1 OpenLab Gene and Cell Technologies, Institute of Fundamental Medicine and Biology, Kazan Federal University, 420008 Kazan, Russia
2 Division of Medical and Biological Sciences, Tatarstan Academy of Sciences, 420111 Kazan, Russia
3 Department of Histology, Cytology and Embryology, Kazan State Medical University, 420012 Kazan, Russia
Abstract
Adeno-associated viruses (AAVs) are established vectors for efficient gene delivery to the central nervous system (CNS). Increasingly, strategies aim to restrict transduction to specific neuronal subtypes defined by the associated functional properties, thereby enhancing precision and therapeutic potential.
Recombinant AAV9 vectors carrying fluorescent reporters under the control of cytomegalovirus (CMV), human synapsin (hSyn), or homeobox 9 (Hb9) promoters were delivered intrathecally in Wistar rats. Transgene expression was evaluated 7 days post-injection by confocal microscopy. Neurons in laminae VII–X were quantified across cervical, thoracic, lumbar, and sacral spinal cord levels. Statistical analysis was performed using the Kruskal–Wallis test followed by Mann–Whitney U tests with Bonferroni correction.
In lamina VII, consistent neuronal expression was mediated by hSyn across all spinal levels, with significantly higher transduction at cervical compared to thoracic and lumbar regions (p < 0.01). CMV and Hb9 showed no detectable tropism for this lamina. In lamina VIII, CMV drove markedly higher expression than hSyn and Hb9, with a 2.8-fold difference at the lumbar level (p < 0.001). In lamina X, CMV expression exceeded hSyn at the lumbar and sacral levels (p < 0.05), while Hb9 showed no activity. In lamina IX, all promoters mediated motoneuron transduction, but only Hb9 restricted expression specifically to motoneuron somata. Notably, CMV induced off-target expression in glial cells.
AAV9-mediated expression patterns in the spinal cord are strongly shaped by promoter choice and segmental level. Hb9 provides high motoneuron specificity, hSyn supports broad neuronal activation across laminae VII–X, whereas CMV drives robust but non-specific expression with significant off-target activity. These findings highlight the importance of rational promoter selection for spinal cord gene therapy and strategies aimed at functional recovery in motor system disorders.
Keywords
- adeno-associated virus
- spinal cord
- motor neurons
- gene therapy
- promoter regions
- intrathecal injections
Adeno-associated viruses (AAV) continue to be considered powerful tools in the field of neurobiology and gene therapy due to their ability to efficiently transfer genetic material into cells, including neurons [1, 2]. According to known studies, the pattern of AAV biodistribution has significant differences due to a number of reasons, including the animal model used, the serotype of the vector, the method used to assess biodistribution, the method of administration of the gene product, determining different results and difficulties in translating the described preclinical results [3, 4, 5, 6, 7]. A review of the literature over the last decade on AAV-based gene therapies targeting the central nervous system (CNS) highlights the limited knowledge available on the biodistribution of AAVs within the spinal cord grey matter laminae and the need to continue these studies [7, 8, 9, 10]. Cell-selective gene expression is a critical element of many AAV-based vector gene therapies. Given the frequent use of highly transcriptionally active promoters as part of genetic constructs that define potent gene expression in neural tissue, neuron-specific promoters are largely more successful at minimising off-target transgene expression [11, 12].
Among delivery strategies, intrathecal administration has emerged as a particularly promising approach, as introduction into cerebrospinal fluid enables broad dissemination of AAV across spinal segments while minimizing parenchymal injury [13, 14]. In preclinical models, this route has achieved robust gene transfer throughout the spinal cord and dorsal root ganglia [15], and has been applied for widespread therapeutic delivery in disorders such as spinal muscular atrophy and hereditary spastic paraplegia [16].
Despite this translational potential, little is known about how intrathecal delivery shapes viral biodistribution across spinal segments and within defined gray matter laminae, particularly with respect to the interaction of neuron-selective promoters with distinct neuronal populations. In particular, it is known that transgene expression under the human synapsin (hSyn) promoter was concentrated in neurons located within laminae IV–VIII and X, with some expression in lamina IX, while the homeobox 9 (Hb9) promoter successfully restricted transgene expression almost exclusively to motor neurons in lamina IX when considering only the lumbar spinal cord [9, 17]. In contrast, quantification of transduced cells in each region of the three spinal cord sections showed that cytomegalovirus (CMV) promoter-driven expression resulted in significantly higher relative numbers of green fluorescent protein (GFP)-positive cells in the cervical spinal cord, but not in the lumbar spinal cord, compared to hSyn, without assessing transduced neurons in grey matter laminae [18, 19]. To further understand the transcriptional activity of the described promoters, it is important to assess the biodistribution of the transgene in grey matter laminae in all parts of the spinal cord, achieved by introducing AAVs with different promoter regions, and to determine whether specific cell types of interest to the researcher will be transduced.
In this study, we focus on comparative analyses of the biodistribution of AAV
depending on the type of promoter used, such as the CMV, hSyn, and Hb9 promoters.
These promoters were chosen because of their specificity to neuronal cells in
different grey matter laminae of the spinal cord. Understanding how promoters
affect gene expression in specific neuronal populations may provide important
data for the development of targeted gene therapy and neuroimaging techniques.
