1 CEDEA & ICGT (Center of Oncology Diagnostic and International Cancer Gene Therapy Foundation), 110010 Bogotá D.C., Colombia
2 Department of Biotechnology, University of Isfahan, 81746-73441 Isfahan, Iran
3 Faculty of Medicine, Autonomous University, Colombia (UNAB University), 681001 Floridablanca, Colombia
4 INSERM UMR 1197, Cancer Center & University of Paris/Saclay, 94802 Villejuif, France
5 National Academy of Medicine–ANM, 75272 Paris, France
Abstract
The brain malignant tumor Glioblastoma multiforme (GBM) has a median survival of 14–16 months using current treatments; thus, understanding the pathology of GBM is crucial for proposing new therapies and increasing overall survival outcomes. Therefore, this study aimed to analyze different elements, particularly growth factors and the related signal transduction pathways, which play a role in brain neoplastic development, from stem cells to established solid brain tumors, and the application of current immunology techniques, molecular biology, and nanotechnology. Targeting growth factors, especially insulin-like growth factor-1 (IGF-I) (the principal neoplastic development factor) using anti-gene technologies—antisense and triple helix—has previously been shown to produce an immune anti-tumor response (CD8, CD28) through the TK/PI3K/AKT pathway. This immune response was increased using phytochemicals (phenolics), especially nanoparticles (theranostic nanoparticles), by modulating IGF-I through common pathways (IGF-I-R and TK/PI3K/AKT/TLR/MAPK and JAK/STAT). This review demonstrates how studies on central nervous system neoplastic development progressively led to establishing clinical cancer gene therapies, increasing GBM survival by 20–24 months. The presented studies compare the results of cancer gene therapy with other current immunotherapies. Moreover, this research chapter briefly describes the investigations of nanotechnology related to neurotumorigenesis and GBM therapies. The presented studies relate to nanotechnology and compare the results of cancer gene therapy with other current immunotherapies.
Keywords
- cancer immuno–gene therapy
- brain neoplastic development
- stem cells
- glioblastoma
- IGF-I-induced signal transduction pathways
- nanotechnology
Glioblastoma multiforme (GBM) is the most common malignant brain tumor diagnosed
in individuals aged 60 years (up to 15 cases per 100,000 people), with a
predominantly low survival rate of less than a year [1, 2, 3, 4]. The pathogenesis of
GBM stems from genetic and growth factor pathway alterations (such as epidermal
growth factor (EGF), transforming growth factor-beta (TGF
This review aimed to present the research processes leading to central nervous
system (CNS)-related tumor therapy, from the hypothesis that neoplastic nervous
system differentiation from stem cells to neuroglial differentiation is related
to the presence of oncoproteins/growth factors, such as alpha-fetoprotein and
IGF-I. Therefore, targeting these factors by suppressing the expression of
related factors on a molecular level (transcription and translation) has promoted
the development of a new form of cancer treatment: Cancer gene therapy [18]. The
cancer gene therapy described in this review is based on anti-gene IGF-1
technology, targeting IGF-I in neoplastic glial cells. The IGF-I receptor induces
the IGF-I signal transduction pathway, transforming GBM cells into immunogenic
cell vaccines (expressing major histocompatibility complex class I (MHC1) and B7
antigens). This signal transduction pathway was further reinforced using
phytochemicals related to IGF-I signaling elements (Toll-like
receptor/mitogen-activated protein kinase/nuclear factor
kappa-light-chain-enhancer of activated B cells (TLR/MAPK/NF
Genetic and environmental causes can promote the induction of CNS malformations and tumors [26]. The neurohistogenesis of malformations is closely related to embryonal tumors. Primitive neuroectodermal tumors (PNETs) were classified as central neuroblastoma, ependymoblastoma, medulloblastoma, and supratentorial PNETs—Medulloepithelioma and atypical teratoid/rhabdoid tumors [2, 3]. The mouse teratocarcinoma model was employed to study the histogenesis of neuroectodermal tumors behind CNS neoplastic development and their transformation into solid tumors, such as glioblastoma. The histogenesis of teratocarcinoma, derived from pluripotent carcinoma cell line 3 (PCC3) and PCC4 embryonal carcinoma cell lines, reproduced normal CNS development (Fig. 1) [18, 27, 28, 29, 30], and, in parallel, the different stages of abnormal nervous tissue histogenesis, which can be presented as follows: The neuroectoblast starts as embryonal carcinoma stem cells, which transform into neuroblastic vesicles mimicking the successful development of a neural tube.
