1 Environmental Process Development, Catholic University of Pernambuco, 50050-900 Recife, Pernambuco, Brazil
2 Northeast Biotechnology Network (Renorbio), Federal Rural University of Pernambuco, 52171-900 Recife, Pernambuco, Brazil
3 Advanced Institute of Technology and Innovation (IATI), 50751-310 Recife, Pernambuco, Brazil
4 School of Health and Life Sciences, Catholic University of Pernambuco, 50050-900 Recife, Pernambuco, Brazil
5 Icam Tech School, Catholic University of Pernambuco, 50050-900 Recife, Pernambuco, Brazil
Abstract
This study aimed to produce, characterize, and apply a biosurfactant as a bioremediation tool for oil-contaminated coastal environments.
The biosurfactant was produced in a medium containing 5.0% corn steep liquor and 1.5% residual frying oil by Candida tropicalis (URM 1150) for 144 hours with 200 rpm agitation. The growth curve and production of the biosurfactant were evaluated. The emulsifying properties and stability of the biosurfactant were tested against pH, temperature, and NaCl variations. Toxicity assays were carried out on seeds, the microcrustacean Artemia salina, and the bivalve Anomalocardia brasiliana. Static and kinetic assays were performed, alongside stone washing to assess the removal capacity.
The produced biosurfactant had a surface tension reduction capacity of 70 to 25 mN/m, yielding 7.1 g/L, and a critical micellar concentration (CMC) of 0.4%. During the initial 4-hour cultivation period, the growth curve showed an exponential phase, marked by a constant increase in production. In the final growth stage, biosurfactant production reached its maximum value of 7.1 g/L. After 120 h of cultivation, the maximum biomass output of 9.93 g/L was reached. The biosurfactant was applied in static and kinetic tests with seawater and sand contaminated with oil. The biosurfactant remained stable in the face of the tested variations. The biosurfactant is anionic in nature and showed no toxicity to the microcrustacean Artemia salina, vegetable seeds, or the bivalve Anomalocardia brasiliana, which exhibited 100% survival rates. The biomolecule removed 97% of the contaminated oil on the stone. The biosurfactant proved efficient and biocompatible for the bioremediation of oil derivatives, showing significant removal rates of hydrophobic compounds.
These results indicate that the biosurfactant has great potential for application as a remediation agent to clean up oil spills in coastal environments.
Keywords
- marine environment
- bioremediation
- bivalve
- Candida
- bioproduct
Pollution caused by oil spills and leaks has a huge detrimental impact on the marine ecosystem. Although all oil spills have catastrophic potential, the long-term damage is more related to the clean-up speed than the volume of oil spilled. Oil causes great damage because hydrocarbons are poorly biodegradable, thus compromising the ecosystems and biodiversity [1, 2].
In this context, oil can be transported to the intertidal zones by currents and winds and when it reaches the coast, the cleaning mechanism becomes complex since oil is made up of around 83–87% carbon, 10–14% hydrogen, 0.05–6% sulfur, 0.05–1.5% oxygen, and 0.1–2% nitrogen, thus, these contaminants have greater hydrophobicity, are less volatile, have lower solubility and have a high absorption capacity [3, 4].
The biological and physical–chemical characteristics of crude oil change after a spill. For example, the viscosity, density, and concentration of nickel, sulfur, and asphaltenes are subject to change during the first few months. In addition, crude oils are known to form sticky water-in-oil emulsions. In nature, it is possible to biodegrade petroleum hydrocarbons adequately, but this differs for branched and long chain hydrocarbons. Due to the increase in pH and salinity, the degradation rate decreases, which is why aliphatic hydrocarbons in crude oil have low degradation under hypersalinity conditions [5].
However, oil spills can be treated using various methods, including chemical, physical, and biological processes. Conventional chemical and physical removal mechanisms can quickly disperse spilled oil; however, they mostly transfer contaminants from one environment to another [6]. Therefore, using dispersants to remediate oil-contaminated environments is one of the critical components in the treatment of spills due to their function of breaking up and dispersing the smaller fractions of oil contained in a body of water [7].
As such, the dispersants used to mitigate areas affected by oil spills are chemically synthesized and can be defined as a mixture of surfactants, solvents, and additives. Moreover, the dispersants reduce the oilwater interfacial tension and, with sufficient mixing energy, can break the oil into smaller droplets and spread them throughout the water column. However, due to their toxicity, the need has arisen to develop environmentally friendly and bio-based dispersants [8].
Accordingly, biosurfactants act as natural surfactants obtained through microbiological processes and consist of secondary metabolites secreted extracellularly and bound to the microbial cell wall. As a result of their diverse structures and properties, biosurfactants perform various activities, including the remediation of water contaminated by oil spills [2, 9]. These molecules have emerged as an alternative technology for mitigating these spills due to the amphipathic nature of their compounds, i.e., they tend to distribute themselves at the interfaces between fluid phases with varying degrees of polarity, decreasing surface and interfacial tensions. In addition to their properties, natural surfactants have advantages compared to their synthetic counterparts, such as low toxicity, short environmental half-life, and specific effectiveness under extreme conditions [5, 8, 10].
