1 Department of Symbiotic Nitrogen Fixation, Institute of Plant Physiology and Genetics of the National Academy of Sciences of Ukraine, 03022 Kyiv, Ukraine
2 Laboratory of Environmental Micropollutants and Microplastics, European Regional Centre for Ecohydrology of the Polish Academy of Sciences, 90-364 Lodz, Poland
Abstract
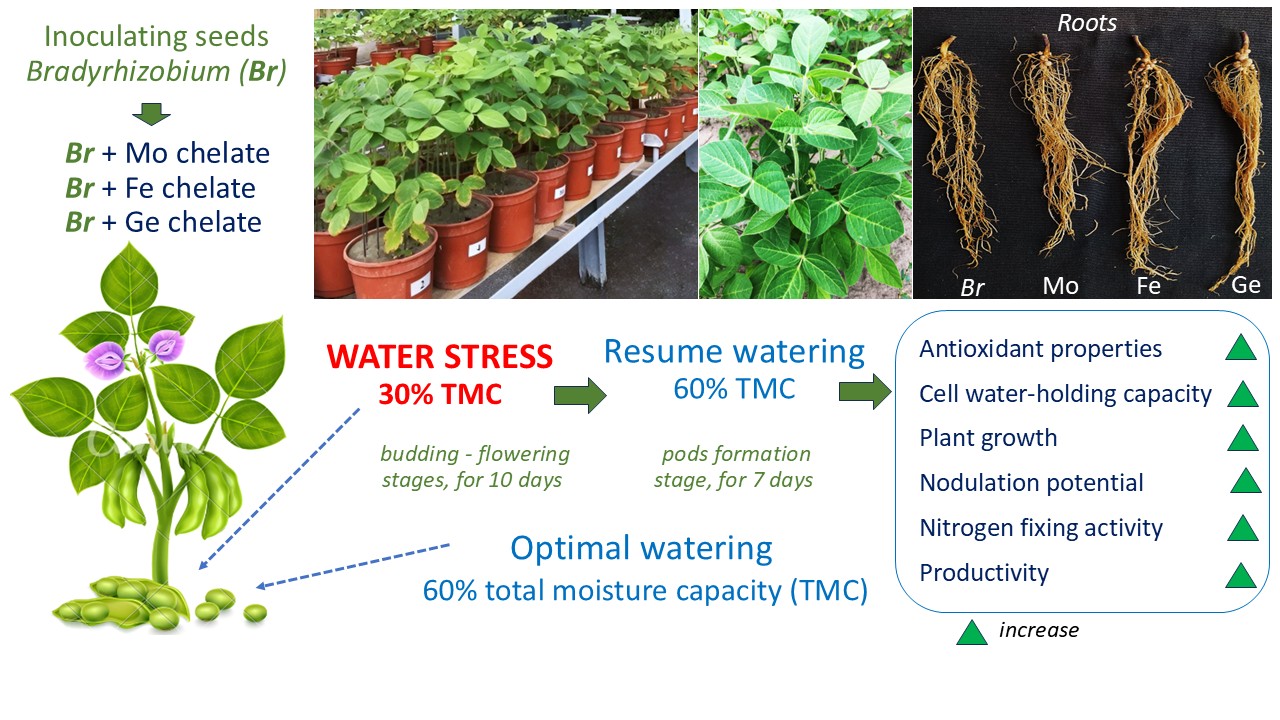
The tolerance and productivity of soybeans under the current climate change conditions can be increased by providing these crops with the necessary macro- and microelements. This can be achieved using effective Bradyrhizobium strains for seed inoculation and adding chelated trace elements.
Soybean Bradyrhizobium japonicum symbioses were cultivated by adding chelates of trace elements, such as iron (Fe), germanium (Ge), and molybdenum (Mo), to the culture medium, after which microbiological and biochemical analyses were performed.
The addition of chelated forms of Fe or Ge to the Bradyrhizobium culture medium promoted a change in the pro-oxidant-antioxidant balance in soybean nodules under different water supply conditions. This is due to the production of hydrogen peroxide in the nodules (an increase of 12.9%), as well as a twofold increase in the ascorbate peroxidase activity and a decrease in the levels of superoxide dismutase (by 40%) and catalase (by 50%) under water stress. Stimulation of nodulation and nitrogen fixation in soybeans (by 40.1 and 73.0%) and an increase in grain productivity (by 47.5 and 58%) were observed when using Bradyrhizobium inoculant containing Fe or Ge chelates. The inoculation of soybeans with Bradyrhizobium modified using Mo chelate causes similar changes in antioxidant processes as Fe or Ge chelates, but the soybean symbiotic capacity decreases under water stress.
Chelated forms of Fe or Ge as additional components in the Bradyrhizobium culture medium are effective in regulating the antioxidant status of soybeans under drought conditions and can simultaneously contribute to increased nitrogen fixation and grain productivity. These findings are important in expanding the current technologies used to grow this legume in risky farming areas caused by climate change.
Graphical Abstract
Keywords
- Bradyrhizobium japonicum
- Glycine max (L.) Merr.
- hydrogen peroxide
- superoxide dismutase
- ascorbate peroxidase
- catalase
- nitrogen fixing activity
- nodulation
- water stress
Sustainable agricultural development requires introducing innovative, environmentally safe agroecological solutions. The modern development of agrotechnological approaches and the emergence of plant protection products with new properties, including low trace element concentrations, can serve as a basis for developing bioecological solutions to reduce anthropogenic environmental impact. This is particularly relevant nowadays, as the food problem caused by climate change is becoming aggravated alongside several agronomic, environmental, and socioeconomic factors [1, 2].
Nanotechnological approaches can improve the quality and quantity of products and simultaneously help reduce the use of agrochemicals that pollute the environment [3]. Moreover, applying micronutrient nanoparticles causes significant changes in physiological and biochemical processes in plants, in particular, increasing enzyme activity, intensifying photosynthetic processes, and improving plant mineral nutrition [4, 5, 6]. As unique carriers of agrochemicals, nanomaterials facilitate targeted, controlled delivery of nutrients with improved plant protection. Therefore, these nanotechnological tools can support the development of high-tech agriculture in precisely managing and controlling resources (fertilizers, pesticides, herbicides) [7].
Presently, promising studies on the biological transformation of molecular nitrogen have been performed using nitrogen-fixing microorganisms alongside plants in natural ecosystems and agrophytocenoses [8, 9]. Soybean (Glycine max (L.) Merr.) is one of the most widely grown and consumed oilseeds; it is grown on an area of about 119 million hectares with a total annual production of 319 million tons [10]. A balanced supply of micronutrients is as important as macronutrients for the intensive growth and development of legumes, including soybeans [11, 12].
