1 Metagenomics and Secretomics Research Laboratory, Department of Botany, Dr. Harisingh Gour University (A Central University), 470003 Sagar, Madhya Pradesh, India
2 Mycorrhizal Research Laboratory, Department of Biochemistry and Microbiology, Rhodes University, 6140 Grahamstown (Makhanda), South Africa
3 Department of Zoology, Dr. Harisingh Gour University (A Central University), 470003 Sagar, Madhya Pradesh, India
4 Centre for Rural Development and Technology, Indian Institute of Technology Delhi, 110016 Hauz Khas, New Delhi, India
5 Ashoka Trust for Research in Ecology and the Environment, 560064 Bangalore, Karnataka, India
6 Department of Botany, Mohanlal Sukhadia University, 313001 Udaipur, Rajasthan, India
7 Microbiology and Genetics Department/CIALE, University of Salamanca, 37007 Salamanca, Spain
Abstract
Climate change, water scarcity, population growth, and food shortage are some of the threatening challenges being faced in today’s world. Among different types of stresses, drought stress presents a persistent challenge for global food production, however, its harshness and intensity are supposed to expand in the imminent future. The most striking effects of drought stress on plants are stunted growth, severe damage to photosynthetic apparatus, reduction in photosynthesis, reduction in seed germination, and nutrient uptake. To deal with the destructive effect of drought stress on plants, it is necessary to consider its effects, mechanisms of action, the agronomic and genetic basis for sustainable management. Therefore, there is an urgent need for sustainable solutions to cope up with the negative impact of drought stress. This review focuses on the detrimental effects of drought stress on plants’ morphological, physiological, and biochemical characteristics and recommends suitable drought management techniques to reduce the severity of drought stress. We summarize the effect of drought stress on physiological and biochemical parameters (such as germination, photosynthesis, biomass, water status, and nutrient uptake) and yield. Overall, in this article, we have reviewed the role of different phytohormones, osmolytes, exogenous compounds, proteins, plant growth-promoting microbes (PGPM), omics approaches, and genome editing technologies like clustered regularly interspaced short palindromic repeats (CRISPR) and CRISPR-associated protein 9 (CRISPR-Cas9) in alleviating drought effects in plants. We also proposed that developing drought-tolerant plant varieties requires the combined use of biotechnological and agronomic approaches and cutting-edge genome editing (GE) tools.
Keywords
- Microbiome
- Abiotic stress
- Drought
- Plant growth-promoting microbes
- CRISPR
Several driving forces are reshaping global food security, including population growth, drastic climate changes, water supplies, arable land availability, food availability, and biodiversity losses [1, 2]. The plants are continually exposed to a wide range of climatic perturbations, leading to different types of abiotic stress conditions like drought, salinity, flooding, heat, cold, heavy metals contamination, and biotic stress conditions [3, 4]. Among these factors, drought stress has become the most important and the key limiting factor that negatively affects crop productivity and eventually compromises food security [5]. Drought not only causes significant losses in agricultural productivity but it also causes desertification, soil erosion, and ecological degradation. As a result, the water shortage has been identified as a critical global and environmental issue [6]. These stress conditions contribute to significant physiological, metabolic, and molecular changes in plants, causing a reduction in agricultural production [7]. Researchers have made significant progress in elucidating the genetic, biochemical, and signalling pathways involved in plant drought responses in recent years with the help of modern technology (such as molecular genetics, genomics, transcriptomics, metabolomics, and proteomics) [3, 6]. However, the complex processes that distinguish resistance from a vulnerability within a species, particularly in crops, are still completely unknown [6]. Drought stress affects several morpho-biochemical parameters, such as leaf area reduction, reduction in total chlorophyll content, wilting of leaf, elongation of the root, and production of reactive oxygen species (ROS) [4, 8, 9]. Plants generally absorb water from the soil effectively through their root system, partially shut their stomata to decrease water loss from transpiration, and change their metabolism to match the present carbon supply in the early stages of drought stress conditions [6, 10]. Under stress conditions, some osmolytes, including prolines, glycine betaine, soluble sugars, and spermine, accumulate inside plant cells to maintain cell osmotic potential [11]. Drought-stressed plants are also reported with alterations in the activity of many oxidation-protective enzymes such as catalase (CAT), superoxide dismutase (SOD), glutathione reductase (GR), and ascorbate peroxidase (APX) [12]. However, drought stress causes changes in the expression of many genes, including those associated with stress signal transduction, as well as the transcription and regulation of hundreds of functional proteins, all of which contribute to the genetic regulation of drought tolerance [13].
Thus, sustainable agro-biotechnology needs to be designed to combat abiotic stress for maintaining or increasing crop production worldwide. Several strategies have been implemented around the globe to improve drought tolerance in crops, i.e., by using genetic engineering and traditional plant breeding methods [14, 15]. Since drought stress tolerance is a multigenic and complex trait, creating transgenic drought-resistant crop varieties is a tedious job [16]. To embark on this, applying drought-tolerant bio-inoculants or plant growth-promoting rhizobacteria (PGPR) is an alternative strategy for sustainable agriculture under water deficit conditions. Many studies conducted by different researchers support the fact that PGPRs can help plants to withstand abiotic stresses by altering the responses at the gene level [17, 18]. PGPRs have great potential for amending physiological responses to water scarcity, thereby enhancing plant endurance under drought stress conditions [8, 19]. Various reports are available that demonstrates the role of PGPRs in extenuating drought stress in many crops, such as tomato, pepper, pea, maize, wheat, mung bean, and chickpea [8, 16, 20, 21, 22, 23, 24].
Interdisciplinary strategies are required to harness the full potential of microbes in agriculture production systems. Through advancements in computational tools, the data-driven science of multi-omics has enhanced our understanding of microbial community structure, dynamics, and composition along with their functional attributions in many complexes and diverse environments like the rhizosphere, where the community level microbial interactions direct the plant responses toward various stresses [25, 26, 27]. However, optimizing plant-microbial partnerships for improving drought tolerance in crop plants is a daunting task given the complexity of plant-microbe and microbe-microbe interactions and the dependence on environmental conditions. This review highlights different management strategies for improving drought stress tolerance in different crop plants that can be achieved by developing drought-tolerant plant genotypes, seed treatments, genetic modifications, application of plant growth microbes, plant mineral nutrients, and the use of compatible solutes.
