1 Department of Symbiotic Nitrogen Fixation, Institute of Plant Physiology and Genetics of the National Academy of Sciences of Ukraine, 03022 Kyiv, Ukraine
2 European Regional Centre for Ecohydrology of the Polish Academy of Sciences, 90-364 Lodz, Poland
Abstract
Background: Rhizobial inoculation in combination with
fungicidal seed treatment is an effective solution for improving soybean
resistance to modern climate changes due to the maximum implementation of the
plant’s stress-protective antioxidant properties and their nitrogen-fixing
potential, which will contribute to the preservation of the environment.
Methods: Model ecosystems at different stages of legume–rhizobial
symbiosis formation, created by treatment before sowing soybean seeds with a
fungicide (fludioxonil, 25 g/L) and inoculation with an active strain of
Bradyrhizobium japonicum (titer 10
Keywords
- Bradyrhizobium japonicum
- soybean (Glycine max (L.) Merr.)
- malondialdehyde
- superoxide dismutase
- catalase
- nitrogen-fixing activity
- nodulation
- drought
Strategically important issues today are the obtaining high and stable crop yields by creating rational technologies for their cultivation. Specifically, crop production is of strategic importance for the world global economy. Legumes are a key ingredient in food and are an increasingly a renewable raw material for industrial and energy purposes [1]. The formation of their high productivity, like most cultivated plants, is largely determined by the complex influence of external factors, among which the main role is played by global climate changes, the spread of phytodiseases, environmental pollution by agrochemicals [2, 3].
Soybean is one of the most highly profitable advanced agricultural crops in the world [http://soystats.com/composition-of-a-soybean/]. It is one of the most widely grown and consumed oil crops, with its uses ranging from human food to animal feed, industrial products, and ingredients to precursor materials [1]. Soybeans are grown on approximately 119 million hectares worldwide, with a total annual production of 319 million tons, and the share of soybeans in global oilseed production is about 55% [FAOSTAT 2013–17].
Global climate changes and frequent droughts worldwide increase agricultural risks [2], including soybean growing. Under modern climate change conditions, a reduction in the yield of cultivated plants by more than 50% of their potential productivity is possible, which can constitute serious food security issues [3]. Therefore, studying the reactions and formation of the adaptation potential of cultivated plants, including soybeans, to climate change is of great economic and social importance.
Realizing the potential productivity of leguminous crops is one of the main concerns in ensuring their high yields and is determined by a harmonious combination of all modern methods: organizational and economic, agrotechnical, immunological, biological, and chemical [4, 5]. One of the important technological techniques used in growing soybeans is poisoning seeds with fungicidal drugs since the loss of seed yield from damage to soybeans by phytopathogens is up to 40% [6, 7].
Decisions regarding the protection of cultivated plants should be aimed at finding ways to reduce their harmfulness to the environment. Hence, the productivity of cultivated plants can be increased via the maximum mobilization of their stress-protective properties and the realization that genetics represent the most effective and economically justified solution for environmental and plant protection. The main criteria for the competitiveness of agricultural products on the world market should be the economic criteria of its value and strict control over the quality and safety of products. In this aspect, active research is being conducted on the possibility of using seed inoculation with nodule bacteria and fungicides to regulate the metabolism of leguminous crops and increase their tolerance and productivity [8, 9, 10, 11, 12].
The efficiency of the functioning of the legume–rhizobial symbiosis under unfavorable growing conditions depends on the ability of the host plant to induce its antioxidant protection systems. This leads to adaptive adjustments in the plant metabolism and increases its tolerance [13, 14]. On the one hand, this can negatively affect the formation and functioning of the symbiosis. Moreover, it can stimulate the development of plant protective reactions and their adaptation to stress.
Antioxidant defense systems play a key role in plant adaptation to unfavorable factors [15]. Among them, considerable attention has been paid to the role of antioxidant enzymatic complexes involved in the neutralization of reactive oxygen species (ROS), the accumulation of which in a plant cell under stress initiates the processes of oxidative destruction of membrane structures [16]. At the same time, it has been proven that ROS can act as signaling molecules involved in activating defense systems under stress, particularly inducing the synthesis of antioxidant enzymes [17]. Therefore, studying the role of antioxidant enzymes in maintaining a stationary level of free radical processes in the cell under the effects of drought is important for developing protective properties of plants, including when establishing mutualistic relationships.
