1 Department of Human Factors and Neurobiology, College of Arts and Sciences, Embry-Riddle Aeronautical University, Daytona Beach, FL 32114, USA
2 Department of Biomedical Sciences, College of Medicine, University of Central Florida, Orlando, FL 32827, USA
3 Department of Molecular and Cellular Biology, The College of Liberal Arts and Sciences (TCLAS), Arizona State University, Tempe, AZ 85287, USA
4 Department of Natural Sciences, Texas A&M University, San Antonio, TX 78224, USA
Abstract
The global rise of antimicrobial resistance necessitates the development of innovative therapeutic strategies beyond traditional antibiotics. Drug repurposing offers a time- and cost-effective approach by identifying new antimicrobial applications for existing medications. Thus, this study aimed to investigate the antimicrobial and anti-virulence potential of several clinically approved drugs, including fluconazole, buspirone, duloxetine, escitalopram, and finasteride.
We evaluated the antimicrobial efficacy of the selected compounds against a panel of microorganisms comprising two Gram-negative bacteria (Escherichia coli, Serratia marcescens), two Gram-positive bacteria (Bacillus megaterium, Staphylococcus epidermidis), and two opportunistic yeasts (Candida albicans, Rhodotorula mucilaginosa). Antimicrobial activity was evaluated using growth inhibition and viability assays. Additionally, we investigated the effects of the selected drugs on fungal virulence traits, including biofilm formation and filamentation, and assessed infectivity using a Caenorhabditis elegans host model.
Duloxetine and escitalopram demonstrated broad-spectrum antimicrobial activity, inhibiting bacterial and fungal growth at concentrations below 512 mg/L. Buspirone exhibited selective antimicrobial effects, particularly against Gram-positive bacteria and C. albicans. Although finasteride exhibited limited direct antifungal activity, it significantly disrupted key virulence traits in yeasts, including biofilm formation, morphological transitions, and host infection capacity.
These findings underscore the potential of serotonin reuptake inhibitors and finasteride as candidates for antimicrobial repurposing. By impairing both microbial viability and pathogenicity, these drugs may provide promising avenues for developing adjunct or alternative therapies against resistant bacterial and fungal pathogens.
Keywords
- finasteride
- serotonin inhibitors
- drug repurposing
- antimicrobial
The escalating global threat of antimicrobial resistance (AMR) has exposed the limitations of traditional antibiotics and antifungal therapies, highlighting the urgent need for innovative and sustainable treatment strategies [1]. One promising approach is drug repurposing, which involves identifying new antimicrobial uses for existing drugs originally developed for non-infectious conditions [2]. This strategy offers several advantages over conventional drug discovery, including well-characterized safety profiles, known pharmacokinetics, and significantly reduced development time and cost [2, 3].
Recent interest has focused on the antimicrobial potential of non-antibiotic drugs, particularly antidepressants and hormone-regulating agents, due to emerging evidence of their off-target effects on microbial growth and viability [4]. Studies have shown that certain antidepressants, such as selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), can inhibit the proliferation of pathogenic bacteria and fungi, potentially through mechanisms unrelated to their neurological targets. Similarly, hormone-modulating compounds have demonstrated the ability to disrupt microbial processes such as biofilm formation and fungal filamentation, suggesting a broader therapeutic potential [4, 5, 6, 7, 8].
However, repurposing these agents for antimicrobial use also raises important considerations regarding their unintended effects on the human microbiome [9, 10, 11, 12]. Many of these compounds, while not inherently antimicrobial, may influence the composition, diversity, or function of commensal microbial communities [12, 13]. Disruption of the microbiota, particularly in the gastrointestinal tract, can lead to dysbiosis, impair immune responses, and negatively impact host metabolism and drug efficacy. Therefore, evaluating the antimicrobial properties of repurposed drugs must be coupled with assessments of their microbiome impact to ensure therapeutic benefits without adverse consequences.
Despite growing evidence supporting the antimicrobial activity of certain
psychotropic agents, antihormonal drugs, such as finasteride, remain relatively
understudied in this context. Finasteride, a 5
Importantly, the antimicrobial spectrum of these repurposed compounds, particularly their activity against a range of Gram-positive and Gram-negative bacteria, as well as clinically relevant fungal pathogens, remains incompletely understood. This knowledge gap is increasingly relevant given the intersection of AMR, drug-induced microbiome changes, and their collective impact on host immunity, therapeutic response, and disease progression. Identifying therapeutic windows in which repurposed drugs can effectively target pathogens without compromising beneficial microbes represents a critical step toward safer and more effective treatment options.
In this study, we evaluated the antimicrobial potential of several repurposed drugs against a panel of representative bacterial and fungal pathogens. The compounds selected include: fluconazole, a well-established antifungal agent [16], used here as a reference standard; buspirone, an anxiolytic acting as a serotonin 5-HT₁A receptor partial agonist [17]; duloxetine, an SNRI indicated for depression and chronic pain [18]; escitalopram, an SSRI commonly prescribed for mood and anxiety disorders [19]; and finasteride, a hormone-modulating agent targeting androgen metabolism [15].
The antimicrobial effects of these compounds were tested against Gram-negative bacteria (Escherichia coli, Serratia marcescens), Gram-positive bacteria (Bacillus megaterium, Staphylococcus epidermidis), and two opportunistic fungal pathogens (Candida albicans, Rhodotorula mucilaginosa). By characterizing their effects on microbial viability and virulence-related traits, this study aims to contribute novel insights into the feasibility of drug repurposing as a complementary strategy to combat infectious diseases and address the growing burden of antimicrobial resistance.
This study utilized six microbial strains: two Gram-negative bacteria, Escherichia coli K12 (American Type Culture Collection [ATCC] 25404 Strain Castellani and Chambers) and Serratia marcescens (ATCC LRA 04.03.73); two Gram-positive bacteria, Bacillus megaterium (ATCC 14581, also known as Priestia megaterium) and Staphylococcus epidermidis (ATCC PCI 1200, FDA strain); and two fungal isolates, Rhodotorula mucilaginosa (ATCC 9449) and Candida albicans (ATCC 18804). The compounds evaluated in this study included fluconazole (Millipore Sigma, PHR1160, Burlington, MA, USA), buspirone HCl (AdooQ Bioscience, A15687, Irvine, CA, USA), duloxetine (AdooQ Bioscience, A11705, Irvine, CA, USA), escitalopram oxalate (AdooQ Bioscience, A16533, Irvine, CA, USA), and finasteride (AdooQ Bioscience, A10387, Irvine, CA, USA). Bacterial cultures were grown and maintained at 30 °C in LB medium (1% tryptone, 1% sodium chloride, 0.5% yeast extract), while fungal strains were cultivated at 30 °C in yeast peptone dextrose (YPD) medium (1% yeast extract, 2% peptone, 2% glucose).
Antimicrobial activity was assessed using growth inhibition assays in 96-well microtiter plates (Corning, Sigma-Aldrich CLS3628, St. Louis, MO, USA) with LB or YPD media, depending on the organism [20]. Bacterial and fungal strains were cultured overnight, then diluted to a final optical density at 630 nm (OD630) of 0.1. Test compounds were applied at serially increasing concentrations (2, 4, 8, 16, 32, 64, 128, 256, 512, and 1024 mg/L), alongside a drug-free positive control for each strain [21]. Wells containing only media and drug (without cells) served as negative controls. Plates were incubated at 30 °C for 24 hours. The minimum inhibitory concentration (MIC) was defined as the lowest drug concentration at which visible growth inhibition was observed. Optical density (OD) measurements were recorded at 630 nm using a microplate reader (Synergy H1, BioTek, Winooski, VT, USA), which is the closest available setting to 600 nm on this instrument and remains within the optimal range for monitoring microbial growth curves, and compared to cell free controls. All experiments were performed independently five times (biological replicates), with each assay conducted in triplicate (technical replicates).
