1 Faculty of Health Sciences, Centre of Excellence for Pharmaceutical Sciences (PharmacenTM), 2520 Potchefstroom, North-West, South Africa
Abstract
Background: Despite attempts to control malaria, poor drug bioavailability means malaria still places enormous pressure on health globally. It has been found that the solubility of highly lipophilic compounds can be enhanced through lipid formulations, e.g., self-emulsifying drug delivery systems (SEDDSs). Thus, quality-by-design and characterization were used to justify the development and determine the feasibility of oral oil-in-water SEDDSs comprising a fixed-dose combination (FDC) of artemether–lumefantrine to treat malaria more effectively without the aid of a fatty meal. These formulations were compared to a commercial product containing the same active compounds. Methods: Excipient compatibility and spontaneous emulsification capacity of different FDC–excipient combinations were identified by employing isothermal microcalorimetry, solubility, and water titration tests. Pseudoternary phase diagrams were constructed, and checkpoint formulations were selected within the self-emulsification region by reviewing formulation properties essential for optimized drug delivery. SEDDSs capable of enduring phase separation within 24 h were subjected to characterization experiments, i.e., drug concentration determination, cloud point, droplet size, size distribution, self-emulsification time, self-emulsification efficacy, viscosity, zeta potential, and thermodynamic stability analysis. SEDDSs with favorable characteristics were identified in the micro or nano range (SNEDDSs) before being subjected to drug release studies. Results: All final formulations depicted enhanced artemether and lumefantrine release compared to the commercial product, which could not release lumefantrine at a quantifiable concentration in this study. The avocado oil (AVO)4:6 and olive oil (OLV)3:7 SNEDDSs overall portrayed the ideal characteristics and depicted the highest percentage of drug release. Conclusions: This study offers evidence that SNEDDSs from selected natural oils comprising an artemether–lumefantrine FDC can potentially enhance the bioavailability of these lipophilic drugs.
Graphical Abstract
Keywords
- artemether (ART)
- lumefantrine (LUM)
- malaria
- lipophilic drugs
- lipid-based dosage forms
- self-emulsifying drug delivery systems (SEDDSs)
- self-nanoemulsifying drug delivery systems (SNEDDSs)
- pseudoternary phase diagrams
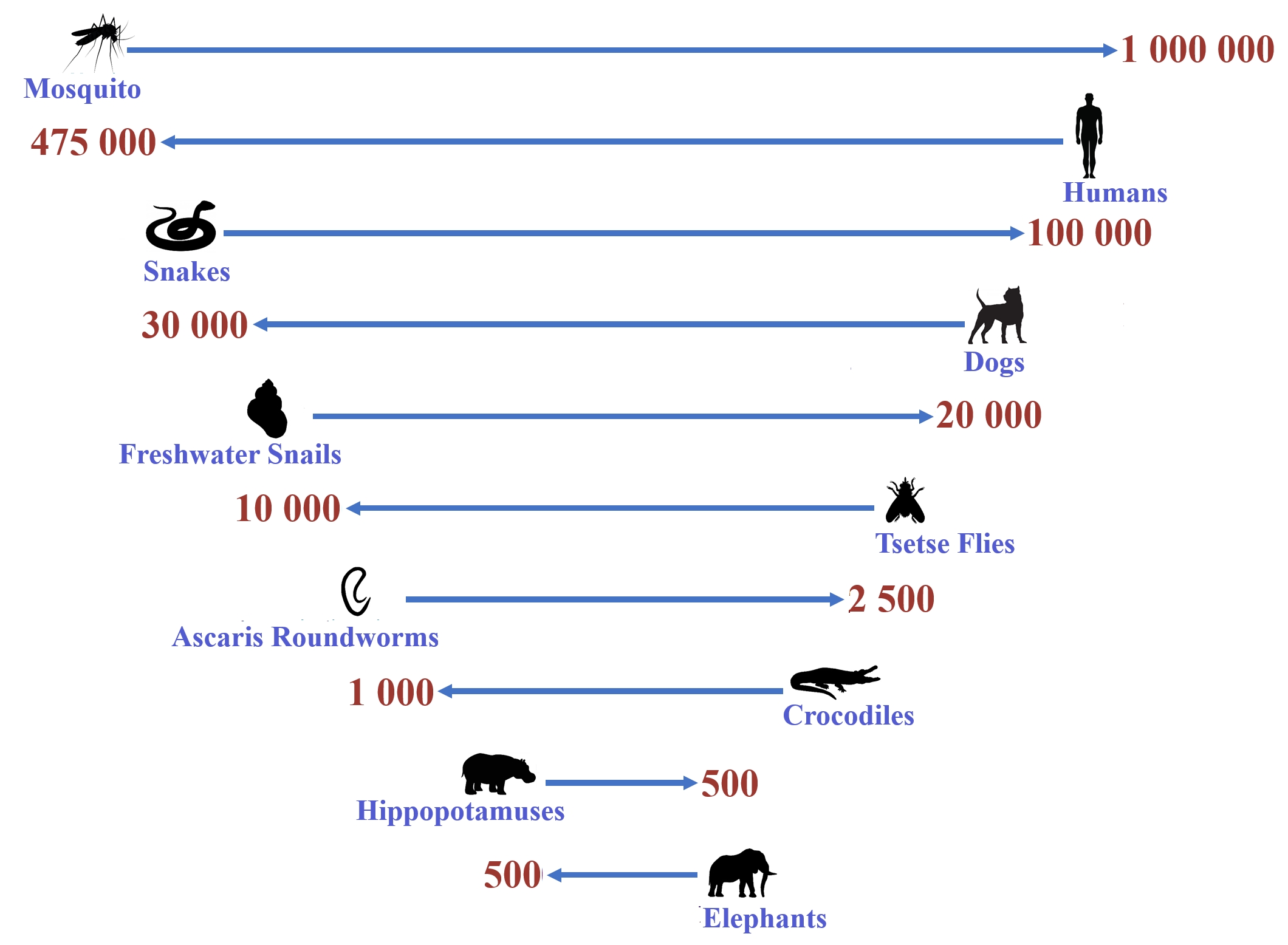
Despite rigorous attempts to control malaria and eradicate it by 2030, it remains an acute febrile infectious disease that is distressing, widespread, and extremely complex, placing enormous pressure on health worldwide [1, 2, 3, 4, 5, 6]. This established vector-transmitted disease is endemic to 97 regions and has shown an unfortunate increase in various countries such as South Africa. According to the World Health Organization (WHO), 247 million occurrences of malaria were recounted in 2021 worldwide. Of these incidents, more than 620,000 individuals died, while the WHO African Region represented approximately 96% of all malaria fatalities. Globally, the mortality rate fluctuates between 0.3 and 2.2%, while regions with tropical climates illustrate mortality figures between 11 and 30% [3, 7, 8]. Unfortunately, it seems that these figures are rising annually despite current interventions. Daniel Vasella, the Chief Executive of Novartis, detailed in 2006 that: “The fight against malaria is a complex one. Availability of the drug is only one element” [9]. However, currently, drug availability is not the main concern anymore as various diverse methods of infection are being observed. A specific malaria infection process has been documented as ‘Odyssean’, ‘minibus’, or ‘baggage’ malaria. This method grants transference of the Plasmodium falciparum parasite, particularly to non-endemic regions, due to the mosquito’s ability to survive inside the luggage of travelers under favorable climatic conditions [10, 11]. In addition, ‘airport malaria’ can occur due to the infectious bite of a tropical anopheline mosquito that has traveled by plane (occasionally also internationally). The probability of this mosquito surviving in a different region is notably high before needing a blood meal, as the hot and humid weather of unlikely areas during summer can favor endurance and breeding. Thus, numerous tourists from developed nations are becoming progressively more prone to contracting or spreading malaria annually. Furthermore, both of these transmission routes are related to the often delayed diagnosis of the patients in question, highlighting the danger of malaria [4, 12, 13] and identifying the mosquito as one of the world’s deadliest creatures (Fig. 1, Ref. [14]), inflicting more harm to humans than most animals [14]. An additional fear is that individuals born in a malaria region develop ‘semi-immunity’ against this parasite owing to enduring various contracted malaria infections; however, when traveling to a non-malaria district, this ‘semi-immunity’ vanishes within 3–6 months, making them just as susceptible to being afflicted by malaria as other individuals [15]. Additionally, the progress of parasitic resistance by P. falciparum and P. vivax to antimalarial drugs furthermore poses one of the greatest dangers to malaria control and has resulted in increased malaria morbidity and mortality [16, 17]. However, it appears that developing drug resistance is not the fundamental distress; it is rather the ‘under-dosing’ of therapeutics that is a significant concern [4, 18].
