1 Molecular Genetics and Microbiomics Laboratory, BIOTROF+ Ltd., 196602 Pushkin, St. Petersburg, Russia
2 Federal State Budgetary Educational Institution of Higher Education “St. Petersburg State Agrarian University”, 196605 Pushkin, St. Petersburg, Russia
3 Federal State Budgetary Educational Institution of Higher Education “St. Petersburg State University of Veterinary Medicine”, 196084 St. Petersburg, Russia
4 All-Russian Research Institute for Agricultural Microbiology, 196608 Pushkin, St. Petersburg, Russia
5 School of Natural Sciences, University of Kent, Canterbury, CT2 7NJ Kent, UK
6 Animal Genomics and Bioresource Research Unit (AGB Research Unit), Faculty of Science, Kasetsart University, 10900 Chatuchak, Bangkok, Thailand
7 L. K. Ernst Federal Research Center for Animal Husbandry, Dubrovitsy, 142132 Podolsk, Moscow Oblast, Russia
Abstract
Significant alterations in feeding, housing, and physiology are observed in dairy cows during the transition period (3 weeks pre- and post-calving), in addition to changes in the composition and abundance of the endometrial microbiota. Thus, this study aimed to evaluate any changes in the composition and predicted metabolic pathways in the cow uterine microbiome during this transition period.
Scrapings were sampled from the endometrial surface of clinically healthy cows (n = 3) in dynamics as follows: in the 10 Days period before, and on Days 3, 5, and 20 after calving. Total DNA was isolated from the samples, and the composition of the microbial community was assessed using targeted next-generation sequencing (NGS) technology. Based on the subsequent NGS data, the dynamics of the predicted metabolic pathways of the microbiota were evaluated.
Seven superphyla and phyla of microorganisms were found in the endometrial microbiota of cows during the transition period. Among these, the phylum Firmicutes (with a dominant class of Clostridia) and the superphylum Fusobacteriota (represented by a single class of Fusobacteriia) can be considered the dominant bacteria in the endometrium, with representation noted from 25.2 to 68.2% and from 12.3 to 51.1%, respectively. The microbiome composition underwent significant changes (p < 0.05) during the transition period. In particular, the high abundance of the Fusobacteriaceae family (up to 68.2%) in the uterus of clinically healthy cows was unexpected, given the potential association of Fusobacteriaceae with the occurrence of metritis in cows. The numbers of microorganisms in two dominant classes, Fusobacteriia and Clostridia, showed generally opposite changes in their relative abundance during the transition period. The predicted functional potential level for 32 pathways in the endometrium changed (p < 0.05) in cows during the transition period. Indeed, the activity of the predicted pathways, such as pyridoxal 5′-phosphate biosynthesis I and teichoic acid (poly-glycerol) biosynthesis, was lowered on day 3 postpartum (p < 0.05).
Microbiota composition and the activity of the predicted metabolic pathways in the cow endometrium underwent significant changes at different critical stages in the transition period. Moreover, even clinically healthy cows exhibited signs of dysbiotic disorders.
Keywords
- Bos taurus
- high-yielding dairy cows
- microbiota
- endometrium
- metabolic pathways
- next-generation sequencing technology
- transition period
Accurate characterization of the dynamics of human and animal microbiomes is
crucial for understanding and predicting the impact of microbial communities on
health and physiological status [1, 2, 3, 4]. For example, uterine inflammatory
abnormalities and reproductive performance in cows (Bos taurus) are
closely related to the composition of the microbiota [5, 6]. Dysbiosis is associated
with the occurrence of endometritis has been shown to be typically characterized
by lower
In recent years, selection for milk productivity and the intensification of animal husbandry has led to a decline in the fertility of dairy cows and an increase in the incidence of postpartum endometritis [8]. Infertility caused by postpartum endometriosis is one of the reasons for culling otherwise healthy animals, however, cows are known to have low levels of lactobacilli in the reproductive tract, and the concept of “normal microbiota” in the vagina and uterus has not been established for them [9]. Thus, studying the microbial ecology of the endometrium of healthy animals is important to identify representatives of microbial populations that ensure good health and fertility [10].
It is well established that, in dairy cattle breeding, the period between 3 weeks before, and 3 weeks after, calving (i.e., the transition period) is one of the most critical stages for animal physiology since most metabolic and infectious diseases occur during this period [11]. Higher energy and nutrient requirements for colostrum and milk synthesis, combined with decreased feed intake, made cows to experience negative energy balance and micronutrient deficiency. During the transition period, cows’ bodies undergo significant metabolic, immunological, endocrine and reproductive changes [12]. As cows’ feed intake naturally declines due to fetal growth, their energy deficit increases and becomes critical on Days 5–10 before the expected calving [13]. In addition to significant changes that are observed in feeding, housing and physiology of animals, changes in the structure, composition and quantity of their endometrial microbiota are also apparent. The composition of the reproductive system microbiome during the transition period is an important factor determining subsequent productivity and reproductive capacity of dairy animals. Usually, endometritis can be suspected on Days 5 to 7 postpartum based on thermometry and other clinical signs. The diagnosis is confirmed, however, when mucopurulent discharge is observed in the postparturient cows within 20–23 days after calving and the cervix is thickened.
Using humans as an example, it has been shown that the normal microbiota of the genital tract has a number of physiological functions that have a positive effect on the host organism, such as maintaining the integrity of the endometrial epithelium and regulating immunity [14]. In turn, the host organism provides optimal conditions for the growth and reproduction of normal microbiota. For example, lactobacilli use genital tract secretions, such as mucin carbohydrates, as a food source. Hormonal fluctuations affect the microbial population of the genital tract mucosa. Thus, estrogen stimulates the deposition of glycogen in the vaginal epithelial tissue, providing a valuable source of nutrients for the microbiota. It is interesting to study the effect of the cows’ reproductive system microbiome on the functions of the body due to changes in its own metabolic profile. In recent years, predicted metabolic pathways of the microbiome in the rumen of cows have been studied using metagenomic methods [15]. Studies on the predicted metabolic pathways of the vaginal and endometrial microbiome of cows are still, however, rare [16].