In the context of environmentally friendly crop cultivation technologies, using nanotechnological approaches in combination with bacterial preparations for legumes is relevant. Biological products based on nitrogen-fixing microorganisms modified using chelated forms of trace elements will help provide plants with nitrogen and, at the same time, the necessary trace elements in an accessible form. This is important for the optimal development of pulses and their productivity in the current climate change conditions. Detailed studies in which chelated forms of trace elements have been used in combination with Bradyrhizobium strains to increase the symbiotic capacity and productivity of soybeans under insufficient water supply are also relevant.
The nodulation process in the interaction of macro- and microsymbionts is regulated by the formation of reactive oxygen species (ROS) and changes in the activity of antioxidant enzymes [8]. ROS, especially hydrogen peroxide (H2O2), plays an important role in signaling processes in various cellular systems. The accumulation of H2O2 is indirectly related to the reaction cascade involved in calcium ion transport and the activation of mitogen-activated protein kinases (MAPKs) [13].
Soybean is a plant with ureide metabolism, in which most of the fixed nitrogen (up to 90%) is transported in the form of ureides—allantoin and allantoic acid [14]. The synthesis of these substances is accompanied by the release of H2O2, which is destroyed in peroxisomes by catalase (CAT). The CAT activity in nodules reflects the metabolic changes in root cortex cells when infected with bacteria [14]. CAT in the cell is a synergist of superoxide dismutase and prevents the excessive accumulation of the superoxide dismutase (SOD) reaction product: H2O2, an inhibitor of SOD.
Different localization of SOD isoenzymes in nodule tissues determines their specific functions [15]. The function of Cu/Zn-SOD may be associated with the growth of cell walls in meristems, infectious filaments, and apoplasts, as well as with the response of plants to bacterial infections. Mn–SOD is present in infected nodule cells and is involved in processes related to the protection and functioning of symbiosis. Iron (Fe)–SOD is localized exclusively in the chloroplast stroma, and its functions in legumes are the least studied.
The ascorbate-glutathione pathway of H2O2 utilization is essential for nodule function [16]. Ascorbate peroxidase (APO) is the key enzyme in this pathway and is found everywhere in nodules, especially in infected and parenchymal cells. In infected cells, APO protects leghemoglobin and other proteins from H2O2. Meanwhile, in the parenchyma of nodule cells, APO can participate in the creation of a diffusion barrier for oxygen, thus controlling the entry of oxygen into the infected zone [16].
It has been shown that legume infection significantly modifies the metabolism of the host plant, creating optimal conditions for nodulation and the effective functioning of symbiosis [8]. Future research in this field will reveal additional aspects of the defense responses that legumes present under stresses induced by climate change.
Indeed, our previous studies demonstrated that inoculating soybeans with Bradyrhizobium containing chelated forms of trace elements led to changes in the activity of SOD and ascorbate peroxidase in soybean nodules and leaves under water stress [12]. However, such studies require an integrated approach to studying the issue in detail, considering the functioning of key components in the antioxidant systems of plants and the implementation of symbiotic capacity under water stress.
The research hypothesis for this study is that an effective Bradyrhizobium inoculant modified with chelated trace elements will increase soybean symbiotic capacity and productivity. This will be achieved by including key plant antioxidant defense system components, which contribute to the tolerance of soybeans to water stress.
This study aimed to evaluate the effectiveness of chelated forms of iron (Fe), germanium (Ge), and molybdenum (Mo) as components of Bradyrhizobiumculture medium for improving soybean development with a focus on (i) pro-antioxidant status in nodules by assessing the activity of key antioxidant enzyme complexes and hydrogen peroxide content; (ii) nodulation and nitrogen fixation processes; (iii) soybean symbiotic capacity and productivity.
Before sowing, soybean seeds (Glycine max (L.) Merr.) of the Almaz variety were inoculated with effective Bradyrhizobium japonicum B1-20. Almaz is an early maturing, medium-oil variety that is highly adaptable to climatic conditions and is recommended for cultivation in the forest-steppe region of Ukraine. Glycine max was created by hybridizing the Moldovan line 3/86 and the Swedish variety Fiskeby 840-5-3 [17]. The B. japonicum B1-20 used for research was obtained from the museum collection of nitrogen-fixing microorganisms at the Institute of Plant Physiology and Genetics, National Academy of Sciences of Ukraine [18].
The inoculation was performed using a culture medium supplemented with chelated forms of trace elements Fe, Ge, and Mo in a dilution of 1:1000 to the culture medium [19, 20]. The chelator was citric acid. In the other treatments, the soybean seeds were inoculated with B. japonicum cultures (titer 108 cells/mL) without any chelated forms of trace elements in the culture medium.
Bradyrhizobium was cultivated in 750 mL flasks on a shaker (220 rpm) at a temperature of 26–28 °C. The microorganisms were cultured on a liquid mannitol yeast medium with the following composition (g/L): KH2PO4: 0.5; MgSO4: 0.2; NaCl: 0.1; yeast extract: 1.0; mannitol: 10.0. Bradyrhizobium cultures were removed in the exponential growth stage (92–96 hours). The amount of inoculum introduced into the flasks was 2% of the medium volume. In total, 108 cells/mL of Bradyrhizobium suspension was added to 200 mL of nutrient medium. The purity of bacterial cultures was evaluated following sowing on meat peptone agar.
The plants were grown under strictly controlled conditions in 5 kg vessels with a sterile substrate (sand) supplemented with Hellriegel’s nutrient mixture (0.25 of the normal nitrogen rate); the plants were cultivated under natural lighting and optimal moisture supply, i.e., 60% of total moisture capacity (TMC). Before sowing the seeds, the substrate was analyzed for moisture and moisture capacity, which was considered when creating different plant watering modes. At the budding and flowering stages, two moisture supply regimes were produced by controlled watering: optimal: 60% TMC and water stress: 30% TMC. Water stress was maintained for 10 days before the pod formation stage for the stressed plants. Then, watering was restored to the optimal level of 60% TMC (Fig. 1). Nodule samples were analyzed at stages: budding, flowering, and pod formation in 8–12-fold repetition.
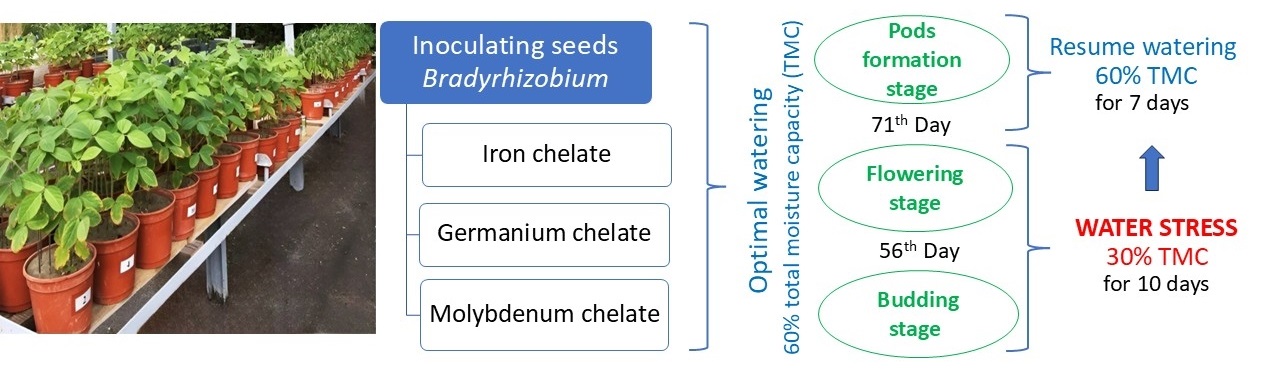 Fig. 1.
