1 Laboratory of Problems of Stable Isotope Spreading in Living Systems, Southern Scientific Center of the Russian Academy of Sciences, 344006 Rostov-on-Don, Russia
2 Department of Biologically Active Substances, Kuban State University, 350040 Krasnodar, Russia
3 Laboratory for the Study of Metabolic Processes, Peoples’ Friendship University of Russia (RUDN), 117198 Moscow, Russia
4 Laboratory of Molecular Biology of Stem Cells, Institute of Cytology of the Russian Academy of Sciences, 194064 St. Petersburg, Russia
5 Scientific Department, Kuban State Agrarian University, 350004 Krasnodar, Russia
6 Department of Genetics, Cytology and Bioengineering, Faculty of Medicine and Biology, Voronezh State University, 394018 Voronezh, Russia
7 Department of Fundamental and Clinical Biochemistry, Kuban State Medical University, 350063 Krasnodar, Russia
8 Experimental Clinic-laboratory of Biologically Active Substances of Animal Origin, The V.M. Gorbatov Federal Research Center for Food Systems of Russian Academy of Sciences, 109316 Moscow, Russia
Abstract
Deuterium is unevenly distributed in natural waters, while the same applies to the content of deuterium in ice on Mars. Moreover, changes in the deuterium content of drinking water are known to affect the bodies of mammals. Thus, since plans are in place to send people to Mars in the coming years, understanding the effects of water with a Martian isotopic composition is necessary. Therefore, this study aimed to evaluate the impact of water with an increased deuterium content of 1200 ppm on the dynamics of indicators in the body of mammals.
The study was conducted on Wistar rats. The metabolic profile of blood and the content of deuterium in it were studied in dynamics by days using nuclear magnetic resonance (NMR) spectroscopy. Additionally, the isotopic composition of brain tissue was studied in dynamics by days using isotope mass spectrometry. A further study was conducted on the functioning of the antioxidant system in blood plasma and brain tissue using PCR analysis, chemiluminescence, and biochemical analysis methods; the intestinal microbiome was also studied. The durations of the animal experiments were 31 (blood and brain study) and 38 (stress-protective activity study) days.
On day 23, the deuterium content in the blood plasma increased to 856 parts per million (ppm), and to 260 ppm in the brain on day 31. This increase led to an imbalance in the antioxidant/prooxidant processes. This effect was accompanied by shifts in the intensity of oxidative processes, alongside changes in enzyme activity and the expression of genes responsible for their synthesis, shifts in amino acid composition, and changes in the concentration of metabolites and microbiome molecules in the blood plasma. By the fifth and eighth days, the number of Bacteroides in the intestines had decreased by 14% and 21.8%, respectively, compared to the values measured on day zero of the experiment. Meanwhile, the population of Firmicutes-type bacteria increased by 12% and 16% on the fifth and eighth days, respectively, compared to the indicators measured on day zero of the experiment.
An increase in the concentration of deuterium in the body promotes the development of a stress reaction and the activation of compensatory mechanisms aimed at adaptation.
Keywords
- deuterium
- water
- immobilization stress
- rats
- adaptation
The isotopic composition of natural waters is unevenly distributed across the surface of our planet [1, 2, 3, 4]. Thus, the deuterium content fluctuates in the approximate range of 89 to 162 parts per million (ppm). The main standard for deuterium content in natural water, the Vienna Standard Mean Ocean Water (VSMOW), is 155.76 ppm [5, 6, 7, 8, 9]. On Mars, the deuterium content in glaciers at the poles, on mountain tops, and at the equator exhibits a much wider range of ~155 to ~1246 ppm [10]. Currently, small fluctuations in the concentration of deuterium in drinking water are known to promote activation (due to the implementation of adaptation reactions of the body) and inhibitory effects [11, 12, 13, 14, 15, 16]. Moreover, these concerns relate to both the reduction and increase of the deuterium content in water [17, 18, 19, 20, 21]. A relatively small decrease in deuterium levels in the body causes changes in the functioning of the nervous system [22, 23, 24, 25]. Additionally, long-term consumption of a drinking diet with a reduced deuterium content leads to a gradual decrease in the concentration of deuterium in the blood plasma and organ tissues [26, 27]. This results in selective 2H/1H substitution in atomic groups with a free unshared pair of electrons capable of forming hydrogen bonds [28, 29]. Such atomic groups in biological systems include hydroxyl, sulfhydryl, and amino groups found in macromolecules such as proteins and nucleic acids, as well as in low-molecular biologically active compounds [30]. The substitution of deuterium for protium can occur in the hydration shell of protein molecules, including enzymes and transporters, as well as proteins involved in the formation of pores and channels, etc. [31]. These alterations cause a change in the thermodynamic and kinetic parameters of macromolecules [32, 33, 34, 35, 36]. The isotopic 2H/1H exchange may affect some regions of the mitochondrial electron transport chain, leading to conformational changes in transport protein structures [37, 38, 39, 40]. Kinetic isotope effects in biological systems have been known for a long time [41]. Moreover, these effects can occur at significant concentrations of deuterium [42, 43]. Meanwhile, studies on the impact of increasing the deuterium concentration in water to levels several times above the natural terrestrial level remain limited. However, seminal work by Xie X and Zubarev R.A. [44, 45] evaluated the effects of increasing the rate of biomass gain by microorganisms with a deuterium content in the environment of 350–370 ppm. The simplification of the system can explain these effects due to the occurrence of isotopic resonance [46]. Our previous work has established an increase in the deuterium content in the blood plasma of laboratory animals from 150 to 487 ppm, with water consumption contributing to a total of 750 ppm over two months [17]. This was shown to increase the antioxidant potential in liver tissues, due to a rise in the content of low-molecular thiol antioxidants and a decrease in the intensity of free-radical processes in the blood and hepatocytes. There was also an increase in the functional activity of the prooxidant–antioxidant component of the non-specific defense system in the body [47, 48, 49].
Thus, there is a need to obtain scientific data on the effect of deuterium content, similar to that of Mars, in drinking water on mammalian organisms. Therefore, this study aimed to evaluate the impact of consuming water with a high deuterium content of 1200 ppm on the dynamics involved in modulating the concentration of deuterium in the blood and brain tissues, the dynamics of oxidative processes, and the functioning of the antioxidant system in the brain and the intestinal microbiome, as well as on the development of the cross-adaptation to immobilization stress.
Experiments were performed on 3-month-old male Wistar rats weighing 280–310 grams. For tissue culture studies, 7–9-day-old rat pups born to 5–6-month-old females were used.
Methods for obtaining cultures were described earlier [13]. The authenticity of the culture was determined by morphological analysis. Rat cerebellar neurons (cerebellar granule cells) in vitro are 8–10 microns in size, have a contrasting edge and characteristic granularity of the cytoplasm, and form a developed network of neurites in the early days of cultivation. The underlying layer of astrocytes is characterized by an irregular polygonal shape, a rounded or oval nucleus, an indistinct edge of the cytoplasm, and dimensions exceeding the linear size of neurons by about 2–4 times. The experiments were performed using an incubation medium (IM) containing 154 mM NaCl, 25 mM KCl, 2.3 mM CaCl2, 1 mM MgCl2, 3.6 mM NaHCO3, 0.35 mM Na2HPO4, 10 mM HEPES, 5.6 mM glucose, pH 7.3. IM with different levels of deuterium was prepared from IM with 150 ppm deuterium, to which “heavy” water with a deuterium content of 99.92% was added to a deuterium concentration of 300, 450, 600 and 750 ppm. Deuterium content was monitored using a 400 MHz Fourier transform NMR spectrometer (FT NMR SYSTEM model JNM-ECA 400, JEOL, Japan) according to the method [50]. To study the effect on the mitochondrial membrane potential and intraneuronal calcium levels, the cultures were incubated with IM for 24 h. Measurements were performed using fluorescent probes: tetraethylrhodamine (TMRE) and Fluo-4 AM, respectively. The probes were added to the cultures for 15 min: 0.1 µM TMRE and 5 µM Fluo-4 AM. The cultures were then washed three times with IM and fluorescence was measured on a Filter Max F5 multifunctional microplate reader (Molecular Devices, USA): TMRE at an excitation wavelength of 535 nm and emission of 595 nm; Fluo-4 at 485 and 535 nm, respectively [51].
There were six animals per cage (294 mm
The study of the response by the body to deuterium intake was performed on 65 rats. The animals were divided into two groups:
Group 1 (n = 30)—rats that received water with a natural deuterium content (150 ppm);
Group 2 (n = 30)—rats that received water weighted with deuterium (1200 ppm).
Five rats were taken from each group of animals on days 5, 8, 11, 16, 23, and 31 of the experiment to collect feces, blood, and the brain was removed following decapitation; five rats were also taken on day zero. For adult specimens, the surgical intervention was performed under anesthesia using the following drugs: Telazol (tiletamine 50 mg/ml and zolazepam 50 mg/ml, total concentration 10% w/v) 20 mg/kg intramuscularly (Zoetis Inc., San Sebastián de los Reyes, Spain) and Xylazine (xylazine hydrochloride, 2% w/v) 5 mg/kg intramuscularly (Interchemie, Enschede, Netherlands). The animals were euthanized in an euthanasia chamber (VET tech) in accordance with Directive 2010/63/EU of the European Parliament and the European Union Council for the protection of animals used for scientific purposes. The feces were used to study the qualitative and quantitative composition of the intestinal microbiome. Plasma and serum were prepared from the blood. The concentration of deuterium was determined in the blood serum, and the plasma was used to study the intensity of the oxidative processes, the leukocyte formula, the metabolic profile, and to research tryptophan and tyrazine fluorescence. In the cold, the cortex was removed from the brain to study the intensity of the oxidative processes, antioxidant activity, and gene expression.
Meanwhile, two groups of 28 rats were formed to study the effects on the behavior of animals of high deuterium concentrations in the drinking water:
Group 1 (n = 14)—rats that received water with a natural deuterium content (150 ppm);
Group 2 (n = 14)—rats that received water weighted with deuterium (1200 ppm).
Seven rats were taken from each group of animals on the 7th and 14th days of the experiment to study the behavioral reactions in the open field test.
An additional three groups of 21 rats were formed to study the protective activity against stress:
Control-150 ppm group (n = 7)—rats that received water with a natural deuterium content (150 ppm);
Stress-150 ppm group (n = 7)—rats that received water with a natural deuterium content (150 ppm) for 32 days and were exposed to immobilization stress;
Stress + deuterium weighted water-1200 ppm group (n = 7)—rats that received water with an increased content of deuterium (1200 ppm) for 32 days and were exposed to immobilization stress.
