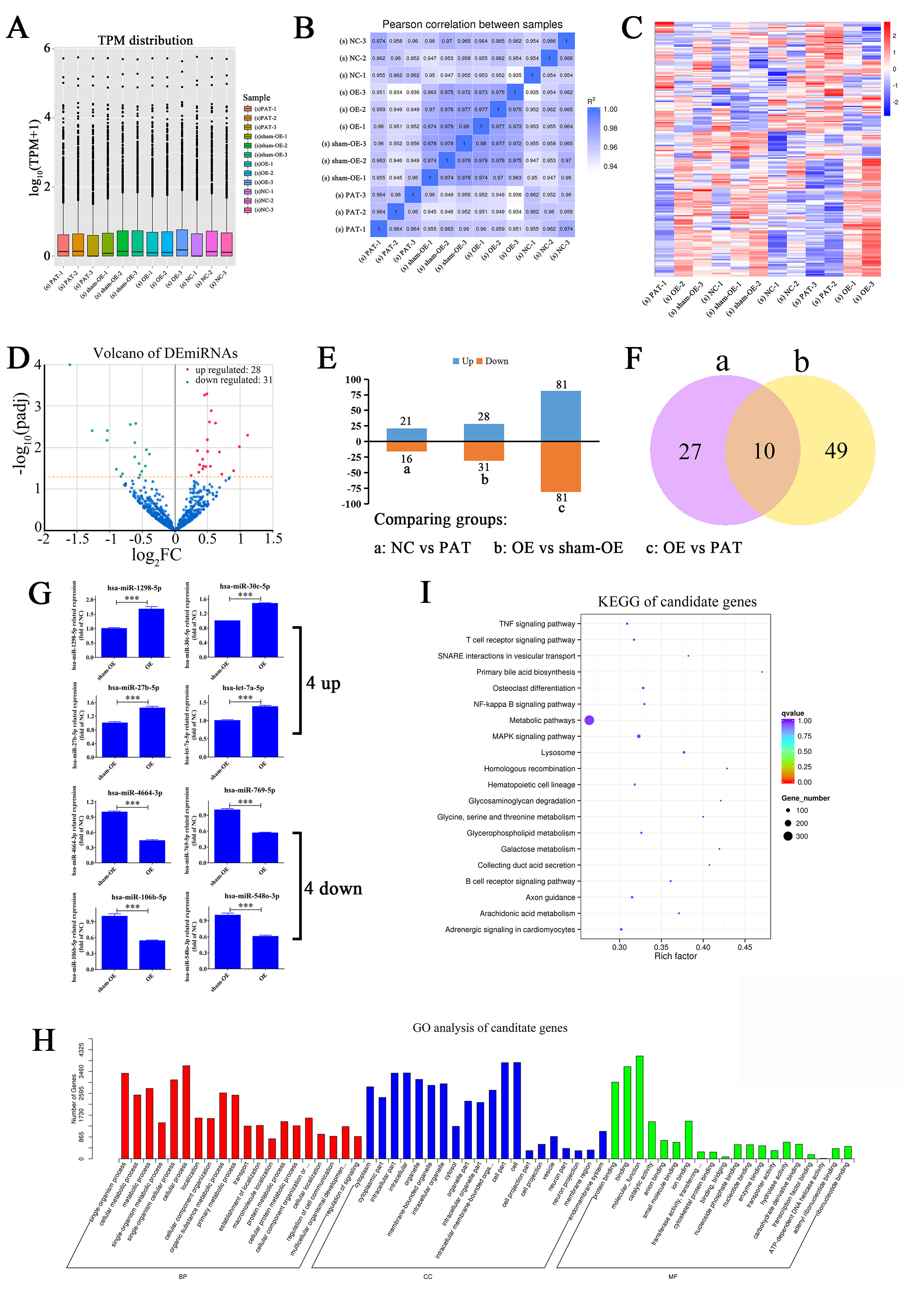 Fig. 3.
Fig. 3.Exploration of DEmiRNAs and functional analysis. (A) The
boxplots of the 12 samples miRNAs, (s) stands for sample, n = 12. (B) OE of
HIF1A-AS1 could markedly regulate the expression of miRNAs. The results of
Pearson’s correlation data were presented. (C,D) Heat map (C) and Volcano plot
(D) of DEmiRNAs expression profiles between OE and sham-OE. (E) The number of
upregulated and downregulated DEmiRNAs among NC vs. PAT, OE vs. sham-OE, and OE
vs. PAT. (F) Venn diagrams of the DEmiRNAs identified in different comparisons.