Fig. 1.
Schematic representation of the pot experiment. TMC, total moisture capacity.
Soybean sowing was conducted in early May. The seeds were sown to a depth of 3–5 cm at the rate of 40 seeds per one linear meter when the upper sowing layer of soil was 10–15 °C (Fig. 2). Plots were placed randomly. The accounting area was 5 m2. Repetition was fourfold. The vegetative mass of plants was measured during the growth period. At the trifoliolate leaves, flowering, and pod formation stages, plants were selected to determine the nodulation ability of B. japonicum in tenfold repetitions. The grain harvest was recorded by hand and then weighed.
 Fig. 2.
Fig. 2.
Schematic representation of the experiment in the field.
To determine the nodulation potential of Bradyrhizobium, 20 typical plants of each treatment were selected, and the roots were washed. Then, nodules were separated, and the mean number and mass per plant were calculated. The results are presented as the number (pcs) and mass (g) of nodules per plant.
Nitrogen-fixing activity (NFA) was measured on a GC system 6850 Gas Chromatograph (Agilent, Santa Clara, CA, USA) with a flame ionization detector [21]. Gas separation was carried out on a column (Supelco Porapak N) at a thermostat temperature of 55 °C and a detector temperature of 150 °C. Helium was used as the carrier gas (20 mL per minute). The volume of the analyzed gas mixture sample was 1 cm3. Pure ethylene (Sigma-Aldrich, No. 536164, St. Louis, MO, USA) was used as a standard. NFA is presented in molar units of ethylene formed (µmol C2H4) per plant per hour.
Hydrogen peroxide (H2O2) content was determined using the ferrothiocyanite method [22]. The plant material was treated with a cooled solution of 5% trichloroacetic acid at a ratio of 1:3 (weight/volume). The supernatant was obtained by centrifugation at 14,000 rpm and 4 °C for five minutes. The peroxide concentration was determined spectrophotometrically at a wavelength of 480 nm following a color reaction with potassium thiocyanate; the result was compared against a calibration curve. The results are presented in µmol per gram of dry weight (DW). The samples were dried to a constant value at +105 °C to determine the dry matter mass.
The plant material was homogenized using chilled 60 mM phosphate buffer (pH 7.5)
in a 1:2 (weight/volume) ratio containing 2 mM ethylenediaminetetraacetic acid
(EDTA), 1 mM phenylmethylsulphonyl fluoride, 5 mM
The SOD (EC 1.15.1.1) activity was determined by its ability to inhibit the
photochemical reduction of nitroblue tetrazolium [24]. The reaction mixture
contained 50 mM phosphate buffer (pH 7.8), 13 mM methionine, 2 µM
riboflavin, 63 µM p-nitroblue tetrazolium, 0.1 mM EDTA, and 100 µL
enzyme extract. The reaction proceeded for 15 min under the illumination of 70
µmol quanta/(m2
The activity of ascorbate peroxidase (APO) (EC
1.11.1.11) was determined by the decrease in optical density at 290 nm over 2
minutes following ascorbate oxidation (
To obtain the enzyme extract, part of the plant material was triturated (ratio
1:2) with cooled 0.5 M Tris-HCl buffer (pH 7.8) containing 5 mM
All statistical analyses were performed using the STATISTICA ver. 13.3 software
package [26]. The Kruskal–Wallis test was used to compare the differences
between the Bradyrhizobium treatments and the Ge, Fe, and Mo chelates
treatments for each growing condition, viz. optimal watering and water
stress. Significant differences are indicated by letters in the figures. The
figures show the arithmetic mean and the standard errors as
Under optimal watering, soybeans inoculated with Bradyrhizobiumcontaining Fe or Ge chelated forms of trace elements had the highest H2O2 content in nodules by 34.8% and 40.2%, respectively, compared to the treatment without chelates (Fig. 3). The use of Bradyrhizobium and Mo chelate treatment resulted in H2O2 production similar to that of the treatment without chelates.
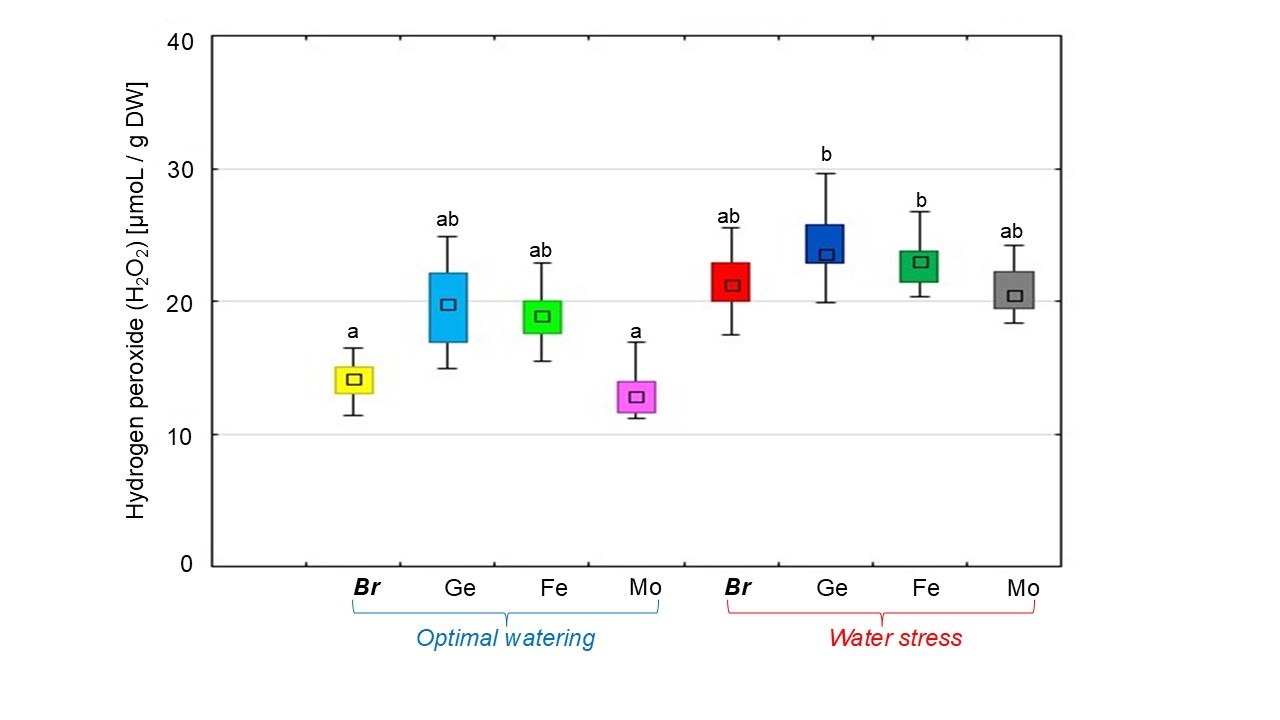 Fig. 3.