Immobilization stress is a traditional model of an acute stress situation, in which, in addition to restricted movement, a pronounced emotional component exists that is associated with the inability to avoid a threatening situation [52]. The use of varying durations of immobilization allows for evaluating changes in the body that occur under the influence of stress factors of different intensities.
Immobilization stress was modeled on the 33rd day of the experiment. Each animal was secured on its back, with its limbs tied to ensure it remained motionless. The stress procedure was repeated four times daily for three days. The animals were monitored daily, with their limbs examined. Behavioral activity was studied in the open field and elevated plus maze tests on the 36th and 37th days, respectively. On the 38th day of the experiment, the animals were sacrificed, euthanasia method is the same as above, blood was collected, and the brain, thymus, adrenal glands, and spleen were removed. Plasma was prepared from whole blood to study oxidative processes. The cerebral cortex was removed in the cold and placed in liquid nitrogen for storage. The brain tissue was ground in the cold, and the supernatant was prepared to study oxidative processes. The thymus, adrenal glands, and spleen were weighed on an analytical balance.
The experiments were conducted in accordance with the requirements of the “Guide for the Care and Use of Laboratory Animals”, European Community Directives 2010/63/EU, and “Guide for working with laboratory animals, including the ethical principles of animal testing (3R principle) of the V.M. Gorbatov Federal Research Center for Food Systems of the Russian Academy of Sciences”. The study was approved by the bioethical commission of the V.M. Gorbatov Federal Research Center for Food Systems of the Russian Academy of Sciences (Protocol No. 7/2023 on 7th March, 2023).
The deuterium content in blood serum was determined on a Bruker Avance NEO 700 MHz NMR spectrometer (Bruker Corporation, Bloomfield, CT, USA) using the method described earlier [50]. The results are expressed in ppm. The experiment was performed at the shared-use center of the Scientific Research Center “Pharmacy” (RUDN University, Moscow).
The metabolic profile was determined using the following method. One volume of cold methanol and one volume of cold chloroform were added to the blood plasma sample and mixed on a vortex (4000 rpm, 30 minutes, 5 °C). The samples were then kept at 5 °C for 30 minutes, after which the samples were centrifuged (12,000 g, 30 minutes, 5 °C). The upper fraction containing water and water-soluble metabolites was transferred to a new tube and dried for 16 hours on a vacuum concentrator. The dry sample was stored at –60 °C. Before measuring the spectrum, 600 µL of buffer solution (0.1 mM disuccinimidyl suberate, 2 mM NaN3, 150 mM Na2HPO4/NaH2PO4, pH 7.07 в D2O) was added to the dry sample, vortexed (4000 rpm, 2 minutes), and centrifuged (12,000 g, 5 minutes). Afterward, 550 µL of the sample was transferred to an NMR ampoule. The spectra were recorded on a Bruker Avance NEO 700 NMR spectrometer equipped with a Prodigy cryoprobe. The Noesypr1d pulse sequence was used under the following conditions: acquisition time of 4.7 seconds, delay time between pulses (D1) of 3 seconds, and number of spectrum accumulations of 64.
The spectra were processed using Topspin 4.1.3 software (version 4.1.3, 2021, Bruker BioSpin, Rheinstetten, Germany), and the identification and quantification of metabolites were performed using Chenomx 8.5 software (version 8.5, 2021, Chenomx Inc., Edmonton, Canada). All results were presented as a percentage of the control sample. The experiment was performed at the shared-use center of the Scientific Research Center “Pharmacy” (RUDN University, Moscow).
Fluorescence was measured for the blood serum using an F-2700 spectrofluorimeter (Hitachi, Tokyo, Japan). The intrinsic combined tryptophan and tyrosine fluorescence was excited by light with a wavelength of 280 nm. The intensity of free radical processes in the blood plasma was determined by chemiluminescent (CL) analysis using a Lum-100 device (Lumex Instruments, Moscow, Russia), which employs the light sum, an integral luminescence indicator expressed in arbitrary units. Blood plasma luminescence was initiated by adding 0.3% hydrogen peroxide [53]. Results are presented as percentages for the control sample.
Leukocyte counting was performed using a veterinary hematology analyzer BC-5000Vet (Mindray Bio-Medical Electronics Co., Shenzhen, Guangdong, China).
The proportion of dominant bacterial phyla in the gut microbiome was assessed using the polymerase chain reaction (PCR) with phylum-specific primers to 16S rRNA. The primer sequences are presented in Supplementary Table 1.
Quantitative PCR analysis was performed on a CFX96TM Real-Time System thermal cycler (Bio-Rad Laboratories, Hercules, CA, USA) using the qPCRmix-HS SYBR kit РК147L (Eurogen, Moscow, Russia). The reaction conditions were as follows: total denaturation at 94 °C for 5 min, followed by 35 cycles of denaturation at 94 °C for 10 s, primer annealing at 55 °C for 10 s, and elongation at 72 °C for 15 s. The quantitative ratio of each phylum was calculated using the formula:
Bacteria count = 100
where E represents the PCR efficiency.
The experimental animals were dissected immediately after sacrifice to remove the thymus, adrenal glands, and spleen. The organs were washed with cold saline, dried with filter paper, and then weighed on an Ohaus Pioneer analytical scale (Ohaus Corporation, Parsippany, NJ, USA).
The hydrogen isotope composition was determined in lyophilized brain tissues by mass spectrometry of light element isotope ratios using a DELTA V Advantage isotope mass spectrometer (Thermo Fisher Scientific Inc., Bremen, Germany). The device was combined with a Flash 2000 elemental analyzer (Thermo Fisher Scientific Inc., Waltham, MA, USA) and equipped with a pyrolytic reactor. Homogenized samples weighing 240–280 µg, packed in silver capsules, were loaded into the autosampler of the Flash 2000 elemental analyzer. The capsule, which entered the pyrolytic reactor heated to 1350 °C, was burned in a carrier gas flow (helium grade 6.0; 90 mL/min). Hydrogen released during pyrolysis entered the DELTA V Advantage mass spectrometer via a capillary through the Conflo II gas distribution system.
The supernatant was obtained from a sample of brain tissue ground in a mortar, frozen in liquid nitrogen, and then placed in a cold phosphate buffer at pH 7.4 using 100 mg of tissue per 1 mL. The sample was homogenized by shaking for 15 min, and then centrifuged at 6000 rpm for 10 min [54]. The intensity of the free radical processes in the supernatant of the cerebral cortex was assessed using the chemiluminescent analysis method and by the number of products formed in the reaction with thiobarbituric acid (TBA method), one of which is the product of lipid peroxidation—malondialdehyde (MDA). Chemiluminescent analysis was performed on the Lum-100 device (DISoft LLC, Moscow, Russia), and the intensity of the oxidative processes was assessed by the light sum, the integral indicator of luminescence, expressed in arbitrary units. Initiation of tissue luminescence in the supernatant was caused by adding 35 mM Fe2+ ions in the form of a solution of iron (II) sulfate.
The content of MDA in the supernatant was determined by the concentration of the colored complex formed during the interaction of MDA with thiobarbituric acid at a wavelength of 532 nm. This indicator was measured in µM per 1 g of protein. All results are presented as a percentage of the control sample.
The supernatant was obtained from a sample of brain tissue ground in a mortar, frozen in liquid nitrogen, and then placed in a cold phosphate buffer at pH 7.4, using 100 mg of tissue per 1 mL. The tissue samples were homogenized by shaking for 15 min, and then centrifuged at 6000 rpm for 10 min. Antioxidant status was determined by catalase activity and the concentration of reduced glutathione (GSH) in the supernatant prepared from the cerebral cortex tissue. The expression of the genes responsible for the synthesis of antioxidant proteins was assessed. Catalase activity was evaluated using the rate of hydrogen peroxide loss in the incubation medium. Hydrogen peroxide is capable of forming a stable-colored complex with molybdenum salts, with an absorption maximum at 410 nm. Catalase activity was proportional to the difference in optical density between the blank and test samples and was expressed as mCat/gram of protein. The content of reduced glutathione was determined spectrophotometrically using Ellman’s reagent (5,5′-dithiobis-(2-nitrobenzoic acid)) at a wavelength of 512 nm; the data are expressed as µmoles per 1 g of protein.
Nucleic acids were isolated using various commercial kits. DNA was isolated
using the Proba-GS kit P-023/4 (DNA-technology, Moscow, Russia). RNA was isolated using
ExtractRNA (Eurogen, Moscow, Russia)—a monophasic aqueous solution of phenol
and guanidine isothiocyanate with the subsequent addition of chloroform for phase
separation and isopropyl alcohol for RNA precipitation. Qualitative analysis of
the isolated nucleic acid samples, as well as PCR products, was performed using
gel electrophoresis in 2% agarose gel with 1
The level of anxiety and exploratory activity of animals was studied in the elevated plus maze test. The elevated plus maze (Open Science, Russia) has two open and two closed arms of 90 cm long and 15 cm high. The number of entries into the closed arms of the maze, the number of vertical rises on the hind legs (vertical activity), hanging from the open arms of the maze, the number of entries into the open arms of the maze, and the number of approaches to the distal edge of the open arms were taken into account. The number of actions aimed at cleaning the fur (grooming), which is an indicator of anxiety in the animal, was also assessed. All indicators in these tests were recorded during 5-minute observations.
The level of anxiety in the animals was studied in the open field test. The open field (Open Science, Russia) is a circle with a diameter of 1 meter and a wall height of 42 cm. The number of entries into the center of the field, the number of crossed squares (horizontal activity), and the number of vertical rises on the hind legs (vertical activity) were taken into account. The number of actions aimed at cleaning the fur (grooming), which is an indicator of anxiety in the animal, was also assessed. All indicators in these tests were recorded during 3-minute observations.
Bovine serum albumin (BSA) was used as a model protein. The structure of BSA in
media with different deuterium contents (150, 1000, 2000, and 4000 ppm) was
studied using intrinsic tryptophan fluorescence spectra and circular dichroism
spectra. For this purpose, a stock solution of BSA was prepared in water with a
natural deuterium content (150 ppm) in 0.01 M potassium phosphate buffer at pH
7.5. The initial water, with a specific resistance of 18.2 MOhm⸱cm and a
deuterium content of 150 ppm, was obtained for the preparation of solutions on a
LaboStar TWF 7 system (Evoqua Water Technologies, Ransbach-Baumbach, Germany).