For the filamentation, colony morphology, and biofilm formation assays, the yeast strains Rhodotorula mucilaginosa (ATCC 9449) and Candida albicans (ATCC 18804), previously used in the MIC determinations, were selected.
The antifungal activity of the tested compounds in both the prevention and treatment of biofilms was evaluated using the XTT [2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide] reduction assay, as previously described [22], with YPD as the growth medium.
For biofilm prevention, planktonic yeast cells from overnight cultures (adjusted to OD630) were incubated with increasing concentrations of each drug (as described previously) in 96-well plates at 37 °C for 24 hours. For biofilm treatment, mature biofilms were first established by incubating drug-free, adjusted cultures in YPD for 24 hours at 37 °C [23]. After incubation, non-adherent planktonic cells were removed and wells were gently washed. Pre-formed biofilms were then treated with increasing concentrations of each drug in YPD and incubated for an additional 24 hours at 37 °C. XTT reduction was measured to assess metabolic activity, and thus biofilm viability. All assays were conducted using three independent biological replicates, each performed in triplicate (technical replicates).
Colony morphology was assessed through two distinct assays: filamentation in Candida albicans and capsule formation in Rhodotorula mucilaginosa.
For filamentation, Candida albicans overnight cultures were adjusted to an optical density at 630 nm (OD630) of 0.5. A 10 µL aliquot of each culture was spotted onto YPD agar plates supplemented with 10% fetal calf serum (FCS) and varying concentrations of the test compounds (4, 8, 16, and 64 mg/L) [24]. Drug-free plates served as positive controls. Plates were incubated at 37 °C for five days, after which colony growth and filamentous morphology were documented and compared to the controls. For capsule formation, Rhodotorula mucilaginosa cultures were similarly adjusted to OD630 = 0.5, and 10 µL aliquots were spotted onto cornmeal agar plates (2% agar, 2% cornmeal, 2% peptone, 2% glucose) [25]. After five days of incubation at 37 °C, colonies were examined microscopically for capsule production and photographed.
Nematode killing assays were performed as previously described [26]. Briefly, overnight cultures of Candida albicans and Rhodotorula mucilaginosa incubated with different concentrations of the drugs (4, 8, 16 and 64 mg/L) were adjusted to an OD630 of 0.5. A 100 µL aliquot of each culture was evenly spread onto YPD agar plates to form a lawn and incubated at 30 °C for 24 hours.
Caenorhabditis elegans were maintained on Escherichia coli K12 at approximately 25 °C and synchronized to the L4 larval stage. For each assay, approximately 100 L4-stage nematodes were transferred to the yeast lawns and incubated at room temperature (~25 °C). Worm survival was monitored daily over a five-day period. Nematodes were considered dead if they failed to respond to mechanical stimulation with an inoculation needle.
Data from growth, biofilm, and nematode killing assays were analyzed using
one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test for
multiple comparisons. Differences between experimental groups and controls were
considered statistically significant at p
The antimicrobial properties (including both antibacterial and antifungal activities) of five pharmacologically active compounds, namely fluconazole (FLU), buspirone (BUS), duloxetine (DUL), escitalopram (ESC), and finasteride (FIN), were systematically evaluated using the MIC dilution method in microplates. The MIC values represent the lowest concentration of the compound required to inhibit 90% of microbial growth in comparison to drug-free positive controls (MIC90). The complete results are illustrated in Fig. 1.
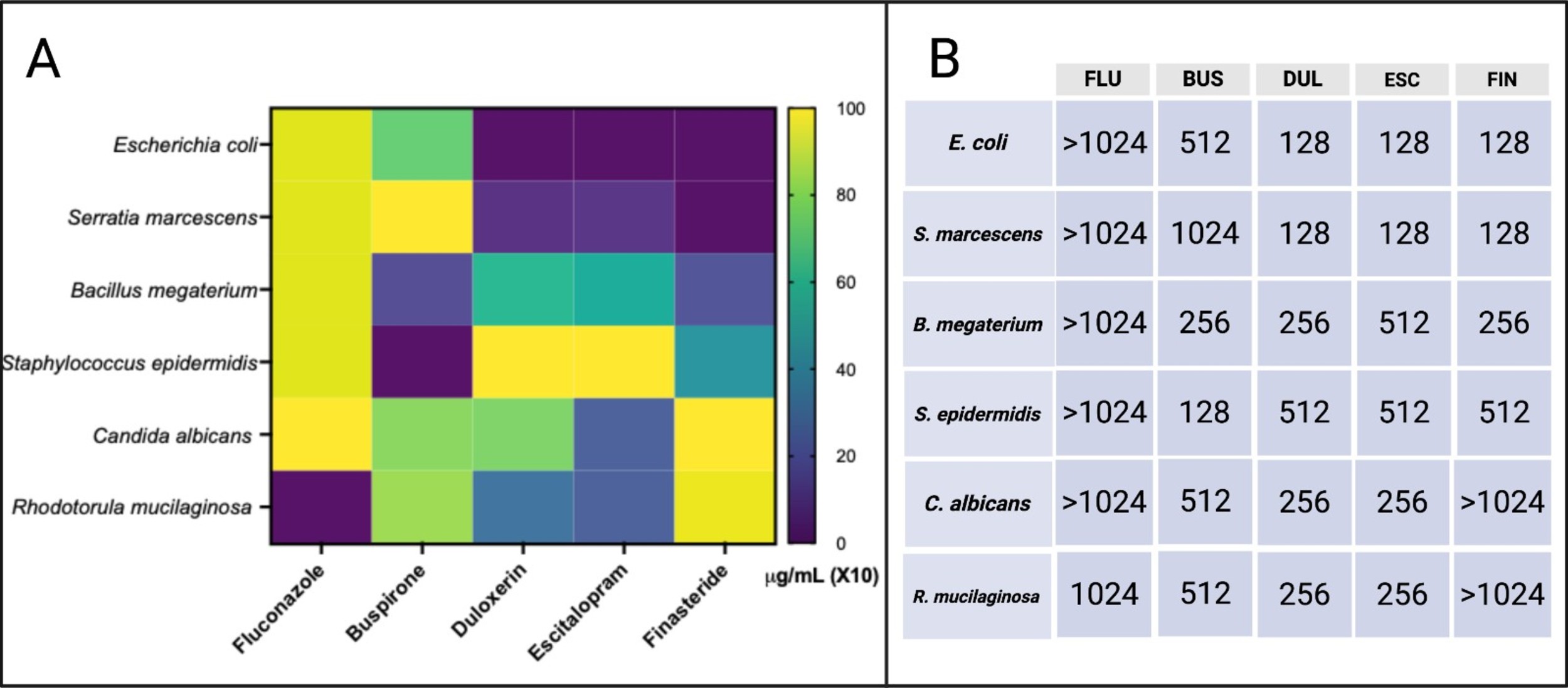 Fig. 1.
Fig. 1.
Antimicrobial susceptibility patterns of planktonic bacteria and yeast. (A) Heatmap illustrating the minimum inhibitory concentration (MIC) required to inhibit 90% of microbial growth (MIC90) relative to untreated controls. Color intensity corresponds proportionally to increasing drug concentrations. (B) Summary table displaying the MIC90 values (mg/L, equivalent to µg/mL) for each antimicrobial agent tested against individual microorganisms. FLU, fluconazole; BUS, buspirone; DUL, duloxetine; ESC, escitalopram; FIN, finasteride.
