1 Department of Experimental Biology and Biotechnology, Institute of Natural Sciences and Mathematics, Ural Federal University Named after the First President of Russia B.N. Yeltsin, 620002 Ekaterinburg, Russia
2 Laboratory of Biotechnology, Institute of Natural Sciences and Mathematics, Ural Federal University Named after the First President of Russia B.N. Yeltsin, 620002 Ekaterinburg, Russia
3 Department of Life Sciences, GITAM School of Science, Gandhi Institute of Technology and Management, 530045 Visakhapatnam, Andhra Pradesh, India
†These authors contributed equally.
Abstract
Amalgamation of metal-tolerant plant growth promoting rhizobacteria (PGPR) with biochar is a promising direction for the development of chemical-free biofertilizers that can mitigate environmental risks, enhance crop productivity and their biological value. The main objective of the work includes the evaluation of the influence of prepared bacterial biofertilizer (BF) on biometric growth parameters as well as physiological and biochemical characteristics of rapeseed (Brassica napus L.) at copper action.
The prepared BF was based on novel metal tolerant strain of PGPR Buttiauxella sp. EA20 isolated from the rhizosphere of orchid Epipactis atrorubens and birch wood biochar (BC). The pot-scale experiments included six treatments: peat-containing control substrate (CS); CS + 200Cu (200 mg Cu/kg of soil); CS + 5% BC (v/v); CS + 5% BC + 200Cu; CS + 5% BF (v/v); CS + 5% BF + 200Cu.
Single Cu treatment caused the decrease in rapeseed leaf area, shoot and root length, fresh and dry biomass, as well as an increase in water saturation deficit, possibly due to damage of cell membranes by lipid peroxidation. Addition of BF or BC alone mitigated these harmful effects of copper. Application of BF, regardless of Cu addition, increased the rapeseed leaf area (1.6 times on average), plant fresh and dry biomass (2.5 times on average), and photosynthetic pigment content (1.8 times on average). In addition, BF treatment along with Cu enhanced the antioxidant activity of B. napus due to the accumulation of non-enzymatic antioxidants such as carotenoids, free proline and soluble phenolic compounds, including flavonoids. Moreover, plant enrichment with copper and essential macronutrients such as nitrogen, phosphorus and potassium was observed.
The study concludes that application of complex biofertilizer based on metal tolerant PGPR strain Buttiauxella sp. EA20 and birch wood biochar mitigated the harmful effects of copper, enhanced the rapeseed growth and increased its biological value. Future perspective includes evaluation of the potential for using the resulting biofertilizer to improve the growth and biofortification of other crop species.
Keywords
- Brassica napus L.
- plant growth promoting rhizobacteria (PGPR)
- essential metal
- bacterial biofertilizer
- nutrient enrichment
- growth parameters
- photosynthetic productivity
- low molecular weight antioxidants
A growing demand for food accompanies steady population growth. Lately, there has been an upsurge in agricultural production attributed to the adoption of high-yielding varieties and an escalation in the utilization of agro-chemicals. These chemicals serve dual purposes, functioning as fertilizers and safeguards against plant pathogens [1, 2]. Nevertheless, the use of chemical fertilizers and pesticides results in an adverse nutrient balance in soils, contributing to environmental contamination and the presence of various pollutants, including heavy metals (HMs), in agricultural production [3, 4, 5]. One of such most common metals is copper (Cu) which is an essential element for humans, animals and plants, when present in adequate amounts, while in excess exerts detrimental effects [6, 7, 8]. Since Cu is a redox-active metal, it catalyzes the formation of reactive oxygen species (ROS), such as superoxide radical, hydrogen peroxide, and hydroxyl radicals, which are involved in free radical chain reactions of membrane lipids and proteins, thus causing oxidative degradation [8, 9]. The formation of ROS leads to oxidative stress and damages DNA, proteins, and lipids, and also affects plant growth [10]. Moreover, high concentrations of Cu can also lead to the development of metal toxicosis (chlorosis, necrosis, inhibition of the growth of roots and shoots), which ultimately causes death of the plants [11]. Plant adaptation to the toxic effect of Cu is based on the functioning of general (low molecular weight organic stress-protective compounds, protective macromolecules, and antioxidant enzymes) and specialized resistance mechanisms (chelation, sequestration, and compartmentation) [8, 12].
Simultaneously, copper plays a crucial role in several biochemical and physiological processes and its insufficiency or excess in soil can have a negative effect on plants and other living organisms [6, 12]. Copper is a cofactor for several important enzymes such as superoxide dismutase, polyphenol oxidase, ascorbate oxidase, etc. [8, 9]. Moreover, it also has a positive effect on the content of chlorophylls and carotenoids in the leaves, and takes an active part in the hormonal regulation of plants [8, 11]. The entry of Cu is carried out in the form of a cation or chelate compound and remains in plants in the form of ions and as part of complex organic compounds [8, 12]. In many agricultural regions, the phytoavailability of copper is low. In particular, copper deficiency is observed in plants growing on calcareous or saline soils, especially in arid and semi-arid conditions [13].
One of the most promising approaches to address the challenge of ensuring balanced nutrition for the human population is biofortification of plant raw materials and crop products. The concept of “biofortification” includes an increase in the content of deficient nutrients in crops, and such biologically active substances as vitamins, antioxidants, etc. It also involves improving the growth and overall productivity of plants [13, 14]. Of particular interest is microbiological biofortification, a process that employs bacteria to enhance the nutritional value of raw materials and food products. One of the most effective agrobiotechnologies that help to increase not only the productivity of cultivated plants, but also their biological value, is the use of biofertilizers based on plant growth promoting (PGP) bacteria [2, 4, 5, 15]. The PGP bacteria can fix atmospheric nitrogen, solubilize inaccessible compounds of phosphorus, potassium, iron, zinc, etc. inaccessible to plants, inhibit the development of phytopathogens (fungi and bacteria), produce biologically active substances, including phytohormones, and also take an active part in the binding of HMs, enhancing the remediation capacity of plants [1, 16, 17]. Meanwhile, their utilization carries no risks of environmental pollution [5, 18]. Various liquid and solid substances can be used as a carrier material for loading bacteria in biofertilizers, however, biochar obtained by oxygen-free pyrolysis from various organic wastes is gaining significant attention [19, 20]. It is evidenced that the addition of biochar into the soil leads to an improvement in its physical structure and chemical characteristics, fosters the development of soil microflora, and mitigates the toxic effect of organic and inorganic pollutants due to its binding properties [21, 22]. Moreover, due to its high water-holding capacity, optimal pH and large surface area, biochar can effectively be used as a carrier material for immobilizing rhizobacteria for the preparation of environmentally safe biofertilizers [17, 23, 24]. Thus, it was hypothesized that the application of composite biofertilizer consisting of metal-tolerant PGP-rhizobacteria (PGPR) and wood biochar in the cultivation of agricultural plants not only enhances nutrient enrichment but also alleviates the detrimental effects of excessive copper accumulation.
The objective of the study was to investigate the morphological and physiological traits of rapeseed (Brassica napus L.) and assess its biofortification through bioaugmentation with a biofertilizer composed of the metal-tolerant PGPR strain Buttiauxella sp. EA20 and birch wood biochar in the presence of copper.