Drought is a meteorological term that signifies a period with no rain, which leads to a significant reduction in soil and atmospheric humidity and elevation in ambient air temperature [28, 29]. This situation arises due to an imbalance between the water intake and evapotranspiration flux from the soil [28]. Drought stress has a direct and indirect effect on plant health. The low availability of water in roots and the great loss of liquid through transpiration affect the osmotic and ionic balance of plant cells, which leads to phenological changes in the plant [30]. It also affects crop development in different ways by reducing the rate of seed germination, which affects crop performance; similarly, the photosynthetic process is repressed, and the absorption capacity of nutrients is reduced, which facilitates the risk of infestation by pests [31, 32]. Drought is major abiotic stress, altering morphological, biochemical, physiological, and molecular responses by regulating protein functions and stress-induced genes [3, 7]. The study conducted by Kanwal et al. [33] reported drought stress adversely affects the morphology, biochemistry, and physiology of the wheat plant. Under drought conditions, cell elongation can be prevented by interrupting water passage from the xylem to the surrounding elongating cells [34]. Drought often reduces leaf area, plant height, and crop development due to impaired mitosis, cell expansion, and elongation [34, 35]. A notable impact of drought is the decrease in photosynthesis due to a decline in leaf development, and early leaf senescence decreased photosynthesis and the related loss in food production. Published research displayed that during drought stress conditions, all the vital processes such as net photosynthesis, biosynthesis of photosynthetic pigments, electron transport rates, the quantum yield of photosystem II (PSII), protein synthesis, lipid, and energy metabolism are generally ceased [36]. Drought impairs plant growth by disturbing the water balance, membrane permeability, mineral nutrition, and enzyme activity of the plant [3, 7].
Indirect effects of drought stress increase reactive oxygen species (ROS)
production, resulting in oxidative damage to macromolecules [37]. The study
revealed that the abscisic acid (ABA) accumulation in plants during drought
stress conditions helps in the regulation of signal transduction pathway by
phosphorylation/dephosphorylation and controls the transportation of K
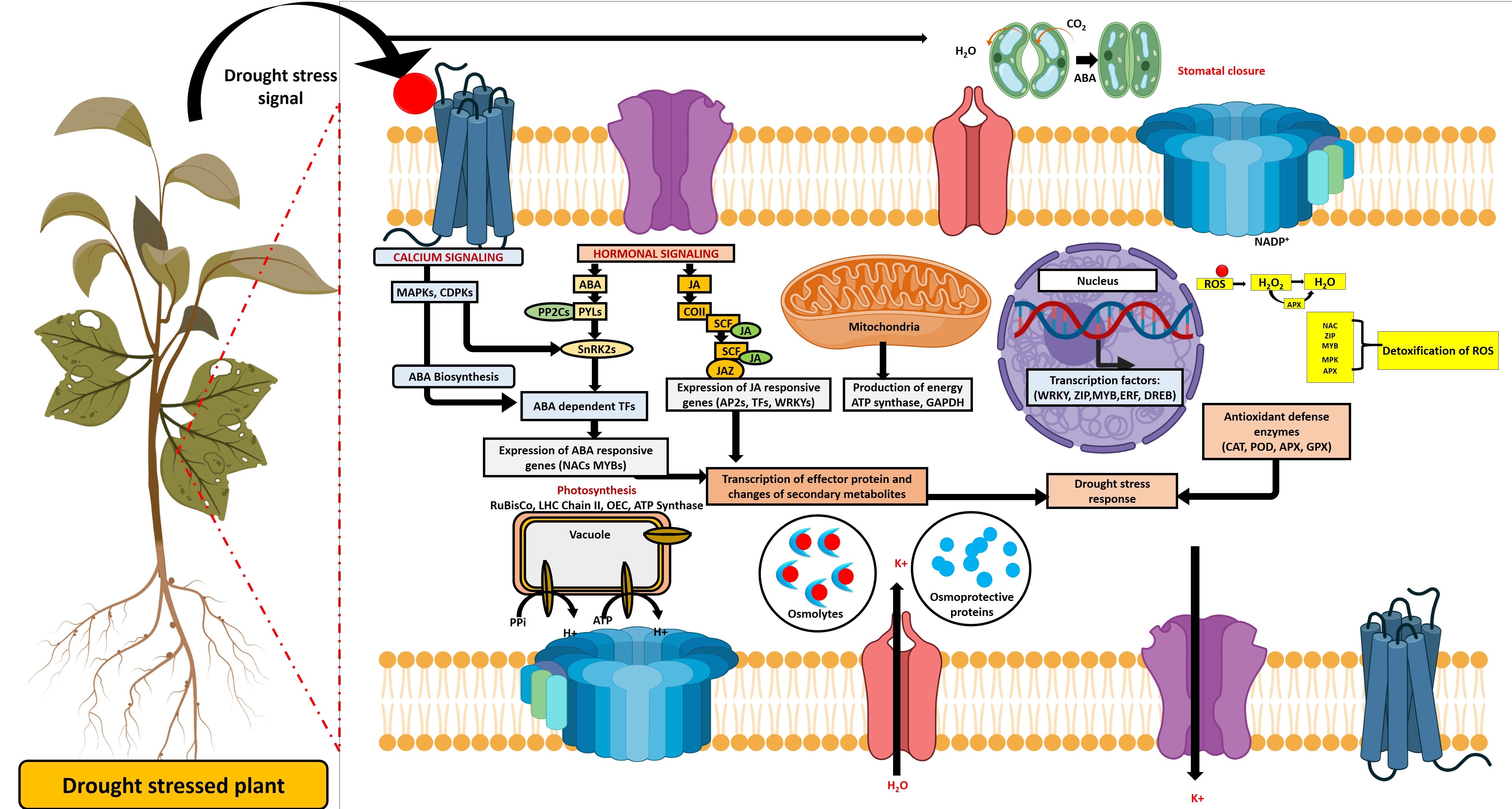 Fig. 1.
Fig. 1.Graphic representation of the mechanism of drought stress tolerance in plants. Abbreviations: ABA, Abscisic acid; APX, ascorbate peroxidase; CAT, catalase; GPX, guaiacol peroxidase; GST, glutathione-s-transferase; JA, Jasmonic Acid; MAPK, mitogen-activated protein kinase; POD, peroxidase; ROS, Reactive Oxygen Species; SOD, superoxide dismutase; TFs, Transcription Factors.