Data are presented as the mean
| Sample name | Raw reads | Clean reads | Total sRNA | Mapped sRNA | Conserved miRNA | Novel miRNA |
|---|---|---|---|---|---|---|
| NC-1 | 10396456 | 10095959 | 10174904 | 8663662 | 630 | 13 |
| NC-2 | 12281806 | 12148322 | 9880267 | 9134235 | 756 | 22 |
| NC-3 | 12371135 | 12173628 | 9043012 | 8697259 | 796 | 23 |
| PAT-1 | 12063104 | 11945895 | 10223291 | 9580570 | 763 | 20 |
| PAT-2 | 11548214 | 11351509 | 14008342 | 12347805 | 747 | 20 |
| PAT-3 | 9967884 | 9844849 | 10032489 | 9188302 | 674 | 19 |
| PAT-sham-OE-1 | 11524964 | 11310117 | 10449928 | 9418015 | 854 | 29 |
| PAT-sham-OE-2 | 12595778 | 12406547 | 10637694 | 9785800 | 888 | 26 |
| PAT-sham-OE-3 | 15271004 | 15102718 | 13704766 | 12264356 | 920 | 31 |
| PAT-OE-1 | 10825192 | 10686483 | 9844659 | 8803245 | 812 | 25 |
| PAT-OE-2 | 10901089 | 10702730 | 9502391 | 8298306 | 792 | 25 |
| PAT-OE-3 | 11455474 | 11268048 | 8817627 | 8188634 | 856 | 27 |
MiRNAs act as key post-transcriptional regulators in multiple cellular
biological processes such as proliferation, differentiation, apoptosis, invasion
and migration [31]. Interestingly, we found that OE of HIF1A-AS1
markedly regulated the expression of miRNAs. The results of the Pearson
correlation data are shown in Fig. 3B. A heat map was generated to reflect the
detailed alterations of miRNAs (Fig. 3C). In addition, the volcano plots in Fig. 3D compared these log
MiRNAs commonly exert their functions through binding to complementary target sites from the target genes. DEmiRNA was obtained from OE vs. sham-OE groups. The DEmiRNA target genes, also called candidate genes, were successively identified by miRBase. As shown in Supplementary Table 3, we obtained a total of 2030 and 2401 predicted targets of the upregulated and downregulated DEmiRNAs, respectively. For the 28 upregulated DEmiRNAs, hsa-miR-193b-3p was found to potentially target the most genes, with a number of 334. For the 31 downregulated DEmiRNAs, hsa-miR-5088-5p possessed the most targets, with a number of 1087.
It was obvious that OE of HIF1A-AS1 globally affected miRNA expression in HUVECs. Consequently, bioinformatics analysis was conducted to identify the key functions in which all candidate genes were involved. GO analysis showed that target genes were mainly enriched in the single-organism process (GO:0044699), cellular process (GO:0009987), and some metabolic processes, especially the organic substance metabolic process (GO:0071704). Furthermore, cellular component analysis indicated that the target genes were mainly enriched in the intracellular regions and shared a heterogeneous spatial distribution across the entire cell. In addition, molecular function analysis showed that most of them performed binding activities and enzymatic reactions (Fig. 3H).
KEGG enrichment analysis of the target genes was performed to gain further insight into their functions (Supplementary Table 4). The results showed that most of the candidate genes were enriched in pathways associated with metabolism including metabolic pathways (ID: hsa01100), glycerophospholipid metabolism (ID: hsa00564), glycosaminoglycan degradation (ID: hsa00531), galactose metabolism (ID: hsa00052), hematopoietic cell lineage (ID: hsa04640), and glycine, serine, and threonine metabolism (ID: hsa00260) (Fig. 3I). It can be proved from the previous discussion that HIF1A-AS1 overexpression in HUVECs can produce numerous complex changes, indicating that it plays an important role in CVD.
RNA-seq experiments were done to further
explore the molecular mechanisms by which HIF1A-AS1 regulates HUVECs.