During the transition period of cow husbandry, the uterine ecosystem thus represents the most dynamic environment in which microorganisms can have a pronounced effect on the physiology of the host. The physiological status of the host, in turn, can significantly affect the composition and function of the endometrial microbiota [17]. Understanding this interaction is key to maintaining the reproductive status of animals. The aim of the present study was therefore to elucidate and evaluate changes in the composition and predicted metabolic pathways of the uterine microbiome during the transition period of cow maintenance using molecular genetic and metagenomic technologies. For this purpose, we employed targeted next-generation sequencing (NGS) of the 16S rRNA gene [18, 19, 20, 21, 22], an approach that has been the widely used technology in recent years for researching the microbiome of the bovine reproductive tract [16, 23, 24].
An experiment was conducted in 2024 on the commercial livestock enterprise SPK Polyany (Vyborg District, Leningrad Oblast, Russia) to analyze the endometrial microbiome in the transition period in Holstein cows kept on a tether. The live weight of the cows was 580–600 kg in the late dry period and 510–530 kg in the fresh period, and the milk yield for 305 days of the previous lactation was 10,000–11,000 kg. The feeding and housing conditions of the cows corresponded to generally accepted recommendations [25]. The cows’ rations are presented in Table 1.
| Diet component, kg/cow/day | Dry period | Fresh period |
| Vitamin and mineral premix for dry cows | 0.10 | – |
| Vitamin and mineral premix for fresh cows | – | 0.10 |
| Hay | 3.50 | 1.20 |
| Silage | 27.00 | 22.00 |
| Rapeseed cake | 0.90 | 2.20 |
| Sunflower meal | – | 1.00 |
| Corn | 0.30 | 3.60 |
| Barley | 0.50 | 3.00 |
| Soy coat | 1.20 | 0.60 |
| Brewer’s grains | – | 2.00 |
| Sugar syrup | 0.25 | 0.40 |
| Sodium bicarbonate | – | 0.20 |
| Sodium chloride | – | 0.10 |
| Chalk | – | 0.18 |
During the transition period, a gynecological veterinary examination of the animals was carried out as described elsewhere [26], on the basis of which clinically healthy cows were selected that had no complications during and after calving and no signs of postpartum endometritis or other inflammatory disorders of the reproductive system. Additional criteria for choosing cows for the current study were no use of antibiotics treatment and hormonal drugs or other drugs and probiotics.
Blood and endometrial samples were collected from clinically healthy cows
(n = 3) dynamically, i.e., on Day 10 before calving and on Days 3, 5 and
20 after calving. Blood tests were performed as they are recommended for
assessing the health and physiological status of dairy cattle [27, 28, 29]. Before
the morning feed, blood was collected from the subcaudal vein of the cows of each
group using vacutainers and immediately delivered to the laboratory for analysis
of hematological and biochemical parameters. The innate immunity (also known as
non-specific blood resistance) level of the cows was assessed dynamically during
the experiment. Innate immunity is a multi-component system of host defense that
operates independently of prior exposure to an antigen. In cows, as in other
mammals, innate immunity plays a key role in maintaining homeostasis, protecting
against infections, and ensuring optimal productivity. Assessing innate immunity
levels is an important tool for monitoring animal health, identifying potential
problems, and developing strategies to improve disease resilience [30, 31, 32, 33, 34, 35]. The
number of erythrocytes and leukocytes was determined using a counting chamber
with a Goryaev grid according to a generally accepted method [36]. The leukogram
was calculated using microscopy with a fixative according to May-Grunwald and
azure-eosin dye according to Romanovsky for staining blood smears [36]. The
hemoglobin level was evaluated using the hemoglobin cyanide method [36] and the
hematocrit level using the centrifugation technique [37]. The color index (CI) of
cow blood was calculated using the following formula: CI = (Hemoglobin (in g/L) /
Count of erythrocytes (in
Endometrial surface scraping was carried out under aseptic conditions using a cytobrush. For this, the animals’ tails were covered with sterile gauze, and the perineum and vulva were washed with soap and water until completely clean and then treated with an antiseptic solution. Samples for microbiota analysis were immediately frozen at –20 °C and sent to the laboratory until examination.
Total DNA from the endometrial microbiota samples was isolated using the Genomic DNA Purification Kit (lot No. 10571031; Thermo Fisher Scientific, Inc., Carlsbad, CA, USA) and according to the manufacturer’s instructions. The composition of the microbial community was evaluated using targeted NGS technology. NGS was performed using the MiSeq platform (Illumina, Inc., San Diego, CA, USA) and the following primers for the V3-V4 region of the 16S rRNA gene: forward primer, 5′-TCGTCGGCAGCGTCAGATGTGTATAAGAGACAGCCTACGGGNGGCWGCAG-5′; reverse primer, 5′-GTCTCGTGGGCTCGGAGATGTGTATAAGAGACAGGACTACHVGGGTATCTAATCC-3′. Metagenomic sequencing was performed using the following reagents: Nextera® XT Index Kit (lot No. FC-131-1001; Illumina Inc.) for library preparation, Agencourt AMPure XP PCR (lot No. A63881; Beckman Coulter Inc., Brea, CA, USA) for product purification, and MiSeq® Reagent Kit v2 (500 cycles; lot No. MS-102-2003; Illumina Inc.) for sequencing.
