1 Food Science Department, Agriculture College, Basrah University, 61001 Basrah, Iraq
Abstract
Flavonoids occur naturally in different types of fruits and vegetables, including tea, cabbage, cauliflower, elderberries, cranberries, red apples, lettuce, pears, spinach, green hot peppers, white and red onions, kale, blueberries, and nuts. Among these flavonoids is quercetin, a potent natural antioxidant and cytotoxic substance with a number of therapeutic functions. Nowadays, quercetin is a common ingredient in many nutraceutical and cosmeceutical products due to its antioxidant properties. Its antibacterial effects and possible action mechanisms have been explored in many studies. From these, it has been established that quercetin stops the activity of numerous Gram-negative and -positive bacteria, fungi, and viruses. This review clarifies the plant sources and extraction methods of quercetin, as well as its medicinal applications as an antibacterial, antifungal, antiviral, and antioxidant agent, with a particular emphasis on the underlying mechanisms of its biological activity. The mechanism of its antimicrobial effect involves damaging the cell membrane—e.g., by changing its permeability, preventing biofilm formation, reducing the mitochondrial expression of virulence factors, and inhibiting protein and nucleic-acid synthesis. Moreover, quercetin has been shown to impede the activity of a variety of drug-resistant bacterial strains, pointing to the possibility of using it as a strong antimicrobial substance against such strains. In addition, it has occasionally been demonstrated that specific structural alterations to quercetin can increase its antibacterial action in comparison to the parent molecule. Overall, this review synthesizes our understanding of the mode of action of quercetin and its prospects for use as a therapeutic material.
Keywords
- quercetin
- antimicrobial
- antifungal
- antiviral
- antioxidant
- anticancer
- anti-inflammatory
Among the many, widely distributed secondary metabolites in plants that are crucial to their growth and reproduction are phenolic compounds. This broad class is comprised of approximately 8000 organic compounds, ranging from simple phenolics like phenolic acids to complex compounds like tannins [1, 2]. Phenolic compounds can be further classified into multiple groups, one of which is the flavonoids. These compounds can only be produced by plants. They perform numerous tasks connected to the physiology of growth in addition to characterizing plants’ flavors and colors. The composition of these substances allows flavonoids to be classified into six primary groups: anthocyanidins, isoflavones, flavones, flavonols, and flavanones [3, 4]. The flavonols are among the most well-known flavonoids, and occur naturally in fruits and vegetables [5]. This subgroup includes quercetin (3,5,7,3′,4′-pentahydroxyfavone), which can be found in apples, red grape, kales, cherries, onions, broccoli, berries, edible seeds, and tea, among others [6]. One of the most widely used crops for both food and medicine, onion (Allium cepa) is among the most abundant source of quercetin. Fig. 1 shows the chemical formula of quercetin, which has the molecular formula C15H10O7. It is a yellow-tinted compound, and its name derives from the Latin word “quercetum”. Quercetin dissolves readily in lipids and alcohol, but is insoluble in cold water and has low solubility in hot water.
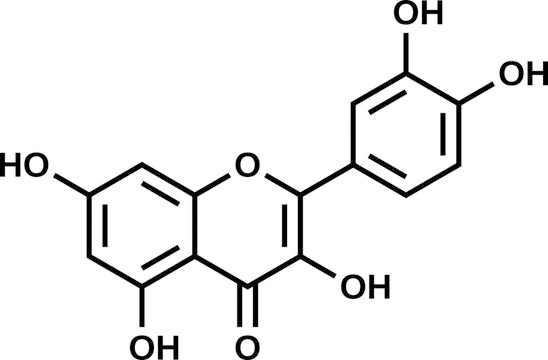 Fig. 1.
Fig. 1.
Molecular structure of quercetin.
Free radicals interact with other substances very rapidly in order to entrap their electron and stabilize. The molecule under attack loses an electron and transforms into a free radical, starting a domino effect that damages biological cells [7]. According to the literature, the hydroxyl groups at the A and B rings 3, 5, 7, 3′, and 4′, the double bond spanning the third and second carbons, and the carbonyl group on the fourth carbon play major roles in quercetin’s antioxidant characteristics [8]. Several researchers have explored quercetin’s antibacterial properties in depth and have proposed it as a potential treatment against a variety of harmful microbes [9]. Moreover, because bacterial diseases are commonly treated with antibiotics, multidrug resistance (MDR) in bacteria has grown to be a pressing issue on a global scale. The pharmaceutical industry has faced substantial difficulties in treating MDR strains [10].
The suppression of
With severely acute and chronic effects, diabetes—the prevalence of which is rising daily—has emerged as a serious global issue. Among its long-term consequences are retinopathy, neuropathy, nephropathy, and podiatric ailments. Furthermore, hypoglycemia, hyperglycemia, and diabetic ketoacidosis are acute effects that require immediate medical attention [16]. Quercetin has been shown to suppress apoptosis and cell growth by blocking enzymes such as protein kinases and cyclooxygenases. Moreover, it was reported that quercetin enhances insulin release, normalizes blood sugar, and encourages beta cell regeneration in pancreatic islets [17].
In cancerous cells, such as those found in breast, lung, ovarian, colorectal, gastric, and hepatic malignancies, quercetin has demonstrated strong inhibitory actions [18]. Its anticancer properties have been shown to occur through a number of mechanisms, including cell death, angiogenesis inhibition, P-gp channel inhibition, reduced oncogene expression, and signaling pathway regulation [19]. Furthermore, inflammation worsens the disease profile in several conditions, such as rheumatoid arthritis, ulcers, and so forth, because it releases inflammatory mediators. Quercetin’s ability to reduce inflammation was found to be mediated by blocking ATP, proinflammatory cytokine, and the binding sites of nuclear factor kappa B (IkBa) [20]. Another key therapeutic characteristic is quercetin’s capacity to bind with various metal ions, such as Fe2+, Fe3+, Cu2+, and Ni2+. The resulting quercetin metal complexes have displayed good activity against inflammation, cancer, ulcer, microbes, and Alzheimer’s disease [21].
Due to the numerous demonstrated benefits of quercetin, it is already being employed in biomedicine for its pharmacological qualities. However, it also has several drawbacks, including low bioavailability, a hydrophobic nature, minimal solubility, and minimal permeability, which make it difficult to be absorbed. To increase quercetin bioavailability and solubility, it is commonly complexed with biodegradable polymers employing chitosan [22]. Recently, the United States Food and Drug Administration (FDA) granted quercetin Generally Recognized as Safe (GRAS) certification [23]. We have outlined the antibacterial, antifungal, antiviral, and antioxidant properties of quercetin in this review, paying particular attention to the underlying molecular concepts. In order to better understand how quercetin prevents microbial infections and how it might be used as a clinically useful antimicrobial drug, future researchers will benefit from reading this review.