Bacterial isolation was carried out in the summer of 2020 from the rhizosphere of Epipactis atrorubens (Hoffm). Besser, belongs to the Orchidaceae family, and thrives in a natural forest community on serpentine soil. Serpentine substrates are known for their unfavorable physico-chemical properties for plant growth such as stoniness, poor water-holding capacity, high insolation, significantly high pH value, and large temperature differences. In addition, they differ by a large amount of certain HMs and low concentration of macronutrients such as nitrogen and phosphorus and high Mg/Ca ratio [25, 26, 27].
The rhizosphere soil was collected in a
UV-sterilized zip-lock bag and brought to the
laboratory. Ten grams of soil sample
(on a dry weight basis) were mixed with 90 mL of
sterilized distilled water and
homogenized by shaking at 180 rpm for 1 hour in an Erlenmeyer
conical flask. After serial dilution in sterilized
distilled water, a 100 µL aliquot of required suspension was plated on
Luria–Bertani (LB) agar (HiMedia,
Mumbai, Maharashtra, India) plate and incubated for 3
days at 28
To determine the minimum inhibitory concentration (MIC), the isolates were cultured on LB agar plates for 5 days at 28 °C, with progressively rising metal concentrations (Cu: 100 to 600; Zn: 100 to 1000; Cd: 100 to 1000; and Ni: 50 to 400 mg/L) for the analysis [17]. Ethylene diaminetetraacetic acid (EDTA) (Sigma-Aldrich, Taufkirchen, Germany) was added to the medium to prevent metal precipitation. Control plates were prepared without the addition of any metals or EDTA. Deionized Millipore water (Milli-Q system, Millipore, Molsheim, France) was used for the preparation of metal stock solutions.
The PGP-activities of the isolated bacterial strains were
evaluated based on their capacity to fix molecular nitrogen, synthesize
indole-3-acetic acid (IAA), solubilize phosphates and
produce siderophores. The N2-fixing activity of the
isolates was determined on solid Ashby medium
containing per liter: 0.2 g KH2PO4, 0.2 g NaCl, 0.1 g K2SO4,
0.2 g MgSO4, 5 g CaCO3, 20 g mannitol, 15 g agar (HiMedia, Mumbai,
Maharashtra, India) with pH adjusted to 7.2
For phosphate solubilization, 100 µL of freshly prepared bacterial
cultures (with optical density adjusted to 108 cfu/mL) were
inoculated into the National Botanical
Research Institute’s phosphate
(NBRIP) liquid growth medium [29]. The
medium consisted of the following per liter: 10 g glucose, 5.0 g
MgCl2
For the analysis of siderophore production, Fe-Chrome Azurol Sulfonate (CAS) (Sigma-Aldrich, Taufkirchen, Germany) indicator solution was mixed with principle buffer in a ratio of 1:8, and then with 70 mL of auxiliary buffer and a solution of casamino acid with agar, which was spot inoculated with 5 µL of freshly grown (108 cfu/mL) bacterial samples. The plates were incubated at 28 °C for 5 days and bacteria producing orange-yellow halo zones were measured and identified as siderophore-producing rhizobacteria [31].
Out of 30 morphologically different pure bacterial colonies, the strain that showed maximum tolerance to multiple HMs along with high numbers of analyzed PGP attributes was selected and identified based on the 16S rRNA gene sequencing.
Total DNA from the selected strain was isolated using a diaGene kit (3318.0250, Diaem LLC, Moscow, Russia) for extracting bacterial genomic DNA on spin columns according to the manufacturer’s instructions. A total of 10 ng of DNA was used as a template for amplifying the 16S rRNA genes. Polymerase chain reaction (PCR) amplification was done by 16S Barcoding Kit (SQK-16S024, Oxford Nanopore Technologies, Oxford, UK) and LongAmp Hot Start Taq 2X Master Mix (NEB, Inc, Ipswich, MA, USA), following the manufacturer’s protocol. PCR amplification conditions were: initial denaturation (95 °C for 60 sec); 25 cycles: denaturation (95 °C for 20 sec), annealing of primers (55 °C for 30 sec) and, chain synthesis (65 °C for 120 sec); final chain elongation (65 °C for 300 sec). The prepared DNA library (11 µL) was combined with 34 µL of sequencing buffer with 25.5 µL of loading beads, and 4.5 µL of deionized Millipore water. This mixture was then loaded into an R9.4 flow cell (FLO-MIN106D; Oxford Nanopore Technologies, Oxford, UK) and sequenced on a GridION Mk1 (Oxford Nanopore Technologies, Oxford, UK). Data collection was performed using the software MINKNOW version 21.05.8 (Oxford Nanopore Technologies, Oxford, UK).
The primary data of the GridION sequences (i.e., data in the FAST5 format) were
converted to FASTQ format files using the Guppy ver. software 5.0.11 (Oxford
Nanopore Technologies, Oxford, UK) with an average reading quality score of
The Neighbor-Joining Method was used for construction of phylogenetic tree [32] and implemented using Mega11 software [33], the evolutionary distances were generated using the Tamura-Nei method [34] and Enterobacter cloacae was used as the outgroup (Fig. 1). The associated taxa were clustered together in the bootstrap test (1000 replicates) [35].
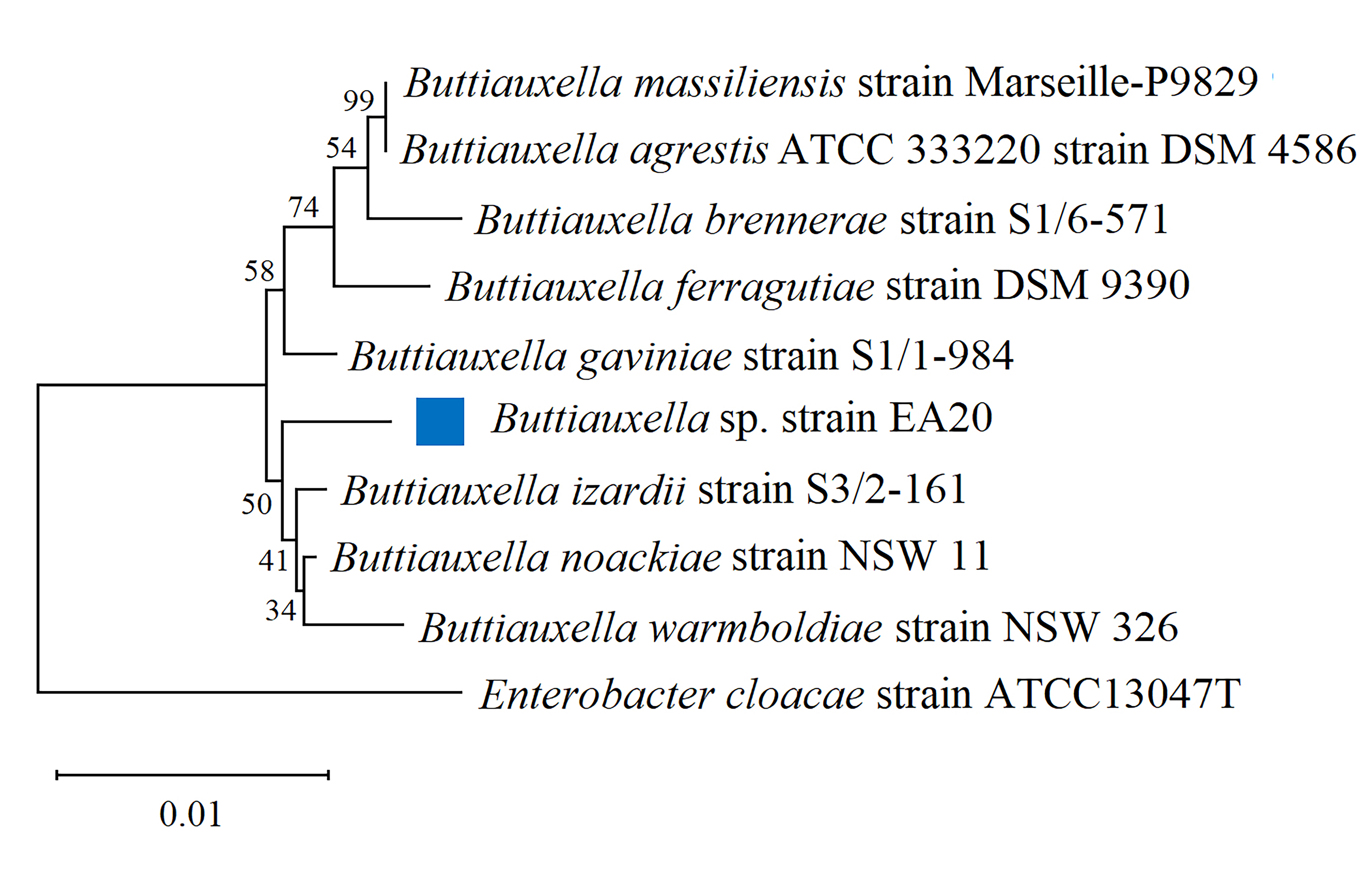 Fig. 1.
