1 Department of Field Plant Breeding and Agroecology, Institute of Agricultural Resources and Economics, LV-4126 Priekuli, Cesis municipality, Latvia
2 Faculty of Agriculture and Food Technology, Latvia University of Life Sciences and Technologies, LV-3004 Jelgava, Latvia
3 Department of Chemical Engineering and Environmental Technology, Faculty of Chemistry, University of Oviedo, 33006 Oviedo, Spain
Abstract
Pseudomonas taetrolens is a highly versatile microorganism that has gained significant attention in biotechnology due to its metabolic adaptability and ability to thrive in diverse environmental conditions. Thus, since P. taetrolens efficiently metabolizes organic compounds, P. taetrolens is a promising candidate for sustainable industrial applications. P. taetrolens demonstrates substantial potential in waste utilization by converting dairy byproducts, such as whey, into value-added compounds, including lactobionic acid, thereby advancing the principles of circular economic frameworks. This review provides a comprehensive analysis of the characterization, structural features, and diverse applications of P. taetrolens. In particular, this review explores the enzymatic mechanisms underlying the metabolic pathways of P. taetrolens, focusing on its role in lactose oxidation into lactobionic acid. Additionally, this review underscores the potential of microorganisms in industrial biotechnology and sustainable development practices by showcasing recent advances and ongoing research. This analysis demonstrates how P. taetrolens contributes to innovative solutions in waste utilization, environmental sustainability, and the production of value-added compounds across sectors, including food, pharmaceuticals, and cosmetics.
Keywords
- Pseudomonas taetrolens
- lactobionic acid
- lactose oxidation
- biotechnology
- whey
- metabolic pathways
Exploring microbial systems has continuously advanced the boundaries of biotechnology, particularly in addressing sustainability and industrial efficiency challenges. The Pseudomonas species, recognized for their metabolic versatility and ecological adaptability, have emerged as essential organisms in industrial and environmental applications. These Gram-negative, rod-shaped bacteria exhibit unique structural and functional features, such as their outer membrane lipopolysaccharides (LPS) and enzymatic repertoire, which enable them to thrive in diverse habitats and interact effectively with complex substrates [1, 2]. Of particular interest is Pseudomonas taetrolens, a non-pathogenic bacterium known for degrading various organic materials and transforming low-value waste into value-added compounds. The flexibility of P. taetrolens in utilizing diverse nutrients and strong enzyme systems makes this bacterium important in environmental and waste recycling efforts [3]. One of the most significant applications of P. taetrolens is its ability to oxidize lactose into lactobionic acid, a compound with extensive utility in the food, pharmaceutical, and cosmetic industries [4]. These capabilities highlight the role of P. taetrolens in advancing circular economy practices by converting industrial byproducts, such as dairy whey, into valuable economic and environmental substances [5].
Nonetheless, despite the growing industrial applications of P. taetrolens, the full potential of this bacterium remains underexplored. Moreover, the increasing demand for sustainable biotechnology solutions, particularly in waste utilization and industrial bioprocessing, underscores the need for an in-depth review of this versatile bacterium. Therefore, this review provides a comprehensive analysis of the taxonomy, structural characteristics, and enzymatic mechanisms of P. taetrolens, along with the implementation of this bacterium in biotechnological applications. Further, this review has specifically focused on the recent studies that have explored the use of P. taetrolens in the sustainable production of lactobionic acid. This review also synthesizes current knowledge of the adaptive strategies performed by P. taetrolens, highlighting its potential for innovative solutions in waste management, industrial biotechnology, and sustainable development. Understanding the enzymatic mechanisms, adaptive strategies, and metabolic pathways through which P. taetrolens function is essential for optimizing the application of P. taetrolens in bioprocessing and other procedures.
This review employs the monographic method and comprehensively examines the application and potential of P. taetrolens. This study primarily focused on information from scientific platforms, including the Web of Science, PubMed, SCOPUS, and ScienceDirect databases. A total of 77 scientific publications, encompassing experimental studies, reviews, and patents, were analyzed. The selected studies span diverse countries and regions, reflecting the global interest in P. taetrolens. Research contributions were identified from countries including Spain, South Korea, the United States, Italy, Latvia, Benin, and Japan. The scope of these studies included optimizing culture conditions, enzymatic pathways, downstream purification techniques, and applying P. taetrolens in sustainable practices. The wide geographical representation of research highlights the global interest in leveraging P. taetrolens for biotechnological innovation.
Pseudomonas species are aerobic, non-spore-forming, Gram-negative, rod-shaped, either straight or slightly curved bacteria, measuring 0.5–1.0 µm in diameter and 1.5–5.0 µm in length. These bacteria exhibit a smooth and shiny appearance due to their outer membrane, a characteristic feature of Gram-negative bacteria. This outer membrane contains lipopolysaccharides, which contribute to their structural integrity and resistance to certain environmental stressors [1, 6, 7]. p. taetrolens is particularly noted for its metabolic versatility, allowing this bacterium to thrive in various environments, including soil and aquatic systems. This bacterium is motile and utilizes one flagella for movement. Additionally, P. taetrolens exhibits a pronounced aerobic respiratory metabolism, although in some cases, it can use nitrates as an alternative electron acceptor, enabling growth under anaerobic conditions. Most species are oxidase-positive [2, 8]. Taxonomy and classification of P. taetrolens:
- Domain: Bacteria
- Phylum: Proteobacteria
- Class: Gammaproteobacteria
- Order: Pseudomonadales
- Family: Pseudomonadaceae
- Genus: Pseudomonas
- Species: Pseudomonas taetrolens.
