1 Nitric Oxide Services, LLC, Akron, OH 44312, USA
2 Bauer Research Foundation, Inc., Akron, OH 44312, USA
Abstract
Nitrosylcobalamin (NO-Cbl) is a vitamin B12 analog designed to exploit the “Trojan horse” vulnerability created by the heightened need of cancer cells for cobalamin and one-carbon metabolism. Building on our recent biophysical studies confirming the affinity of NO-Cbl for intrinsic factor, this work aimed to investigate the mechanistic basis for the selective anticancer activity of NO-Cbl through the cobalamin transport axis and lysosomal processing.
Human cancer cell lines (NIH-OVCAR-3, MCF-7, WM9, and DU145) were cultured and transfected to overexpress transcobalamin II (TCII). Cell proliferation and cytotoxicity were measured using the sulforhodamine B (SRB) assay. TCII-R (CD320) expression was quantified by flow cytometry. The impact of anti-CD320 antiserum and lysosomal alkalization (chloroquine) on NO-Cbl activity was assessed.
Antiserum inhibition of the TCII receptor resulted in dose-dependent inhibition of NIH-OVCAR-3 and MCF-7 cell proliferation. Lysosomal alkalinization by chloroquine pretreatment abrogated NO-Cbl-induced cytotoxicity in OVCAR-3 cells. Flow cytometric analysis demonstrated an inverse correlation between TCII-R (CD320) expression (MFI ratio) and NO-Cbl ID50. TCII overexpression significantly reduced NO-Cbl ID50 in NIH-OVCAR-3 cells.
NO-Cbl utilizes tumor cell cobalamin transport and processing pathways to deliver nitric oxide selectively to cancer cells. These results, integrated with recent binding studies, validate NO-Cbl as a cobalamin-based targeted anticancer agent with efficacy in tumors expressing high levels of TCII and CD320.
Keywords
- nitrosylcobalamin
- cobalamin
- transcobalamin
- TCII
- CD320
- lysosome
- cancer
The pivotal role of vitamin B12 (cobalamin, Cbl) in human health was first highlighted in 1926 when Minot and Murphy demonstrated that a liver-rich diet could effectively treat pernicious anemia, suggesting the presence of a specific therapeutic agent in liver tissue [1]. Subsequent research led to the isolation and characterization of this “anti-pernicious anemia” factor. By 1948, Karl Folkers and colleagues achieved the further purification of vitamin B12, identifying it as a cyano-cobalt coordination complex, subsequently named cyanocobalamin [2].
Early modifications of vitamin B12 involved substituting the cyano group with various ligands, resulting in derivatives such as hydroxo-, chloro-, bromo-, sulfato-, nitro-, and cyano-cobalamin [3]. These studies laid the groundwork for the concept of modifying vitamin B12 to create biologically active analogs. The rationale behind this approach is supported by numerous studies demonstrating that cobalamin is essential for maintaining cellular differentiation, proliferation, and metabolic functions [4]. Therefore, derivatization of cobalamin could yield pharmacologically effective analogs [5].
Cancer cells, by virtue of their relentless proliferation and metabolic rewiring, exhibit a dramatically increased demand for cobalamin and one-carbon metabolism [6, 7, 8]. Uptake and intracellular processing of cobalamin are orchestrated by a highly specific molecular relay: absorption with intrinsic factor, plasma transport via transcobalamin II (TCII), cellular internalization through the TCII receptor (CD320), and trafficking into lysosomal compartments for cobalamin release and utilization [9, 10, 11, 12, 13].
This cobalamin-centric metabolic axis presents a unique vulnerability, a “gateway”, that can be exploited for therapeutic purposes. Tumors upregulate TCII, CD320, and downstream one-carbon metabolic pathways, increasing their reliance on cobalamin for DNA synthesis, methylation, and redox homeostasis [14, 15]. Thus, cobalamin-dependent malignancies represent ideal targets for “Trojan horse” strategies that masquerade as cobalamin, infiltrating cancer cells through their own transport machinery.