The inability to successfully deliver highly lipophilic therapy to a patient is likely the cause of subtherapeutic drug concentrations achieved during treatment (i.e., erratic and/or inadequate drug absorption) [19, 20, 21, 22, 23]. For this reason, studies revealed that a fatty meal is required for more productive treatment when taking antimalarial medication to achieve adequate therapeutic drug concentrations in subjects who are too unwell to eat for the duration necessary to obtain ample improvement [19, 20, 21, 22, 23]. This ‘food effect’ is said to enhance the absorption of lipophilic compounds that have been co-administered because of the induction of bile secretion. Sequentially, a solubilizing setting consisting of acid and homogenized fat is provided, and the formed milieu stimulates nutritional lipid absorption [24]. For example, it has been described that oral lumefantrine (LUM) bioavailability can be improved up to 16 times when lipids are provided in conjunction with LUM during treatment [21, 25]. In addition, studies showed that lipid amounts as few as two grams can promote adequate quantities of bile salt to increase intraluminal lipid refining and drug solubilization [23]. However, these inferences are problematic. The frequently experienced gastrointestinal disorders associated with malaria, such as nausea, vomiting, abdominal pain, and appetite loss, deter patients from eating adequately—particularly fatty foods [21, 26, 27]. Subsequently, erratic drug bioavailability, insufficient drug–blood levels for effective treatment as well as patient nonconformity occur [4]. Furthermore, it is extremely challenging for patients living in a low or middle-income country who are not only continuously nauseated but also reside in impoverished regions (most of these regions are tropical) and do not have the resources to obtain appropriate food to eat adequately [28]. It can, therefore, be asserted that under-dosing is likely due to an insufficient drug delivery system that is unable to increase lipophilic drug absorption [4]. Furthermore, the following three considerations should be taken into account when wanting to enhance the effect of antimalarials against the spread of the parasite:
• the active ingredient effect on early gametocyte and nonsexual phases;
• sporontocidal consequences ensue in the mosquito; and
• drug results are achieved on advanced transmittable gametocytes [29].
Treatment of malaria throughout the early phases of the parasite is beneficial
because it confidently destroys the parasite during its nonsexual stages.
Nevertheless, research should also be conducted on compounds that can eradicate
the parasite during the gametocyte phase. For instance, LUM should be considered
as it can play a vital role in malaria management [29]. Studies have shown that
P. falciparum has developed resistance to various artemisinins,
especially when utilized as a monotherapy [30]. Although new chemical
entities/drugs/compounds are being developed, lack of funding and constraints in
developing time cause delays in new treatments reaching the market. This forced
the WHO to reflect on alternate dosage treatments that can resist this endemic
disease more constructively [1, 3, 8, 17, 31]. One such effort by the WHO is the call
for developing and employing oral artemisinin-based combination therapies to
fight drug resistance, as these combinations have substantially decreased the
malaria mortality rate in recent years. Combination therapy has subsequently
developed into an area of importance and is now extensively utilized. Artemether
(ART) and LUM are, for example, not newly synthesized composites; nevertheless,
these two drugs have been prepared into a fixed-dose drug combination (FDC), the
commercially available product, Coartem
Amid the various economically viable technologies that have been researched and proven to enhance drug solubility, address bioavailability difficulties, and improve patient compliance, lipid formulations have been significantly utilized in recognized oral drug products since about 70% of recently discovered drugs and around 40% of commercially available oral drugs are practically insoluble [4, 47, 48]. One such example is self-emulsifying drug delivery systems (SEDDSs). Researchers have proposed that there are several mechanisms through which SEDDSs can improve hydrophobic drug bioavailability, including the following:
• the fluidity of membranes is increased, which subsequently enables transcellular drug absorption;
• tight junctions between cells are opened, permitting paracellular transport;
• P-glycoprotein-mediated drug efflux and/or metabolism is/are constrained due to gut membrane-bound CYP450 enzymes;
• lymphatic transport together with lipoprotein/chylomicron production stimulation are enhanced;
• in vivo dispersion using supplementary surfactants is accelerated; and
• constituent lipids are metabolized, for example [4, 23, 24, 49, 50].
However, SEDDSs are a broad expression for certain lipid-based drug delivery
systems that should rather be separated into three classes concerning droplet
size. Characteristic SEDDSs that form cloudy emulsions with a droplet size of
more than 300 nm are called SEDDSs. The second class is self-microemulsifying
drug delivery systems (SMEDDSs), followed by self-nanoemulsifying drug delivery
systems (SNEDDSs), which are both deemed translucent and possess a droplet size
range of 100–250 nm and smaller than 100 nm, respectively. Furthermore, these
three classes vary in composition: SEDDSs generally have an oil percentage
between 40 and 80% and are prepared by including hydrophobic surfactants with
hydrophilic–lipophilic balance (HLB) values lower than 12. SNEDDSs and SMEDDSs
generally comprise an oil component lower than 20%. These systems are created by
including hydrophilic surfactants with HLB values of
Presently, no antimalarial FDC has been developed into specific oral hydrophobic
drug delivery systems, including SEDDSs, SMEDDSs, or SNEDDSs. Therefore, for the
reasons mentioned, this exploratory research aimed to establish oral oil-in-water
(o/w) SEDDSs or SNEDDS formulations containing an ART/LUM FDC that comprises
certain natural oils to effectively treat malaria by enhancing the solubility of
the included drugs and subsequently, meaningfully improve their pharmaceutical
availability without the aid of a fatty meal. The natural oils included were
selected on their safety profile for oral use, their relative accessibility, and
their probability of improving the solubilization of both ART and LUM. Not only
was it attempted to improve the solubilization of this highly lipophilic FDC
significantly, but also to improve the erratic release, dissolution, and
subsequent absorption [56] of a commercial product that normally requires
consuming a fatty meal during the treatment regime [19, 31, 57]. However, the
effectiveness of a lipid formulation to form a robust self-dispersible SEDDS or
SNEDDS is typically incident-explicit; therefore, the composition of a SEDDS or
SNEDDS formulation should be determined [50, 58, 59, 60]. Consequently, this study
employed a valuable assessment tool, namely, pseudoternary phase diagrams, to
evaluate the effect of the tested formulation constituents on the in
vitro performance of the formulations. This tool offers a scientific basis to
decide which excipients and in what concentrations they should be included during
formulation [61]. To facilitate the reading of this paper, the authors used the
term ‘SEDDSs’ to collectively refer to both SEDDSs and SNEDDSs until a
distinction between the obtained formulations could be made. Chosen natural oils
(peanut: PNT; olive: OLV; coconut: CCT; castor: CAS; and avocado: AVO oil),
surfactants (sodium lauryl sulfate and Tween
ART was donated by DB FINE CHEMICALS (Pty) Ltd. LUM was offered by Cipla
Pty Ltd. (Mumbai, India). Peanut (PNT), olive (OLV), coconut (CCT), castor
(CAS), and avocado (AVO) oil were acquired from Scatters Oils (Johannesburg,
South Africa). Sodium lauryl sulfate (SLS) was secured from Merck Chemicals (Pty)
Ltd. (Johannesburg, South Africa); Tween
IR was employed to identify the donated ART and LUM samples and verify that
these powders contained no impurities. The positions of the absorbance bands at
different wavelengths of the received ART and LUM batches were individually
compared to the positions of the absorbance bands of the known, pure reference
standards. An Alpha sample compartment RT-DLaTGS IR (ATR) spectrometer (Bruker,
Billerica, MA, USA) was used, and analysis was conducted in the spectral range
between 4000 and 400 cm
Excipient compatibility was recognized according to a previously published
method [63, 64] employing a 2277 thermal activity monitor, TAMIII (TA Instruments,
New Castle, DE, USA) furnished with an oil bath (stability:
An excess amount (
Samples were analyzed employing an Agilent
Pseudoternary phase diagrams of the drugs, oils, surfactant phase (surfactant and co-surfactant), and water are valuable in concluding the most suitable combination of excipients to form effective SEDDSs. When combined, these illustrations recognize the self-emulsifying areas and verify the ideal concentrations and fractions of the aforementioned components [63, 66, 67].