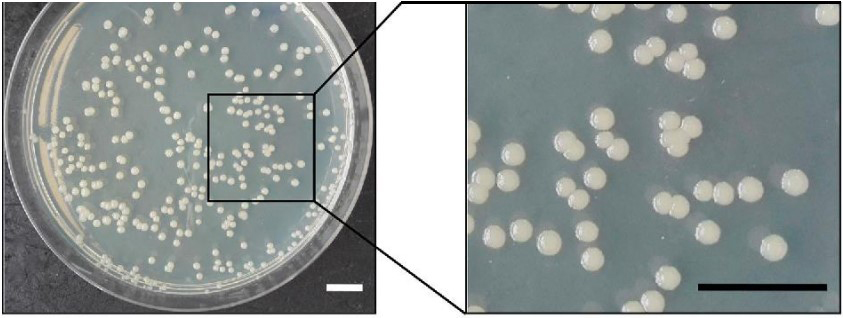
The metabolic capabilities of P. taetrolens include the ability to utilize a wide range of organic compounds as carbon and energy sources. Various species within the Pseudomonas genus, including P. taetrolens, possess a diverse enzymatic repertoire that enables the degradation of complex organic materials, making them valuable in bioremediation processes across different environmental contaminants [9, 10, 11, 12]. Members of this genus are characterized by their ability to grow in simple, neutral environments. Pseudomonas spp. colonies are typically colorless but can also exhibit white, greyish-white, creamy, or yellow pigmentation, as seen in Fig. 1 (Ref. [13]), which depicts P. taetrolens colonies on agar plates; organisms from older cultures may appear slightly pleomorphic. Fluorescent colonies can be easily observed under ultraviolet light [2, 4].
The optimal growth temperature for P. taetrolens is 30 °C [14], though this bacterium can survive at 4–5 °C for several weeks. Moreover, pseudomonas spp. resume active proliferation upon transfer to fresh media [1]. P. taetrolens has a three-layered cell envelope; see Table 1 (Ref. [1, 15, 16, 17]).
| Cell component | Description |
| Cell wall | Inner membrane: Phospholipid bilayer that controls substance flow. Peptidoglycan layer: Thin layer that provides shape and stability. Outer membrane: Lipopolysaccharide-rich barrier against toxins and antibiotics, which triggers immune responses. |
| Pili | Aid in attachment, biofilm formation, and cell interactions. Enhance adaptability in diverse habitats. Involved in conjugation, promoting genetic exchange and potential antibiotic resistance. |
| Cytoplasm | A gel-like substance that contains enzymes, ribosomes, nucleic acids, and other molecules essential for cell metabolism and growth. |
| Nucleoid | DNA is located in the nucleoid region as a single circular chromosome with genetic information. |
| Ribosomes | The cell contains 70S ribosomes, which are responsible for protein synthesis and are composed of proteins and ribosomal RNA (rRNA). |
| Flagella | Enable movement in liquids, helping P. taetrolens respond to chemical stimuli. |
| Extracellular polymeric substances | Capsules or extracellular polymeric substances that help the bacteria adhere to surfaces and survive in adverse conditions. |
| Plasmids | Small, circular DNA fragments that often possess genes for antibiotic resistance or other advantages. |
Based on the information in Table 1, the authors created an image of P. taetrolens (Fig. 2).
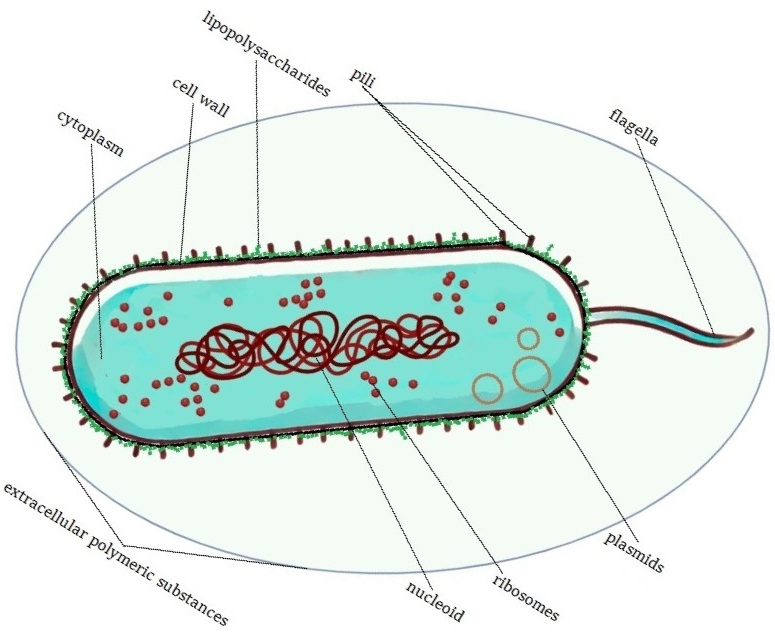 Fig. 2.
Fig. 2.
Structure of Pseudomonas taetrolens. Created in BioRender. Sarenkova, I. (2025) https://BioRender.com/egvekit.