Quercetin is present in vegetables and fruits such as red and white onions (22 mg/100 g), elderberries (26.77 mg/100 g), cranberries (14 mg/100 g), green hot peppers (2 mg/100 g), kale (22.58 mg/100 g), blueberries (14 mg/100 g), red apples (5 mg/100 g), lettuce (31 mg/100 g), pears (4.24 mg/100 g), spinach (27.2 mg/100 g), tea (2.63 mg/100 g), cabbage (0.28 mg/100 g), and cauliflower (0.54 mg/100 g) [24]; Table 1 (Ref. [25]) shows other plant sources of quercetin. The compound is frequently utilized as a dietary supplement due to its diverse range of pharmacological properties. From the intake of fruit, vegetables, and tea, varying between 50 to 800 mg/day in several nations, varying daily dietary intakes were observed. According to reports, the average daily intake of quercetin was 18, 16.2, 18.48, and 9.75 mg in China, Japan, Spain, and the USA, respectively [24]. Fig. 2 illustrates the various sources and biological properties of quercetin.
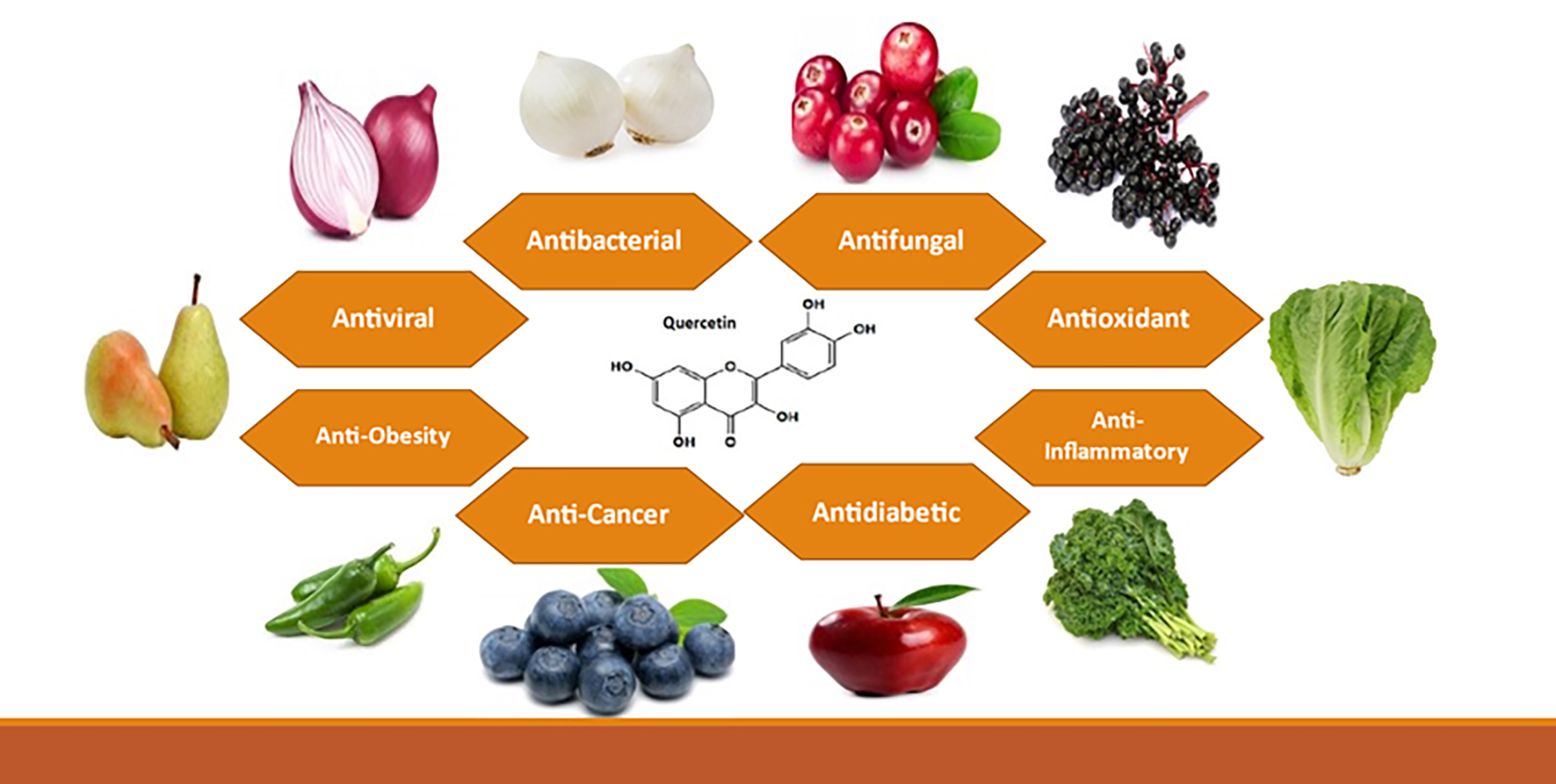 Fig. 2.
Fig. 2.
Sources and biological properties of quercetin.
| Natural resource | Quercetin content | Natural resource | Quercetin content |
|---|---|---|---|
| Apples | 4.01 | Asparagus | 14.0 |
| Broccoli | 13.7 | Chili pepper | 32.6 |
| Kale | 22.6 | Leeks | 0.9 |
| Lettuce | 14.7 | Onions | 45.0 |
| Spinach | 27.2 | Chives | 10.4 |
| Dill | 79.0 | Fennel leaves | 46.8 |
| Oregano | 42.0 | Blueberry | 14.6 |
| Cherry | 17.4 | Cranberry | 25.0 |
Many bacterial strains, especially those that impact the gastrointestinal, respiratory, urinary, and integumentary systems, are sensitive to quercetin’s antibacterial properties. This compound’s capacity to inhibit microorganisms has been connected to its solubility [26] and how it interacts with the membrane of the bacterial cell [27], which is mostly influenced by the quantity of hydroxyl groups in quercetin [9]. In general, Gram-negative bacteria are more resistant to quercetin’s antibacterial action than Gram-positive bacteria [28]. The variation in cell membrane structure between these two bacterial types may be indirectly responsible for their difference in susceptibility to quercetin. However, some quercetin derivatives have displayed higher activity against Gram-negative than Gram-positive bacteria [9]. Quercetin’s solubility might be changed by phosphorylation and sulfation at various hydroxyl groups, which would change its effectiveness against specific bacteria [9, 29].
In an intriguing study conducted in India, Sarangapani and Jayachitra [30] employed a strain of the fungus Aspergillus niger to convert rutin to quercetin at a faster pace and with less-harmful byproducts. The generated quercetin was tested for a number of properties, including its ability to fight off Staphylococcus aureus, pseudomonas aeruginosa, and Escherichia coli. The three bacterial strains’ biofilms were disrupted and the microorganisms died as a result of the production of reactive oxygen species (ROS) from quercetin at their surface. Quercetin was found to have a wider zone of inhibition when used against S. aureus [30].