Pseudoternary phase diagrams of the selected oils included were created
employing the water titration method [58, 59, 63, 68]. The surfactant phase was set
at a 1:1 concentration fraction as the literature resolved it to produce more
stable SEDDSs. Higher fractions expand the emulsion range; however, these ratios
aid a decrease in stability that might trigger the precipitation of included
drugs [66, 67]. ART and LUM concentrations (% w/w) differed according to their
individual solubility in a selected oil [69]. The surfactant phase and the chosen
oils were primed in set fractions (9:1, 8:2, 7:3, 6:4, 5:5, 4:6, 3:7, 2:8, and
1:9), during which water (varying element) was supplemented dropwise at
All formulations were visually examined for transparency and optical
isotropicity. Each microscopic appearance of a SEDDS was observed using
cross-polarized light microscopy (Nikon
Selected SEDDS formulations were prepared utilizing aqueous phase titration and
using the results from the erected pseudoternary phase diagrams. The ART/LUM FDC
was dispersed in each selected oil and exposed to sonication for 2 min with a
UP400St (400 W, 24 kHz) Hielscher’s digital ultrasonic device (Hielscher
Ultrasonics, Teltow, Germany). A chosen surfactant phase was separately organized
through constant stirring and heating for 25 min. Then, each oil phase was added
to the surfactant phase according to the ratios determined while incessantly
stirring for an extra 25 min. Small additions (less than 5% w/w) of deionized
water were subsequently included with lag times in between until the
predetermined amount was incorporated. Before storage at room temperature
(25
A 5 mL SEDDS sample from each batch was filtered through a 0.45 µm membrane filter. Each deposit was first diluted with THF and then further diluted with methanol to attain a final volume of 10 mL. These samples were positioned in HPLC vials and analyzed in triplicate using HPLC, as detailed in Section 2.2.3.
Mean droplet size, zeta potential, and size distribution were evaluated using
dynamic light scattering conducted at 25 °C by a Zetasizer
Nano
A 1 mL sample of each SEDDS was added to 100 mL of distilled water, maintained
at 37 °C (
| Grading | Description |
| Grade A | Immediate emulsion formation, exhibiting a clear/bluish emergence within 60 sec |
| Grade B | Instant bluish emulsion establishment within 60 sec |
| Grade C | Delicate milky emulsion that formed within 120 sec |
| Grade D | Cloudy, greyish to white emulsion that depicts a vaguely oily presence, and which is slow to cream (prolonged: |
| Grade E | Defective or negligeable emulsification seen. Sizable oil droplets detected on the emulsion surface |
A Brookfield
Each fixed-dose SEDDS combination was diluted (1:100) with purified water, and the sample was initially positioned in a water bath at 25 °C. Progressively, the temperature was increased at 2 °C/min until an opaque presence (i.e., the cloud point) of the tested formulation was depicted, indicating dehydration of the included excipients [77, 78].
The formulated SEDDSs were subjected to fluctuating temperatures (freeze-thaw sequences) during three sequences of about 4 °C and 45 °C, not surpassing 48 h. Each SEDDS was accordingly inspected for potential phase separation or drug precipitation. Following, these formulations were centrifuged at 3500 rpm for 30 min; whereafter, they were again evaluated for any signs of instability, for example, cracking, creaming, and/or phase separation [79]. Lastly, SEDDS formulations were diluted 100 times using distilled water. Each sample was retained at 25 °C for 24 h preceding visual assessment for drug precipitation and/or phase separation [59].
The effect of exposing SEDDSs to both gastric and intestinal pH levels upon drug
release was controlled in progressive dissolution settings (100 rpm stirring
rate; regulated temperature: 37
DDSolver software (freely accessible menu-driven add-on program for
Microsoft
For this study, the fit factors and mean dissolution time (MDT) [86] were calculated, followed by the construction of the release profiles. Subsequently, the dissolution profiles of all SEDDSs and the commercial product (the control) were related and reflected according to the individual MDTs and fit factor results. Moreover, the fit factors for the correlating formulations are offered in the Supplementary Material (Supplementary Tables 1,2). MDT indicates the mean time interval for an entire drug dose to be emitted from a dosage form into a solution (Eqn. 1).
where n is the total number of samples; j is the sample number; t
The fit factor, f
where R
Statistica software (ver.12; TIBCO Software Inc., New York, NY, USA) was
utilized to perform a one-way analysis of variance (ANOVA) where
p-values
IR spectroscopy is a trustworthy procedure to detect and verify different samples by comparing their unique wavelengths, intensities, number of bands present, and band contours [88]. The IR spectra of standard ART and LUM references and those of donated ART and LUM batches were individually measured. The resulting IR spectra were compared through data overlays of the spectra (Supplementary Material, Supplementary Figs. 1,2). Both active ingredients were identified and verified to be pure compounds without contamination since the peaks and intensities of the samples correlated with those of the individual reference standards. Moreover, the absence of additional peaks at different intensities ruled out the presence of any contaminants.
IMC is a highly sensitive and useful instrument for studying thermal activities
by measuring heat flow and temperature fluctuations (
Table 2 shows that no incompatibilities could be identified for any tested oil, drug, and/or surfactant combination. Likewise, no significant differences in heat flow or additional sloping were noticed (Supplementary Material, Supplementary Figs. 3–7), which may be ideal for formulating SEDDSs containing an ART/LUM FDC. Overall, combinations comprising either OLV or CAS depicted slightly less heat flow during testing, whereas combinations that included AVO displayed heat flow values that were marginally higher, comparatively. Nonetheless, no trough or slope was observed in the interaction curves, meaning no incompatibilities could be detected, thereby deeming all the selected mixtures unanimous (Supplementary Material, Supplementary Figs. 3–7). Therefore, all excipients could be further utilized in this study.
| ART/LUM FDC with Specified Surfactants | Average heat flow (µW/g) | ||||
| Avocado oil | Castor oil | Coconunt oil | Olive oil | Peanut oil | |
| Tween |
4.78 (4.15) | 4.92 (9.93) | 12.67 (12.78) | 5.377 (6.55) | 0.70 (0.01) |
| Tween |
9.58 (3.85) | 9.70 (11.4) | 11.99 (12.40) | 1.15 (1.49) | 16.91 (17.31) |
| Sodium lauryl Sulfate + Span |
14.12 (1.42) | 20.58 (27.06) | 15.32 (16.60) | 24.75 (21.52) | 21.57 (24.54) |
| Sodium lauryl sulfate + Span |
19.91 (19.52) | 1.29 (4.02) | 5.44 (6.42) | 7.12 (6.58) | 8.77 (9.34) |
ART, artemether; LUM, lumefantrine; FDC, fixed-dose combination.