In addition to extracellular polymeric substances (EPS), Pseudomonas species produce various metabolites that significantly influence biofilm formation. For instance, Pseudomonas aeruginosa synthesizes pyocyanin, a redox-active pigment that enhances biofilm development by modulating gene expression and generating reactive oxygen species (ROS), promoting bacterial adhesion and structural integrity. Similarly, pyoverdine and pyochelin, both siderophores, facilitate iron acquisition under iron-limited conditions, which is crucial for biofilm maturation and stability. While specific studies on P. taetrolens are limited, the close relationship between p. taetrolens and P. aeruginosa suggests that P. taetrolens may produce analogous metabolites contributing to biofilm formation. Hence, understanding these mechanisms is vital for developing strategies to control biofilm-related issues in industrial and clinical settings [18, 19]. LPS in Pseudomonas spp. comprise three main components: lipid A, a core polysaccharide, and an O-antigen. LPS play a crucial role in structural integrity and act as endotoxins. Moreover, LPS can be released upon the death of Gram-negative bacteria, triggering strong immune responses in hosts. In both pathogenic and non-pathogenic bacteria, LPS contribute to survival by protecting against environmental stressors such as pH changes, temperature shifts, and antimicrobial compounds. In Pseudomonas spp., LPS form a dense layer on the outer membrane, covering up to 75% of the surface and acting as a barrier [20, 21]. Pili, also present in Pseudomonas spp., facilitate surface attachment and biofilm formation, aiding nutrient acquisition and stability in natural environments such as soil and water. Additionally, pili can mediate horizontal gene transfer, enhancing bacterial adaptability by sharing genes related to stress resistance and metabolic functions [22, 23]. While LPS and pili are both surface structures, they serve distinct roles; LPS provide protection, whereas pili protrude above the LPS layer to interact with the environment [21, 23]. The cytoplasm in P. taetrolens, similar to other bacterial cells, contains several essential components that support cell metabolism and growth, including enzymes, ribosomes, nucleic acids, metabolites, precursors, small molecules, ions, and other macromolecular complexes. The enzymes in P. taetrolens are crucial for metabolic processes, enabling efficient oxidation–reduction reactions essential for energy production and amino acid metabolism. These enzymes also allow the bacterium to degrade complex molecules, such as carbohydrates, into simpler forms for energy utilization while further supporting DNA replication, repair, and other vital metabolic pathways [16, 24]. Pseudomonas spp. can oxidize amino acids and carbohydrates [25], including lactose, to produce lactobionic acid, a compound with significant biotechnological applications [5, 26]. The ribosomes in P. taetrolens are of the 70S type, typical of prokaryotic cells, and consist of a large 50S subunit and a small 30S subunit. These ribosomes are responsible for synthesizing proteins by translating mRNA into polypeptides, which are essential for cell structure, enzymatic functions, and various cellular processes. The nucleic acids in P. taetrolens include a single circular chromosome in the nucleoid region, which contains all the genetic information required for cell growth, metabolism, and reproduction. Various forms of RNA are also present in the cytoplasm, such as mRNA, which carries genetic instructions from DNA for protein synthesis; transfer RNA (tRNA), which delivers specific amino acids to the ribosomes during protein synthesis; rRNA, which forms part of the ribosome structure, facilitating protein synthesis. Cytoplasmic metabolites and precursors play critical roles in metabolic pathways. ATP serves as the primary energy currency in the cell, while NADH and NADPH act as electron carriers in redox reactions essential for respiration and biosynthesis. The cytoplasm also contains free amino acids for protein synthesis and intermediates for other metabolic pathways, along with lipids and fatty acids that serve as precursors for membrane synthesis and modification [16, 24, 27]. Small molecules and ions, such as K+, Na+, Mg2+, and Ca2+, are vital for maintaining osmotic balance and enzyme activity [28]. Cofactors, including iron–sulfur clusters, heme, and coenzymes, such as coenzyme A (CoA), flavin adenine dinucleotide (FAD), and flavin mononucleotide (FMN), assist in various enzymatic reactions. Finally, other macromolecular complexes, such as proteasomes and chaperonins, play a role in protein folding and degradation, ensuring the maintenance of protein quality control within the cell [29, 30]. This structural composition enables P. taetrolens to survive and adapt to diverse substrates and environmental conditions. Moreover, the ability of P. taetrolens to thrive in such varying settings is attributed to its robust cellular mechanisms, including versatile metabolic pathways and enzymatic systems that efficiently utilize available resources, even under challenging conditions.
P. taetrolens is commonly found in naturally spoiled products [31]. Stodola and Lockwood [32] were the first to discover strains of the Pseudomonas genus capable of producing lactobionic acid by oxidizing lactose. These authors demonstrated that lactobionic acid could be obtained microbiologically [32]. Among the 15 studied Pseudomonas species, only Pseudomonas graveolens (now known as P. taetrolens) could oxidize lactose into lactobionic acid, with 75% of the initial lactose in rotary drums converted into lactobionic acid within 165 hours [32]. Pseudomonas species are typically cultured in bioreactors or flasks using orbital shakers, as agitation ensures proper contact between nutrients and cells, facilitating oxygen uptake. These bacteria are sensitive to high salt concentrations and extreme pH levels, which can reduce their growth capacity [1].