Two working solutions of BSA were prepared one day before the measurements. The
first working solution was prepared by diluting the stock solution with a
phosphate buffer (pH 7.5, 150 ppm) to achieve the desired protein concentration.
The second working solution was obtained by adding D2O to the stock
solution. The BSA concentration was 5 µM and was determined using a Hitachi
U-2000 spectrophotometer (
Statistical analysis of the obtained data was performed using the STATISTICA 10
software package (version 10, 2010, StatSoft, Inc., Tulsa, USA). The
Kruskal–Wallis H-test was used to test the hypothesis of equality of medians in
multiple comparisons of several independent samples. The nonparametric
Mann–Whitney U test was used to assess the reliability of differences in
quantitative parameters between two independent small samples. The Wilcoxon
W-test was used to test differences between two small samples with paired
measurements. Differences between samples were considered statistically
significant at a value of p
Fig. 1A shows that the animals in the control group at day 0, before the start
of the exposure, possessed a 2H level in the blood of 152
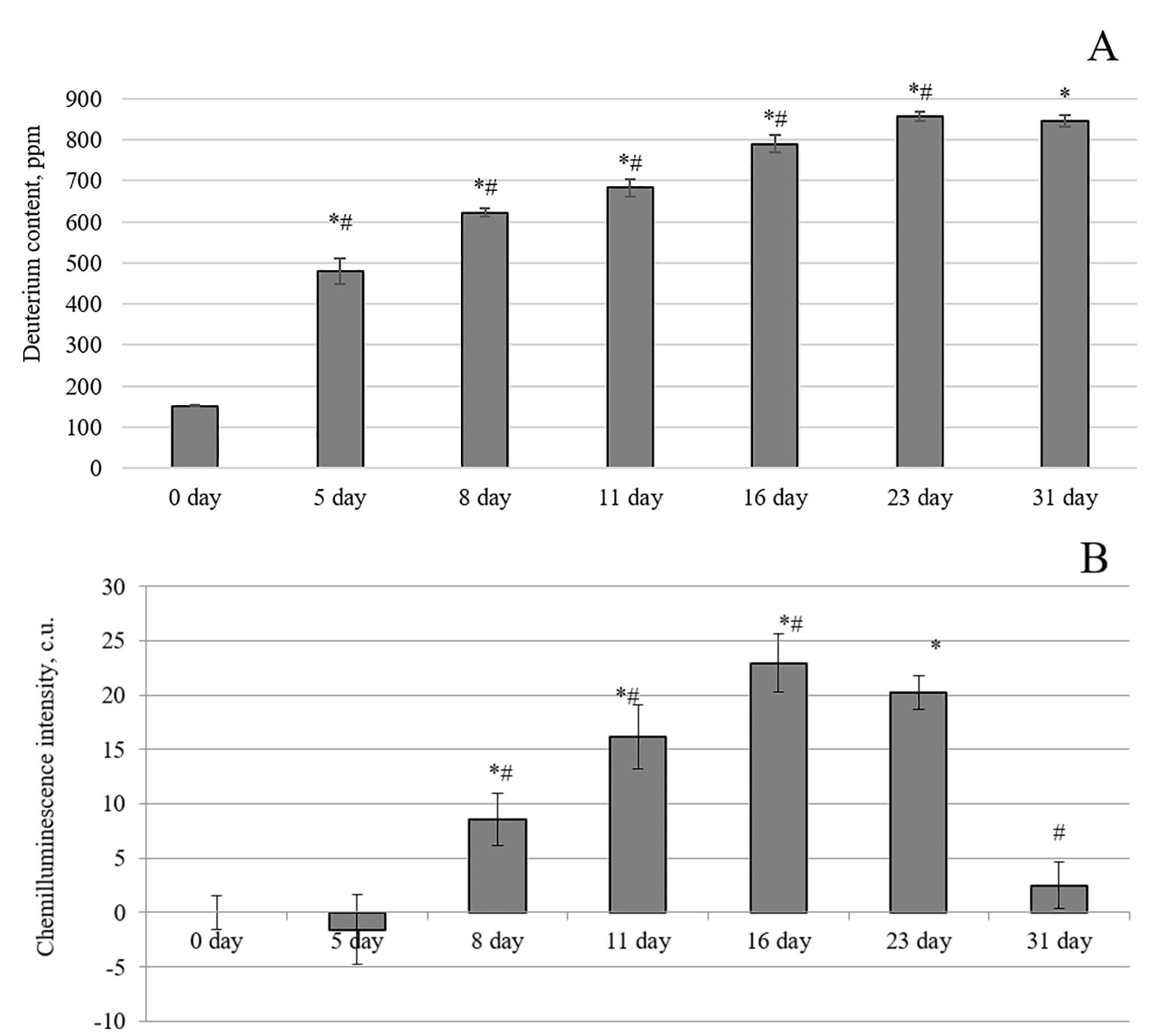 Fig. 1.
Fig. 1.
Changes in blood plasma parameters of laboratory animals after
the consumption of water with an altered isotopic composition (2H/1H =
1200 ppm). (A) Change in deuterium content. (B) Chemiluminescence intensity. *,
p
The deuterium content was 623
The intensity of chemiluminescence (light sum) is proportional to the rate of free radical formation in the model system. Thus, an increase in the light sum of blood plasma luminescence may result from changes in the antioxidant potential in the blood plasma and the activation of free radical processes. A noticeable increase in redox processes in the blood plasma of laboratory animals was observed after the fifth day of the experiment. On the eighth day of the experiment, the CL intensity exhibited a statistically significant increase of 9% compared to the control animals. The maximum effect was achieved on the 16th and 23rd days, with values of 23% and 20%, respectively, relative to the blood plasma of the control animals. The intensity of the luminescence of the blood plasma from the animals decreased after 23 days of drinking water with a 1200 ppm concentration. On the 31st day of the experiment, the intensity did not significantly differ from the indicators of animals whose diet included water with a natural deuterium content. Thus, an increase in the deuterium content in the blood plasma of rats at the beginning of the experiment led to an imbalance in antioxidant/prooxidant processes in favor of the latter, which was restored with longer exposure.
Table 1 presents the leukocyte composition data of the blood from the laboratory animals in dynamics by day when the animals were exposed to a drinking ration with a modified isotopic composition. While consuming a drinking ration with increased deuterium content, there were fluctuations in the overall level of leukocytes in the blood throughout the experiment, along with unstable changes in the elemental composition of immune system cells.
| Parameter | Day 0 | Day 5 | Day 8 | Day 11 | Day 16 | Day 23 | Day 31 |
| Leukocytes, total, |
3.9 |
4.3 |
4.3 |
5.1 |
4.9 |
4.5 |
6.0 |
| Segmented neutrophils, % | 7 |
9 |
8 |
3 |
1 |
7 |
11 |
| Monocytes, % | 7 |
6 |
6 |
6 |
6 |
12 |
12 |
| Lymphocytes, % | 87 |
85 |
87 |
91 |
93 |
81 |
77 |
| Basophilic leukocytes, % | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Eosinophilic leukocytes, % | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
Note: *, p
Statistically significant changes in the leukocyte formula were observed after eight days of the study. Moreover, there was an increase in the total concentration of leukocytes from the eleventh to the sixteenth day of observation, caused by a decrease in the number of segmented neutrophils and a sharp increase in the number of lymphocytes. In contrast, an opposite trend was observed from day 23 to day 31 of the experiment, whereby an increase in the total number of leukocytes occurred due to a sharp rise in segmented lymphocytes and monocytes, while the level of lymphocytes decreased. Notably, the changes in the leukocyte profile of laboratory animals that we established correspond to the reference values of the physiological norm for Wistar rats.
Thus, a drinking diet with an increased content of deuterium (1200 ppm) relative to a natural content causes minor changes in the leukocyte profile of these experimental animals.
An increase in the deuterium content in the blood of laboratory animals against the background of their consumption of water with a modified isotopic composition led to a change in the spectra of the intrinsic fluorescence of the proteins in their plasma (Supplementary Fig. 1). These changes were expressed by a shift in the intensity index and a bathochromic shift in the radiation emitted by tryptophan and tyrosine amino acid residues.
Induced isotopic 2H/1H exchange in the blood plasma of laboratory animals caused statistically significant changes in fluorescence indices on the fifth day of the experiment. On day 5 of this study, the intensity of the emitted wavelength of radiation was 31% higher, and the maximum shifted 3 nm to the long-wave region of the spectrum, compared to the radiation of the blood plasma of animals that drank water with a natural content of deuterium (control). On day 8 of the experiment, the fluorescence indices of blood plasma proteins did not differ from those on day 5, while the maximum effect was observed on day 11. The bathochromic shift of the emission maximum was noted as 7 nm, and the intensity increased by 88% compared to the blood plasma of animals in the control group. On day 16 of the experiment, the fluorescence parameters did not differ from the control indices, and these parameters were maintained until the end of the experiment. Thus, a change in the isotopic composition of the blood plasma proteins at the initial stages of exposure led to a change in the fluorescence indices of the tryptophan and tyrosine amino acid residues. At the later stages of exposure, there was a complete restoration of the intensity and maximum wavelength of radiation to control values.
The blood metabolic profile allows for a qualitative and quantitative assessment of hundreds of small circulating molecules that provide information about changes in the physiological parameters of the body. Therefore, this study established the effect of changes in the deuterium content to protium ratio in the body of laboratory animals on the levels of some metabolites. A shift in the amino acid composition, changes in energy metabolism, and microbiome molecules in the blood plasma were noted in the body of laboratory animals following the daily intake of deuterium (Fig. 2).
