1 Department of Environmental Biotechnology, Biotechnology Research Center, Al-Nahrain University, 10018 Baghdad, Iraq
2 Department of Medical Laboratory Techniques, College of Health and Medical Techniques, Al-Esraa University, 10018 Baghdad, Iraq
Abstract
Contamination with crude oil and hydrocarbons has become a global threat. Such threats have urged us to invent solutions to deal with this dilemma. However, chemical treatment comes with limited benefits. The use of bioremediation proved to be the optimal approach to face this problem since it is cost-effective, time-conserving and may improve the quality of soil and increase its fertility.
Soil samples were collected from three sites with the highest degree of pollution in Iraq: Al-Latifia, Al-Begei, and Basra. These were the source of novel Streptomyces isolates that could degrade contaminants and be used as a source of nutrients. The isolation principle was the degradation of petroleum in these soils as a carbon source.
The most efficient isolate was obtained from Basra soil, characterized by the highest degree of contamination. The bacterium grows on minimal medium with crude oil, diesel fuel, aromatic, and non-aromatic hydrocarbons as the only source of carbon and showed the ability to reduce a hydrocarbon mixture containing 23 analytes with C8-C40 and C13-C30 representing total crude hydrocarbons effectively and with a high mineralization capability reaching 84%. This isolate was found to produce biosurfactants and reduce surface tension significantly.
Using chemical treatment of crude oil contaminants, burying the contaminated areas, or sometimes flooding them with water to eliminate these contaminants may not be an effective solution. Thus, using nonpathogenic microorganisms, especially those indigenous to the soil, maybe the most effective solution to achieve this goal.
Keywords
- bioremediation
- Streptomyces
- crude oil contamination
- polyaromatic hydrocarbons
- soil decontamination
Globally, crude oil is the strategic vein and natural energy resource for civilization. Excavation, geological survey, and oil production resulted in an increased rate of ecological pollution found in both terrestrial and aquatic habitats [1]. Since most hydrocarbon components are toxic and resistant to degradation regardless of the environments in which they are found, the spillage of hydrocarbons into the ecosystem through accidents or human activities is a major hazard that tragically influences ecological and environmental systems [2]. Polyaromatic hydrocarbons (PAHs) result from thermal geological reactions that are sometimes associated with the fossil fuel and mining industries and naturally during the burning of vegetation in forests and bushfires. Human activities, industry, fuel combustion, transportation, spillage of petroleum products, and incineration of waste are potentially significant sources of releasing PAHs into the environment [3]. However, with large-scale oil production, large amounts of polycyclic aromatic hydrocarbon sludge from crude oil characterized by a highly complicated chemical structure, high molecular weight, strong molecular bonds, and hydrophobic characteristics can be found in the ecosystem. These properties make the biodegradation process more difficult since they are less vulnerable to this process [4, 5]. As they are persistent and difficult to degrade, they may exert chemical and physical alterations in the ecosystem, influencing land and aquatic habitats [6]. For this reason, these compounds were considered hazardous and require treatment to minimize their effect, if possible, before release into the environment [7, 8]. Among the highly recommended methods and bioremediation techniques, the use of microorganisms stands as a promising approach that is considered a greener method and cost-effective way to control and decontaminate the ecosystem with minimum disruptive side effects potentially occurring to the environment. The principle behind these methods lies in activating metabolic activity that involves oxidation–reduction activity pathways for contaminants and converting them to simpler compounds that may become CO2 and water [9]. Several physical and chemical methods have been accommodated and performed to decontaminate the environment. However, the drawback of such methods is that they need intensive labor and often involve the probability of expanding the pollution due to a need to deposit the waste elsewhere [10]. Bioremediation is divided into two types: in situ, where decontamination techniques include land tilth, the addition of microorganisms, bio-culturing, and bio-ventilation, and ex situ, which includes prefabrication foundation and bioreactor devices [11, 12]. The natural attenuation process, which is the main principle in bioremediation, depends on the ability of indigenous microflora to reduce contamination and eliminate or convert pollutants in the soil to harmless compounds [13]. Still, PAHs in soil may be the main source of abundant carbon necessary for microorganisms; however, limitations in nutrients, nitrogen, and phosphorus may restrict the activity and propagation of microorganisms, leading to less effective decomposition of petroleum and PAH contaminants. Adding such nutrients led to a significant increase and higher efficiency of microbes to degrade what are considered contaminants when such chemicals do not naturally occur in the ecosystem [14, 15]. Most crude oil pollution and oil derivatives are observed in oil-producing countries, especially when the country suffers from unsecured circumstances, such as Iraq, one of the leading countries in oil production. The lack of environmental awareness, low management of oil wells maintenance, and feeble control of production methods and transportation of crude oil and oil derivatives, in addition to terrorist attacks, rendered vast lands around oil wells and storage facilities contaminated with PAHs. Thus, this work was designed to isolate environmentally, PAH degrading, and nonpathogenic bacteria that may be developed as a promising bioremediation agent for a healthy ecology.
Soil samples from storage facilities at Al-Latifia, Al-Begei refinery, and Basra crude oil extraction wells were obtained at a depth of 5–20 cm and dried by hot air as an initial step to the quantification of PAHs and the study of their chemical and physical properties. The control soil sample (blank) was collected from contamination-free areas around Baghdad, Latifia ranches, and counties north of Baghdad distant from oil storage and processing facilities. Soil samples were refined through a 2 mm sieve to eliminate pupils, stones, plant remains, and other impurities.