Fig. 1.
Phylogenetic tree showing the 16S rRNA sequence of Buttiauxella sp. strain EA20 and closely related sequences of all the strains of the genus Buttiauxella. Enterobacter cloacae is used as the outgroup. The bootstrap value (1000 replicates were used in the test) is shown as a percentage below the branches.
Biochar (BC), produced by a domestic manufacturer from high-density birch wood
by oxygen-free pyrolysis, was crushed and passed through a 0.2 mm sieve, followed
by drying for 2 days at a temperature of 70 °C and double sterilized at
a temperature of 110 °C for 20 min in an autoclave. An assessment of its
physicochemical parameters showed circumneutral pH (6.4
Metal-tolerant PGPR strain Buttiauxella sp. EA20 was used to prepare the biofertilizer (BF). Strain EA20 was grown overnight at 28 °C in 250 mL LB broth at 150 rpm in an orbital shaker incubator (Biosan ES-20/60, Latvia). The bacterial culture (108 cfu/mL), prepared as previously described [17], was mixed with double sterilzed biochar (130 °C for 1 hour) and left overnight in a laminar flow chamber until the level of moisture reaches between 25–35%. The BF was stored in perforated disinfected bags at 27 °C for its use in pot-scale experiment [17].
In April and June of 2021, two separate pot-scale experiments were conducted with spring rapeseed plants, and the averaged outcomes are reported in this study. The B. napus seeds underwent surface sterilization by exposure to 70% ethanol for 30 seconds, followed by a 2-minute wash with 4% sodium hypochlorite, and ultimately, thorough rinsing with sterile double-distilled Millipore water.
The experiment comprises two distinct lines: one without copper (0Cu) and another involving the introduction of 200 mg of Cu (in sulfate form) per kilogram of soil (200Cu) on the 21st day of the plant growth period. Six treatment conditions were applied, namely: peat-containing control substrate (CS), CS + 5% BC (v/v); CS + 5% BF (v/v); CS + 200Cu (200 mg Cu kg-1 soil); CS + 200Cu + 5% BC (v/v); CS + 200Cu + 5% BF (v/v).
Double sterilized (130 °C for 20 min) peat-containing substrate with pH
6.7
Pre-dried and homogenized samples, weighing 0.2 g underwent wet digestion with
70% nitric acid (AR, analytical grade)
in a MaRS 5 Digestion
Microwave System (CEM, Matthews, NC, USA). The
atomic absorption spectrometer (AAS)
AA240FS (Varian Australia Pvt Ltd., Mulgrave, Victoria, Australia) was employed
to determine the total Cu and potassium (K)
content in both
aboveground and underground rapeseed
biomasses. JSC Ural Plant of Chemical Reagents (Verkhnyaya Pyshma, Russia)
was used as Standard Reference Materials; GSS 7998-93 for Cu(II) and GSS 8092-94 for K(I) were utilized for the preparation, calibration and analysis of each analytical batch. Calibration
coefficients were maintained at
The biometric parameters of plants such as shoot and root fresh and dry biomass, length, and leaf area were studied [17]. The water content in the organs was calculated by weighing the samples before and after drying at 75 °C for 24 hours. The germination rate of seedlings was also computed following the method reported by Wijewardana et al. [36].
The CO2 assimilation (µmol CO2/m2 sec) on the
4th leaf (excluding cotyledons) of rapeseed plant was measured in a
LeD Light Source chamber (3
Malondialdehyde (MDA), a marker of lipid peroxidation, was quantified in 0.3 g
of fresh rapeseed leaves. The leaves were homogenized with 4 mL of a reaction
medium comprising 0.25% thiobarbituric acid in 10% trichloroacetic acid
(v/v) as per Heath and Packer [39]. After boiling the extract for 30
min, cooling it on ice, and centrifuging it at 12,000
The total content of soluble phenolic compounds and flavonoids was determined in an ethanol extract from freshly crushed rapeseed leaves (0.3 g) pre-filled with 80% ethanol for 24 h. The resulting extract was filtered through the suitable size filter paper and analyzed. The total phenolic content was determined by spectrophotometer (Infinite 200 PRO, Tecan Austria GmbH, Grödig, Austria) at 760 nm after incubating the reaction mixture for 1 h (0.1 mL of extract interacting with 0.5 mL of 0.2 M Folin–Ciocalteu reagent for 5 min, followed by 0.4 mL 7.5% Na2CO3 (w/v) solution) at room temperature [40]. Gallic acid sourced from Sigma-Aldrich Chemie GmbH in Germany served as the reference standard in the analysis, with the outcomes reported as milligrams of gallic acid/g DW.
The content of flavonoids was measured by reacting the extracted sample with an equivalent amount of 10% aluminum chloride ethanolic solution followed by a 15 min incubation period at room temperature [38]. The absorbance of the reaction mixture was measured by spectrophometer (Infinite 200 PRO, Tecan Austria GmbH, Grödig, Austria) at 415 nm. The 10% aluminum chloride in the blank was replaced with an equal volume of 80% ethanol. Likewise, a standard solution of rutin from Sigma-Aldrich Chemie GmbH (Taufkirchen, Germany) was treated with aluminum chloride to establish a calibration curve, and the findings were presented as milligrams of rutin/g DW.
The data presented in the Table 1 represent the mean values (Means) of 3
replicates for measurements of PGP-attributes (IAA, siderophore production and
phosphate solubilisation). Fig. 1 shows the bootstrap value from 1000 replicates. Fig. 2A demonstrates the appearance of 36-day-old rapeseed plants grown under various treatments. Fig. 2B and Table 2 represent the mean values (Means) from 5 replicates, composed of composite samples from each pot. Fig. 3 represent the Means obtained from 20 replicates, each consisting of four randomly selected plants from every pot, for biometric characteristics. Figs. 4,5,6 represent the Means of 5 composite replicates.