For example, for a number of neurodegenerative diseases, gene therapy involves
transduction of specific populations of spinal cord neurons [1], such as target
gene therapy for motor neurons located in laminae VIII and IX (ventral horn) of
the grey matter of the spinal cord in severe spinal muscular atrophy (SMA),
targeted therapy for the loss of lumbar interneurons between laminae IV and VII
grey matter of the spinal cord and
The purpose of this study is to assess the biodistribution of AAV vectors in the central nervous system, with a focus on the spinal cord and neuronal transduction of different grey matter laminae. We analysed the activity of the CMV, hSyn, and Hb9 promoters on the specificity of fluorescent protein expression in neurons, allowing us to determine their efficacy and targeting for potentialtherapeutic applications.
The plasmids pAAV-Hb9-LMO3 (sbGLuc-VChR1-eYFP) and pAAV-hSyn-mCherry were synthesized by Addgene, Watertown, MA, USA (Addgene plasmid: 114103, catalog #114472) according to a previously reported specification [17]. A schematic representation of the developed constructs is shown in Fig. 1A. Luminopsin (LMO3) is a fusion protein of Gaussia luciferase variant (sbGLuc), Volvox canalorhodopsin-1 (VChR1) and enhanced yellow fluorescent protein (eYFP) under the regulation of the Homeobox 9 (Hb9) promoter. The plasmid pAAV-CMV-eGFP was provided by Marlin Biotech, Moscow, Russia.
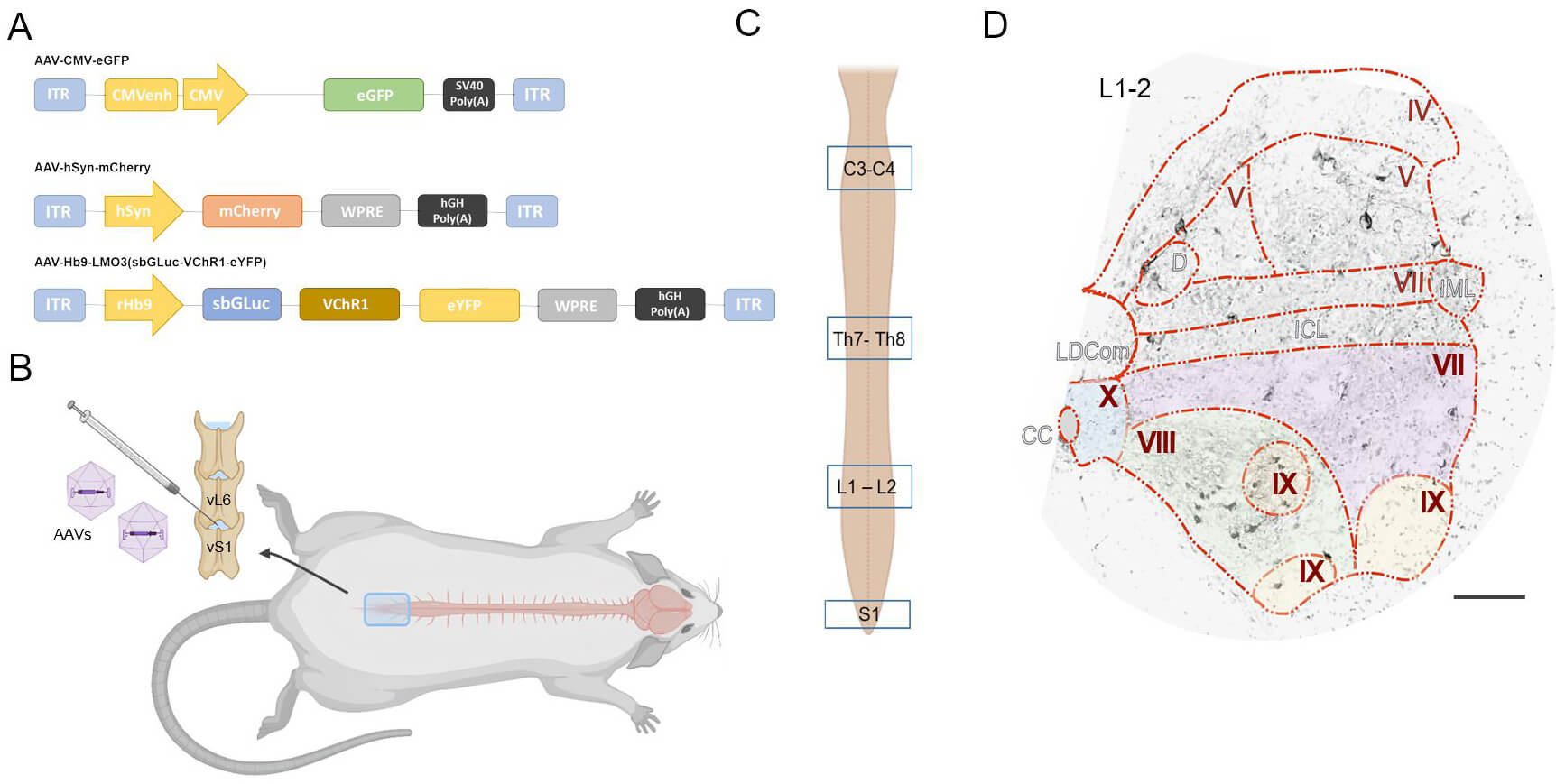 Fig. 1.