Different studies have revealed strategies to cope with drought stress, such as (i) plant breeding and marker assistant selection [46]; (ii) improving osmotic adjustment by application of exogenously applied compounds and osmolytes like glycine, betaine, proline, or other amino acids, and polyols (sorbitol, pinitol, myo-inositol, and mannitol, among others) [47]; (iii) solicitation of plant hormones (salicylic acid, auxins, gibberellins, cytokinins, brassinosteroids, jasmonic acid, and ABA) [48, 49]; (iv) enhancing antioxidant function such as glutathione, polyamines, ascorbate, and enzymes (SOD, CAT, APX, GR, etc.) [50]; (v) generating transgenics for drought tolerance in plants and [51]; (vi) applying drought-tolerant microbes [52, 53]. Some of the strategies are discussed in the next section in detail.
Phytohormones are recognized to have a key role in controlling various plant responses to varied drought conditions. Additionally, salicylic acid (SA), jasmonic acid (JA), auxin (indole-3-acetic acid, IAA), ethylene (ET), cytokinins (CKs), brassinosteroids (BRs), and gibberellins (GAs) are essential plant hormones to encounter drought stress [54]. Auxins are a class of phytohormones that play an essential role in plant growth, development, and stress response [55]. Auxin production occurs mainly in leaf primordia, young leaves, developing seeds, and its biosynthesis route is mainly conserved among plants. These are delivered from the site of synthesis to the target by phloem or cell-to-cell transfer [11]. Several studies conducted by different researchers have found a positive role of auxin in developing drought tolerance. In research, Aux/IAA genes were discovered in rice, and some genes were triggered by drought stress [56].
Furthermore, one of these genes, OsIAA6, has been linked to drought stress [56]. Another study conducted by Ke et al. [57] and Kim et al. [35] in poplar and potato, wherein YUC6 gene was overexpressed, resulting in auxin overproduction phenotypes and improved drought tolerance. They proposed that the functioning of the YUCCA6 gene in the auxin production pathway in potatoes might be used to manipulate plant responses to the environment [35, 57]. Auxin also increased the ramification of roots that might be critical for improving drought tolerance [58].
Cytokinins (CKs) are among the major phytohormones needed to control plant growth, development, and adaptation to environmental stressors, like drought stress [59]. Several studies reported CKs to have both beneficial and negative impacts on drought tolerance [59, 60, 61, 62]. CK levels can also increase or decrease according to the duration and intensity of the drought. CK levels were increased endogenously in transgenic plants expressing an isopentenyl transferase gene. Drought tolerance was improved in the transgenic plants by delaying senescence and suppressing drought-induced leaf senescence. However, some studies reported that CK has a detrimental influence on drought tolerance [63]. The CK oxidase/dehydrogenase (CKX) system is well-known for the breakdown of CKs, and overexpression of CKX in Arabidopsis often results in the decreased endogenous CK level [63, 64]. The role of CK in drought stress and its signalling route are yet unknown.
Gibberellins (GAs) are tetracyclic diterpenoid carboxylic acids that act as growth hormones and respond to biotic and abiotic stressors. Various studies have demonstrated that the exogenous application of GA improves drought tolerance in maize [65], soybean [49], wheat [66, 67]. The study conducted by Moumita et al. [66] reported foliar application of Gibberellic acid stimulated glyoxalase I (Gly I) and glyoxalase II (Gly II) activity for protecting the wheat seedlings against drought stress. The combined application of GA & ABA improves stress tolerance, scavenging of ROS, and carbon-nitrogen balance in Tobacco [68].
Ethylene (ET) is a natural gaseous hormone with several activities, including fruit maturation, growth, senescence, germination of seeds, flowering, and reaction to different stimuli [69]. ET plays an active role in drought stress tolerance in plants; for example, the study conducted by Scarpecit et al. [70] reported that the overexpression of AtERF019 had delayed growth and senescence of plants improved Arabidopsis’ drought tolerance. In another study conducted by the overexpression of ERF (ethylene response factor), soybean gene GmERF3 exhibited higher drought tolerance in tobacco due to higher osmolyte content than wild-type plants [71].
Different drought-responsive metabolites produced by microbes act as precursors
of plant hormones, such as phenylalanine, which is a precursor for the
biosynthesis of salicylic acid (SA), and another drought stress-responsive
secondary metabolites [72]. For example, in the initial phases of drought, the
oak tree produces secondary metabolites that play a significant role in
rhizosphere signalling whereas primary metabolites are produced during recovery
[73]. Abscisic acid (ABA) is a strongly induced phytohormone generated in
response to drought signals. ABA is generated in the roots of plants and
subsequently transported to the leaves to regulate stomatal aperture, expression
of ABA-responsive genes, and channel activity [73]. ABA also maintains water
status in the plant by guard cell regulation and induction of genes coding for
enzymes and protein associated with dehydration tolerance. By regulating stomata,
guard cells respond to ABA and decrease water loss. Various membrane receptors
like G-protein perceive the environmental stress signals coupled receptors
(GPCRs), receptor-like kinases (RLKs), ion channels, and histidine kinases, which
brings changes in the cytoplasmic Ca
Moreover, genes responsible for jasmonic acid (JA) and salicylic acid (SA) are down-regulated during drought in sweet sorghum [81]. Salicylic acid is a potential non-enzymatic antioxidant that plays a crucial role in eliminating ROS injury. Exogenously applied SA increases SOD, APX, POD, and CAT activities in stressed plants [39]. Salicylic acid-associated exudation signals are involved in systemic resistance and the host plant-mediated selection of microbes in the rhizosphere [81]. The study conducted by Hayat et al. [82] and Khan et al. [83] reported that the drought stress tolerance is stimulated when treated with a low concentration of SA and resulted in improved photosynthetic parameters, chlorophyll content, leaf water potential, relative water content (RWC), membrane stability index (MSI), carbonic anhydrase (CA), and nitrate reductase (NR) activity. The combined application of PGPR and SA enhances the phytoremediation and drought tolerance potential of Sunflower (Helianthus annus) [83].