This work aimed to produce a biosurfactant using the yeast Candida tropicalis URM 1150 and apply it to bioremediation of coastal environments affected by oil spills.
Candida tropicalis URM 1150 was obtained from the culture bank of the Federal University of Pernambuco, Recife, Brazil, and used as the biosurfactant-producing microorganism. The cultures were kept in Petri dishes with Yeast–Mold Agar (YMA) and repeated once after 30 days.
The yeast inoculum was standardized by transferring the cultures to a Petri dish containing YMA to obtain new cultures. The sample was transferred to an Erlenmeyer flask with 100 mL of Yeast Malte Broth (YMB) medium and grown at 28 °C under agitation at 200 rpm for 24 h (Shaking refrigerated incubator TE-424, TECNAL, Piracibaca, Brasil). Dilutions were made to a diluted concentration of 109 cells/mL.
A fermentation containing 5% corn steep liquor and 1.5% residual frying oil was performed, and the mixture was incubated with a cell suspension of 109 cells/mL to produce the biosurfactant. The flasks were kept at pH 5.6 (pH meter TEC-7, TECNAL, Piracicaba, Brasil) and shaking orbitally at 200 rpm for 144 hours.
For extraction, the biosurfactant was separated from the metabolic liquid using ethyl acetate (C4H8O2), and the process was repeated twice. The solvent was then removed to a separating funnel, and discarded before the aqueous phase was eliminated. The solvent phase was dried using anhydrous sodium sulfate (Na2SO4) and then filtered and evaporated [11].
Surface tension was measured on a tensiometer (KSV Instruments Ltd, Sigma 700, Helsinki, Finland) with a du Noüy ring. Subsequently, the CMC was determined by measuring the surface tension of a water sample to which the surfactant was progressively introduced until a constant tension value was reached. The CMC value was determined as g/L of the extracted biosurfactant [12].
The emulsification activity was determined by analyzing samples of the biosurfactant produced by Candida tropicalis using the methodology previously proposed by Cooper and Goldenberg [13]: 2.0 mL of the cell-free metabolic liquid was added to 2.0 mL of the oily substrate and mixed using a vortex for 2 minutes (Vortex Mixer AP-56/1, TECNAL, Piracicaba, Brasil). After 24 hours of stability, the E24 index was given as a percentage of the height of the emulsified layer (mm) divided by the total height of the liquid column (mm).
Aliquots were collected during fermentation at intervals of 2, 4, 6, 8, 12, 24, 48, 72, 96, 120, and 144 hours and subjected to centrifugation (4500 rpm and 9 °C for 15 minutes, Megafuge 16R centrifuge, Thermo Fisher Scientific, Waltham, MA, USA), then filtered, after which the cell-free metabolic liquids were used to measure surface tension, pH, and biosurfactant yield. To determine the biomass by dry weight, 50 mL of the culture was first centrifuged at 5000 rpm for 15 minutes while the supernatant was discarded. After centrifugation, the biomass was dried in an oven at 105 °C for 24 hours and measured [14].
Various temperatures (0 °C, 5 °C, 25 °C, 70 °C, 100 °C, and 120 °C), NaCl concentrations (2.0%, 4.0%, 6.0%, 8.0%, and 10.0%) and pH values (2.0, 4.0, 6.0, 8.0, 10.0, and 12.0) of the biosurfactant were analyzed to determine surface tension and emulsification activity. The assays were carried out in triplicate.
For the ionic charge of the biosurfactant, a Zeta potentiometer was used with the Zeta-Meter 4.0 + ZM3-DG Direct Imaging system (Zeta Meter, Inc., Harrisonburg, VA, USA).
The biosurfactant’s chemical structure was characterized by Fourier-transform infrared spectroscopy (FTIR), using Perkin Elmer’s FT-IR 400 spectrum (Spectrum 400, Perkin Elmer, Shelton, CT, USA), with a resolution of 4 cm-1 in the region of 400 to 4000 wave numbers (cm-1).
The toxicity of the isolated biosurfactant was assessed using microcrustacean (Artemia salina) larvae as an indicator. After 24 hours of incubation, the larvae were used. The biosurfactant solutions were used in different concentrations (½CMC, CMC, and 2CMC). The analyses were conducted in 10 mL penicillin vials containing 10 larvae in 5 mL saline solution per vial. The larvae in each flask were exposed to 5 mL of the tested concentrations for 24 hours, and the mortality was observed [15]. Sea salt was used as a control. The threshold toxic concentration was defined as the lowest concentration capable of causing the death of the microcrustacean in 24 hours.
In this study, Solanum lycopersicum (tomato) seeds purchased from the
Ferreira Costa store (Ferreira Costa, Recife, Brazil) were used to evaluate the
phytotoxicity of the biosurfactant using a static test employing seed germination
and root growth [16]. Solutions of the isolated biosurfactant were prepared in
distilled water at different concentrations (½CMC, CMC, and 2CMC).