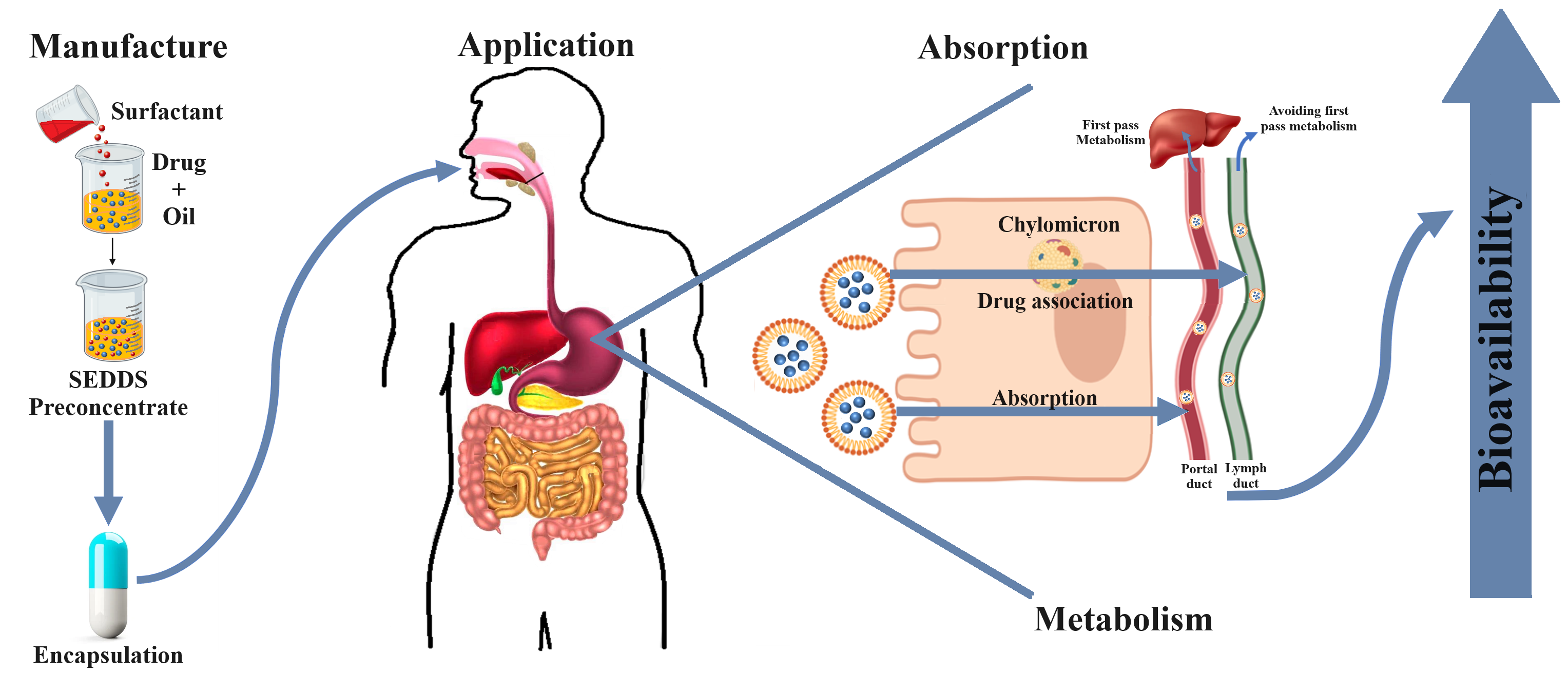
The failure of highly lipophilic drug treatment, such as ART and LUM, has been associated with incomplete absorption of active ingredients due to poor aqueous solubility [20, 45]. For this reason, the key challenge remains to construct a delivery system that can effectively enhance the solubility of both drugs. Christian et al. [91] proposed that the stability and solubility of these compounds could be improved in the presence of oils or lipids. SEDDSs improve drug absorption by distributing the drug in a dissolved state, thus evading the conventional drug dissolution action. Furthermore, drugs can remain solubilized during preparation, dispersion, and digestion [92, 93]. The existence of lipids or lipid-based formulations in the duodenum acts on the principle that the presence of exogenous lipids will cause the creation of colloids into which the active ingredients can divide (i.e., into which the drugs can partition, solubilize, and in turn, enhance absorption), thereby increasing their solubilization within the microenvironment and affording a concentration increase for lipid absorption [93]. Furthermore, due to their microscopic droplet size, emulsified drugs are straightforwardly absorbed by the lymphatic system, thus avoiding hepatic first-pass metabolism. In general, by increasing drug solubilization through the formulation of SEDDSs, absorption over the intestinal epithelial tissue is also substantially increased for BCS Class II drug molecules [92, 94, 95, 96]. According to the BCS, ART and LUM are categorized as Class II drugs, indicating that both active compounds are very sparingly soluble [97, 98], and when solubilizing these drugs in any of the selected oils, their solubility will increase significantly, which, in turn, will enhance absorption and subsequent bioavailability.
Generally, ART showed noticeably higher solubility in the selected oils than LUM
(Table 3). The solubility of the drugs in CCT is considered the highest
comparatively. Even so, the solubility of both drugs was markedly higher in all
of the assessed lipophilic agents, and the subsequent rank order regarding
lipophilic vehicle solubility may be reflected as CCT
| Vehicle | Solubility (mg/mL)* | |
| ART | LUM | |
| Avocado oil (AVO) | 127 |
1.00 |
| Castor oil (CAS) | 74 |
0.70 |
| Coconut oil (CCT) | 135 |
2.00 |
| Olive oil (OLV) | 118 |
1.00 |
| Peanut oil (PNT) | 113 |
1.00 |
| Water | 2.00 |
0.13 |
*Data are presented as the mean
Among the many tools available to advance the solubility of highly lipophilic compounds and surpass bioavailability concerns, economically sustainable lipid formulations, such as SEDDSs, have been highly retained in several recognized oral drug products [4, 47, 48]. SEDDSs are uniform mixtures of active compound(s) with a blend of lipids, surfactants, and co-surfactants able to generate spontaneous thermodynamically stable oil-in-water dispersions (emulsions) [102]. This pharmaceutical technology has become a promising strategy for developing improved oral drug delivery systems incorporating active pharmaceutical ingredients with notably low aqueous solubility features (i.e., biopharmaceutical classification system (BCS) Class II drugs). The ability of these systems to yield emulsions post-exposure to modest agitation and dilution through the addition of a water phase offers an uncomplicated process for advancing lipophilic drug delivery, where continued drug dissolution in the aqueous environment of the gastrointestinal tract is termed the rate-limiting phase, which influences drug absorption [49, 55]. Moreover, SEDDSs can significantly improve drug bioavailability as these systems have been reported capable of circumventing the dissolution phase after oral administration, and they may even evade first-pass metabolism. Moreover, SEDDSs, and more specifically, SNEDDSs, have become a promising tactic permitting the advancement in oral drug delivery system development that contain active compounds with remarkably poor aqueous solubility as it has been found that these systems can improve the bioavailability of such drugs. What is more, and as stated previously, lipid amounts as little as two grams are required to effectively solubilize lipophilic compounds [21, 26, 27], thus demonstrating that SEDDSs and/or SNEDDSs will be capable of enabling increased antimalarial drug solubilization and absorption when a fatty meal is highly unlikely or impossible for a patient to take during treatment due to economically straining circumstances [43, 55, 102].