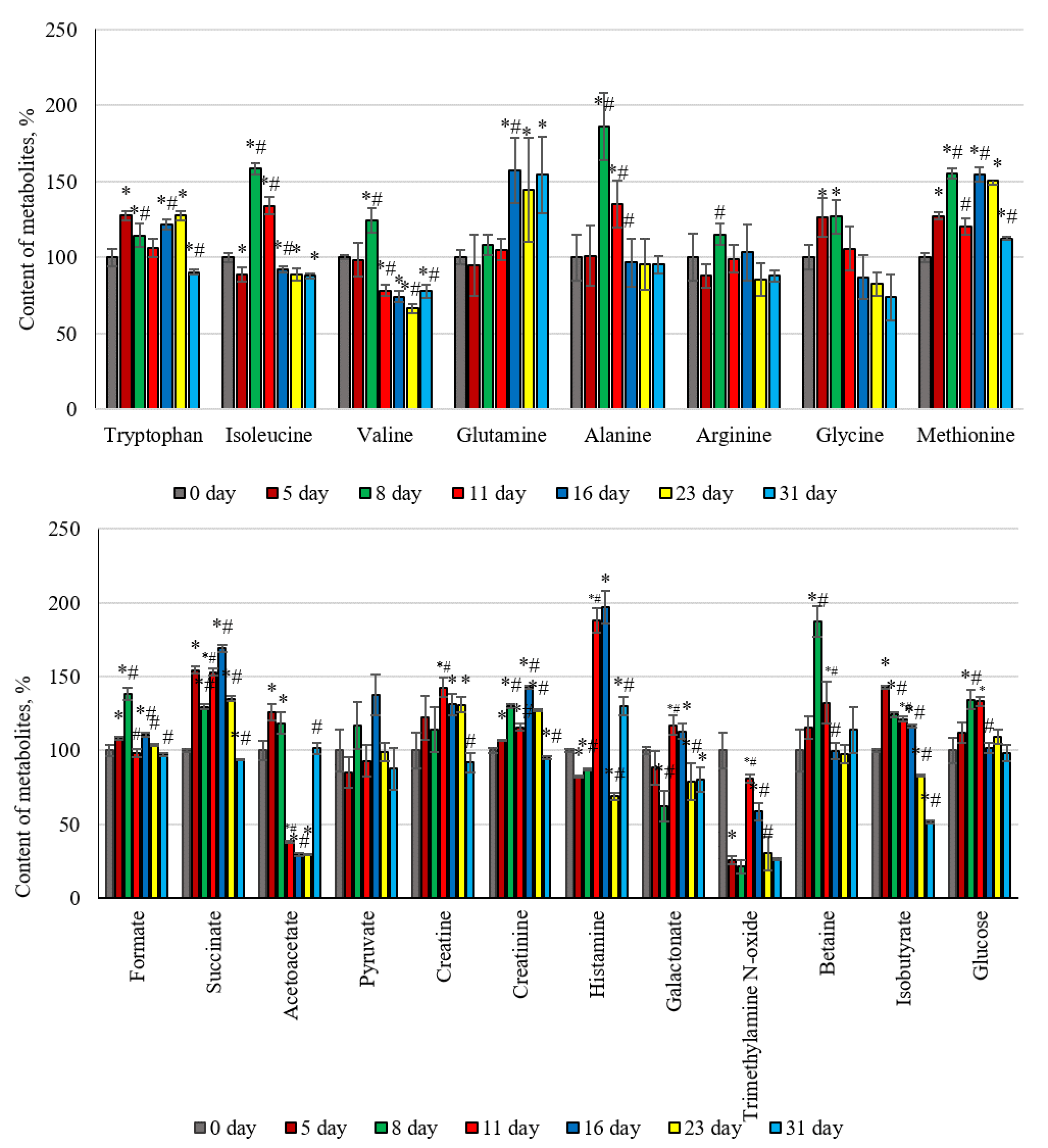 Fig. 2.
Fig. 2.
Changes in the metabolic parameters of the blood plasma of
laboratory animals against the background of their consumption of water with a
modified isotopic composition (2H/1H = 1200 ppm). *, p
Fig. 2 presents compelling evidence that the dynamics involved in the changes to
the amino acid profile have a certain pattern. A marked increase in the
concentration of aminocarboxylic acids in the blood was observed at the initial
stages of exposure, followed by a decrease in the control values at later stages
of the experiment. This trend was noted for tryptophan, alanine, glycine,
isoleucine, arginine, and methionine. The content of tryptophan and methionine
increased on the first day of the experiment and remained high until day 23. The
levels of isoleucine, alanine, and glycine characteristically increased on days 8
and 11 of the intake of heavy hydrogen atoms into the body. Valine and arginine
were the exceptions, consistently showing high concentrations relative to the
control on day 11 of the experiment. The quantitative profile of the studied
compounds involved in catabolism also changed over time, depending on the
duration of the experiment. Molecules such as glucose, succinate, formate,
creatine, creatinine, betaine, and isobutyrate are characterized by a certain
dependence on the background of changes in the isotopic 2H/1H exchange
[56]. The concentration of these metabolites increased in the bodies of the
experimental animals by day 5 and remained high for up to 16–23 days from the
start of the experiment. The concentration of glucose in the blood increased on
days 8 and 11 of the experiment by an average of 30% compared to the control
animals. The glucose concentration leveled out on the 16th day of exposure. The
content of succinate, a participant in the Krebs cycle, increased by 54% on the
fifth day and remained high until the 23rd day; only on day 31 of the experiment
did the succinate concentration not differ from the control parameters. The level
of nitrogen-containing carboxylic acid, creatine, increased by 22% (p
The daily increase in deuterium in the intracellular fluid and tissues of laboratory rats resulted in changes in the quantitative composition of the intestinal microbiome (Fig. 3A,B). These changes most significantly affected the main representatives of the intestinal microflora, such as Bacteroides and Firmicutes. Fig. 3A shows that the ratio of the Firmicutes/Bacteroides abundance has a bell-shaped dependence. The maximum increase in Firmicutes bacteria and the maximum decrease in Bacteroides representatives occurred on days 5 and 8 of the experiment, respectively. By the fifth and eighth days, the number of Bacteroides decreased by 14% and 21.8%, respectively, compared to the values measured on day zero of the experiment. In contrast, the population of Firmicutes bacteria increased by 12% and 16%, respectively, on these days compared to the values measured on day zero.
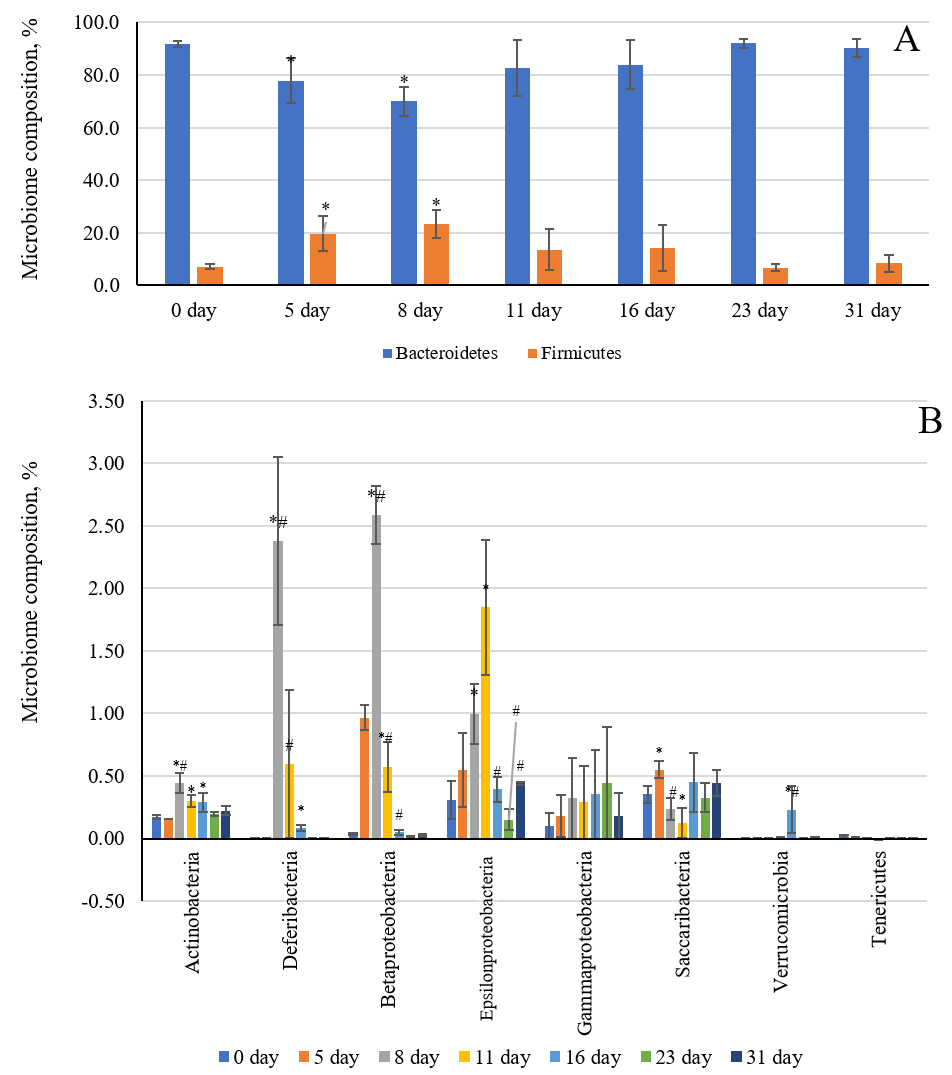 Fig. 3.
Fig. 3.
Changes in the intestinal microbiome of laboratory animals
against the background of their consumption of water with an altered isotopic
composition (2H/1H = 1200 ppm). (A) Bacteroides and
Firmicutes. (B) Actinobacteria, Deferibacteria,
Betaproteobacteria, Epsilonproteobacteria,
Gammaproteobacteria, Saccaribacteria, Verrucomicrobia,
and Tenericutes. *, p
Quantitative changes affected the populations of small representatives of the intestinal microflora. The increase in the number of bacterial types, such as Actinobacteria, Deferibacteria, Betaproteobacteria, and Epsilonproteobacteria, follows a bell-shaped pattern throughout the entire duration of the experiment. Indeed, the maximum growth of these microorganisms occurred between days 5 and 11 of the isotopic substitution of protium for deuterium in the internal environment of the body (Fig. 3B). For the Deferibacteria family, the maximum growth was 2.4%, 2.6% for Betaproteobacteria, and 1.8% for Epsilonproteobacteria, compared to the initial day (day 0) of the experiment. Small quantitative shifts toward an increase were observed in the Actinobacteria phylum, which includes Bifidobacterium and Collinsella. Meanwhile, ambiguous changes in population growth were observed in Saccaribacteria, amid an increase in deuterium within the host organism. By day 5 of the experiment, Saccaribacteria number decreased; from day 8 to 11, the numbers increased, and the numbers remained at the control level in the following days.
