1 Department of Biotechnology, Maharishi Markandeshwar University, Mullana, 133207 Ambala, India
2 Department of Environmental Science SP College, 190001 Srinagar, India
3 Department of Molecular Biology, Biotechnology and Bioinformatics, Chaudhary Charan Singh Haryana Agricultural University, 125004 Hisar, India
4 Department of Seed Science and Technology, Chaudhary Charan Singh Haryana Agricultural University, 125004 Hisar, India
5 Department of Environmental Science, Government Degree College, 190017 Eidgah, India
6 Department of Soil Science and Agricultural Chemistry, Sri Karan Narendra Agriculture University, 303329 Jaipur, India
7 Department of Biochemistry, Chaudhary Charan Singh Haryana Agricultural University, 125004 Hisar, India
8 Department of Microbiology, PSGVP Mandal's S I Patil Arts, G B Patel Science and STKV Sangh Commerce College, 425409 Shahada, India
9 Department of Pharmacology, College of Pharmacy, Umm Al-Qura University, 24382 Makkah, Saudi Arabia
10 Finnish Museum of Natural History, University of Helsinki, FI-00014 Helsinki, Finland
Academic Editor: Naeem Khan
Abstract
In the present era of climate change and global warming, high temperatures have increased considerably, posing a threat to plant life. Heat stress affects the biochemistry, physiology and molecular makeup of the plant by altering the key processes, i.e., photosynthesis, respiration and reproduction which reduces its growth and development. There is a dire need to manage this problem sustainably for plant conservation as well as the food security of the human population. Use of phytohormones to induce thermotolerance in plants can be a sustainable way to fight the adversities of heat stress. Phytohormone-induced thermotolerance proves to be a compelling approach to sustainably relieve the damaging effects of heat stress on plants. Salicylic acid (SA) is an essential molecule in biotic and abiotic defense response signal transduction pathways. When supplied externally, it imparts heat stress tolerance to the plants by different means, viz., increased Heat Shock Proteins (HSP) production, Reactive oxygen species
(ROS) scavenging, protection of the reproductive system and enhancing photosynthetic efficiency. The effect of SA on plants is highly dependent on the concentration applied, plant species, plant age, type of tissues treated, and duration of the treatment. The present review paper
summarizes the mechanism of thermotolerance induced by salicylic acid in plants
under heat stress conditions. It includes the regulatory effects of SA on heat
shock proteins, antioxidant metabolism, and maintenance of Ca
Keywords
- antioxidant system
- heat shock proteins
- heat stress tolerance
- photosynthesis
- salicylic acid
Salicylic acid (SA) is a hepta-carbon phenolic compound used effectively as an analgesic for more than 2000 years. It occurs in nature as an endogenously synthesized ubiquitous plant growth regulator. Plants synthesize SA through two main pathways: phenylalanine and isochorismate, which are part of the shikimic acid pathway [1, 2]. SA balances plant growth and development along with critical physiological processes [3]. It was not before 1987 when SA (earlier known as calorigen) caused thermogenesis in treated flowers of voodoo lily (Sauromatum guttatum) [4]. Being a plant signaling molecule, it has a role in local and systemic plant defense responses against pathogens through its direct or indirect involvement in signaling networks [5]. It helps in providing resistance to biotic and tolerance toward abiotic stress in plants [6, 7], such as temperature extremes [8], heavy metals [9], drought [10, 11, 12], osmotic [13] and salinity [14]. The stress caused by heat is a potential global risk to food security caused due to accelerated temperature and anthropogenic activities, which have aggravated the problem by degrading the agricultural system. Heat stress influences the plant from seed germination to maturity and causes drastic economic yield losses. In plants, many interconnected changes like morpho-anatomical, physiological and biochemical modifications occur due to the adverse effects of transient or constant high temperature. In a broad sense, these changes could be categorized as direct and indirect injuries. The continuous high temperature may cause immediate injuries, including protein denaturation, aggregation, and increased fluidity of membrane lipids. On the other hand, slow or transient heat produces indirect damage such as inactivation of chloroplastic and mitochondrial enzymes, suppression of protein synthesis, protein degradation, loss of membrane integrity, and photosynthetic machinery arrest [15].
Numerous approaches are being used to enhance crop tolerance and reduce the negative impacts of heat stress on plants [16, 17, 18]. Exogenous application of plant growth regulators such as Abscisic acid (ABA), Jasmonic acid (JA), SA, and others can induce thermotolerance [19, 20]. The SA can be applied through different means, including seed priming, irrigation or foliar spray [21, 22]. SA initiates the abiotic stress tolerance mechanisms when provided exogenously to stressed plants. The influence of SA on the plant is dose-dependent, where low and high SA concentrations can result in different outcomes for the same plant species. The specific high or low concentarion of SA treatment depends on many factors i.e., the duration of the treatment, plant species, plant age, and treated plant part [23].