The toxicity of the biosurfactant was determined in sterile Petri dishes (10 cm)
containing Whatman No. 1 filter paper discs. After five days of incubation in the
dark, seed germination, root growth (
Relative seed germination (%) = (number of seeds germinated in the
extract/number of seeds germinated in the control)
Relative root length (%) = (average root length in extract/average root
length in control)
GI = ((% seed germination)
The oil was added until the stones were completely immersed, noting all volumes utilized. The stones were carefully transferred to a 100 mL beaker using tweezers and subjected to the following treatments: Addition of the biosurfactant isolated in ½CMC (0.2%), CMC (0.4%), 2CMC (0.8%), the crude biosurfactant, a chemical surfactant (Sodium dodecyl sulfate, VETEC, Jacaré, Brasil), and a control sample. The volumes used were recorded, and the washing percentages were calculated. The oil remaining in the washed stones was determined by gravimetry after extraction with hexane and expressed as a percentage.
For this test, 40 mL of water was added to a Petri dish with a diameter of 15 cm. The crude and isolated biosurfactant was tested with concentrations of ½CMC (0.2%), CMC (0.4%), and 2CMC (0.8%). The dispersion capacity of the biosurfactant was evaluated. The results of the experiments, performed in triplicate, were expressed as the average diameter of the clear zones that appeared in the Petri dishes, while the dispersion index was defined as the percentage of the average diameter in relation to the diameter of the dish (15 cm) [12].
Samples of 50 g of standard sand were contaminated with 10% motor oil and placed in 250 mL Erlenmeyers flasks, then autoclaved. Next, 100 mL of each surfactant solution was introduced into the contaminated soil flask. The biosurfactant and the synthetic surfactant (sodium dodecyl sulfate) were used in concentrations corresponding to half the CMC (½CMC), the CMC, and double the CMC (2CMC). The crude biosurfactant (cell-free metabolic liquid) was also tested in the same quantity (100 mL). A bottle with sand and 100 mL of water, without added surfactants, was used as the control. The flasks were agitated at a stirring speed of 150 rpm at 28 °C for 24 hours. After treatment, the sand and the cleaning solution were separated for analysis [12].
Glass columns (55
Experiments to remove engine oil were carried out in 250 mL Erlenmeyer flasks containing 50 mL of seawater and 1% engine oil. The medium was sterilized and then inoculated with 5% biosurfactant-producing yeast. The experiments were carried out under three different conditions: Condition 1: seawater + engine oil + C. tropicalis; condition 2: seawater + engine oil + C. tropicalis + biosurfactant at CMC (0.4 g/L); condition 3: seawater + engine oil + C. tropicalis + biosurfactant at twice the CMC (0.8 g/L). The flasks were incubated on a rotary shaker at 150 rpm for 10 days.
The 120-hour acute toxicity experiment was carried out at the Invertebrates and
Marine Biology Laboratory in the Catholic University of Pernambuco (Pernambuco,
Brazil), using beakers with a volume of 2 L, duly filled according to the tested
dilutions. The bivalves used in the experiment were collected at low tide in the
Rio Formoso estuary (8º39′45′′S and
35º06′15′′W) in the municipality of Rio Formoso,
Pernambuco. After collection, the bivalves were taken directly to the laboratory
and kept in 2L beakers with seawater at a salinity of 26 and temperature of 27
Three dilutions of the biosurfactant (1:40, 1:80, and 1:160) conducted with seawater were tested over three replicates. Five specimens were exposed for 120 hours without any change in food or water. The controls only included seawater, similar to Santos et al. [18]. The dilutions were made from the CMC (0.4 g/L) of the biosurfactant produced by Candida tropicalis.
The lethality of the biosurfactant was expressed based on the survival of the specimens tested, as shown in Table 1 below (Ref [18]).
| Degree of toxicity | |
| Survival (S) | Classification |
| S |
High survival |
| 50% |
Medium survival |
| S |
Low survival |
Source: Adapted from Santos et al. [18].
Although their applications are promising, large-scale production of biosurfactants is restricted due to their high production costs, mainly due to expensive growth media. To ensure profitable production, by-products, and agro-industrial waste have been used as alternative substrates to guarantee production profitability [19].
The growth and production of biosurfactant produced by the yeast Candida tropicalis were evaluated in a medium containing 5% corn steep liquor and 1.5% residual frying oil for 6 days (Fig. 1).