However, spontaneous emulsification is specifically governed by the selection of excipients that complement each other to form a vehicle designed to improve drug delivery [55, 63, 103, 104]. Self-emulsification will only be accomplished if the additives are well-suited and competent to prompt spontaneous emulsification when subjected to mild agitation. Therefore, a careful selection of excipients is vital. Self-emulsification has been shown to depend on the nominated lipid/surfactant blend, the concentration of surfactant, and the ratio of lipid to surfactant included in the formulation [105]. Research has also established that only certain additive mixtures can induce the configuration of unprompted emulsions [52, 55, 105]. For this reason, pseudoternary phase diagrams, which are a systematic development approach, are frequently utilized to aid in establishing the most appropriate composition of compounds to formulate SEDDSs. Pseudoternary phase diagrams classify the self-emulsifying regions and conclude the ideal concentrations and proportions of oil, surfactant, and co-surfactant when combined. When the area of a SEDDS is determined, the viability of producing an emulsion can be established [58, 59, 106]. Construction of these diagrams enables a schematic depiction of the zone of self-emulsification when specific excipients are used in a mixture [106, 107]. Pseudoternary phase diagrams are practical tools that can project the phase behavior of a potential SEDDS or SNEDDS, as different regions of the illustration expose different behavioral properties of emulsions. For example, they can explain the strength of dilution of a SEDDS or SNEDDS in the gastrointestinal milieu [55, 106].
As stated, the purpose of this study was to formulate stable, o/w nanoemulsions, which include natural oils that can be utilized for oral lipophilic drug delivery. Fig. 2 represents the pseudoternary phase diagrams created after testing the different combinations of selected excipients deemed feasible to formulate for oral drug delivery. The pseudoternary phase diagrams that indicated unstable SEDDS formulations can be viewed in the Supplementary Material (Supplementary Figs. 8,9).
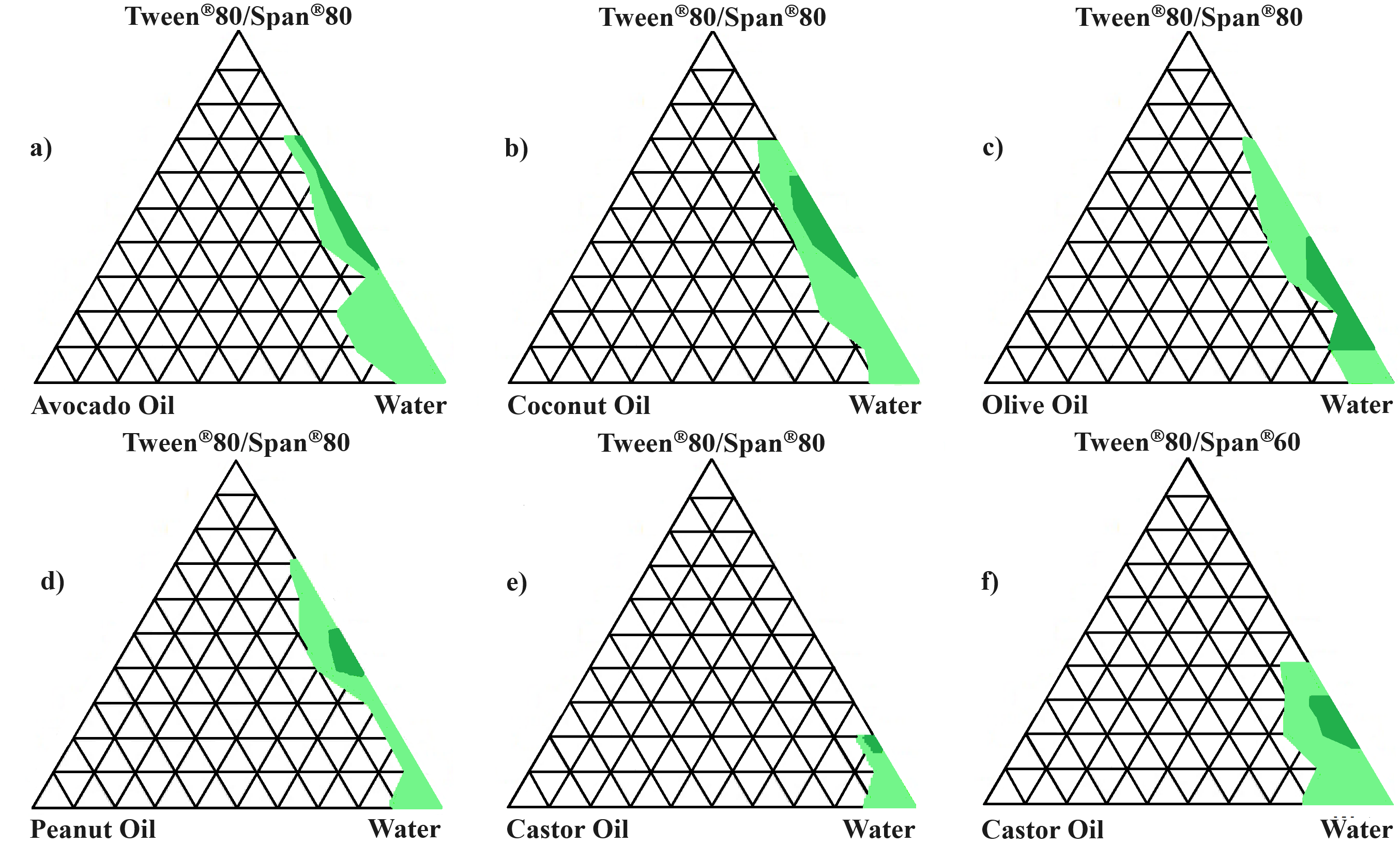 Fig. 2.
Fig. 2.Pseudoternary phase diagrams. (a) Avocado oil, ART/LUM,
Tween
All SEDDSs containing sodium lauryl sulfate (SLS) as the surfactant were omitted from further analysis because these systems produced either a narrow range to experiment with or unstable spontaneously formed emulsions. SLS was initially chosen as a surfactant due to its significant hydrophilicity (HLB value = 40) and ability to form o/w emulsions [60]. However, the oils tested possess HLB values between 6 and 12, making them notably more lipophilic [108]. Generally, the HLB system is a simple scientific method for forecasting the optimal surfactant/co-surfactant combination needed to create an ideal emulsifying system. An optimal and stable emulsifying system is founded when the HLB values of the selected oil and surfactant phases match [61, 108, 109, 110, 111]. In this case, the unstable formulations obtained are probably due to the HLB values not complementing one another; thus, the hydrophilicity of the components does not correspond.
The remaining SEDDSs were also visually examined as described during the preformulation phase. Optically clear emulsions were deemed acceptable for further analysis (Fig. 3a), whereas SEDDS formulations depicting crystallization, precipitation (Fig. 3b), or other instabilities were disregarded.
 Fig. 3.
Fig. 3.Microscopic SEDDS examples. (a) Acceptable SEDDS formulation. (b) Undesirable SEDDS that either started to precipitate or form crystals.