Fluconazole, a reference antifungal agent, showed no significant inhibitory effect against any of the tested microbial strains, including yeast species such as Candida albicans and Rhodotorula mucilaginosa, even at the highest concentration tested (1024 mg/L). In contrast, buspirone exhibited antibacterial activity within a concentration range of 128 to 1024 mg/L. Notably, its antibacterial effects were more pronounced against Gram-positive bacterial strains such as Bacillus megaterium and Staphylococcus epidermidis, compared to the Gram-negative strains Escherichia coli and Serratia marcescens.
Duloxetine and escitalopram demonstrated the most potent antifungal activity among the tested compounds, effectively inhibiting fungal growth at 256 mg/L. Moreover, escitalopram also exhibited considerable antibacterial activity. It showed greater efficacy against Gram-negative bacteria, with a MIC90 of 128 mg/L, in contrast to a MIC90 of 512 mg/L observed for Gram-positive species, suggesting selective potency.
Finasteride displayed the strongest antibacterial activity against Gram-negative bacteria with a MIC90 of 128 mg/L, while its inhibitory effect on Gram-positive bacteria was comparatively weaker, with MIC90 values of 256 mg/L for Bacillus megaterium and 512 mg/L for Staphylococcus epidermidis. However, finasteride exhibited no antifungal activity against C. albicans or R. mucilaginosa at the highest tested concentration (1024 mg/L).
In our study, cultures were maintained at 30 °C for MIC determinations and at 37 °C for biofilm and filamentation assays. The decision to use 30 °C for MIC testing was based on our laboratory’s standard culture conditions for both fungal and bacterial strains, as well as the longer incubation times required, 24 hours in our protocol compared to the 12–18 hours typically reported for assays conducted at 37 °C [27]. While many antimicrobial susceptibility studies, particularly those involving clinical isolates, are conducted at 37 °C, the effect of temperature on MIC values has been well-documented, with reports showing notable differences in susceptibility at higher temperatures such as 35 °C and 37 °C. Our current study is limited by the use of a single incubation temperature for MIC testing; however, this choice provides consistency with our existing culture workflows and may also capture resistance patterns that have been observed more frequently at 30 °C in certain non-clinical bacterial isolates. Nevertheless, to enhance clinical relevance and comparability, future studies will incorporate MIC testing at 35 °C and 37 °C, alongside optimized media, to produce more standardized and generalizable results across strains.
Fluconazole is a well-established antifungal agent with extensively documented
efficacy against a broad spectrum of fungal species. However, the potential
antifungal activity of non-traditional agents, such as antidepressants and
5
It is important to note that fungal pathogenicity is not solely dependent on viability. Numerous studies have shown that sub-inhibitory concentrations of various drugs can impact critical virulence traits without necessarily causing cell death. Such traits include biofilm formation, filamentation, and tissue invasion which are key processes involved in host colonization and disease progression. Given the clinical relevance of fungal biofilms, which are notoriously resistant to conventional antifungal therapies, we also focused our attention on the antibiofilm potential of the selected compounds.
To this end, we employed a metabolic activity assay based on the reduction of the tetrazolium salt XTT to formazan, which provides an indirect but quantifiable measure of biofilm viability and metabolic activity. Biofilms of Candida albicans and Rhodotorula mucilaginosa were subjected to increasing concentrations of the test compounds (ranging from 2 to 1024 mg/L), and two experimental approaches were used. In the first approach, drugs were added simultaneously with fungal cells to assess their ability to prevent the initial attachment and establishment of biofilms. In the second approach, drugs were introduced after the formation of mature biofilms to evaluate their potential in disrupting established microbial communities.
Fig. 2 presents the results for C. albicans biofilms. When drugs were
added during the initial adhesion phase, all compounds tested demonstrated a
measurable inhibition of biofilm development. At a low concentration of 8 mg/L, a
modest reduction of approximately 20% in biofilm metabolic activity was
observed. More substantial inhibition, exceeding 50%, was achieved with
concentrations of 16 mg/L and above. In contrast, when applied to mature
C. albicans biofilms, duloxetine and escitalopram exhibited the
strongest antibiofilm activity, showing significant reductions in metabolic
activity starting at concentrations as low as 8 mg/L. Buspirone and finasteride
also demonstrated inhibitory effects on mature biofilms, although higher
concentrations (
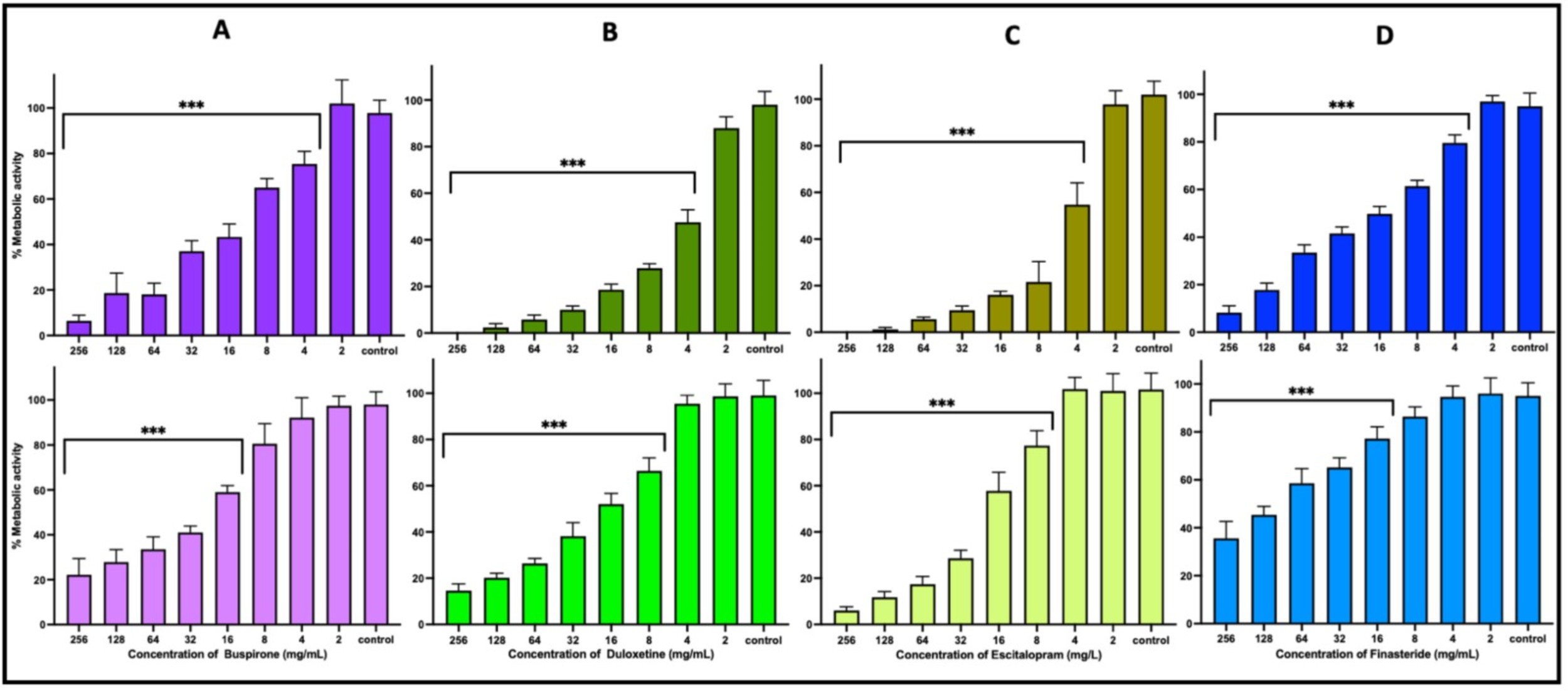 Fig. 2.