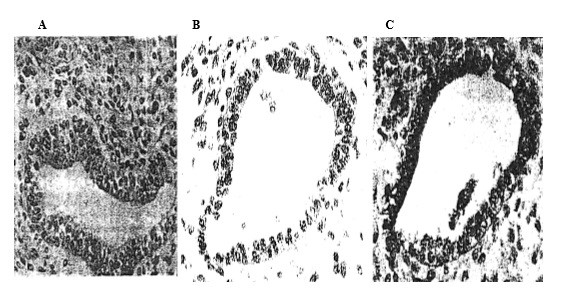 Fig. 1.
Fig. 1.
Histogenesis of neuroectodermal structures in the mouse
teratocarcinoma model. The tumor is derived from PCC4 stem cells. (A) A cyst of
nervous origin is a pathological neural tube surrounded by neuroepithelial stem
cells, and exhibits characteristics of neuroglia. The cyst shows a basal limit.
Hematoxylin and eosin stain (HE) (
These so-called neuroblastic structures follow different stages: (1) Undifferentiated carcino–embryonic structures; (2) medulloepithelial structures (composed of a mixture of ectoblastic and neuroectoblastic components); (3) neuroblastic structures; (4) neuroepithelial structures [2, 27, 28, 31]. These structures present either a diffused growth of neuroblastic stem cells or a mixture of neuronal and glial precursors. Final differentiation results in encephaloid tissue (a positive biomarker in staining for alpha-fetoprotein (AFP), serum albumin (SA), and IGF-I) [27]. Implementation of the mouse teratocarcinoma model, containing stem cells and neuroglial structures, is useful in understanding human CNS tumors, which can differentiate into both neuronal and glial lineages [22, 32, 33], as well as in GBM-related cancer gene therapy [18].
Glioblastomas are usually located in the subcortical white matter and deep grey
matter, and the histopathology of GBM is characterized by cellular pleomorphism,
nuclear atypia, and areas of hemorrhage and necrosis. Generally, we distinguish
between giant cell glioblastomas, gliosarcomas, and epithelioid glioblastomas
[2, 34]. GBM pathogenesis involves gene suppression, expression changes, and
abnormal dysregulation of growth factors, such as IGF-I, TGF
GBM treatment involves complex molecular characteristics and an
immunosuppressive microenvironment [35]. The tumor microenvironment involves
immunosuppression, astrogliosis, and angiogenesis. While treatments, such as
immune checkpoint inhibitors or chimeric antigen receptor T cell (CAR-T) therapy,
have made progress, tumor vaccines based on dendritic cells (DCs) have also
appeared in immunotherapy [36]. DCs activate tumor-specific T cells and initiate
adaptive immune responses [37, 38]. Nevertheless, the immunosuppressive
microenvironment in GBM is known to inhibit the function of DCs by secreting
immunosuppressive factors, such as TGF-
Magnetic resonance imaging (MRI) and computed tomography (CT) exams are used primarily in emergency laboratory diagnoses. Meanwhile, positron emission tomography (PET) and single-photon emission computed tomography (SPECT) facilitate early detection and treatment monitoring [10]. Regarding GBM biomarkers, IGF-I, IDH, P53, olig 2, and S100 are positive, and stem cell markers CD44 and CD133 are generally positive [2, 42, 43, 48, 49]. EGFR amplification, the PI3K pathway-related genes, PTEN genes, and RTK gene alterations should also be considered [50]. For the prognosis of GBM, the giant cell subtype has a better prognosis, whereas gliosarcoma and epithelioid GBM are associated with a poor prognosis; nonetheless, the GBM treatment using temozolomide (MGMT promoter methylated tumor) promotes improved survival [1, 13, 51]. When considering IGF-I as a diagnostic and prognostic biomarker, this growth factor also constitutes the principal target for immuno–gene therapies [18]. Moreover, cell-free microRNAs in the blood exhibit potential in diagnosing, prognosticating, and monitoring treatment. The cell-free miRNA profile within CSF demonstrates high potential in delivering precise and specific evaluations of brain tumors [52].