Fig. 1.
Experimental design and targeting strategy for AAV-mediated gene
delivery to the spinal cord. (A) Schematic representation
of the main components of the gene cassette packaged into the recombinant AAV
vector. (B) Intrathecal injection of gene constructs (6
To produce plasmid DNA of pAAV-Hb9-LMO3 (sbGLuc-VChR1-eYFP), pAAV-hSyn-mCherry,
pAAV-CMV-eGFP in preparative quantities, the Escherichia coli strain
NEB® Stable (Thermo Fisher Scientific Inc., Waltham, MA, USA) was
transformed. For this purpose, bacterial cells were cultured in Luria-Bertani
(LS-LB) medium without addition of antibiotics at 37 °C and vigorous
shaking (200–300 rpm) until OD600 = 0.30–0.35. In this work, low-salt
Luria-Bertani LS-LB medium was used for cultivation of E. coli
NEB® Stable cells (per 1 L of deionized water): salt-free yeast
extract (Sigma-Aldrich, St. Louis, MI, USA USA)—5 g, tryptone (Sigma-Aldrich,
USA)—10 g, 5 M NaCl (Sigma-Aldrich, USA)—5 g, pH 7.5. Agarized LS-LB medium
was also used (per 1 L of deionized water): salt-free yeast extract—5 g,
tryptone—10 g, 5 M NaCl—5 g, agar (Sigma-Aldrich, USA)—15 g. Sterilization
of nutrient media was carried out in autoclave in liquid or agar mode. Competent
cells were prepared using CaCl2 method. Plasmid DNA from bacterial cells was
isolated by alkaline lysis using a commercial GeneJET Plasmid MiniprepKit (Thermo
Fisher Scientific Inc., USA) according to the methodology recommended by the
manufacturer. For this purpose, a single bacterial colony of recombinant
Escherichia coli NEB® Stable strain containing the required
plasmid DNA was seeded in a flask with 2–5 mL of LS-LB medium in the appropriate
selective antibiotic. The culture was incubated in a shaker (300 rpm) at 37
°C for 8 hours. Next, the initial bacterial culture was diluted from
1:500 in 50 mL of LB medium. After seeding, the culture was incubated in a shaker
(300 rpm) at 37 °C for 12–16 hours. The culture should reach a cell
density of approximately 3–4
SH-SY5Y cells were cultured in DMEM/F-12 medium (PanEco, Moscow, Russia)
supplemented with 10% fetal bovine serum (Bioserum, South America), 2 mM
L-glutamine (PanEco, Russia) and 1
Human embryonic kidney 293 cells (HEK293T; ATCC CRL-11268, American Type Culture Collection, Manassas, VA, USA) were used in this study. The cells were cultured in high-glucose DMEM (Gibco, Waltham, MA, USA) supplemented with 10% (v/v) fetal bovine serum (FBS; Biosera Europe, France) and Penicillin-Streptomycin (Pen-Strep). All cell lines were maintained at 37 °C in a humidified atmosphere of 5% CO2 and 95% relative humidity, and were confirmed to be free of mycoplasma contamination by PCR testing. Cell line authentication was performed by the American Type Culture Collection using short tandem repeat (STR) profiling.
Primary motor neurons were isolated from neonatal Wistar rats at postnatal day 3
(P3). Animals were euthanized by decapitation (a detailed description of the
procedure is provided in Section 2.9), after which the spinal cord was extracted
and transferred into sterile physiological saline supplemented with antibiotics.
Tissue was then incubated in 0.25% trypsin solution (PanEco, Russia) containing
antibiotics at 37 °C for 20 minutes with gentle trituration every 10
minutes. Following enzymatic digestion, the suspension was centrifuged at 2000
rpm for 10 minutes. The supernatant was discarded, and the pellet was resuspended
in calcium- and magnesium-free Dulbecco’s phosphate-buffered saline (DPBS, 450
mL), followed by a second centrifugation under the same conditions. The DPBS wash
was repeated twice. After the final centrifugation, cells were resuspended in
DMEM/F12 medium without glutamine (PanEco) supplemented with B27 (ncB27
Supplement, serum-free, Shownin, Hefei, China) and EGF (1:4464, Cloud-Clone, catalog #APA560Hu02, USA). Cells were seeded into 6-well plates pre-coated 24 hours in
advance with human fibronectin (IMTEK, Moscow, Russia). An initial small volume
of the cell suspension was added to allow attachment; after 10–15 minutes of
incubation at 37 °C, the final volume of culture medium was added. These
preparations were established as de novo primary motor neuron cultures.