Thermo protection using SA was first induced in mustard with low concentration (0.01–0.1 mM) [24]. SA can have different effects based on concentration given and may even vary on the basis of plant species studied [25, 26]. The exogenous SA application can increase the levels of endogenous SA (in free or glycosylated forms) and mimick heat acclimation. Hence, both the acclimation process and external application of SA can improve the plant survival under stress [27]. Larkindale and Knight [28] concluded that SA is vital in protecting the plant from heat-induced oxidative stress. SA signaling helps in acquiring basal thermotolerance in Arabidopsis and insulates against heat stress and the subsequent recovery [29]. It can also increase levels of Phenylalanine Ammonia Lyase (PAL) mRNA, new PAL protein production, and accumulation of considerable amounts of phenolics, all of which contribute to thermotolerance [30]. In Triticum aestivum, heat stress was alleviated using 0.5 mM SA by reducing the activity of 1-amino cyclopropane carboxylic acid synthase (ACS) and restricting ethylene formation under heat stress [31]. Plant physiology and yield attributes were improved under heat stress by seed priming and external SA application [32]. Studies have also attempted to annotate functional components and signaling networks linked with molecular mechanisms involved in SA-mediated heat stress response in plants [33].
This review paper summarizes the studies conducted to better understand the
SA-induced plant tolerance to heat stress (Table 1, Ref.
[24, 25, 26, 31, 32, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65]). Therefore, those aspects
are emphasized that possibly underpin heat stress tolerance in the presence of SA
and its crosstalk with other factors indulged in the signal transduction pathway.
The primary focus is to support the important functions of SA (applied
externally) in modulating different plant systems to achieve heat stress
tolerance (Fig. 1). Various sub-sections briefly discuss the interconnections of
SA with heat shock proteins, photosynthetic machinery, involvement of SA in the
signaling of reactive oxygen species (ROS) along with the alteration of
antioxidants, major osmolytes like proline, Ca
| Species | Applied SA concentration | Mode of application | Parameters studied | Reference |
| Sinapis alba | 10–500 µM | Foliar spray | H |
[24] |
| Sinapis alba | 100 µM | Foliar spray | Endogenous SA level, antioxidant metabolites, antioxidant enzymes | [47] |
| Solanum tuberosum | 10 |
Culture medium | H |
[54] |
| Nicotiana tabacum | 10 and 100 µmol·L |
Culture medium | H |
[25] |
| Phaseolus vulgaris and Lycopersicon esculentum | 0.1–0.5 mM | Seed priming | Survival percentage | [26] |
| Nicotiana tabacum | 70 µM | Protoplast incubation | Hsp 70 | [55] |
| Agrostis stolonifera | 10 µmol·L |
Foliar spray | Turf quality, leaf photosynthetic rate, TBARS (Thiobarbituric Acid Reactive Substances), antioxidant enzymes | [43] |
| Triticum aestivum | 1 mM | Seedling treatment | Dry weight of seedlings, H |
[49] |
| Cicer arietinum | 100 µM | Foliar spray | Relative injury of membranes, protein and proline content, antioxidant enzymes | [50] |
| Poa pratensis | 0, 0.1, 0.25, 0.5, 1 and 1.5 mmol | Foliar spray | Turf quality, leaf water content, AOS (active oxygen species) production, O |
[48] |
| Pisum sativum | 100 µM | Foliar spray | Membrane marker assay, PIP |
[56] |
| Cucumis sativa | 1 mM | Foliar spray and nutrient solution | Lipid peroxidation, chlorophyll fluorescence, antioxidant enzyme activity | [40] |
| Vitis vinifera | 100 µmol·L |
Foliar spray | Relative electrolyte leakage, TBARS, antioxidant metaboltites, antioxidant enzymes, Ca |
[44] |
| Vigna radiata | 0.5 and 1 mM | Foliar spray | H |
[57] |