Standard errors (SE) are included. Statistical
analysis was conducted using STATISTICA 13.0 (StatSoft, Palo Alto, CA, USA) and
Excel 16.0 software (Microsoft Office 2016, Microsoft, Redmond, WA, USA),
following verification of normality using the Shapiro–Wilk test and homogeneity
of variance using Levene’s test. Differences between treatment groups were
assessed using the non-parametric Kruskal–Wallis H-test at a
significance level of p
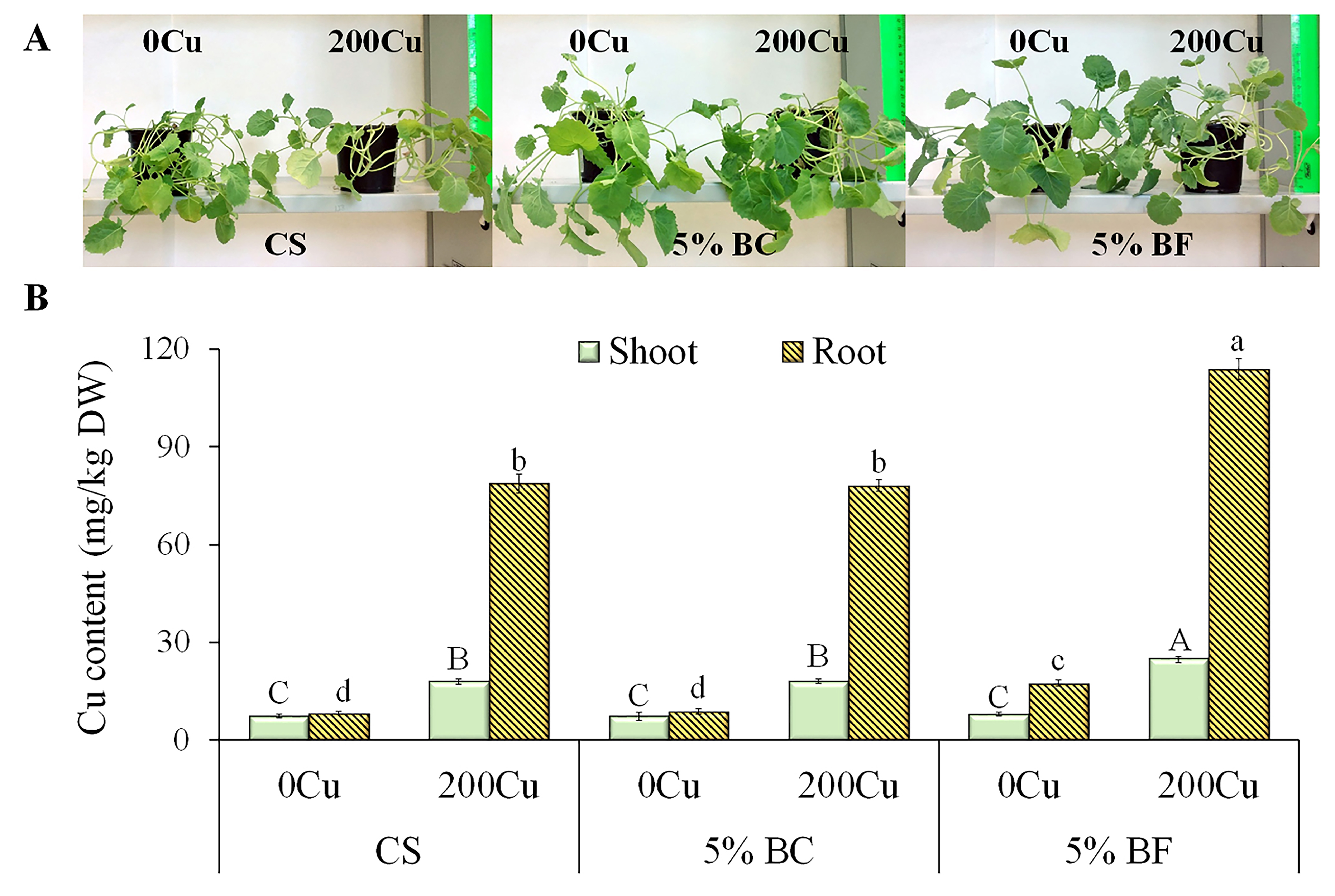 Fig. 2.
Fig. 2.
Effect of 5% biochar (BC) and biofertilizer (BF)
based on PGPR Buttiauxella sp. strain EA20 and BC on plant appearance
and copper accumulation in
shoot and root of
B. napus after 36 days of vegetation without Cu
(0Cu) and with the addition of 200 mg Cu/kg of soil
(200Cu). (A) Plant appearance. (B) Copper content. CS, control
substrate. Data are presented as Means
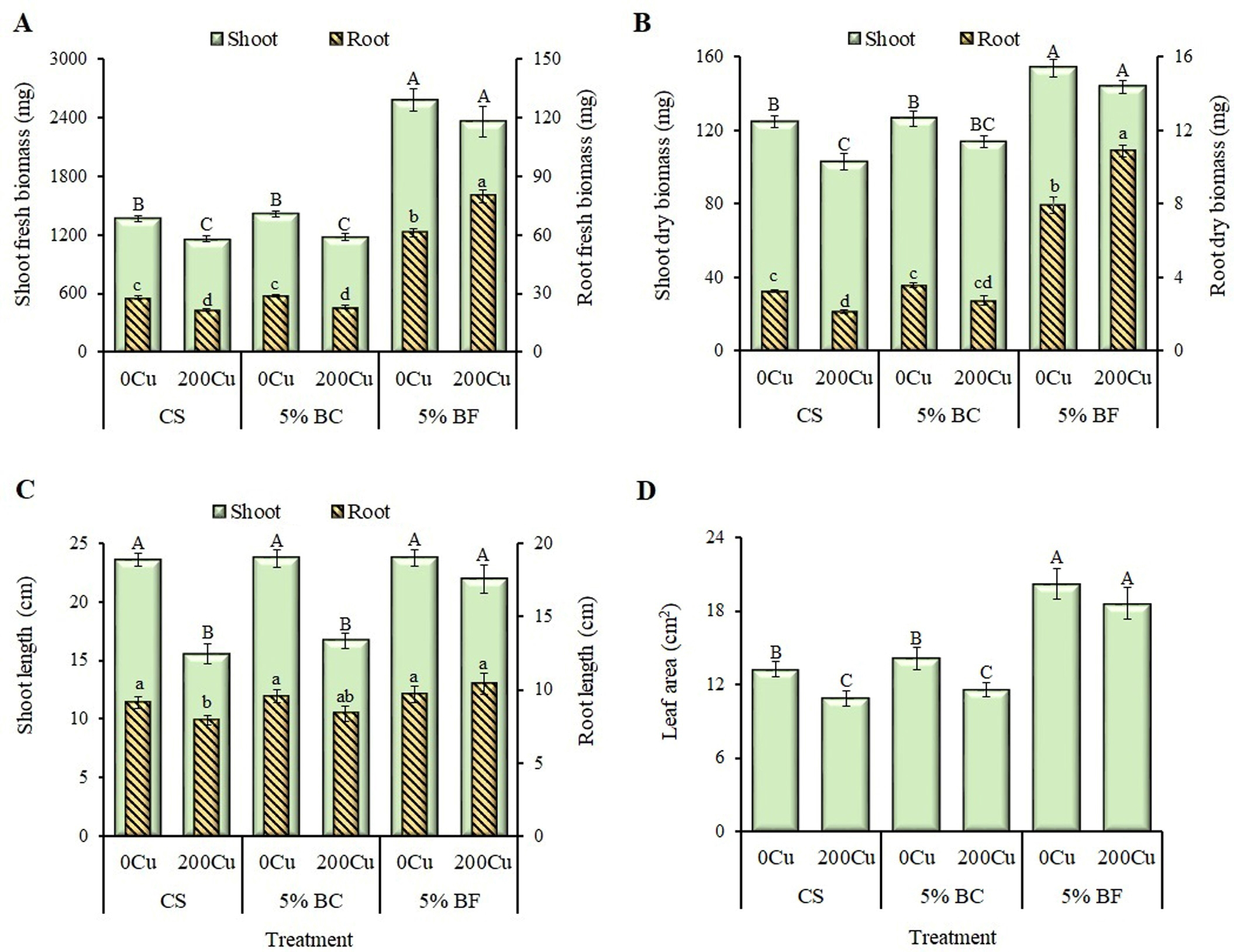 Fig. 3.