More than 1.20 billion pairend reads were generated, corresponding with
~100 million sequence reads per sample. Using Hisat2 software,
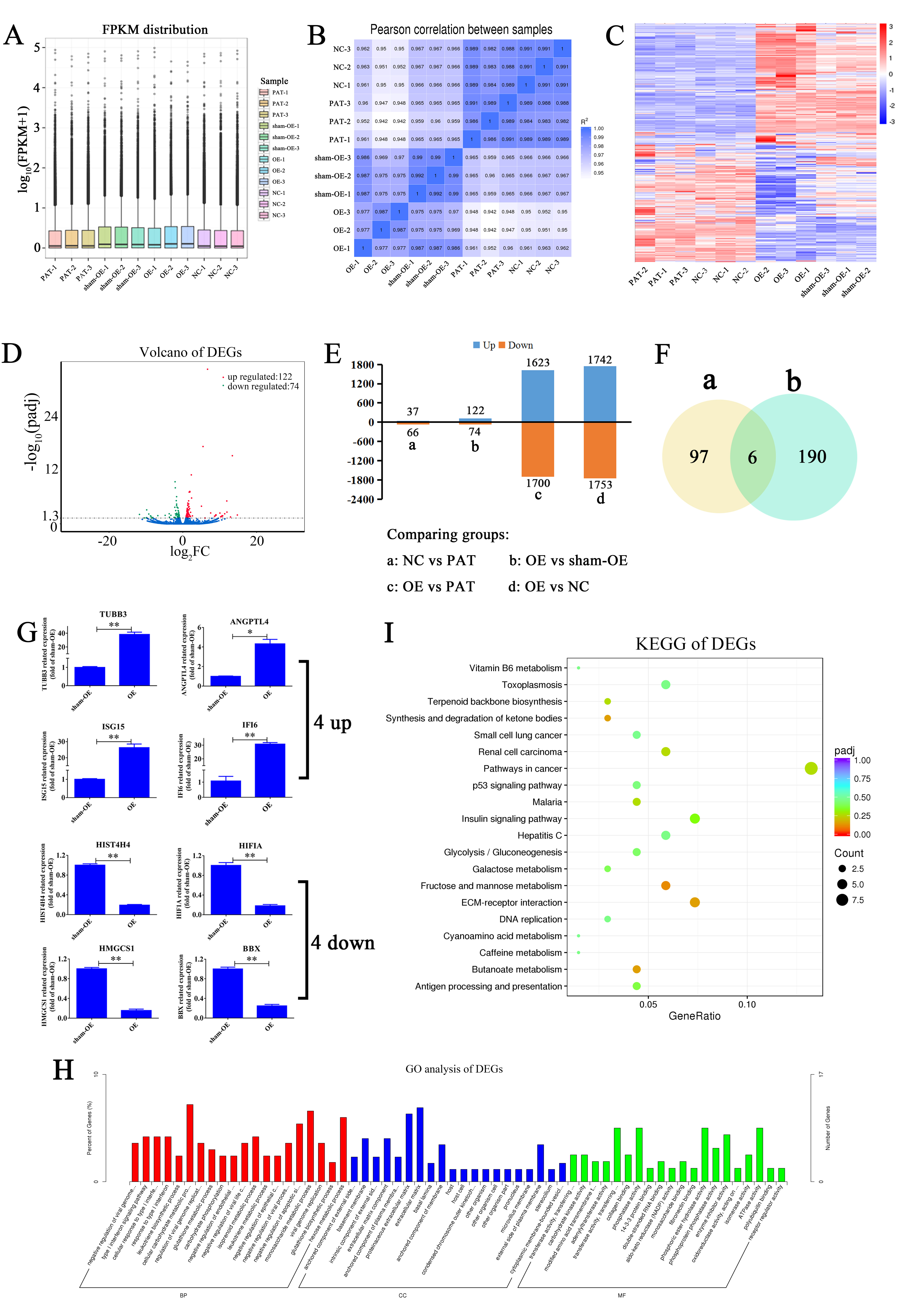 Fig. 4.
Fig. 4.Exploration of DEGs and functional analysis. (A) Boxplots of
the 12 samples RNA-seq; no significant difference was found among these groups.