Methyl Jasmonate (MeJA) is a volatile derivative, a methyl ester of jasmonic
acid (JA) and it is used as plant growth regulator. The study conducted by Anjum
et al. [84] investigated the role of MeJA in enhancing drought stress
tolerance in five different wheat cultivators and found that MeJA effectively
mitigates the detrimental effects of drought stress. Glutathione plays a diverse
role in physiological functions for removing harmful ROS and metal detoxification
in stressed plants. Combined seed and foliar application of Methyl jasmonate (20
Brassinosteroids (BRs) modulate plant growth and development, including cell division and elongation, senescence, photomorphogenesis, vascular differentiation, and response to abiotic and biotic stresses [86]. BRs interact significantly with GA to regulate plant development. Previous studies displayed a positive role of BRs for alleviating the negative effect of drought stress in Brassica napus, Arabidopsis, and rice [87, 88, 89].
To address global challenges in agriculture, the studies should be focused on improving the
germplasm, exogenous application of hormones, and developing crop management
practices to increase water use efficiency [84, 90]. ROS are valuable messengers,
leading stressed plants to oxidative signaling, systemic acquired acclimation
(SAA), and systemic acquired resistance (SAR). Oxidative signaling initiates a
cascade of defense mechanisms and growth regulation under stress. ROS (superoxide
and H
Osmoprotectants are generally known as osmolytes due to their cellular defensive
functions against dehydration injury. Different types of osmolytes have been
identified (Fig. 2). Osmolytes mainly comprise of sugars (glucose, sucrose,
fructose, raffinose, trehalose, and fructans), amino acids
(
 Fig. 2.
Fig. 2.Classification of different osmolytes/osmoprotectants.
| Exogenously applied Compounds | Compounds and their combinations | Concentration of Compounds Used | Plant Species | Application | Mechanism of action | References |
| Osmolytes | Glycine betaine (GB) | 0, 50, 100, and 200 mM | Phoebe hunanensis | Foliar application | Enhances permeability of cell membrane and inhibited membrane lipid peroxidase accumulation | [103] |
| Maize, barley | Photosynthetic efficiency maintenance, thylakoid membrane protection, and osmotic adjustment | [104] | ||||
| Flax (Linum usitatissimum) | Foliar application | Increased osmolytes accumulation and enhanced antioxidative enzyme activities | [105] | |||
| Glycine betaine (GB) | (0, 50 and 100 mM) | Wheat | Foliar application | Enhancing plant biomass, transpiration rate, nitrogen and phosphorus content in roots under drought stress | [106] | |
| Glycine betaine + Salicylic acid (SA) | GB 100 mM and Salicylic acid (SA) 0.724 mM | Sunflower | Foliar application | Increased the free leaf proline and oil content under drought | [107] | |
| Glycine betaine + Salicylic acid + Zinc | 11.5 g/L GB+ 140 mg/L SA+ 4 g/L Zn | Maize | Foliar application | Increased proline and sugar accumulation as well as enhanced antioxidative enzyme activities | [108] | |
| Proline | Rice | Foliar application | Up-regulated antioxidants enzyme activity and glycine betaine contents and decrease in lipid peroxidation | [109] | ||
| Maize (Zea maysL.) | Foliar application | Proline improved the antioxidative enzyme activities | [110] | |||
| Proline +ABA | ABA (10 |
Wheat | Foliar application | Improved drought and salinity stress tolerance | [111] | |
| Proline + Salicylic acid | 10 mM proline and 0.5 mM Salicylic acid | Barley | Foliar application | Increased plant biomass, chlorophyll concentration, relative water content, antioxidant enzyme activity, and reduces lipid peroxidation, and hydrogen peroxide (H |
[112] | |
| Trehalose | (0 and 30 mM) | Brassica, Maize | Foliar application | Induced drought stress tolerance by up-regulation of photosynthetic and water relation characteristics as well as antioxidant enzyme activity. | [113] | |
| 40 mM trehalose | Wheat | Foliar application | Improves the physiological status of wheat | [114] | ||
| (0, 10, 20 and 30 mM) | Sunflower | Foliar application | Induced drought stress tolerance | [115] | ||
| GABA and proline (0, 0.25, 0.5, 0.75 and 1 mol/L) | Tobacco | GABA acts as an effective osmolyte to reduce the production of reactive oxygen species (ROS) under drought stress | [116] | |||
| Mannitol + thiourea | M (15 and 30 mM) or T (3.5 and 7.0 mM) | Maize (Zea mays L.) | Foliar application | Scavenge the stress induce oxygen radicals and osmotic adjustment | [92] | |
| Mannitol | 30 mM | Blackgram (Vigna mungoL.) | Foliar application | Scavenge the stress induce oxygen radicals and osmotic adjustment | [117] | |
| D-Ononitol | Arabidopsis | Prevent water loss in plants | [118] | |||
| D-pinitol | Arabidopsis | Prevent water loss in plants | [119] | |||
| Spermidine (Spd) | (0.1 mM) | Maize (Zea mays L.) | Foliar application | Increased plant biomass, and polyamine content | [90, 120] | |
| 0.5 mmol/L | White clover (Trifolium repensL.) | Foliar application | Promotion of ascorbate–glutathione cycle and higher activities of antioxidant enzymes | [121] | ||
| 30 |
White clover (Trifolium repensL.) | Foliar application | Reduction in lipid peroxidation and enhancing the antioxidant defense system | [122] | ||
| SA + Spd | SA + Spd (0.25 mM + 0.05 mM) | Maize (Zea mays L.) | Foliar application | Enhancing the antioxidant defense system and regulating endogenous polyamine metabolism | [120] | |
| Putrescine | 20 and 40 mg/ L | Thymus vulgarisL. | Foliar application | Enhanced leaf water content and dry matter, reduction in cell injury indices; upregulation in antioxidant enzymatic activities. Essential oil content was found to be increased by 23% | [123] | |
| Putrescine + salicylic acid | Putrescine (1 ppm) +SA (100 ppm) | Trachyspermum ammi | Total sugars, phenolics, free amino acids, chlorophyll, and carotenoids were enhanced in shoots and roots | [124] | ||
| Phytohormones | Methyl jasmonate (MeJA) + salicylic acid (SA) | MeJA (20 |
Maize (Zea mays L.) | Seed and foliar application | Prevented drought-induced oxidative stress by modulating ABA and osmolytes level and antioxidant enzymes activity. | [85] |
| Gibberellic acid (GA) | Wheat | Foliar application | Stimulated activities of glyoxalase I (Gly I) and glyoxalase II (Gly II) for protecting the wheat seedlings against drought stress | [66] | ||
| GA & ABA | Tobacco | Improves stress tolerance, scavenging of ROS, and carbon-nitrogen balance | [68] | |||
| Methyl jasmonate (MeJA) | 250 mg L |
Citrus | Increased chlorophyll, sugar and proline contents and significantly decreased the H |
[125] | ||
| Jasmonic acid + abscisic acid | JA (100 |
Soybean | Relieving the adverse effects of drought stress and regulating trypsin inhibitor | [126] | ||
| Brassinosteroids (BRs) | 0.01 |
Brassica napus, Arabidopsis, and rice | Seed application | Improved leaf water economy and CO |
[87, 88, 89] |
Abiotic stress, mainly drought stress is usually linked with the dysfunctioning of different proteins and enzymes. So, under abiotic stress conditions, it is essential to avoid accretion of non-native proteins and retain the proteins’ functional conformation. Therefore, the plant generally activates different genes under drought stress, producing different kinds of protein. Usually, the drought stress response is associated with the production of two kinds of proteins: (1) late embryogenesis abundant (LEA) proteins and (2) heat-shock proteins (HSPs) [77].