Fig. 2.
Effect of drug concentrations on Candida albicans
biofilm integrity. The upper panel illustrates the prevention of biofilm
formation, while the lower panel shows the treatment effects on mature biofilms.
Biofilm viability is represented in terms of metabolic activity, with each drug
treatment compared to the untreated control. Statistical analysis was performed
using one-way analysis of variance (ANOVA) followed by t-tests;
statistically significant differences (***p
Similarly, Fig. 3 illustrates the antibiofilm effects of the tested compounds on
R. mucilaginosa biofilms. In the biofilm prevention model, even low
concentrations (8 mg/L) of all drugs resulted in a notable reduction in biofilm
formation, with duloxetine and escitalopram again producing the most pronounced
effects, achieving over 50% reduction in biofilm metabolic activity. When
targeting pre-formed, mature R. mucilaginosa biofilms, duloxetine and
escitalopram maintained superior efficacy at concentrations of 16 mg/L or higher.
Finasteride was effective at reducing biofilm activity at 32 mg/L, while
buspirone required higher concentrations (
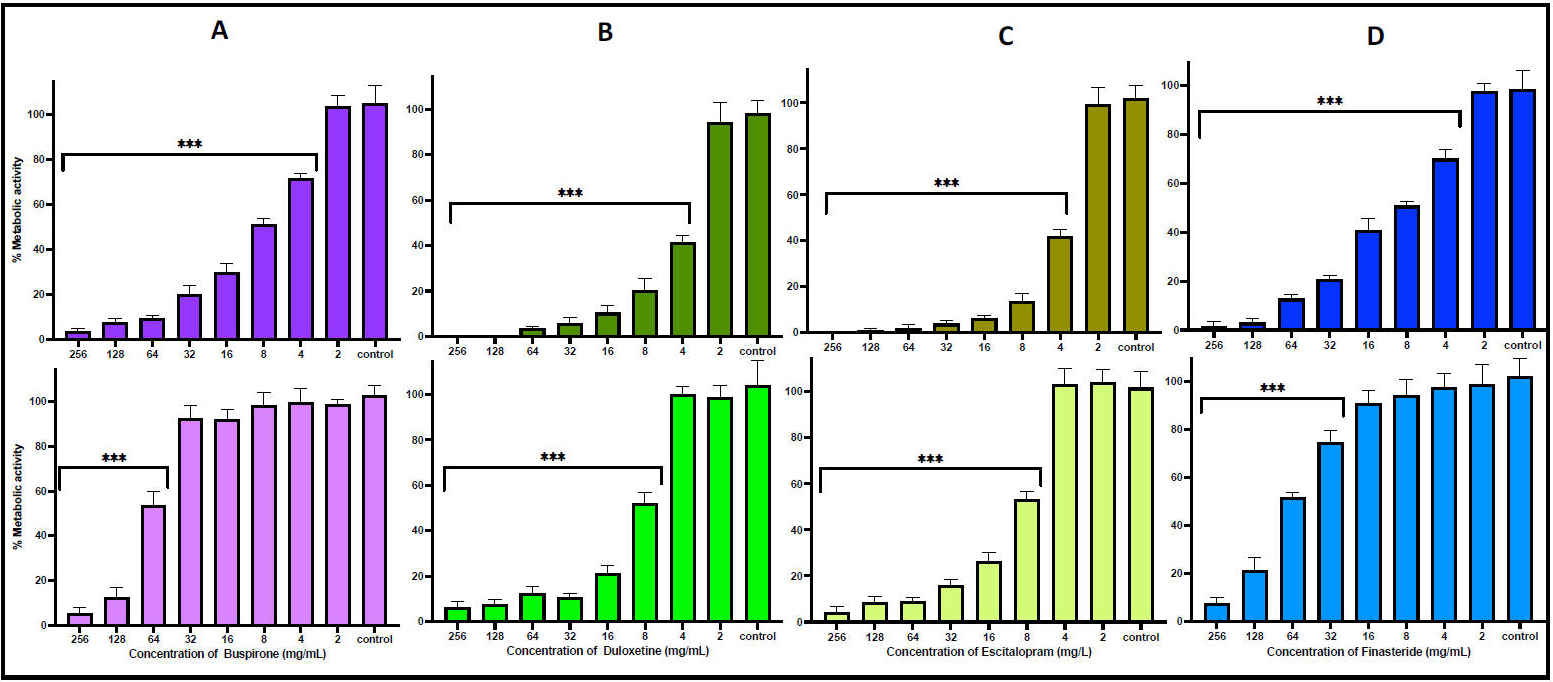 Fig. 3.
Fig. 3.
Effect of drug concentrations on Rhodotorula
mucilaginosa biofilm integrity. The upper panel illustrates the prevention of
biofilm formation, while the lower panel shows the treatment effects on mature
biofilms. Biofilm viability is represented in terms of metabolic activity, with
each drug treatment compared to the untreated control. Statistical analysis was
performed using one-way ANOVA followed by t-tests; statistically
significant differences (***p
Collectively, these findings support the potential of certain antidepressants and finasteride as modulators of fungal virulence, particularly in the context of biofilm inhibition. These results warrant further investigation into the underlying mechanisms of action and raise the possibility of repurposing these drugs as adjunctive therapies for fungal infections characterized by robust biofilm formation.
Candida albicans plays a crucial role in infection dynamics, notably through its ability to undergo a phenotypic switch from a yeast form to filamentous hyphae. This transition is critical for host cell invasion and the progression of infection, as the hyphal structures contribute to tissue penetration and pathogenicity. Filamentation can be observed both macroscopically and microscopically. Macroscopically, it is evident through the formation of wrinkled colonies on agar plates, particularly when supplemented with media that promote filamentation. Microscopically, filamentation is characterized by the elongation of hyphae, which can be further analyzed in terms of hyphal length, branching, and density.
In this study, we focused on the macroscopic examination of colony morphology, specifically the presence of wrinkled versus smooth colonies, as a straightforward indicator of filamentation and mycelia formation. Colonies were incubated for five days on YPD agar supplemented with fetal calf serum, which promotes the filamentous phenotype. Fig. 4 presents representative results, where a wrinkled colony of C. albicans was observed in the absence of any drug treatment, indicating normal filamentation. In contrast, smooth colonies, indicative of inhibited filamentation, were observed upon the addition of various drugs. The concentrations at which filamentation was significantly inhibited, resulting in a smooth phenotype, were 4 mg/L for DUL and ESC, 8 mg/L for FIN, and 16 mg/L for BUS. Notably, these concentrations were lower than those required to inhibit biofilm formation, suggesting that the effects of these drugs on filamentation occur at concentrations that are more potent than those necessary for biofilm inhibition.