The dynamics of predicted metabolic pathways in the microbiota were assessed
based on NGS data using the QIIME 2 (version 2020.8;
https://forum.qiime2.org/t/recommendations-for-installing-older-versions-of-qiime-2/28956
(Accessed: 31 March 2025)) metagenomic analysis platform [40, 41]. After the
initial transfer of sequences to the Qiime2 format, paired read lines were
aligned. Then, they were chosen by quality using the default settings. Noise
sequences were filtered using the Deblur method [42]. The MAFFT (version 7;
https://mafft.cbrc.jp/alignment/software/ (Accessed: 31 March 2025)) software
package was used to reconstruct a de novo phylogeny, followed by masked
read alignment [43, 44]. The SILVA 138
(https://www.arb-silva.de/documentation/release-138/ (Accessed: 31 March 2025))
reference database [18, 45, 46] was used to analyze the taxonomic affiliation of
microorganisms. Indices of
Using Microsoft Excel XP/2003 and RStudio (version 1.1.453;
https://web.archive.org/web/20220806090006/https://docs.rstudio.com/ide/server-pro/1.1.453/index.html
(Accessed: 31 March 2025)) [55], including multivariate analysis of variance
(multi-factor ANOVA), the results were processed statistically and
mathematically. Results involved the means (M) and standard errors of the mean
(
Hematological and biochemical parameters of the cows’ blood during the
transition period are presented in Supplementary Tables 1–8. Analysis
of the leukogram and biochemical parameters of the blood characterizing nitrogen
metabolism, pigment metabolism, acid-base balance (Supplementary Tables
2,7,8) did not reveal any significant deviations indicating a
possible disturbance of the general physiological state of the cows during the
transition period based on the parameters studied. Nevertheless, the number of
leukocytes on Day 10 before calving (8.04
The endometrial microbiota can influence the host’s physiology but can also be impacted by it. We thus assessed physiological characteristics using hematological and biochemical blood parameters, since changes in them in birds and mammals are important indicators reflecting the state of the internal environment of the body, especially under the influence of external factors [67]. Blood analysis allows to elucidate indirectly the degree of immunological protection, metabolic rate, the presence of inflammatory processes, and determine the duration of these processes [68]. The alterations in blood parameter we observed here may suggest an increased load on the liver during the studied periods, which is probably associated with the stress of metabolic processes during the transition period, as well as the state of negative energy balance caused by high-concentrate feeding adopted in industrial farm conditions.
NGS of the bovine endometrial microbiome performed on all animals in this study generated a total of 403,010 16S rRNA gene sequences (with a median of 33,694 reads, a minimum of 21,259 and a maximum of 43,242 reads). The raw 16S rRNA sequence data were deposited into the NCBI Sequence Read Archive (SRA; BioProject Number: PRJNA1230390, https://www.ncbi.nlm.nih.gov/bioproject/PRJNA1230390 (Accessed: 31 March 2025)).
Based on the values of the Chao1 and Fisher’s
| Biodiversity indices and ASV | Transition period stages | |||
| 10 days before calving | 3 days postpartum | 5 days postpartum | 20 days postpartum | |
| Chao1 | 117.70 |
63.37 |
112.70 |
88.50 |
| Fisher’s |
15.80 |
7.40 |
15.20 |
10.90 |
| Shannon entropy | 4.10 |
4.20 |
4.70 |
4.50 |
| Simpson | 0.88 |
0.93 |
0.94 |
0.94 |
| ASV (feature count) | 31,457.00 |
41,280.00 |
26,113.00 |
35,485.00 |
1 n, number of cows studied per each stage; M
Our findings may indicate a restoration of microbiome balance by 20 days
postpartum. Recently, it was shown [24] that both
Our data had some limitations related to a rather small sample size. The main factor limiting the number of animals in this study was the traumatic nature of the endometrial scraping sampling protocol. Obtaining endometrial samples is an invasive procedure that may cause discomfort and potential risks to animals including the risk of infection and tissue damage as well as associated long-term negative consequences. We nonetheless believe that our dataset was sufficient to illustrate trends in the microbiome composition during the transition period. On the other hand, this would intensify the need for further studies with larger numbers of animals, once less invasive endometrial scraping sampling methods are developed. In any event, research using invasive sampling methods is often limited in the number of animals studied due to ethical and practical considerations. For example, a recent metagenomic investigation by Rashid et al. [74] also used a limited number of postpartum cows to examine their endometrial microbiome due to the invasiveness of the sampling procedure. In contrast, studies of the vaginal microbiota, which use less invasive sampling methods (e.g., swabs), often allow the use of larger numbers of animals.
Seven superphyla and phyla of microorganisms were identified in the endometrial microbiota of cows during the transition period (Fig. 1). Among them, the phylum Firmicutes (with the predominant class Clostridia) and the superphylum Fusobacteriota (represented by the single class Fusobacteriia) can be considered the “dominant” endometrial bacteria (from 25.2 to 68.2% and from 12.3 to 51.1%, respectively). This is consistent with the results of previous studies conducted on small groups of dairy [75] and beef cattle [76]. In these studies, Firmicutes turned out to be the dominant phylum of microorganisms in the reproductive system microbiota, with a relative abundance of 32.4 to 65.9%. This range is consistent with the relative abundance of Firmicutes observed in our investigation.