Cultures were maintained under standard conditions (37 °C, 5% CO2,
95% relative humidity) and monitored daily. Medium replacement was performed as
needed but not earlier than day 3; only 50% of the medium volume was replaced
during each change. Culture was confirmed to be free of mycoplasma
contamination by PCR testing. On day 7 of culture, cells were transduced with
recombinant AAV9 vectors (AAV9-Hb9-LMO3 or AAV9-hSyn-mCherry) at a multiplicity
of infection (MOI) of 50,000. Transgene expression was evaluated 48 hours
post-transduction. For immunofluorescence analysis, cells were fixed with 4%
paraformaldehyde in PBS, permeabilized with 0.1% Triton X-100 in PBS, and
stained with DAPI (10 mg/mL, Sigma-Aldrich) to visualize nuclei. Confocal imaging
was performed using an LSM 780 microscope (Carl Zeiss, Germany). To verify the
cellular identity within the cultures, immunofluorescent staining was performed
using antibodies against neurons (NeuN, catalog #DF614, Affinity, Shanghai, China)/
Recombinant viral vectors AAV9-Hb9-LMO3, AAV9-hSyn-mCherry, AAV9-eGFP were
produced in HEK293T cells by the triple transient transfection method on a total
area of 7500 cm2. Viral preparations were purified from cellular debris,
impurity proteins, and empty viral capsids according to the methodology
previously developed in the laboratory [22, 23, 24]. Viral vector preparations were
sterilised by filtration using a syringe filter with a pore diameter of 0.22
µm, then frozen in viral preparation storage buffer:
1
The study was conducted on 16 female Wistar rats (weight: 200–250 g, 3–4
months; source - KrolInfo LLC, Moscow region, Orekhovo-Zuyevsky city district,
Russia). The animals were kept in standard conditions (23 °C
Prior to the intrathecal injection procedure, the animals were anesthetized with
a combination of Zoletil (Virbac Laboratories, Inc., 100 mg/mL, Karros, France)
and Xylanite (Nita-Farm, Saratov, Russia). The anesthetic mixture was
administered via intramuscular injection at a dose of 40 mg/kg for Zoletil and 10
mg/kg for Xylanite. All rats had the abdomen and lumbosacral area shaved at the
start of the injection. The shaved areas were cleaned with a diluted solution of
the alcohol-based antiseptic «Chisteya Plus» (LLC
«VITA-PUL», Moscow, Russia). Rats were positioned
in sternal recumbency with their pelvic limbs brought under the abdomen as
cranially as possible in order to arch the lumbosacral area. The intrathecal
injection was performed using a Hamilton syringe (75N 20 µL SYR
25G). The intrathecal injection of gene constructs was administered in a 20
µL, 6
Euthanasia was performed by intramuscular administration of Zoletil (40 mg/kg) and Xylanite (10 mg/kg), followed by transcardial perfusion with cold 0.01 M phosphate-buffered saline (PBS; pH 7.4), followed by 4% paraformaldehyde (PFA) in PBS. For neonatal rat pups (P3), anesthesia was induced with the same drug combination at equivalent doses prior to decapitation.
For histological analysis, spinal cords were extracted and post-fixed in 4% paraformaldehyde in PBS. Tissues were cryoprotected by sequential immersion in 15% and 30% sucrose solutions. After embedding in Tissue-Tek O.C.T. Compound, 20 µm transverse sections were cut using a RWD Minux® FS800A. For each animal, five slides from each analyzed spinal cord segment (cervical C3–4, thoracic Th8–9, lumbar L1–2, and sacral S1), each containing five sections, were selected for analysis. Segment choice was based on their functional relevance: C3–4 for forelimb control, Th8–9 for trunk coordination, L1–2 for the locomotor rhythmogenic kernel, and S1 for autonomic and urinary control [25, 26, 27, 28]. Sections were rinsed in PBS, counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich), and mounted with medium for confocal microscopy.
Spatial analysis of neurons expressing reporter proteins was conducted in the
gray matter of the spinal cord at the level of the studied segments Fig. 1C. Detailed imaging was carried out using an LSM 780 confocal
microscope (Carl Zeiss, Germany). Visualization was performed using confocal
microscopy with a 20
Spatial analysis of neurons expressing reporter proteins was conducted in the
gray matter of the spinal cord at the level of the studied segments.
Visualization was performed using confocal microscopy with a 20
Statistical analyses were performed using Origin 10.0 SR0 (OriginLab
Corporation, Northampton, MA, USA). Differences among groups were first assessed
using the Kruskal–Wallis test, and significant results were further examined by
pairwise Mann–Whitney U tests with Bonferroni correction. Significance levels
were set at *p
To confirm the integrity and correct structure of the developed plasmid constructs, restriction digestion was performed. Analysis of pAAV-CMV-eGFP, pAAV-hSyn-mCherry, and pAAV-Hb9-LMO3-eYFP revealed restriction fragments of expected sizes, consistent with the theoretical plasmid maps (Fig. 2A). These results confirm the successful cloning and structural accuracy of the designed vectors.
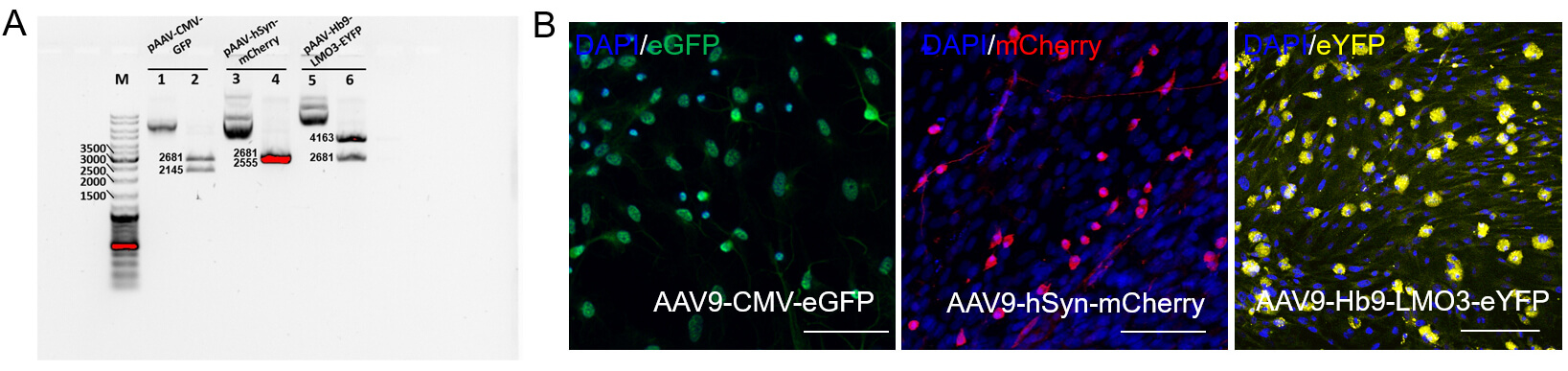 Fig. 2.