Both soil pH and salinity were measured. The soil pH was measured in a suspension of 1:5 (soil:deionized water) and measured using a pH meter (model: HI99163, Hanna, Padua, Italy). Salinity was measured using a salinity meter from the same company. The salt content was measured by drying the soil in an oven at 100 °C for 4 h; then, 10% H2O2 was added as an oxidant to eliminate organic materials. This step was repeated thrice until the entire residue turned white. The mixture was allowed to stand for one night and heated at 104 °C to remove H2O2. The remaining salt was collected and weighed to calculate the salt content in each sample.
The total hydrocarbon content of the soil was measured gravimetrically. A weight of 5 g aliquant of moist soil was extracted by Soxhlet using 200 mL dichloromethane for 12 h with a temperature of 54 °C [16]. The extract concentration was measured by a rotary evaporator (model: R-114 RE, Buchi R 124, Marshall Scientific, Hampton, NH, USA) until dryness alongside the mass of TPAHs, according to the method of [17]. Quantification of the extract was performed thrice for accurate results, and saturated hydrocarbons were subjected to separation using a silica gel alumina column and quantified using Shimadzu’s premier gas chromatograph Nexis GC-2030 gas chromatograph (model: Nexis GC-2030, Shimadzu, Kyoto, Japan), as in the previous study [18].
The method from [19] was used to isolate streptomycetes from PAHs contaminated soil from which 1 g was suspended in 500 mL MNGA liquid medium (ingredients per liter; glucose 10 g, beef extract 3 g, peptone 5 g, sodium chloride 3 g, and yeast extract 3 g with final pH 7) [20]. Both naphthalene and phenanthrene (0.1%) were supplemented into the medium as crystals. Inoculates were incubated in an orbital shaker incubator at 200 rpm and 30 °C without light for 10 days. After the incubation period, 10 mL was transferred to be inoculated in 100 mL of freshly prepared liquid MNGA medium with the same hydrocarbon used previously. After the enrichment step, a series of dilutions of 10-1 to 10-6 was made, from which 0.1 mL was cultivated by spreading evenly on starch casein agar supplemented with nalidixic acid 100 mg /mL and cycloheximide 100 mg/mL to inhibit non-spore-forming bacterial, yeast and fungal growth. Plates were incubated for 14 days.
The bacteria genomic DNA of bacteria was extracted using a Favorgen Total DNA Isolation kit (Favorgen, Taiwan, type: FAMBD 001). The primers Str-F: GAACCACTTCGGTGGGGATT and Str-R: CAGGTACCGTCACTTTCGCT were designed in this study depending on the sequence accession no. PP855663.1 to give a product size of 400 bp of 16S ribosomal RNA gene by polymerase chain reaction (PCR) in a final PCR reaction mixture of 20 µL. The PCR amplification program conditions were as follows: Initial denaturation step (one cycle) at 94 °C for 4 min; after this step, 30 cycles of amplification started at 94 °C for 30 sec, annealing at 52 °C for 30 sec, and concluded by extension at 72 °C for 1 min. The final step of PCR amplification was an extension step at 72 °C for 10 min. Amplified samples were cooled to 4 °C until removed from the device. Amplified PCR products were resolved by electrophoresis using 2% agarose at a field strength of 8 v/cm. The gel was stained with red-safe dye (Promega, Madison, WI, USA, cat no.: H1181), visualized under a ultraviolet (UV) transilluminator, and photographed. The PCR products were sent to Macrogene Biotech Corp. (Korea) to be sequenced and compared using the BLAST tool available at https://www.ncbi.nlm.nih.gov/. All Streptomyces isolates were grown separately on ISP-2 medium as pure cultures.
Washed spores from Streptomyces were cultivated on an MNGA medium for 10 days, and their ability to degrade PAHs was tested. The medium was supplemented with naphthalene and phenanthrene as the main carbon source. After the cultivation period, cells were harvested by centrifugation, inoculated in IM medium supplemented with 100 mg naphthalene and phenanthrene, and incubated in the absence of light in a shaker incubator at 200 rpm, 30 °C for 10 days. The control medium was not supplemented with PAHs, allowed to grow simultaneously in the same conditions as before, and served for comparison. This experiment was triplicated for accurate results. The growth of microorganisms was monitored spectrophotometrically at optical density 600 (OD600) nm, and cells were collected by centrifugation at 8000 g for 15 min. The supernatant was transferred to a new sterile tube acidified with 2 N HCl to pH 2 and extracted with equal volume ethyl acetate [21]. The ethyl acetate layer was separated and dried using anhydrous Na2SO4 to dryness with the aid of a vacuum evaporator and a temperature of 40 °C.
Using high-performance liquid chromatography (HPLC model: Shimadzu LC-2050 series, Shimadzu, Kyoto, Japan, equipped with UV absorbance detector), polycyclic aromatic hydrocarbons were identified and quantified. PAH standards were obtained from Sigma-Aldrich. The separation conditions were performed according to [22].
The bacterial isolate with the highest growth rate on a hydrocarbon-rich medium was investigated for biosurfactant production. Using 100 mL Bushnell–Hass medium composed of (g/L) from MgSO4 0.20; CaCl2 0.02; KH2PO4 1.00; K2HPO4 1.00; NH4NO3 1.00; FeCl3 0.05; dextrose 15. The bacterium was inoculated and cultivated for 10 days in a shaker incubator at 200 rpm. Production of biosurfactants was estimated using the following methods: oil displacement test [23], drop collapse rest [24], and E 24 emulsification index percentage was determined [25]. Surface tension change due to biosurfactant was measured using the tensiometer and drop weight method [26].