The presented research approach proves that using active Bradyrhizobiumvirulent strains in combination with a fungicide is important for increasing the implementation of protective and nitrogen-fixing properties in soybeans under drought conditions. Research in this aspect is important for creating effective symbiotic systems involving active strains of nodule bacteria using the treatment of seeds with fungicides as an alternative means to provide plants with environmentally friendly nitrogen while increasing their tolerance to appropriate growing conditions.
This work aimed to investigate the effect of the pre-sowing seed treatment with fungicide and inoculation with rhizobia on the development of stress-protective properties in soybeans and the efficiency of the symbiosis functioning under optimal plant growth and drought conditions. Our research includes elements of modern soybean cultivation agrotechnologies in the context of obtaining ecologically and economically safe crops.
The study was based on the following hypothesis: pre-sowing seed treatment with fungicide and active rhizobia will increase the antioxidant properties of plants, which will ensure improved tolerance and productivity of soybean symbiosis with B. japonicum under drought conditions.
The presented study emphasizes the need for a comprehensive and multidimensional understanding of the abovementioned processes when choosing the most effective means for creating stress-resistant legume–rhizobial systems.
Soybeans (Glycine max (L.) Merr.) of the Almaz variety (early maturing variety) were selected for the study (originator—Poltava State Agrarian Academy, Ukraine) from the 2007 Register of Plant Varieties of Ukraine and recommended for cultivation in the forest steppe of Ukraine. Before sowing, soybean seeds were treated with a solution of fludioxonil (25 g/L), after which they were inoculated with a suspension of rhizobia from the active virulent Tn5 mutant B1-20 for an hour. The inoculation titer was 200–300 thousand rhizobia cells per seed. The control was soybean plants of the variant with inoculation of seeds with rhizobia without fungicidal treatment, which were grown under optimal moisture supply conditions.
An active, virulent Tn5 mutant of Bradyrhizobium japonicum B1-20 was created by transposon mutagenesis from the original strain 646 using Escherichia coli S17-1 with the plasmid pSUP5011, which contains the Tn5 transposon (pSUP5011::Tn5mob) at the Institute of Plant Physiology and Genetics of the National Academy of Sciences of Ukraine.
Nodule bacteria were cultivated in liquid yeast mannitol agar in 750 mL flasks
in a shaking device (220 rpm) at 26–28 °C. The B. japonicum
culture in the exponential growth stage (92–96 h) was used as an inoculum. The
amount of inoculum added to the flasks was 2% of the medium volume. The amount
of rhizobia in the suspension added to 200 mL of culture medium was 10
The research was conducted as a pot experiment. Soybean seeds were planted in pots without holes at the bottom, weighing 10 kg. The substrate for plant growth was sterile sand. Plants were grown on Herligel’s nutrient mixture of macro- and microelements with a nitrogen rate of 0.25, which was added to sterile sand.
Soybean plants inoculated with bacteria were grown under natural light and optimal water supply, which was maintained at 60% total moisture content (TMC) by controlled watering. The amount of watering the plants received was calculated based on the mass of the empty pot, the dry mass of sand (per pot), and its moisture content. Before filling the pots, sterile sand was analyzed for moisture content (3.12%) and capacity (19.67%).
Three stages characterized the period of active nitrogen fixation by the soybeans: third trifoliate (40 days after sowing), beginning flowering (46 days after sowing), and full bloom (52 days after sowing). The plants were subjected to drought for 12 days, comprising the five days of the third trifoliate stage, which continued for seven days into the beginning flowering stage. Control plants received 60% TMC—optimal water supply (control) during this period. After this period, i.e., in the soybean full bloom stage, the stressed plants were provided optimal watering (60% TMC) for a seven-day restoration period (Fig. 1). Nodule samples for analysis were taken in stages: third trifoliate (V3), beginning flowering (R1), and full bloom (R2). Plants grown under conditions of optimal water supply (60% TMC) were used as the control.