LEA proteins were initially categorized in seed, but they are also present in other vegetative tissues of the plants. The Late embryogenesis abundant (LEA) gene is either induced by applying ABA or environmental stress signals such as drought, low temperature, and salinity, etc. It has been suggested that they behave as water-holding molecules in ion sequestration and can stabilize membrane and protein. LEA proteins that play an important role in the mechanism of drought stress tolerance are known as dehydrin [133, 134]. The study conducted by different scientists has characterized LEA proteins in a variety of plants like cotton [134], barley [133, 134], rice [135], and wheat [136] for investigating their functional role in drought stress tolerance.
Plants under field conditions need to deal with many abiotic and biotic stresses simultaneously [137]. Plants respond to these stressors through continuous transcriptional and translational regulation of diverse metabolic pathways [138]. Understanding these complex plant stress response pathways is essential and involves a combined effort of a squad of molecular chaperones like Heat Shock Proteins (HSPs) [139]. HSPs are highly overexpressed in plants responding to abiotic stress, comparatively very little attention has been given to recognize their functional role in stress tolerance. The roles of these HSPs belonging have been identified in different plants like Arabidopsis thaliana [140], Soybean [141, 142], and Rice [143, 144]. The two Nuclear/cytosolic HSPs, GmDjp1 and DNAj in soybean, have been associated with abiotic [142] and biotic [141] stress tolerance. Proteins present in endoplasmic reticulum (ER) HSP90s is also named as glucose-regulated protein 94 (GRP94), which is responsible for drought stress tolerance in Xerophyta viscosa, a drought-tolerant plant [145]. Another ER-resident Hsp70s known as BiPs (luminal Binding Proteins) the overexpression of SoyBiPD (soybean BiPs) conferred drought stress tolerance in tobacco.
Nanoparticles are highly reactive due to their high surface area, tunable pore size, and particle structure. Nanoparticles are designed to target exact cell organelles in the plant for releasing their content [146]. They are generally involved in the up-regulation of antioxidant enzymes such as CAT, SOD, POD, APX etc. Silicon-based nanoparticles (SNPs) can efficiently alleviate various abiotic stresses such as drought, heat, chilling, salt stress, and heavy metal toxicities [77]. The mechanisms involved in alleviating stresses involve (1) activation of antioxidant defense systems in plants, (2) co-precipitation of Si with toxic metal ions, (3) toxic metal ions immobilization in growth media, (4) uptake, and compartmentation of metal ions inside plants. The study conducted by Linh et al. [147] reported that metal-based (copper, iron, cobalt, zinc oxide) nanoparticles could enhance drought stress tolerance in soybean by enhancing relative water content, biomass reduction rate, and drought tolerance index. Another study conducted by Sedghi et al. [148] reported that the application of zinc oxide nanoparticles increases seed germination (%) and overall germinates rate, suggesting that ZnO nanoparticles are more effective for using seed reservoirs for seedling growth and improve drought stress tolerance. A recent study by Ali et al. [149] used chitosan nanoparticles to improve drought stress tolerance in Catharanthus roseus (L.) by biochemical and modulation of gene expression.
Plants are intimately associated with an enormous diversity of microbes present in the rhizosphere, endosphere, phyllosphere, and other components such as nectar and pollen are collectively known as the plant microbiome [9, 150]. Root exudate release by plants comprises numerous organic compounds which provide better plant absorption, increase growth, provide higher yields, and provide the home for these microbes [151, 152]. Several studies have shown that plant microbiomes play essential roles, such as the enhancement of biotic (pathogens) and abiotic (drought, salinity) stress tolerance, nutrient cycling and transformation of organic matter, and the improvement of the plant growth and yields by promoting nutrient availability, biological nitrogen fixation, and production of phytohormones and other secondary metabolites [47, 153, 154]. Plant recruitment of a drought-tolerant microbiome might be an evolved attribute, with generations of recurrent drought responses leading to the establishment of beneficial and stable plant-microbe interactions that benefit both microbe and host plant [155, 156]. The study conducted on Brassica rapa plants subjected to drought for generations enhanced bacterial diversity and richness around roots in dry conditions than control plants [157]. Examining previously reported plant growth-promoting microbes (PGPM) from drought-stressed plants could reveal which microbial features are likely to be beneficial to plants. Indeed, bacteria with plant growth-promoting (PGP) capacities have been detected more in soils and roots [15], particularly in persistently drought-stressed areas [155]. The use of plant growth-promoting rhizobacteria (PGPR) for alleviating the harmful effect of drought stress has been extensively studied by different scientists in different crops, such as soybean [4, 158], wheat [159, 160], chickpea [8, 161], maize [162, 163], rice [164], and mung bean [24, 165] (Table 2, Ref. [4, 167, 166, 161, 168, 159, 30, 165, 164, 8, 98]). A number of PGP abilities confers drought resistance, and the most studied is the enzyme 1-aminocyclopropane-1-carboxylate deaminase (ACCd). The plant hormone ethylene is maintained below inhibitory levels by ACCd activity, allowing normal root development and delaying senescence during drought [162, 169]. The study conducted by different scientists reported use of ACCd producing microbes in alleviating the negative effect of drought stress in Vigna mungo L. and Pisum sativum [21, 170], maize [162], soybean [4], and Capsicum annuum [171]. These PGPR microbes also synthesize other plant hormones, like auxin analog indole-3-acetic acid (IAA), which improve root and shoot growth and help in other plant developmental processes [172]. PGPB may have a role in nutrient cycling during droughts, such as diazotrophy, siderophore production, and phosphorus solubilization [173].