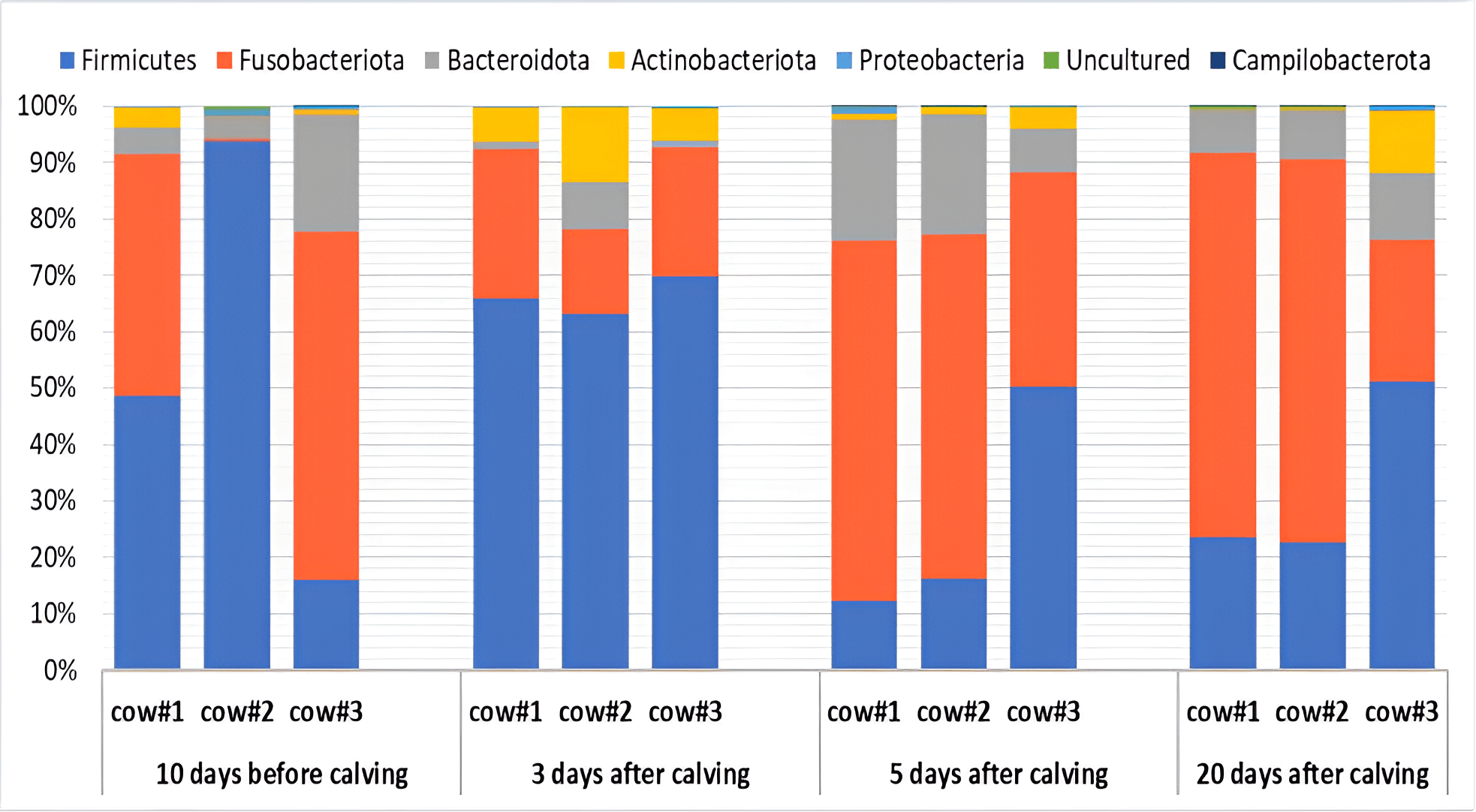 Fig. 1.
Fig. 1.
Dynamics of the taxonomic composition (%) of the endometrial microbiota in Holstein dairy cows during the transition period (10 days before calving; 3, 5 and 20 days postpartum) at the phylum level based on the next-generation sequencing (NGS) data. Cow #1, 2, 3, individual animal numbers.
Our study revealed that successional microbiological processes in the uterus of cows had an oscillatory nature depending on the technological period of reproduction. This confirms the complexity of the relationships in the endometrial microbial ecosystem with the sequential passage of critical transition period stages for the animal’s physiology. For example, the number of two dominant classes of microorganisms, i.e., Fusobacteriia and Clostridia, basically demonstrated opposite changes in their relative abundance during the transition period in cows. According to the theory of oscillations in the mathematical compositional model by Lotka–Volterra [77, 78], the antiphase oscillations of the numbers of microorganisms in an ecosystem may indicate competition between these microorganisms, e.g., for a nutrient resource [79]. Some researchers [80, 81] are of the opinion that the etiological onset of endometritis may be due to an increase in the proportion of individual taxa, including the species Fusobacterium necrophorum, a representative of the class Fusobacteriia. Among the class Clostridia, in contrast, we found typical representatives of the normal microbiota of mammals, e.g., Lachnospiraceae_NK4A136_group, Peptococcus, Murdochiella, Anaerococcus, Finegoldia, etc. (Fig. 2), producing volatile fatty acids with antimicrobial properties [82]. In addition, previous results have shown that an increase in the number of Lachnospiraceae in the vagina was associated with the reproductive health of cows [75]. In all likelihood, representatives of this taxon can prevent colonization of the reproductive system by unwanted microorganisms.