Nitrosylcobalamin (NO-Cbl), a nitric oxide–modified cobalamin analog, is designed to capitalize on one carbon metabolism in cancer cells [16]. Our recent biophysical studies established that NO-Cbl maintains high-affinity binding to intrinsic factor, indicating preservation of key recognition sites for physiological cobalamin transport proteins [17]. This enabled NO-Cbl to mimic natural B12 culminating in the selective delivery and release of cytotoxic nitric oxide within tumor cells [18].
The specific aims of this study were to assess possible mechanisms associated with cobalamin uptake, specifically NO-Cbl, in human cancer cell lines. Our goal was to assess the functional blockade of TCII-mediated uptake along with the overexpression of TCII and the effects of lysosomal processing to encourage continued research into one-carbon metabolism as potential exploits for future anticancer treatments.
Nitrosylcobalamin was synthesized as previously described [16]. Hydroxocobalamin (vitamin B12a) acetate (Hebei Huarong Pharmaceutical Co. Ltd., Shijiazhuang, Hebei, China) was dissolved in dichloromethane (OmniSolv, EMD Chemicals, Gibbstown, NJ, USA) and exposed to CP grade NO gas (Praxair, Bethlehem, PA, USA) at 150 PSIG (1034 kPa). The reaction proceeded in a closed system within a high-pressure stainless-steel reactor (Parr Instrument Co., Moline, IL, USA). The system was purged daily and evacuated prior to NO exposure. The NO gas was scrubbed prior to entering the system using a stainless-steel cylinder (Midwest Process Controls, Bay Village, OH, USA) containing NaOH pellets. The solid NO-Cbl product was collected following rotary evaporation of the solvent and stored under argon at –80 °C prior to use.
Human tumor cell lines: HTB-161, HTB-22, TIB-152 (ATCC, Manassas, VA, USA) and
WM9-01-0001 (Rockland Immunochemicals, Inc. Limerick, PA, USA) were grown in
RPMI 1640 and Dulbecco’s modified Eagle medium (DMEM, Cellgro, Mediatech,
Herndon, VA, USA) as appropriate, both supplemented with 10% fetal bovine serum
(FBS, Mediatech) and 1% Antibiotic-Antimycotic (GIBCO BRL, Invitrogen, Carlsbad,
CA, USA). Cells were maintained in 5% CO2 at 37 °C in a humidified
incubator. Cells were confirmed to be mycoplasma free using a
MycoAlert
TCII cDNA (a gift from Dr. Rothenberg, SUNY Brooklyn, NY, USA) [19, 20] was sub-cloned into the pCXN2myc mammalian expression vector using XhoI and BstXI restriction sites. Cells were transfected using Lipofectamine Plus Reagent (Invitrogen); GFP was used as a positive control. Chloroquine diphosphate salt was purchased from MP Biomedicals, Inc. (Solon, OH, USA).
Cells were harvested with 0.5% trypsin/0.53 mM EDTA, washed with PBS and resuspended in media containing 10% FBS. Cells were plated in 96-well plates in 0.2 mL aliquots containing 2000 cells. Cells were allowed to adhere to the plate for 4 h and then NO-Cbl was added in different dilutions to the assay plate. Replicates of eight were performed for each treatment. Growth was monitored using a modified sulforhodamine B (SRB, Sigma Chemical, St. Louis, MO, USA) colorimetric assay [21, 22].
After 96 h growth, the medium was removed, and cells were fixed with 10% trichloroacetic acid and stained with SRB. Bound dye was eluted from cells with 10 mM TRIS (pH 10.5) and absorbance was measured at 570 nm using a Lab Systems Multiskan RC 96-well plate reader (Lab Systems Multiscan RC, Thermo Lab Systems, Franklin, MA, USA).
To quantify growth of the cells, experimental absorbance values (Aexp ) were
compared with initial absorbance readings representing the starting cell numbers
(Aini). To determine the starting cell number, an additional 96-well plate
was seeded with cells and fixed at the beginning of the experiment. The
absorbances derived from the initial plate and from the untreated cells at the
end of the growth period (Afin) were defined as 0% and 100% growth,
respectively. The percentage control growth (100%
Cells were treated with increasing dilutions of anti-TCIIR (CD320) antiserum [23] (1:40 to 1:2000; a gift from Dr. Bellur Seetharam) for 72 hours to assess effects on proliferation. For lysosomal alkalization, cells were pretreated with chloroquine (20 µM) for 30 min prior to NO-Cbl exposure and quantified by SRB.