Several studies have investigated whey fermentation using P. taetrolens [3, 4, 5, 13, 14, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42]. These studies found that the biomass of P. taetrolens affects lactobionic acid productivity; higher initial biomass leads to greater lactobionic acid production [4]. Maintaining an appropriate pH during fermentation is crucial, as P. taetrolens growth decreases below a pH of 6, negatively impacting lactobionic acid yield. Similarly, pH levels above 7 adversely affect both bacterial growth and lactobionic acid production [4]. In addition to pH, the oxygen supply during fermentation significantly influences the outcome. Alonso [4] found that excessive oxygen presence negatively affects lactobionic acid production. Furthermore, high stirring speeds (over 350 rpm) adversely affected lactose oxidation by P. taetrolens, with intense agitation reducing lactobionic acid yield despite continued bacterial growth [4]. Since P. taetrolens is aerobic, this bacterium requires oxygen for reproduction; thus, as the bacteria grow, more dissolved oxygen is consumed from the substrate. Hence, the oxygen supply is crucial in producing bacterial metabolites and cell growth [43]. Another study explored how different wavelengths and light intensities could enhance the enzyme activity of P. taetrolens in converting lactose into lactobionic acid using whey [33]. P. taetrolens is a versatile microorganism with significant applications in agriculture and biotechnology; however, this bacterium is primarily recognized for its role in lactobionic acid production. Thus, various bioproduction approaches for lactobionic acid have been established, yet optimizing microbial incubation conditions can greatly enhance its productivity [44]. These capabilities make P. taetrolens a valuable organism for enhancing cell growth and producing bioactive compounds (Table 2, Ref. [3, 4, 5, 6, 13, 14, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54]).
| Pseudomonas taetrolens subspecies | Information | Year published, [reference] |
| Pseudomonas graveolens *; ** | This study investigated whether certain Pseudomonas subspecies can metabolize reducing disaccharides directly, without hydrolysis, by exploring their potential to produce bionic acids. This study found that P. graveolens oxidized lactose to calcium lactobionate with a 75% yield in 165 hours. | 1947, [32] |
| Pseudomonas graveolens * NRRL B14 | This study examined the ability of P. taetrolens to oxidize isomaltose without breaking its 1–6 glycosidic bond, producing isomaltobionic acid. The chromatographic analysis confirmed the product, highlighting the unique enzymatic activity of the strain in carbohydrate metabolism. | 1969, [45] |
| ATCC 4683 | This study explored the regulation of de novo pyrimidine biosynthesis in P. taetrolens, focusing on enzyme activity and transcriptional control. The study examined how pyrimidine supplementation affects enzyme activity and analyzed pyrimidine limitation in an auxotrophic mutant. Additionally, the study assessed aspartate transcarbamoylase inhibition by nucleotides and compared regulatory mechanisms between P. taetrolens and P. fragi. | 2004, [31] |
| Y-30 | This study characterized P. taetrolens, a methylamine-assimilating bacterium, and evaluated its theanine production from glutamic acid and ethylamine. Growth conditions were optimized, and a coupled fermentation system with baker’s yeast enhanced ATP regeneration. The 16S rDNA analysis confirmed its similarity to P. taetrolens, supporting its identification. | 2004, [6] |
| ATCC 4683 | This study isolated and characterized the broad substrate racemase ArgR from P. taetrolens, investigating its localization and translocation. ArgR was found in the periplasm, processed via signal peptide cleavage, and translocated via the Sec system. ArgR plays a key role in D-arginine and D-lysine catabolism, showcasing unique features among amino acid racemases. | 2009, [46] |
| LMG 2336 | This study developed a cost-effective bioconversion process for lactobionic acid production from cheese whey, optimizing P. taetrolens inoculum age and size using a pH shift-cultivation strategy. This study also explored the impact of dissolved oxygen levels under oxygen-limited and oxygen-sufficient conditions and evaluated various feeding strategies, including co-feeding, continuous feeding, and multi-pulse feeding. Additionally, an innovative flow cytometry approach was used to assess the physiological state of P. taetrolens and enhance production efficiency. | 2013, [4] |
| DSM 21104 | This study optimized lactobionic acid production from whey-derived lactose using P. taetrolens, examining the effects of time, temperature, and inoculum concentration. | 2014, [34] |
| LMG 2336 | This study developed a co-fermentation system using glucose, lactose, and glycerol to enhance lactobionic acid production, emphasizing cheese whey as a cost-effective feedstock. This study also analyzed P. taetrolens adaptive responses to carbon source shifts, providing insights into process stability and efficient carbon uptake. | 2015, [35] |
| TCCC 11892 | This study explored P. taetrolens resting cells for L-rhamnose biotransformation into L-rhamnonic acid, thereby reporting this capability for the first time. Optimization of temperature, cell density, and shaker speed enhanced the conversion rate. | 2016, [47] |
| LMG 2336 | This study evaluated temperature-controlled and temperature-shift strategies for optimizing lactobionic acid production from cheese whey by P. taetrolens. This study focused on balancing growth-coupled and uncoupled formation and examined how temperature and initial cell density influence this ratio. | 2017, [14] |