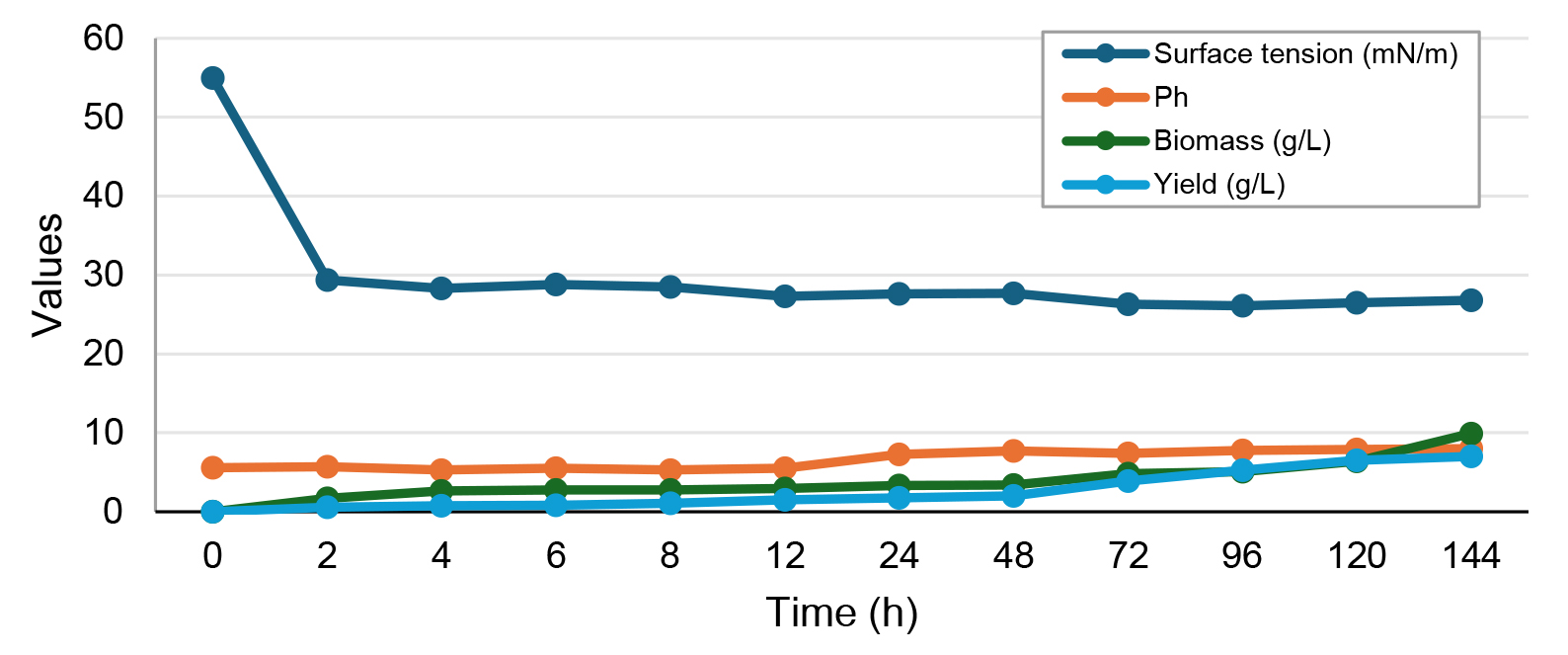 Fig. 1.
Fig. 1.
Growth and production kinetics of the biosurfactant produced by Candida tropicalis grown in 5% corn steep liquor and 1.5% residual frying oil.
Over a 4 hour cultivation period, the growth curve showed an exponential phase characterized by a continuous increase in production. During the final growth phase, biosurfactant production peaked at 7.1 g/L. An increase in pH was observed after 24 hours of cultivation. As for biomass, its maximum value was 9.93 g/L after 120 hours. Furthermore, a significant reduction in the surface tension of the medium was noted, which went from 55 mN/m to 27.6 mN/m over 6 hours of cultivation.
Almeida et al. [20] evaluated the growth curve of the biosurfactant produced by Candida tropicalis in a medium supplemented with 2.5% residual frying oil, 2.5% corn steep liquor, and 2.5% molasses. After 120 h of cultivation, the maximum biomass production of 23.75 g/L was reached, and the lowest surface tension was observed, reducing from 55 to 30.4 mN/m.
da Silva et al. [21] analyzed a commercial biosurfactant produced with the yeast Starmerella bombicola. The results during the process showed that after fermentation, the medium’s surface tension was reduced to 32 mN/m. The extraction process resulted in a significant yield of 32.5 g/L.
The critical micelle concentration (CMC) represents the minimum concentration of biosurfactant required to reduce the surface tension of water to the maximum and initiate the formation of micelles. This concentration was used to measure the effectiveness of the biosurfactant [22].
The surface tension reduction capacity of the biosurfactant produced by Candida tropicalis was satisfactory since the water tension was reduced from 70 mN/m to 25 mN/m as the biosurfactant concentration increased to 0.4 g/L (Fig. 2), demonstrating that the CMC was reached.