Trypsinized cells (250,000) were resuspended in PBS pH 7.4 and incubated with
rabbit anti-human TCII-R polyclonal antibody 1:20 (a gift from Dr. Bellur
Seetharam) [23] on ice for 30 min. Cells were washed three times and incubated
with Goat Anti-Rabbit IgG (H+L) Antibody (Alexa Fluor
All data are expressed as mean
Treatment of NIH-OVCAR-3, MCF-7, and WM9 cell lines with serial dilutions of anti-TCII-R (CD320) antiserum (1:40 to 1:2000) for 72 hours resulted in a marked, concentration-dependent inhibition of cell proliferation as measured by the SRB assay (Fig. 1). The greatest inhibition was observed at the highest antibody concentrations (lowest dilution), with OVCAR-3 and MCF-7 exhibiting the most pronounced response.
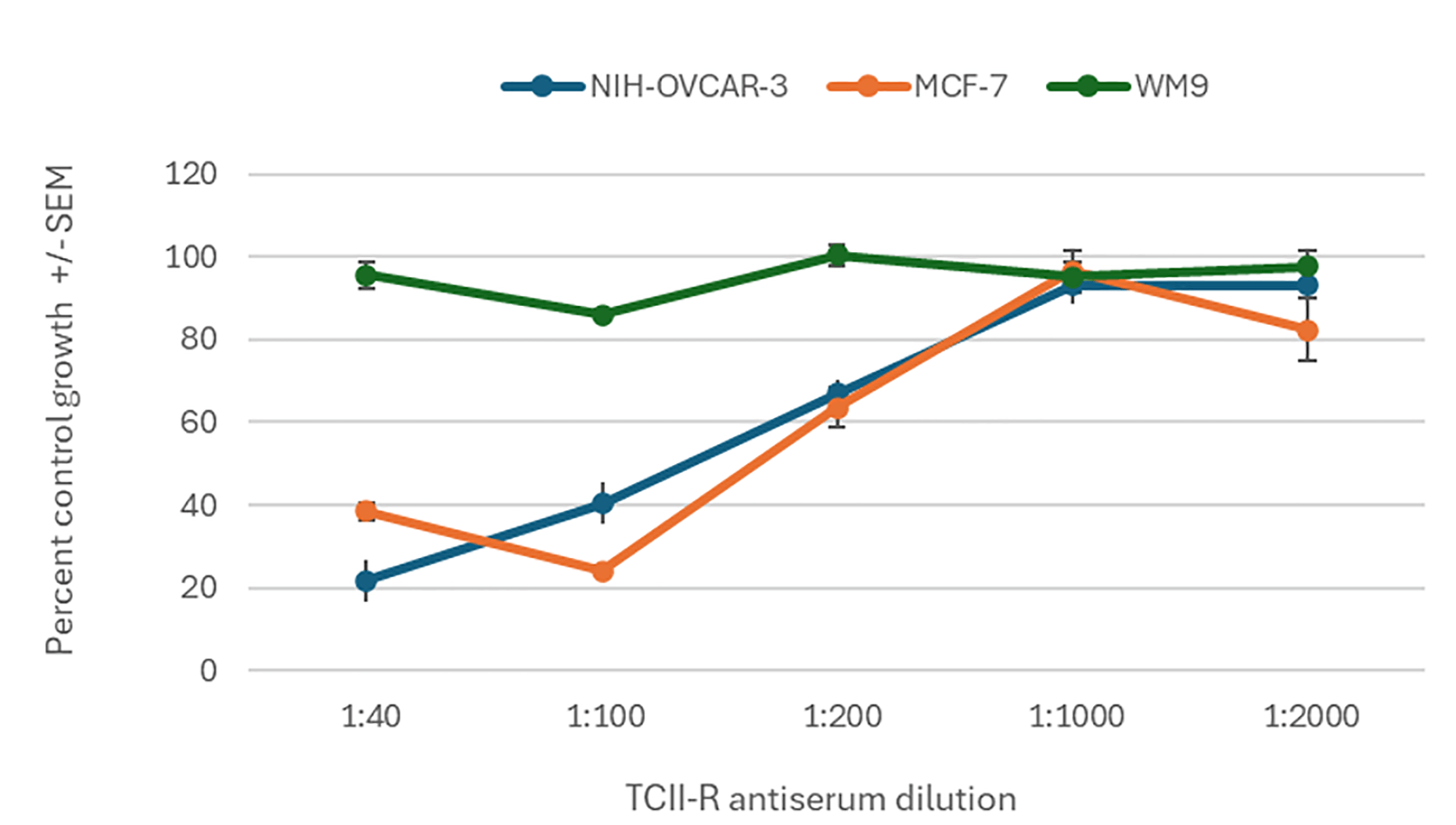 Fig. 1.