| LMG 2336 | This research paper examined the simultaneous fermentation of residual cheese whey by P. taetrolens and Lactobacillus casei to co-produce lactic acid and lactobionic acid. The experimental results highlighted the critical role of interactions between these two microorganisms. | 2017, [13] |
| LMG 2336 | This study developed a sequential fermentation process to create a synbiotic functional food by combining Lactobacillus casei with lactobionic acid. P. taetrolens first fermented dairy whey for high lactobionic acid yield, followed by L. casei inoculation to produce lactic acid while retaining lactobionic acid as a prebiotic. | 2017, [36] |
| BCRC 12815 | This study examined the impact of light on lactobionic acid production by P. taetrolens using cheese whey, analyzing how different wavelengths and intensities affect growth and yield. This study explored light as a potential modulator to enhance efficiency and reduce fermentation time. | 2017, [33] |
| LMG 2336; DSM 21104 | This study explored lactobionic acid production from ricotta cheese whey, a byproduct mainly used as cattle feed. Moreover, the study assessed the suitability of whey use as a substrate and identified two P. taetrolens strains as effective producers. | 2018, [37] |
| KCTC 12501 | This study involved culturing P. taetrolens in an optimized synthetic medium, which is different from the traditional whey medium, to achieve high productivity of lactobionic acid. | 2019, [48] |
| LMG 2336 | This study explored encapsulation to reduce the inhibitory effect of lactic acid bacteria on P. taetrolens, enabling co-cultivation for improved lactobionic acid production. Various encapsulating agents were tested, highlighting their potential to protect sensitive strains in mixed fermentations. | 2020, [49] |
| ATCC 4683 | This study optimized lactobionic acid production by P. taetrolens through pH control, lactose concentration, and temperature adjustments. The study also developed an efficient recovery method using ethanol precipitation and ion exchange chromatography for high-purity lactobionic acid. | 2020, [38] |
| KCTC 12501 | This study enhanced lactobionic acid production in P. taetrolens through genetic engineering by expressing quinoprotein glucose dehydrogenase 1. A pDSK519-based genetic tool was developed to boost intracellular lactose oxidation and production efficiency. | 2020, [50] |
| LMG 2336 | This study evaluated acid whey as a substrate for lactobionic acid production by P. taetrolens in shake-flask and bioreactor systems. The study also compared uncontrolled and pH-controlled (6.5) fermentation and assessed purification methods, such as activated carbon adsorption and lyophilization, to enhance production efficiency for industrial use. | 2021, [39] |
| KCTC 12501 | This study developed a two-step biocatalytic process to convert waste office paper into cellobionic acid. Commercial cellulases first hydrolyzed paper into cellobiose, which was then oxidized by recombinant P. taetrolens expressing quinoprotein glucose dehydrogenase. The research highlights a cost-effective approach with potential industrial applications. | 2022, [51] |
| NCIB 9396, DSM 21104 | This study investigated the impact of acid whey permeate concentration and essential ions (potassium, magnesium, and manganese) on P. taetrolens lactobionic acid production. Analyzing the variations in the total solids and optimizing ion levels to enhance lactose oxidation and maximize yield in microbial synthesis. | 2022, [5] |
| LMG 2336 | This study aimed to optimize lactobionic acid production from acid whey, addressing low bacterial growth in this substrate. The study also explored strategies such as periodic P. taetrolens inoculum addition to improve lactose conversion and yield, enhancing acid whey utilization. | 2022, [40] |
| DSM 21104 | This study evaluated factors influencing lactobionic acid production from dairy waste, focusing on whey quality, inoculum volume, and cultivation time. The study also explored acid-to-sweet whey ratios to optimize P. taetrolens lactose conversion, promoting the sustainable use of dairy byproducts. | 2022, [41] |
| LMG 2336 | This study explored traditional cheese whey from Bénin as a substrate for producing lactobionic and lactic acids using P. taetrolens and Lactobacillus casei. The study also aimed to support the dairy sector economically while mitigating the environmental impact from whey disposal procedures. | 2022, [3] |
| KCTC 12501 | This study investigated whether P. taetrolens has additional lactose-oxidizing enzymes beyond the known GCD1, GCD3, GCD4, and MQO1. Gene inactivation experiments confirmed that all lactose-oxidizing enzymes in P. taetrolens depend on pyrroloquinoline quinone (PQQ) for activity. | 2024, [52] |
| ULE-PH5 | This study isolated and characterized phosphate-solubilizing bacteria, including P. taetrolens, from hop plant rhizospheres to address phosphorus legacy in soils. This study identified high-performing strains with potential as biofertilizers to enhance phosphorus uptake and plant growth. | 2024, [53] |
| ** | This study developed an inline monitoring approach using electrical conductivity to track lactobionic acid production by P. taetrolens. A simplified mathematical model was introduced to predict lactobionic acid concentration, demonstrating its effectiveness in optimizing fermentation conditions. | 2025, [54] |
| DSM 21104 | This study aimed to optimize lactobionic acid production using P. taetrolens in whey-based bioreactor cultures. The study also evaluated the prebiotic properties of lactobioni acid by microencapsulating Bifidobacteria and assessing their survival in an in vitro gastrointestinal model. | 2024, [42] |
* now known as Pseudomonas taetrolens.
** does not specify the exact strain of Pseudomonas taetrolens used.
GCD1, glucose dehydrogenase 1; GCD3, glucose dehydrogenase 3; GCD4, glucose dehydrogenase 4; MQO1, malate:quinone oxidoreductase 1; rDNA, ribosomal deoxyribonucleic acid.