Fig. 3.
Effect of 5% biochar (BC) and biofertilizer (BF) based on PGPR
Buttiauxella sp. strain EA20 and BC on biometric growth parameters of
B. napus after 36 days of vegetation without Cu
(0Cu) and with the addition of 200 mg Cu/kg of soil
(200Cu). (A) Shoot and root fresh biomass. (B) Shoot and root dry
biomass. (C) Shoot and root length. (D) Leaf area. CS, control substrate. Data
are presented as Means
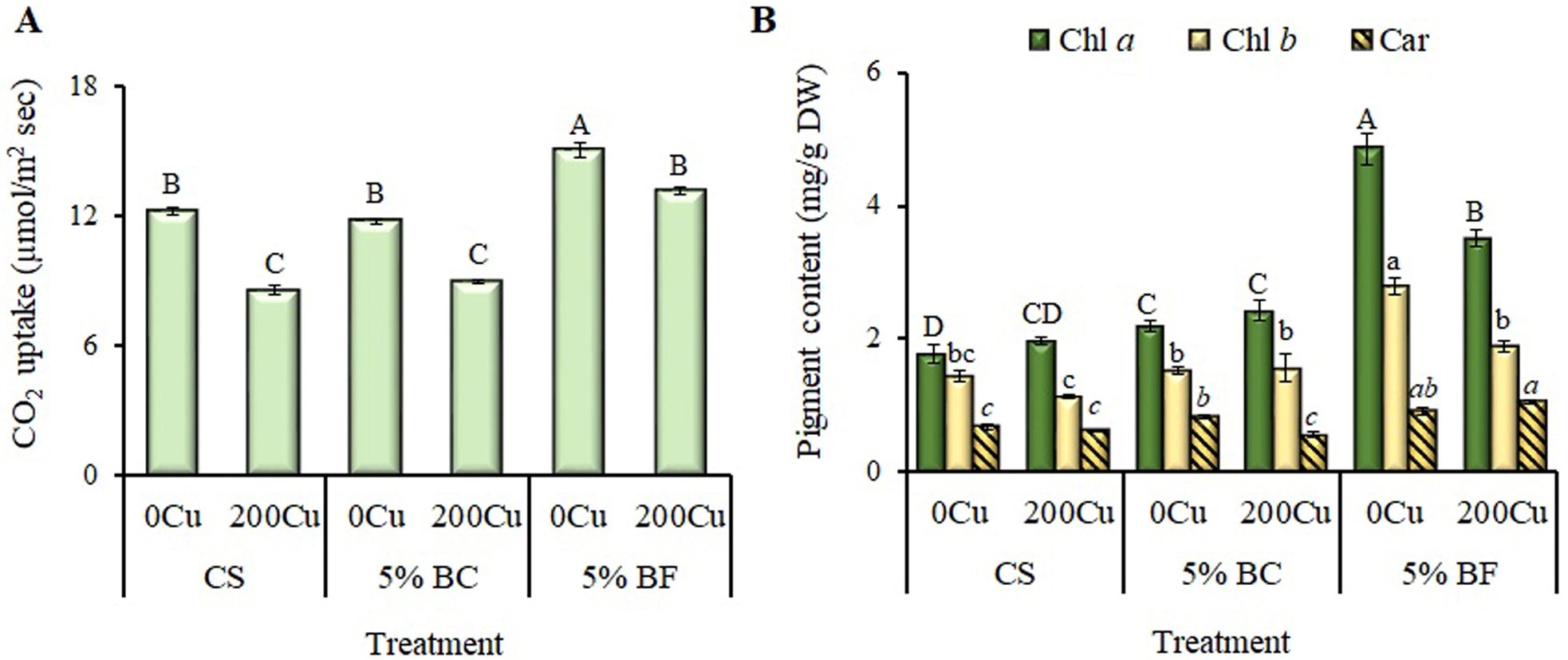 Fig. 4.
Fig. 4.
Effect of 5% biochar (BC) and biofertilizer (BF)
based on PGPR Buttiauxella sp. strain EA20 and BC on photosynthetic
characteristics of B. napus after 36 days of vegetation without
Cu (0Cu) and with the addition of 200 mg
Cu/kg of soil (200Cu). (A) Rate of CO2 uptake.
(B) Photosynthetic pigment content. CS, control substrate; Chl a,
chlorophyll a; Chl b, chlorophyll b; Car, carotenoids.