Fig. 3.
The effect of adding chelated trace elements to the
Bradyrhizobium cultivation medium on hydrogen peroxide
(H2O2) production in soybean nodules under different water supply
conditions. The total value of the indices analyzed during the budding,
flowering, and pod formation stages; the letters indicate significant differences
between the treatments (p
Water stress induced an over-generation of H2O2 in nodules by 53.1% compared to soybean plants under optimal irrigation (Fig. 3). However, dehydration of soybeans caused H2O2 production in nodules formed by Bradyrhizobium with Ge or Fe chelates (by 12.9%) (Fig. 3). During the treatment with the Mo chelate, the accumulation of H2O2 in nodules was at the same level as when soybeans were treated under stress conditions without chelates.
Under optimal conditions of soybean watering, an increase in the SOD activity in nodules formed by Bradyrhizobium with the addition of chelated forms of trace elements was revealed compared to the level of its activity in soybean treatment without chelates (Fig. 4). The highest enzymatic activity was observed in the soybean nodules treated with the Ge chelate.
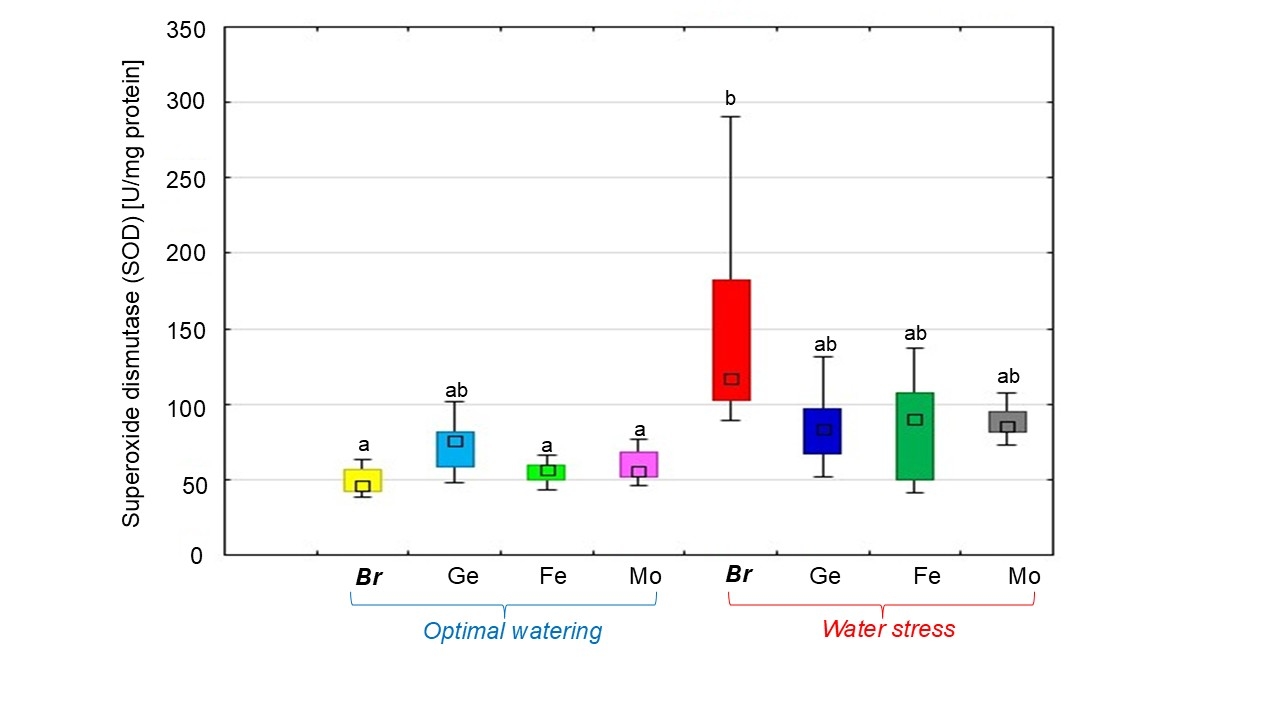 Fig. 4.
Fig. 4.
The effect of adding chelated forms of trace elements to the
Bradyrhizobium cultivation medium on superoxide dismutase (SOD) activity
in soybean nodules under different water supply conditions. The total value of
the indices analyzed during the budding, flowering, and pod formation stages; the
letters indicate significant differences between the treatments (p
Water stress resulted in SOD activity being three times higher in soybean nodules during the treatment without chelates compared to under optimal watering (Fig. 4). In contrast, the use of chelated trace elements in the bacterial culture decreased SOD activity by 40% under water stress. Moreover, in the treatments with chelated trace elements, the enzyme activity under dehydration approached the same level as in soybeans under optimal watering (Fig. 4).
A decrease in the CAT activity level in soybean nodules under inoculation with Bradyrhizobium with chelated forms of Ge, Fe, or Mo under optimal plant watering was detected (Fig. 5). At the same time, this index was almost the same in soybean nodules under optimal plant growing conditions. Water stress led to an increase in CAT activity in soybean nodules during the treatment without chelates by 56% compared to optimal watering (Fig. 5). The use of chelated forms of trace elements in combination with Bradyrhizobium reduced CAT activity in soybean nodules under dehydration by 50% (Fig. 5). At the same time, the levels of CAT activity in the treatments with chelates were almost at the same level.