Fig. 2.
Analysis of plasmid DNA integrity and transgene expression in primary motor neuron cultures. (A) Restriction analysis of plasmid DNA and electrophoresis in 0.8% agarose gel. Lanes 1, 3, 5—circular plasmid DNA; lanes 2, 4, 6—plasmid DNA after restriction digestion with SmaI. (B) Transgene expression analysis 48 hours after transduction of primary motor neuron cultures with AAV9-CMV-eGFP, AAV9-hSyn-mCherry or AAV-Hb9-LMO3-eYFP. Scale bar: 100 µm.
To evaluate the functionality of the constructs, primary motor neuron cultures were transduced with AAV9-CMV-eGFP, AAV9-hSyn-mCherry or AAV9-Hb9-LMO3-eYFP vectors. Confocal imaging revealed expression of eGFP, mCherry or eYFP in transduced cells, confirming that both constructs drive effective transgene expression under control of the respective promoters (Fig. 2B). Notably, fluorescence was localized to neuronal pericarion, suggesting successful transduction of motor neuron populations in vitro.
On day 7 after intrathecal administration of recombinant AAV9 vectors, the number of transduced neurons was analyzed in lamina VII at four spinal cord levels: cervical (C3–4), thoracic (Th8–9), lumbar (L1–2), and sacral (S1). No expression of reporter proteins eGFP or eYFP was detected at lamina VII in the AAV9-CMV-eGFP and AAV9-Hb9-LMO3-eYFP groups at any of the examined levels, indicating a lack of tropism of these constructs for neurons in this lamina. In contrast, the AAV9-hSyn-mCherry group exhibited consistent mCherry expression in neurons at all analyzed levels, with notable segmental variability (Fig. 3A,3A’).
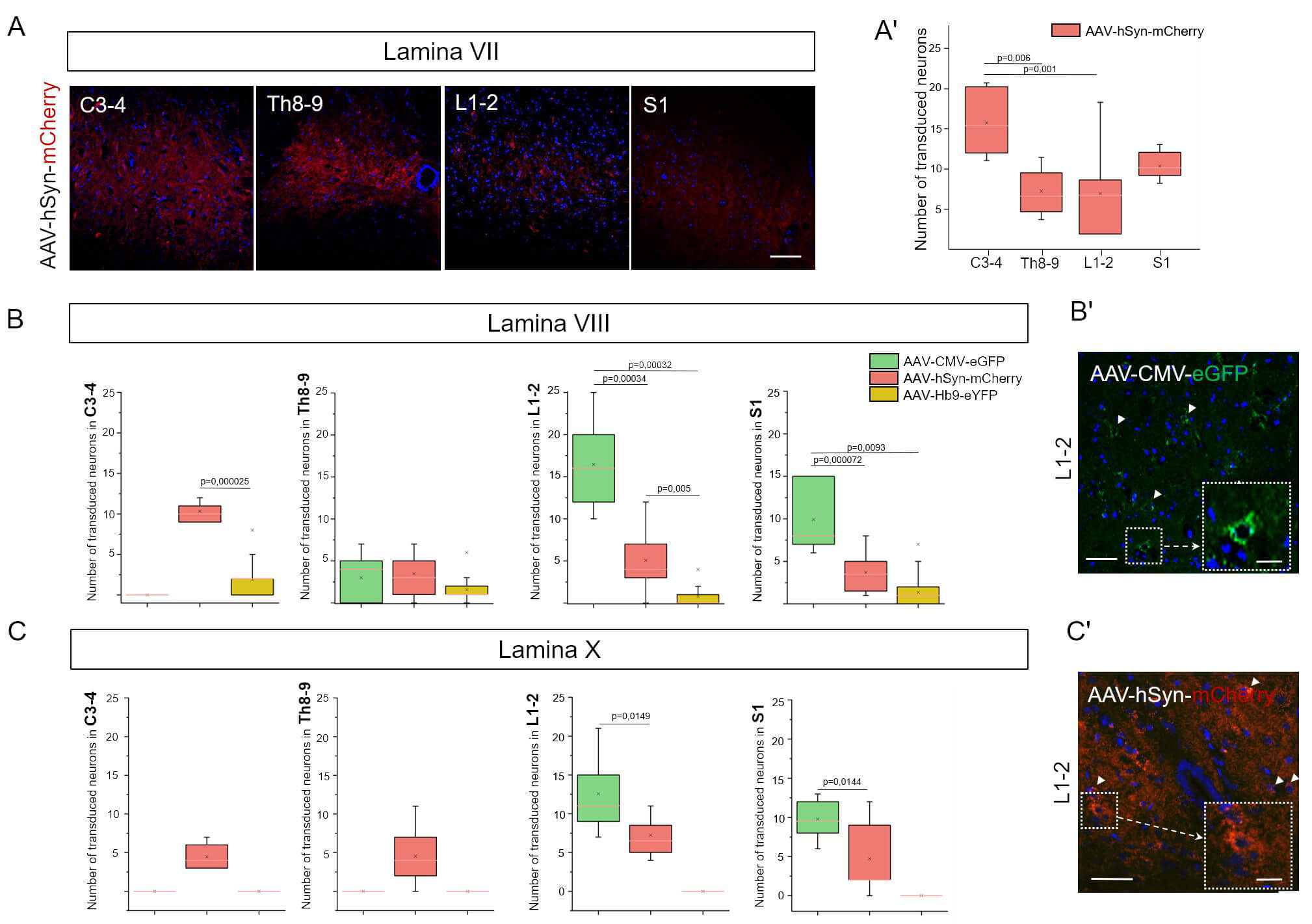 Fig. 3.