| Lycopersicon esculentum | 0.1 mM | Foliar spray | Hsp 70, Hsf A1, Hsf A2, Hsf B1, Hsf-DNA binding | [36] |
| Brassica juncea | 10 |
Foliar spray | Plant growth, chlorophyll and photosynthesis measurements, leaf water potential, proline content, antioxidant enzymes, carbonic anhydrase, nitrate reductase, leaf nutrient status | [58] |
| Brassica spp. | 10 and 20 µM | Foliar spray | Electrolyte leakage, total soluble sugar, protein profiling, soluble neutral invertase, antioxidant enzymes | [59] |
| Arabidopsis thaliana | 250 µM | Culture medium | Hsp 101 and Hsp 17.6 | [37] |
| Vitis vinifera | 100 µM | Foliar spray | Net photosynthesis rate, substomatal CO |
[38] |
| Triticum aestivum | 0.5 mM | Foliar spray | Proline metabolism enzyme, TBARS, H |
[31] |
| Triticum aestivum | 0.1, 0.3 and 0.5 mM | Foliar spray | Chlorophyll content, chlorophyll fluorescence, net photosynthesis rate, psbA transcription, lipid peroxidation, O |
[41] |
| Cicer arietinum | 0.2, 0.4, 0.6, 0.8 and 1.0 mM | Foliar spray | Heat tolerance assessment, H |
[60] |
| Triticum aestivum | 100 mM | Foliar spray | Differential protein profiling, heat responsive transcription factors, stress associated genes, soluble starch synthase activity, morphology of starch granule and starch content | [35] |
| Solanum lycopersicum | 0.25 and 0.5 mM | Foliar spray | Growth and yield characteristics, chlorophyll content, carotenoid content, total soluble sugar, proline content, MDA content, POX (peroxidase) and PAL activity | [61] |
| Gossypium hirsutum | 0, 0.5, 1.0 and 1.5 mM | Foliar spray | H |
[52] |
| Matricaria chamomilla | 0, 1, 10, 26 and 100 mg·L |
Foliar spray | Physiological characters, chlorophyll content, free proline concentration, essential oil | [62] |
| Brassica juncea | 0, 50, 100 and 150 ppm | Foliar spray | Relative water content, photosynthetic parameters, chlorophyll content, heat susceptibility index, membrane stability index | [63] |
| Zea mays | 10–800 µM | Foliar spray | Seedling growth, antioxidant metabolites and antioxidant enzymes | [45] |
| Oryza sativa | 0.01, 0.1, 1, 10 and 50 mM | Foliar spray | Pollen viability, antioxidant enzymes, H |
[34] |
| Triticum aestivum | 10 |
Seed priming and foliar spray | Electrolyte leakage, chlorophyll content, proline content, soluble sugar, soluble protein, yield attributes | [32] |
| Triticum aestivum | 10 % | Foliar spray | Leaf growth, yield attributes, relative water content, chlorophyll content, proline content, total soluble sugar, total phenolics, antioxidant enzymes, grain quality | [53] |
| Oryza sativa | 100 mg·L |
Foliar spray | Seedling parameters, total soluble sugar, total soluble proteins, nitrate reductase, nitrite reductase, uptake of nutrients (N,P,K, Mg) | [64] |
| Mentha |
2, 3 and 4 mM | Foliar spray | Relative water content, antioxidant enzymes, essential oils | [51] |
| Solanum lycopersicum | 1 mM | Foliar spray | Photosynthetic efficiency, water use efficiency, electrolyte leakage, antioxidant enzymes, chlorophyll content, MDA content, H |
[42] |
| Euphorbia pulcherrima | 200 and 400 µM | Foliar spray | Electrolyte leakage, MDA content, antioxidant enzymes, plant appearance | [65] |
| Capsicum annuum | 0, 0.002, 0.01, 0.05 and 0.25 mM | Petri plate medium and foliar spray | Antioxidant content and enzymes, seedling parameters, relative electrical conductivity, relative water content, root activity, MDA content, soluble sugar and protein, proline content, chlorophyll content, photosynthesis parameters | [46] |
| Lablab purpureus | 0.5 and 1 mM | Foliar spray | Photosynthetic efficiency, chlorophyll content, leaf water content, electrolyte leakage, yield attributes, carotenoid content, MDA content, proline content, H |
[39] |
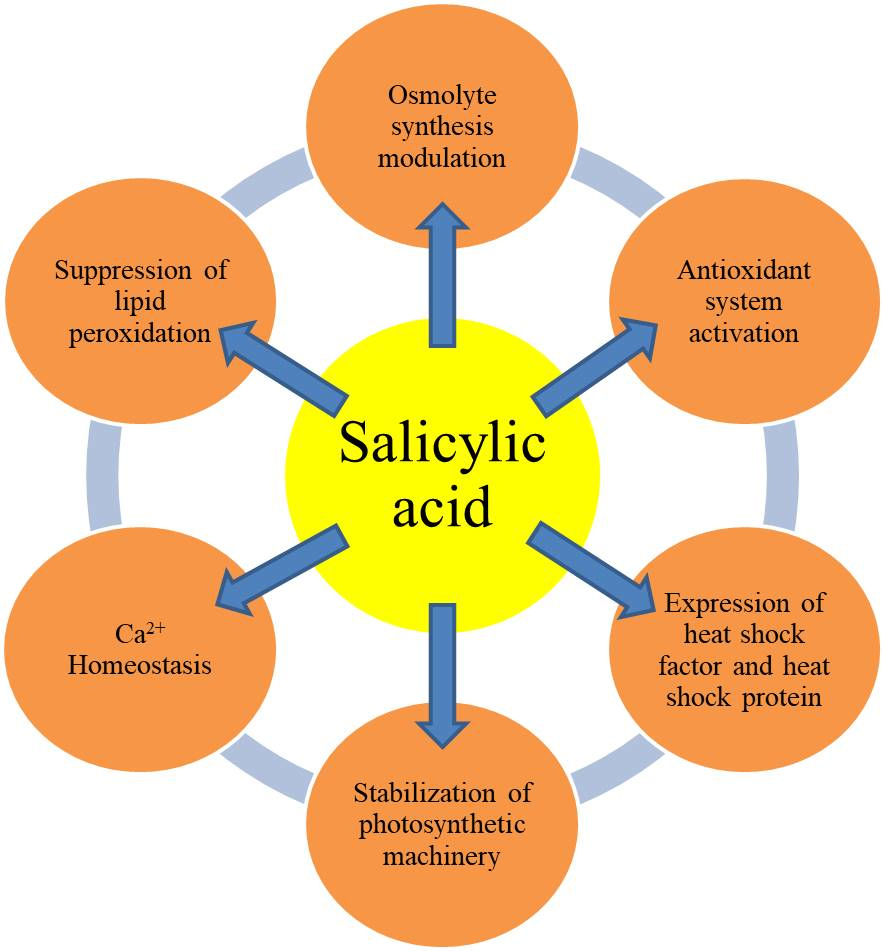 Fig. 1.