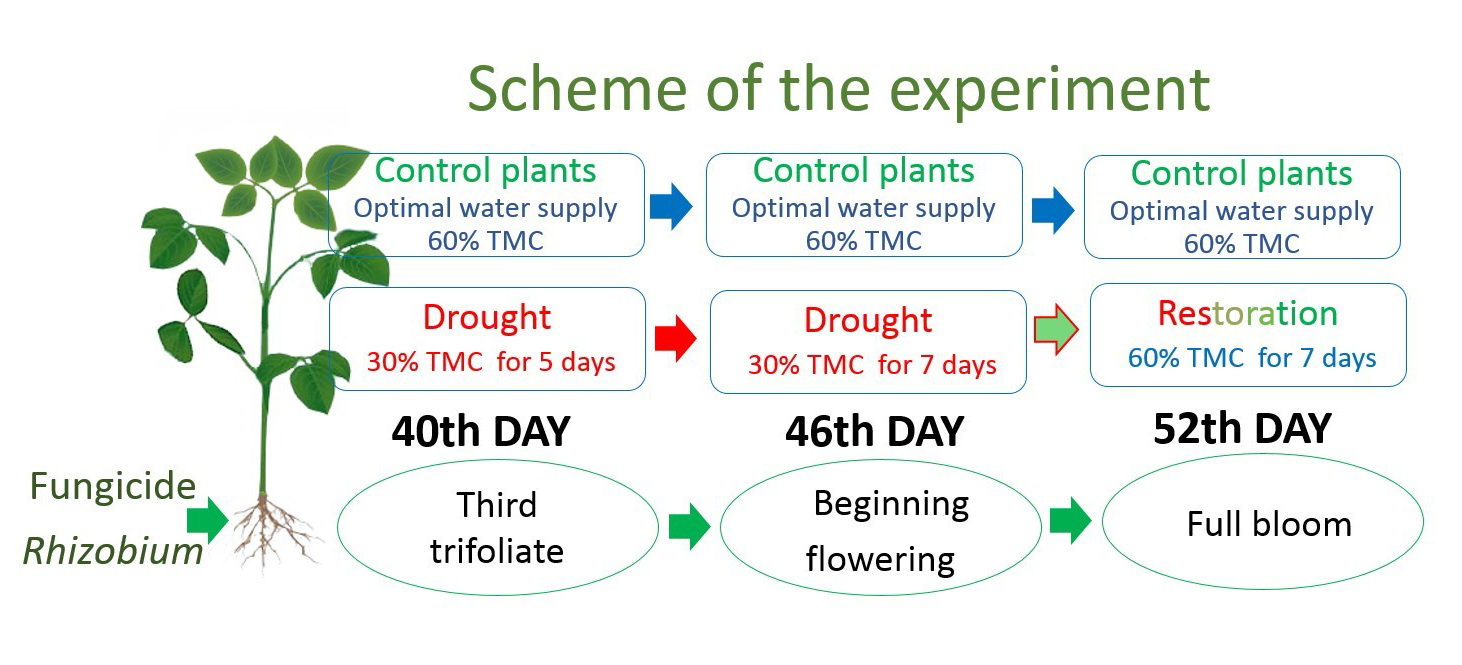 Fig. 1.
Fig. 1.Schematic representation of plant growth experiment under controlled conditions. Soybean under optimal water supply (60% TMC) and drought (30% TMC). 40th day, 46th day, 52nd day—days after soybean growth stages: third trifoliate (V3), beginning flowering (R1), and full bloom (R2). TMC, total moisture content.
Soybean sowing was carried out in early May to a depth of 3–5 cm at a rate of
40 seeds per 1 linear meter, while the upper sowing layer of soil was heated to
10–15 °C (Fig. 2). Plots were placed randomly. The
accounting area was 5 m
 Fig. 2.
Fig. 2.Schematic representation of the field experiment with soybeans. Data were recorded on the 56th and 72nd days, days after soybean growth stages: full bloom (R2) and pod formation (R4).
The nitrogen-fixing activity was analyzed using an Agilent Gas chromatograph (GC) System 6850 gas
chromatograph (Agilent, Santa Clara, CA, USA) with a flame ionization detector [18]. Gas separation was
carried out on a column (Supelco Porapak N) at a thermostat temperature of 55
°C and a detector temperature of 150 °C. The carrier gas was
helium (20 mL/min). The analyzed gas mixture sample volume was 1 cm
The amount and mass of the soybean nodules determined the nodulation activity.
The level of lipid peroxidation (LPO) was determined by changing the content of
its final product, malondialdehyde (MDA), as a result of a reaction with
thiobarbituric acid by the Shimadzu UV-1900 scanning two-beam spectrophotometer
(Shimadzu Corporation. Kyoto, Japan) at
The dry matter mass was determined by drying samples of aboveground and underground plant parts to a constant value at +105 °C.
To obtain the enzyme extract, a portion of the plant material was triturated
(ratio 1:2) with a cooled 0.5 M Tris HCl buffer (pH 7.8) containing 5 mM
The Superoxide Dismutase (SOD) activity in the supernatant (EC 1.15.1.1) was determined by its ability
to inhibit the photochemical reduction of nitroblue tetrazolium [21]. The
reaction mixture contained 50 mM phosphate buffer (pH 7.8), 13 mM methionine, 2
µM riboflavin, 63 µM p-nitroblue tetrazolium, 0.1 mM EDTA, and 100
µL of enzyme extract. The reaction proceeded for 15 min at a light
intensity of 70 µmoL quanta/(m
Total soluble protein content was determined using the Bradford assay [22]. The soluble protein content was measured using bovine serum albumin based on the standard curve method.