Using the isotope mass spectrometry method (Fig. 4), the changes in 2H content in the brain tissue of rats were analyzed after implementing a drinking diet with an increased content of deuterium (1200 ppm).
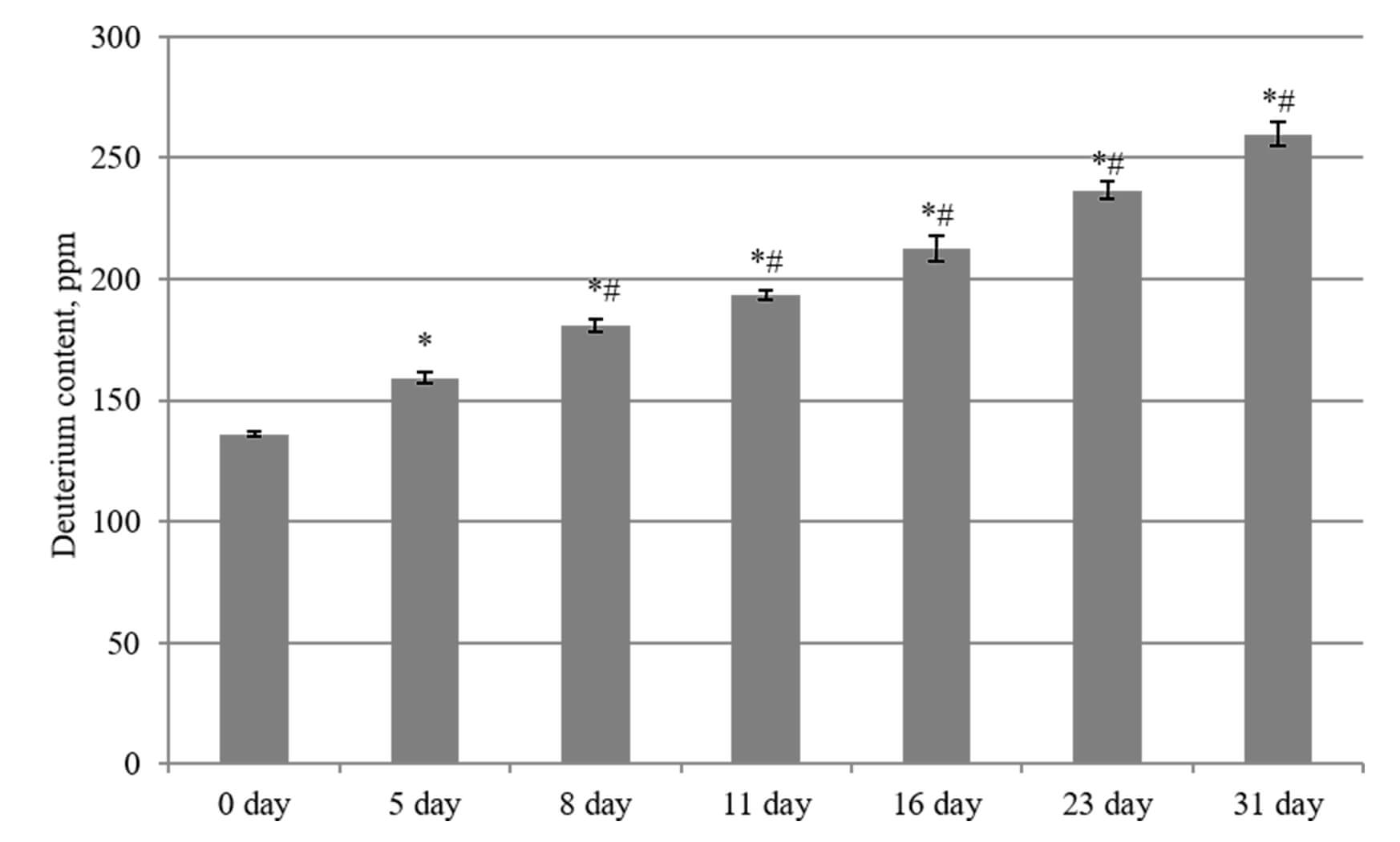 Fig. 4.
Fig. 4.
Changes in deuterium content in the brain tissues of laboratory
animals during their consumption of water with an altered isotopic composition
(2H/1H = 1200 ppm). *, p
The starting 2H level in the brain tissues of the control group (day 0) was
136
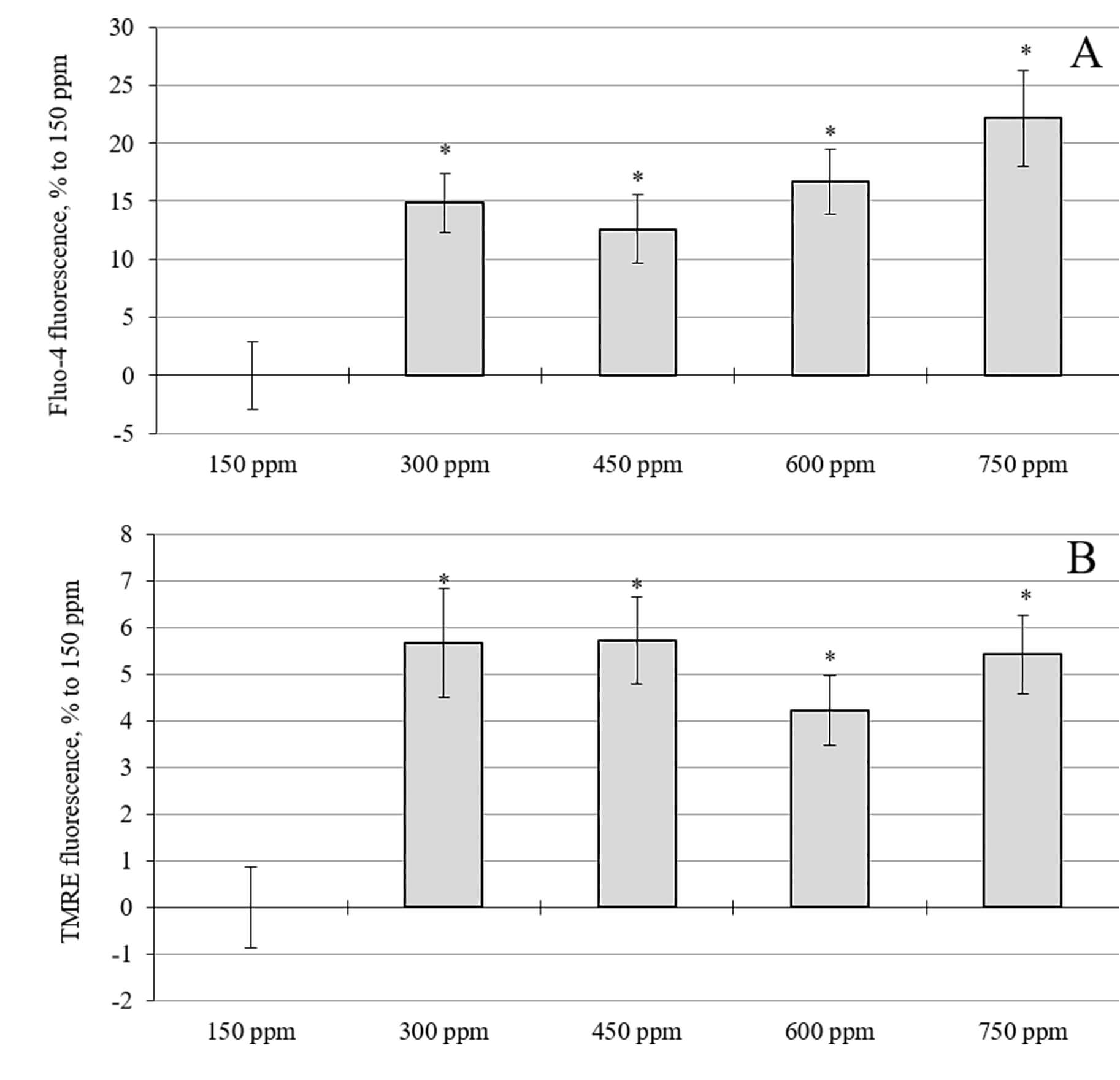 Fig. 5.
Fig. 5.
Effects of the increase in deuterium concentration in the
incubation medium. (A) Intracellular calcium levels in rat cerebellar neuron
cultures. (B) Mitochondrial membrane potential in rat cerebellar cell cultures.
*, p
The effect on neuronal indices of a medium with an increased deuterium content was studied. The minimum deuterium content in the medium was 300 ppm, which approximately matched the content in the brains of rats consuming water with 1200 ppm at the end of the experiment. The results for the calcium level and mitochondrial membrane potential are presented in Fig. 5. Interestingly, an increase in the deuterium concentration in the incubation solution for 24 hours led to a statistically significant increase in the intracellular calcium level in relation to cultures with a standard (natural) concentration of deuterium (150 ppm) (Fig. 5A). The most significant increase in the calcium level was observed at 750 ppm, rising by 19% to 150 ppm, which was significantly higher than the 10–11% increase observed at 300–600 ppm.
Increasing the concentration of deuterium in the incubation solution for 24 hours resulted in a statistically significant, increase in the mitochondrial membrane potential (Fig. 5B). When deuterium was added to the incubation medium at concentrations of 300, 450, 600, and 750 ppm, an average increase of 4–6% was observed in the mitochondrial membrane potential after 24 hours compared to cultures with a deuterium concentration of 150 ppm.