Bacterial cells were grown in liquid Bushnell–Hass medium in a shaker incubator at 200 rpm, 37 °C for 10 days, and collected by centrifugation at 10,000 rpm for 20 min to remove biomass. A solvent extraction method detected biosurfactants produced by Streptomyces sp. supernatant obtained from the culture medium. The solution was acidified using 1 N HCl to pH 2 and stored at 4 °C overnight for precipitation. After one night of storage in the refrigerator, an equal volume of diethyl ether was used for extraction using a separation funnel. The resulting organic layer was transferred to a beaker, and the solvent was removed by evaporation, during which biosurfactant was retained [27]. Production of biosurfactants, oil displacement ability [28], drop collapse test [29], and emulsification index E24 [30] with hexan and xylene were estimated after this process.
Thin layer chromatography (TLC) using silica gel with dimensions of 20 cm
The physical and chemical properties of Iraqi oil were determined. It is
characterized by specific gravity of 15.6/15.6 pour point (“C), degree American Petroleum Institute (API) gravity of 33.07,
sulfur content of 2.74 (wt. %), Condradson carbon residue 4.60 (wt. %), ash
yield 0.007 (wt. %), pour point (“C)
The chemical and physical properties of each collected soil type were determined. Each site has a unique physical and chemical structure, as listed in Table 1.
| Site | Acidity (pH) | Salinity (g/kg) | TOC (mg/g) | Particle size (%) | ||
| Clay ( |
Silt (2–50 µm) | Sand ( | ||||
| Al-Begei refinery | 8.5 | 3.7 | 20.56 | 0.8 | 51 | 12 |
| Al-Latifia | 7.7 | 18.5 | 21.36 | 3 | 25 | 23 |
| Basra | 5.3 | 38.1 | 2.23 | 0.25 | 87 | 96.2 |
TOC, total organic carbon.
A significant difference in both pH and salinity was found among samples collected from different parts of Iraq. The soil of the Northern part (Al-Begei) is characterized by alkalinity with pH 8.5, low salinity calculated to be less than 10 g/kg and high total organic carbon (TOC). Meanwhile, the Basra site to the south of Iraq was characterized by a low pH of 5.3 with high salinity that reached 38 gm/kg. The pH with moderate alkalinity in the Al-Latifia site located south of Baghdad in the middle of Iraq has acceptable salinity but a high TOC concentration. Basra contained fewer amounts of TOC, less than 17 mg/g, with the identical particle size distribution as 3.6% clay, 77% silt, and 53.5% sand as an average value.
Total concentrations of PAHs (TPAHs) in soils were estimated gravimetrically.
The test started at (t = 0 d) and continued after cultivating microorganisms (t =
120 d) to investigate the degradation potential of PAHs. Microbial flora from
Al-Begei and Al-Latifia soils showed higher potential to perform this process
than the Basra soil, with reduction reaching 1.97
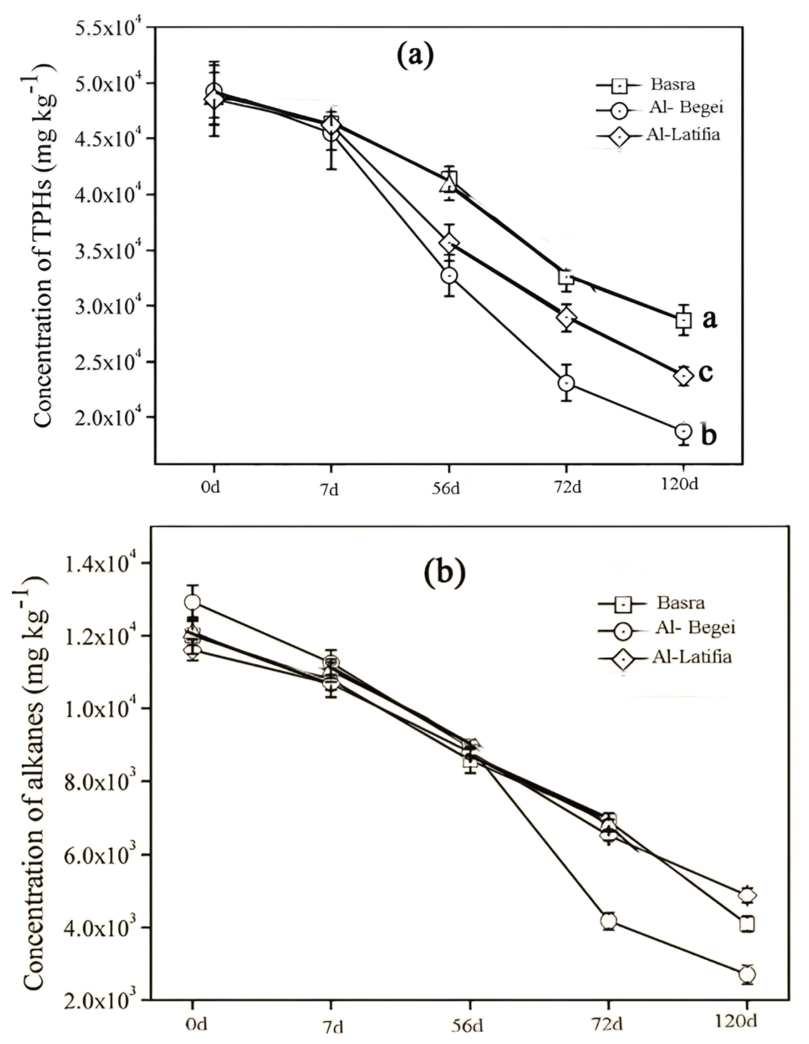 Fig. 1.