Fig. 1.
Effect of TCII-R antiserum on percent control growth in
NIH-OVCAR-3, MCF-7, and WM9 cell lines. Cells were treated for
72 hours with serial dilutions of anti-TCII-R (CD320) antiserum (1:40 to 1:2000),
and cell proliferation was measured by the SRB assay. Percent control growth
(mean
To investigate the role of lysosomal processing in NO-Cbl cytotoxicity, OVCAR-3
cells were pretreated with chloroquine (CQ, 20 µM) for 30 minutes, followed
by pulsed NO-Cbl exposure (3, 4, or 5 hours). After 96 hours, cell growth was
assessed by the SRB assay (Fig. 2). Chloroquine pretreatment significantly
protected against NO-Cbl-induced cell death at all time points, with percent
control growth restored to baseline levels in CQ+NO-Cbl groups compared to robust
cytotoxicity from NO-Cbl alone (p
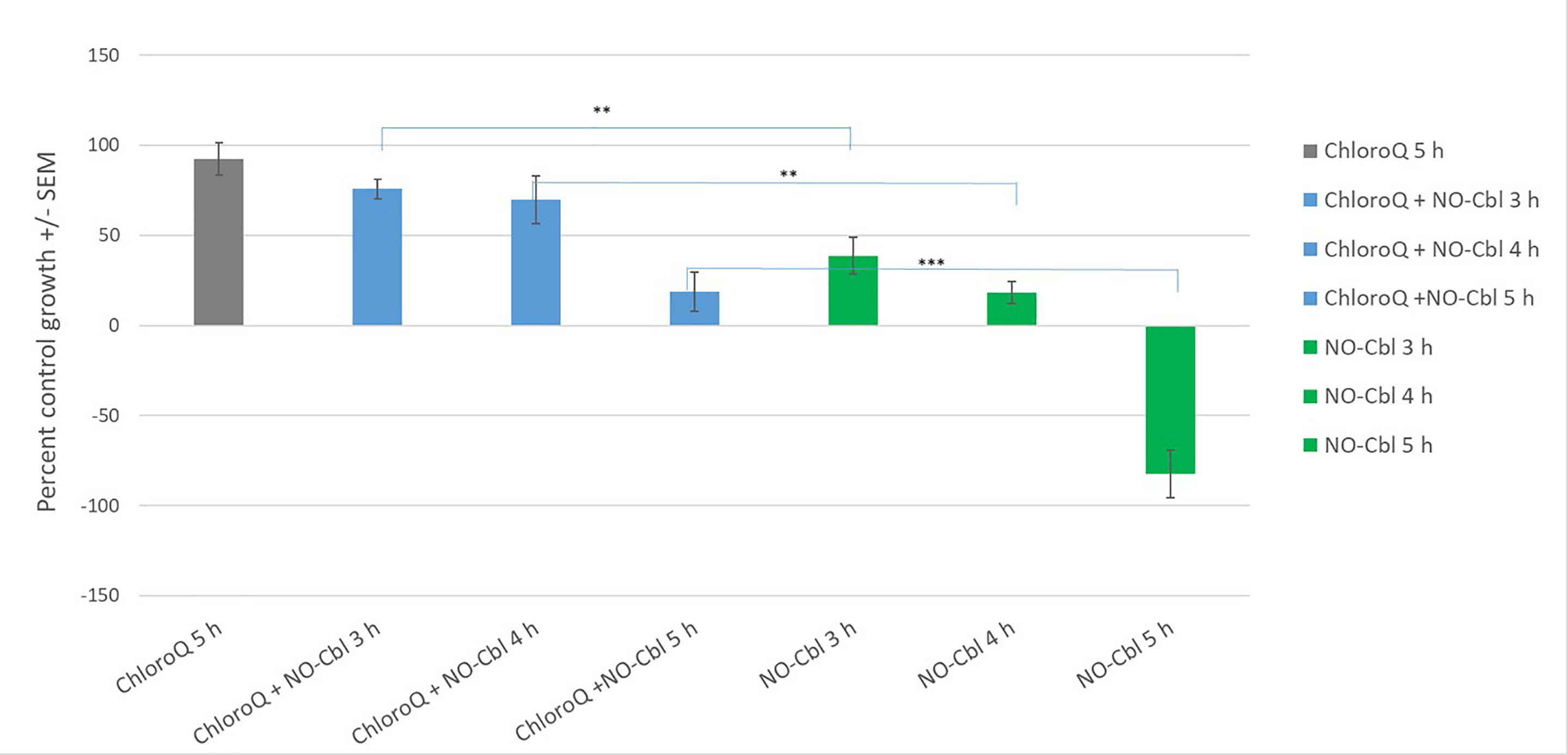 Fig. 2.