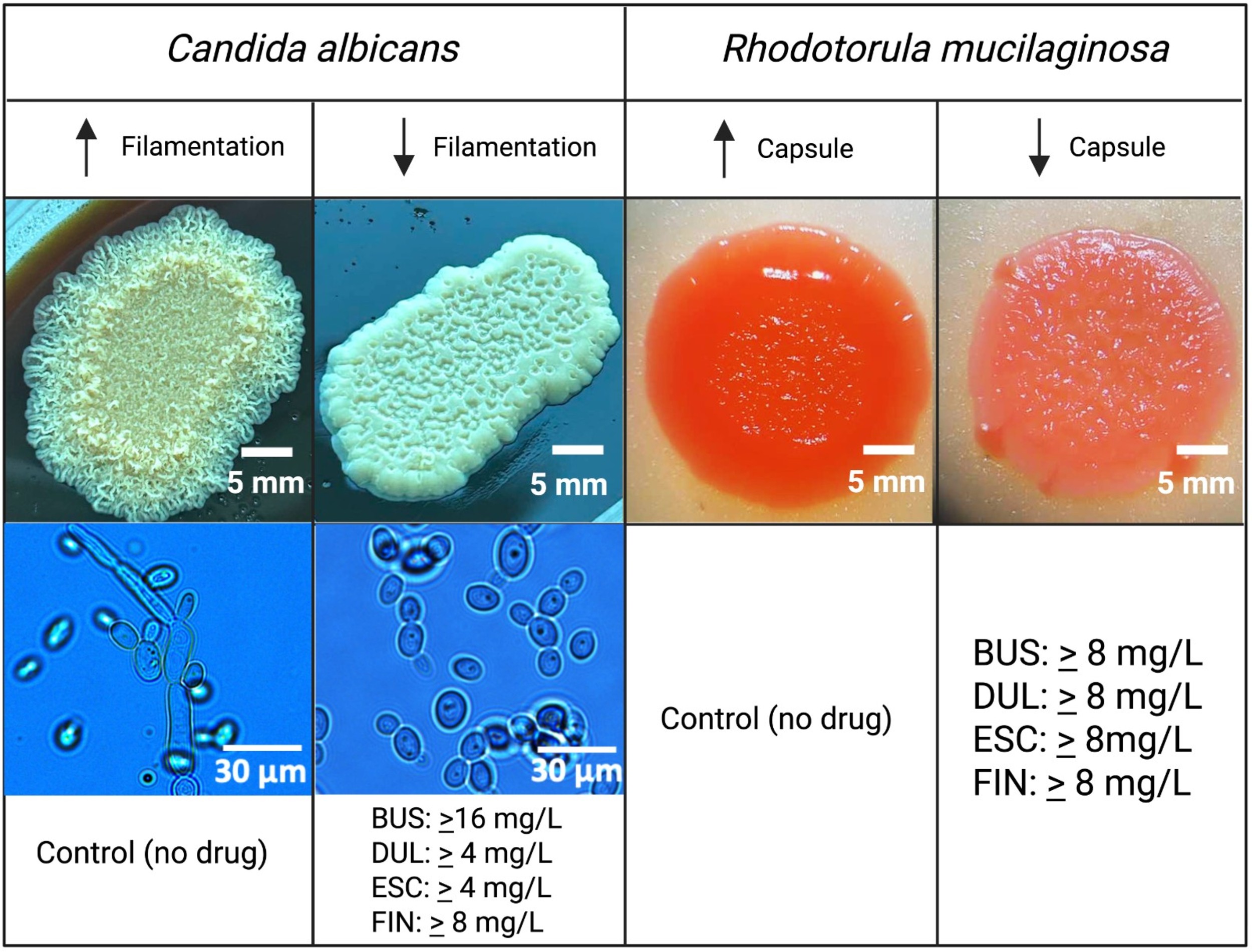 Fig. 4.
Fig. 4.
Effect of drug concentrations on Candida albicans
filamentation and Rhodotorula mucilaginosa capsule formation.
Qualitative results are presented, highlighting the specific drug concentrations
that induced observable changes in filamentation in C. albicans both
macroscopically or microscopically (400
Rhodotorula mucilaginosa, in contrast to C. albicans, does not significantly form hyphal structures that are visually detectable at the colony level. Instead, this yeast species is characterized by the formation of a protective capsule, which plays a key role in immune evasion. This capsule is typically observed as a shiny, smooth colony with a pronounced pink pigment, distinguishing it from non-capsulated strains or those with less developed capsular structures. Fig. 4 demonstrates that the presence of the capsule was significantly reduced or absent when the drugs were administered at concentrations of 8 mg/L or higher. This observation follows a similar pattern to the reduction in biofilm formation, suggesting a parallel effect on the virulence traits of R. mucilaginosa.
Yeast strains were cultured in the presence of varying drug concentrations (2,
4, 8, 16, and 64 mg/L) and subsequently used to infect Caenorhabditis
elegans, following previously established protocols. The resulting nematode
survival rates after five days are shown in Fig. 5. Treatment with 16 mg/L
significantly increased survival, producing a
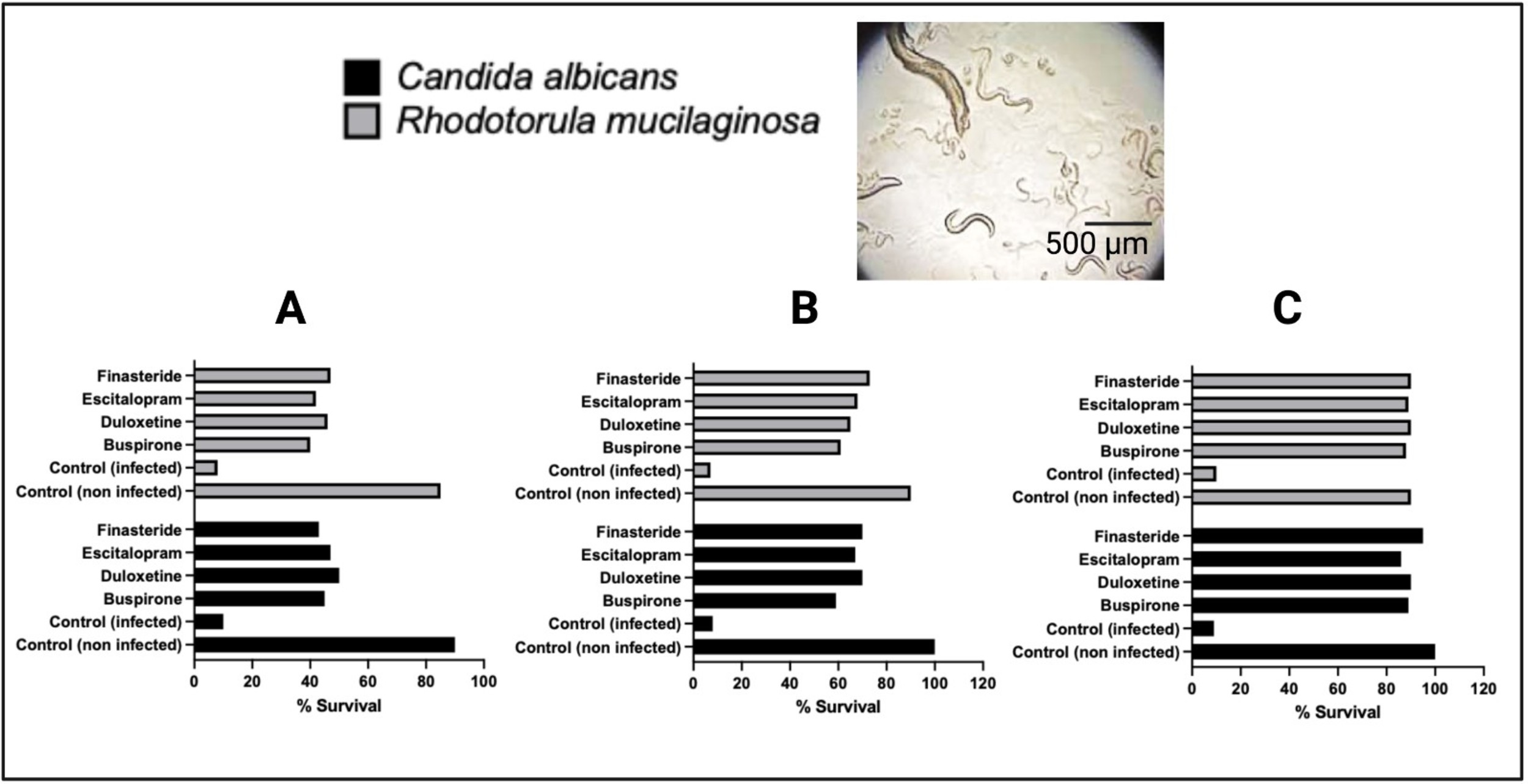 Fig. 5.