Based on the pseudoternary phase diagrams and photographic examination, SEDDSs
that adhered to the set principles, and in which the quantity of oil phase
entirely solubilized the ART/LUM FDC, were reserved at 25 °C for 24 h to
identify any phase separation. All SEDDSs comprising a surfactant phase of
Tween
| SEDDS | Assay (%) | Droplet size (nm) | Zeta potential (mV) | Self-emulsification grading | Self-emulsification time (s) | Cloud point (°C) | Viscosity (mPa.s) | |
| ART | LUM | |||||||
| AVO4:6 | 105.513 | 96.553 | 241.3 | –43.8 | A | 35 | 62 | 402.1 |
| CAS2:8S80 | 95.493 | 95.200 | 524.1 | –44.6 | B | 59 | 55 | 372.0 |
| CAS3:7S60 | 96.723 | 95.735 | 249.4 | –20.8 | C | 120+ | 55 | 312.0 |
| CCT6:4 | 105.600 | 96.310 | 776.2 | –46.6 | A | 55 | 45 | 200.0 |
| OLV3:7 | 107.588 | 96.000 | 241.2 | –54.7 | A | 40 | 60 | 510.2 |
| PNT6:4 | 112.375 | 96.211 | 1452.7 | –45.7 | A | 50 | 60 | 278.8 |
The International Pharmacopoeia (IP) affirms that oral dosage forms comprising
ART and LUM should have an individual drug concentration between 90% and 110%
[112]. From Table 4, it is clear that, in general, ART depicted a higher drug
content percentage in SEDDS formulations compared to LUM. Nonetheless, the drug
content percentage for both drugs fell well within the set criteria, except for
the SEDDS that comprised PNT, i.e., PNT6:4 (112.375% ART). Furthermore, the
concentrations of ART and LUM (to a lesser extent) in CAS SEDDSs were notably
lower compared to the other SEDDS formulations, regardless of the surfactant
included. This is possibly due to the reduced solubility of both ART and LUM in
CAS (Table 3). Moreover, no significant (p
Droplet size, size distribution, and zeta potential mainly influence drug
delivery employing SEDDSs [42, 113, 114]. Size classification is one of the most
perceptive trials carried out during SEDDS development since size affects drug
release, SEDDS stability, and absorption [114]. As the droplet size decreases,
the available interfacial surface area increases, which sequentially advances
abrupt drug release into an aqueous milieu for drug absorption [58, 115, 116].
Gershanik et al. [117] explored the effect of emulsion droplet size on
the diffusion capacity of SEDDSs through the intestinal mucosa. They established
that the best possible droplet size range is between 100 and 500 nm. AVO4:6,
CAS3:7S60, and OLV3:7 SEDDSs each depicted a droplet size smaller than 250 nm,
rendering them in the nanosize range and defining these formulations as SNEDDSs
(Table 4). Additionally, these SNEDDS formulations depicted a %RSD of
Only PNT6:4 presented an average droplet size in the micro range (1452.667 nm) and a relatively wide droplet size distribution (16.305% RSD). Therefore, PNT6:4 will most probably not be able to diffuse into the intestinal mucosa to the same degree as the SNEDDSs that make up the other oil phases and could also be unstable. Moreover, although the CCT6:4 and CAS2:8S80 formulations fall within the nano size range, they will presumably also not be efficient in traversing the intestinal film to the same limit as the accepted SNEDDSs. The larger average droplet sizes achieved with PNT6:4, CCT6:4, and CAS2:8S80 could be acredited to the oil–surfactant phase fraction. Normally, the droplet sizes enlarged as the oil phase concentration increased and the surfactant phase declined. When droplet size increases, it can lead to flocculation or coalescence of the oil droplets, which, in turn, will probably cause SEDDS instabilities [109]. Thus, these SEDDSs were not deemed acceptable in terms of droplet size and size distribution in this study.
Progressively positive or negative zeta potential values (in other words
Self-emulsification efficacy may be denoted as dispersibility assessment [123, 124, 125]. Instantaneous emulsification is highly promising if detected using SEDDSs aimed at oral administration. This is because spontaneous emulsification is acknowledged as the rate-limiting step that has to transpire before effective absorption [125]. Additionally, when self-emulsification is initiated quickly, it indicates that only a small sum of energy was needed to form the said emulsion; that is, no heating was necessary, for example [58, 126, 127]. In other words, the self-emulsification time of a SEDDS formulation demonstrates the free energy required to permit self-emulsification that can be induced by the energy-reducing aptitudes of emulsifying agents, which directs the collapse of formulations [128]. The literature established that natural emulsification could transpire immediately or be delayed, subject to the existence of kinetic obstacles between excipients contained in SEDDSs [71]. Furthermore, kinetic barriers can be overcome by applying mild agitation or heat [71], but this should not be required for forming oral SEDDS (mild agitation will be present within the gastrointestinal tract). Thus, given the emulsification behavior grading system for SEDDSs, as demonstrated in Table 1, it could be concluded that SEDDSs, which achieved an A or B grading, could be judged favorable for successful oral delivery of the ART/LUM FDC. However, SEDDSs that showed slow and/or weak emulsification characteristics, with resulting C, D, or E grading, were reasoned inapt.
Hence, CAS3:7S60 (Table 4, highlighted and in bold) was rejected in terms of oral drug delivery owing to an acquired C grading (Table 1). As stated previously, a stable emulsifying system is normally established when the surfactant phase’s HLB value, especially when synthetic surfactants are used, is near the HLB value required of the oil phase [61, 108, 109, 110, 111]. This study calculated the HLB value of the surfactant phase included in CAS3:7S60 as 9.85. CAS, however, has an HLB value of 14, thus indicating a significant difference in HLB values and, therefore, consenting a safe hypothesis to be formed, namely, that the surfactant phase is not effective enough to successfully lower the interfacial tension. The necessary energy to naturally emulsify the formulation consequently favored the diffusion surface area and not the entropy variations of the emulsion.
Viscosity describes the internal resistance of a fluid that can affect flow
resistance as well as spontaneous emulsification [129, 130]. In general, the lower
the viscosity, the faster the rate of emulsification, which, sequentially, may
impact the in vivo release of a drug and succeeding bioavailability
[6, 78]. Overall, it was found that viscosity increased as the surfactant phase
ratio increased (Table 4). The included natural oils also notably influenced the
viscosity but did not influence it to the same extent as the surfactant phase.
According to the type of oil incorporated, these obtained viscosity values could
be graded as OLV
All the SEDDSs additionally depicted plastic rheological properties. Most pharmaceutical formulations are manufactured to be pseudoplastic, which makes them advantageous. The justification is that the high viscosity at a lower shear allows for the insoluble particles in the SEDDSs to be stabilized, which then inhibits these particles from promptly sedimenting. Moreover, pseudoplastic flow has a shear-thinning property that grants simple flow when emptying the emulsion container [131]. Consequently, this rheological performance is valuable as it permits the formation of a more stable SEDDS, especially during storage. It furthermore assists with simple use and pouring during administration.
The cloud point indicates the temperature at which SEDDS formulations cannot
maintain their spontaneous emulsification properties [132]. Erratic drug release
and irreversible phase separation occur due to dehydration of the incorporated
constituents at elevated temperatures above the identified cloud point. These
occurrences can harshly influence drug absorption [77, 133]. For these reasons,
the cloud point for an oral SEDDS ought to be greater than body temperature
(i.e.,
The surfactants incorporated into the SEDDS formulations cannot guarantee physical stability due to the intricacy of the emulsified system supported by the included surfactants. These compounds generate interfacial tension slopes and offer adjustment to the mobility of droplets [134]. Therefore, physical SEDDS stability was examined during thermodynamic stability testing [58, 63, 79].
No physical instabilities were observed when the AVO4:6, CAS2:8S80, CAS3:7S60,
and OLV3:7 SNEDDSs were exposed to differing temperatures. Alternatively, the
CCT6:4 SEDDS solidified once refrigerated owing to the modest melting point
(
The different SEDDSs were then exposed to centrifugation to determine if these formulations could withstand kinetic stress conditions. On visual inspection, no kinetic instabilities were identified, which signified that all tested SEDDSs could be considered acceptable when exposed to the centrifugation experiment. For the last thermodynamic stress test, the selected SEDDSs were diluted to evaluate their robustness, as this simulates comparable circumstances that arise in the gastrointestinal tract upon oral administration [78]. Again, no indication of any physical instabilities of the SEDDSs could be detected.