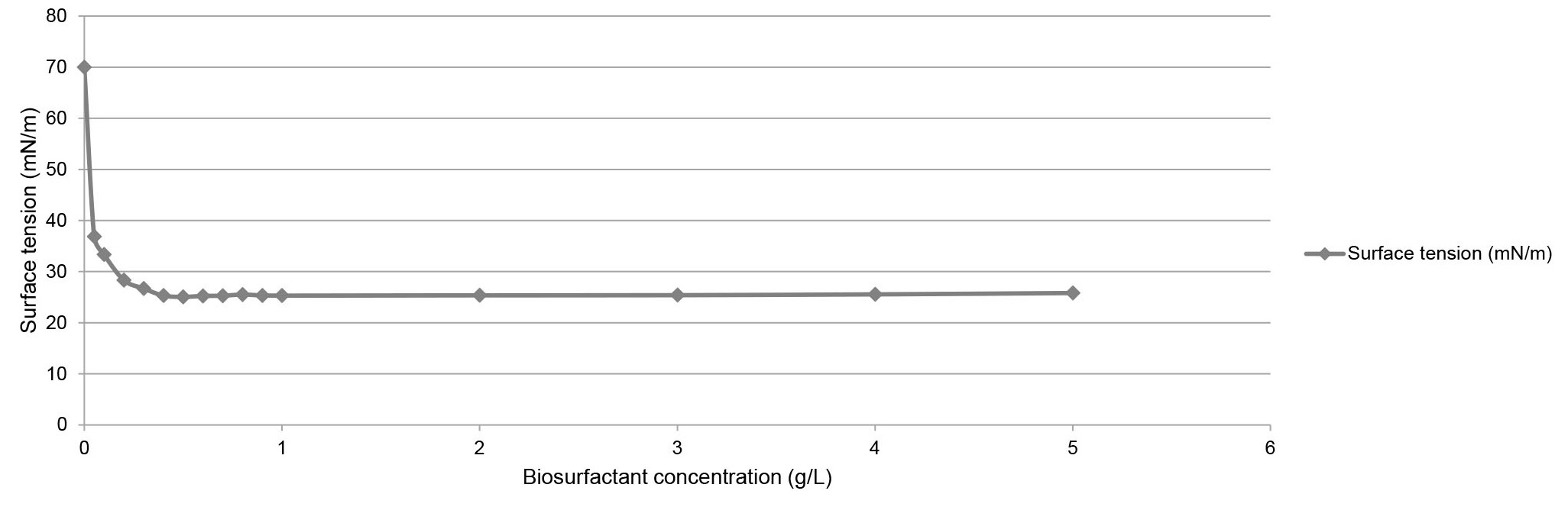 Fig. 2.
Fig. 2.
Critical micelle concentration of the biosurfactant produced by Candida tropicalis grown in medium supplemented with 5% corn steep liquor and 1.5% residual frying oil.
Compared with other produced biosurfactants, the Candida tropicalis biosurfactant obtained a low CMC. Different species of the genus Candida, such as C. bombicola (0.5 g/L) [22], C. lipolytica (0.5 g/L) [12], C. guilliermondii (0.72 g/L) [23], presented the highest CMC.
According to Akbari et al. [24], the CMC is when the surface of the liquid is completely saturated with surfactant molecules in an aqueous solution, and, at the same moment, the surface tension reaches its minimum value. The CMC indicates the efficiency of the surfactant; a lower CMC represents the presence of fewer surfactant molecules to saturate the interface and micelles.
The CMC is one of the main aspects capable of interfering with removing contaminants from matrices. The formation of micelles may be the key player in the contamination process, as the micelles immobilize the contaminating molecules in their hydrophobic core for subsequent removal [25].
A biosurfactant must preserve its surfactant characteristics in all environmental conditions. Environmental factors such as pH, temperature, and salinity affect the activity and stability of a biosurfactant; consequently, studying the influence of these variables is of major importance [26].
The emulsification stability of the biosurfactant produced by Candida tropicalis was analyzed using cell-free metabolic liquid after 144 hours of fermentation under various conditions. For this evaluation, different pH values (2, 4, 6, 8, 10, and 12), temperature variations (0, 5, 25, 70, 100, and 120 °C), and variable NaCl concentrations (2, 4, 6, 8, 10 and 12%), were employed.
As shown in Table 2, the biosurfactant demonstrated significant stability when subjected to emulsifying activity, maintaining more than 80% emulsification of the engine oil under all temperature variations. The efficiency varied between 80% and 100% at different pH values and between 88% and 100% in the NaCl concentrations tested.
| pH | Emulsification of burnt engine oil (%) | Temperature (°C) | Emulsification of burnt engine oil (%) | NaCl (%) | Emulsification of burnt engine oil (%) |
| 2 | 95 |
0 | 90 |
2 | 88 |
| 4 | 100 |
5 | 80 |
4 | 100 |
| 6 | 90 |
25 | 85 |
6 | 80 |
| 8 | 90 |
70 | 85 |
8 | 90 |
| 10 | 80 |
100 | 85 |
10 | 100 |
| 12 | 80 |
120 | 90 |
12 | 92 |
When comparing these results with those in the literature, Almeida et al. [20], the yeast Candida tropicalis UCP 0996 has been recognized as an osmotolerant microorganism capable of surviving in high salt concentrations. This ability is crucial for the fungus to persistin saline environments. This characteristic highlights the potential of yeast for future uses in various biotechnological processes under variable and extreme environmental conditions.