Fig. 1.Interaction among SA and various components of the plant system contributes to heat stress tolerance.
The occurrence of high-temperature stress in the course of the reproductive phase significantly affects the production of pollens and seeds in some botanical species, viz. rice [66], wheat [67], tomato [68], cowpea [69] and Arabidopsis [70]. Heat stress is not suitable for flower development, especially the development of stamens. The extent of vulnerability varies between male and female reproductive systems; female reproductive organs are less susceptible to heat stress than male reproductive organs, as evidenced by the significant susceptibility of tapetal cells present in the anther [71]. The most sensitive events in the reproductive phase of plants are pollen mother cells (PMC) meiosis, microspore development, tapetum development, anther dehiscence/pollen release, and the fertilization process [72, 73]. Degeneration of microspores and increased cell size (hypertrophy) of tapetum are the most prevalent effects which result in male sterility [74]. In rice, exposure to heat for 4 days or more during the early stages of anther development caused the tapetum cells to degrade prematurely and to undergo programmed cell death (PCD), which resulted in male infertility [75]. Additionally, downregulation of some crucial genes linked to male sterility, viz. YY1, YY2, TGMS, and tms5 due to heat stress have also been reported in rice [76]. Similarly, the YUCCA gene in Arabidopsis exhibited lower expression under heat stress conditions, particularly in tapetum cells and PMC, resulting in male fertility reduction [77].
SA has successfully reduced the harmful impacts of this stress in vegetative
stages, but the reproductive stage is far less explored in this respect. Most
works on the protective effect of SA during the reproductive stage have been
performed predominantly on rice than on any other plant species. Fu et al. [78]
reported the protective results of SA in alleviating some damages caused in the
process of spikelet differentiation by heat stress in rice. SA increased the
actual light quantum efficiency and P
Furthermore, genes involved in tapetum development, viz. EAT1, MIL2, and DTM1 were also engaged in heat tolerance mechanisms mediated by SA. The pathway might
have also been dependent on hydrogen peroxide (H
Heat shock proteins (HSPs) are multi-family proteins that act as chaperones in nature and play a key role in the survival mechanism of organisms under various stresses [81]. They exist in five families based on their molecular weight, namely HSP100, HSP90, HSP70, HSP60/40, and HSP20 [82]. Heat shock proteins from all of these families have distinct and essential roles in different developmental processes in plants, especially under abiotic stress conditions [83]. Depending on their location, substrate selectivity, and transcript level pattern, HSPs can perform additional tasks. Small heat shock proteins (sHSPs) express when plants are subjected to heat stress. The sHSPs protect photosystem II (PS II) and the whole electron transport chain by binding to thylakoid membranes [84]. Many processes involving HSP101 and class I & II sHSPs help acquire thermotolerance [85]. Kumar et al. [35] discovered that when SA was applied to wheat plants under stress, it increased the expression of transcription factors [Heat stress transcription factors (HSF4 and HSF7) and Drought responsive element binding protein (DREB)] as well as heat-responsive proteins [HSP17, oxygen-evolving enhancer protein, ATP synthase small subunit, rubisco activase, cyclin-dependent protein kinase (CDPK), and superoxide dismutase (SOD)]. Also, the SA-treated plants exhibited a lesser reduction in starch content and soluble starch synthase (SSS) alongside protection to the starch granule morphology of thermotolerant wheat cultivar (C 306) compared to susceptible (PBW 343). SA increased the accumulation of HSP/HSC70 in tomato seedlings [36]. However, the presence only of SA did not affect the HSP70 level, and heat shock was also required. In both SA and heat shock, the seedlings showed a high Hsf-DNA binding, suggesting that the SA modulates Hsf, leading to the HSP70 accumulation. It also enhances the expression of hsp70, hsfA1, hsfA2, and hsfB1. Earlier, Cronjé et al. [86] also observed while working with tobacco protoplasts that SA alone did not influence HSP70 proliferation. SA could either induce or inhibit HSP expression depending on ambient conditions. It was observed that at room temperature, i.e., 26 °C, it did not influence the expression of hsp101 and hsp17.6. However, at mild heat shock temperature (37 °C), it remarkably reduced the induced expression of hsp101 and hsp17.6 [37]. A clone designated Oshsp18.0 CII is rare in class II sHSPs and gets induced by SA [87]. However, SA did not cause Oshsp18.0 CII protein accumulation, or the level was too low to be detected through western blot analysis. Wang et al. [38] observed the accelerated recovery of plants when treated with SA and noticed high levels of HSP21 during the recovery period. SA also enhanced the expression of hsp17, and this expression was higher in thermotolerant (C 306) in comparison to thermosensitive (PBW 343) wheat [35]. Similarly, Rai et al. [39] also observed upregulated expression of hsf in Lablab purpureus under heat stress.