Fig. 2.
Chloroquine protects OVCAR-3 cells from NO-Cbl–mediated
cytotoxicity. Percent control growth (mean
Flow cytometric analysis of Jurkat, NIH-OVCAR-3, MCF-7, and WM9 cells revealed variable expression of TCII-R (CD320), measured as the ratio of mean fluorescence intensity (MFI) for primary + secondary antibody versus secondary antibody alone. Representative histograms are shown for each cell line (Fig. 3A–D). Quantitative analysis (Fig. 3E) demonstrated a negative correlation between TCII-R relative fluorescence (MFI ratio) and NO-Cbl ID50 values (Pearson r = –0.92, p = 0.0779). Specifically, cell lines with higher TCII-R expression were more sensitive to NO-Cbl-induced growth inhibition.
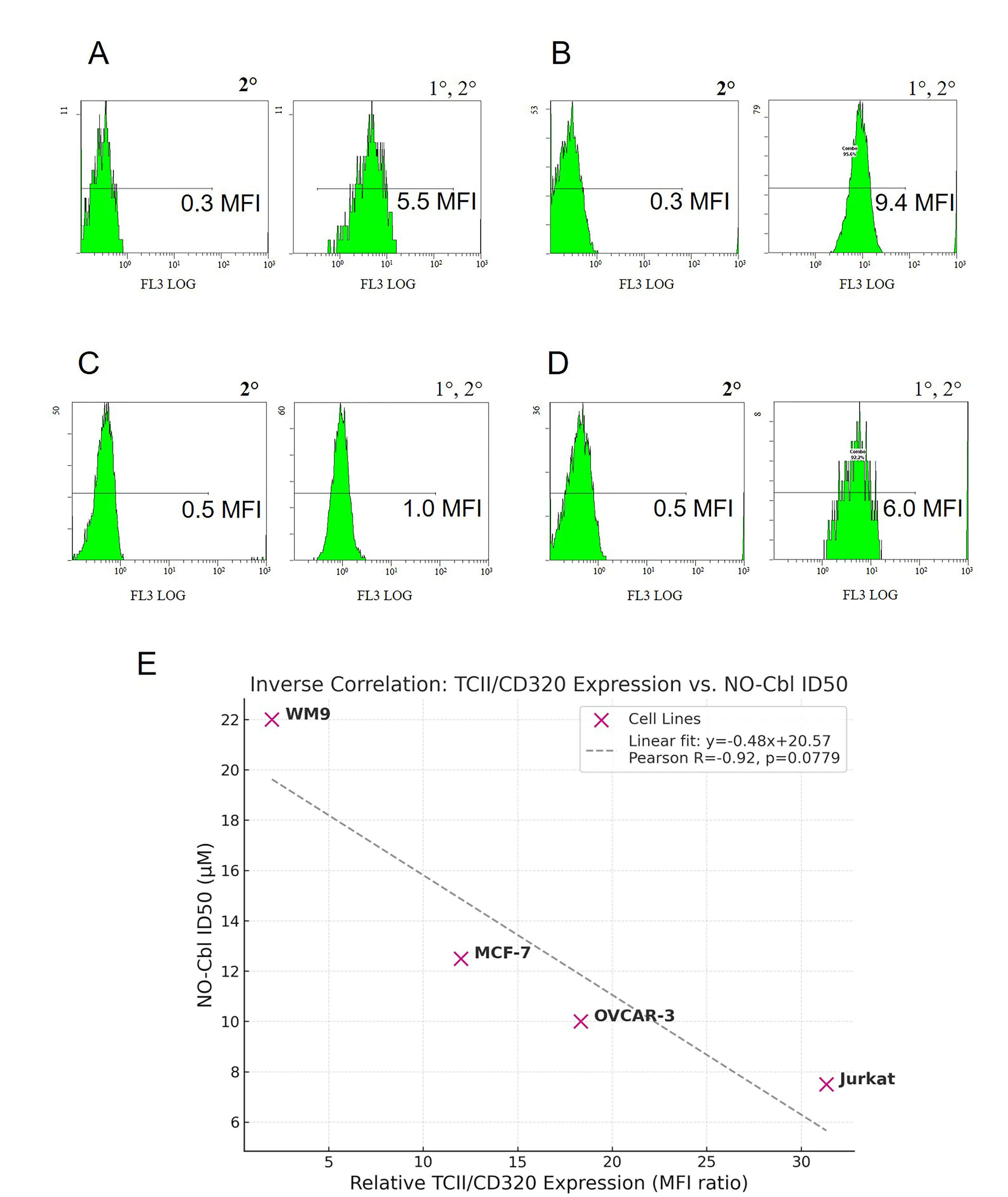 Fig. 3.
Fig. 3.
Relationship between relative TCII-R (CD320) expression and NO-Cbl sensitivity in human cancer cell lines. (A–D) Representative flow cytometry histograms for OVCAR-3 (A), Jurkat (B), WM9 (C) and MCF-7 (D) cells stained with secondary antibody only (left) or with both anti-CD320 primary and secondary antibody (right). (E) Correlation between relative TCII-R