Classical therapies include surgery, radiotherapy, and chemotherapy; however, the maximalist tumor resection remains the first-line treatment [1]. Nevertheless, high doses of glucocorticoids (GCs) improve neurologic deficits, and patients receiving excessive doses of GCs present high CD8 T cell expression, with the latter playing a role in the anti-tumor response [1, 11, 12, 53]. On the other hand, nuclear medicine has introduced theranostic approaches—the therapeutic potential of targeted radionuclide therapy [54, 55, 56, 57]. In parallel with radiotherapy, a postoperative radiation, i.e., chemotherapy, such as temozolomide (TMZ), can be administered to improve the effective survival [58, 59]; however, many GBM patients are resistant to temozolomide, affecting the immune response of the host [17, 59, 60]. Moreover, chemotherapy is often replaced by anti-tumor vaccines such as bevacizumab (Bev) and anti-VEGF-A, although genetic variations in VEGF expression influence treatment resistance [61]. Neoadjuvant Bev treatment decreases the volume of GBM and improves clinical symptoms. Meanwhile, the responsiveness of the patient to Bev might depend on the level of VEGF expression [62]. Other growth factors, such as EGFR and EGFRvIII, can also be considered for therapeutic purposes. EGFR and EGFRvIII amplification are frequently observed in GBMs and contribute to tumorigenesis and progression. However, therapies directed against EGFR and EGFRvIII have yet to present clear clinical benefits; combination therapies are needed to improve outcomes [15]. Moreover, studies have investigated the clinical intravenous delivery of a single dose of autologous T cells redirected toward the EGFRvIII mutation by a chimeric antigen receptor (CAR). These investigations may improve the efficacy of EGFRvIII-directed strategies in GBM [63].
Immunotherapeutic strategies involving immune checkpoint inhibitors, adoptive T cell therapy, and viral immunotherapy have increased interest in immune and virus studies [1, 64, 65, 66, 67, 68, 69]. The adoptive T cell (ACT) strategy (CAR-T cells), a more recent cancer therapy [66], dramatically improved tumor volume. However, further research is needed to resolve challenges related to the mechanisms involved [14, 66, 70, 71, 72]. In addition to technologies, such as immune checkpoint inhibitors (such as anti-PD-1/PD-L1 and CTLA-4 antibodies), CAR-T therapy, and oncolytic viruses, demonstrating progress, tumor vaccines based on DCs have become an important part of the immunotherapy field; nevertheless, individual differences in efficacy suggest that applying DC vaccines to treat GBM requires additional optimization [39, 40, 41].
Cancer immunotherapy and nanotechnology have become increasingly intertwined in recent years. Examples of nanotechnology, NPs, used in cancer immunotherapies can be mentioned as follows [21, 73]: NPs can be used to deliver immunotherapeutic agents such as checkpoint inhibitors or CAR-T cells to the tumor; NPs can be loaded with tumor antigens that activate dendritic cells; NPs administered in immuno–gene therapy can promote the delivery of agents to APCs, or participate in immunomodulation by delivering cytokines.
New glioblastoma therapies are mainly based on immune or immuno–gene strategies: cancer immunotherapy was established by Townsend and Allison [74] and cancer gene therapy by Trojan et al. [75], followed by others [64, 65, 66, 70, 71, 72, 76, 77, 78]. Cancer gene therapy targeting growth factor genes, especially IGF-I, the principal neoplastic development factor, was performed in tumor cells using antisense [79, 80] or triple helix technologies [81, 82] (Fig. 2, Ref. [75]).