Data are presented as Means
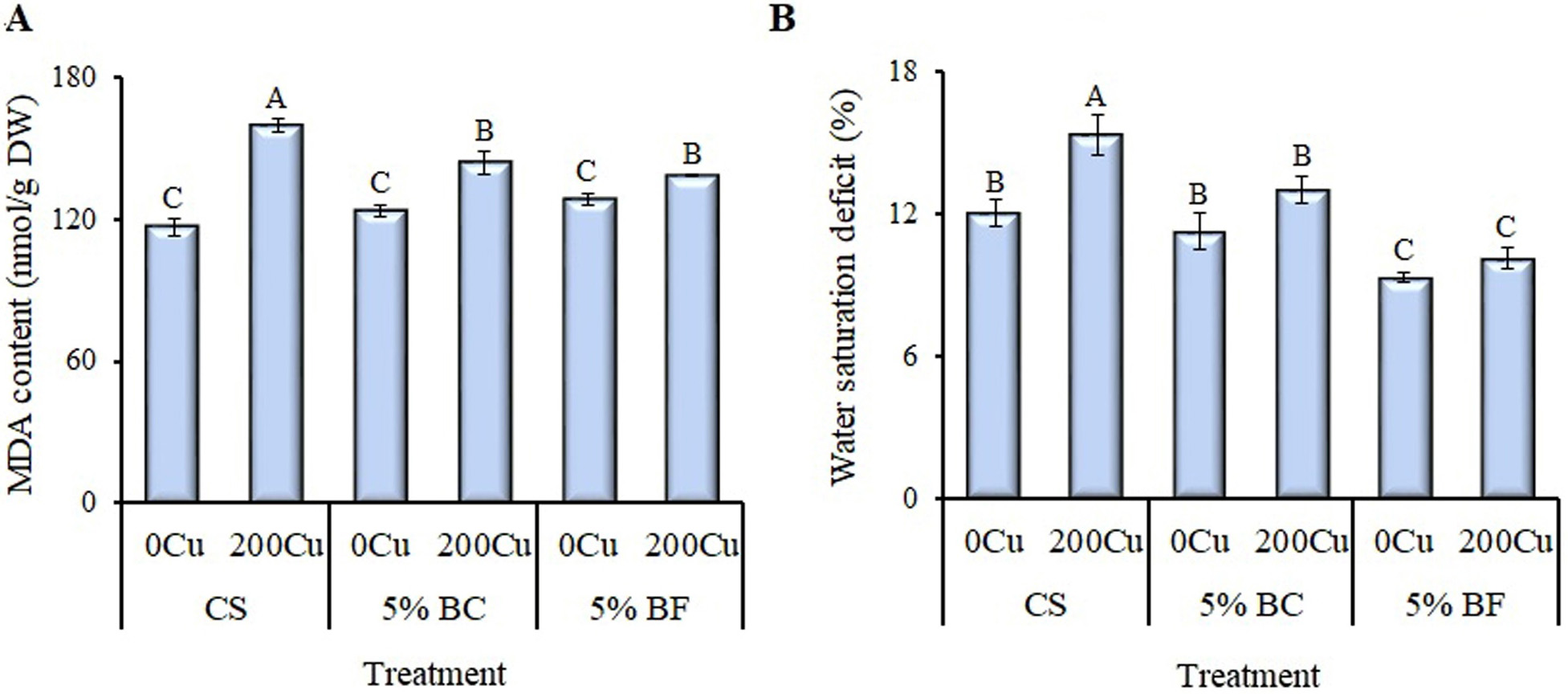 Fig. 5.
Fig. 5.
Effect of 5% biochar (BC) and biofertilizer (BF)
based on PGPR Buttiauxella sp. strain EA20 and BC on the content of
malondialdehyde (MDA) and water
saturation deficit in the leaves of B. napus after 36 days of vegetation
without Cu (0Cu) and with the addition of 200 mg
Cu/kg of soil (200Cu). (A) MDA content. (B) Water
saturation deficit. CS, control substrate. Data are presented as Means
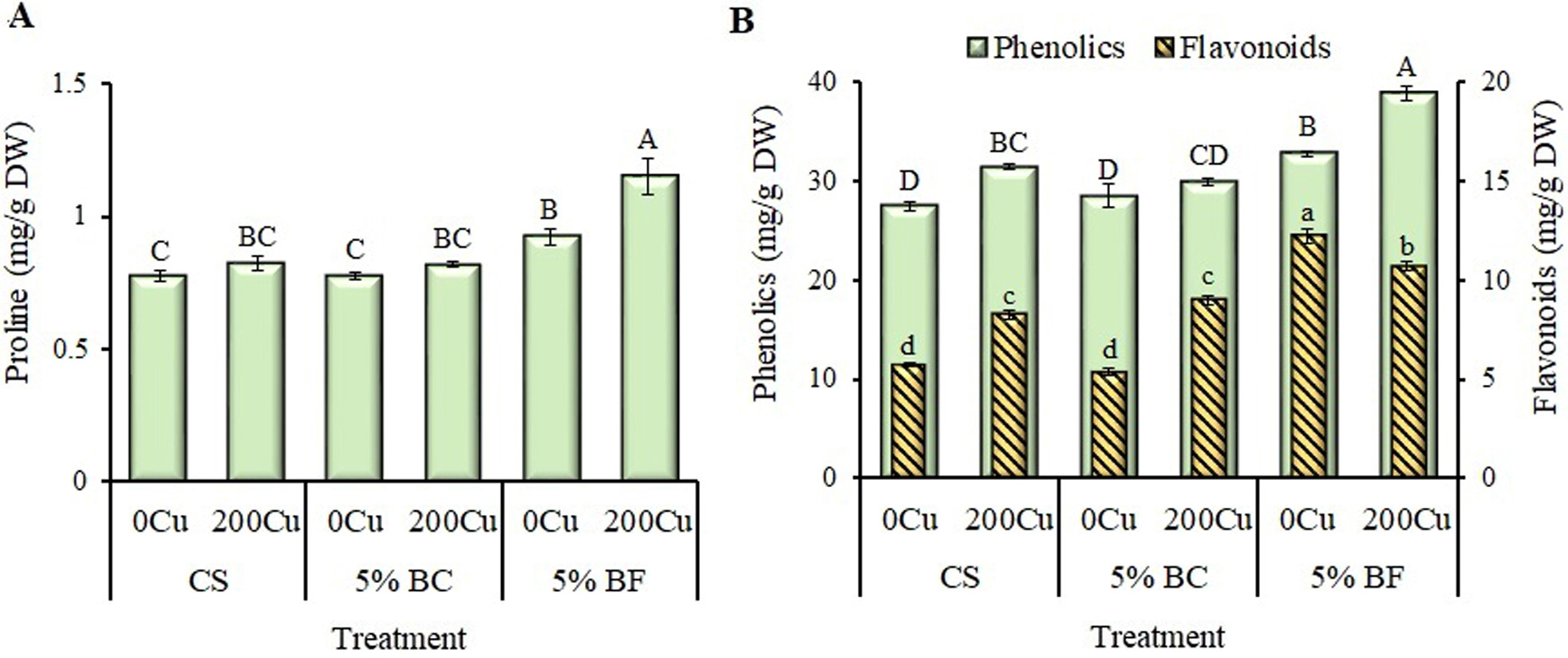 Fig. 6.
Fig. 6.
Effect of 5% biochar (BC) and biofertilizer (BF) based on PGPR
Buttiauxella sp. strain EA20 and BC on the content of low molecular
weight antioxidants in the leaves of B. napus after 36 days of
vegetation without Cu (0Cu) and with the addition of
200 mg Cu/kg of soil (200Cu). (A) Free proline. (B)
Total soluble phenolic compounds and flavonoids. CS, control substrate. Data are
presented as Means
| Bacterial strain | MIC | PGP attributes | ||||||
| Cu (mg/L) | Ni (mg/L) | Zn (mg/L) | Cd (mg/L) | N2-fixation | 1IAA production (mg/L) | 1Phosphate solubilization (mg PO43–/L) | 1Siderophore production | |
| Buttiauxella sp. strain EA20 | 600 | 400 | 1000 | 1000 | + | 12.7 |
1110.7 |
2.73 |
1Data are presented as Means
| Organ | Nutrients (mg/g DW) | Treatments without Cu | Treatments with 200Cu | ||||
| CS | 5% BC | 5% BF | CS | 5% BC | 5% BF | ||
| Shoot | N | 32.28 |
36.98 |
44.85 |
29.87 |
34.71 |
49.22 |
| P | 1.98 |
2.13 |
2.57 |
1.67 |
2.09 |
2.89 | |
| K | 29.25 |
33.45 |
42.64 |
27.67 |
32.89 |
38.10 | |
| Root | N | 26.12 |
26.37 |
29.78 |
22.76 |
26.21 |
33.15 |
| P | 1.21 |
1.35 |
2.18 |
1.02 |
1.30 |
2.04 | |
| K | 33.87 |
35.40 |
45.16 |
31.69 |
34.65 |
44.68 | |
Data are presented as
Means
From the 30 strains isolated from the rhizosphere of the orchid E. atrorubens, one particular isolate i.e., strain EA20, exhibited the highest resistance to metals such as Cu, Ni, Zn, and Cd. EA20 also demonstrated exceptionally positive results across all tested plant growth-promoting attributes, including N2-fixation, IAA production, phosphate solubilization, and siderophore production. The preliminary identification was performed by assessing its morphological characteristics. The isolate was identified as Gram-negative and aerobic and the pure colony exhibited a smooth, crateriform elevation, light beige color, and a round shape with a rolled appearance around the edge.