(B) Subsequently, the RNA-seq results revealed that OE of HIF1A-AS1 not only
regulate the expression of miRNAs but also genes. (C,D) Heat map (C) and Volcano
plot (D) of DEGs expression profiles between the OE and sham-OE groups. (E) The
number of upregulated and down-regulated DEGs among NC vs. PAT, OE vs. sham-OE
and OE vs. PAT groups. (F) Venn diagrams of the DEGs identified in different
comparisons. Data are presented as the mean
| Sample name | Raw reads | Clean reads | Total mapped | Multiple mapped | Uniquely mapped |
|---|---|---|---|---|---|
| NC-1 | 92950742 | 90845448 | 85500273 | 3714836 | 81785437 |
| NC-2 | 88259194 | 86224692 | 81248247 | 4094785 | 77153462 |
| NC-3 | 96525162 | 94188860 | 88406416 | 4294222 | 84112194 |
| PAT-1 | 101172612 | 98776918 | 92458131 | 4556970 | 87901161 |
| PAT-2 | 106123908 | 102950612 | 95273281 | 6263675 | 89009606 |
| PAT-3 | 105586716 | 102833318 | 95759913 | 5149250 | 90610663 |
| sham-OE-1 | 101559260 | 99046714 | 90953710 | 4024188 | 86929522 |
| sham-OE-2 | 103669156 | 101555434 | 91930479 | 4509528 | 87420951 |
| sham-OE-3 | 101384686 | 99407612 | 90783970 | 4071846 | 86712124 |
| OE-1 | 105385816 | 103382496 | 88847622 | 4617749 | 84229873 |
| OE-2 | 99645746 | 95951214 | 84091509 | 5573479 | 78518030 |
| OE-3 | 102004516 | 97888796 | 86382747 | 5694658 | 80688089 |
Subsequently, the RNA-seq results revealed that OE of HIF1A-AS1 regulated the expression for both miRNA and mRNA. Pearson’s correlation data are shown in Fig. 4B. The heat map reflected the DEGs (Fig. 4C). By adopting the same method, a total of 196 DEGs were obtained from the sham-OE vs. OE groups (Supplementary Table 5), with 122 upregulated and 74 downregulated, respectively (Fig. 4D,E). We observed six reliable core mRNAs across two groups (PAT vs. NC and OE vs. sham-OE) from the Venn diagram (Fig. 4F). qPCR was conducted to verify the four upregulated mRNAs (TUBB3, ANGPTL4, ISG15 and IFI6) and four downregulated mRNAs (HIST4H4, HIF1A, HMGCS1 and BBX) (Fig. 4G). The qPCR results were in accordance with that of mRNA-seq. Interestingly, 23.5% DEGs overlapped with DEmiRNA target genes. The overlapping DEGs are listed in Supplementary Table 6. According to previous reports [27, 28, 29], both mRNA and miRNAs had the ability to regulate the apoptosis and proliferation of HUVECs. To further explore the role of miRNA in HUVECs, we conducted this study.
Since some DEGs overlapped with DEmiRNA target genes, bioinformatics analysis was conducted to identify the biological function of DEGs. GO enrichment analysis showed that most of the enriched pathways were associated with multiple metabolic process, signaling pathways, and apoptosis with high confidence. Remarkably, the outcome also highlighted large-scale alterations in a variety of metabolic pathways and apoptosis processes upon the elevation of HIF1A-AS1 levels. Similarly, the results also demonstrated that a larger number of DEGs were related to extracellular matrices with overlapping distributions, and others were restricted to particular cellular loci. The majority had binding functions and enzymatic activity, especially transferase activity, with a fraction of them having other housekeeping functions (Fig. 4H and Supplementary Table 7).
The most significantly enriched KEGG pathways are shown in Fig. 4I and Supplementary Table 8. The results showed that fructose and mannose metabolism (ID: hsa00051), synthesis and degradation of ketone bodies (ID: hsa00072) and butanoate metabolism (ID: hsa00650) were the most significant pathways for enrichment. KEGG results showed that some DEGs were more uniformly enriched in tumor-related pathways such as p53 signaling pathway (ID: hsa04115), renal cell carcinoma (ID: hsa05211), Vitamin B6 metabolism (ID: hsa00750), pathways in cancer (ID: hsa05200), malaria (ID: hsa05144), and caffeine metabolism (ID: hsa00232). The main signaling and metabolism pathways determined by KEGG analysis will provide further insight into future research directions on mRNA.