Fig. 5.
Effect of drug-treated yeast strains on Caenorhabditis elegans viability. Effect of drug-treated yeast strains on Caenorhabditis elegans viability. Yeast strains were pre-treated with varying concentrations of each drug: (A) 4 mg/L, (B) 8 mg/L, and (C) 16 mg/L, and subsequently used to infect C. elegans L4 larvae. Nematode viability was assessed after 5 days. Uninfected control worms (Control, non-infected) exhibited full survival, while worms infected with untreated yeast strains (Control, infected) showed approximately 90% mortality. Infection with drug-treated yeast strains resulted in a concentration-associated increase in worm survival: ~30% survival with 4 mg/L, 50–70% survival with 8 mg/L, and 85–95% survival with 16 mg/L treatments. These findings indicate a potential protective effect of yeast pre-treatment on host survival, although more detailed statistical analyses are required to confirm the relationship between concentration and survival outcome. Scale bar corresponds to 500 µm.
Drug repurposing (defined as the process of identifying new therapeutic uses for existing, approved, or investigational drugs outside the scope of their original medical indication) has emerged as a promising strategy in the fight against microbial infections [2]. This approach has gained significant momentum over the past decade, driven largely by the global rise in antimicrobial resistance, which continues to outpace the development of novel antibiotics. Leveraging the established pharmacokinetics, pharmacodynamics, and safety profiles of known compounds, drug repurposing offers a time- and cost-efficient alternative to traditional drug discovery pipelines [28]. Despite its growing appeal, the application of repurposed drugs in infectious disease contexts remains relatively underexplored, warranting further research to elucidate their mechanisms of action and therapeutic potential, particularly in translational and clinical settings.
In this study, we evaluated the antimicrobial efficacy of a select group of
pharmacological agents with established clinical use in non-infectious diseases:
the SSRIs duloxetine and escitalopram, the serotonin receptor agonist
buspirone, and the 5
To contextualize the activity of these repurposed agents, we included fluconazole, a well-characterized antifungal, as a reference compound [16]. Although fluconazole is widely used to treat Candida infections, its efficacy is frequently compromised by resistance, particularly in Candida albicans and other opportunistic yeasts [30]. In some clinical cases, elevated doses are required to achieve fungistatic or fungicidal effects. By comparing the effects of fluconazole to those of the tested drugs, we aimed to benchmark the antimicrobial potential of the repurposed compounds against clinically relevant fungal pathogens.
To further investigate the potential antimicrobial effects of the selected repurposed drugs, we examined their impact on virulence-associated phenotypes in Candida albicans and Rhodotorula mucilaginosa which are two opportunistic fungal pathogens commonly found in the human microbiota. R. mucilaginosa is typically associated with mild superficial infections, such as onychomycosis, whereas C. albicans exhibits a broader pathogenic potential, ranging from mucocutaneous infections (e.g., oral thrush) to life-threatening systemic infections, particularly in immunocompromised individuals [31, 32]. Although viability assays are frequently used to assess antifungal efficacy, recent studies have emphasized that the attenuation of virulence traits (that are independent of direct fungicidal activity) can be a valuable therapeutic strategy. Even in cases where fungal viability is not significantly compromised, sub-inhibitory concentrations of certain compounds can disrupt key virulence mechanisms, such as the yeast-to-hyphal transition (filamentation), biofilm formation, and host tissue invasion [33, 34, 35]. These phenotypic changes can reduce the pathogen’s ability to establish infection and evade the host immune system. Given this, we focused on evaluating the effects of selected compounds on filamentation and biofilm formation using well-established qualitative and quantitative assays. Additionally, we employed a Caenorhabditis elegans infection model to assess pathogenicity and host survival, thereby providing a comprehensive view of how these repurposed drugs may influence fungal virulence beyond conventional viability endpoints.
While many studies have explored the synergistic potential of fluconazole in combination with other agents [36, 37, 38, 39, 40], our investigation focused exclusively on the independent effects of each compound. Nonetheless, the robust antimicrobial and anti-virulence activity observed in this study highlights the potential for future exploration of combination therapies involving these agents. Our findings contribute to the growing body of evidence supporting drug repurposing as a viable approach in antimicrobial research and offer new insights into the potential translational applications of these commonly prescribed medications in managing resistant infections.
The antibacterial and antifungal effects observed in this study varied depending on the drug tested. As expected, fluconazole (an antifungal agent) did not affect bacterial viability at any of the concentrations tested. Fluconazole exerts its antifungal activity by inhibiting ergosterol synthesis [41], a key component of fungal cell membranes that is absent in bacterial membranes and cell walls. This lack of a shared target likely explains its ineffectiveness against bacterial strains in our assay. Interestingly, although fluconazole is not traditionally considered an antibacterial agent, previous studies have reported potential off-target effects on certain bacterial species. For example, some studies [41, 42, 43] demonstrated that fluconazole, at high concentrations (2000 mg/L), inhibited both growth and biofilm formation in selected Gram-negative and Gram-positive bacteria, including Escherichia coli and Streptococcus mitis. However, no such effects were observed in other species such as Lactobacillus salivarius, indicating that fluconazole’s off-target activity may be species-specific. The mechanisms underlying these antibacterial effects remain poorly understood. It is plausible that, at high concentrations, fluconazole may interfere with bacterial membrane integrity or metabolic pathways through non-specific interactions. Further research is warranted to elucidate these potential mechanisms and to determine whether similar effects occur across a broader range of bacterial species and strains. In parallel, the viability of the yeast strains tested in our study was also not compromised by the highest concentration of fluconazole used. This is consistent with prior reports indicating that fluconazole has limited efficacy against certain yeast species [44, 45], while other antifungals (such as ketoconazole and nystatin) often exhibit greater potency [42]. Susceptibility to fluconazole is known to vary among Candida albicans strains, with MICs ranging widely from 32 to 2500 mg/L, particularly among clinical isolates, which tend to exhibit higher levels of resistance [44, 45, 46, 47]. The Candida albicans strain used in this study (ATCC 18804) is reported by the ATCC to be susceptible to fluconazole at concentrations of 64 mg/L and has been widely used as a reference strain in multiple studies. However, previous research has shown variability in susceptibility patterns depending on the growth medium employed. For example, the medium optimized for fluconazole susceptibility testing in clinical isolates RPMI medium with L-glutamine (GIBCO), buffered with morpholinepropanesulfonic acid (MOPS) and supplemented with 2 g/L glucose, has been identified as particularly suitable for standardized assays, especially when multiple C. albicans clones are tested either alone or in combination with other antifungals [45, 48]. In our study, the absence of detectable fluconazole activity under our assay conditions likely reflects the use of a different medium and temperature, as well as the drug concentration tested. We acknowledge that this limited the role of fluconazole as an effective positive control in our antimicrobial susceptibility testing. To address this, future work will incorporate the optimized RPMI/MOPS medium, include incubation at 37 °C, and expand testing to additional strains. We will also evaluate a broader range of fluconazole concentrations to establish a precise MIC for the strain used and to better characterize its susceptibility profile. These adjustments will help ensure more reproducible and clinically relevant results in line with established susceptibility data. To more accurately determine the susceptibility of the Candida strain used in our experiments, future studies will incorporate higher fluconazole concentrations in combination with to establish the precise MIC and to further characterize resistance profiles.