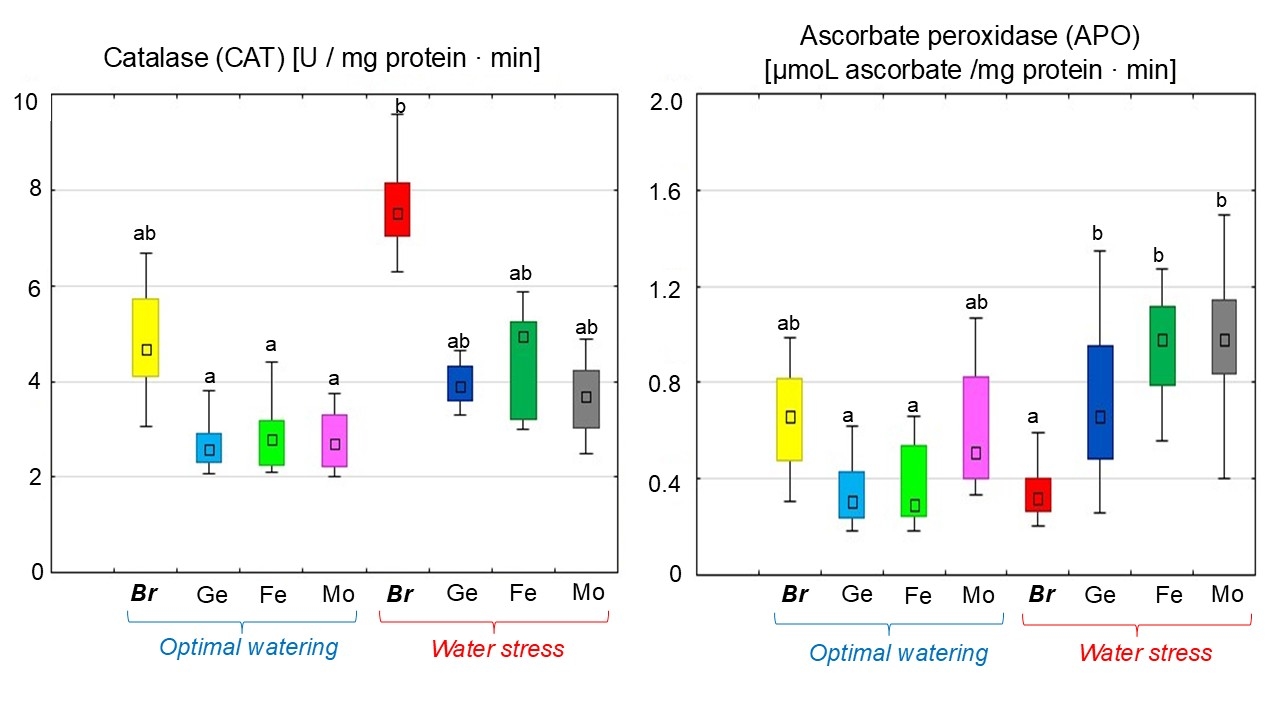 Fig. 5.
Fig. 5.
The effect of adding chelated trace elements to the
Bradyrhizobium cultivation medium on CAT and APO activity
in soybean nodules under different water supply conditions. The total value of
the indices analyzed during the budding, flowering, and pod formation stages; the
letters indicate significant differences between the treatments (p
A decrease in APO activity in soybean nodules formed by Bradyrhizobium containing chelated forms of Ge (by 46.5%) and Fe (by 43.2%) was observed; meanwhile, there were no significant changes following the treatment with Mo chelate compared to without chelates under optimal plant watering (Fig. 5). Water stress induced a decrease in APO activity in soybean nodules inoculated with Bradyrhizobium without chelate (by 46.3%) (Fig. 5). The addition of chelated forms of trace elements to the bacterial culture increased APO activity in soybean nodules by twofold under dehydration; meanwhile, the enzyme levels remained similar for all treatments with chelates.
The nodulation processes were stimulated following an optimal water supply in all treatments with Bradyrhizobium seed inoculation containing chelated trace elements. The largest number of nodules (28 pcs/plant) was recorded for soybeans with the Ge chelate, which was 92.2% higher than for the treatment without chelates (Fig. 6). In the treatments with the Fe or Mo chelates, the nodular numbers were increased by 65.8% and 38.8%, respectively (Fig. 6). An increase in the nodules weight was detected under inoculation with Bradyrhizobium and Ge (0.36 g/plant) and Fe (0.40 g/plant) chelates (Fig. 6). At the same time, the Mo chelate treatment under optimal watering reduced the nodule weight by 34.7% (Fig. 6).
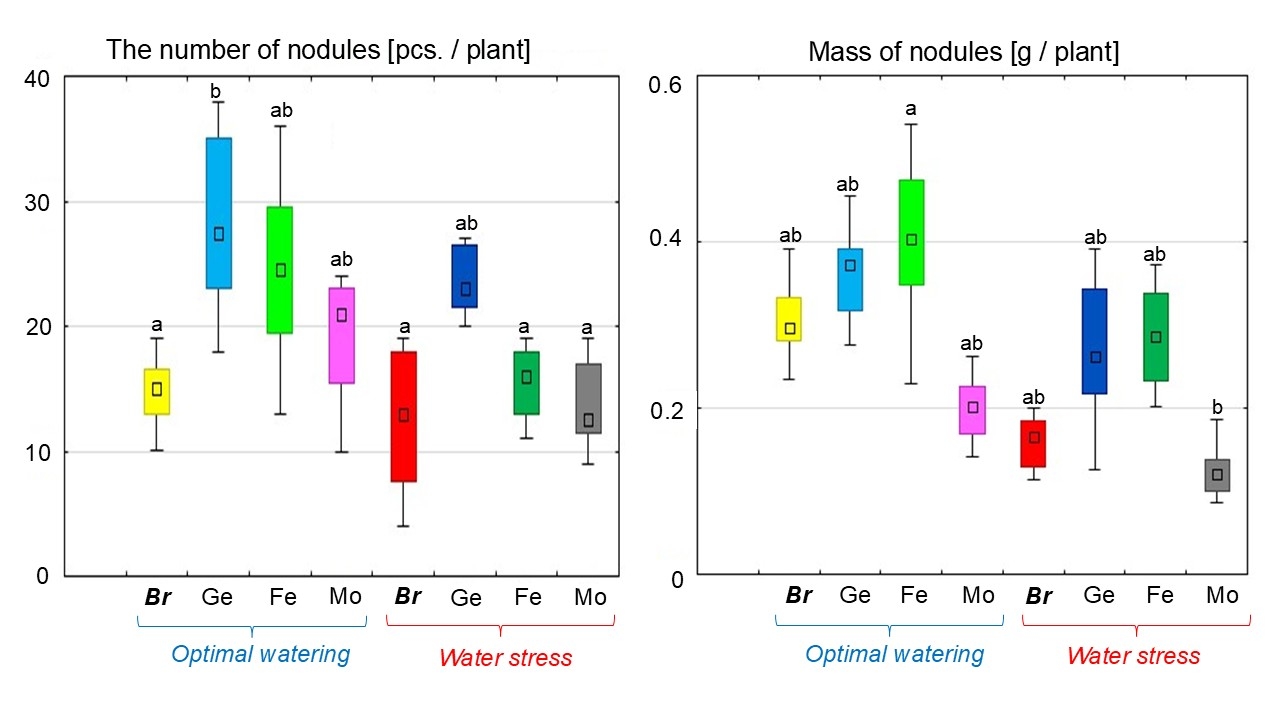 Fig. 6.