Many types of bacteria have shown susceptibility to the antibacterial activity of quercetin and its derivatives at various minimum inhibitory concentrations (MICs). Quercetin was found to inhibit the growth of six out of eleven examined oral harmful bacteria, which included Streptococcus mutans, S. sobrinus, Lactobacillus acidophilus, S. sanguis, Actinobacillus actinomycetemcomitans, and Prevotella intermedia, with MICs ranging from 1 to 8 mg/mL [31]. Its antagonistic effect on Staphylococcus aureus and Pseudomonas aeruginosa was also reported [32], with a MIC of 20 mcg/mL. Furthermore, quercetin demonstrated antimicrobial activities against Shigella sonnei and Micrococcus luteus at MIC of 25 mcg/mL [33]. Quercetin has also been shown to have antagonistic effects on a variety of Gram-positive bacteria, including methicillin-resistant Staphylococcus aureus, methicillin-sensitive Staphylococcus aureus, and standard Enterococcus [34].
Additionally, quercetin has been found to inhibit bacterial growth by working in synergy with other therapeutic substances and antibiotics [35, 36]. For example, in combination with many other antibiotics, quercetin showed its antibacterial efficacy against methicillin-resistant S. aureus [37].
Quercetin’s antibacterial capabilities against resistant Staphylococcus saprophyticus and S. aureus were reported by da Costa et al. [38], who observed its antibacterial action against methicillin-susceptible S. aureus (MSSA) (MIC = 250 µg/mL), methicillin-resistant S. aureus (MIC = 500 g/mL), vancomycin-intermediate S. aureus (MIC = 125 and 150 µg/mL), and vancomycin-resistant S. aureus.
The antibacterial characteristic profile of thirteen flavonoids was detailed by Adamczak et al. [39], namely isoorientin, apigenin, rutin, naringin, chrysin, flavones, isovitexin, kaempferol, luteolin, orientin, quercetin, vitexin, and vitexin 2-o-rhamnoside, together with six organic acids: quinic, malic, salicylic, citric, chlorogenic, and rosmarinic acid. Four clinical strains, comprising the Gram-positive bacteria Enterococcus faecalis and S. aureus and Gram-negative bacteria E. coli and P. aeruginosa, were employed to calculate the MIC values for each tested substance. Overall, all tested compounds showed stronger antibacterial efficacy against Gram-negative than Gram-positive bacteria [39].
Quercetin has been reported to have the ability to combat Aspergillus fumigatus (at 16–64 µM) [36] and Aspergillus niger [40]; however its antifungal actions are not as well studied as those against bacteria.
Gao et al. [41] demonstrated the treatment of vulvovaginal candidiasis (VVC) with fluconazole and quercetin. Among patients with VVC, 15 clinical strains of Candida albicans were identified. The study verified the quercetin and fluconazole combination’s synergistic inhibition of antifungal biofilms at dosages of 64 and 128 µg/mL. By using 5 and 20 mg/kg of quercetin, the fungal burden in the vaginal mucosa of mice with VVC was reduced [41]. Also in Candida, quercetin and kaempferol MIC was evaluated against C. metapsilosis, C. orthopsilosis, and C. parapsilosis strains in a study by Rocha et al. [42]. The quercetin and kaempferol were found to reduce the metabolism rate and activity of all three fungi isolates, with quercetin’s MIC being between 0.5 and 16 µg/mL. This demonstrated quercetin’s suitability as an antifungal agent [42]. Moreover, Saccharomyces cerevisiae, C. Albicans, and other Candida spp. were subjected to quercetin’s antifungal action [34].
The survival strategies of Aspergillus flavus throughout the germination phase were described in the results of an Indian study by Shishodia et al. [43], who investigated quercetin’s impact on the Aspergillus flavus signaling pathways during germination using Scanning Electron Microscopy (SEM). Real-time quantitative Polymerase Chain Reaction (PCR) was used to detect a significant increase in calcium signaling pathways and heat shock proteins at 4- and 7-h intervals in response to quercetin. The fungal colonies that received quercetin treatment showed higher levels of the genes that code for calcium kinase, cAMP, Rhogdp, Plc, and Pkc. Hence, the results supported quercetin’s potential as an antifungal [43].
Quercetin-capped gold nanoparticles with antifungal properties were reported by Milanezi et al. [44]. These were made using quercetin, aqueous sodium citrate solution, and trichlorogold-hydrochloric acid. Fourier-transform infrared spectroscopy (FTIR), Ultraviolet (UV), and Transmission electron microscopy (TEM) examination were used to confirm the presence of the nanoparticles, after which these nanoparticles were tested for antifungal activity. Significant activity against fungus was detected against Aspergillus fumigatus at doses between 0.1 and 0.5 mg/mL [44].
Quercetin has been found to increase the effectiveness of other medications against resistant fungi [41, 45]. The antifungal potential of quercetin against Cryptococcus neoformans and Candida albicans was deemed minimal by Oliveira et al. [46]. However, quercetin greatly increased amphotericin B’s antifungal activity against C. neoformans strains when paired with it, as well as significantly decreasing amphotericin B’s side effects, which may have been related to the antioxidant properties of quercetin [46]. Similarly, when only quercetin was applied to treat fluconazole-resistant C. albicans, there was essentially no cell death, but when quercetin and fluconazole were combined, there was a considerable increase in cell death [41].
Long-term research has been conducted on the activity of quercetin and its derivatives against viruses. Many viruses, including the poliovirus, Sindbis virus, respiratory viruses, and the human immunodeficiency virus (HIV) [15, 27], and Mayaro virus [47], were susceptible to quercetin and its derivatives when tested in vitro.
Those with modest COVID-19 symptoms recovered more rapidly from the virus when medicated with quercetin phytosome, according to a human trial [48]. Due to the lack of specific antiviral medications available for the treatment of dengue virus infection (DENV), quercetin was studied for its ability to fight this virus. The DENV strain-2 New Guinea C-infected human cell line Huh 7 it-1 was used for the investigation. The quercetin IC50 and CC50 values were 18.41 g/mL and 217.113 g/mL, respectively, according to the MTT test that was utilized to determine these values. The findings validated quercetin’s potential as a DENV-2 antiviral medication [49]. In mice afflicted with the Mengo virus, oral administration of quercetin resulted in a certain degree of defense against fatal infections in vivo [15].
In Spain, Rojas et al. [50] investigated quercetin’s anti-hepatitis C (HCV) virus properties. Quercetin demonstrated antiviral effects in primary hepatocytes (PHH) and Huh-7.5 cells infected with HCVcc at different phases of the HCV life cycle. Both cell types experienced significant viral load reductions of up to 85% in Huh7.5 and 92% in PHH. When virus particles were treated with quercetin directly, there was a 65% decrease in their ability to spread disease. Curiously, quercetin also prevented the HCV core protein from localizing to lipid beads’ surface and from upregulating diacylglycerol acyltransferase (DGAT). Consequently, it was found that the antiviral actions of quercetin against HCV were host-mediated [50].