Generally, miRNAs have the capacity to recognize and bind to complementary 3’-untranslated regions of target mRNAs, which can lead to the degradation or transcriptional repression of mRNAs [32]. To explore the relationship between DEmiRNA and DEGs, a co-expression network was generated by calculating the PCC for the expression levels of DEGs and DEmiRNAs. It showed a close correlation between the expression levels of DEmiRNAs and DEGs, which were enriched in some initial pathways (Fig. 5). A total of 37 DEmiRNAs and 33 DEGs were filtered into the co-expression network complex. The network manifested that lncRNA HIF1A-AS1 mediated the function of HUVECs by not only regulating the expression of some genes directly but also influencing a few miRNAs to indirectly mediate the expression level of mRNA. These findings may explain the underlying mechanism of HIF1A-AS1 in CVD.
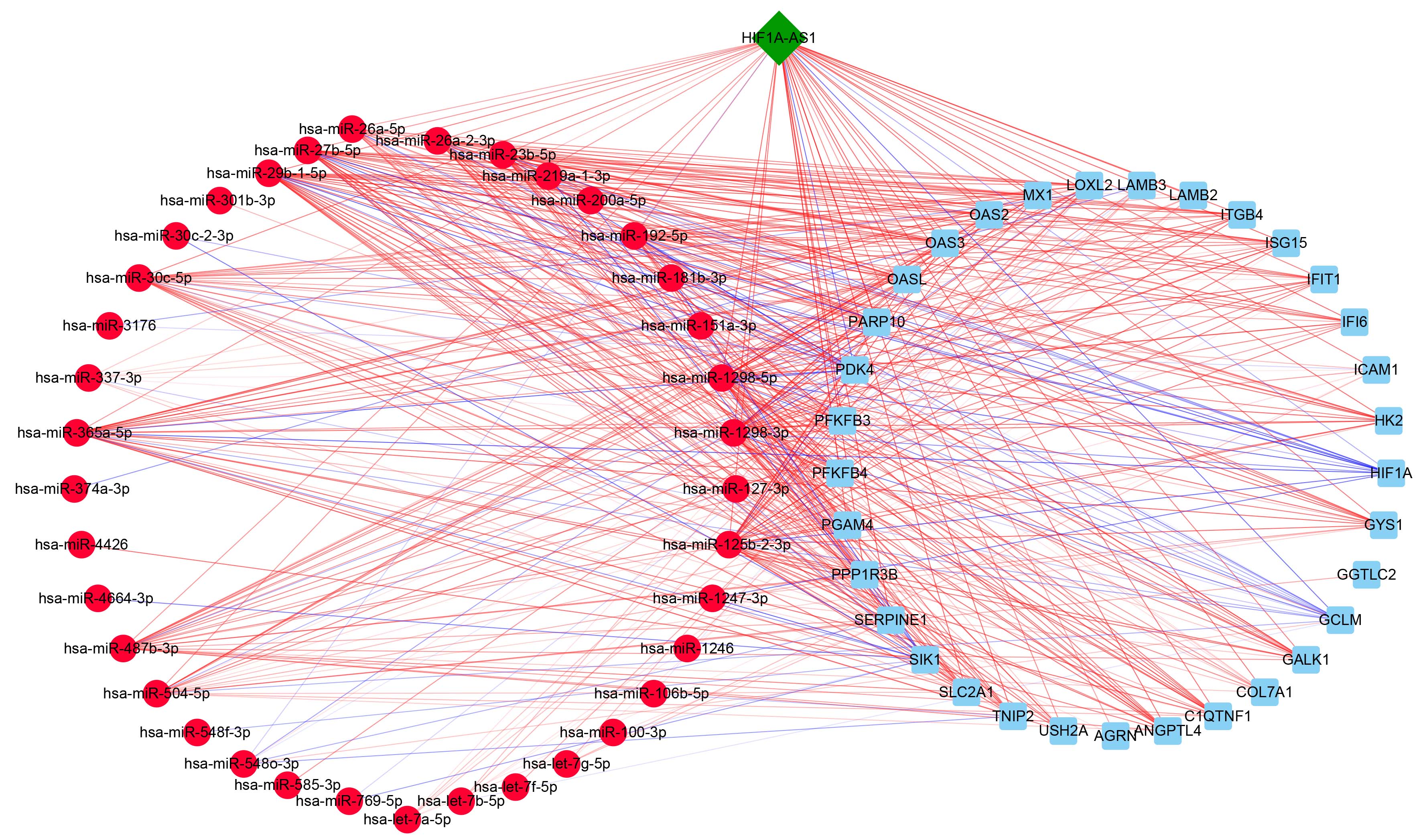 Fig. 5.