Fig. 6.
The effect of adding chelated forms of trace elements to the
Bradyrhizobium cultivation medium on the nodulation in soybeans under
different water supply conditions. The total value of the indices analyzed
during the budding, flowering, and pod formation stages; the letters indicate
significant differences between the treatments (p
Adding chelated trace elements to the Bradyrhizobium culture under dehydration promoted nodulation on soybean roots. The highest number of nodules was recorded following the treatment with Bradyrhizobium containing the Ge chelate (23 pcs/plant), which was 88.8% higher than for the treatment without chelates under stress (Fig. 6). In the Fe or Mo chelate treatments, the number of nodules increased by 23.8% and 10.0%, respectively (Fig. 6).
Water stress without chelates reduced the weight of soybean nodules by 48% (Fig. 6). Under dehydration in the treatments with complex inoculation of Bradyrhizobium and Ge or Fe chelates, nodule weight increased by 80% compared to the soybeans without chelates (Fig. 6). However, the lowest nodule weight was recorded under water stress for the treatment with Bradyrhizobium and Mo chelate (Fig. 6).
Soybean inoculation with Bradyrhizobium containing chelated forms of Fe or Ge was found to increase nitrogen-fixing activity (NFA) levels by 70.2% and 59.6%, respectively, under optimal watering conditions (Fig. 7). At the same time, the NFA level was similar for soybeans treated with Mo chelate and soybeans treated without chelates.
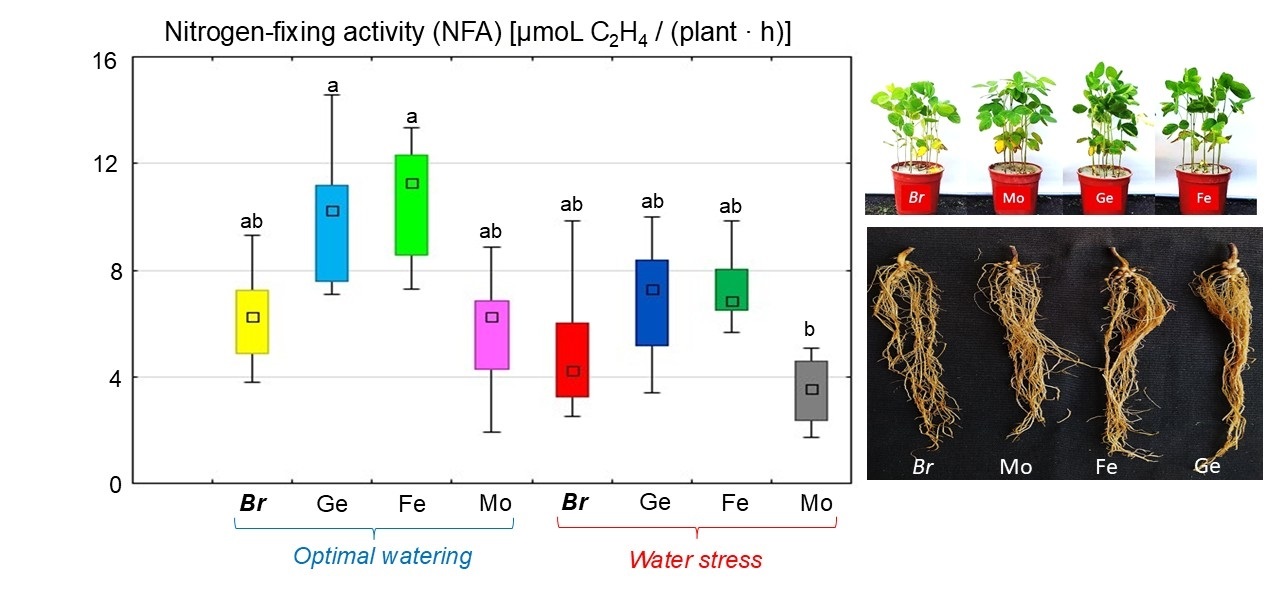 Fig. 7.
Fig. 7.
The effect of adding chelated trace elements to the
Bradyrhizobium cultivation medium on the NFA in soybean nodules under
different water supply conditions. The total value of the indices analyzed
during the budding, flowering, and pod formation stages; the letters indicate
significant differences between the treatments (p
Under water stress, a 21.2% decrease in NFA was observed in soybeans without chelates (Fig. 7). In contrast, adding chelated trace elements to the Bradyrhizobium culture promoted NFA in soybean nodules under dehydration. In particular, for these treatments, NFA increased by 39.9% (Ge chelate) and 48.2% (Fe chelate) as in soybean without chelates (Fig. 7). Meanwhile, the Mo chelate treatment produced the lowest level of NFA under water stress.
In the plot field experiment, inoculation of soybean with Bradyrhizobium containing chelated forms of trace elements increases the efficiency of nitrogen fixation by 73.0% (Ge chelate) and 40.1% (Fe chelate) compared to soybeans treated without chelates (Fig. 8). An increase in grain weight was found in treatments using pre-sowing seed inoculations of Bradyrhizobium modified with Ge chelate (by 58%) and Fe chelate (by 47.5%) compared to soybeans inoculated without chelates (Fig. 8).
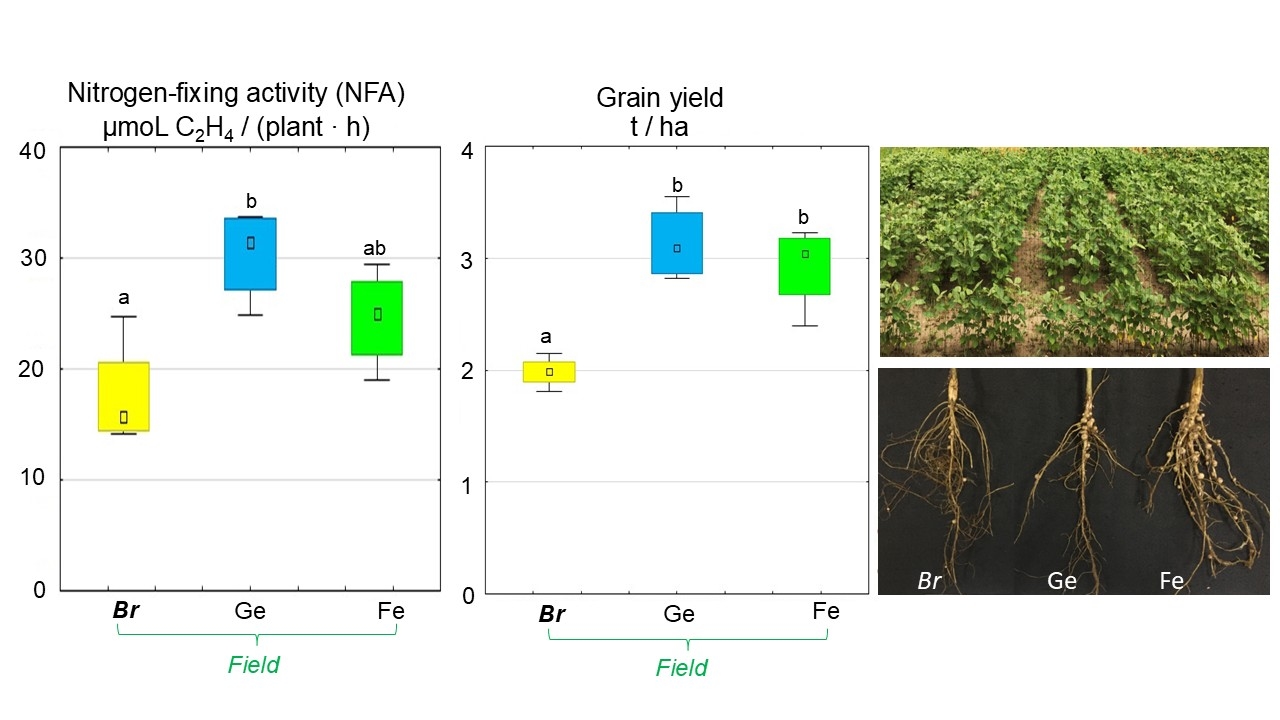 Fig. 8.