The chemiluminescent method and the TBA method can provide information on the intensity of oxidative processes in biological tissues. The chemiluminescence intensity is proportional to the number of free radicals generated by the system after adding the initiator. The chemiluminescence intensity can increase for several reasons: the production of reactive oxygen species (ROS) by various cell organelles, lipid peroxidation, the accumulation of lipid peroxidation products in tissues, and decreased activity of the enzymatic and non-enzymatic antioxidant systems. The TBA method is not specific and is used to supplement other methods for studying the redox state in biological tissues, particularly in the analysis of chemiluminescence. This biochemical method allows for the indirect determination of the malonic dialdehyde concentration, which is a product of membrane lipid oxidation. The malonic dialdehyde concentration increases in tissues due to oxidative stress. Table 2 shows that the intensity of chemiluminescence in the brain tissues of laboratory animals increases throughout the experiment as deuterium atoms enter the body. On day 5 of the experiment, there was a sharp 28% increase in the total light sum of the glow. By day 8, this indicator had decreased to the initial pre-experiment (day 0) value. The maximum glow in the tissues of the cerebral cortex was observed on days 11 and 16, reaching 121% and 129%, respectively, of the relative control (day 0). Following further consumption of the isotope-modified water, a decrease in the chemiluminescence intensity was observed on the 23rd and 31st days of the experiment. Notably, there was only a tendency toward an increase in this indicator on days 23 and 31 of the experiment; however, no reliable differences were established relative to the control day.
| Parameter | Day 0 | Day 5 | Day 8 | Day 11 | Day 16 | Day 23 | Day 31 |
| Сhemiluminescence intensity, c.u. | 86 |
106 |
85 |
100 |
107 |
94 |
95 |
| Specific catalase activity, mM/min × g | 0.09 |
0.14 |
0.29 |
0.31 |
0.51 |
0.55 |
0.57 |
| Malondialdehyde concentration, nM/mg | 2.2 |
2.1 |
2.4 |
1.9 |
2.7 |
2.5 |
2.7 |
*, p
The concentration of malondialdehyde in the cerebral cortex of the experimental animals remained at the control level (before the start of the experiment) throughout the experiment.
Thus, the intake of deuterium into the mammalian organism promoted a shift in the redox state in the brain tissues. This process was also accompanied by the activation of the antioxidant system, which included an increase in catalase activity and the expression of some genes responsible for synthesizing antioxidant proteins (Fig. 6).
 Fig. 6.
Fig. 6.
Dynamics of changes in gene expression in the brain tissues of
laboratory animals after the consumption of water with an altered isotopic
composition (2H/1H = 1200 ppm). Cat, catalase; Gclc,
glutamate cysteine ligase; Gfap, glial fibrillary acidic protein;
Tnf, tumor necrosis factor; Il-1
It was found that following an increase in the concentration of deuterium in the
cerebral cortex of laboratory animals, the expression of the genes responsible
for synthesizing catalase (Cat), glutamate cysteine ligase
(Gclc), glial fibrillary acidic protein (Gfap), and
Nrf2 (Nfe 2I2) also increased. Notably, a regular pattern was
observed in the changes in the activity of these genes. In the first week of the
experiment, the genes of all proteins listed in Fig. 6 were activated following
the isotopic 2H/1H exchange in the cerebral cortex. In the second and
third weeks of the experiment, the expression of the genes Cat,
Gclc, Gfap, and Nrf2 in this part of the brain was
observed to be passivated. After 4 weeks of the experiment, an increase in the
concentration of deuterium in the brain tissues led to another rise in the
activity of the above genes, except for the Gclc gene. A different
nature of the dependence was observed in the expression of the genes responsible
for the synthesis of proinflammatory molecules, such as tumor necrosis factor
(Tnf) and interleukin 1
Daily deuterium supply to rat brain tissue did not cause any pronounced changes in the behavior of experimental animals in the open field test (Supplementary Table 2). On the 14th day of the experiment, the animals whose diet included deuterium-weighted water with a deuterium content of 1200 ppm had higher horizontal and vertical activity indices compared to control animals. These indices reflect the number of squares crossed and rearing on the hind legs. Notably, the animals in the control group groomed more than those in the experimental group (deuterium-weighted water for 14 days).
The differences in these parameters between the two groups indicate that the animals that drank deuterium-weighted water for 14 days had higher locomotor and exploratory activity than the animals that consumed water with a natural deuterium content (150 ppm). On the 31st day of the experiment, both groups of animals (control and deuterium-weighted water) showed a decrease in both locomotor and exploratory activity compared to the 14th day. Perhaps this pattern is because the animals were repeatedly exposed to an open field. As is known, an open, brightly lit space is a stress factor for rats. No obvious differences in behavior were observed between the two groups of animals on the 31st day of the experiment.
Fig. 7A shows the intensity of the chemiluminescent reaction of rat blood plasma oxidation. The maximum luminescence of 29% occurred in the group of animals that consumed water with a natural deuterium content and were subjected to immobilization stress (group stress-150 ppm).
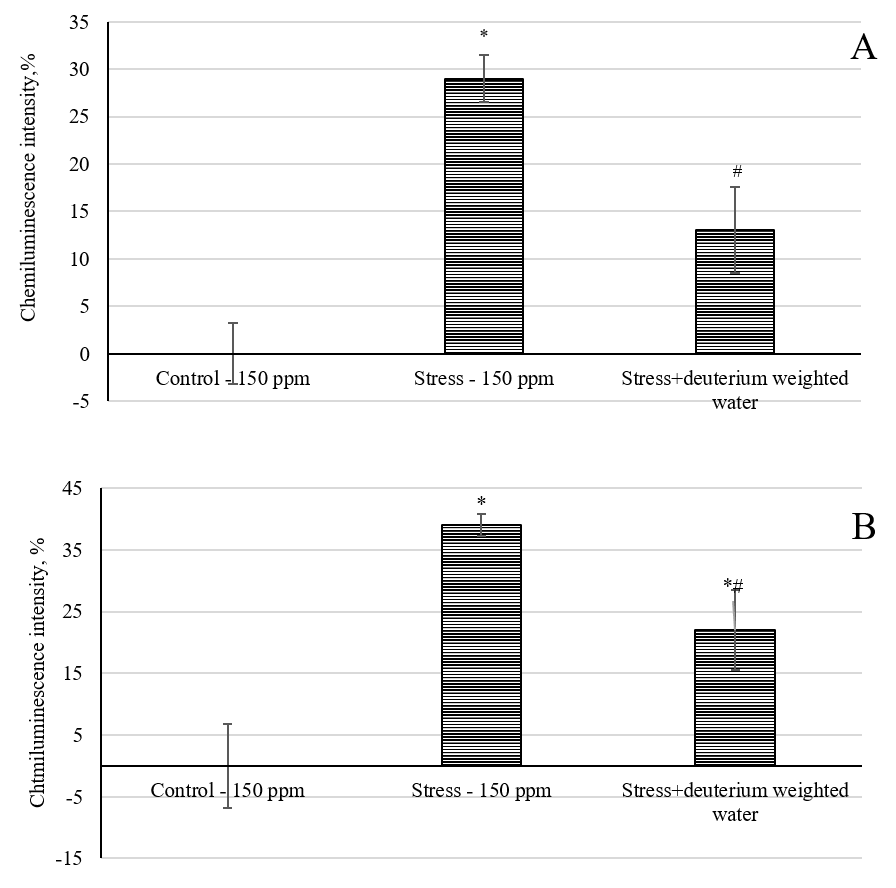 Fig. 7.
Fig. 7.
Effect of deuterium-weighted water (2H/1H = 1200 ppm)
on the chemiluminescence intensity. (A) Blood plasma of laboratory animals under
immobilization stress. (B) Rat cerebral cortex tissues under immobilization
stress. *, p
The increase in the light sum of chemiluminescence for the blood plasma reflects the development of oxidative stress in the animals in this group. Animals that consumed deuterium-weighted water (1200 ppm) for 32 days and were subjected to immobilization stress had a 13% higher light sum of chemiluminescence compared to the control-150 ppm group and a 55% lower value compared to the stress-150 ppm group. Fig. 7B shows the light sum of the chemiluminescent oxidation reaction in the brain tissues of the laboratory animals. The maximum luminescence of 39% (p ˂ 0.05) was observed in the group of animals that consumed water with a natural deuterium concentration of 150 ppm and were subjected to immobilization stress (stress-150 group).
The increase in the chemiluminescence light sum reflects a shift in the antioxidant/prooxidant balance toward prooxidants. Rats that drank deuterium-weighted water (1200 ppm) for 32 days and were subjected to immobilization stress had a lower oxidative stress intensity index, which was accompanied by an increase in the chemiluminescence light sum of 22% relative to the control-150 ppm group, and, accordingly, by 43% less than in the stress-150 ppm group (p ˂ 0.05). Thus, prolonged intake of deuterium-weighted water for 32 days contributed to a decrease in the intensity of the oxidative processes in the blood and brain tissues of laboratory animals.
This study measured the weight of the thymus, adrenal glands, and spleen, and calculated the ratio of these weights to the weight of the animals. Supplementary Table 3 shows that after the induction of immobilization stress, the specific weight of the organs changed in all experimental groups. In stressed animals, whose diet included water with a natural deuterium content (stress-150 ppm group), compared with the control group (control-150 ppm), the specific weight of the thymus decreased by 17% (p ˂ 0.01), and the particular weight of the adrenal glands increased by 20% (p ˂ 0.01). The spleens in the animals in the stress-150 ppm group were also enlarged, although the weights did not differ significantly from the control group.
A similar trend in the change in the specific gravity of the thymus, adrenal glands, and spleen was observed in the stressed animals that consumed deuterium-weighted water for 32 days, as well as in the stress-150 ppm group. Notably, a more pronounced depletion of the thymus (p ˂ 0.05) was observed in the stress + deuterium-weighted water (1200 ppm) group than in the stress-150 ppm group. No statistically significant difference was observed for the other two organs.
Thus, following the induction of stress, there was a change in the specific gravity of the endocrine glands that produce the main stress hormones, as well as the organs associated with the immune system.
The behavioral reactions of the animals were studied in the open field and elevated plus maze tests (Supplementary Tables 4,5). Both tests provide information on the emotional state and locomotor activity of the animals.
A decrease in horizontal and vertical activities, a decline in entries into the center of the field, and an increase in grooming were observed in the open field test for the stressed animals, whose diet included water with a natural content of deuterium (stress-150 ppm), compared to the control group (control-150 ppm). In the elevated plus maze test, the stressed animals, whose diet included water with a natural content of deuterium (stress-150 ppm), made fewer entries into the closed and open arms, as well as fewer hangings from the open arms and vertical rises on the hind legs, compared to the control group (control-150 ppm). However, no approaches to the distal edge were observed in this group of animals, while the number of groomings significantly increased. Thus, a decrease in motor and exploratory activities was observed in the animals in the stress-150 ppm group, due to the development of a depressive state induced by immobilization stress.