The antiviral efficacy of a quercetin derivative containing a glucoside molecule against Zika virus strains was examined in vitro and in vivo. Using Vero cells with the PLCal-ZV strain, an in vitro assay of quercetin3—O-D-glucoside (Q3G) was carried out. After exposing the infected Vero cells to various concentrations from 0.67 mol/L to 10.76 mol/L, viral RNA was isolated and subjected to real-time quantitative Polymerase Chain Reaction qPCR analysis after 2 and 4 days. The cytopathic effects of Q3G were seen at EC50 and EC90 concentrations of 1.2–1.3 mol/L and 1.5 mol/L, respectively. Immunodeficient mice lacking the type I interferon receptor underwent the in vivo assay while being administered the strain PRVABC59. Q3G and the Zika virus strain were administered intraperitoneally for up to 7 days at 50 mg/kg each. According to the findings, animals fed Q3G had higher survival rates and lower weight loss [51].
Based on quercetin’s ability to fight viruses, an innovative pharmacological dosing technique comprising quercetin and lecithin quercetin phytosome (QP) was developed in Italy and investigated on patients with SARS-CoV-2. An open-label, random, systematic clinical study was carried out over a period of two weeks. Forty-two patients with COVID-19 were enrolled; 21 of them received standard care (SC), and the other 21 received QP. Findings revealed that sixteen patients within the QP group had a completely negative profile after one week, with twelve individuals exhibiting completely reduced symptoms. More than two weeks passed before 17 patients in the SC group delivered a COVID-19 negative result; after the third week, one patient had a negative result, and one had a positive result up until day 20, when they died. As a result, it was determined that QP shortened the period of time for a negative test result to be obtained, in addition to reducing COVID-19 symptoms and intensity [48]. Similarly, in individuals with early respiratory tract infections, particularly those with COVID-19, quercetin, when combined with vitamin C, assisted in both prevention and treatment in vivo [15, 52].
Influenza A virus infections regularly cause scourges and pandemics with high
mortality rates and devastation. The glycoprotein hemagglutinin plays a crucial
function during infection, making it a possible target of anti-flu medications.
Quercetin’s antiviral activity was examined in three strains of Influenza
A—A/FM-1/47/1 H1N1, A/Aichi/2/68 H3N2, and A/Puerto Rico/8/34 H1N1—resulting
in IC50 values of 7.756
In other studies, the antiviral effectiveness of acyclovir against herpes viruses was improved in vitro by quercetin [47], while quercetin’s antiviral properties were also found to actively combat a variety of serotypes of the poliovirus (type 1 Sabin), coxsackievirus (A21 and B1), rhinovirus, and echovirus (types 7, 11, 12, and 19) [54].
Reactive oxygen species (ROS) and pro-inflammatory cytokines lead to oxidative stress, one of the earliest processes connected to gout and its comorbidities [55]. At the two last steps of the purine metabolic pathway, xanthine oxidoreductase (XOR), particularly the xanthine oxidase (XO) form, oxidizes hypoxanthine to yield xanthine, which is then transformed into urate, releasing H2O2 and O2.
Quercetin is well known as a strong antioxidant substance. Studies have shown strong in vitro antioxidant activity in a variety of tests, including those using OH radicals, 2,2-Diphenyl-1-picrylhydrazyl (DPPH), and Ferric Reducing Antioxidant Power (FRAP) [56, 57]. The existence of a 30, 40-catechol configuration in B-ring, a 4-carbonyl group conjugated to a double bond between C-2 and C-3 in the C-ring and a free OH group at C-3 are some of the structural components of quercetin that are thought to support its antioxidant effects [58]. Furthermore, there are indications that quercetin improves the antioxidant system of defense. In both normal and hyperuricemic rats, using quercetin at 5 mg/kg substantially elevated the total antioxidant capacity of the serum (FRAP value) and greatly decreased the increased levels of malondialdehyde in hyperuricemic rats. This was accomplished by its ability to inhibit the liver enzymes XO and XDH, which produce uric acid, and thus significantly decrease the serum uric acid levels of hyperuricemic rats [59]. In mice with galactose-induced oxidative stress, the serum total antioxidant capacity increased after treatment with 0.3 mmol/kg quercetin, superoxide dismutase, catalase, and glutathione peroxidase levels along with the increase in the mRNA expression levels of these antioxidant enzymes. In addition, the quercetin also reduced serum levels of malondialdehyde and nitric oxide [57]. Another investigation into the inflammation caused by monosodium urate crystals in rats found that quercetin at 200 and 400 mg/kg doses reduced malondialdehyde levels while increasing the activity of antioxidant enzymes such as glutathione peroxidase, catalase, and superoxide dismutase [60].
Nanotechnology has made it feasible to handle substances that are smaller than 100 nm, which helps them be absorbed by cells and work well in small amounts. Applications of nanotechnology in different fields have grown recently [61, 62], particularly in aquaculture [63, 64, 65], including in health management via nutrient and vaccine supply, fish breeding, pollution cleanup, and water purification [63, 66]. Despite its many beneficial effects, the volatility and limited absorption of quercetin limit its use. To overcome these limitations, the beneficial properties and increased bioavailability of quercetin nanoparticles have been explored [67].
The drawbacks of quercetin include its low solubility in water (about 60 mg/L), poor intestinal permeability, and physiological instability [68, 69, 70]. Several studies have made an effort to overcome these restrictions, as doing so is essential to providing safe and efficient solutions to the evaluated formulations. Consequently, a variety of nanostructures have been created, such as liposomes, cyclodextrin, mesoporous silica nanoparticles, chitosan nanoparticles, PLGA polymer nanoparticles [71], biogenic nanomaterials [72], and other technologies. The size and shape of the nanoparticles affect the solubility of bioactive compounds; for example, particles of smaller size with larger surface area lead to improved dissolution rates [72, 73, 74].
Aghapour et al. [75] used a wet impregnation technique with ultrasonic assistance to create quercetin-conjugated silica nanoparticles. Their objective was to assess the impact on the induction of apoptosis and growth inhibition in MCF-7 cells, a cell line used to treat breast cancer. The authors found that the system contained 45% quantifiable quercetin and that the spherical nanoparticles had an average size of 84 nm. Their subsequent biological experiments showed that 100 µM quercetin and 10 µM quercetin nanoparticles were efficient against breast cancer cells; by causing apoptosis at lower doses than the isolated drug, the method prevented cell division. Similarly, to test quercetin chitosan nanoparticles’ efficacy in cancer treatment, Baksi et al. [76] employed an ionic gelling technique and conducted controlled-release research, finding that the system was highly successful in raising the bioavailability of compounds that were poorly soluble.
Studies conducted both in vitro and in vivo were used to
investigate the potential of free material and nanoparticles. These found that
the release of flavonoids occurred over a 12-hour period at pH 7.4 (67.28%), and
that the IC50 of the nanoparticles was substantially lower than that of
quercetin alone (p
Tumor growth is largely influenced by the processes of mitosis and cell proliferation; this is why two successful cancer treatments involve blocking cell proliferation and stopping the cell cycle. When quercetin was applied to PC-3 human prostate cancer cells for 24 and 48 hours at different concentrations (50–200 µM), it was observed that the vitality of the cells gradually declined. Additionally, in a dose-dependent manner, quercetin therapy boosted the G2/M phase population in PC-3 and LNCaP cells as well as the S phase population in PC-3 cells [77].