The full-length sequence of the 16S rRNA gene was used for genus identification. Utilizing the cloud-based platform EPI2ME Agent, the strain’s genus was determined to be Buttiauxella with an average accuracy of 91.8%. Additionally, a phylogenetic investigation revealed that strain EA20 was situated in a shared cluster with strains from the Buttiauxella genus, including B. izardii, B. noackiae, and B. warmboldiae. This supports the conclusion that strain EA20 certainly falls within the Buttiauxella genus (Fig. 1).
The bacterial strain Buttiauxella sp. EA20 showed resistance against
various metals. The strain EA20 exhibited maximum tolerance for Zn = Cd
The application of BC or BF showed no significant effect on the germination of B. napus seeds, which averaged 76%. Fig. 2A shows the appearance of B. napus plants grown under different treatments at the end of the experiment. It can be visually seen that the appearance of rapeseed shoots was much better in the variants with the addition of 5% BC or BF, regardless of copper treatment, compared to the control substrate (CS). The addition of 200 mg/kg of Cu into CS resulted in a 2.4-fold increase in B. napus shoot content and a 9.4-fold increase in the root, corresponding to 18 and 79 mg/kg DW, respectively (Fig. 2B).
A similar trend was observed in the plants treated with 5% BC. The application of BF without Cu did not affect its content in the shoot, rather a 2-fold enhanced accumulation was observed in the root (compared to CS and 5% BC). When copper was applied (BF + Cu), it mainly accumulated in rapeseed root (4.6 times higher than in shoot). The addition of BF also contributed to an increase in the accumulation of Cu in shoot and root by 3.3 and 13.3 times, respectively, as compared to CS or BC without Cu, and by 1.4 and 1.5 times as compared to CS or BC with 200Cu addition (Fig. 2B).
The use of BC without copper did not significantly affect the phosphorus content, however slightly increased the nitrogen and potassium content in the shoot (15%), while there were no significant differences observed for NPK concentration between CS and BC in root (Table 2). Simultaneously, plants treated with BF demonstrated 1.4-fold higher NPK content in their shoot and root as compared to CS. The combined application of 200Cu and BC increased NPK content in the shoot by 20% compared with single copper. Wherein, the nitrogen content in the root increased by 15%, but the phosphorus and potassium content remained unchanged. The application of Cu and BF resulted in a significant increase of 1.5 times on average in NPK content in both aboveground and underground organs (Table 2).
Both in control and BC treated plants, the addition of Cu showed a negative effect on the growth parameters of rapeseed (Fig. 3). An average decrease of 18% was observed for fresh and dry biomass of shoot while root biomass was reduced by 35% as compared to CS and CS + 5% BC treatments (Fig. 3A,B). Moreover, a significant decrease of 28% (on average) in the shoot and root length as well as leaf area was also observed (Fig. 3C,D). In the absence of Cu, BF enhanced the aboveground and underground fresh biomass by an average of 1.9 times as compared to control and only BC treated plants (Fig. 3A,B). Simultaneously, shoot and root length did not change (Fig. 3C), while the leaf surface area increased significantly (by 47%) compared to CS and BC (Fig. 3D). When Cu was applied together with BF, the fresh shoot biomass was increased almost by 2-fold (Fig. 3A), while the dry shoot biomass upsurge only by 30% (Fig. 3B), compared to CS and CS + Cu. At the same time, an almost quadruple increase in underground biomass for both fresh and dry was observed (Fig. 3A,B). The addition of BF along with Cu did not have a significant effect on the shoot and root length compared to the CS, however, it was 1.4 times higher than CS + Cu (Fig. 3C). Simultaneously, the leaf area increased by 1.4 and 1.7 times compared to CS and CS + Cu, respectively (Fig. 3D).
The rate of CO2 assimilation in the control plants decreased by 40% in the presence of copper (Fig. 4A), while the content of photosynthetic pigments remained more stable (Fig. 4B). Similar trend was observed for 5% BC treated plants. The addition of BF (both with and without copper) had a positive effect on photosynthetic activity and pigment content in rapeseed leaves which increased them by 1.4- and 1.8-fold, respectively (Fig. 4).
The application of copper contributed to a significant (37%) increase in the MDA content of control rapeseed plants (Fig. 5A). In BC + Cu and BF + Cu treated plants, the MDA content increased to a lesser extent (by 17% from CS on average). In the presence of copper, a significant increase of 25% in water saturation deficit was observed for control plants (Fig. 5B). BC and especially BF caused a significant decrease in water deficit after its application.
The content of free proline in rapeseed leaves significantly increased (by 30% on average) when BF was applied, especially in the presence of copper (Fig. 6A). The Cu addition led to a significant increase in the content of soluble phenolics in the CS-plants, and especially (almost 1.5 times) flavonoids (Fig. 6B). The application of BF also activated the synthesis of soluble phenolics, while the proportion of flavonoids increased by almost 2-fold compared to the control. The copper in the presence of BF stimulated the synthesis of phenolics even more, however the proportion of flavonoids decreased.
The present research work aimed to assess the morphophysiological traits of B. napus at individual and combined application of copper and BF based on the metal-tolerant PGP-rhizobacteria Buttiauxella sp. strain EA20 and wood biochar. Utilizing BF based on PGPR represents a promising avenue in agricultural biotechnologies, particularly in techniques focused on enhancing crops with essential micro- and macronutrients [41, 42]. Rapeseed is one of the important agricultural plants and the largest source of edible oil in the world [43, 44]. In the present study, the copper treatment (200 mg/kg) caused a decrease in leaf area, shoot and root length as well as fresh and dry biomass of B. napus. Due to excess Cu, similar deleterious changes in the growth parameters of rapeseed and other crops of Brassicaceae were also reported earlier [43, 45]. In contrast, Kumar et al. [17] also reported the positive effect of copper (100 mg/kg) which stimulated rapeseed root growth and caused a significant increase in their biomass.