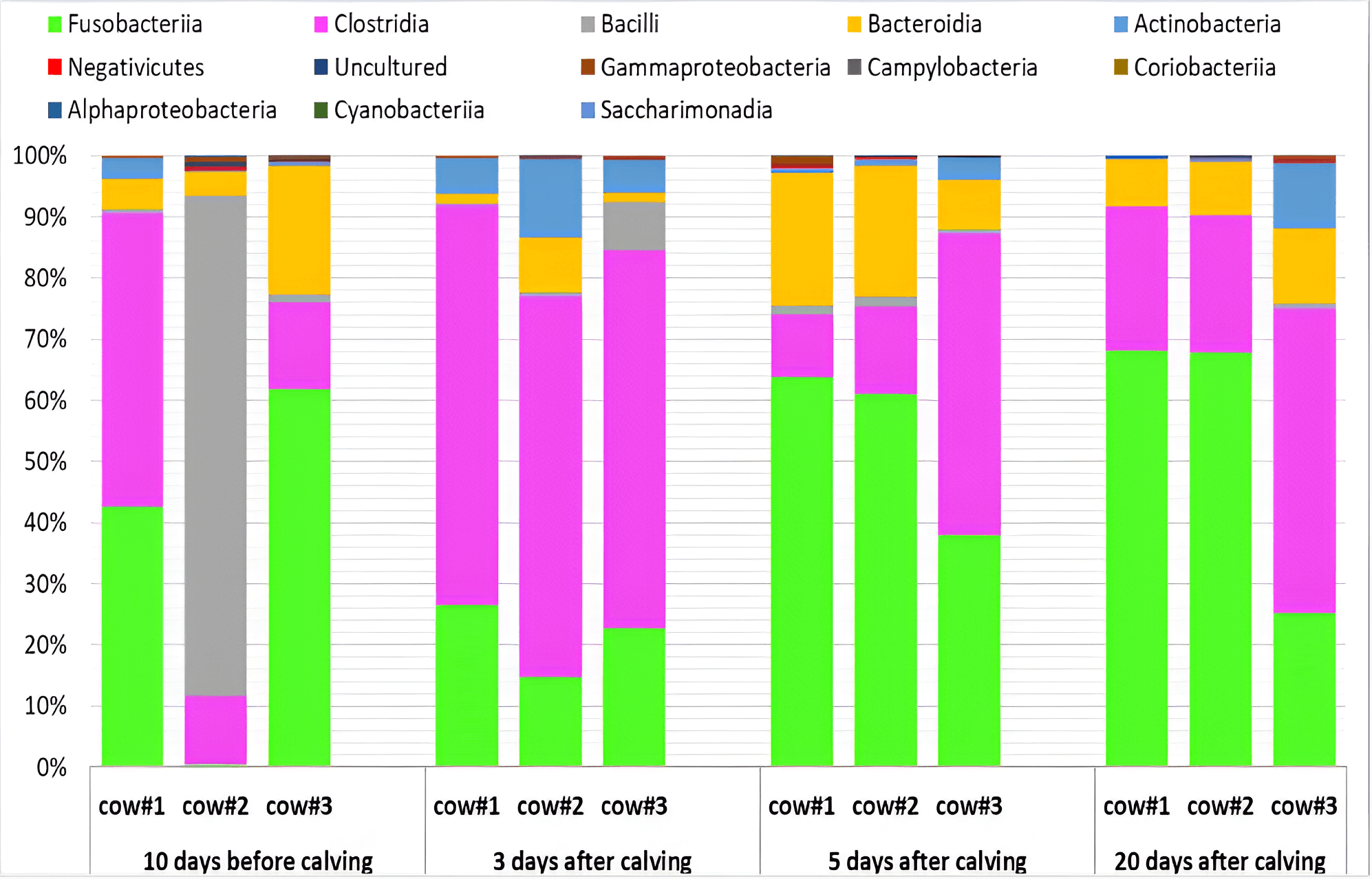 Fig. 2.
Fig. 2.
Dynamics of the taxonomic composition (%) of the endometrial microbiota in Holstein dairy cows during the transition period (10 days before calving; 3, 5 and 20 days postpartum) at the class level based on the NGS data. Cow #1, 2, 3, individual animal numbers.
It should be noted that our data demonstrate significant differences in the
abundance of various phyla, classes, and genera of endometrial microorganisms
between individual cows, especially pronounced on Day 10 before calving (Figs. 1,2,3), even though they belong to the same breed and were kept under the same
conditions. For example, in the period of 10 days before calving, cow #2 had a
sharply reduced Fusobacteriota content and an increased Firmicutes count
(p
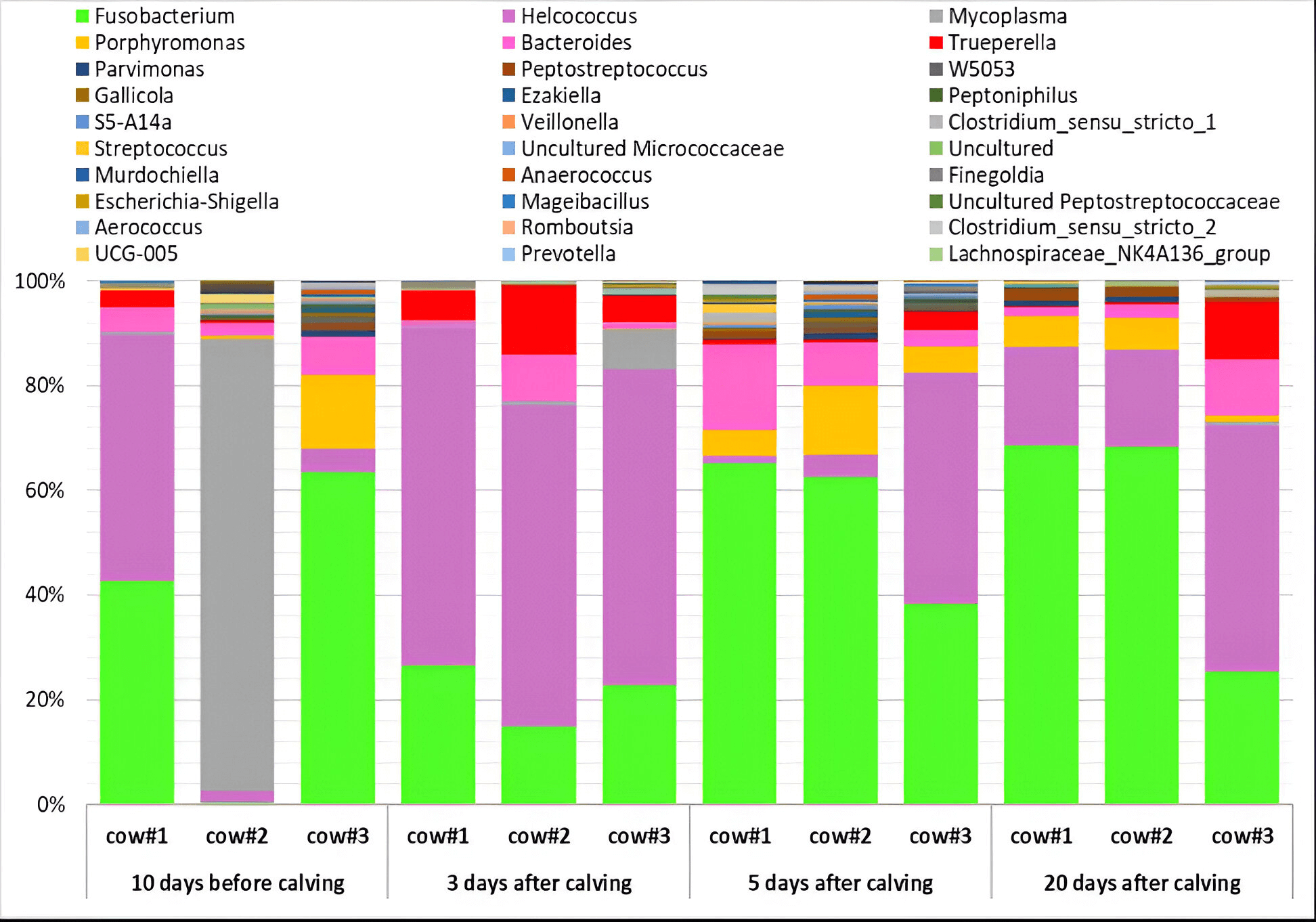 Fig. 3.
Fig. 3.
Dynamics of the taxonomic composition (%) of the endometrial microbiota in Holstein dairy cows during the transition period (10 days before calving; 3, 5 and 20 days postpartum) at the level of major genera (with a proportion of more than 0.3%) based on the NGS data. Cow #1, 2, 3, individual animal numbers.
Overall, the evidence suggests that the variation in microbial abundance observed between individuals may be due to several factors, including animal’s life history since birth. Differences in previously occurred diseases, use of antibiotics or other drugs, and stress throughout life [83] can all affect the microbiome. A study by Jernberg et al. [84] showed that even a single course of antibiotics can lead to significant and long-lasting changes in the composition of the gut microbiome. The authors found that some bacterial species can disappear from the microbiome and not recover for months or even years after antibiotic treatment.