Fig. 5.Interaction network analysis of DEmiRNAs and DEGs associated
with some important pathways. Network analysis on the basis of PCCs for DEmiRNAs
and DEGs enriched in ‘negative regulation of endothelial cell apoptotic process’
(GO:2000352), ‘cellular carbohydrate metabolic process’ (GO:0044262), ‘type I
interferon signaling pathway’ (GO:0060337) and so on. Circular nodes represent
DEmiRNAs and rectangular nodes signify DEGs, while these solid lines represent
significant correlations between DEmiRNAs and DEGs. The red
lines represent negative correlation, while the blue lines
represent positive correlation. p
Most CVDs are related to the apoptosis of VECs, which is the main form of vascular injury [33]. Previous studies have shown that the broken balance between VECs apoptosis and proliferation markedly contributes to the pathogenesis of CVD [23, 34]. An increasing number of studies has shown the critical effect of lncRNAs on regulating the proliferation and apoptosis of VECs in CVD [3, 35, 36].
Only about 2% of sequences in the human genome possess the ability of encoding proteins. Accumulating evidence has revealed that lncRNAs are related to human diseases as a biomarker or therapeutic target [37, 38]. Postnatally, lncRNAs have attracted a lot of attention due to their variety of biological roles including cell cycle control, cell proliferation, apoptosis, transwell invasion, embryonic development, and carcinogenesis by mediating the gene expression at the transcriptional, splicing, transportation, and translational levels [39, 40].
MiRNAs involved in mRNA degradation or translation inhibition [41] are a group of evolutionarily conserved ncRNAs about 20–22 nt in length from hairpin pre-miRNA precursors [42]. Accumulative evidence has revealed that miRNAs, alone or in combination with lncRNAs, are involved in regulating specific gene expression at the translation or transcription level. Then they can alter cell signaling pathways associated with different physiological and pathological processes [43, 44, 45, 46].
A study found that HIF1A-AS1 TFR2 forms triplexes with EPH receptor A2 (EPHA2)
and adriamycin (ADM) double-stranded DNA under regular and triplex-stabilized
conditions upon DNA hairpin formation. Increasing the expression of HIF1A-AS1 can
inhibit the expression of EPHA2 and ADM, whereas the downregulation of HIF1A-AS1
produces opposite results. These results suggest that the trimer formation region
can mediate EPHA2 and ADM inhibition [47]. Another study showed that the
HIF1A-AS1 was significantly increased in gemcitabine (GEM)-resistant pancreatic
cancer cells. HIF1A-AS1 enhanced the GEM resistance of pancreatic cancer cells by
upregulating the expression of HIF1
Another study, which was published recently, showed that some lncRNAs regulate various cellular processes by acting as competing endogenous RNAs (ceRNAs) and binding proteins. For example, HIF1A-AS1, acting as a ceRNA, absorbed miR-204 to evaluate Suppressor of Cytokine Signaling 2 expression in cardiac function [21]. This lncRNA participates in the regulation of proliferation, apoptosis, and the activity of the extracellular matrix proteins of VSMCs [18, 49]. Abundant evidence has indicated that this lncRNA might participate in the development of CVD by regulating the PA-induced apoptosis of HUVECs [23]. However, the molecular mechanism by which HIF1A-AS1 interacts with miRNAs and mRNA and their regulatory roles of pathogenesis are unclear. HIF1A-AS1 has potential as a novel therapeutic target in CVD, but underlying information about the regulatory mechanisms in HUVECs is lacking.