Photosynthesis is a delicate process in green plants [88]. When stress hits a
plant, photochemical reactions are the primary site of injuries in the thylakoid
lamellae in chloroplast stroma [89]. As an impact of heat stress, the
dissociation of the oxygen-evolving complex (OEC) imbalances the flow of
electrons from OEC to the acceptor of PS II [90]. Other susceptible reaction
center parts, such as D1 and D2 proteins, are also impaired by heat stress [91].
Higher quantum yield efficiency (F
Additionally, the PS I was more protected during the recovery phase than the
control. When SA is applied, photosynthesis is protected against heat stress by a
combination of physiological and biochemical mechanisms, including increased
water potential (due to Rubisco protein stabilization), higher nitrogen uptake,
and higher N allocation to Rubisco protein, i.e., higher photosynthetic nitrogen
use efficiency and reduced stress ethylene production with elevated
F
According to Jahan et al. [42], F
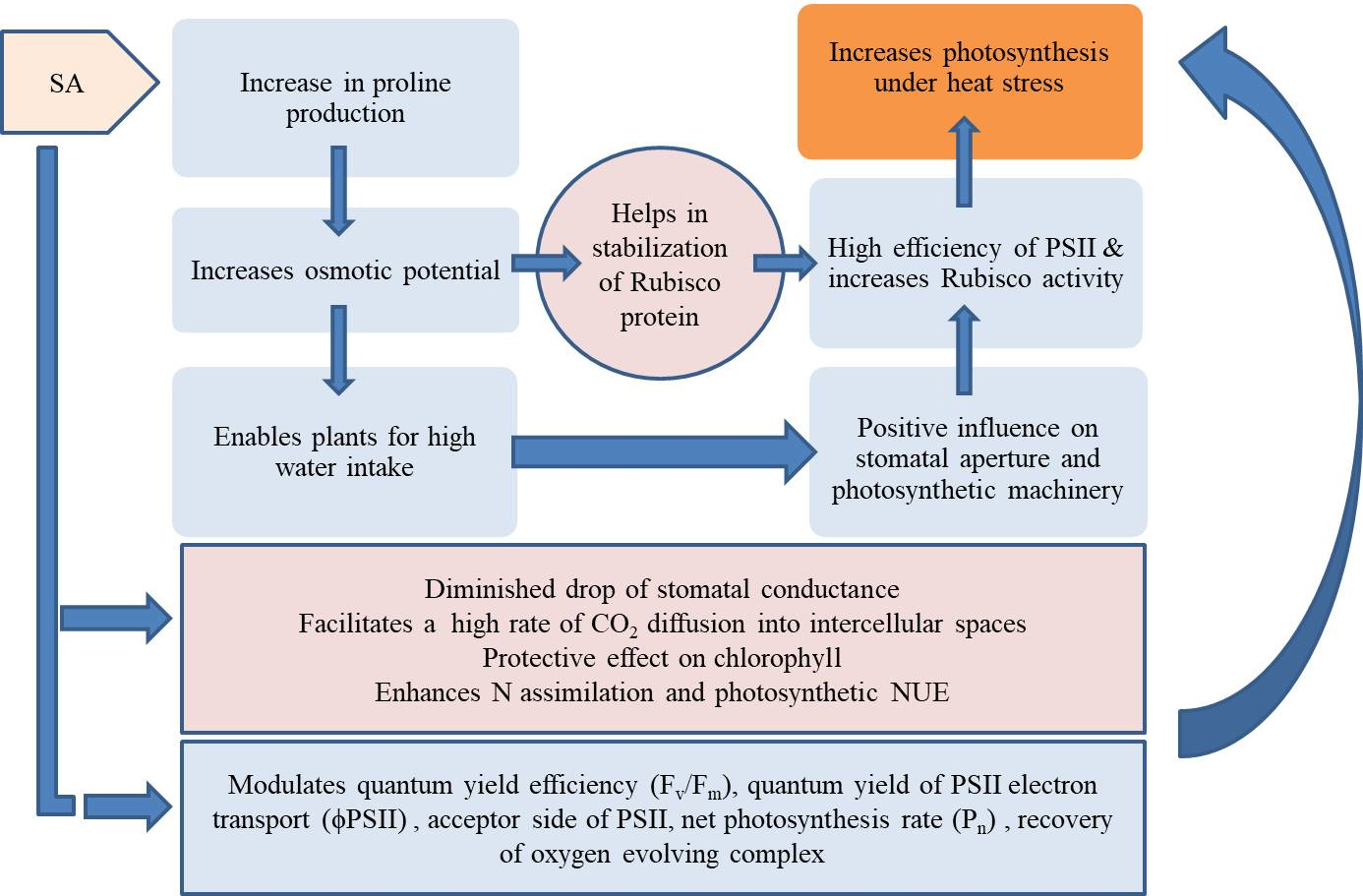 Fig. 2.
Fig. 2.SA mediated protection of Photosystem II and enhanced photosynthesis under heat stress.