Fig. 3.
Selective transduction patterns of AAV vectors in distinct spinal cord lamina. (A) Confocal images of spinal cord sections at the L1–L2 level for experimental groups. Scale bar: 60 µm. (A’) Transduction of neurons in lamina VII was observed following intrathecal injection of AAV-hSyn-mCherry, whereas AAV-CMV-eGFP and AAV-Hb9-LMO3-eYFP showed no tropism for these neurons. (B) Number of transduced neurons in lamina VIII (C) and X at C3–4, Th8–9, L1–L2, and S1 spinal cord levels. Statistical analysis was performed using the Mann–Whitney U test with Bonferroni correction. (B’) Confocal imaging of spinal cord lamina VIII (C’) and X at the L1–L2 level in experimental groups. White arrowheads indicate transduced neurons. Insets with dashed borders show enlarged views of selected regions. Scale bar: 60 µm; inset scale bar: 15 µm.
The highest mean number of transduced neurons was observed at C3–4 (15.88
Transduction of neurons in lamina VIII was analyzed in the C3–4, Th8–9, L1–2, and S1 spinal cord segments on day 7 after intrathecal administration of recombinant AAV9 vectors (Fig. 3B,3B’). In the AAV9-Hb9-eYFP group, eYFP expression in neurons was minimal (on average, fewer than one cell per section), consistent with the motoneuron specificity of the Hb9 promoter and the absence of motoneurons in this region.
In the groups using a strong ubiquitous promoter (CMV) and a neuron-specific
promoter (hSyn), a moderate number of transduced cells were observed. At the
L1–2 level, the mean number of eGFP-positive neurons in the AAV9-CMV-eGFP group
was 16.46
At the C3–4 level, a significant difference was found only between the AAV9-hSyn-mCherry and AAV9-Hb9-eYFP groups (p = 0.0000025), with markedly higher expression observed using the hSyn promoter. No statistically significant differences were detected between groups at the Th8–9 level.
Neuronal transduction in lamina X was analyzed in spinal cord segments C3–4,
Th8–9, L1–2, and S1 on day 7 after intrathecal administration of recombinant
AAV9 vectors (Fig. 3C,3C’). In the AAV9-CMV-eGFP group, eGFP expression in lamina
X neurons was observed only at the L1–2 and S1 levels, where the mean number of
transduced cells was 12.73
In the AAV9-Hb9-eYFP group, no eYFP expression was detected in lamina X neurons at any spinal level, which is consistent with the motoneuron-specific activity of the Hb9 promoter and the absence of such cells in this region.
Transduction of neurons in lamina IX was evaluated at the spinal cord levels
C3–4, Th8–9, L1–2, and S1 on day 7 after intrathecal injection of recombinant
AAV9 vectors (Fig. 4A,4A’). All three groups demonstrated consistent reporter
expression in accordance with the expected localization of motoneurons in this
lamina. The average number of transduced neurons ranged from 3.58
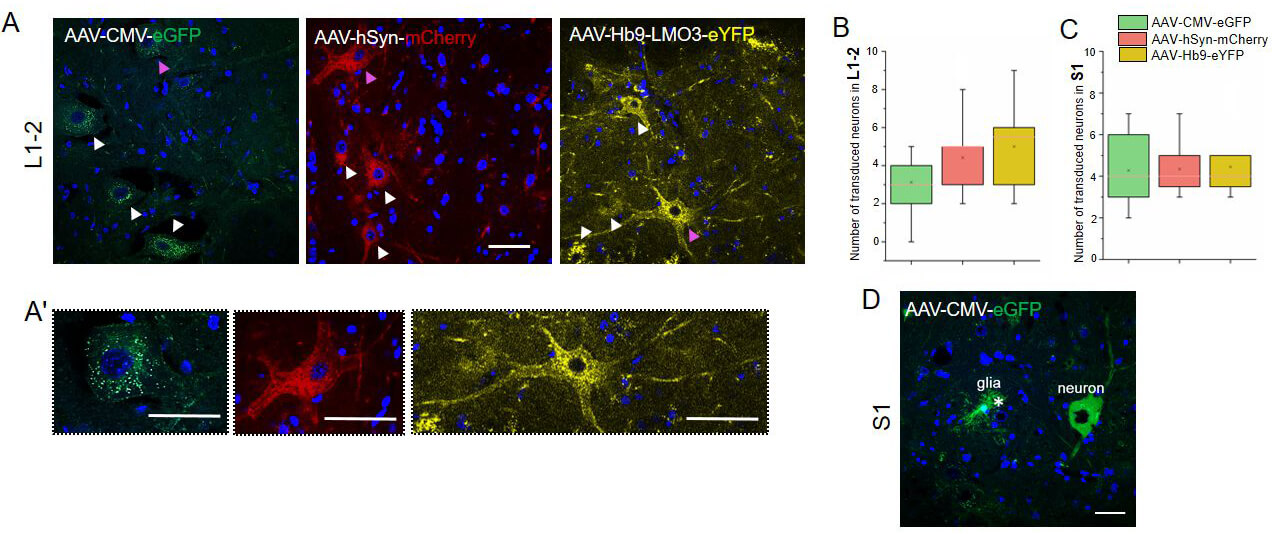 Fig. 4.