The class Bacilli was represented by a single order, Lactobacillales, that
accounted for up to 1.41%. These bacteria are typically dominant in the human
reproductive tract microbiome, creating an acidic environment that is thought to
protect against pathogens and opportunistic infections. The dominance of
lactobacilli appears to be unique to humans, however, in that the relative
abundance of lactobacilli in the human reproductive tract is typically over 70%,
whereas, in other mammals, lactobacilli rarely exceed 1% [85]. We observed here
that the abundance of Lactobacillales was decreased in cows on Days 3 and 20
postpartum compared to other sampling points (p
The high abundance of the genus Fusobacterium (up to 68.2%) (Fig. 3)
in the uterus of clinically healthy cows was unexpected, given their potential
association with the occurrence of metritis in cows [86]. On Days 5 and 20
postpartum, the abundance of Fusobacterium spp. was the highest compared
to previous sampling points (p
Judging from the obtained data, the relative stabilization of the number of uterine microorganisms in clinically healthy animals during the transition period occurred by Day 5 postpartum. This is confirmed by the analysis of the factor map (Fig. 4) using such analyzed variables as the relative representation of microorganism families in the uterus. These findings coincide with the traditional concepts and recommendations of veterinary and obstetric practitioners [89]. After all, according to the recommendations, cows with inflammatory abnormalities of the uterus can be identified precisely on Days 5–7 postpartum, mainly based on thermometry data and other clinical signs.