Quercetin derivatives have been identified in several patents as a viable candidate for a variety of medicinal uses, including as anti-aging, anti-inflammatory, anticoagulant, and antioxidant agents [78]. A quercetin patent was filed based on new quercetin derivatives that were proposed as anticancer agents and could be used to make medications that reduce the chance of getting cancer. Quercetin derivatives with hydrogen, benzyl, or substituted benzyl structures have demonstrated cytotoxic effects on cancer cell lines, including those from prostate, ovary, and lung cancers [79]. Furthermore, a derivative of quercetin was produced from sinapinic acid, and metal salt and quercetin showed anticancer activity against human myeloid leukemia cells and human promyelocytic leukemia HL-60 cells [80]. Another chemical was created when quercetin and p-coumaric acid reacted; when tested on SCC-4 human tongue squamous cell carcinoma cells, the substance demonstrated anticancer activity [81, 82].
In a randomized, double-blind, 10-week clinical experiment, 72 women received an
individual daily dosage of 500 mg quercetin. Their high-density lipoprotein
cholesterol (HDL-C), systolic blood pressure (SBP), interleukin-6 (IL-6), and
tumor necrosis factor-
Another randomized, double-blind, placebo-controlled clinical trial explored
quercetin’s effect on 50 women with rheumatoid arthritis. For eight weeks, they
were divided into two groups: one received a placebo and the other received
quercetin (500 mg/day). It was shown that quercetin substantially lowered
high-sensitivity tumor necrosis factor-
The effects of quercetin (500 and 1000 mg/day) in comparison to placebo were assessed in 1002 patients with upper respiratory tract infection (URTI) during the course of a 12-week randomized, double-blind, placebo-controlled experiment. The study demonstrated that quercetin had no discernible impact on URTI rates when compared to the placebo [86]. Additionally, a randomized controlled clinical study with an open-label design was carried out to assess the efficacy of quercetin and curcumin at daily doses of 260 mg and 168 mg, respectively, against COVID-19. Twenty-five patients with early to mid-stage COVID-19 received the medication twice a day at home. The results showed that the treated patients’ problems resolved rapidly [87]. Furthermore, due to their multi-target binding capabilities with many SARS-CoV-2 viral proteins, quercetin and some quercetin derivatives are essential for disrupting viral replication. To boost therapeutic efficacy, quercetin and its derivatives can be employed alone or in conjunction with other treatments to target the aforementioned target proteins and impede viral replication and transcription [88].
To elucidate how quercetin affects patients with Type 2 diabetes mellitus postprandial hyperglycemia, a randomized, blind, cross-over study was conducted. It found that patients with type 2 diabetes might successfully lower their postprandial hyperglycemia when taking 400 mg of quercetin [89]. A different randomized, double-blind, placebo-controlled trial recruited overweight participants, who were divided into two groups—the placebo group, which consisted of six men and 30 women, and the 100 mg quercetin group, which consisted of five men and 30 women—for a period of 12 weeks. Both the control and treatment groups had a reduction in blood glucose and leptin levels, and quercetin significantly lowered the participants’ weight and body fat percentage [90].
Nineteen participants with prehypertension and 22 participants with hypertension participated in a double-blind, placebo-controlled, cross-over trial to investigate the effects of 730 mg quercetin over a 28-day period. It was demonstrated that while oxidative stress was not affected, there was a significant drop in both the systolic and diastolic blood pressures in the hypertension participants following quercetin supplementation [91]. In contrast, blood pressure did not alter in the prehypertensive subjects.
The use of natural antimicrobials for food preservation is a recent trend followed by both consumers and food manufacturers. Despite great efforts to improve production and distribution techniques, hygiene standards, and consumer education, food-borne pathogenic and spoilage-causing microorganisms continue to cause huge economic losses and human costs. The increase in consumption of fresh, ready-to-eat, and processed foods to ensure its microbial safety has encouraged the development of innovative food preservation concepts that are primarily based on the use of natural antimicrobial substances rather than synthetic preservatives. This increasing demand and concern about food are leading to increased interest in natural antimicrobials that exhibit effective antagonistic activity against a wide range of undesirable microorganisms in foods. Recent studies have shown that the growth of pathogenic and spoilage-causing microorganisms may be significantly reduced or inhibited by many plant extracts, and their effective antimicrobial activities make them an interesting alternative to synthetic preservatives [92].
Phenolic compounds are considered natural functional components that can be used as antimicrobial food preservatives. These work according to different antimicrobial mechanisms, with at least three mechanisms agreed upon by many authors: (1) Modification of the permeability of cell membranes and rupture of the cytoplasmic membrane; (2) changes in various intracellular functions caused by hydrogen bonding of phenolic compounds with enzymes through their OH groups; and (3) modification of cell morphology (cell wall rigidity and loss of integrity) caused by various interactions with cell membranes [93]. As one of the most powerful antioxidants among phenolic compounds, in addition to possessing other properties such as antibacterial, antiviral, and antifungal activity, quercetin has the ability to act as a biologically active additive in food products [94]. Quercetin works according to various antibacterial mechanisms. These mechanisms involve destroying bacterial cell walls, changing the permeability of the bacterial cell, inhibiting the synthesis of nucleic acids—which affects the synthesis and expression of protein—and reducing enzyme activity [95].
The human diet includes large levels of flavonoids, as this family of polyphenolic chemicals is widely dispersed in the plant kingdom. Numerous studies have demonstrated the potential advantages of flavonoids as antibacterial agents when included in a healthy diet. These include apigenin (40,5,7,-trihydroxyflavone), myricetin (3,5,7,3′,4′,5′-hexa-hydroxyflavone), luteolin (30,40,5,7-tet-rahydoxyflavone), morin (2-(2, 4-dihydroxiphenyl)-3,5,7-trihydroxychromen-4-one), quercetin (3,5,7,3′,4′-pentahydroxyfavone), kaempferol (3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-one), and many others [96, 97]. Their beneficial bioactive properties include antiviral, antibacterial, antifungal, anti-inflammatory, and cardio-protective qualities, leading to their increasing importance in research.
For example, pharmaceutical studies have shown quercetin and kaempferol to be potential antibacterial drugs inhibiting a variety of harmful microorganisms. The antibacterial mechanisms of these compounds is well documented in literature based on microbial research, and includes bacterial membrane rupture. It has been suggested that flavonoids may prevent bacterial growth in a number of ways, including by rupturing and permeabilizing membranes, directly interacting with membrane proteins, preventing unchecked production of ROS, and preventing the formation of membranes and cell walls [98, 99].