Buspirone demonstrated notable antibacterial and antifungal activity in this study, showing greater efficacy against Gram-positive bacteria compared to Gram-negative strains and fungi. Although primarily prescribed as an anxiolytic due to its unique pharmacological profile as a serotonin 5-HT₁A receptor partial agonist [17], buspirone has also been reported to exhibit anti-inflammatory properties, which have been explored in the context of alternative effects [49, 50]. However, to our knowledge, its direct antimicrobial effects have not been previously reported, and the underlying mechanism of action remains unknown. To elucidate the pathways involved in buspirone’s antimicrobial activity, future studies employing metabolomic and transcriptomic analyses are warranted. These approaches may help identify disrupted microbial biochemical pathways and reveal potential molecular targets. One hypothesis is that buspirone’s antimicrobial activity may be linked to its anti-inflammatory properties, as other anti-inflammatory drugs have previously been shown to possess antimicrobial effects and are increasingly being considered as promising repurposing candidates [51, 52, 53, 54, 55]. Beyond its effects on microbial viability, buspirone significantly disrupted key virulence-related traits in Candida albicans, including biofilm formation, filamentation, and host infection. Remarkably, these phenotypic changes occurred at concentrations over 100-fold lower than those required to inhibit fungal growth, suggesting that buspirone may interfere with signaling or regulatory mechanisms essential for fungal pathogenicity. These findings underscore the potential of buspirone as a candidate for antifungal repurposing and highlight the need for further molecular and phenotypic investigations to better understand its mechanism of action and therapeutic potential.
Duloxetine and escitalopram emerged as the most effective antimicrobial agents among the tested compounds, exhibiting broad-spectrum activity at relatively low concentrations (128–512 mg/L). Both drugs demonstrated notable efficacy against all microbial species evaluated in this study, with particularly strong activity against Gram-negative bacteria. These findings align with prior research suggesting the antimicrobial potential of SSRIs and SNRIs against enteric pathogens [56, 57, 58]. For instance, structurally similar antipsychotics and antidepressants, such as atomoxetine, have shown effectiveness against Salmonella and Yersinia species. Moreover, a study conducted by Chavez-Dozal AA et al. [59] reported that escitalopram significantly inhibited the growth of 47 clinical isolates of Gram-positive bacteria, highlighting its potential as a repurposed antibacterial agent with activity across a wide microbial spectrum.
In the context of fungal pathogens, escitalopram has received less attention, particularly with respect to Rhodotorula mucilaginosa, for which, to our knowledge, this is the first study investigating its susceptibility to SSRIs. Conversely, Candida albicans has been extensively studied as a model organism in drug repurposing research. A recent study by Patangia DV et al. [11] demonstrated that escitalopram at concentrations as low as 87 mg/L compromised the viability of C. albicans. Consistent with those findings, we observed that while a concentration of 256 mg/L was required to significantly inhibit fungal growth, much lower doses (4–8 mg/L) were sufficient to disrupt critical virulence-associated phenotypes such as biofilm formation, filamentation, and host lethality in a C. elegans infection model. These results suggest that escitalopram may exert anti-virulence effects at sub-inhibitory concentrations, a property that could be leveraged therapeutically to reduce pathogenicity without exerting selective pressure for resistance.
Duloxetine, an SNRI more frequently studied for antimicrobial purposes, has shown documented activity against a variety of microbial taxa, including Staphylococcus aureus, Escherichia coli, Bacteroides, and Lactobacillus spp. [60]. Its antifungal effects have also been validated: Cantón E et al. [36] reported that duloxetine, when combined with fluconazole, exhibited synergistic inhibition of fluconazole-resistant C. albicans, with effective concentrations as low as 3 mg/L (FLU) and 160 mg/L (DUL). Our findings corroborate these results, demonstrating that duloxetine alone (at comparably low concentrations) can disrupt fungal biofilm integrity, inhibit filamentation, and reduce virulence in host models. Based on this, we hypothesize that combining duloxetine with fluconazole may produce enhanced antifungal effects even in resistant strains. Such synergistic combinations will be prioritized in our future research to assess their clinical potential.
Nevertheless, while the antimicrobial efficacy of these antidepressants is compelling, it is critical to consider potential risks associated with their long-term use or environmental accumulation. A study has cautioned that antidepressants, including duloxetine, may contribute to the emergence of antimicrobial resistance through bioaccumulation in microbial communities [61]. This selective pressure can lead to genetic adaptations and shifts in microbial composition, particularly within the human gut microbiota, potentially altering both therapeutic outcomes and side-effect profiles. Thus, while drug repurposing offers a promising strategy to combat multidrug-resistant infections, it must be pursued with careful risk-benefit assessments and a commitment to monitoring unintended microbiological consequences.
Despite its well-established clinical use in hormonal regulation, particularly in the treatment of conditions such as benign prostatic hyperplasia (BPH) and androgenetic alopecia [62, 63], finasteride has not been extensively studied for its antimicrobial or antifungal properties. Nevertheless, recent findings indicate that finasteride may exert biological effects beyond its primary hormonal targets. For instance, Chavez-Dozal et al. [59] demonstrated that finasteride can modulate the virulence of Candida albicans, suggesting interference with key fungal pathogenic mechanisms. Such findings provide a rationale for exploring the broader pharmacological potential of finasteride, particularly its role in modulating microbial phenotypes in opportunistic infections.
In our study, we observed that while finasteride did not compromise the viability of Candida and Rhodotorula even at the highest tested concentrations; however, lower concentrations (32 mg/L) significantly reduced biofilm formation. Moreover, sub-inhibitory concentrations (8 and 16 mg/L) impaired fungal filamentation and capsule formation, which correlated with a reduction in host infection potential. These observations support the hypothesis that finasteride can affect fungal virulence without exerting direct fungicidal activity. Future research will focus on evaluating the synergistic potential of finasteride in combination with established antifungal agents such as fluconazole to further understand its impact on pathogenic fungal phenotypes.
Beyond its known effects in reducing prostate gland volume (reportedly by up to 20%, although not always correlating with symptomatic improvement) [61], finasteride has shown potential in modulating bacterial infections. Notably, an intriguing study revealed its antibacterial activity against Escherichia coli, the primary causative agent of bacterial prostatitis [61]. More recent investigations have demonstrated that finasteride treatment alters the composition of the gut microbiota, including changes in the abundance of genera such as Barnesiella, Acetatifactor, Butyricimonas, Desulfovibrio, Anaerobacterium, and Robinsoniella, all of which have been implicated in BPH pathophysiology. Notably, altered levels of Desulfovibrio and Acetatifactor were linked to the promotion and inhibition of apoptosis in prostate tissue, respectively. Furthermore, the levels of beneficial bacteria such as Lactobacillus and Acetatifactor normalized following finasteride treatment. These findings suggest that finasteride-induced changes in gut microbial composition, particularly those associated with apoptosis regulation, could serve as valuable indicators for the diagnosis, prevention, and therapeutic management of BPH.
Although antidepressants and finasteride have demonstrated promising antimicrobial activity, their exact mechanisms of action remain incompletely understood. One prevailing hypothesis is that these compounds inhibit microbial efflux pumps. SSRIs, along with certain tricyclic antidepressants (TCAs), are known to target the human serotonin transporter, a Na+-dependent solute carrier protein embedded in the cell membrane with 12 predicted transmembrane domains. This transporter facilitates serotonin uptake into cells, a function that is disrupted by antidepressants. By analogy, it is believed that in microbial cells, these drugs may reduce the activity of membrane-associated proteins involved in xenobiotic transport, such as efflux pumps [7].