The dissolution profiles of the commercial product and the SEDDS formulations
were related. It was found that ART release from all the SEDDSs differed
statistically significantly (f
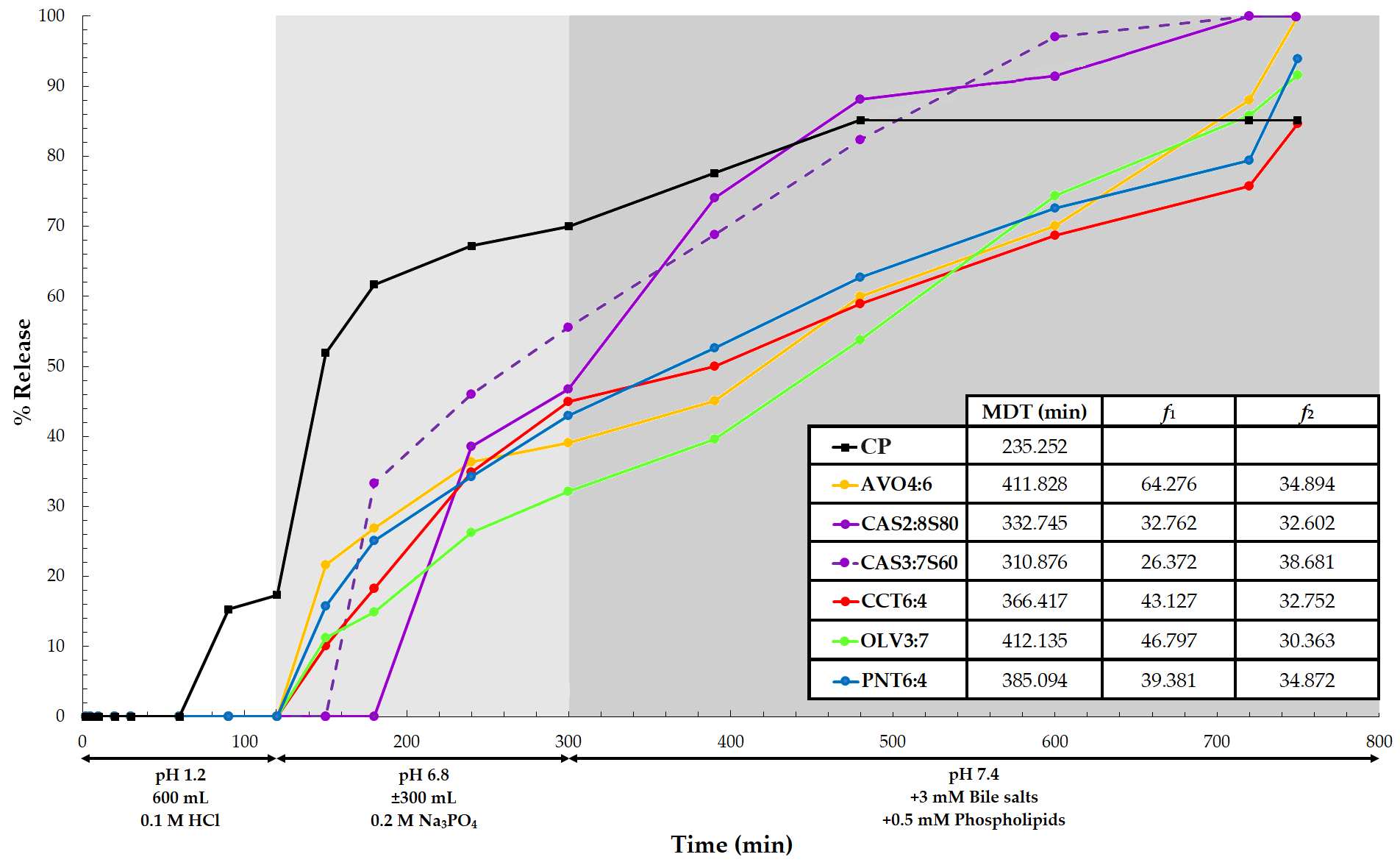 Fig. 4.
Fig. 4.ART release profiles for the respective SEDDS formulations tested. The shaded areas and interpretations express the time points where changes in the dissolution media were made (n = 6). The abbreviation CP indicates the drug release of the commercial product.
In contrast, all the tested SEDDS formulations depicted initial delayed ART
release profiles until approximately 120 min, despite being liquid formulations,
and this coincided with when the pH of the dissolution medium was changed from
1.2 to 6.8. This is further highlighted by the notably high MDT values (Fig. 4)
achieved for the different SEDDSs. Furthermore, all SEDDSs portrayed release
characteristics dependent on an increase in pH and, to a lesser extent, the
presence of biorelevant media at 300 min when pH was again adjusted from 6.8 to
7.4 after adding phospholipids and bile salts. Moreover, SEDDS formulations that
included CAS showed a slower onset of drug release, signifying that this oil had
the most retardation properties of ART release. However, once ART release was
initiated, these SEDDSs were able to release more ART (100%) faster compared to
the other SEDDSs (f
Considering the fit factors (Supplementary Material, Supplementary Table 1) between the selected SEDDS formulations tested, only the SEDDSs comprising CAS, irrespective of the surfactant phase included, differed statistically significantly from the other SEDDS formulations. A possible explanation for the differences observed in the various ART release profiles may be the noticeable varying ART solubility in the selected oil phases. ART is meaningfully less soluble in CAS, whereas higher and more comparable solubility results were obtained for the other oils. Meager solubility can cause faster drug release from a preparation (CAS MDT values are relatively faster than most other formulations), whereas improved solubility in a dispersed phase can deter a drug from diffusing from the formulation [135].
Regarding the dissolution profiles constructed for LUM from the different formulations (Fig. 5), it is again clear that LUM was released only after the biorelevant medium was added, with a subsequent meaningful increase in the pH to 7.4 (i.e., after 300 min and even slower than ART). Furthermore, LUM could not be quantified for the commercial product tested, and thus, no comparison to the control could be made.