One investigation found several flavonoids, including rutin, myricetin, naringin, catechin, luteolin, and aparin, to have antibacterial and antifungal properties. The study employed nine strains found in dental plaque—Streptococcus oralis, Streptococcus sanguinis, Lactobacillus casei, Escherichia coli, Enterococcus faecalis, Aggregatibacter actinomycetemcomitans, Actinomyces naeslundii, Actinomyces viscosus, Staphylococcus aureus—and the fungal strain Candida albicans. The findings showed that rutin, luteolin, morin, naringin, and quercetin all successfully slowed the growth of bacteria and fungi, with the most potent of these being morin [96].
The effects of flavonoids on sheep erythrocyte membrane structure and on the
diameter, zeta-potential, and membrane organization of viable S. aureus
cells were compared by Veiko et al. [100]. According to flavonoid
lipophilicity, fluorescence tests showed that naringenin and catechin had
substantially lower access to the membrane interior than quercetin. Depending on
their ability to alter the bacterial membrane structure, naringenin, catechin,
and quercetin (100–200 µM) all suppressed the development of
S. aureus cells. S. aureus-produced,
For decades, flavonoids have been extracted using traditional techniques like Soxhlet, maceration, heat-reflux extraction, and hydro distillation. Unfortunately, the methods require prolonged treatment durations, high temperatures, and more solvents; in addition, the apparatus must be used by qualified persons, the methods are not eco-friendly, and the systems are not cost-effective. These techniques include enzyme-aided extraction (EAE), supercritical fluid extraction (SFE), microwave-assisted extraction (MWAE), and ultrasound-assisted extraction (UAE). Every technique has different processing principles, parameters, and approaches to improving extraction. The new extraction methods are less time-consuming, use less organic solvents, and are safe for the environment [101]. The table below (Table 2, Ref. [102, 103, 104, 105, 106, 107, 108]) shows some methods of extracting quercetin from plant sources in general.
| Extraction method | Plant and extraction conditions | Reference |
|---|---|---|
| Soxhlet Extraction | A study was conducted in Malaysia in which researchers were able to extract quercetin from the leaves of the Melastoma malabathricum plant. The Soxhlet method was used for a period of six hours with the trialed solvents (deionized water, ethanol, ethyl acetate, and hexane), with the highest quercetin content (0.77 ppm) obtained in the ethanol extract. | [102] |
| Maceration Extraction | A 48-hour soaking method was used to extract quercetin from the fenugreek plant using a group of extraction solutions (ethanol, hexane, ethyl acetate). The results showed that the best extraction solution was ethanol. | [103] |
| Pressurized Liquid Extraction | It was found that quercetin-4′-o-glucoside (G4′) could be extracted from (white and red) onions (Allium cepa) by using a pressurized liquid method and a mixture of methanol and water as a solution of extraction. | [104] |
| Ultrasound Extraction | It was reported that (waste) onion peels have a high content of quercetin, which could be extracted using ultrasound technology with aqueous ethanol and under specific conditions, such as a percentage of ethanol of 59% and an extraction temperature of 49 °C, which led to obtaining a total quercetin content of 11.08 mg per gram of dry weight of solid onion waste. | [105] |
| Microwave Extraction | When extracting quercetin from the stems of the Euonymus alatus plant, it was observed that the microwave (MAE)-assisted method was a very effective and rapid way to extract quercetin from plants compared to methods such as Soxhlet, soaking, and ultrasound when using 50% ethanol as the extraction solution and W170 in addition to 6 minutes of saturation time. | [106] |
| Enzyme-Assisted Extraction | Enzymatic extraction was studied using cellulose, xylanase, and |
[107] |
| Supercritical Fluid Extraction | This study used the supercritical fluid extraction method to obtain quercetin from sumac fruits using supercritical carbon dioxide modified with ethanol, in addition to many factors that affect the output of supercritical extraction, such as temperature, pressure, flow rate, and supercritical fluid. | [108] |
UAE is a newly developed method of extraction with a lower time of reaction that releases the least amount of hazardous solvents into the environment of the described extraction methods, in addition to being easy to use. During extraction, ultrasound may improve solvent penetration by rupturing cell walls, increasing the area of the contact surface, and promoting the solvation of soluble compounds [109]. The extraction of flavonoids using UAE is influenced by the temperature of treatment, duration, power, and the ratio of liquid–solid. When the ratio of liquid–solid is increased in the UAE, the rate of extraction rapidly increases. However, the breakdown of active substances by ultrasonic waves is a drawback of UAE.
An extraction technique for obtaining quercetin from onion solid waste utilizing an ultrasonic treatment and a solution of aqueous ethanol was developed by Jang et al. [105]. The study found that the most significant factors impacting the recovery rate were the ethanol content (40–80%, v/v) and temperature (40–60 °C) of extraction, whereas the extraction’s integrity was unaffected by the ratio of liquid/solid extraction, pH, or extraction duration. With ethanol (59%) and extraction temperature (49 °C), the solid onion waste had the highest quercetin extraction, yielding 11.08 mg/g dry weight. Similar to the latter study, another was conducted to compare different methods of extracting quercetin from Raphanus sativus leaves. Findings indicated that using UAE at 50% ultrasonic intensity for 10 minutes in methanol was the optimal method for extracting flavonoids. In comparison to Sharifi et al.’s [110] Soxhlet, maceration, and digesting methods, a higher quercetin level was obtained using UAE.
Another study was conducted by Wang et al. [28] to improve the extraction of quercetin, chrysanthemum, pine cortex, apigenin, and luteolin from Schisandra sp. To modify the UAE’s process parameters, the Taguchi method and Plackett–Burman design were combined. The Plackett–Burman design was initially used based on a single-factor analysis, which showed that the particle size, temperature, ultrasonication time, and intensity significantly affected the rate of extraction. The Taguchi method was also helpful for further determining the optimal conditions. These conditions (16.26 mg/g of sample) were achieved at 70 °C, 0.18 mm particle size, 60% ethanol, 35 min ultrasonication time, 25 mL/g ratio of liquid–solid, and a 3.3 W/cm2 adjustment of ultrasonication density.
Moreover, Wei et al. [111] investigated the quercetin, taxifolin, and diosmin extraction characteristics from Abies nephrolepis bark and leaves. A first-order kinetic model was used to perform the synchronized kinetic of the extraction yields of these three flavonoids. According to the results of the thermodynamic analysis, quercetin, taxifolin, and diosmin extraction from the bark and leaf samples was a natural endothermic process in which the disorder deteriorated. In another study employing UAE, Thuy et al. [112] extracted and identified quercetin from the peel and bulb of shallots, obtaining a greater yield with UAE compared to the solvent extraction method. UAE increased the shallot peel’s extraction yield of quercetin by 13.38–15.64%, and the bulb’s by 49.46–56.88%.