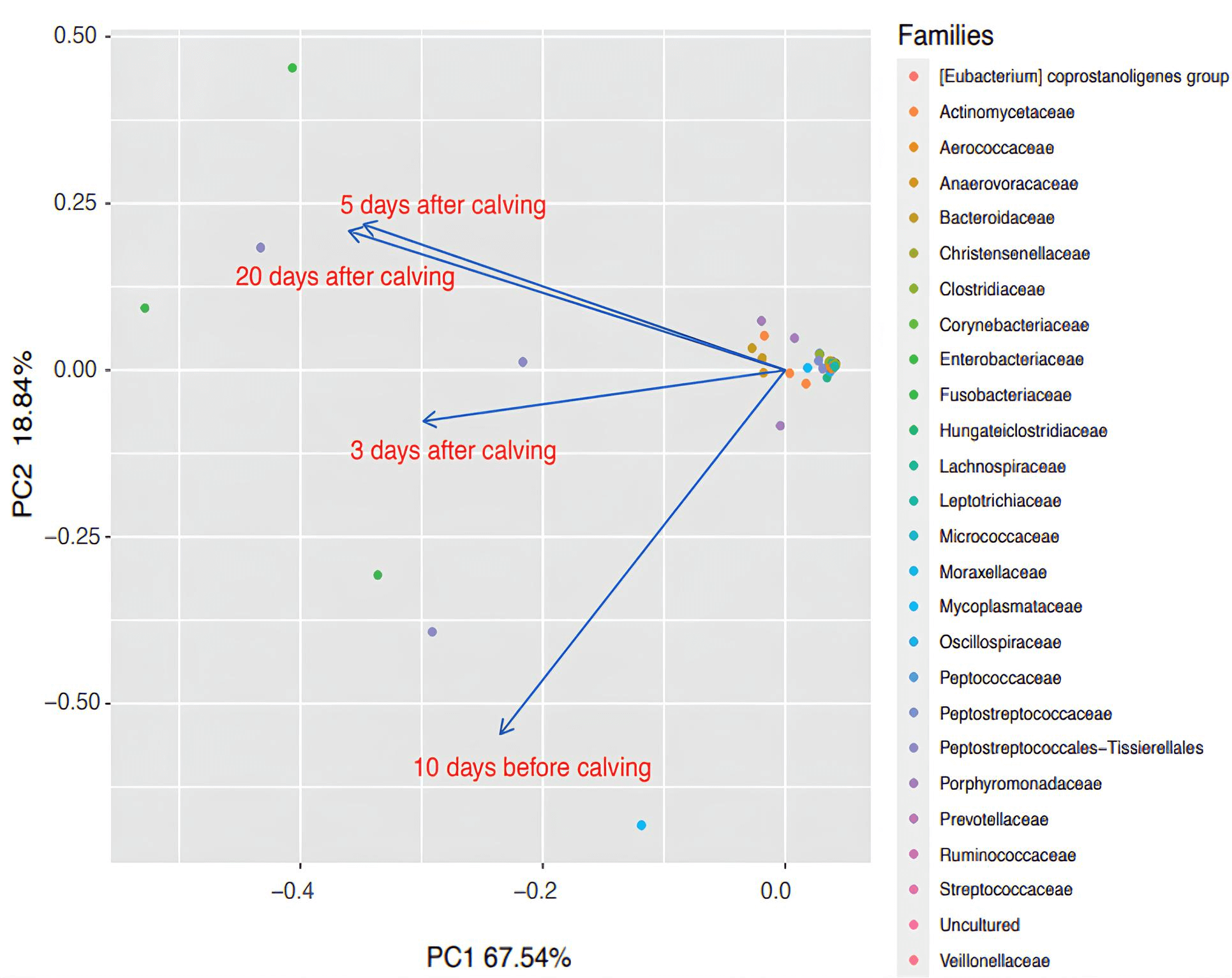 Fig. 4.
Fig. 4.
Factor map reconstructed using principal component analysis (PCA) and based on the values of relative representation of microbial families in the cow uterus as assessed using NGS technology. PC1, principal component 1; PC2, component 2. Arrows are vectors denoting representatives of microbial families (marked with colored dots according to the legend right) at different transit period stages (10 days before calving; 3, 5 and 20 days postpartum). The vector length corresponds to the contribution that a bacterial taxon makes in determining the two principal components. The angles formed by the two vectors represent the degree of correlation between the variables: small angles mean a high positive correlation, and angles close to 90° have no correlation.
Based on the abundance of microbial taxa, the stabilization of the microbiome
did not mean, however, that it had returned to its prepartum state. For example,
the abundance of Fusobacteriota was generally higher (p
As a result of the metagenomic analysis, 362 predicted metabolic pathways of the
endometrial microbiota were identified and annotated. The level of the predicted
functional potential of 32 pathways in the endometrium changed (p
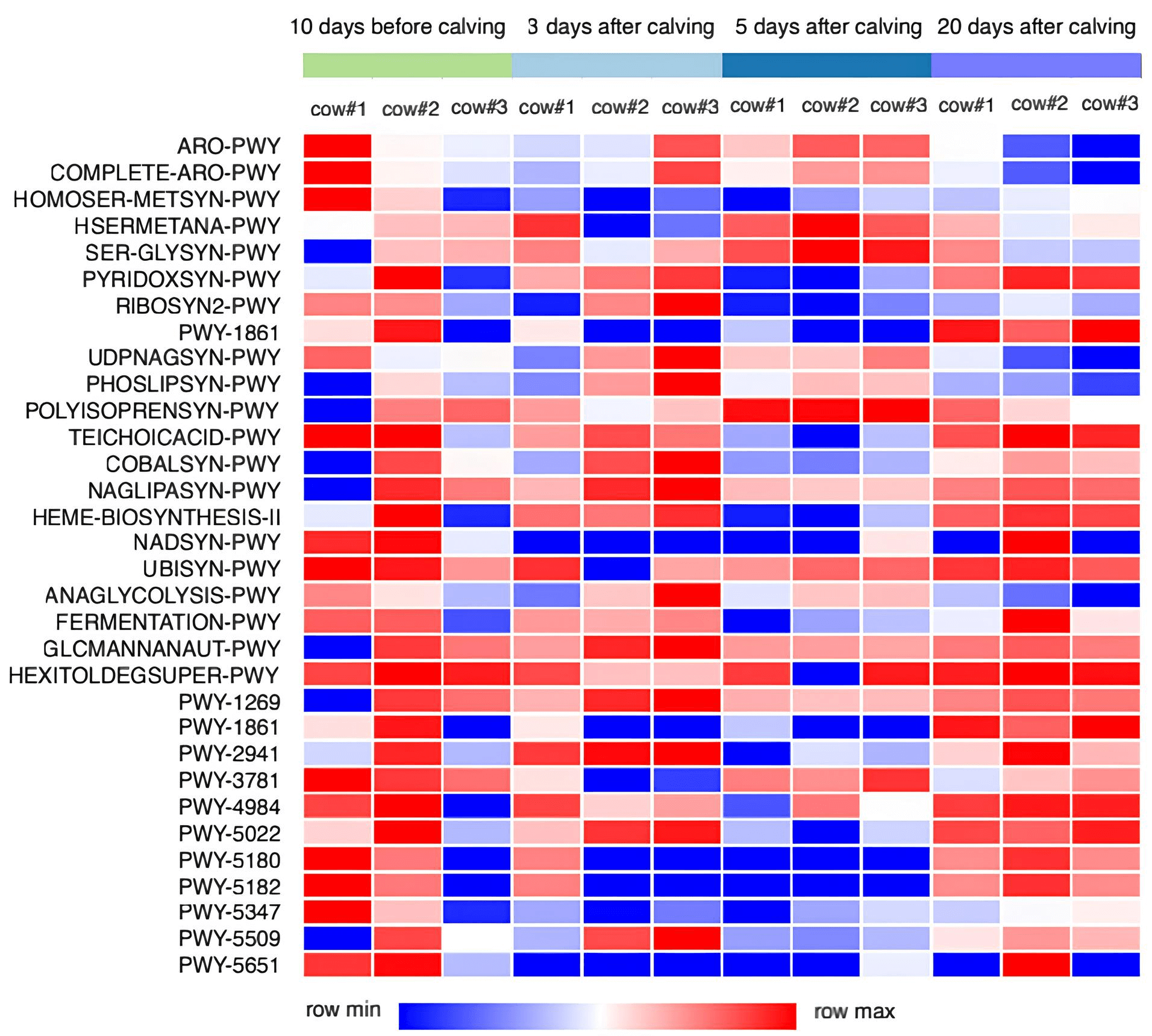 Fig. 5.
Fig. 5.
Data of functional annotation of metabolic pathways of the endometrium microbial community in Holstein dairy cows based on NGS data. Cow #1, 2, 3, individual animal numbers.
In the functional prediction of the endometrial microbiome composition, we
discovered that the activity of some pathways involving amino acid biosynthesis
(e.g., HOMOSER-METSYN-PWY: L-methionine biosynthesis I) was reduced on Days 3 and
20 postpartum (p
We herein demonstrate that both the microbiota composition and the activity of predicted metabolic pathways in clinically healthy cows during the transition period of the intercalving cycle undergo significant changes associated with various critical transition stages. This may indicate that communities of microorganisms in the reproductive system of cows exist in a finely tuned relationship with the host and its physiology. Our results show dynamic changes in the endometrial microbiome of cows in the prepartum and early postpartum periods, with marked individual differences between individual animals. Changes in endometrial microbial diversity after calving were characterized by wave-like dynamics, which presumably reflect a complex interaction of hormonal, physiological and metabolic factors, as well as environmental and intramicrobial interactions. Relative stabilization of the microbiome was noted by Day 20 postpartum. We established that even clinically healthy cows demonstrated signs of dysbiotic disturbances in the microbiome composition (e.g., fusobacteria dominance) and predicted functions of the endometrial microbiota throughout almost all transition period stages. Despite cows of the same breed being kept in the same housing conditions