Fig. 1.
The residual concentrations of petroleum hydrocarbons (PAHs) in soils from three geographic regions. (a) Concentrations of total petroleum hydrocarbons (TPAHs) in microcosm soils. (b) Concentration of n-alkanes (C8–C40) in microcosm soils.
There are several methods to isolate streptomycetes with active enzyme systems. Among these methods, using phenol as a selective agent for streptomycetes and actinomycetes proved efficient in achieving this goal. Using hydrocarbons as a carbon source helped cultivate Streptomyces isolates, which can grow and assimilate PAHs as an energy source. The morphology of Streptomyces isolate was as follows: No fragmentation of substrate mycelium and aerial mycelium with recti flexible (RF) arrangement. Cultural morphology on ISP2 agar was white leathery brown and showed brown pigment. A photo of Streptomyces isolates illustrating colony morphology is shown in Fig. 2.
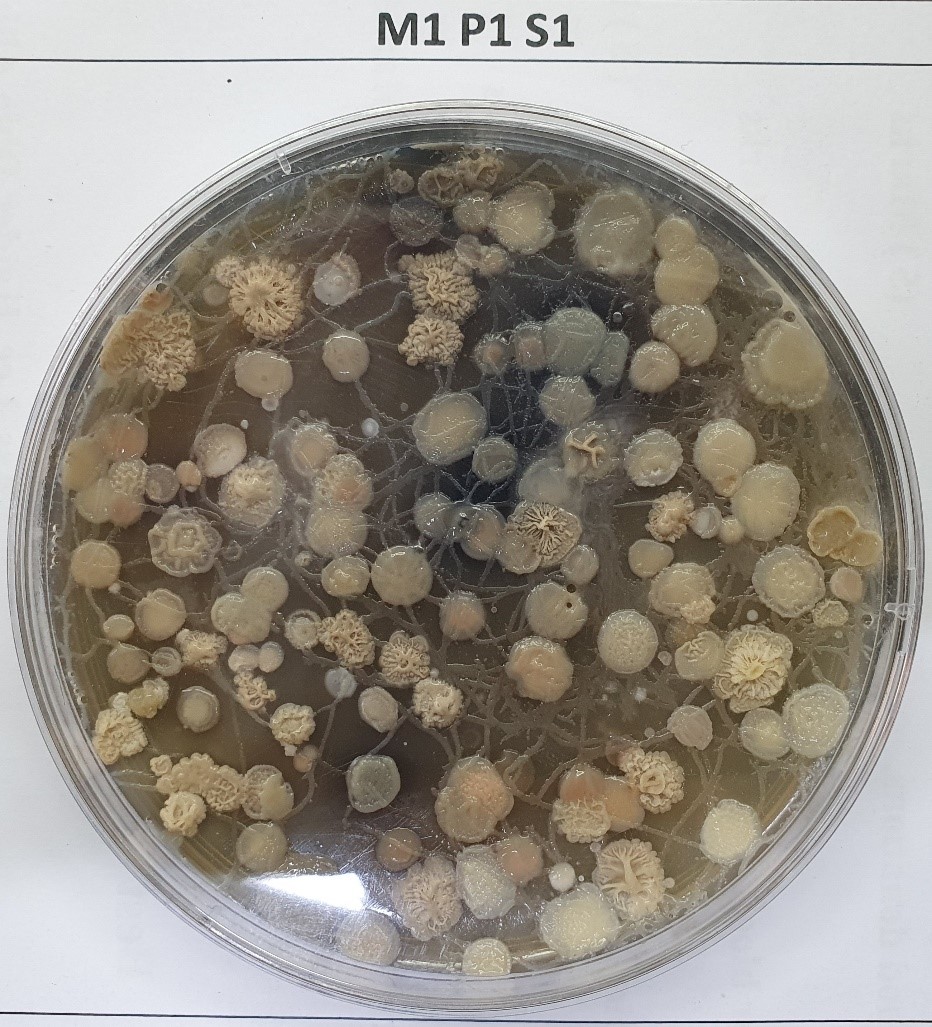 Fig. 2.
Fig. 2.
Culture of Streptomyces isolates with a high ability to utilize different types of hydrocarbons cultivated on ISP2 medium. The biochemical properties of the bacterium showed the ability to grow on Czapeck medium with white substrate mycelia and grey aerial mycelia, a utilizer of glucose, arabinose, xylose, sucrose, and mannose but not rhamnose and inositol. It can produce organic acid and catalase and assimilate urea.
Bacterial isolates selected for this study were identified using 16S RNA primers specifically designed for streptomycetes [32]. The resulting amplicon was 400 bp in size and was sequenced for further confirmation of bacterial identity. The 16S RNA PCR amplicon is shown in Fig. 3.
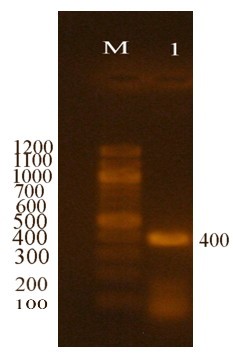 Fig. 3.
Fig. 3.
The 16S RNA amplification of Streptomyces isolates under study. The first lane (M) is a 100 bp DNA marker. The (1) lane is a 16S RNA amplified site.