| Bacterial | |||||
| S.No | Microbes | Isolated from | Target plant | Reported role | References |
| 1. | Bacillus cereus, Pseudomonas otitidis, and Pseudomonas sp. | Rhizosphere | Soybean | Increased plant biomass, accumulation of proline, sugar, and decrease osmotic stress under drought stress condition | [4] |
| 2. | Bradyrhizobium japonicum and thuricin-from Bacillus thuringiensis | Soybean rhizosphere | Soybean | Increase in nodule biomass, and photosynthetic rate | [167] |
| 3. | Bacillus cereus UFGRB3, B. subtilis UFGS2, and B. thuringiensis UFGRB2 | Soybean rhizosphere | Soybean | Differential gene expression and increased plant growth and biomass | [166] |
| 4. | Pseudomonas putida and Bacillus amyloliquefaciens | Chickpea | Chickpea | Increased root biomass, which leads to higher nutrients uptake | [161] |
| 5. | Burkholderia phytofirmans PsJN | Wheat rhizosphere | Wheat | Improved the antioxidant levels, ionic balance, and also increased the phosphorus, potassium, nitrogen, and protein concentration in wheat grains | [168] |
| 6. | Klebsiella sp., Flavobacterium sp, and Enterobacter ludwigii | Wheat | Wheat | Increased plant biomass, accumulation of proline, sugar, and decrease osmotic stress under drought stress condition | [159] |
| 7. | Azotobacter chroococcum, Azospirillum brasilense, and Rhizobium japonicum | Soybean rhizosphere | Soybean | Increased phytohormones production and decreases polyamine accumulated under drought stress condition | [30] |
| 8. | Pseudomonas aeruginosa GGRJ21 | Mung bean rhizosphere | Mung bean | Increased plant biomass, upregulation of drought stress-responsive genes, and accelerated production of proline | [165] |
| Fungal | |||||
| 9. | Piriformospora indica | Rice | Rice | Increased chlorophyll fluorescence, accelerated production of proline and improved the antioxidant activity | [164] |
| 10. | AMF: Glomus etunicatum, Glomus mosseae, and Glomus intraradices | Maize | Chickpea | Increased the weight and number of nodules, leghemoglobin content and nitrate reductase enzyme activity under drought stress condition | [8] |
| 11. | AMF: Rhizophagus clarus | Strawberry plant | Strawberry plant | Increased plant biomass by increasing water content, water use efficiency, antioxidant enzyme activity, photosynthesis rate, and the nutritional status of mainly Zinc | [98] |
Plant endophytes residing in the intercellular spaces have been shown to relieve drought stress by evolving several mechanisms to sustain plant growth even under stressful conditions [174, 175]. Even when drought significantly modifies the microbial communities associated with plants, this drought-tolerant microbiota has a great positive influence on the phenological development of stressed plants [31, 153]. Thus, endophytes are considered as one of the most promising and easily available resources to improve plant tolerance under drought stress [4]. However, understanding the biochemical and physiological mechanisms of drought tolerance that act at the whole-plant level is necessary before employing these endophytes on a large scale [176]. Plants under drought stress events experience several physiological, metabolic, and molecular changes regulated or influenced by microbiota. Endophytes produce several secondary metabolites, many similar to those of the host, or regulate gene expression to overcome abiotic and biotic stresses [176, 177]. The plant microbiome positively alters the production of primary and secondary metabolites in plants during water stress conditions [47]. Endophytic microbes can synthesize different “protective” compounds like antioxidants, carbohydrates, and proline to cope with abiotic stress conditions [178]. In addition to those mechanisms detailed above, microbial endophytes also seem to confer stress tolerance by induction and regulation of stress-responsive genes, production of anti-stress metabolites as well as generation of ROS scavengers [76]. Enhanced accumulation of osmoprotectants proline, glutamine, and leucine under drought stress has been shown to impart stress tolerance [179]. Interestingly, Etesami et al. [180] reported that superoxide dismutase, catalase, and peroxidase enzymes showed greater activity in plants inoculated with beneficial microorganisms (i.e., Bacillus cereus, B. subtilis, and Serratia sp.). These enzymes are highly related to decreasing the negative effects of drought; thus, these bacterial species could be used as agents for mitigating this type of abiotic stress in plants, improving their development. Sadeghi et al. [181] found that the fungal endophyte (Penicillium citrinum, Aureobasidium pullulans, and Dothideomycetes sp.) inoculation enhances drought tolerance and reduces ROS accumulation in mandarin (Citrus reticulata) by increasing their scavenging via improving the redox state of glutathione and ascorbate, and promotion of antioxidant enzymes activity such as superoxide dismutase (SOD), glutathione reductase (GR), and, ascorbate peroxidase (APX). The study conducted by Martins et al. [166] using the three bacterial strains Bacillus cereus, B. subtilis, and B. thuringiensis, for mitigating the effect of drought stress in soybean and found a decrease in quantum efficiency of PSII (Fv/Fm) in control plants as compared to plants inoculated with bacterial strains. Another study conducted by Maxton et al. [171] used ACC deaminase-producing strains (Bulkhorderia cepacia, and Citrobacter feurendii) to mitigate the adverse effect of drought and salinity stress on Capsicum annuum. Melatonin accumulation, when inoculated with Bacillus amyloliquefaciens counter-balancing the adverse effects of drought, salinity, and cold stress [182]. This bacterial strain also reduces the transcription of tryptophan decarboxylase and serotonin. However, melatonin-producing endophytic microbes may have long-term effects on the endogenous melatonin levels in plants once they enter inside plant tissues.