In MWAE, dipole molecules rotate as a result of the application of microwave energy. The solvent’s temperature rises rapidly at the same time, breaking or rupturing the sample’s cell wall and promoting the extraction of the desired chemicals [113]. Powerful microwave irradiation during MWAE operation will raise the extraction solvent temperature, matrix of the sample, and improve mass transfer rate, all of which have an impact on the extraction yield. A further crucial component for extraction yield is the microwave irradiation period. As time is increased toward a certain limit, the yield of extraction clearly increases. One to three cycles of extraction are adequate to provide a better yield while saving solvent, energy, and time [114, 115]. Moreover, the choice of solvent is one MWAE restriction; only microwave-absorbing solvents, like methanol and ethanol, are appropriate [115].
An earlier study on quercetin and rutin extraction from Euonymus alatus stalks, applying the MWAE method, was published by Zhang et al. [106]. The results from MWAE were compared to those from UAE and Soxhlet extraction, showing that the MWAE yield of quercetin was higher, at 0.016 mg/g.
To extract quercetin from the peel of onion, three extraction techniques (MWAE, UAE, and solvent extraction) were also reported. Higher than was achieved in previous extraction techniques by Jin et al. [116], MWAE achieved a maximum extraction yield of 20.3–30.8%. A simple renewable energy-based technique was created earlier by Kumar et al. [117] to achieve the optimal conditions for UAE and MWAE. For the quercetin extraction rate, extraction time effects, pH, and other operating parameters were examined. Commercial silica was chemically modified using synthetic ionic liquids to create MWA silica adsorbents with ionic liquid bases, which were used as a unique adsorbent for the separation of quercetin and its glycosides from the solid onion peel. Moreover, the silica adsorbent with an ionic liquid base showed improved selectivity when compared to conventional C18 adsorbents.
Mukhaimin et al. [118] recently used MWAE and 80% methanol to extract quercetin from dried Carica papaya flowers. Several microwave (MW) powers (120, 200, 280, and 400 W) and extraction times (1–5 min) were used in the study. The maximum yield (0.214%) was obtained at 400 W MW power and 4 minutes of treatment.
An alternative innovative approach used to extract flavonoid molecules from plant sources is UMWAE. By rupturing cell walls and increasing surface area contact during extraction, ultrasound may improve the penetration of solvents and increase soluble materials’ solvation [119]. Furthermore, microwave energy increases sample temperature rapidly, boosting the solute solubility and mass transfer rate, and improving the effectiveness of extraction by promoting the effectiveness of the targeted compound’s activity during desorption [115]. Therefore, the simultaneous application of microwave and ultrasonic irradiation would provide the extraction solvent with adequate heat and energy; however, this requires a system that has been specifically developed to incorporate both types of irradiation.
According to Sun et al. [119], using microwave and ultrasound together produces a good extraction yield and is useful for obtaining chemicals that are sensitive to heat. In another study, quercetin was extracted from powdered Anoectochilus roxburghii using the UMWAE method. Ethanol concentration, extraction time, temperature, and the ratio of liquid to solid were deemed crucial factors for the efficient extraction of quercetin. Consequently, the maximum quercetin yield was achieved with a temperature range of 50 °C, 50% ethanol concentration, a 15-minute extraction time, and an 8:1 liquid-solid ratio [120].
Results from a different study showed that high-speed countercurrent chromatography (HSCCC) was effective at purifying quercetin, while UMWAE was effective for extracting it. This detachment approach was more effective than some conventional methods. A solution of two-phase solvent composed of methanol, ethyl acetic acid derivatives, n-hexane, and water (3:6:4:3, volumes) was used to conduct the HSCCC test. In a separating funnel, every soluble mixture was thoroughly equilibrated at ambient temperature [120].
One of the more recent methods of extraction is SFE. Any materials that have reached their thermodynamic critical point consequently have low viscosity and high diffusivity, which aids in increasing matter transfer; this state is referred to as a supercritical fluid. SFE is now widely used in a variety of industries, including the chemical, toxicological, environmental, petrochemical, textile, and polymer sectors [111], in addition to the food and pharmaceutical industries. The extraction of bioactive substances has been supported by notable SFE implementations in several research areas during the past three decades. Moreover, SFE showed a greater extraction yield of flavonoids, particularly quercetin, according to Chávez-González et al. [121].
Previously, by extracting biological solvents through SFE, Lévai et al. [122] created submicron encapsulated quercetin particles. The dispersed organic phase was rapidly oversaturated as a result of the rapid exclusion of the organic solvent, which led to precipitation and entanglement of the submicron quercetin particles with the surfactant. In a pluronic experiment, approximately 1-m sized, needle-shaped quercetin particles were created, albeit with low encapsulation efficiency. With no isolated quercetin crystals, quercetin-loaded multivesicular liposomes for soybean lecithin were able to encapsulate up to 70% of the compound with a 100 nm particle size.
Soxhlet extraction and modified SFE with ethanol were used to extract quercetin from Rosa damascena by Ghoreishi et al. [123]. In the investigation, the temperature of extraction ranged from 35 to 55 °C, the rate of CO2 flow was set to 0.3–1.5 mL per minute, and the extraction time ranged from forty minutes to two hours. According to their response surface methodology (RSM) test, the model’s modified R2 and R2 were, respectively, 87.1% and 93.1%. The maximum quercetin yield in the study, of 32.0%, was achieved using the RSM test, which indicated an optimal operating temperature of 46.3 °C, 25.5 MPa pressure, 2 h extraction time, and 0.7 mL/min CO2 flow rate.
Quercetin is a natural flavonoid found in many fruits, vegetables, and grains, such as apples, onions, broccoli, berries, and green tea. Quercetin has attracted the attention of researchers for its potential health benefits, including its antioxidant, anti-inflammatory, antiviral, and anti-cancer properties. The mode of action of quercetin involves multiple mechanisms and different levels of action on cells and molecules, making it a promising compound in the prevention and treatment of many diseases.
The antioxidant activities of quercetin come mainly from its ability to neutralize free radicals and chelate metal ions that can catalyze the formation of reactive oxygen species (ROS). Free radicals are unstable molecules that contain unpaired electrons and can cause cell damage by reacting with proteins, lipids and nucleic acids. By neutralizing these reactive species, quercetin contributes to protecting cells from oxidative stress that is associated with aging and many chronic diseases such as cardiovascular disease and cancer [124]. This antioxidant property is largely attributed to the hydroxyl groups in its chemical structure, which can provide hydrogen atoms to neutralize free radicals.
In addition to its direct antioxidant effects, quercetin modulates the expression of antioxidant enzymes through activation of the Nrf2 transcription factor pathway. Nrf2 is a transcription factor involved in regulating the expression of antioxidant genes. When Nrf2 is activated, it translocates to the nucleus and binds to the antioxidant moiety in the promoter regions of a variety of genes encoding antioxidant proteins such as superoxide dismutase (SOD), catalase, and glutathione peroxidase [125]. This enhances the cells’ antioxidant defense system, providing additional protection against oxidative damage.