In Candida albicans, resistance to antifungal agents is frequently
mediated by the overexpression of efflux transporters like Cdr1, Cdr2, and Mdr1,
which help the fungus eliminate toxic compounds. Another proposed mechanism
involves mitochondrial dysfunction or disruptions to membrane integrity,
particularly in fungal pathogens. For example, neuroleptic drugs such as
aripiprazole have been shown to interfere with lanosterol
14
When considering the potential of these compounds for drug repurposing, it is essential to account for both the administered dose and the serum concentrations achieved in patients. While our in vitro results demonstrate antimicrobial effects at relatively high concentrations, these levels are substantially different from those typically reported in patient plasma. In clinical use, the reported plasma concentrations of the drugs tested in this study are markedly lower than the MIC values observed here, indicating that the antimicrobial activity seen in vitro would require concentrations far exceeding those typically achieved in patients. For example, fluconazole achieves serum levels of approximately 18 mg/L in patients treated for cryptococcosis and other fungal infections [64], while buspirone, administered at 15–20 mg/day, reaches a mean peak plasma concentration of only ~2.5 ng/mL within 1 hour of dosing [65]. Duloxetine serum concentrations range from 47–93 ng/mL depending on therapeutic response, and escitalopram reaches mean steady-state concentrations of 11–21 ng/mL under standard dosing (10–20 mg/day) [66]. Finasteride, at therapeutic dosing, is quantifiable in plasma within a range far below the MIC values observed in this study, with assay detection limits as low as 0.17 mg/L [65]. Given these disparities, it is unlikely that these compounds, at standard clinical doses, would exert direct fungicidal or fungistatic effects. However, their concentrations may still be sufficient to modulate fungal virulence factors, such as biofilm formation and filamentation, without necessarily killing the cells. This suggests potential utility in attenuating pathogenicity rather than directly inhibiting growth. To enhance translational relevance, future work will focus on testing these compounds in combination with low concentrations of traditional antifungals to assess possible synergistic effects. Such combinations may lower the required concentrations of both the repurposed drug and the conventional antimicrobial, thereby improving clinical applicability while minimizing toxicity risks.
While the antimicrobial activities of buspirone, duloxetine, escitalopram, and finasteride have not been fully characterized, their shared lipophilic chemical structures suggest a common and highly plausible primary mechanism: disruption of microbial membrane fluidity, leading to the loss of proton motive force and membrane potential. This disruption compromises cell envelope integrity, perturbs ion gradients, impairs ATP synthesis, and can increase permeability to other compounds, potentially sensitizing microbes to additional stressors or antimicrobials [7].
Beyond membrane perturbation, these compounds may also interact with nucleic acid–binding enzymes or interfere with transcriptional machinery, modulate oxidative stress response pathways, or alter quorum-sensing networks, thereby impairing the expression of key virulence factors. Another potential mechanism (particularly relevant for combination therapy) is inhibition of multidrug efflux pumps, which could enhance the intracellular retention of co-administered antimicrobials and lower the effective dose required.
For buspirone, these effects may be partly linked to its anti-inflammatory
activity, as certain anti-inflammatory agents have been shown to possess
antimicrobial and antivirulence properties [47]. Duloxetine and escitalopram,
given their structural features and high membrane-targeting potential [66], may
act primarily through envelope destabilization, but could also contribute to
efflux pump inhibition and regulatory interference. Finasteride, a synthetic
5
Future work will employ metabolomic, transcriptomic, and phenotypic analyses to systematically evaluate these hypotheses for each compound. In the case of duloxetine, escitalopram, and finasteride, as with our ongoing studies on buspirone, we will prioritize confirming membrane disruption as the main antimicrobial action while also investigating efflux pump inhibition through synergistic combination testing. These mechanistic investigations will clarify whether the primary impact of these drugs in microbial systems is direct growth inhibition, attenuation of virulence, enhancement of susceptibility to existing antimicrobials, or a combination of these effects.
While these preliminary findings are promising and suggest new directions for repurposing non-antimicrobial drugs, comprehensive studies are still needed to fully assess their safety and efficacy as antimicrobial agents. Caution is warranted, as prior research has highlighted potential drawbacks, including the risk of promoting antimicrobial resistance and disturbing the balance of the gut microbiota.
This study highlights the promising potential of drug repurposing as an alternative strategy to combat fungal and bacterial pathogens, particularly in the face of rising antimicrobial resistance. By investigating the off-target effects of clinically approved agents such as buspirone, duloxetine, escitalopram, and finasteride, we demonstrate that these compounds can significantly impair microbial virulence factors (including biofilm formation, filamentation, and host pathogenicity) at sub-inhibitory concentrations. While their direct effects on microbial viability varied, the ability to disrupt key virulence mechanisms offers a valuable therapeutic avenue that may complement existing antifungal and antibacterial treatments. Nonetheless, further research is necessary to fully elucidate their molecular mechanisms, assess potential synergistic effects with standard antimicrobials, and evaluate long-term implications such as microbiome disruption and resistance development. Our findings support the expanding scope of drug repurposing in infectious disease research and provide a foundation for future translational and clinical investigations.
All data and materials produced in the course of this study can be obtained upon reasonable request from the corresponding author.
RT conceived the original idea for the study, selected the antidepressants, and conducted viability assays, filamentation studies, nematode infection assays, and statistical analyses. SCV carried out minimum inhibitory concentration (MIC) determinations and contributed to the nematode assays. AJ performed MIC assays with bacterial strains, while KD conducted yeast growth experiments and selected MIC assays with Rhodotorula mucilaginosa. DS substantially helped with designing the research study and provided critical revisions of the manuscript and contributed expert insight into the finasteride literature. LR conducted biofilm assays with yeast strains. HC contributed substantially to the analysis and interpretation of data, and offered expert feedback, editorial input, and overall guidance. AC supervised the research, performed biofilm, MIC, and filamentation assays, authored the manuscript, and secured funding for the study. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This article does not contain any studies with human participants performed by any of the authors. Caenorhabditis elegans is a free-living, non-parasitic nematode and is not subject to animal ethics regulations that require formal protocol numbers. Therefore, no policy ethics number is applicable to the experiments described here. Although strict institutional or governmental ethical approval is not required for C. elegans research, all authors involved in this study received training in invertebrate worm culturing, maintenance, and safe disposal procedures. Throughout the study, experiments were conducted in accordance with accepted ethical principles for invertebrate research, ensuring that all handling, experimental manipulations, and disposal were performed responsibly, humanely, and with minimal environmental impact.
We gratefully acknowledge the valuable input and constructive feedback provided by the students of the BIO 111 (Research Symposium) course at Embry-Riddle Aeronautical University.
This Research was funded by the Summer Undergraduate Research Fellowship (SURF NSF grant DMS-1345499) program, supported by the Office of Undergraduate Research at Embry-Riddle Aeronautical University.
The authors declare no conflict of interest.
During the development of this manuscript, the authors utilized the ChatGPT-4 model, specifically the GPT-4o architecture, as a tool to assist with refining the text. This included tasks such as identifying and reducing redundancy, correcting grammar and spelling, and enhancing overall clarity. Following the use of this AI-based assistance, the authors thoroughly reviewed and manually edited the content to ensure accuracy, coherence, and alignment with the scientific intent of the work. The authors accept full responsibility for the final version of the manuscript and its contents.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