Moreover, increased anxiety was observed in stressed animals that consumed
deuterium-weighted water for 32 days, as well as in the stress-150 ppm group,
compared to the control group; this was also indicated by a decrease in the motor
and exploratory activities of these animals. However, the motor and exploratory
activity indices were higher in the deuterium-weighted water group than in the
stress-150 ppm group. Animals in the stress + deuterium-weighted water group
(1200 ppm) crossed a 30% significantly (p ˂ 0.05) higher number of
squares in the open field (horizontal activity) than animals in the stress-150
ppm group; meanwhile there was a tendency (p
When assessing the intrinsic tryptophan fluorescence of BSA, the fluorescence intensity of albumin was shown to decrease in a dose-dependent manner as the concentration of deuterium in the medium increased (Supplementary Fig. 2).
There is also a decrease in the intensity of the difference in absorption of left- and right-handed polarized light in the circular dichroism spectra (Fig. 8).
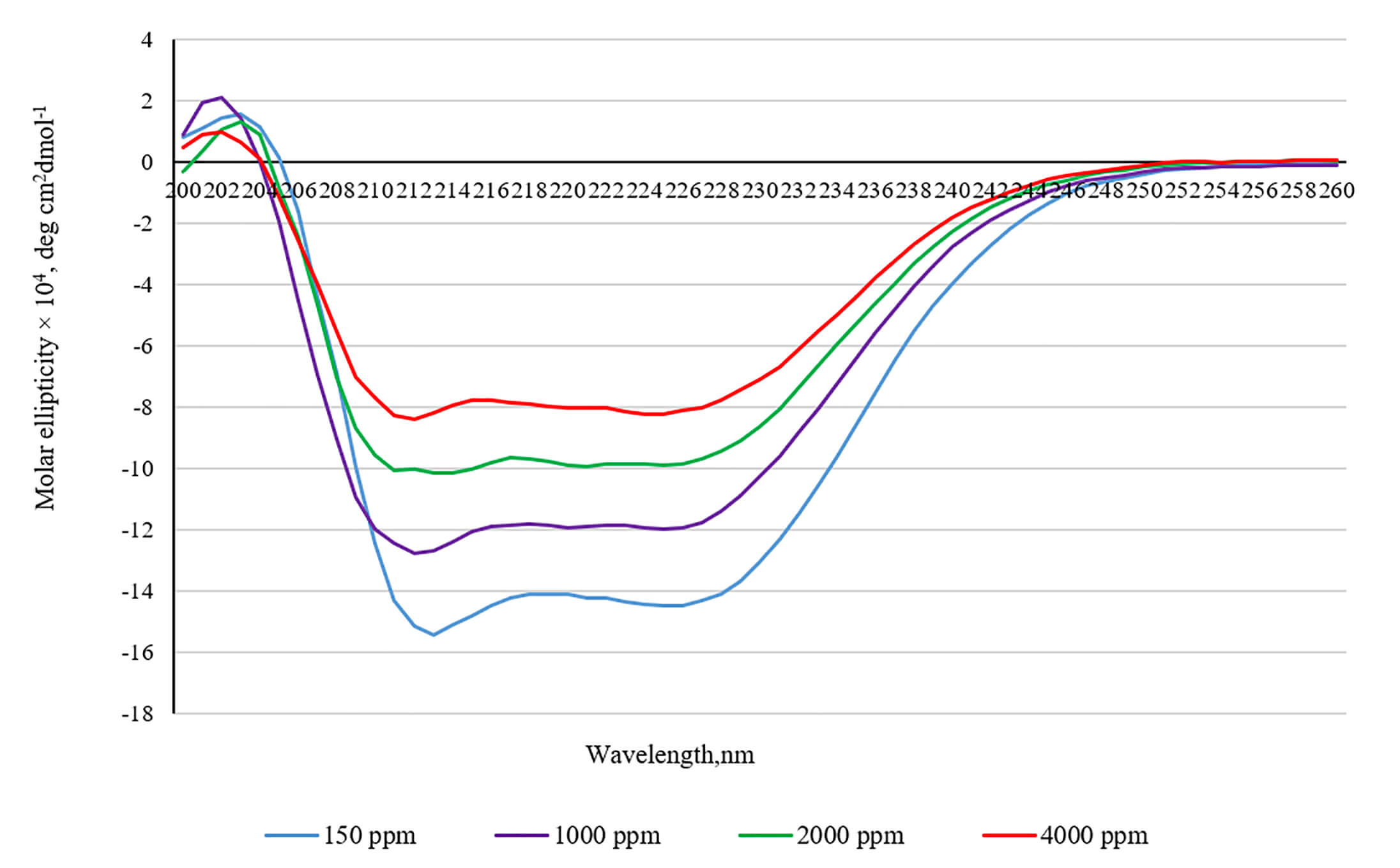 Fig. 8.
Fig. 8.
Circular dichroism spectra of bovine serum albumin in the media with increased deuterium concentration.
The intensity of the CD spectra at 208 and 220 nm can be employed to determine
the percentage of
There is a gradual increase in the concentration of deuterium in the blood plasma and brain tissues of laboratory animals after consuming water with increasing contents of deuterium. The rise in deuterium concentration in the extracellular fluid and tissues of the body occurs due to the isotopic exchange of 2H/1H in proteins, lipids, and nucleic acids that form cells. It is known that such substitution occurs most actively in functional groups with a lone electron pair, which are capable of forming hydrogen bonds. Such atomic groups include hydroxyl (-OH), carboxyl (-COOH), and amino groups (-NH2) [57, 58]. The transition of protons and deuterons from one biomolecule to another is realized along chains of hydrogen bonds using the Grotthuss mechanism. Moreover, the active substitution of deuterium for protium in the body is facilitated by isotopic exchange, occurring between these groups of biomolecules and the hydrate shell through hydrogen bonds [21, 59, 60, 61]. Thus, due to the active isotopic 2H/1H exchange in biomacromolecules, the internal fluid and tissues in the body are enriched with deuterium [19].
Meanwhile, any change in the constancy of the internal environment of the body leads to the development of a stress reaction. When the intensity of the stress reaction does not exceed the stress-limiting system of the body, the effect of the stress is reduced through compensatory mechanisms and adaptation to this stressor or a different one. This process is ensured by the activation of regulatory systems and effector organs, which contribute to a new level of functional state in the body under changed environmental conditions [62]. Our study identified that a change in the constancy of deuterium in the body leads to the development of a mild stress response and an adaptation syndrome, and we also suggest several mechanisms through which these responses are implemented. Data in the literature indicate that low and high concentrations of deuterium at the cellular level may impact mitochondria [38]. It has been shown experimentally that changing the deuterium concentration in the medium shifts the redox potential of mitochondria in cancer and normal cells [11, 63, 64, 65, 66]. This is expressed by changes in the level of oxidized mitochondrial proteins and the activation or inhibition of ROS production in these organelles [67, 68], depending on the deuterium content in the environment [58, 69]. Moreover, a shift in the 2H/1H ratio in the medium reduced the efficiency of oxidative phosphorylation in rat liver mitochondria [70]. In our studies on primary cerebellar cell cultures, we also found that incubation in a medium with a high deuterium content (300–750 ppm) increased the mitochondrial membrane potential and intracellular calcium level. The deviation in these metabolic parameters by the cell may be associated with changes in the activity of proteins in the electron transport chain, which are involved in forming a proton gradient on both sides of the inner mitochondrial membrane, as well as changes in the function of Ca2+ exchangers. As is known from prior research, the isotopic 2H/1H exchange in proteins and nucleic acids leads to a change in their kinetic and thermodynamic parameters [71]. Therefore, isotopic 2H/1H exchange may increase the possibility of finding deuterium in the proton channels of complexes I and III, as well as in the ATP synthase of complex IV in the electron transport chain [72]. As is known, proton transfer in proton pumps, ETC translocase is conducted along a chain of hydrogen bonds between amino acid residues. The entry of deuterium into one of the specified carrier proteins leads to the strengthening of the hydrogen bond and a change in their conductivity. Maintenance of intracellular calcium ion homeostasis is mainly associated with the Na+/Ca2+ pump in the cytoplasmic membrane and the Na+/Ca2+ pump in the endoplasmic reticulum; the Na+/Ca2+ exchanger is an ATP-dependent enzyme [12]. Hence, the disruption of ATP synthesis caused by an increase in the mitochondrial membrane potential can lead to an imbalance in the activity of calcium transporters. The subsequent increase in calcium ions in the cytoplasm may be associated with its release from the endoplasmic reticulum. However, this assumption requires additional research. Additionally, we assume that the above processes may contribute to the development of a stress reaction in brain cells associated with an increase in deuterium content. As is known, a change in the mitochondrial membrane potential and an increase in the concentration of cytoplasmic calcium contribute to the production of active oxygen species in mitochondria [73, 74, 75]. Our experimental results show that an increase in the production of free radicals in brain tissues accompanies an increase in the deuterium concentration. The maximum shift in redox balance is observed at the initial stages of exposure, when the 2H/1H exchange rate in the blood and brain tissues peaks. Meanwhile, the intensity of active oxygen and nitrogen species production decreases with longer exposure, as indicated by a reduction in the chemiluminescence light sum. Notably, despite the increase in the intensity of the chemiluminescent reaction, the concentration of malondialdehyde in brain tissues remained unchanged throughout the experiment. These data may indicate a moderate activation of free radicals in the cerebral cortex tissues, which does not lead to oxidative damage to the lipids in the cytoplasmic membrane of neurons. The observed shift in redox balance in brain tissues may be associated with an increase in mitochondrial ROS production, as well as an increase in membrane potential and calcium levels in the cytosol. As is known, even moderate production of ROS in the cell promotes the activation of endogenous antioxidant proteins. The results of our studies show that an imbalance in the antioxidant system occurs at the initial stages of exposure. Indeed, the catalase activity increased and remained high until the end of the experiment. Changes in catalase activity in brain tissues result from a compensatory response to a shift in redox balance. During the experiment, it was noted that changes in the expression of genes responsible for the synthesis of Cat, Nrf2, and Gfap have a U-shaped dependence. The expression of these proteins was activated in the first and last weeks of the experiment, whereas their inactivation was observed during the second and third weeks. The activity of the gene encoding Gclc was only observed during the first week. Therefore, the dynamics of expression of these observed proteins could be the result of adaptation development. The dynamics of gene expression of the above proteins correlate with those of free radical production and are a consequence of their production. The transcription factor Nrf2 activates the genes of antioxidant proteins that are required to restore the redox balance [76]. Gamma-glutamylcysteine synthetase (Gclc) is an enzyme required for synthesizing reduced glutathione. Thus, the Gclc gene expression is enhanced in response to stress factors that lead to increased production of reactive oxygen and nitrogen species [77]. Increased expression of Gclc in the initial phase of the experiment is also one of the links in the development of the adaptation syndrome against the background of changes in the deuterium content. The glial fibrillary acidic protein (Gfap) is involved in forming the cytoskeletal structure of glial cells. Astrocytes possess the highest Gfap activity. Moreover, Gfap is considered a marker of neurodegenerative processes, and the expression of Gfap is increased in Alzheimer’s disease, cerebral ischemia, and gliomas [78], which is cause for concern. However, Gfap levels have also been shown to increase under moderate influences: chronic mild stress and social isolation in mice [79], and moderate physical activity [80].