BLAST results from 16S rRNA showed that the isolate is similar to Streptomyces pini, Streptomyces radiopugnans, Streptomyces niveiscabiei, Streptomyces turgidiscabies, and Streptomyces radicis with query coverage of 100%, e-value 0.15, and per. ident of 100%, respectively. Fig. 4 shows the distance tree of the newly isolated Streptomyces isolates.
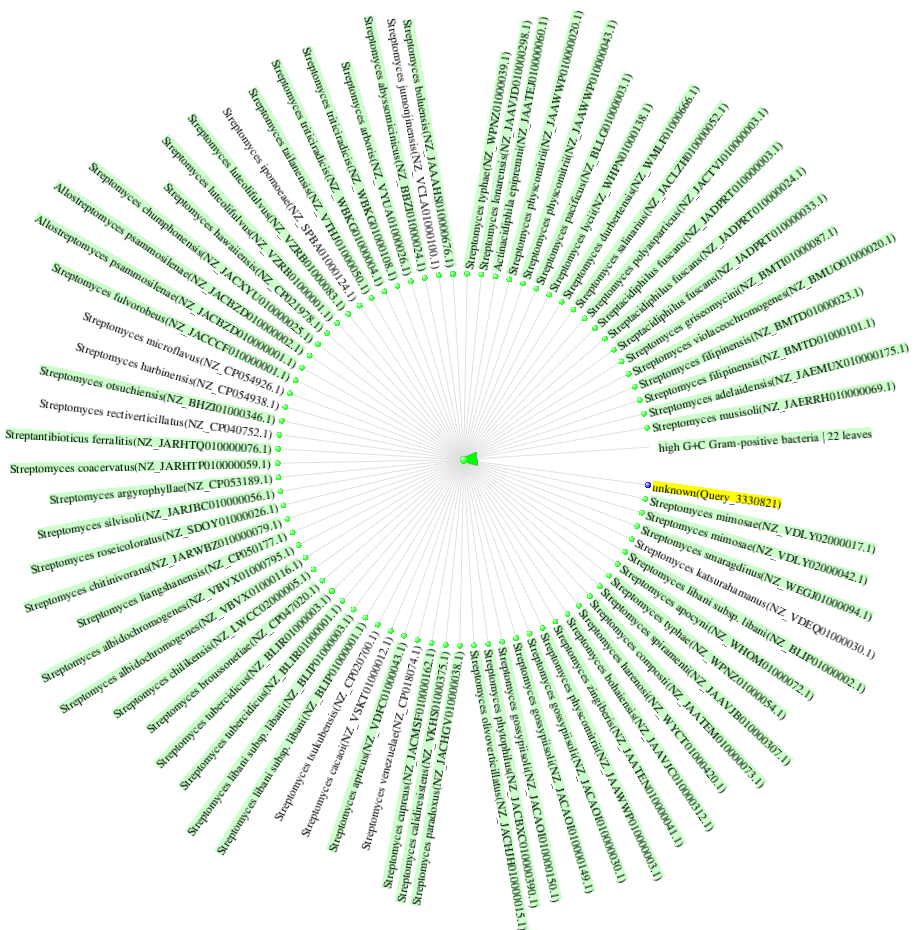 Fig. 4.
Fig. 4.
Distance tree of Polyaromatic hydrocarbons (PAHs) degrading Streptomyces isolate.
The degradative potential of Streptomyces against different hydrocarbons was investigated by cultivation on media supplemented with other types of these compounds. The ability to flourish and produce viable cells was the criteria for utilizing hydrocarbons, as illustrated in Table 2. The percentage of biodegradation of PAHs depends on the number in Table 3.
| Hydrocarbon compound and concentration | Bacterial growth measured by optical density 600 (OD600) |
| Hexadecane (6 mg/L) | ++++ |
| Sodium benzoate (300 mg/L) | ++ |
| Naphthalene (100 mg/L) | +++ |
| Anthracene (1000 mg/L) | +++ |
| Cyclohexane (0.8 v/v) | ++ |
| Phenol (1000 mg/L) | + |
| Tetrahydrofuran (30 mM) | + |
| Benzene (1% v/v) | ++ |
| Toluene (500 mg/L) | ++ |
| Phenanthrene (1000 mg/L) | +++ |
(+) Low growth (OD600 = 0.1–0.3).
(++) Moderate growth (OD600 = 0.4–0.6).
(+++) Good growth (OD600 = 0.7–0.9).
(++++) Heavy growth (OD600 = 1–2).
(Control) Cell cultivated in MSM without hydrocarbons (OD600 = 0.01).
| PAH compound | No. of rings | Abiotic control residual amount µg/mL |
Residual amount 120 days µg/mL |
Biodegradation % |
| Naphthalene | 2 | 49.34 |
5.53 |
85.98 |
| Acenaphthylene | 3 | 49.23 |
4.51 |
87.96 |
| Fluoranthene | 4 | 49.16 |
3.26 |
88.94 |
| Benzo (b) | 5 | 49.40 |
3.01 |
93.49 |
| Benzo (g,h,i) | 6 | 49.33 |
2.35 |
95.47 |
SD, standard deviation.
We found that identifying PAHs for the control sample represented by soils collected from sites under study contained high molecular weight PAHs, which exist in very high percentages, while low molecular weight PAHs existed in traces. For Streptomyces sp., the maximum biodegradation percent of PAHs was 93.49 and 95.47, respectively. This means it can assimilate most crude oil waste in spills. A further test was conducted on high molecular weight PAHs that reached C44 to calculate the range in which our isolate can utilize complex PAHs, as shown in Fig. 5.