To ensure the stability of the biosurfactant produced by Candida tropicalis, the cell-free broth (crude biosurfactant) was subjected to experimental conditions, including different pH values, temperatures, and NaCl concentrations. The surface tension reduction properties were analyzed in each condition, allowing a comprehensive assessment of its stability in the face of variations.
The assays did not vary following changes in pH, temperature, or NaCl, demonstrating that the biosurfactant obtained by Candida tropicalis did not show significant changes in surface tension (Table 3).
| pH | Surface tension (mN/m) | Temperature (°C) | Surface tension (mN/m) | NaCl (%) | Surface tension (mN/m) |
| 2 | 29.5 |
0 | 27.9 |
2 | 28.8 |
| 4 | 28.9 |
5 | 27.3 |
4 | 28.1 |
| 6 | 27.6 |
25 | 27.4 |
6 | 27.6 |
| 8 | 26.4 |
70 | 27.6 |
8 | 27.4 |
| 10 | 28.2 |
100 | 27.4 |
10 | 27.5 |
| 12 | 29.7 |
120 | 27.2 |
12 | 27.3 |
Lira et al. [14] analyzed the biosurfactant produced by Candida guilliermondii and obtained similar results relating to stability. In another study, da Silva et al. [22] evaluated the surface tension reduction capacity of the biosurfactant produced by Candida bombicola, which varied between 30 and 35 mN/m in relation to pH, by 29 to 32 mN/m at different temperatures, and 30 to 31 mN/m for the NaCl concentrations.
The biosurfactant produced by the yeast Candida tropicalis showed a negative charge after zeta potentiometer analysis, with –75.8 ZPmV and 135.7 µS/cm at 25 °C, which indicates that it is an anionic surfactant. According to the literature, other biosurfactants produced by different Candida species also showed an anionic character when subjected to this analysis [11, 22].
According to the infrared spectrum (Fig. 3), the signals found in the 2915 cm-1 and 2849 cm-1 regions correspond to the asymmetric stretching of the C–H bonds. These signals are typical of aliphatic chains, confirming the presence of carbon chains in the molecule that makes up the apolar fraction of the biosurfactant. The peak observed at 1709 cm-1 is associated with the carbonyl function (C=O) present in the molecular structure, suggesting the presence of a carboxylic acid function since the molecule has acidic characteristics and is soluble in basic media. The signal at 1582 cm-1 corresponds to a possible double bond between carbon atoms (C=C), indicating that the chain responsible for the apolar fraction is unsaturated.
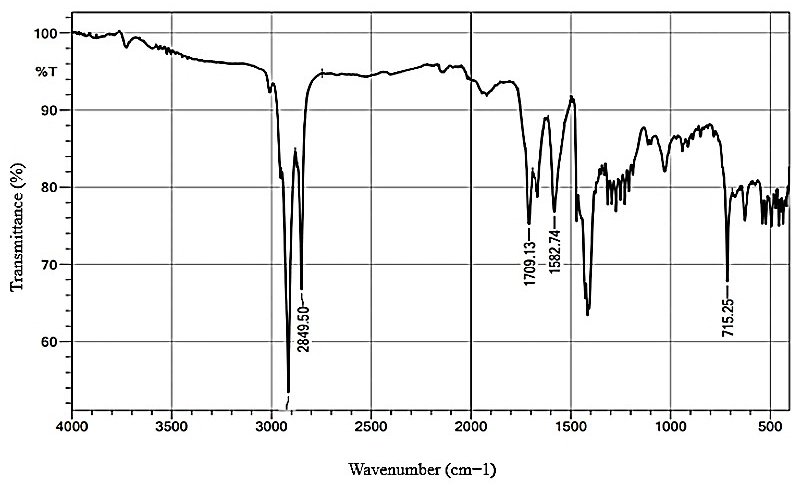 Fig. 3.
Fig. 3.
Fourier-transform infrared spectroscopy (FT-IR) spectrum for the semi-purified biosurfactant produced by Candida tropicalis grown in a medium containing 5% corn steep liquor and 1.5% residual frying oil.
Therefore, it can be concluded that the biosurfactant is possibly a fatty acid metabolized by the microorganism, as evidenced by the presence of aliphatic chains, carbonyl groups, and characteristic double bonds.
In this study, stones from the coastal environment were impregnated with motor oil and washed with different concentrations of the isolated biosurfactant, the crude biosurfactant, and a chemical surfactant. Removal rates of 97%, 93%, 97%, 83%, and 97% were observed for the crude biosurfactant, ½CMC, CMC, 2CMC, and the chemical surfactant sodium dodecyl sulfate, respectively. These values demonstrate the effectiveness of the biosurfactant, even matching the results obtained by its chemical counterpart.
Durval et al. [10] evaluated the effectiveness of the biosurfactant produced by Bacillus cereus in removing engine oil. The authors used a concentration of 2CMC, and the results showed a removal capacity of the present engine oil of 69%, demonstrating significant degradation of the pollutant.
In another study, Soares da Silva et al. [6] investigated the properties of the biosurfactant produced by Pseudomonas cepacia. The tests resulted in a removal rate of 84%, indicating performance for applications in oil-contaminated environments.