Table 2 summarizes a range of studies on P. taetrolens, focusing on the enzymatic activities and applications of P. taetrolens subspecies in sustainable production. Most studies demonstrate the ability of P. taetrolens to efficiently convert lactose into lactobionic acid using various substrates, including cheese whey, acid whey, and synthetic media. For example, Goderska et al. [34] yielded 15.79 g/L of lactobionic acid during a 50-hour fermentation at 30 °C with an initial lactose concentration of 30.29 g/L. Alonso [4] reported a 100% conversion yield of lactose to lactobionic acid from 60 hours in sweet whey, while Sarenkova et al. [40] demonstrated complete lactose conversation in 48 hours using acid whey. These studies emphasize the efficiency of P. taetrolens in producing lactobionic acid, a compound with significant industrial applications. Studies also focused on adapting production processes for specific substrates, such as ricotta cheese whey and traditional Bénin whey [3, 37]. Following an analysis of the growth of P. taetrolens in acid whey, it was hypothesized that Na+ ions influence the proliferation of P. taetrolens. Acid whey naturally has a low pH, which is adjusted to 6.5 by adding 6 M NaOH, resulting in a higher concentration of Na+ ions than cheese whey [40]. Studies have shown that the presence of Na+ and/or Cl⁻ ions in the environment can inhibit the growth of Gram-negative bacteria, including Pseudomonas spp. This ion influx alters the osmotic pressure within the cell, affecting cell viability. Research has demonstrated that elevated Na+ and/or Cl⁻ concentrations reduce the activity of enzymes, such as lipase and collagenase, in P. aeruginosa JCM5962(T) and other species within the Pseudomonas genus [55, 56, 57]. Environmental factors, including ion concentrations, pH, and substrate composition, significantly affect the growth and productivity of P. taetrolens [4]. Although high salt concentrations initially inhibit bacterial proliferation, a small portion of cells gradually adapts, indicating phenotypic adaptation [40], similar to P. aeruginosa [58]. This adaptation may allow P. taetrolens to survive in acid whey, making it a viable substrate for lactobionic acid production. Moreover, introducing biomass containing partially adapted bacteria can synergistically enhance lactose bioconversion and lactobionic acid production in acid whey [40]. Magnesium and manganese ions play critical roles in microbial growth and enzyme activity. Magnesium stabilizes nucleic acids, promotes peptide hydrolysis, and supports cell division, while manganese is essential for enzymes such as superoxide dismutase and lactase [28]. These ions enhance lactobionic acid yield and expand the pH range for enzymatic activity, underscoring their importance in optimizing fermentation processes [5]. The colony-forming unit (CFU) count of P. taetrolens during fermentation reflects its growth dynamics, with concentrations reaching 107 to 1010 CFU/mL during the stationary phase in cheese whey, typically within 30 hours [34, 37]. Meanwhile, factors such as inoculum size, pH, and substrate composition influence bacterial growth and productivity. Research confirmed that a high content of solids in whey, exceeding 20%, reduces lactobionic acid yields due to limited lactose absorption and enzyme inactivation [5]. Subsequently, moderate lactose concentrations, such as 150 g/L, have been found optimal for maximizing yields compared to higher concentrations [59].
Co-fermentation approaches using Lactobacillus casei were investigated
to produce lactobionic and lactic acids simultaneously [49]. P.
taetrolens exhibits unique enzymatic activities, such as oxidizing lactose to
lactobionic acid without hydrolysis and metabolizing isomaltose into
isomaltobionic acid [4, 45]. A previous study also revealed the roles of specific
lactose-oxidizing enzymes and their dependence on cofactors such as
pyrroloquinoline quinone [52]. The racemase enzyme ArgR was characterized for its
broad substrate specificity, highlighting its role in the catabolism of amino
acids such as D-arginine and D-lysine [46]. Pyrimidine biosynthesis regulation in
P. taetrolens was studied, providing insights into transcriptional and
enzymatic control mechanisms [27]. The capacity of the organism to utilize
unconventional feedstocks, such as waste office paper and acid whey, into
value-added compounds, e.g., cellobionic acid and lactobionic acid, has also been
demonstrated [40, 41, 51]. P. taetrolens Y-30 was found to produce
In addition to these industrial applications, Parker et al. [60] identified P. taetrolens as a microbial biomarker for assessing the effectiveness of polychlorinated biphenyl (PCB) remediation in the Hudson River. Parker et al. [60] analyzed the cytoplasmic redox states of microorganisms from various river sites, focusing on NADH and the H+ ratio as indicators of PCB-induced oxidative stress. Among 33 bacterial isolates, P. taetrolens was identified as a PCB-degrading bacterium. Parker et al. [60] also found that a reduced NADH and H+ ratio in P. taetrolens from remediate sites approached statistical significance (p = 0.065), suggesting the potential of using this ratio as a biomarker for successful PCB remediation.
Recent studies also propose that P. dantrolene can serve as a plant growth-promoting rhizobacterium, enhancing phosphorus solubilization and producing growth-stimulating metabolites, such as auxins and siderophores. This dual role highlights the versatility of P. taetrolens in industrial and agricultural applications [53]. The findings summarized in Table 2 underscore the importance of optimizing environmental conditions and substrate composition to enhance the efficiency of P. taetrolens for diverse applications. These studies underscore P. taetrolens as a versatile organism with significant potential for sustainable industrial applications, offering solutions in waste utilization, functional food production, biofertilizers, and pharmaceuticals.
The biotechnological production of lactobionic acid relies on the oxidation of
lactose using microorganisms or specific enzymes. The key reaction mechanism
involves the formation of lactobiono-
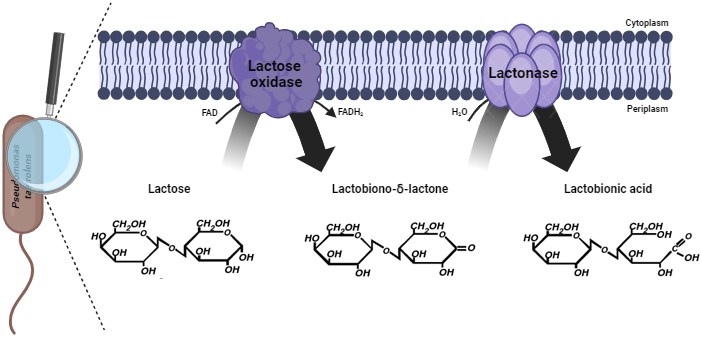 Fig. 3.