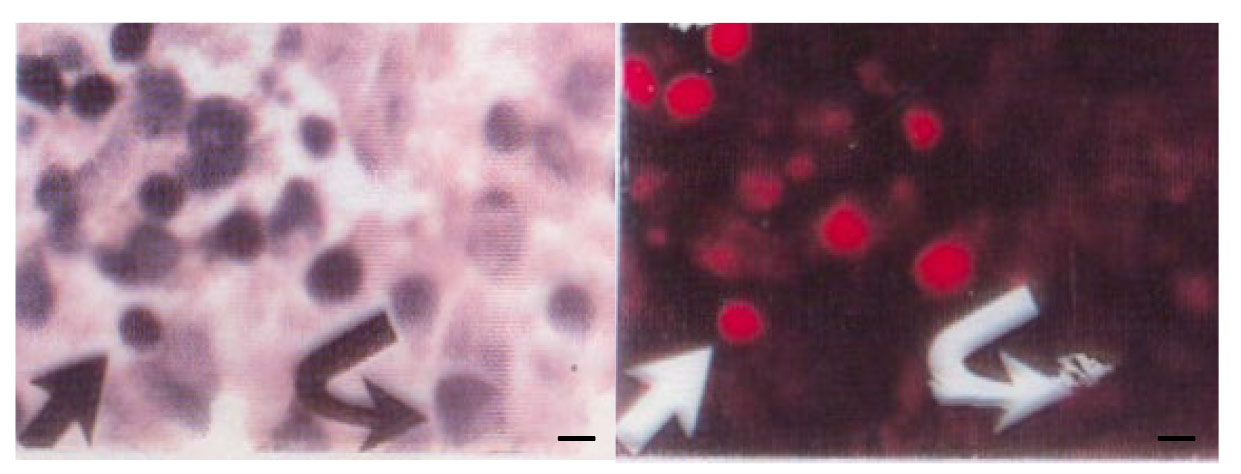 Fig. 2.
Fig. 2.
Rat glioma tumor. (left) The tumor derived from C6
glioma cells one week after subcutaneous injection of transfected IGF-I antisense C6
cells. HE (
Other promising approaches involve targeting growth factors such as TGF-beta or
VEGF and EGF, their receptors, and their downstream transduction signaling
elements [6, 16, 83, 84]. Recent neuro-oncology research has highlighted the role of
the PI3K/AKT pathway in glial cells: the simultaneous arrest of at least two
links, either IGF-I, TGF
An anti-gene IGF-I strategy that blocks IGF-I synthesis at the transcription or translation levels [90] has been introduced into clinical trials, with the median survival of patients reaching 18 to 20 months [91]. Immune and gene therapies, particularly anti-gene IGF-I/phytochemical immuno–gene treatment, increased the average survival span of GBM patients up to 20–24 months.
These promising clinical results were obtained using cell vaccines (anti-IGF-I
antisense and triple helix transfection) presenting strong immunogenicity: CD8
and CD28 molecules. Immunogenicity was induced by a common signal transduction
pathway produced by anti-gene and phytochemical technologies (TK/PI3K/AKT and
TLR/MAPK/NF
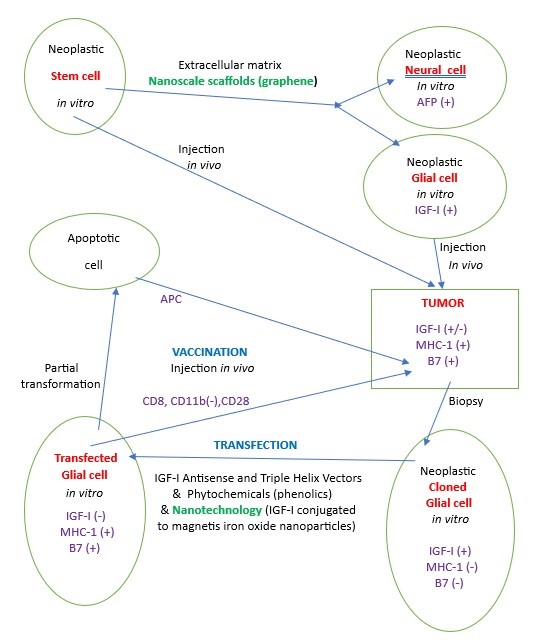 Fig. 3.