Fig. 8.
The plot field experiment of the effect of pre-sowing
inoculation with Bradyrhizobium containing chelated trace elements on
nitrogen fixation and grain yield in soybeans. The total value of the indices
analyzed during the budding, flowering, and pod formation stages; the letters
indicate significant differences between the treatments (p
Micronutrient deficiencies for optimal plant function have recently become an increasingly common problem due to environmental factors caused by, among other things, frequent droughts and insufficient moisture. In this regard, it is easy to predict that a regular increase in plant micronutrient deficiencies can lead to a significant deterioration in crop quality in the near future; therefore, this problem is the subject of intensive research [27]. Micronutrients can also be toxic in high concentrations, threatening plant growth and development. Hence, our research aimed to study the peculiarities of using chelated forms of trace elements to improve the function of soybean, Bradyrhizobium japonicum, and symbiotic systems under optimal and insufficient moisture supply conditions.
The data analysis showed that the addition of chelated forms of trace elements Fe or Ge to the Bradyrhizobium culture medium induced, under optimal moisture conditions, changes in the pro-antioxidant status in soybean nodules by increasing the content of hydrogen peroxide and SOD activity and simultaneously reducing the levels of APO and CAT activity in nodules. This was accompanied by stimulation of nodulation and nitrogen fixation processes in soybeans under inoculation with Bradyrhizobium containing Ge or Fe chelates. Inoculation of soybeans with Bradyrhizobium and Mo chelate revealed similar changes in antioxidant processes; however, these changes were not as intense as in the treatments with Fe or Ge chelates. At the same time, nodulation stimulation and the absence of effective changes in nitrogen fixation were recorded in soybeans inoculated with Bradyrhizobium containing the Mo chelate.
Limited Fe availability in soils is one of the key factors affecting crop yields and quality, especially in alkaline and calcareous soils. This is due to the physical, chemical, and biological processes in the rhizosphere that occur due to the interaction between soil, microorganisms, and plants. An optimal amount of iron is believed to be necessary for plant growth; however, the chemical form of the metal is also crucial for metabolic activity and other plant functions [28].
Legumes in symbiosis with nodule bacteria have an increased need for iron in the synthesis of Fe-containing proteins in the host plant, including leghemoglobin, as well as bacteroids for the functioning of nitrogenase and cytochrome, affecting nodule formation and development [29]. In root cells, Fe is chelated with organic acids, such as citrate and nicotinamide, and distributed to other parts of the plant in the forms of Fe (II) and Fe (III) [30]. The formation of symbiosis is accompanied by bacteroids, which are surrounded by a plant-derived membrane, the symbiosome, which regulates the flow of nutrients to the bacteroid. To date, only one Fe (II) transporter has been characterized in nodules, GmDMT1 (Glycine max divalent metal transporter 1), and is located on the symbiosome in the soybean [29]. When analyzing the genomes of legumes, including Medicago truncatula and Glycine max, other members of the NRAMP (natural resistance-associated macrophage protein), YSL (yellow stripe-like), VIT (vacuolar iron transporter), and ZIP (Zrt-, Irt-like protein) transport family were identified, which show increased expression in nodules and are important in transporting Fe and other metals across symbiotic membranes [31, 32].
Ge in plants has been shown to improve plant growth and development due to its ability to directly absorb ROS because of the 4S24P2 electronic configuration [33]. It has a high effect on nutrient uptake, plant photosynthesis, photosynthetic pigments content, activity of endogenous antioxidant enzymes and non-enzymatic compounds, and promotes the accumulation of polysaccharides and amino acids in plants [30]. At the same time, the excessive accumulation of Ge in the roots and shoots of plants disturbs the metabolic processes [34]. Therefore, studying the mechanisms through which Ge uptake, transport, and distribution are regulated in plants requires further in-depth research.
The highest level of H2O2 production under water stress was observed in soybean nodules inoculated with Bradyrhizobium containing Ge or Fe chelates compared to other treatments. This was not observed in the symbiotic system with Bradyrhizobium modified with Mo chelate. A similar trend in the H2O2 content in soybean nodules was observed under optimal watering of plants. Meanwhile, a decrease in the levels of SOD and CAT activity and an increase of APO in nodules under the influence of water stress were recorded in all treatments with chelated forms of trace elements. Thus, the chelated forms of trace elements combined with Bradyrhizobium significantly changed the direction of antioxidant processes in soybeans under insufficient and optimal water supplies.
ROS play a key role in plant protection against biotic and abiotic stress factors. One of the fastest protective responses to stress factors is the formation of H2O2 [13]. It has been proven that H2O2 is induced in the response by plants to environmental factors [35]. H2O2 can act as a signaling molecule and induce (apparently through the activation of protein kinases and protein phosphorylation) the expression of protective genes, which is important for the adaptation of plants to stressful conditions. Methodological DNA approaches allowed us to assess gene expression in response to ROS. In particular, exposure of Arabidopsis cells to H2O2 resulted in changes in about 175 genes, of which 113 encoded proteins with antioxidant functions or associated with protective responses to stress, and the remaining genes encoded proteins with signaling functions [36].
There is convincing evidence that ROS are important in signaling processes for establishing legume-rhizobial symbiosis [8]. This interaction involves complex molecular signaling between the host plant and symbiont, leading to the infection of root hairs by nodule bacteria and the formation of nodules, which fix atmospheric nitrogen. Our studies have shown that chelated forms of trace elements as additional components of Bradyrhizobium culture medium affect plant metabolism, causing changes in antioxidant processes both under optimal and insufficient watering conditions. However, these changes have a positive impact on the realization of plant defenses, as well as improve soybean symbiotic capacity and productivity (Fig. 9). Earlier, we found [12] that Bradyrhizobium inoculation with chelated forms of trace elements increased the water-holding capacity of cells and optimized water status and growth processes in soybean under water stress (Fig. 9).