Stress causes injuries to membranes, which leads to oxidative damage. Lipid peroxidation is indicated by measuring thiobarbituric acid reactive substances (TBARS) concentration, a standard assay for oxidative membrane damage [94]. A large proportion of linolenic acid (polyunsaturated fatty acids) in the leaves is confined to the thylakoid glycolipids. The oxidative degradation of linolenic acid specifically leads to TBARS formation. Therefore, its development in the leaves is a possible measure of peroxidative harm to the chloroplast membrane [95]. Elevated amounts of TBARS were present in plants exposed to high oxidative stress levels. SA pre-treated plants had lower TBARS than untreated plants in Agrostis stolonifera [43]. This ultimately led to 3-fold lower oxidative damage than controls by the end of treatment. Specific proteins are protected by the foliar spray of SA, characterized by reduced TBARS which results in reduced peroxidation [40]. In a study on grape plants under heat stress, SA pre treatment resulted in reduced TBARS content and protected the plants [44]. Similarly, Khan et al. [31] also observed that SA treatment reduced TBARS content in heat-stressed wheat plants.
Malondialdehyde (MDA) is a marker of oxidative stress resulting from ROS degeneration of polyunsaturated lipids under stress conditions. When SA was applied to stressed wheat plants, the MDA levels were considerably reduced compared to plants only subjected to stress [41]. It also enhanced the decline of MDA content in the recovery period. The MDA content declined with 1.5 mM SA, which helped develop thermotolerance in cotton seedlings. The MDA content of maize seedlings was reduced under heat stress due to the foliar spray of SA [45]. Declined MDA content was also reported by Zhang et al. [46] in ornamental pepper by SA foliar spray under heat stress. This could be credited to an enhanced antioxidant defense system and alleviated heat stress-induced membrane damage.