Among the other mechanisms, endophytes have been shown to influence
osmoregulation of tissues, regulation of stomatal conductance, and maintenance of
cell-wall elasticity leading to drought tolerance [4]. In a study, arbuscular
mycorrhizal fungi (AMF) colonize both C
Similarly, Pandey et al. [186] showed significant alleviation of drought stress tolerance in rice by the endophyte Trichoderma harzianum, which significantly modulated proline levels, SOD, and upregulation of aquaporin, dehydrin, and malondialdehyde genes. A transcriptome analysis of B. phytofirmans PsJN colonizing potato in response to drought stress revealed the upregulation of transcripts related to transcriptional regulation, cellular homeostasis, and ROS detoxification [187]. The integration of microbiota to agricultural practices represents a promising sustainable alternative to address the food security issue under abiotic stress conditions. However, the use of these microorganisms requires several issues addressed in the field, such as agricultural practices, microbial establishment, colonization of soil, and plant by microbial inoculants, and biosynthesis of bioactive metabolites produced by microbes involved in the regulation of plant growth, under specific conditions of soil, climate, and plant genotypes.
Droughts in coming time are likely to be more enduring, common, and severe than they have been in recent times, hence some novel and quickly deployable solutions for improving and convalescing drought tolerance in crops are urgently required. Terrestrial organisms, including plants, must be potent enough to adapt to the dry conditions resulting from diurnal and seasonal water fluctuations [188, 189]. Although adapted to resist, tolerate and evade the drying environments, plants tend to modify the root architecture and growth, alter their physiology, as well as shut down their stomata on their aboveground segments [190]. Recent studies have demonstrated that the interaction of plants with their above and below ground microbial partners plays an important role in augmenting plant’s ability to thrive and endure under dry conditions [191, 192] and that drought significantly influences these interactions, thus altering both the structure and function of the root microbiome [193, 194]. Therefore, it can be predicted that an enhanced understanding of the complex mechanism and feedback between the plants and their microbial partners during and after drought will pave the way for harnessing the rhizosphere microbiome to augment the resilience of crop production to drought. Several studies conducted in diverse plant species have revealed that during the drought, the plant root microbiome shifts in favor of some particular microbial taxa and supplant the majority of other microbial lineages residing there in the root [194, 195, 196, 197].
This shift in microbial communities is mainly governed by the abundance of genes involved in the degradation of complex plant polysaccharides, thus suggesting the proliferation of oligotrophic bacteria [155]. One possible reason for the proliferation of Gram-positive bacteria is that the bacteria of this group are metabolically more active and hardy enough to degrade compounds present in the drought environment [155]. Moreover, studies have also revealed that this enrichment is proportional to the duration and strength of the drought and is believed to disappear once water returns to the root system rapidly [195]. Recent advancements in omics-biology such as metagenomics, proteomics, and metabolomics allow us to binning of genomes, proteomes, and metabolomes from diverse environments to better understand the dynamics, structure, and functional including secondary metabolite biosynthesis and genes in soil microbial communities [198, 199].
Such studies will help to identify different genes, carbohydrate and secondary metabolites, transport, and metabolism pathways associated with bacterial enrichment under drought conditions. Additionally, the obtained datasets will help identify the genetic traits associated with these bacterial communities. A series of recently conducted studies revealed the role of multi-omics approaches in understanding the microbial traits in drought conditions. For example, while using metagenome-guided comparative genomics to under the dynamics of iron metabolism in drought-induced rhizosphere microbiome, concluded a linkage between drought, iron metabolism, and the plant root microbiome [200]. Similarly, Michaletti et al. [201], while studying the proteomics and metabolomics of spring-wheat leaf tissues, provided a detailed account of some of the key metabolites that can be used to develop ameliorated models to establish linkage between yield-associated traits and several metabolic pathways. In another study, Chmielewska et al. [202], while studying the proteomic and metabolomic changes in leaves and roots of two barley genotypes of different origins, indicated molecular chaperones’ activity as well as osmoprotectants, elevated the resistance of these genotypes under drought stress. Gundaraniya et al. [203] studied the metabolomic alterations of drought-tolerant and susceptible Peanut (Arachis hypogaea L.) revealed that the two peanut genotypes possess different sensitivity to drought with accumulation and regulation of some metabolites, including organic acids, sugars, sugar alcohols, and fatty acids. The study conducted by Das et al. [204] studied metabolic alteration in different vegetative tissues of soybean under drought and heat stress conditions. This study revealed metabolites that are needed for various cellular processes, like the tricarboxylic acid (TCA) cycle, glycolysis, starch biosynthesis, and pentose phosphate pathway for regulating amino acid metabolism, peptide metabolism, carbohydrate metabolism, biosynthesis of purine and pyrimidine, were affected under heat and drought stress condition [204]. Another study conducted on soybean by Cao et al. [205] studied the metabolomic and transcriptomic profiling of soybean treated with melatonin under drought stress conditions during the grain-filling period through secondary metabolite biosynthesis pathways regulation.
It is challenging to understand the mechanisms involved during damage caused due to drought stress for the development of drought stress plant varieties. Therefore, the applications of transgenic or genome editing approaches will help to introduce desired drought stress-resistant character into different crop varieties. CRISPR has emerged as an innovative tool for genome editing in plants with desired characters and improves crop production under abiotic stress conditions (Fig. 3). Recently the application of CRISPR/Cas9 has been reported in different crops: Bread wheat [206, 207], potato [208], and model plants like Arabidopsis thaliana [209] and rice [210]. Overexpression of different transcription factors (TFs) and genes associated with drought stress signalling helps to accumulate different metabolites, signalling molecules, and osmolytes, enhancing drought stress tolerance in plants.