Quercetin also exhibits anti-inflammatory effects by modulating key signaling
pathways associated with inflammation. One of the main mechanisms is inhibition
of the NF-
Furthermore, quercetin inhibits the MAPK pathway, which is involved in cell proliferation, differentiation, and stress responses. The MAPK pathway involves several protein kinases that are activated in response to external signals, and plays a critical role in the regulation of many cellular processes. By inhibiting the MAPK pathway, quercetin reduces the production of inflammatory mediators and reduces the expression of enzymes such as Cyclooxygenase-2 (COX-2) and Inducible Nitric Oxide Synthase (iNOS), which are involved in the inflammatory process [128]. COX-2 is an enzyme that catalyzes the formation of prostaglandins, which play a key role in promoting inflammation and pain. iNOS is an enzyme that catalyzes the production of nitric oxide, a molecule that plays a dual role in inflammation and vascular regulation.
The antiviral properties of quercetin also deserve attention. It interferes with different stages of viral replication and can inhibit the activity of viral enzymes. For example, quercetin has been shown to inhibit the protease activity of hepatitis C virus, preventing viral replication [129]. In addition, quercetin modulates signaling pathways in host cells, enhancing the antiviral response of the immune system. It can increase the production of interferons, which are cytokines that play a crucial role in antiviral defense by activating immune cells and preventing virus replication [130]. Interferons are proteins secreted by virus-infected cells that activate immune cells such as natural killer cells and cytotoxic T cells, enhancing the ability to fight viruses.
The anticancer effects of quercetin are attributed to its ability to induce apoptosis, inhibit cell proliferation, and suppress proliferation. Apoptosis is a programmed process that leads to the death of unwanted or damaged cells, and is essential for maintaining tissue health and preventing cancer. Quercetin induces apoptosis in cancer cells through activation of both mitochondrial (intrinsic) and death receptor (extrinsic) pathways of apoptosis.
It increases the expression of cell death-inducing proteins such as Bax and reduces the expression of anti-apoptotic proteins such as Bcl-2, leading to the release of cytochrome c from mitochondria and the activation of caspases, which are enzymes that carry out the process of apoptosis [131]. In addition, quercetin inhibits the PI3K/Akt pathway, which is often dysregulated in cancer, leading to reduced cell survival and proliferation [132]. The PI3K/Akt pathway is a cellular signaling pathway that plays a key role in regulating cell growth, survival, and metabolism.
Quercetin also affects the tumor environment by inhibiting angiogenesis, the process of creating new blood vessels to supply nutrients to tumors. It reduces the expression of vascular endothelial growth factor (VEGF) and other angiogenesis-stimulating factors, starving the tumor of its blood supply and thus inhibiting its growth and metastasis [133]. VEGF is a protein secreted by cancer cells to promote the growth of new blood vessels, and is essential for tumor nutrition and growth.
Furthermore, quercetin has the ability to modulate gene expression in cancer cells. It can reduce the expression of genes related to cell proliferation and invasion, while increasing the expression of genes inducing apoptosis. This balance between inducing cell death and inhibiting cell proliferation is one of the key factors that makes quercetin a promising ingredient in anti-cancer therapies [134].
Besides its antioxidant, anti-inflammatory, antiviral and anti-cancer properties, quercetin also has beneficial effects on neurological health. Studies show that quercetin can cross the blood-brain barrier and exert protective effects on neurons. By reducing oxidative stress and inflammation in the central nervous system, quercetin can contribute to the prevention of neurodegenerative diseases such as Alzheimer’s and Parkinson’s [135]. Research suggests that quercetin can inhibit the buildup of beta-amyloid protein, which is linked to the development of Alzheimer’s disease, and reduce nerve cell damage caused by oxidative stress [136].
Furthermore, research shows that quercetin can improve cardiovascular health. It can improve vascular endothelial function, reduce blood pressure, and improve blood flow. By inhibiting low-density lipoprotein (LDL) oxidation and reducing inflammation, quercetin may contribute to the prevention of atherosclerosis, a disease characterized by the accumulation of fats and plaques on the walls of arteries [137]. In addition, quercetin can improve blood vessel elasticity and reduce the risk of clot formation, promoting overall cardiovascular health.
Regarding the effects of quercetin in foods, it plays an important role as a natural preservative due to its antioxidant and antimicrobial properties. Quercetin helps extend the shelf life of foods by preventing oxidation and spoilage caused by microorganisms. Quercetin’s antioxidant effects enhance the stability of fats and oils in foods, preventing decomposition and oxidation that lead to rancidity. In addition, quercetin can be used as an antimicrobial agent in foods, contributing to reducing the growth of harmful microorganisms such as bacteria and fungi [27].
Quercetin exhibits antibacterial and antifungal effects, making it a promising candidate for use in combating infections. Its mechanism of action against bacteria is thought to involve inhibiting enzymes necessary for bacterial reproduction and growth.
For example, quercetin inhibits the activity of enzymes such as DNA gyrase and topoisomerase IV, which play a crucial role in bacterial DNA replication [138]. For fungi, quercetin is thought to weaken the fungal cell wall and disrupt membrane functions, leading to fungal death [139].
Quercetin also shows anti-type 2 diabetes effects. Research suggests that quercetin can improve insulin sensitivity and reduce blood glucose levels. It does this by modulating insulin-related signaling pathways and enhancing glucose uptake into cells [140].
Numerous phytochemicals, derived from a large number of natural sources, have demonstrated therapeutic efficacy against a variety of pathogenic species in multiple notable research projects carried out in recent years. Among them is quercetin, one of the most well-known and biologically active antioxidants. Quercetin has been shown to possess strong antagonistic properties against a range of bacteria, fungi, and viruses. This review examined quercetin’s safety as a nutritional supplement and its range of biological effects in both humans and animals. The majority of published studies demonstrated the safety profile of its antibacterial, antifungal, antiviral, and antioxidant activity in animals. However, more thorough analyses in this regard, with reliable results, is urgently required. A key issue with the usage of quercetin is its poor oral bioavailability and solubility. Because of its synergistic actions with other substances, quercetin is a promising chemical for creating modern, more effective, and less harmful treatment modalities for both acute and chronic human disorders. The available research reveals quercetin to have broad-spectrum antimicrobial properties, making it a potential nutraceutical for fighting bacteria, fungi, and viruses. Unfortunately, its low oral absorption and bioavailability in the human body hampers its usage as an efficient antibacterial agent. To fully utilize its medicinal potential, additional study is needed to improve its bioavailability. It is also important to understand the potential future role of quercetin, as subsequent research may reveal it to have greater potential in the treatment of chronic diseases such as diabetes, cancer, and heart disease, and new treatments may be developed based on our increasing understanding of this important protein. Thus, investing in research on quercetin and understanding its underlying role may be key to improving overall health and increasing quality of life in the future.
SGA-S, SHB, and ZKA-Y performed the literature search and wrote the manuscript. SHB contributed to the figure design. SGA-S provided oversight and direction for the manuscript. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity. All authors read and approved the final manuscript. All authors contributed to editorial changes in the manuscript.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