The increase in Gfap gene expression may be associated with the activation of astrocytes and their participation in adaptation processes in response to a rise in the concentration of deuterium in brain tissue. Thus, based on studies of cerebellar cell culture and biochemical analyses, a picture of the development of adaptation syndrome is observed due to changes in deuterium homeostasis. However, notably, the deuterium drinking ratio did not negatively affect the behavior of the experimental animals in the open field test. After two weeks of exposure, positive behavior dynamics were observed relative to the animals that had water with a natural deuterium content. This was expressed as an increase in exploratory and motor activities in the open field test. After four weeks of exposure, the behaviors of the experimental animals did not differ from those of the control animals. It can be assumed that a more positive emotional state in these animals is associated with the action of signaling molecules, which are activated as a result of a compensatory response to deuterium stress and affect the sensory–motivational and motor-reinforcing links of the integrative brain system [81].
Clinical and experimental evidence exists of a bidirectional relationship between the gut and the brain. The gut microbiota influences brain development and the production of neurotransmitters, metabolites, and hormones into the blood and lymphatic system through neuronal, endocrine, and immune pathways [82]. A disrupted microbiome in laboratory animals caused the development of an acute stress response, anxiety, and anhedonia [83]. Experimental models of gut microbiota alterations in laboratory animals can alter responses to various types of stress, activate or reduce anxiety and depression, and influence the hypothalamic–pituitary–adrenal axis [84, 85, 86]. A correlation has also been established between the likelihood of developing depression and schizophrenia in people and changes in the composition of their microflora [87].
Following an increase in the deuterium content in the blood and tissues of
laboratory animals, a change was observed in the ratio of the number of bacteria
inhabiting the intestines of these animals. It is possible that a change in the
metabolic state of brain tissues, resulting from the compensatory response to an
increase in deuterium, causes a change in the quantitative composition of the
microbiome. The results show that the Firmicutes/Bacteroides
population ratio, along with changes in the numbers of Actinobacteria,
Deferibacteria, Betaproteobacteria, and
Epsilonproteobacteria, exhibit a bell-shaped dependence and correlate
with alterations in the antioxidant/prooxidant balance in brain tissues.
According to the brain–gut relationship theory, changes in the quantitative
composition of the microbiome in the intestines of laboratory animals result from
alterations in biochemical and metabolic processes in brain tissues. These
alterations occur as a compensatory response to a change in deuterium
homeostasis. Increased production of free radical processes and an increase in
intracellular calcium ions lead to the activation of astrocytes (an increase in
the expression of the gene encoding Gfap was established) [88].
Activated astrocytes produce numerous proinflammatory cytokines, such as
interleukins and tumor necrosis factor (an increase in the expression of genes
encoding Tnf and Il-1
In addition, it is necessary to note another important aspect related to deuterium and the microbiome. The possibility of deuterium accumulation by intestinal microorganisms, through the simultaneous synthesis and entry into host cells of organic molecules with a reduced content of deuterium (butyrate, acetate, propionate) has been shown. Then, butyrate is included in the tricarboxylic acid cycle in mitochondria [95]. Thus, fractionation of deuterium occurs. Meanwhile, the studied metabolites of creatine, betaine, and isobutyrate are substrates for the synthesis of low-deuterium hydrogen gas by intestinal bacteria. It has been established that microbial hydrogenase produces hydrogen gas, which is 80% depleted in deuterium [96]. The Bacteroidetes and Firmicutes we studied are also hydrogen-producing bacteria [97]. Although the total number of these bacteria did not change, the fluctuations in the numbers of each individual were quite significant. Hence, consuming deuterium-enriched water causes fluctuations in the number of hydrogen-producing bacteria potentially involved in the fractionation of deuterium in biological molecules. Additionally, changes in the levels of creatine, betaine, and isobutyrate in the blood observed in our experiments may be associated with the activation of certain representatives of intestinal microflora.
We assume the presence of another possible mechanism for the development of the adaptation syndrome against the background of deuterium homeostasis disturbance. This mechanism may be associated with a change in the native structure of blood plasma proteins and cellular protein components. The results of our studies show that an increase in the concentration of deuterium in the medium promotes a change in the structure of the model protein. Indeed, an increase in the deuterium content in the medium to 1000, 2000, and 4000 ppm altered the spectra of intrinsic tryptophan fluorescence of BSA and induced changes in the spectra of circular dichroism. Thus, considering the obtained results of optical studies, it can be concluded that the strengthening of hydrogen bonds leads to conformational rearrangements in the BSA molecule. Furthermore, the loss of a percentage of alpha helicity indicates changes in the secondary structure of BSA. Interestingly, BSA is known to possess two tryptophan residues: one on the surface of the molecule and the other inside the molecule. The main quenchers of tryptophan fluorescence are solvent molecules (water) and tyrosine residues, which quench fluorescence through energy transfer via the dipole–dipole mechanism. It can be assumed that the decrease in the intensity of tryptophan fluorescence may be a consequence of the increased availability of tryptophan residues to the solvent and the increased probability of energy transfer from tryptophan to tyrosine residues. Thus, the optical studies indicate a modification of the BSA structure in a medium with a high deuterium content.
This work also established a change in the intrinsic tryptophan fluorescence spectra of blood serum proteins, indicating possible structural changes in these proteins. The literature contains data on the effect of high and low concentrations of deuterium on the structure and functional properties of protein molecules [40]. A change in the native structure of proteins in the body leads to the activation of a system aimed at their restoration. This system includes a group of chaperone proteins, which include several families classified according to their molecular weight. The main representatives of this group are heat shock proteins, which are activated by stress factors [98]. In addition to their canonical function, heat shock proteins participate in cellular signaling, regulating important cellular processes [99]. Therefore, partial modification of the protein components of the cell caused by an increase in the concentration of deuterium can lead to the development of adaptive potential due to the activation of heat shock proteins.
The change in the metabolic profile of blood plasma and the leukocyte formula we have established is a response to stress caused by an increase in the content of deuterium in the blood and tissues of laboratory animals. Our study observed an increase in lymphocyte numbers and a decrease in segmented neutrophils from days 10 to 16 of the experiment, with a complete recovery to control values noted by the 3rd and 4th weeks. The results we obtained are in good agreement with the experimental data of Yaglova et al. [100]. The authors of this work found that a decrease in deuterium in the body of laboratory animals leads to a reduction in the content of CD3+ cells in the thymus at the initial stages of the experiment; however, on the 14th day of water consumption with a modified isotopic composition, the percentage of mature T cells again exceeded control values in the animals. The changes in the metabolic profile of blood plasma that we observed may be a consequence of changes in the functioning of the endocrine system [12, 101] or alterations in hematopoiesis resulting from a response to deuterium stress [102].
Interest in the distribution and properties of Martian water is increasing in connection with a possible expedition to Mars in the near future [103, 104]. In our study, after consuming an increased deuterium content, a moderate stress reaction developed, associated with the adaptation to an altered H2/H1 homeostasis in the internal environment. A change in the redox potential of cells, an increase in catalase activity, an increase in the expression of the Cat, Gclc, and Nrf2 genes responsible for the synthesis of antioxidant proteins, and the Gfap gene are compensatory responses to deuterium stress. Fluctuations in the number of representatives of the intestinal microbiome, as well as changes in the metabolic profile, also reflect the adaptation process. The established stress-protective effect of water, which is similar to Martian water in terms of deuterium content, results from the development of cross-adaptation. The activation of the antioxidant system, a compensatory effect on deuterium stress, contributed to a decrease in the intensity of oxidative stress and normalization of behavioral reactions in stressed rats whose diet included a drinking ration with an increased content of deuterium. It should be noted that, although prolonged consumption of water with an increased content of deuterium caused the development of stress-protective mechanisms, the change in the mass of the adrenal glands and thymus was more pronounced in this group. This effect occurs because stressed animals, whose diet included water with an increased content of deuterium, experienced double the stress compared to those with a natural drinking ration. The first stress is linked to a change in deuterium homeostasis, and the second is related to the effect of immobilization stress.
Thus, an increase in the concentration of deuterium in the body leads to the development of a stress reaction and the inclusion of compensatory mechanisms aimed at adapting to these conditions. Therefore, the antioxidant system is activated, and the protective properties of the body against immobilization stress increase.
The results of our work provide the prerequisites for further research to study the effects of longer-term use of deuterium-enriched water, including throughout the life of laboratory animals.
VSMOW, Vienna Standard Mean Ocean Water; TBA, thiobarbituric; MDA, malondialdehyde; GSH, reduced glutathione; BSA, bovine serum albumin; CL, chemiluminescence; ROS, reactive oxygen species; Cat, catalase; Gclc, glutamate cysteine ligase; Gfap, glial fibrillary acidic protein; Tnf, tumor necrosis factor.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Conceptualization, SK, AK and SD; methodology, SK, AK and OL; validation, EC, AM and LF; formal analysis, VN and PB; investigation, KP; data curation, OL and VI; writing—original draft preparation, SK, AK and SD; writing—review and editing, AD and SD; visualization, AD; supervision, LF. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The experiments were carried out in accordance with the requirements of the “Guide for the Care and Use of Laboratory Animals”, European Community Directives 2010/63/EU and “Guide for working with laboratory animals, including the ethical principles of animal testing (3R principle) of the V.M. Gorbatov Federal Research Center for Food Systems of the Russian Academy of Sciences”. The study was approved by the bioethical commission of the V.M. Gorbatov Federal Research Center for Food Systems of the Russian Academy of Sciences (Protocol No. 7/2023 on 7 Mart 2023).
Not applicable.
This work was supported by the State Task Force of the Southern Scientific Center of the Russian Academy of Sciences for 2025, state registration number 125011700394-5.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBE39086.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