Fig. 3.
Schematic of the experimental cancer gene therapeutic process
(anti-IGF-I/phytochemical/nanotechnology). The stem cells, i.e., PCC4 cells
[30], in the presence of nanoscale scaffolds (graphene), transition to neural and
glial cells [23, 93]. The stem cells, if injected in a mouse model in
vivo, induce a teratocarcinoma tumor reproducing CNS neoplastic development
containing neural cells (alpha-fetoprotein positive) and glial cells (IGF-I
positive) [27, 32]. On the other side, the glial cells (i.e., C6), if injected
in vivo, induce another type of tumor: glioma murine tumor [19, 75]. The
neoplastic glial cells removed by biopsy from the glioma tumor are transfected
in vitro using IGF-I antisense or IGF-I triple helix vectors
[17, 79, 82, 90, 94], combined with phytochemicals derived from essential oils of
Acmella and Geraniaceae [18, 95, 96] and nanotechnology (conjugation of human IGF-I
to magnetic iron oxide nanoparticles (IONPs)) [92]. All three technologies
combine to increase cell immunogenicity (expression of MHC-1 and B7) due to a
common signaling pathway: anti-IGF-I tyrosine kinase—PI3K/AKT/PKC/GSK3
[17, 20, 97]; phytochemicals (TLR/MAPK/NF
New neurons are created throughout life through a process known as neurogenesis [101, 102]. Neurogenesis occurs when neural stem cells (NSCs) generate new multipotent cells. This process begins during fetal development and persists in adulthood [103, 104]. The first description of the central nervous system development was performed between 1979 and 1981 by applying an alpha-fetoprotein marker using a rat brain fetal model and then a neoplastic model of CNS in mouse teratocarcinoma derived from PCC4 stem cells. A comparison of the two models demonstrated the existence of convergence between embryonic/fetal development and neoplastic development [29, 30].
Immunologic factors play an important role in normal and neoplastic development, especially when related to insulin-like growth factor-1 (IGF-I), which plays a role in normal NSCs by inducing differentiation, proliferation, or survival of neurons [105, 106, 107]. As the main neoplastic development factor, IGF-I was proposed as a target to treat glioblastoma progression [44, 108].
Stem cell research offers great promise for regenerative medicine, tissue engineering, drug screening, and clinical therapies. However, the success of such applications relies on the ability to accurately control the differentiation and growth of stem cells into functional tissues. Meanwhile, the distinction of stem cells can be directed by controlling their microenvironment, including substrate stiffness, topography, and chemical cues. However, methods for controlling traditional microenvironments lack the precision required for successful and reproducible differentiation. Nanotechnology provides a way to precisely control the behavior of stem cells differentiating into neuroglial cells by manipulating their cellular microenvironment at the nanoscale [32].
The unique properties of nanomaterials, including their small size, high surface area, and tenable physicochemical properties, have enabled researchers to manipulate and control cellular behavior with a high degree of precision. In particular, nanomaterials have been developed to mimic the extracellular matrix (ECM) and provide physical and chemical cues to guide stem cell behavior. Additionally, nanoparticles can act as delivery vehicles for therapeutics or genetic material, enabling targeted gene therapy. Furthermore, one of the major applications of nanotechnology in stem cell research is the development of nanoscale scaffolds that mimic the ECM found in the tissues. Nanotechnology has been used to create scaffolds that support the growth and differentiation of neurons and glial cells, with potential applications in treating neurodegenerative diseases and spinal cord injuries. These scaffolds can be designed to provide physical support and chemical and mechanical cues to guide stem cell differentiation and tissue regeneration. For example, nanofibrous scaffolds have been shown to support the distinction of mesenchymal stem cells into bone, cartilage, and other tissue types [23, 24, 93, 109, 110, 111, 112, 113].