In the typical aerobic metabolism of plants, ROS like superoxide (O
Externally applied SA controls antioxidant enzymes activities and increases
plant tolerance to abiotic stress. For example, an increase in the concentration
of H
SA transiently increased APX and lowered CAT but did not affect SOD and
peroxidase (POX) in creeping bentgrass [43]. On the other hand, suggestions also
prevail regarding the involvement of SA in scavenging active oxygen species (AOS)
under heat stress as it increases the activities of CAT and SOD [48]. It also
suppressed the O
Induced heat-stress tolerance response of 100
Among the stress survival adaptive mechanisms, the accumulation of osmolytes
like proline is worth discussing [101]. Plants evolved this mechanism to adapt to
osmotic and ionic stresses and aid in the turgor maintenance in stressed plants
[102]. Under stressful environments, proline biosynthesis via glutamate and
pyrroline-5-carboxylate is enhanced in chloroplasts [103]. Proline regulates
cellular osmotic homeostasis, safeguards biological membranes, stabilizes enzymes
and proteins, and detoxifies excess ROS [104]. SA serves as a signaling molecule
by inducing stress-associated genes, which generate osmolytes like proline,
ultimately enhancing thermotolerance in plants [105]. In this regard, Chakraborty
and Tongden [50] observed significantly higher proline content in SA-treated
Cicer arietinum seedlings than heat acclimatized seedlings. High proline
accumulation is indicative of enhanced tolerance to heat-induced oxidative
damage. SA (0.5 mM) treatment to heat-stressed wheat plants increased
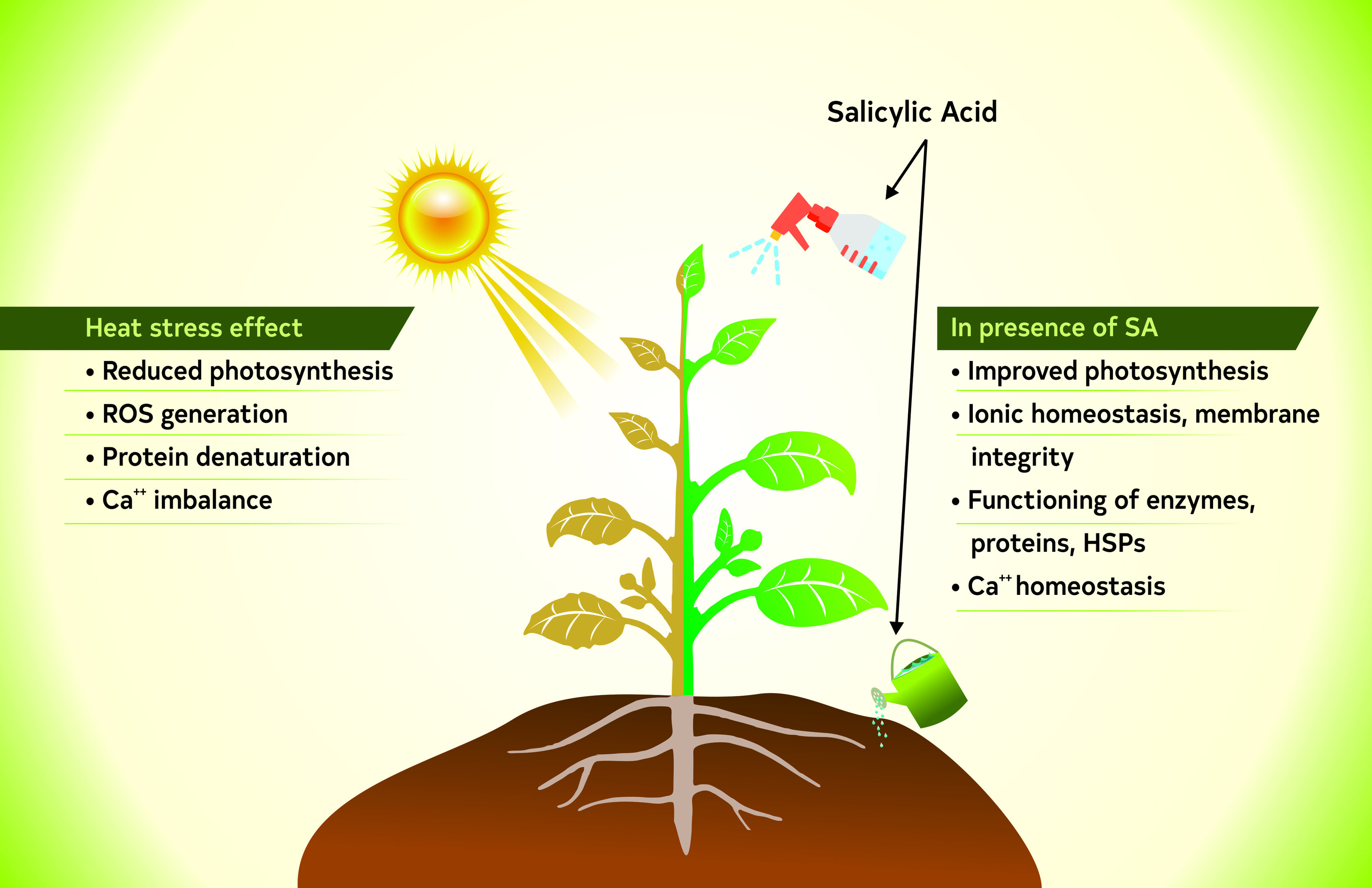 Fig. 3.
Fig. 3.Effects of heat stress and salicylic acid application in plants.
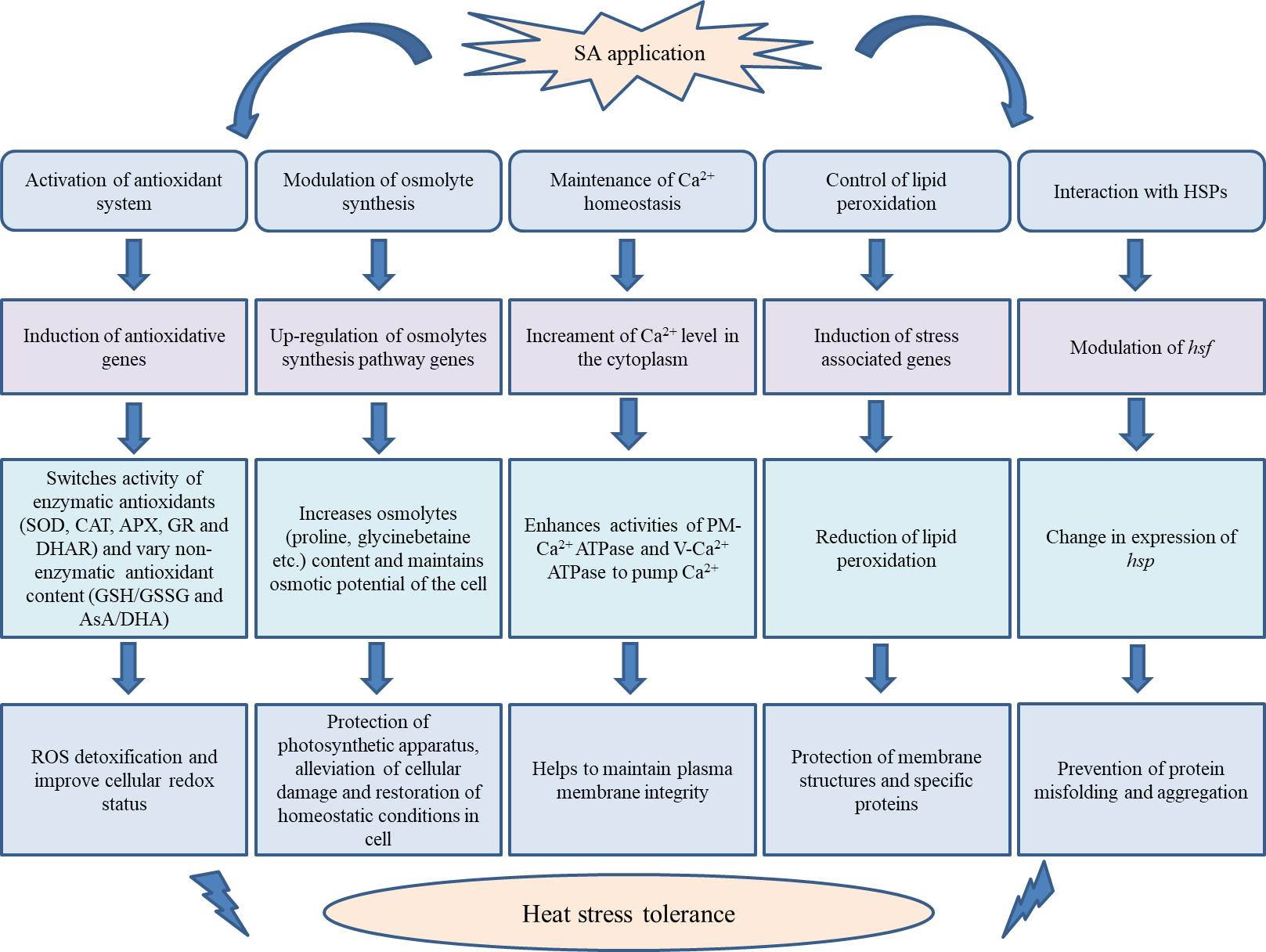 Fig. 4.