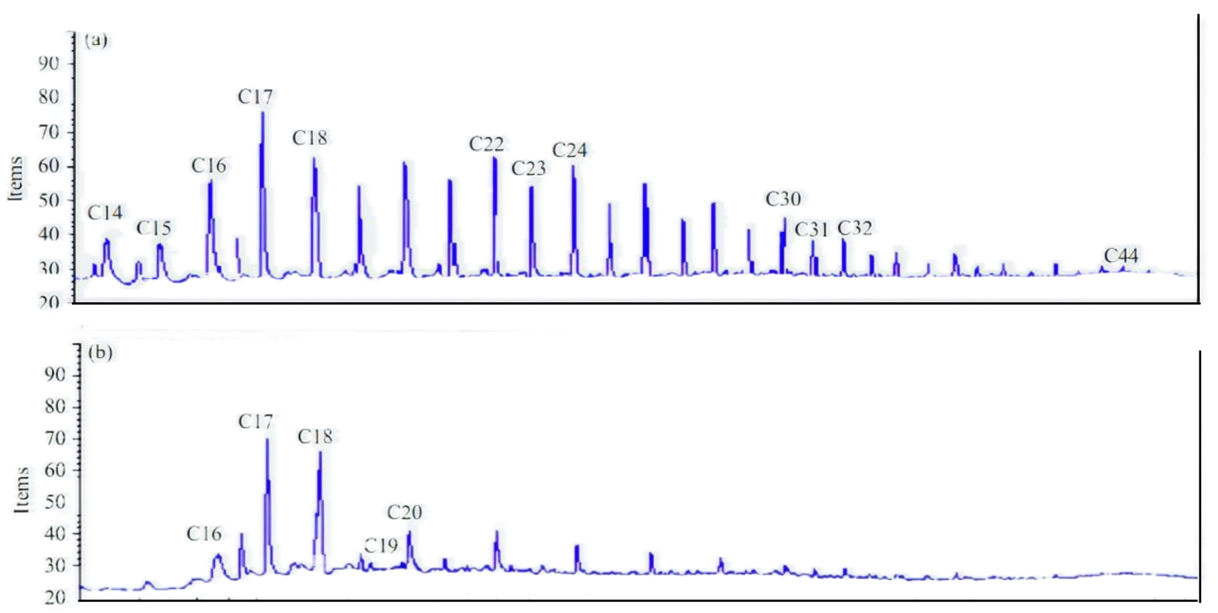 Fig. 5.
Fig. 5.
Gas chromatographs of the high molecular weight PAHs assimilated by Streptomyces sp. (a) Untreated control, (b) Streptomyces sp. The figure shows that the spectrum of complex PAHs began from C5 and declined at C20.
The acid precipitation method was used to isolate and purify biosurfactant-produced Streptomyces. The yield was 1 g/L of biosurfactant. The nature of this substance was found to be lipopeptide, as determined by Fourier transfer infrared spectroscopy and biochemical tests. The surface-active compound was found to be white in color when precipitated and contained peptide bonds in its structure and lipid. The biosurfactants tested were found to be of two types: Polar and non-polar lipids, carbohydrates, and protein. Biosurfactants were produced after 10 days of cultivation on the ISP2 medium and were detected by TLC, as shown in Fig. 6.
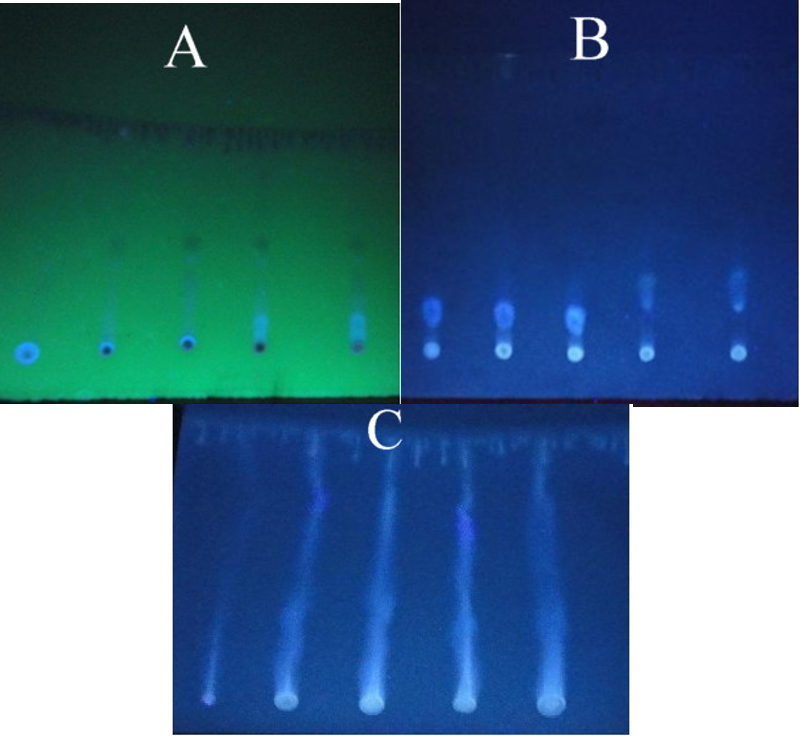 Fig. 6.
Fig. 6.