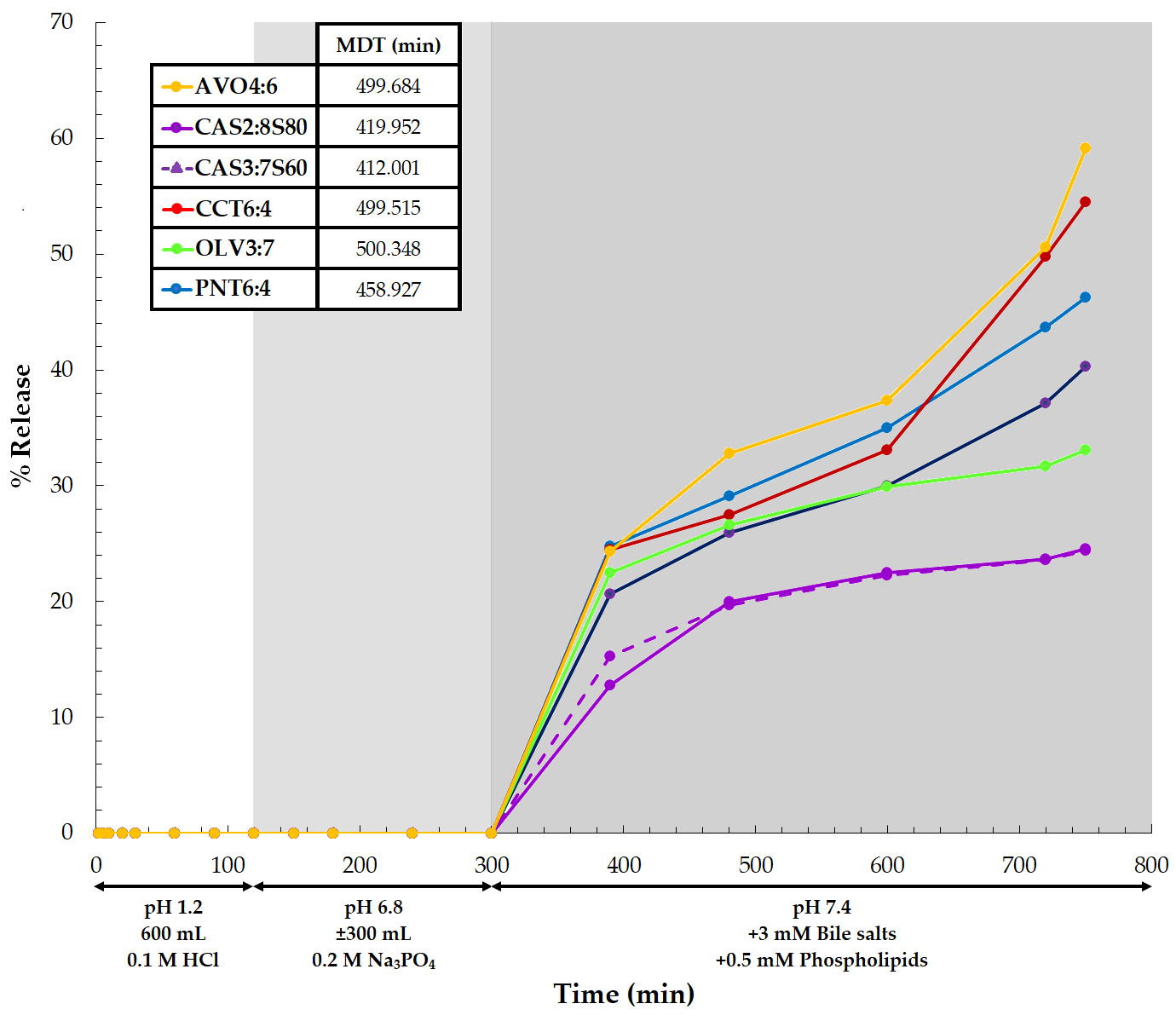 Fig. 5.
Fig. 5.LUM release profiles for the individual SEDDS formulations analyzed. The colored regions and explanation direct the time points for changes in the dissolution media (n = 6).
LUM possesses a log P of 9.19 [60] and log D values (distribution coefficient) of 8.9 and 10.1 at pH 6 and 7.4, correspondingly [136]. These values reflect its noticeable affinity for an organic or lipophilic phase, which, in turn, provides a basic explanation for its poor detection during performed dissolution studies when compared to ART. However, it could be concluded that the type of phospholipid and its concentration included in the progressive dissolution media were adequate to enhance the wettability of LUM. The increased wettability led to high enough LUM concentrations in the aqueous phase for quantifiable detection when the SEDDS formulations were analyzed. Bile salts and phospholipids are said to implicitly affect the solubility and ensuing bioavailability of poorly soluble compounds owing to bile salts and phospholipids being physiologically significant surfactants. Therefore, the addition of synthetic surfactants to SEDDS formulations appeared to noticeably decrease the interfacial friction between the SEDDS excipients and the immediate fluid [137]. The interfacial tension could subsequently be reduced due to the entropy changes that favor the dispersion because of the combination of surfactants in the dissolution medium.
However, the biorelevant media employed was a simple preferential constitution to imitate the milieu in the small intestine and not to amend or enhance the dissolution of the two drugs utilized. Literature normally suggests the use of two different biorelevant media types when analyzing drug dissolution, that is, Type I and Type II. Type I simulates the fasting state in the gastrointestinal tract, whereas Type II media mimics the fed state. Here, Type I dissolution media was utilized for drug release experiments because (and as mentioned before) patients contracting malaria normally reside in rural areas; taking a suitable nutritious fatty meal during treatment with the relevant medication is seldom plausible [138]. Thus, it was attempted to establish whether ART and LUM could be released in the fasted state. However, the use of Type I media can be unfavorable in terms of the effective release of highly lipophilic drugs such as LUM during dissolution studies. Comparatively, Type II dissolution media include lecithin, which facilitates lipid digestion, making this dissolution medium more advantageous for the solubilization of lipophilic drugs. Nevertheless, various enzymes are found in the gastrointestinal tract that will normally facilitate lipid digestion and probably improve LUM release from the SEDDS formulations after oral dosing [138]. In addition, there are lipid transport systems in which chylomicrons and micelles aid in carrying the lipids to the absorption region [139]. Therefore, numerous additional mechanisms in the digestive system can contribute to the normally improved bioavailability of LUM in situ. Consequently, although the LUM release profiles attained during experimentation show inapt drug release, it could still be considered effective as LUM was sufficiently released for quantification from all the SEDDS formulations (though significantly slowly). Literature additionally stated that LUM displays delayed absorption and elimination [140]. This property is beneficial as LUM targets the blood schizonticide stage of malaria and presents no antimalarial activity against the pre-erythrocytic liver stages. Consequently, when taken together with ART, LUM will eliminate all residual parasites once ART has diminished the initial parasites [140, 141]. For this reason, having a drug delivery system with delayed LUM release characteristics may aid in treating malaria.
Overall, the LUM concentrations released from the different SEDDSs were
significantly lower compared to the ART released from the same formulations.
Nonetheless, all the SEDDS formulations again fit the Peppas-Sahlin 2 with
T
In summary, all tested SEDDSs could release both ART and LUM more effectively
than the commercial product. An average lag time (ALT) and MDT of 135 min and
369.849 min, respectively, were shown for when ART was released from the analyzed
SEDDSs. These results indicate a faster release rate compared to the release of
LUM (ALT = 300 min; MDT = 465.071 min). Moreover, a significantly higher amount
of ART (95%) was released relative to LUM (40.3%). No distinct proportional
trends could be recognized for the drug concentrations released from the chosen
SEDDSs. Both drugs also displayed release kinetics from the different SEDDS
formulations that could be related to the Peppas-Sahlin 2 with T
Preformulation studies indicated that individual solubilities were exponentially
improved by simply dissolving both ART and LUM in the selected natural oils.
Moreover, the stability of the six chosen emulsions and nanoemulsion areas
depended on the type of surfactant phase incorporated. Except for the SEDDS
formulation that included PNT as an oil phase, these emulsions could be
classified as SNEDDSs. Additionally, all six formulations exhibited pseudoplastic
rheological flow behavior, which is ideal for pharmaceutical preparations, as
this type of flow behavior can stabilize any insoluble particles in an emulsion,
preventing sedimentation of the said particles [143]. These formulations also
exhibited quantifiable ART and LUM release. However, all release profiles
indicated that delayed responses for both drugs ensued until the pH of the
dissolution fluid was augmented, rendering the drug release pH-dependent.
Furthermore, all release profiles of both drugs fit the Peppas–Sahlin 2 with
T
ANOVA, Analysis of Variance; ART, Artemether; AVO, Avocado Oil; BCS,
Biopharmaceutical Classification System; CA, California; CAS, Castor Oil; CCT,
Coconut Oil; f
All data and materials used have been included in this paper.
Conceptualization of the study was done by JMV and LHdP. Methodology was planned and executed by JMV and LC. LC furthermore performed the research/investigation. JMV was responsible for data curation and software. Validation of the study was completed by JMV, LC and LHdP. Following, LC and JMV conducted the formal analysis for this research. Resources were procured by JMV. JMV was responsible for the writing — original draft preparation; and JMV and LHdP were responsible for the writing — review and editing. JMV and LC were in charge of the visualization. Overall supervision and project administration were conducted by JMV and LHdP. Funding was acquired by JMV. All authors have read and agreed to the published version of the manuscript. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity.
Not applicable.
The authors would like to sincerely thank the Centre of excellence for
Pharmaceutical Sciences (Pharmacen
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.