Fig. 3.
Biocatalytic conversion of lactose to lactobionic acid by Pseudomonas taetrolens. Created in BioRender. Sarenkova, I. (2025) https://BioRender.com/j52e087.
Lactose dehydrogenase catalyzes the conversion of lactose to
lactobiono-
Lactonases, commonly found in Pseudomonas species, are involved in quorum sensing and the degradation of lactones, which are cyclic esters, including acyl-homoserine lactones that serve as signaling molecules in bacterial communication. While lactonase activity is not directly related to lactose metabolism, it is a characteristic feature of many Pseudomonas species. In contrast, P. taetrolens is particularly noted for its lactose-oxidizing capability, where it catalyzes the conversion of lactose to lactobionic acid, a process driven by lactose oxidase or functionally similar enzymes. Indeed, quinoprotein glucose dehydrogenase (GDH) is central to this activity and exists in two forms: membrane-bound GDH1 and soluble GDH2. GDH1 is primarily responsible for lactose oxidation. Subsequent advances in genetic engineering have successfully enhanced the expression of GDH1 in P. taetrolens, resulting in significantly increased lactobionic acid production. These data demonstrate the potential of using this organism in industrial applications, making it a promising candidate for the sustainable production of this valuable compound [50, 63]. A study on lactonase activity in Pseudomonas revealed the potential use of these bacteria in biotechnological applications, such as antifungal treatments, by targeting fungal quorum-sensing molecules [63].
Lee et al. [52] recently revealed that P. taetrolens possesses more than four enzymes capable of producing lactobionic acid, all of which are pyrroloquinoline quinone (PQQ)-dependent. Research has identified four key enzymes involved in the lactose oxidation process (Table 3, Ref. [52]).
| Enzyme | Acronym | Cofactors/regulation | Function |
| Glucose dehydrogenase 1 | GCD1 | PQQ-dependent | Oxidizes lactose to lactobiono- |
| Glucose dehydrogenase 3 | GCD3 | PQQ-dependent | Secondary carbohydrate oxidation |
| Glucose dehydrogenase 4 | GCD4 | PQQ-dependent | Broader carbohydrate oxidation |
| Malate:quinone oxidoreductase 1 | MQO1 | PQQ-dependent | Facilitates electron transfer in the respiratory chain |
PQQ, pyrroloquinoline quinone.
Further studies involving the inactivation of specific genes associated with these enzymes and PQQ synthesis confirmed that all lactose-oxidizing enzymes in P. taetrolens are PQQ-dependent. These enzymes are encoded by genes regulated by transcriptional factors responsive to environmental conditions, such as carbon availability and oxygen levels. The presence of lactose in the environment induces the expression of these enzymes, likely through a regulatory system involving catabolite repression and PQQ biosynthesis pathways. Additionally, PQQ production is tightly regulated by genes involved in redox balance, ensuring optimal enzymatic function during lactose oxidation [52]. This finding underscores the unique metabolic capabilities of P. taetrolens and highlights the critical role of PQQ in its biochemical pathways.
P. taetrolens plays a critical role in the circular economy by transforming low-value byproducts, such as whey, into high-value compounds, such as lactobionic acid. This process adds economic value and addresses significant environmental challenges. Whey, a byproduct of cheese, curd, and Greek yogurt production, is often produced in quantities exceeding its utilization capacity. Despite its high nutritional value [64], surplus whey is frequently discharged into wastewater systems, leading to severe environmental pollution due to its high chemical (~70,000 mg/L) and biological oxygen demands (~40,000 mg/L) [65, 66].
Particularly challenging is the management of acid whey, which contains higher lactic acid and salt concentrations than cheese whey due to the transfer of colloidal calcium during fermentation. These characteristics limit the reuse of acid whey and increase disposal costs, contributing to the overall processing expenses for dairy producers [67, 68]. On average, nine liters of whey are generated for every kilogram of dairy product produced [69]. Thus, the ability of P. taetrolens to utilize whey for lactobionic acid production presents a sustainable solution for using this byproduct.
Lactobionic acid is a polyhydroxy acid with two hydroxyl groups per molecule formed during the oxidation of lactose [26, 44, 70]. The production of lactobionic acid through a biotechnological process involving P. taetrolens is illustrated in Fig. 4 (Ref. [68]). The different downstream processes of this bacterium were performed after fermentation.
 Fig. 4.
Fig. 4.
Lactobionic acid samples were obtained through microbial fermentation using Pseudomonas taetrolens. After bioreactor fermentation, the product was centrifuged and microfiltered to remove bacteria, then subjected to further processing: (A) Dried at 40 °C in a thin layer. (B) Concentrated to 40% total solid content and dried at 40 °C. (C) Precipitated with 96% ethanol from the concentrated product (40% total solid content), followed by drying at 40 °C. (D) Freeze-dried from the concentrated product (40% total solid content) [68].