Nanotechnology has also been applied to studying neural cells, including neurons and glial cells. Similar to stem cells, neurons and glial cells require specific microenvironments for growth and differentiation. Thus, nanotechnology has been used to create scaffolds that support the development and differentiation of these cells. Nanofibers can be used as scaffolds to promote neurite outgrowth and synapse formation. Additionally, nanoparticles can be engineered to deliver therapeutic molecules or drugs directly to neurons or glial cells. In neuroscience, nanoelectronics devices are being developed to interface with neural cells and circuits. These devices can record and stimulate neural activity with high spatial and temporal resolution, enabling researchers to improve understanding of brain function. For example, researchers have developed nanowire arrays that can be implanted into the brain to record neural activity. These devices have been used to study various phenomena, including learning and memory, perception, and motor control. Nanoparticles have also been used as contrast agents for imaging techniques such as MRI and PET, allowing researchers to visualize and track neural cells in vivo [25, 114].
Neurons and glial cells require specific microenvironments for growth and differentiation, and nanotechnology can be used to create scaffolds that support their growth and differentiation. In addition to scaffolds, nanoparticles have also been used to deliver therapeutic agents directly to stem cells or the tissues that require regeneration. These nanoparticles can be functionalized with targeting ligands or imaging agents to improve specificity and efficacy. Moreover, these nanoparticles can be engineered to release drugs or growth factors in a controlled manner, enabling precise temporal and spatial control over the biological response [23, 25, 115].
Nanomaterials have been developed to mimic the ECM and provide physical and chemical cues to guide stem cell behavior, followed by their ulterior neuroglial differentiation. For example, nanofibers can replicate the collagen fiber structure in the ECM, providing a scaffold for stem cells to attach and grow on. Similarly, nanoparticles can be functionalized with specific ligands that bind to cell surface receptors, directing stem cell differentiation. One example of a nanomaterial used in stem cell research is graphene, a two-dimensional material of carbon atoms arranged in a hexagonal lattice. The unique properties of graphene, including its high surface area, mechanical strength, and electrical conductivity, make it an attractive material for creating scaffolds for tissue engineering applications. Researchers have demonstrated that graphene-based scaffolds can support the growth and differentiation of various cells, including neural stem cells. In one study, graphene oxide nanosheets were incorporated into a hydrogel scaffold to promote the regeneration of spinal cord tissue in rats following injury. These researchers found that the graphene oxide nanosheets increased the adhesion and proliferation of neural progenitor cells, improving functional recovery in the injured animals [23, 32, 93, 109].
Nanomaterials can also be used as therapeutics or genetic material delivery vehicles, enabling targeted gene therapy and drug delivery. Nanomaterials can be engineered to release drugs or other therapeutic agents in response to specific stimuli, such as changes in pH or temperature. This allows precise control over drug release timing and location, improving therapeutic efficacy and reducing side effects. In one study, researchers developed gold nanoparticles functionalized with a protein that targets cancer cells. When these nanoparticles were loaded with a chemotherapy drug and injected into mice with brain tumors, the nanoparticle–drug combination selectively targeted and killed cancer cells while sparing normal cells. This approach can potentially improve the effectiveness of chemotherapy while minimizing toxicity to healthy tissues [25, 110, 114, 115].
Nanotechnology offers unique opportunities to control stem and neuronal/glial cell behavior by manipulating their microenvironment. Nanomaterials can mimic the ECM and provide physical and chemical cues to guide stem cell behavior, while nanoparticles can act as delivery vehicles for therapeutics or genetic material. Furthermore, nanotechnology has been used to create scaffolds that support the growth and differentiation of neurons and glial cells. Overall, nanotechnology in stem cell and neuronal/glial cell research holds great promise for regenerative medicine, tissue engineering, and drug screening. Meanwhile, integrating nanotechnology with stem cell and neuronal/glial cell research could revolutionize regenerative medicine and neuroscience. Therefore, with continued innovation and development, researchers may be able to create more effective therapies for a wide range of diseases and injuries [17, 23, 114, 115].