Increasing the amount of MDA in B. napus leaves with the addition of 200Cu indicated a violation in the integrity of cell membranes. This was also confirmed by a significant increase in water saturation deficit. A water deficiency in metal-stressed plants can be explained by reduced water uptake. These changes in copper-stressed plants are consistent with the results of other authors who investigated the impact of abiotic stress caused by increased Cu concentration [9, 46, 47, 48]. An excess of HMs causes osmotic disturbances in plants due to the negative impact on the development of the absorbent surface of the roots. Moreover, metals can inhibit water transport both in the symplast and in the apoplast, which negatively affects the water supply of the shoot. Water deficiency often occurs due to a decrease in the size of xylem cells, as well as due to the inhibition of transpiration, because of the reduction of leaf area and decreasing stomatal density under metal stress [49, 50]. A decrease in the rate of CO2 assimilation in copper-stressed plants was also noted earlier. Excess copper is known to disrupt the photosynthetic apparatus by affecting pigment complexes and chloroplast structure. This disruption leads to a decrease in the net photosynthetic rate, limits the activity of Rubisco, and inhibits the photosynthetic electron transport chain [8, 11, 12].
Application of BF increased shoot length, leaf surface, and biomass of B. napus indicating its positive effect on the growth and development of plants. Since, the use of only BC did not have a significant influence on growth parameters, it can be assumed that namely, rhizobacteria Buttiauxellasp. strain EA20 had this positive action due to its growth promoting attributes like N2-fixation, IAA synthesis, phosphate solubilization, and siderophore production. To date, information on endophytic and rhizospheric bacteria of the genus Buttiauxella is limited. Previously, the endophytic bacteria Buttiauxella agrestis was found in the orchid Epipactis helleborine [51]. Ruangsanka [52] noted that bacteria of the genus Buttiauxella increase the availability of phosphates for plants. It has been also reported that the strain Buttiauxella sp. SaSR13 is cadmium resistant and capable of producing IAA, siderophores, and inhibiting ethylene production (due to 1-aminocyclopropane-1-carboxylic acid deaminase, ACCD). Inoculating Sedum alfredii plants with strain SaSR13 increased biomass when treated with Cd, enhancing the phytoextraction of cadmium [53].
When copper was added together with BC and BF, the level of MDA decreased, indicating the prevention of oxidative stress. The application of BC alone and especially with PGPR mitigated the negative effect of copper on water status of plants. This may be due to the ability of biochar to retain moisture in its porous structure [22, 23]. The use of BF significantly increased the rate of CO2 assimilation and the content of photosynthetic pigments in B. napus. It also mitigated the toxic effects of copper on these important physiological characteristics. The positive effect of biofertilizers based on different species of PGP-bacteria on the photosynthetic apparatus of plants was also previously reported by other researchers [47, 54]. Moreover, it was observed that the PGP-bacteria help to protect plants from the HMs toxicity [17, 46, 54].
One of the non-specific protective reactions of plants to abiotic stress is the increased synthesis of low molecular weight antioxidants [10, 55]. Combined application of BF and 200Cu alleviated the Cu impact by synthesizing non-enzymatic antioxidants (carotenoids, free proline and soluble phenolic compounds including flavonoids). To date the information concerning the influence of associative PGP-bacteria on antioxidant status of stressed plants is limited. In what way diverse bacteria species activate plant tolerance strategies is yet not properly explicated. However, various studies revealed stimulating effects of bacteria on plant antioxidant systems under abiotic stress. Chen et al. [46] found that rhizobium inoculation in Medicago sativa enhanced copper tolerance by influencing copper uptake, regulating antioxidant enzyme activities, and modulating the ascorbate-glutathione cycle. Additionally, they observed an impact on the expression of genes related to phytochelatin biosynthesis in M. sativa seedlings. Application of BF based on Bacillus altitudinis strain TF16a and BC increased the soluble phenolics content in B. napus plants and mitigated copper stress [17]. Bacterial inoculation with Paenibacillus polymyxa and Bacillus circulans also caused mitigation of Cu-stress in maize due to increase of antioxidant enzyme activity [47]. Ansary et al. [56] reported that different strains of Pseudomonas sp. enhanced the free proline content in maize under water deficit stress.
It was noted that BF based on PGPR Buttiauxella sp. strain EA20 and wood biochar increased Cu accumulation, especially in rapeseed root, compared to single metal application. A similar trend was noted in prior investigations when examining the influence of the copper-tolerant endophyte Pseudomonas lurida strain EOO26 on the growth of Helianthus annuus and its accumulation of Cu under copper exposure [11]. Nevertheless, in the present study, Cu concentration in aboveground rapeseed biomass (about 25 mg/kg DW) remained within the physiological norms [57]. It is established that copper ranks among the seven vital mineral elements in food crops essential for the human diet [13, 14]. Therefore, the use of biofertilizer based on PGPR Buttiauxella sp. strain EA20 and biochar can compensate for the deficiency of this essential micronutrient. Furthermore, the application of BF, both with and without copper, led to a significant increase in the concentration of crucial macronutrients such as nitrogen, phosphorus and potassium in rapeseed biomass.
Altogether, our research leads us to conclude that combined application of BF based on metal-tolerant PGPR Buttiauxella sp. strain EA20 and biochar with copper is an effective strategy for enhancing the growth as well as increasing the content of Cu and NPK in rapeseed plants. The use of biofertilizer also contributed to the biofortification of rapeseed with important antioxidants such as carotenoids, free proline and soluble phenolic compounds, including flavonoids, which is of particular importance in the production of functional foods.
The positive effect of biofertilizer (BF) based on metal tolerant PGPR Buttiauxella sp. strain EA20 and wood biochar (BC) for Brassica napus growth and biofortification at copper action was reported for the first time. Single copper treatment (200 mg/kg) caused a decrease in rapeseed leaf area, shoot and root length, fresh and dry biomass, activated lipid peroxidation, and increased water saturation deficit. The application of BF as well as single BC mitigated these harmful effects of copper. Biofertilizer, regardless of Cu addition, augmented B. napus biomass with a simultaneous increase in the leaf area and photosynthetic pigment content, especially chlorophylls. Moreover, plant biofortification with copper and essential macronutrients such as nitrogen, phosphorus and potassium was observed. In addition, BF treatment enhanced the antioxidant activity of rapeseed due to the accumulation of such non-enzymatic antioxidants as carotenoids, free proline and soluble phenolics, including flavonoids, which can probably increase plant resistance and their biological value. Future perspective includes evaluation of the potential for using the resulting biofertilizer to improve the growth and biofortification of other crops.
All data points generated or analyzed in this study are presented in this article. Further information can be obtained by contacting the corresponding author upon request.
GB, MM designed the research study, analyzed the data, wrote the original draft. GB, MM, NC performed plant pot-scale researches. T, OV, AK performed microbiological researches. AT provided molecular genetic identification of the selected bacterial isolate and construction of a phylogenetic tree. MM, T were involved in data curation; provided project administration and funding acquisition. All authors have sufficiently contributed to the work to accept public responsibility for appropriate sections of the content and have agreed to be accountable for all aspects of the work in ensuring the accuracy and integrity of the content. All authors have reviewed and approved the final manuscript. All authors contributed to editorial revisions in the manuscript.
Not applicable.
The authors express their gratitude to Sinenko O.S., leading engineer of the Department of experimental Biology and Biotechnology, Ural Federal University (Ekaterinburg, Russia) for measuring of CO2 assimilation, and Sobenin A.V., scientific researcher at the Institute of Mining of the Ural Branch of RaS (Ekaterinburg, Russia) for the analysis on copper content. The authors also thank two anonymous reviewers for their valuable recommendations and comments that helped improve the article.
The work was supported and funded by Russian Science Foundation, Project No. 23-26-00292, https://rscf.ru/project/23-26-00292/.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