This compound combines galactose and gluconic acid into an ether-like structure, making it highly versatile. The universal properties of this compound, such as antioxidant activity, chelating ability, and moisturizing effects, make it valuable across multiple industries. Lactobionic acid exhibits strong antibacterial effects against various pathogens, whereby it effectively inhibits biofilm formation and virulence in pathogens by disrupting biofilm architecture and reducing virulence secretion [26, 71]. In cosmetics, lactobionic acid prevents signs of aging by mitigating free radical damage and preserving skin elasticity [72]. In pharmaceuticals, lactobionic acid is a component of organ preservation solutions, such as the University of Wisconsin solution; lactobionic acid has also been explored as a contrast agent for applications in chemotherapy and magnetic resonance imaging [73]. Furthermore, lactobionic acid is important in the chemical industry in cleaning agents and anti-corrosion coatings [70]. From a nutritional perspective, lactobionic acid is a low-calorie sweetener (2 kcal/g) with prebiotic properties, promoting beneficial gut microbiota and enhancing calcium absorption [74]. Moreover, lactobionic acid has demonstrated prebiotic effects by improving the survival of Bifidobacteria in the gastrointestinal tract [42, 75]. Additionally, lactobionic acid supplementation in the diet of laying hens has been shown to enhance laying quantities and egg quality. A study involving 700 hens demonstrated that a diet enriched with 2% lactobionic acid significantly enhanced egg production and fatty acid accumulation, improving the nutritional quality of the eggs [76]. The global market for lactobionic acid is steadily growing, with approximately 15,000 tons produced annually for medical, industrial, and research purposes [77]. The broad applicability of lactobionic acid underscores the strategic importance of efficient production methods using cost-effective and recyclable raw materials, such as whey.
Despite its promising potential in biotechnology, large-scale industrial applications of P. taetrolens face several challenges. These challenges must be addressed to optimize the use of P. taetrolens in sustainable production and bioprocessing. The efficiency of P. taetrolens in converting lactose to lactobionic acid is highly dependent on fermentation conditions, such as pH, temperature, dissolved oxygen levels, and substrate concentration. Maintaining optimal conditions for high yields remains challenging, as variations in these factors can lead to reduced productivity or undesired byproducts. Study indicates that excessive oxygen supply or high stirring speeds negatively affect lactobionic acid production, requiring careful bioreactor control strategies [4]. While P. taetrolens can utilize dairy byproducts, including whey, industrial-scale fermentation requires consistent and cost-effective feedstocks. Variability in whey composition, particularly in acid whey, can impact bacterial growth and lactobionic acid yield due to changes in pH and salt concentrations. This involves additional pretreatment steps, increasing production costs [40]. The metabolic pathways of P. taetrolens are not yet fully optimized for industrial applications. For instance, the dependence of P. taetrolens on PQQ-dependent dehydrogenases limits enzymatic efficiency under suboptimal conditions. Additionally, gene regulation mechanisms affecting lactose oxidation need further investigation to enhance strain performance through metabolic engineering [52]. The separation and purification of lactobionic acid remain cost-intensive due to the need for specialized methods such as ion exchange chromatography and electrodialysis. Meanwhile, efficient recovery methods that maintain high purity while reducing energy consumption and processing time are needed for commercialization [62, 68]. Transitioning from lab-scale to industrial-scale production requires overcoming aeration, agitation, and bioreactor design challenges. However, ensuring consistent bacterial growth and metabolite production across large-scale fermentation setups remains a significant engineering challenge [14]. Although P. taetrolens is non-pathogenic, regulatory approvals for its use in food, pharmaceuticals, and cosmetics production require extensive validation. Strain safety assessments and compliance with global regulations, such as those imposed by the U.S. Food and Drug Administration (FDA) and European Food Safety Authority (EFSA), must be addressed before large-scale adoption in consumer products [78, 79].
P. taetrolens demonstrates exceptional versatility as a microorganism,
with the ability to thrive in diverse environments and metabolize a wide range of
organic compounds, including lactose, glucose, maltose, and various amino acids.
This adaptability and robust enzymatic systems establish P. taetrolens
as a cornerstone for industrial biotechnology. Furthermore, the ability of
P. taetrolens to oxidize lactose into lactobionic acid, a compound with
extensive applications in pharmaceuticals, cosmetics, and functional foods,
highlights the importance of this bacterium in sustainable production processes.
Moreover, P. taetrolens has proven effective in utilizing dairy
byproducts, such as whey, offering an eco-friendly solution for waste utilization
within the framework of a circular economy. The enzymatic mechanisms in this
bacterium, particularly using PQQ-dependent glucose dehydrogenase enzymes, are
essential for its metabolic efficiency. These enzymes facilitate the conversion
of lactose into lactobionic acid through well-characterized pathways involving
lactobiono-
EPS, extracellular polymeric substances; LPS, lipopolysaccharides; PQQ, pyrroloquinoline quinone; CFU, colony-forming unit; FAD, flavin adenine dinucleotide; PCB, polychlorinated biphenyl; GDH, quinoprotein glucose dehydrogenase; GCD1, glucose dehydrogenase 1; GCD3, glucose dehydrogenase 3; GCD4, glucose dehydrogenase 4; MQO1, malate:quinone oxidoreductase 1.
IS, IC and MR conceptualized and designed the review structure. IS conducted the literature search and compiled data from the sources. IC and MR contributed expertise and critical insights to the content analysis. IS wrote manuscript. IC and MR contributed to the editorial revisions. IS, IC and MR approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT to check spelling and grammar. After using this tool, the authors reviewed and edited the content as needed and took full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.