The results obtained for engine oil dispersion in seawater showed capacities of 94%, 97%, and 98% for the ½CMC, CMC, and 2CMC concentrations, respectively; the dispersion capacity for the crude biosurfactant was 74%. These results indicate the potential for bioremediation applications to recover marine waters contaminated by oils.
Santos et al. [27] evaluated the oil dispersion capacity of a biosurfactant produced by the yeast Candida lipolytica through a test developed by the US Environmental Protection Agency and reported a dispersion index that ranged from 50% to 100%, depending on the concentration of the biosurfactant.
The ability of petroleum hydrocarbons to bind physically and chemically to soil particles is one of the main obstacles to the bioremediation of contaminants in soil, ultimately preventing the removal and degradation of these compounds [26].
The results of a kinetic test regarding the removal of engine oil from sand by the biosurfactant produced by the yeast Candida tropicalis are described in Table 4.
| Removal agent | Oil removal (%) |
| Control (distilled water) | 17 |
| Crude biosurfactant | 72 |
| Biosurfactant concentration at ½CMC | 81 |
| Biosurfactant concentration at CMC | 90 |
| Biosurfactant concentration at 2CMC | 92 |
| SDS (sodium dodecyl sulfate) | 29 |
The assays were carried out using crude and isolated biosurfactants, with the chemical surfactant (SDS) and distilled water used as controls. It can be seen that the higher concentration of the isolated biosurfactant influenced the removal percentage, demonstrating the biosurfactant’s increased ability to solubilize the oil present in the sand. The highest removal percentages of 92% and 90% were obtained for the condition containing 2CMC and CMC concentrations.
The crude biosurfactant (cell-free metabolic liquid) produced from C. tropicalis was also tested to assess oil removal, where by 72% removal was observed. The results obtained for the chemical surfactant SDS showed 29% oil removalcompared to the biosurfactant produced by C. tropicalis.
Durval et al. [10] achieved pollutant removal rates of 84% for the biosurfactant produced by Bacillus cereus, used at a concentration of 2CMC in a kinetic test with beach sand.
These results reinforce the use of different microorganisms in the production of biosurfactants, contributing to the development of efficient bioremediation methods.
Spills of petrochemical products cause widespread contamination of ecosystems due to their high toxicity and are one of the main causes of soil pollution. Remediation techniques for oil-contaminated soils are divided into physical, chemical, and biological, which can be applied in situ or ex situ [26].
In this study, both crude and isolated biosurfactants could remove the motor oil adsorbed on the sand. The biosurfactant achieved a high removal rate regardless of the concentration used. The removal rate for CMC, ½CMC, and 2CMC varied between 93%, 94%, and 97%, respectively. The crude biosurfactant (cell-free metabolic liquid) produced by the yeast Candida tropicalis was also tested to assess its oil removal potential, where a removal rate of 92% was observed. The result obtained by the chemical surfactant sodium dodecyl sulfate showed a removal rate of 96%.
Santos et al. [28] obtained similar results with the biosurfactant produced by Candida sphaerica, with removal rates of 80%, 67%, 70%, and 70% for the raw biosurfactant, ½CMC, CMC, and 2CMC, respectively. Durval et al. [10] obtained a lower removal rate (63%) for an oil adsorbed by sand using a biosurfactant produced by Bacillus cereus.
Due to their amphipathic nature, biosurfactants can form micelle aggregates with the contaminants, increasing the removal percentage. In short, these results indicate the ability of the biosurfactant produced by the yeast Candida tropicalis to reduce the interfacial tension of the oil, favoring its mobilization.
Various processes conducted by companies involved in the petroleum sector are responsible for polluting the marine environment. During these cases, oil occasionally reaches the seawater, requiring the use of different containment technologies, which include the application of surfactants [20].
The removal percentage of the contaminant corresponds to the effectiveness of different concentrations of the biosurfactant and methods that can remove the oil from the water. It was possible to observe 100% removal of the oil in seawater in conditions 2 and 3 containing the biosurfactant in CMC and 2CMC over the 10 days, meaning the biosurfactant produced by C. tropicalis acted as a solubilizing agent for the engine oil, facilitating its removal. In comparison, condition 1, containing only the microorganism, could remove only 37% of the oil.
Silva et al. [29] analyzed the percentage degradation of two biosurfactants, one produced by the bacterium Bacillus cereus and the other by the yeast Candida sphaerica, over 30 days. The degradation capacity of the microorganisms increased as the experiment developed. However, biodegradation of approximately 60% and 80% occurred in the first 10 days for the inoculum using the CMC and 2CMC conditions of the biosurfactant produced by B. cereus.
The phytotoxicity assay involving the biosurfactant produced by Candida tropicalis using Solanum lycopersicum (tomato) seeds showed no inhibitory effect on seed germination or root growth. Germination rates varied between 89%, 91%, and 87% for the ½CMC, CMC, and 2CMC concentrations, respectively. It was also found that leaf and secondary root growth occurred at all the tested concentrations.