Fig. 4.
Analysis of transduction specificity and distribution of AAV vectors in lamina IX of spinal cord. (A) Confocal images of spinal cord sections at the L1–L2 level showing lamina IX in experimental groups. (A’) Transduced neurons are indicated by arrowheads. Higher-magnification images show motoneurons, highlighting differences in pericarion and process transduction among constructs with different promoters. (B) Number of transduced neurons in lamina IX of the L1–L2 and (C) S1 spinal cord segments on day 7 after intrathecal injection of AAV constructs. Statistical analysis was performed using the Mann–Whitney U test with Bonferroni correction. (D) In the AAV-CMV-eGFP group, reporter expression was observed in both glial cells (asterisk) and neurons within lamina IX of the S1 segment. (A,A’,D) Scale bars: 50 µm. In Fig. 4A, the pink arrows indicate the neurons, which are represented on an enlarged scale in Fig. 4A’.
At the L1–2 level, eGFP expression in the AAV9-CMV-eGFP group (5.92
The pattern of eYFP expression under the control of the Hb9 promoter displayed distinct characteristics compared to the other vectors: the fluorescent signal was predominantly confined to motoneuron somata, with minimal labeling of neuronal processes, in contrast to the more diffuse labeling observed in the groups with CMV and hSyn promoters.
Intrathecal administration of AAV9 has proven to be a promising minimally invasive method for delivering genetic constructs to the spinal cord. This approach ensures broad vector distribution in the cerebrospinal fluid, minimizing tissue damage and increasing accessibility to lumbar enlargement neurons—a key region for restoring motor function after injury [31, 32]. Unlike systemic administration, which requires overcoming the blood–brain barrier [33], intrathecal injection directly targets spinal cord networks, making it particularly important in chronic injuries when the reorganization of descending pathways (cortico–reticulo–spinal) is critical for recovery [1]. Our results are consistent with studies demonstrating that AAV9 ensures long‑term expression in lumbar motoneurons [32, 34, 35], confirming its clinical relevance. The unique ability of AAV9 to efficiently transduce the spinal cord distinguishes it from less permeable serotypes such as AAV2 or AAV5 [34]. Our results indicate that intrathecal delivery provides an efficient means of distributing AAV9 in all segments of the spinal cord.
We observed markedly reduced transduction in cervical segments C3–4 especially under CMV and Hb9 promoters. This finding is consistent with comparative neuroanatomy, as lumbar segments contain a higher density and greater molecular diversity of motor-related interneurons such as V1 interneurons, while thoracic and cervical levels include fewer of these rhythmogenic populations [36]. The thoracic segments Th8–9 are characterized by a lower density of motor-related interneurons than lumbar enlargement, which likely reduces the number of available cellular targets for promoter-driven expression and is consistent with the modest transduction observed in these regions [37]. In addition, intrathecal AAV9 delivery into caudal spinal segments produces a rostrocaudal gradient, with vector distribution diminishing rostrally and leading to lower titers in upper cervical regions [13].
The laminar patterns we observed in VII–X align with the reported positioning of locomotor CPG candidates: ventromedial interneurons in L2 (including commissural and V-class subsets) that project caudally toward L4–L5 motoneuron pools, and Hb9-positive excitatory neurons contributing to the rhythmogenic kernel [27]. This organization provides a plausible substrate for the promoter-dependent distributions detected here. In the sacral segment S1, transduction levels of all three vectors were comparable to those at lumbar L1–2. This corresponds with the functional importance of the sacral cord, where rhythmogenic circuits include the sacral micturition center localized to S1–S4 and concentrated at S1 [28, 38].
The extent and selectivity of transduction is shaped by promoter choice, with our interlaminar analysis across laminae VII–X revealing distinct patterns aligned with the functional organization of spinal circuits. In combination with the Hb9 promoter, which selectively drives expression in motoneurons of lamina IX [34], our AAV9-Hb9-LMO3-eYFP construct showed high specificity. By contrast, the hSyn promoter induced broader neuronal expression across laminae VII–X, while CMV, although highly active, lacked selectivity and also transduced glial cells [39]. Although different fluorescent reporters (eGFP, mCherry, eYFP) were used, their properties do not influence cellular tropism; all vectors shared the same AAV9 backbone, production protocol, and delivery route. The distinct laminar patterns therefore reflect promoter specificity, consistent with previous studies reporting broad CMV activity [39], pan-neuronal hSyn expression, and Hb9 restriction to motoneurons [34].