Resulting biosurfactant from hydrocarbon degradation. (A) shows the polar LPS, (B) the non-polar LPS, and (C) the carbohydrate chromatography. LPS, lipoply saccharide.
Furthermore, the isolate under study was found to have a high emulsification index, the ability to displace oil, and to collapse oil drop when mixed with it. Table 4 illustrates these results.
| Test | |||||
| Emulsification activity | Oil displacement test | Drop collapse test | |||
| + | 65–70% | + | 3.14 cm | + | Flatened |
Fig. 7 shows the bioemulsification of hydrocarbons results.
 Fig. 7.
Fig. 7.
Biosurfactant production test. (A) oil displacement test, (B) drop collapse test in which an oil drop is disperesed on the glass after adding the biosurfactant and (C) emulsification activity. These tests were performed using Streptomyces isolate, which was obtained in this study.
The spectrum from which the Fourier-transform infrared spectroscopy (FTIR) began was 500–700 nm to cover all compounds involved in this detection method. The results showed a widening in specific areas as an indicator that these compounds transformed their chemical structure due to the bacterial effect of assimilation on crude oil contaminants. In Fig. 8, the fraction CH2Cl2 suffered a change in the CH bands at 3383, 3387, and 3414 cm-1, suggesting the presence of an aliphatic chain (CH3CH2)n, while the change in compounds at wavelength 1643 cm-1 suggested a stretching in N–C amid the group.
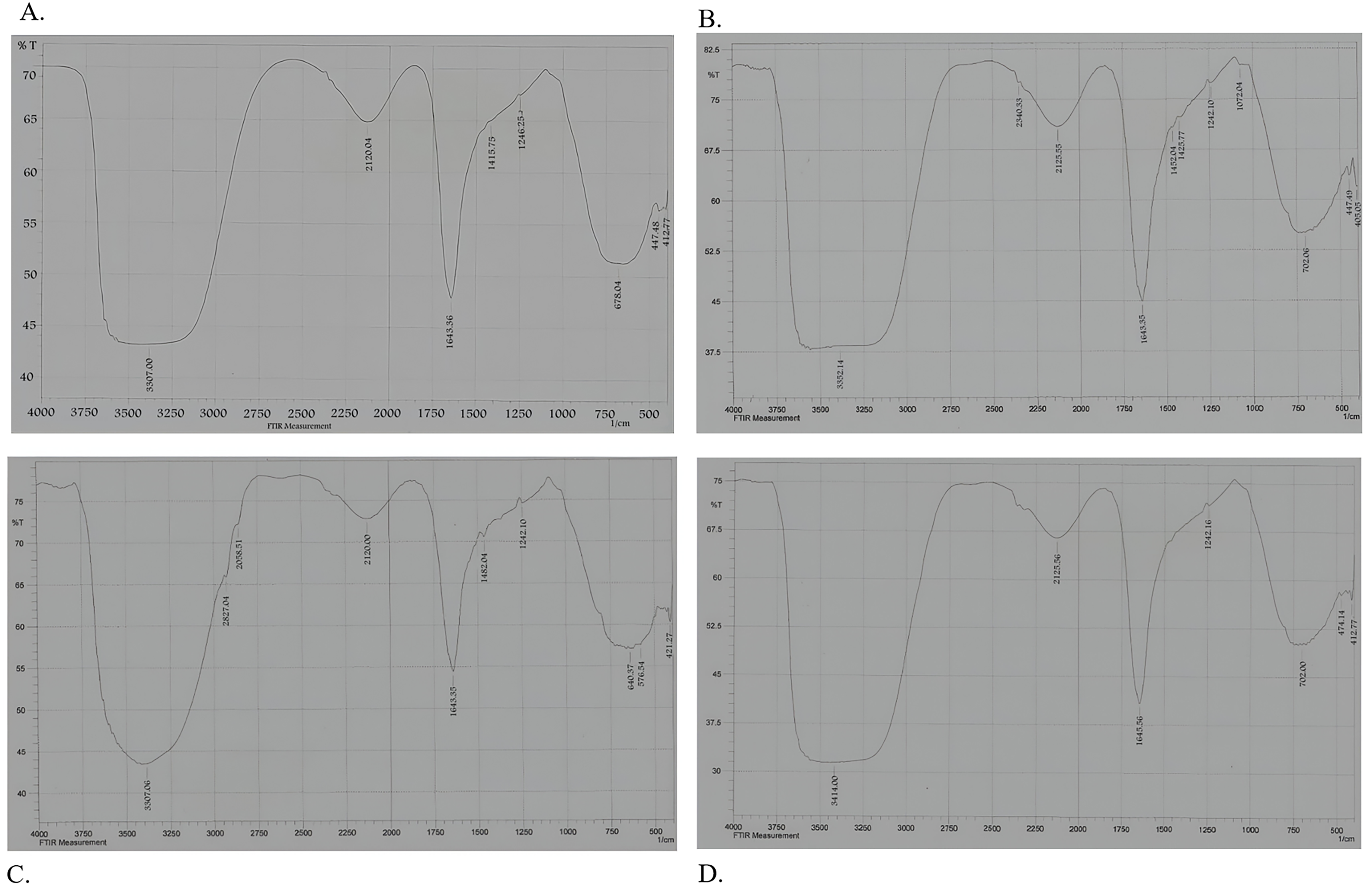 Fig. 8.
Fig. 8.