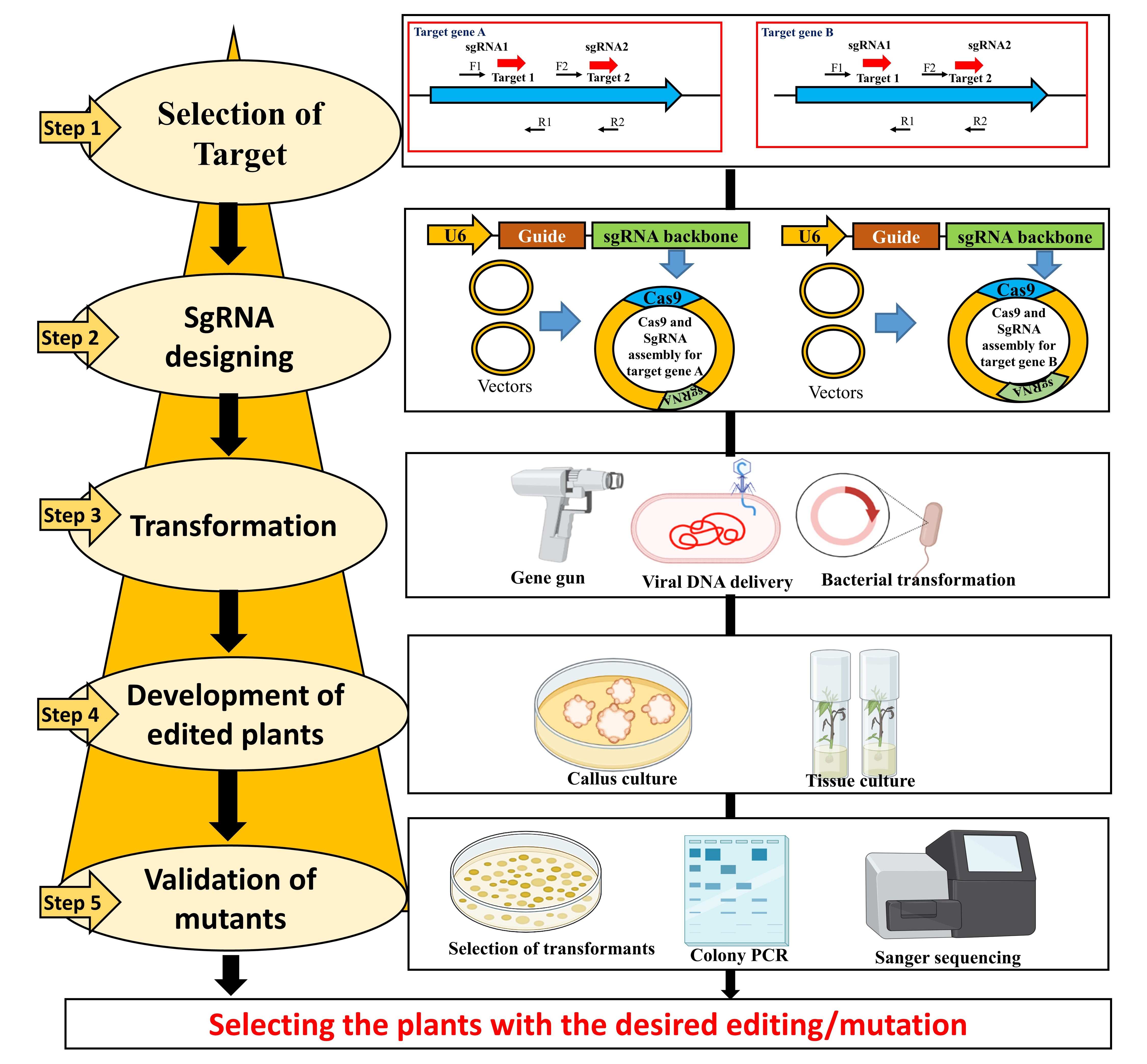 Fig. 3.
Fig. 3.Diagrammatic representation of Cas9/sgRNA genome editing (GE) approach. Sequential steps: (1) selection of target gene; (2) designing of sgRNAs using online tools; (3) Agrobacterium-mediated transformation into target plant species; (4) selection of transformants for the presence of Cas9 and sgRNA; (5) finally, selecting the plants with the desired editing/mutation by using, colony PCR and DNA sequencing.
On the other hand, sensitive (S) gene expression increases drought situations in
plants by hormonal imbalance, inducing ROS production, and reducing antioxidant
activity. For example, the study conducted by Fang et al. [210] reported
Oryza sativa stress-related ring finger protein 1 (OsSRFP1) present in
rice works as a negative regulator of drought stress, and its silencing often
results in enhanced drought stress tolerance by decreasing H
Ethylene plays a fundamental role in regulating water scarcity conditions, therefore, the silencing 1-aminocyclopropane-1-carboxylic acid synthase 6 (ACS6), an ethylene biosynthetic gene present in maize, has resulted in high yield under drought stress conditions [212]. Another study conducted by Shi et al. [213] reported that the ARGOS family genes, i.e., AUXIN REGULATED GENE INVOLVED IN ORGAN SIZE 8 (ARGOS8), negatively regulates the ethylene signalling pathway and confers enhanced yield and drought stress tolerance under drought conditions. However, the endogenous expression of ARGOS8 transcript is spatially fluctuating and comparatively low in maize. Comprehensive molecular analysis has shown that ABA act as a primary factor of drought response in plants by regulating the expression of stress-related genes and controlling stomatal closure to prevent water loss [214]. Overall, these genome editing approaches will deliver new opportunities for creating nucleotide-specific alterations and might be widely explored in the future for improving drought tolerance in crops and thus will be a powerful tool for maintaining global food security.
Different approaches like PGPR and endophytes inoculation, seed priming with exogenous compounds, synthetic growth regulators, and genetic engineering are required to counter the negative impact of drought stress on crop plants. The emergence of an omics approach including genomics, metabolomics, proteomics, and transcriptomics might improve our understanding of the essential drought-tolerant genes and untangling the complex signalling cascades and gene networks involved in drought tolerance in different plants. Notably, ground-breaking methods, like CRISPR/Cas9 genome editing tools, will provide a deeper understanding and help to develop drought-resilient crops to minimize the risk of global food insecurity. Understanding of the processes through which soil microorganisms impact plant drought tolerance and recovery, as well as their relevance and application in field settings, holds a lot of promise for making crop production systems more resilient to drought. Implementing novel methods combining physiological, molecular, and genetic methods will deliver an effective solution for combating stress-related problems in the near future.
AD and AK conceived, designed, and edited the article; MM, KC, GS, GR, H, ZSS, EM, SS, and JFD contributed to different sections of the article.
Not applicable.
Anamika Dubey would like to acknowledge the DST, New Delhi, India, for providing financial support through Inspire Ph.D. Fellowship (IF160797). Prof. Dames and Dr. Kumar acknowledges the research support provided by Rhodes University.
This research received no external funding.
The authors declare no conflict of interest.