Brain development is marked by a complex interplay of molecular, cellular, and structural adaptations, which promote the sophisticated functions performed by the brain. Neoplastic development, such as gliomas and glioblastomas, often exploits pathways similar to those involved in brain evolution, such as cellular plasticity, angiogenesis, and metabolic adaptability. Hence, nanotechnology provides a unique vantage point for studying both the development processes of the brain and the mechanisms underlying tumorigenesis, enabling advancements in diagnostics, research, and therapeutic applications [23, 24, 109, 116].
The human brain is a product of millions of years of evolution, marked by increased cortical size, neural complexity, and functional specialization. Parallelly, neoplastic brain diseases, such as glioblastoma, highlight the vulnerabilities of this organ, exploiting developmental and evolutionary mechanisms for malignant growth. Thus, applying nanotechnology offers a dual advantage: Studying the evolutionary intricacies of the brain while addressing its pathological transformations. This chapter delves into the convergence of these fields and the role of nanotechnology in advancing research and treatment paradigms. The evolution of the brain from simpler structures in early vertebrates to the complex human brain involves key genetic, structural, and metabolic changes. Nanotechnology offers tools to dissect these processes with unprecedented precision as follows [116].
Gene expression studies: Nanoparticles conjugated with fluorescent probes have enabled gene expression patterns to be traced in ancestral and modern brains. For example, nanoscale sensors track the activity of evolutionarily significant genes across species, such as FOXP2 (language development) and SRGAP2 (neural connectivity) [116, 117].
Epigenetics: gold nanoparticles map histone modifications and DNA methylation, revealing epigenetic changes that underlie brain evolution [110, 118].
Neural circuitry mapping: (a) Nanoelectrodes. High-resolution nanoelectrodes have mapped evolutionary changes in synaptic structures, highlighting how interspecies variations in connectivity influence cognition and behavior [119]. (b) Brain organoids. Nano-scaffolds facilitate the growth of organoids that mimic ancestral and modern brains, enabling comparative studies on structural and functional evolution [93, 120].
Brain tumors, especially gliomas and glioblastomas, share several hallmarks with neoplastic development processes, including (a) cellular plasticity: tumor cells exhibit stem-like behavior, a feature central to neural development during evolution; (b) angiogenesis: tumors co-ordinate blood vessel formation and developmental pathways; (c) metabolic adaptability: similar to the brain evolutionary processes, tumors have evolved to adapt to limited oxygen and nutrient supplies [18, 116].
To understand tumor progression using nanotechnology we can consider (a) the tumor microenvironment: Nanoparticles equipped with biosensors detect pH, oxygen, and nutrient levels, providing real-time data on the tumor microenvironment [121]; (b) genomic mutations: nanoscale sequencing tools can identify key mutations driving tumorigenesis, such as those in the TP53 and IDH1/2 genes [122]; (c) three-dimensional tumor models: nano-engineered scaffolds replicate brain tumor growth, enabling studies on tumor invasion and drug resistance mechanisms [93, 123]; (d) single-cell analysis: nanoparticle-based techniques isolate and analyze individual tumor cells, uncovering heterogeneity and identifying therapy-resistant populations [124, 125].
The insights gained from studying brain tumoral development in relation with tumoral therapies translate into innovative nanotechnology-driven therapies: (a) Drug delivery systems: Nanocarriers bypass the blood–brain barrier to deliver targeted therapies, such as chemotherapeutics or RNA-based treatments, to tumor sites [111, 125, 126, 127]; (b) gene editing: CRISPR–Cas9 systems, delivered via nanoparticles, target oncogenes while offering insights into gene functions critical for brain evolution [128]; (c) Immunotherapy: Nanoparticles, present tumor antigens to the immune system, enhancing responses against glioblastoma. Immunotherapy performed using nanotechnology constitutes the presently employed promising approach in cancer treatment [21, 73, 112, 125, 129, 130, 131, 132, 133]; NP-related therapy was recently approved for GBM treatment—NanoThermTM [134]—and is based on iron oxide NPs and the thermal ablation of the tumor using a magnetic field. In parallel, other types of NPs are being evaluated, including polymer and lipid-based nanoformulations, nanodiscs, dendrimers, metallic, and silica. The advantages of these nanoscale drug carriers include improved penetration across the blood–brain barrier and lower systemic toxicity.