It is known that miRNAs are involved in the progression and pathogenesis of VECs [50]. In this study, it was found that OE of HIF1A-AS1 reduced the cellular growth rate and led to the robust apoptosis of HUVECs. We further studied the molecular mechanism of this phenomenon using miRNA-seq and RNA-seq. More than 852 conserved miRNAs were identified and about 29 novel miRNAs were found by miRNA-seq in the sham-OE group. When HIF1A-AS1 was overexpressed, the expression levels of some miRNAs markedly changed, indicating that this lncRNA may play a critical role in miRNA-based therapies. The target genes of those DEmiRNAs were successively predicted by miRase. Additionally, multiple target genes of the DEmiRNAs were associated with the apoptosis, proliferation, and migration of HUVECs, suggesting that OE of HIF1A-AS1 could inhibit proliferation and promote the apoptosis of HUVECs by mediating miRNA expression. As previously reported, lipids have important functions in maintaining normal physiological cellular functions [51]. Glycosaminoglycan can promote wound healing. The administration of d-galactose to animals decreases the proliferation of cells and reduces the migration and survival of new neurons in the granule cell layer [52]. Researchers have found a potential involvement of the glycine-serine-threonine metabolic axis in longevity and related molecular mechanisms [53]. Thus, DEmiRNA may serve a regulatory role in the molecular functional analysis of HUVECs.
Subsequently, RNA-seq was performed to identify the DEGs. Cluster of differentiation, which leads to endothelium apoptosis, was not identified. Individual differences may be responsible for this unusual phenomenon. Many DEGs were found to overlap with the target genes of DEmiRNA. Furthermore, both mRNA and miRNAs could regulate the apoptosis and proliferation of HUVECs [54, 55, 56]. Therefore, HIF1A-AS1 has the ability to regulate the expression of some miRNAs, which could target some apoptosis-related genes by degrading mRNAs or inhibiting their translation. The crosstalk among miRNAs, lncRNAs, and mRNA shows a complex network of gene expression regulation [57]. Hence, in the present study, a co-expression network was systematically constructed to explore the relationship among lncRNA HIF1A-AS1, DEmiRNA, and DEGs. The findings revealed that the expression levels of the DEmiRNAs was tightly linked to the apoptosis-related DEGs. However, this network has not been systematically validated, which limits the comprehensive understanding of the mechanisms underlying the role of HIF1a-AS1 in the treatment of CVD. In addition, accumulative evidence has indicated that ANGPTL4 is directly correlated with the risk of CVD, especially atherosclerosis [58]. SERPINE1 may serve as a potential therapeutic target or new biomarkers in acute myocardial infarction [59]. Interferon Alpha Inducible Protein 6, which is a mitochondrial localized antiapoptotic protein, contributes to promoting the metastatic potential of certain cancer cells through mitochondrial reactive oxygen species [60]. However, in this co-expression network, the expression levels of these CVD-related genes were tightly related to certain miRNAs. Therefore, HIF1A-AS1 can modulate the expression of DEGs by mediating miRNA expression. The present study reveals a novel mechanism by which HIF1A-AS1 regulates the apoptosis of HUVECs.
In summary, our study showed that HIF1A-AS1 regulated HUVEC function by not only regulating the expression of some genes directly but also influencing some miRNAs to indirectly mediate the expression level of mRNA, indicating that it may play a key role in the pathogenesis and progression of CVD. The current study also provides some new insights and directions for the prevention and treatment of CVD. Although the clinical applications need to be further explored, these results additionally provide insight into the molecular mechanisms by which HIF1A-AS1 affects HUVECs and a scientific experimental basis for treating CVD. Thus, it is feasible that the co-expression network could be applied for the prevention, diagnosis, treatment, and prognosis of CVD. However, further studies are being conducted to more systematically elucidate the role of HIF1A-AS1 in CVD and further determine the potential clinical role of the co-expression network.
The datasets analyzed in this study can be obtained from the NCBI public repository under accession numbers GSE107409 and GSE85610.
ZG, JY designed and managed the project. JD, HL, BW, KD drafted the manuscript. CZ, LC participated in sample collection and manuscript revision. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
This study was supported by National Natural Science Foundation of China (NSFC) (Grant Nos.: 81860093 and 81660215) and Associated Project of Yunnan Province Science & Technology Department and Kunming Medical University Basic Research for Application [Grant Nos.: 202101AY070001-031, 2019FE001(-207), and 202101AT070226].
The authors declare no conflict of interest.