expression and NO-Cbl ID50 values for the four cell lines. Pearson’s r = –0.92, p = 0.0779. Higher TCII-R relative fluorescence is associated with greater NO-Cbl sensitivity. Relative MFI was calculated as the mean fluorescence intensity (MFI) of the CD320-stained sample divided by the MFI of the secondary-only control.
To directly test the functional consequence of increased TCII expression, SRB dose–response assays were performed in wild-type and TCII-transfected OVCAR-3, WM9 and DU145 cells (Fig. 4). Overexpression of TCII increased sensitivity to NO-Cbl, as evidenced by a substantial reduction in ID50 values across cell lines.
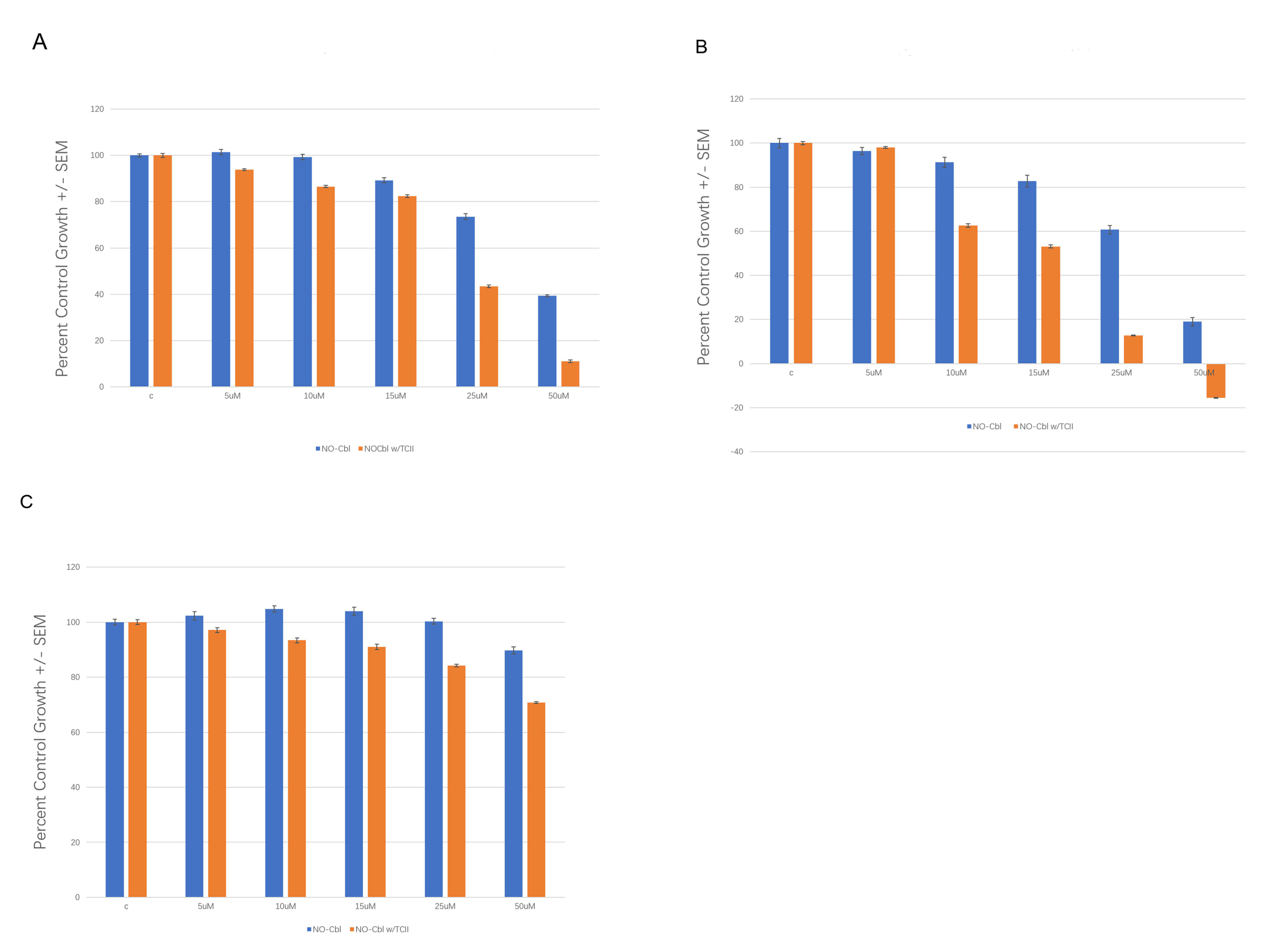 Fig. 4.
Fig. 4.
Sensitization to NO-Cbl by TCII Overexpression in Human Cancer Cell Lines. An SRB assay dose–response using NO-Cbl was performed in TCII transfected cells compared to wild type. (A) OVCAR-3. (B) WM9. (C) DU145.
This study demonstrates that nitrosylcobalamin (NO-Cbl) leverages the native cobalamin transport and processing machinery—particularly transcobalamin II (TCII) and its receptor (TCII-R/CD320)—to achieve potent and selective antitumor activity, confirming the “Trojan horse” hypothesis. Various components of the cobalamin uptake and utilization pathway, long understood for its essentiality in normal cell function and disease [9, 24] is revealed here as a mechanistic determinant for NO-Cbl sensitivity in tumor cells.
Our first line of evidence comes from antibody-mediated blockade of TCII-R (Fig. 1), where we observed a robust, dilution-dependent inhibition of cell proliferation in NIH-OVCAR-3 and MCF-7 cell lines. WM9 cells were unresponsive which is consistent with non-TCII mechanisms associated with cobalamin uptake by melanoma [25]. These findings are consistent with earlier studies demonstrating that antibody or genetic disruption of the cobalamin uptake machinery inhibits tumor cell growth and DNA synthesis [9], and further support the notion that tumor dependence on TCII-mediated B12 import can be therapeutically exploited. Notably, our findings expand on work by others that show TCII-R blockade limits vitamin B12 utilization [26, 27] by demonstrating the same vulnerability with an engineered cobalamin analog.