and receiving the same diet, a significant inter-individual variation in microbiome composition was detected, particularly on Day 10 before calving. This highlights the importance of considering such potential differences when developing reproductive health management strategies in cows. Again, our findings are limited by the small sample size associated with the invasive nature of endometrial scraping sampling. This highlights the need for further studies with larger numbers of animals when less invasive sampling methods are developed.
In recent years, the intensification of dairy farming has significantly affected the structure of cows’ diets and has led to an increased provision of concentrated feeds. Our findings are consistent with the fact that the recently adopted colossal body “overload” with available forms of energy (starch and sugars), while reducing the proportion of fiber in the fodder, appears not to correspond to the physiological needs of animals. This aggravates the stress of metabolic processes in the body of animals. Because of this, highly productive animals are vulnerable to stress and infectious diseases, and respond inappropriately to negative environmental factors [97]. Our data are pivotal for a better understanding of how symbiotic microbiota affects the physiology, immunity, and health of the reproduction system in dairy cows [86]. In the future, it would be of interest to conduct further experiments in cows kept in eco-farms with a housing and feeding system close to their natural habitat in order to assess the true representatives of the normal microbiota of the reproductive tract.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Conceptualization, EAY and GYL; methodology, EAY, VAF, LAI and EAK; software, ESP; validation, ESP and EAY; formal analysis, EAY, ESP, EAB, NIV, DKG and MNR; investigation, KAS, IAK, VAZ, VNB and EAK ; resources, VNB and EAK; data curation, EAY, NIN and DGT; visualization, ESP and EAB; supervision, GYL and DKG; project administration, EAY, MNR and GYL; funding acquisition, EAY and VAF; writing— original draft preparation, EAY; writing—review and editing, EAY, MNR and DKG. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study received ethical approval from the Bioethical Commission of the L. K. Ernst Federal Research Center for Animal Husbandry (Protocol No. 2023-10/1, dated October 31, 2023). This study was conducted in accordance with the ARRIVE guidelines.
Not applicable.
The study was financially supported by the Russian Science Foundation (Grant No. 24-16-00131, Development of a New Biotechnological Approach to the Prevention and Treatment of Endometritis in Cows).
The authors declare no conflict of interest. EAY, GYL, DGT, VAF, LAI, NIN, KAS, ESP, VAZ and VNB are employees of BIOTROF+ Ltd. The judgments in data interpretation and writing were not influenced by this relationship.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBE39439.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