Fig. 4.A potential mechanism underlying SA-conveyed plant heat stress tolerance.
Studying signal transduction during plant response and adaptation to stress is
fascinating. During the most recent three decades, broad scope of extracellular
signals to specific responses has been elucidated, including Ca
Heat stress adversely affects almost all plants’ vegetative and reproductive health. Mitigation of the negative impact of heat stress involves applying various plant growth regulators/chemical agents. However, in recent times, salicylic acid gained a popular place in heat stress mitigation strategies. The crucial role of salicylic acid in plant growth and development is the main factor behind the popularity of SA as an ‘effective therapeutic agent’ for plants growing under diverse environmental conditions. It performs multiple roles, as evident by different studies conducted to understand its mechanism of action. Literature appraised herein confirmed the role of SA in protecting the plants from heat by modifying various physiological/biochemical processes. Its application improves the efficiency of almost every mechanism crucial for a plant’s survival under heat, i.e., reproductive system, especially pollen health, HSPs production, which are involved in mitigation signaling throughout the plant system, photosynthetic machinery, antioxidative system to relieve the stress, osmolytes and calcium metabolism which improves chemical homeostasis of the plant cells.
It should be noted that the specifics of the mechanisms that alleviate heat stress and the relationship between various aspects of SA in plant immunity and abiotic stresses are unknown. The area is still an open challenge at both the physiological and molecular levels. Further, extensive genomics and proteomics studies must reveal SA-responsive genes and proteins expressed upon exposure to stress. The complex mechanisms involving SA in producing and signaling specific plant metabolites and their influence on SA-endogenous levels can be unraveled by genomic and metabolomic approaches. The perception, transduction, and retrieval of knowledge regarding SA targets during signaling in the presence of heat stress remain ambiguous. It can be discovered using a multidisciplinary approach that includes genetics, molecular biology, biochemistry, genomics, metabolomics, bioinformatics, and computational biology.
An adequate understanding of its regulatory mechanism is indispensable to encourage future use. Various studies have presented SA as a capable and budding tool in mitigating the adverse effects of abiotic stress in plants. SA is maximally provided as a foliar spray. The development of its nanostructures such as nanoparticles, nano-formulations, nano-conjugates, or nano-encapsulation can be done to have better penetration efficiency at specific sites in the plant system. This innovative delivery system could allow the slow and constant release of the active component. Since SA application and its possible outcomes are highly dependent on the dose/amount applied, it is a fact that higher SA concentration doesn’t profit plants. Therefore, additional research into the heat stress-regulated SA-catabolism process may be worthwhile to exploit SA as a potential phytoprotectant molecule. Another exciting area of study is to understand ecological implications related to SA application to plants as well as studying its signaling mechanism through the angle of ecology and evolution. These potential studies indicate several prospects for creating approaches that can improve overall sustainability in agricultural production systems through effective and productive management of abiotic and biotic problems posed by climate change.
ABA, Abscisic acid; ACS, 1-amino cyclopropane carboxylic acid synthase; AOS,
Active oxygen species; APX, Ascorbate peroxidase; ASA, Acetylsalicylic acid; AsA,
Ascorbate reduced; ATP, Adenosine triphosphate; Ca
Conceptualization and supervision—SY, HSJ, JAP; Writing—SS (Sonali Sangwan), NS, HT, SS (Sushma Sharma); Writing-Review and editing—SY, JAP, PP, HP, RZS; Formal analysis—PP, WHA, RZS; Fund acquisition—PP, WHA. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
The authors extend their appreciation to the Deputyship for Research and lnnovation, Ministry of Education in Saudi Arabia for funding this research work through the project number “20-UQU-IF-P1-001”. The authors would like to thank the Deanship of Scientific Research at Umm Al-Qura University for supporting this work by Grant code (22UQU4310387DSR33).
This work was funded by the Deanship of Scientific Research at Umm Al-Qura University by Grant Code (Project Code: 22UQU4310387DSR33). Open access funding provided by the University of Helsinki, Helsinki, Finland.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