Secondly, our lysosomal alkalinization experiments (Fig. 2) demonstrate the
effects of chloroquine pretreatment, by neutralizing the acidic environment of
the lysosome, to protect OVCAR-3 cells from NO-Cbl-induced cytotoxicity at all
pulse durations. For example, NO-Cbl alone caused a pulse-duration-dependent
decrease in cell growth (to –82.5% at 5 hours), while chloroquine plus NO-Cbl
resulted in protection (69.8–92.4% growth across all time points, p
Third, flow cytometric analysis (Fig. 3) revealed a negative correlation between TCII-R expression (measured as MFI ratio) and NO-Cbl ID50 across four cell lines (Pearson r = –0.92). Jurkat cells (high TCII-R) were most sensitive (ID50 = 7.5 µM), while WM9 (lowest TCII-R) were most resistant (ID50 = 22 µM). This aligns with recent findings in both clinical and preclinical studies, where CD320 (TCII-R) expression is upregulated in proliferative tissues and in a wide variety of cancers, correlating with cobalamin demand and malignant potential [7, 29, 30].
Finally, our TCII transfection experiments (Fig. 4) show that expression of TCII in OVCAR-3, WM9, and DU145 cells reduced the NO-Cbl ID50 (the concentration needed to inhibit cell growth by 50%). This is in agreement with animal and tissue studies that show TCII upregulation in tumors and in endothelial cells within the tumor microenvironment [14].
Taken together, our data support a model in which NO-Cbl exploits the cobalamin
pathway to selectively target tumor cells. This “Trojan horse” mechanism
requires that an upper-axial (
When considering clinical translation, it is important to note that normal human plasma cobalamin concentrations are in the nanomolar to picomolar range and rarely exceed ~10 µM even with high-dose parenteral therapy. While our prior murine xenograft study [18] demonstrated no histologic evidence of toxicity after 54 days of daily dosing at 100 mg/kg, and our long-term canine case series [43] tracked up to 61 months of daily NO-Cbl treatment without clinically significant adverse effects, these supraphysiologic exposures may not fully predict human tolerance. Careful, stepwise dose-escalation studies will therefore be required to define the maximum tolerated dose (MTD) and confirm safety under physiological conditions.
Our work helps elucidate the mechanism of NO-Cbl action in cancer suggesting new opportunities for biomarker-driven, precision therapies using vitamin B12 analogs. Future directions include the exploration of biomarkers of cobalamin pathway activity to guide patient selection, combination approaches with lysosome-targeting agents, and investigation of resistance mechanisms. Future clinical development should incorporate careful dose-finding studies to establish safety parameters in humans, recognizing that physiological plasma cobalamin levels differ substantially from those achieved in experimental systems. These steps will be essential to translate the favorable preclinical profile into a safe and effective therapeutic strategy.
The data sets used and analyzed during the current study are available from the corresponding author upon request.
JAB designed the research study, conducted experiments, and performed statistical analysis. JAB and AMS wrote and edited the manuscript, analyzed the data and created the figures. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and have agreed to be accountable for all aspects of the work.
Not applicable.
The authors wish to express their deepest gratitude to Dr. Daniel J. Lindner, MD, PhD, for his invaluable support and mentorship throughout the course of this research, and to Dr. Bei H. Morrison, MD, for her contributions and expertise in the TCII experiments—both of the Taussig Cancer Institute, Center for Hematology and Oncology Molecular Therapeutics, Cleveland Clinic, Cleveland, Ohio. We extend our heartfelt thanks to Dr. Bellur Seetharam, Division of Gastroenterology and Hepatology, Departments of Medicine and Biochemistry, Medical College of Wisconsin and Zablocki Veterans Administration Medical Center, Milwaukee, Wisconsin, for his guidance, enthusiastic collaboration, and enduring encouragement, especially in advancing our CD320 studies and fostering a shared love of vitamin B12 research. His inspiration and generosity have profoundly shaped the field. We also gratefully acknowledge Dr. Rothenberg (SUNY Downstate Health Sciences University, Brooklyn, NY) for providing the TCII construct used in these experiments.
This research received no external funding.
JAB is employed by Nitric Oxide Services, LLC. AMS is affiliated with the Bauer Research Foundation, Inc. However, the judgments in data interpretation and manuscript writing were not influenced by these affiliations. Both authors declare no conflicts of interest.
ChatGPT 4o was used as an editing tool to create a more concise structure. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