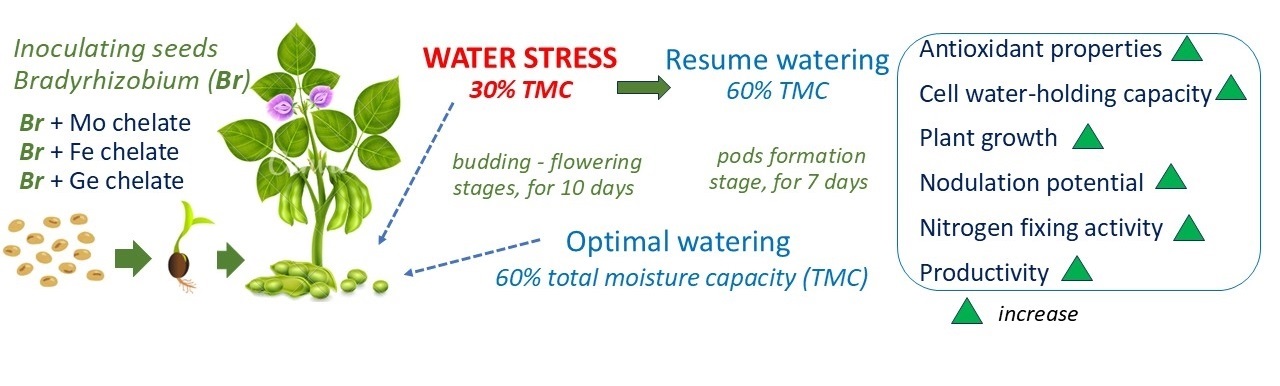 Fig. 9.
Fig. 9.
Schematic representation of the influence of chelated forms of trace elements in Bradyrhizobium culture medium on the formation of soybean symbiotic capacity. The directions of the arrows indicate the changes in the levels of the specified parameter: increase (green).
It has been shown that under different stress conditions, the Nramp and YSL gene families help translocate Fe, which acts as a regulatory element and protects plants from stresses caused by salinity, drought, and heavy metals. This is because Fe activates plant enzymatic antioxidant complexes such as CAT, peroxidase, and SOD isoforms, which act as ROS scavengers [37]. In addition, Fe deficiency and excess can disrupt the homeostasis of the plant cell and lead to a decrease in the photosynthetic and respiration rate and an increase in the accumulation of Na⁺ and Ca2+ ions, resulting in excessive ROS formation [38, 39].
Molybdenum is one of the most widely consumed elements by plants and provides optimal conditions for maximizing the efficiency of enzymes involved in nitrogen assimilation, as well as other Mo-containing enzymes in legumes [40]. Molybdenum acts as a cofactor for the nitrogenase and nitrate reductase enzymes, which are directly involved in the biological fixation and further nitrogen assimilation in legumes [11]. Adequate molybdenum supply avoids competition for this trace element between nitrogenase and nodule nitrate reductase, improving the nitrogen supply to the plant [41]. The current understanding of how plants take up Mo from the soil solution and later redistribute it in the plant remains unclear. It is known that its uptake by the root system, transport in the plant, and the manifestation of functions depend on soil pH and the influence of environmental factors [11, 42]. It has been shown that limited water resources and lack of access to sufficient water for agricultural production in terms of quantity and quality risk jeopardizing Mo uptake [43]. Therefore, a thorough analysis of existing studies and a re-examination of the known mechanisms of Mo anion transport in plants will help determine how this important trace element affects the functioning of the symbiosis of legumes with Bradyrhizobium under insufficient water supply conditions.
The intensity of metabolism and growth process recovery after stress factors impact, particularly insufficient water supply, indicates the development of plant self-regulation processes and its ability to restructure metabolic processes and adapt to growing conditions [8]. The capacity of legumes to fully realize their symbiotic properties under appropriate growing conditions is important for forming reproductive organs and future yields [9].
Adding chelated forms of Ge or Fe to the Bradyrhizobium culture medium enhanced nodulation and NFA in soybean nodules under different water conditions. And their use in the field positively affected soybean grain yield. Inoculating soybeans with Bradyrhizobium containing Mo chelate did not affect nitrogen fixation; therefore, we did not use this complex in the field. It is worth noting that we did not detect any negative impact of soybean inoculation with Bradyrhizobium containing Mo chelate. Moreover, in the treatment with Mo chelate, similar changes in pro-antioxidant processes in soybean nodules under insufficient and optimal water supply were recorded as in the case of Fe or Ge chelates; however, these were not as intense. The response of the symbiotic apparatus of soybean in the treatment with MO chelate to watering conditions was similar to that of soybeans without chelates.
Co-inoculation of soybean with Bradyrhizobium and chelated forms of trace elements Ge, Fe, or Mo leads to changes in pro-antioxidant processes in nodules and simultaneously affects the soybean symbiotic capacity under optimal and insufficient water supply. Chelated forms of Fe or Ge in combination with Bradyrhizobium optimized antioxidant status in nodules under water stress and improved nodulation and nitrogen fixation in soybean, as well as grain productivity. Inoculation of soybean with Bradyrhizobium with Mo chelate causes changes in the antioxidant status in nodules, which are not accompanied by a positive effect on the symbiotic apparatus under water stress.
Inoculating soybeans with Bradyrhizobium containing Ge or Fe chelated forms effectively enhances protective antioxidant properties and realizes symbiotic capacity under insufficient water supply. Using such a seed inoculation complex improves soybean grain yield, which can be especially important for growing the crop in risky farming areas, especially under current climate change conditions.
ROS, reactive oxygen species; H2O2, hydrogen peroxide; SOD, superoxide dismutase; CAT, catalase; APO, ascorbate peroxidase; TMC, total moisture content; NFA, nitrogen-fixing activity; Ge, germanium; Fe, iron; Mo, molybdenum; EDTA, ethylenediaminetetraacetic acid.
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reason able request.
TN and SK contributed to the study conception and design. Material preparation, data collection and analysis were performed by TN. The first draft of the manuscript was written by TN and SK. Supervision: SK. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work to take public respon sibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity. Both authors contributed to editorial changes in the manuscript.
Soybeans (Glycine max (L.) Merr.) of the Almaz variety were used in this study. Soybean seeds were kindly provided by Poltava State Agrarian Academy (Poltava, Ukraine). Originator–Poltava State Agrarian Academy, Ukraine. The variety from the 2007 Register of Plant Varieties of Ukraine and recommended for cultivation in the forest steppe of Ukraine.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBE33505.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