Nanotechnology in relation to signal transduction pathways plays a role in GBM immune and gene treatment mechanisms: (1) Nanostructured scaffolds can mimic the ECM, influencing signaling pathways [109, 113, 135]; (2) gold NPs, AuNPs, and those conjugated with EGF can activate the EGF receptor [110]; (3) nanocarriers for targeted cancer therapy—the liposomes coated with antibodies against the HER2 receptor—can deliver doxorubicin specifically to HER2-positive cancer cells, and modulate the PI3K/AKT pathway; (4) NPs can deliver small interfering RNAs (siRNAs), either small molecule inhibitors directly to cancer, or reinforce the IGF-I-R signal engaged in anti-IGF-I/phytochemical strategy [109, 135]. Ongoing cancer immune and gene therapeutic studies are analyzing nanotechnology mechanisms related to signal transduction pathways and immune response [131, 132, 136, 137, 138].
Future research should focus on: (a) integrating brain organoid studies with evolutionary genomics and tumor modelling; (b) enhancing biocompatibility and minimizing off-target effects; (c) refining nanocarriers for personalized and adaptive therapies, particularly immunotherapies [21, 112, 125, 131, 139].
This review describes long-term research surrounding GBM treatment and analyzes brain neoplastic development from stem cells differentiating into neuroglial cells, followed by the establishment of cancer gene therapy. This cancer gene therapy is based on immunogenic vaccines originating from brain neoplastic glial cells. These vaccines were prepared for the first time according to a strategy using three technologies in parallel: anti-gene, phytochemical, and nanotechnologies, all of which induce immunogenicity. The anti-tumor vaccine immunogenicity (CD8, CD28) is mediated by the overexpression of the common IGF-R signal transduction pathway, which was reinforced by nanotechnology. In the future, this approach will be applied by using a brain neoplastic stem cell therapy in relation to nanotechnology. The ability of nanotechnology to analyze, model, and intervene at the molecular level offers transformative potential for understanding the neoplasia of the brain and combating fatal malignant brain tumors—glioblastomas. Cancer immunotherapy, gene therapy, and nanotechnology have recently become increasingly intertwined, and the inclusion of nanotechnology in GBM immunotherapy is undergoing permanent progress [94, 132, 140, 141, 142].
GQ: Conceptualization; Formal analysis; Investigation; Methodology; Roles/Writing — original draft; and Writing — review & editing. MR: Formal analysis; Methodology; Validation; Roles/Writing — original draft. YCL: Formal analysis; Methodology; Validation. JT: Conceptualization; Formal analysis; Investigation; Methodology; Project administration; Supervision; Validation; Roles/Writing — original draft; and Writing — review & editing. All authors have read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We thank Dr W. Timmer (NIH - NCI, Bethesda, USA) - Program Director, Adult Brain Tumor Consortium), recently deceased, for his help in recognition of cancer gene therapy as a new oncology treatment proposed by J. Trojan thirty years ago. We need to underline the engagement in the described basic CNS research by Drs J. Uriel (CNRS, Villejuif) and in the clinical cancer gene therapy by Dr D. Anthony (CWRU University, USA), H. Kasprzak (Nicolaus Copernic University, Poland), and Pedro J. Penagos (National Cancer Institute, Colombia). We are greatful for a research collaboration in stem cells and neoplastic development to Dr S.J. Bueno (UNAB, Floridablanca, Colombia). We thank Dr A. Trojan (university License in English Literature) for verification of English spelling and grammar. We need to underline the logistic support of W. Arenas (GTF SA, USA) in the publication of previous experimental results.
This research received no external funding.
All authors declare no conflicts of interest. Despite receiving sponsorship from CEDEA & ICGT (Center of Oncology Diagnostic and International Cancer Gene Therapy Foundation), the judgments in data interpretation and writing were not influenced by this relationship.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