For example, in lamina VIII, CMV led to overexpression in V3 neurons, which regulate locomotor symmetry [35], potentially distorting motor patterns. Our data revealed pronounced differences in transduction across spinal cord laminae, highlighting their functional specialization. In lamina VII, expression was observed only with hSyn, which is associated with the presence of V2a and V2b interneurons. V2a neurons, being excitatory, participate in rhythm generation and left–right coordination [40], whereas V2b interneurons regulate the balance of flexors and extensors [41]. In lamina VIII, CMV promoter dominance is associated with the high density of V3 neurons, which ensure symmetric motor output [35]. In lamina IX, Hb9 specificity for motoneurons confirms their central role in motor signal generation, and the selective activation of these cells via LMO3 avoids interference with inhibitory circuits (e.g., V2b), which is critical for preventing spasticity [34]. In lamina X, CMV expression in the lumbar region, particularly in V0 subpopulations, correlates with their role in coordinating forelimb and hindlimb movements [42]. At the same time, the absence of transduction in thoracic segments emphasizes the segmental differences in the organization of motor networks.
The use of luminopsin LMO3 in combination with CTZ allows for non‑invasive activation of motoneurons, avoiding risks associated with optical fiber implantation [43]. The bioluminescence peak at approximately 35 minutes corresponds to CTZ pharmacokinetics [44, 45], limiting the duration of stimulation but making it compatible with short rehabilitation sessions. Importantly, combining optogenetics with motor training promotes activity-dependent synaptic plasticity [17]. For instance, activation of V2a interneurons associated with respiratory and locomotor networks may synergize with our approach, further enhancing functional recovery [40]. Despite these advances, a key limitation remains the dependence on exogenous CTZ. Potential solutions include nanocarriers for intranasal delivery [46] or self‑activating luminopsins [47, 48, 49]. Moreover, the long-term safety of AAV9 requires further investigation, especially in the context of immunogenicity [50, 51]. Nevertheless, our strategy, combining Hb9 selectivity and the non‑invasiveness of bioluminescence, paves the way for personalized therapy targeting spinal cord intrinsic networks in both injury and neurodegenerative diseases [17, 52].
Our study confirms that intrathecal administration of AAV9-Hb9-LMO3-eYFP is a highly specific method for modulating motoneurons critical for movement restoration. Differences in laminar transduction reflect their functional specialization, while the combination with bioluminescent stimulation and rehabilitation establishes a foundation for novel therapeutic strategies.
This study demonstrates that the specificity of AAV9-mediated transgene expression within the spinal cord is critically determined by the choice of promoter and the targeted grey matter lamina. The Hb9 promoter reliably confined expression to motoneurons within lamina IX across lumbar, thoracic, and cervical segments, providing a highly targeted approach compared with the broader neuronal transduction achieved by hSyn or the nonspecific expression driven by CMV. These findings highlight the importance of matching promoter elements to cellular populations and laminar architecture when designing gene therapy strategies for motor system disorders.
AAV, adeno-associated virus; SCI, spinal cord injury; CMV, cytomegalovirus; hSyn, human synapsin; Hb9, homeobox 9; GFP, green fluorescent protein; SMA, spinal muscular atrophy; ALS, amyotrophic lateral sclerosis; CPG, central pattern generator; LMO3, luminopsin; sbGLuc, gaussia luciferase variant; VChR1, Volvox canalorhodopsin-1; eYFP, yellow fluorescent protein; DNA, deoxyribonucleic acid; LS-LB, Luria Bertani Broth; DAPI, 4’,6-Diamidino-2-phenylindole; LPEI, linear polyethylenimine; DMEM/F-12, Dulbecco’s modified eagle medium/nutrient mixture F-12; PBS, phosphate buffered saline; DPBS, Dulbecco’s phosphate-buffered saline; MOI, multiplicity of infection; PCR, polymerase chain reaction; ITR, inverted terminal repeats; LDCom, the lateral dorsal commissural nucleus; IML, intermediolateral nucleus; D, dorsal nucleus; ICL, intercalated nucleus; CTZ, coelenterazine.
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to the evolving nature of the project.
TA and YM designed the research study. TA, YM, AR curated the data. TA, ED, EP, and YM performed the formal analysis. AR and YM obtained funding. ED, EP, RS, and YM conducted the investigation. TA and YM developed the methodology. TA and YM managed the project. AR and YM provided resources. AR and YM supervised the work. TA wrote the original draft. YM reviewed and edited the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All experiments involving animals were performed in accordance with the guidelines set forth by the European Communities Council Directive 86/609/EEC. The experimental protocols were approved by the Animal Care and Use Committee of the Kazan Federal University (protocol No. 50, dated 26.09.2024).
Not applicable.
This work was supported by a grant from the Russian Science Foundation, No. 23-75-10041; https://rscf.ru/en/project/23-75-10041/ (to Y.M.)
The authors declare no conflict of interest.
During the preparation of this work the authors used ChatGPT-4o in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBE44274.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