Spectral graph of crude oil contaminant degradation by Streptomyces isolate. The graph was obtained from Fourier-transform infrared spectroscopy (FTIR) analysis at a wavelength range of 500–700. (A) Streptomyces isolates grown on crude oil as a source of carbon, (B) Streptomyces isolate grown on hexadecane as a source of carbon, (C) Streptomyces isolate grown on diesel, and (D) Streptomyces isolate grown on naphthalene.
Iraq is considered one of the leading countries in oil production. Almost the entire economy and its revenues depend on processing, exporting, and trading oil with nations worldwide. With abundant oil wells and ongoing excavation, contamination with oil spills, leaks, and transportation occurs at a higher rate. During the last decades, Iraq faced an embargo, unprecedented terrorist attacks, and political disputes that influenced the oil industry. Owing to a lack of containment protocols and low consciousness about the risk of spreading PAHs in soil, rivers, sewers, and around storage facilities resulted in ecological problems that may affect many coming generations and should be addressed at the highest level of government. In many research facilities, using natural methods was the means to reduce this threat. Using plants that may grow in contaminated soils and naturally occurring microorganisms is considered a better way than using chemicals or burying contaminated areas that might reach underground water, eventually causing more risk and damage. This work focused on the spore-forming, widely abundant, nonpathogenic, and enzymatically rich Streptomyces that constitute a major part of the soil and can be isolated from almost any site.
Three sites were selected to isolate the bacteria under study: Al-Begei refinery, Al-Latifia storage facility, and Basra, where oil wells are. With increased activity and contamination in these sites, they were considered ground zero to begin this research. In addition, the difference in soil texture (Table 2) may expand the search for efficient streptomycetes that may be used for bioremediation. Differences in salinity, pH, and soil particles can be attributed to geographic factors. Most of the lands north of Iraq (Al-Begei) are characterized by water abundance, high clay content, low salinity, and elevated pH due to agricultural activities that differ from the mid-Iraq provinces where precipitation of both Tigris and Euphrates plays the main role in soil composition. However, areas in the south of Iraq are characterized by high salinity and reduced pH due to low water resources, sands from the western region, and limited farming and agriculture activities. With such variable factors, the chance to isolate different streptomycetes was high. Since the Al-Begei region was characterized by high organic content and favorable conditions to enumerate bacterial communities, isolating active Streptomyces was possible. The physicochemical properties of soil can be altered with oil contamination, causing dramatic effects on soil fertility, biodistribution and divergence, and plant life. Microorganisms are the primary members of soil and play a key role in the decomposition of organic pollutants, meaning bioremediation is an effective technology for petroleum-polluted soil [33]. Streptomycetes are major components of soil microbes, and their ability to utilize most organic matters, including hydrocarbons and nonpathogenic criteria, makes them perfect candidates for the bioremediation process. With a wide range of hydrocarbons tested for degradation, it might indicate that the isolated Streptomyces harbor highly efficient enzymes dedicated to this purpose. The emulsification activity determines the productivity [34], which in previous reports was 100% [35]. Our bacterial isolate reached 75% emulsification capability, which indicates high enzymatic activity. Furthermore, FTIR spectral data and TLC results demonstrated the ability of Streptomyces to produce streptofactin, an extracellular compound with different types of peptides [36, 37]. Depending on the IR spectra and previous reports, detailed information about the already known biosurfactants was studied by several investigators, which have demonstrated the lipopeptide nature of these compounds as our results indicated [38, 39, 40], which is the same that was found in Bacillus [41, 42] and Pseudomonas [43].
Our study showed the ability of Streptomyces isolates to degrade complex hydrocarbons (hexa, penta, and tetra) ring compounds with the production of a simpler and emulsified compound that are easier to be reformulated to nontoxic compounds (Fig. 6) or lost the characteristics of toxicity [44]. This study follows with other studies that showed Streptomyces as a reliable source of biosurfactant and retains the ability to degrade oil spills and other hydrocarbon pollutants [45, 46]. These compounds may be the substrate for the growth of other microorganisms that lead to an enriched microbial environment in treated soils. The ability to reduce surface tension through biosurfactant production will eventually help enrich soils and allow water and nutrients to spread and go through soils, turning contaminated soils into rich farming soils.
In oil production countries, oil spills and contamination with oil derivatives have become major problems that must be addressed. In Iraq, one of the leading countries in oil production, oil storage facilities, production wells, and transportation along the country, facing sabotage and terrorist attacks for years, had increased contamination with PAHs that sometimes extended to river banks, farming land, and was detected in underground water urged the need to find the mean to overcome this increasing problem. Since chemical treatment and burial of contaminated areas is not an effective method, using naturally occurring microorganisms presents a reasonable solution. The use of streptomycetes may be a more favorable solution since it is a nonpathogenic bacteria, naturally occurs in soil, has a high tolerance for toxic substances, is rich with enzymes and secondary metabolites, and maintains the texture of the soil.
Data, samples, and genomic materials used during this study are available and stored at the Biotechnology Research Center, Al-Nahrain University.
GHA handled microbial isolation, identification, cultivation, and preservation ATD conducted the planning of the research, RAK performed bacterial isolation and preservation, RNA performed PCR amplification, sequencing, data analysis, correspondence, and writing the paper, RSR data curation, RAK data recording, and SST preservation of bacterial identification. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity. All authors read and approved the final manuscript. All authors contributed to editorial changes in the manuscript.
This research project was approved by the Ethic Committee at Biotechnology Research Center/Al-Nahrain University with ref. no. E. B. 8- 2.1.23.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