Lira et al. [14] analyzed the toxicity of the Candida guilliermondii biosurfactant on Solanum lycopersicum seeds and found that the biosurfactant had no inhibitory effect on seed germination, with rates varying between 43%, 57%, and 89% for the ½CMC, CMC, and 2CMC concentrations, respectively. Pinto et al. [11] observed that there was no inhibitory effect on the germination rates of tomato seeds against the biosurfactant produced by Candida bombicola, with germination rates of 85%, 38%, and 22% for ½CMC, CMC, and 2CMC, respectively.
Due to its speed, reliability, and low cost, the Artemia salina toxicity experiment is one of the most widely used biological assays. Artemia salina is a microcrustacean used for this type of test because it is sensitive to environmental variations, easy to acquire, and an alternative to animal tests [30].
After 24 h of exposure to the ½CMC, CMC, and 2CMC concentrations of the biosurfactant, the Artemia salina larvae showed a survival rate of 100%. These results indicate that the biosurfactant was not toxic at the concentrations tested.
The results obtained by Almeida et al. [20] for the Candida tropicalis biosurfactant were not toxic to Artemia salina, indicating toxicity only for the 10CMC concentration. Pinto et al. [11] assessed the toxicity of the biosurfactant produced by Candida bombicola and obtained results of 50% and 100% lethality rates at concentrations of CMC and 2CMC, respectively.
Bivalve mollusks are organisms capable of bioaccumulating pollutants and pathogens, demonstrating why they are important bioindicators in aquatic environments. The shellfish Anomalocardia brasiliana, popularly known as cockles, is a marine bivalve mollusk with a wide geographical distribution from the West Indies to Brazil and Uruguay. It is a species adapted to a great diversity of habitats, such as coves, bays, estuaries, and shallows without vegetation, in which it lives buried superficially in sediments, mainly sandy mud [31].
Experiments using bivalves allow for the prior identification of the potential ecotoxicological impacts of biosurfactants, such as sublethal and lethal effects, changes in behavior, and reproduction, ensuring that these bioproducts are safe for the environment before their large-scale application. This guarantee is essential as these compounds are increasingly used in various sectors, including food, pharmaceuticals, and environmental remediation [32].
The results of the acute toxicity experiment using the bivalve Anomalocardia brasiliana with different dilutions of the biosurfactant produced by Candida topicalis are shown in Table 5. The high survival rate (100%) obtained in this study classifies the biosurfactant as low in toxicity, guaranteeing its safety for environmental use.
| Dilution (v/v) | I | II | III | Survival |
| Control | 5 | 5 | 5 | 100% |
| 1:40 | 5 | 5 | 5 | 100% |
| 1:80 | 5 | 5 | 5 | 100% |
| 1:160 | 5 | 5 | 5 | 100% |
In a previous study, Durval et al. [33] subjected the same bivalve mollusk to the crude biosurfactant and the formulated biosurfactant produced by Bacillus cereus. The results showed low toxicity, especially when the biosurfactant was diluted at a ratio of 1:20 (v/v) or even higher dilutions. Low toxicity was observed after 96 hours of exposure of Anomalocardia brasiliana specimens to the biosurfactants used. Santos et al. [18] obtained similar 95%, 95%, and 100% results for the 1:40, 1:80, and 1:160 dilutions, respectively, of the biosurfactant produced by C. lipolytica in a 96-hour experiment.
Developing and using new biosurfactant toxicity experiments alongside bivalve mollusks is extremely important for the environmental analysis of new biotechnological compounds. Aquatic invertebrates such as mussels and oysters are filter-feeding organisms that accumulate substances in the water, making them highly sensitive bioindicators that quickly reflect water quality changes and the presence of toxic products [34]. Due to this sensitivity and capacity for accumulation, bivalves are ideal for acute or chronic toxicity studies.
The anionic biosurfactant produced by Candida tropicalis URM using industrial waste as a substrate has emerged as a promising and sustainable solution for the remediation of coastal environments contaminated by oil. Its lack of proven toxicity and high oil removal capacity opens the way for its application, contributing to preserving marine biodiversity and recovering degraded coastal ecosystems.
The dataset is available from the corresponding author upon request.
Conceptualization, JML and LAS; methodology, MCFC; RRS; HMM and GSS; validation, JML; MCFC and RRS; formal analysis, MCFC; RRS and HMM; investigation, MCFC and RRS; writing—original draft preparation, MCFC; RRS and JML; writing—review and editing, MCFC and JML; visualization, JML; supervision, JML; project administration, JML and LAS. All authors have read and agreed to the published version of the manuscript. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
According to local regulations of the state of Pernambuco, the experiments involved in this experiment do not require ethical approval.
The authors would like to thank the Science and Technology Support Foundation (FACEPE), the Catholic University of Pernambuco (UNICAP) and the Advanced Institute of Technology and Innovation (IATI), Brazil.
This research was funded by the following agencies: Science and Technology Assistance Foundation of the state of Pernambuco (Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco – FACEPE), National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq and Higher Education Personnel Improvement Coordination (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – CAPES, Finance Code 001).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