The acidic forms of peroxidase were separated by disc electrophoresis in a 10% polyacrylamide gel in vertical blocks at 4–6 °C in 0.2 M Tris–glycine (pH 8.3) buffer [23]. Bromophenol blue was used as the leading colorant. To visualize the peroxidase isoforms, the gel was incubated for 10 min in a cooled 5 mM benzidine solution in Na–acetate buffer (pH 5.2), washed with distilled water, and incubated in 0.3% hydrogen peroxide solution until blue coloration appeared. Electrophoresis was performed in 5-fold repetition.
The grain harvest was recorded by manual harvesting and then weighed.
All statistical analyses were performed using STATISTICA ver. 13.3 software
package [24]. The significance of differences in the physiological parameters was
tested, and the results of the tests are presented in the Figures and Tables as
Detailed information about the reagents and equipment used in this study is provided in the supplementary materials (see Supplementary Table 1).
Prolonged drought promoted an increase in MDA in soybean nodules inoculated with rhizobia without fungicide seed treatment by 126.8% (third trifoliate stage) and by 153.7% (beginning flowering stage), compared to its concentration in the control soybeans under optimal watering, 60% TMC (Fig. 3). The use of seed inoculation combined with fungicide treatment led to a 93.6% increase in MDA content in soybean nodules under moderate drought conditions in the third trifoliate stage and a 138.5% increase under prolonged drought in the beginning flowering stage compared to the control (60% TMC) (Fig. 3).
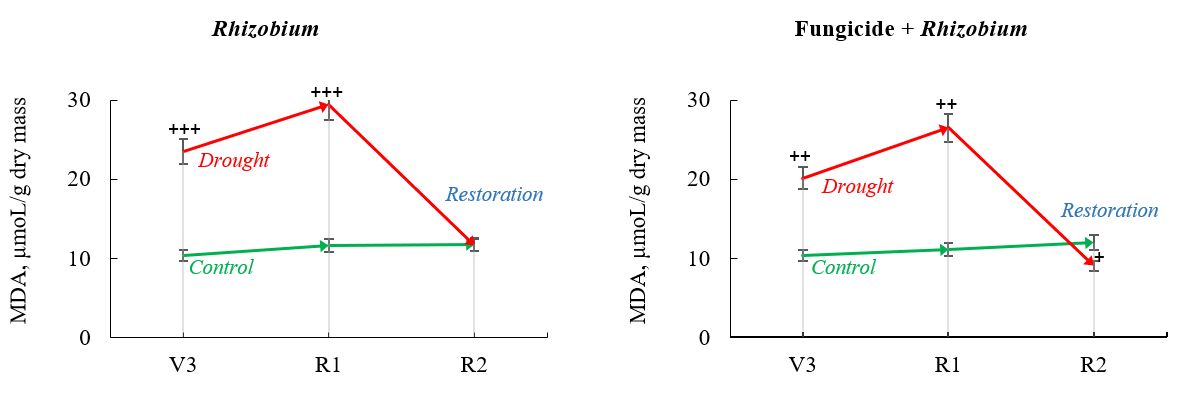 Fig. 3.
Fig. 3.Malondialdehyde (MDA) content in soybean nodules after
fungicide seed treatment and Rhizobium inoculation under optimal
watering—control (green) and drought/restoration (red) conditions. The arrows
indicate trends progressing from the third trifoliate (V3) stage to the beginning
flowering (R1) and full bloom (R2) stages. Data compared to the control (optimal
watering) are probable by
In the post-stress period, nodule MDA decreased by 24.3% when seeds were treated with both fungicide and inoculant. In the variant with rhizobia inoculation without fungicide, the MDA concentration was similar to that in plants unaffected by drought (Fig. 3).
The drought caused an increase in the SOD activity by 38.2–40.6% in soybean nodules inoculated with rhizobia without fungicide treatment, compared to the activity of the enzyme in the control variant with optimal watering (Fig. 4A). An increase in SOD activity was also found in soybean nodules formed by active rhizobia after seed treatment with a fungicide. Moreover, there was 32.7% higher enzyme activity under moderate moisture deficit and 16.5% under long-term stress compared to the control (Fig. 4A). In the post-stress period, SOD activity in soybean nodules was 20% higher than the control in both variants exposed to drought.
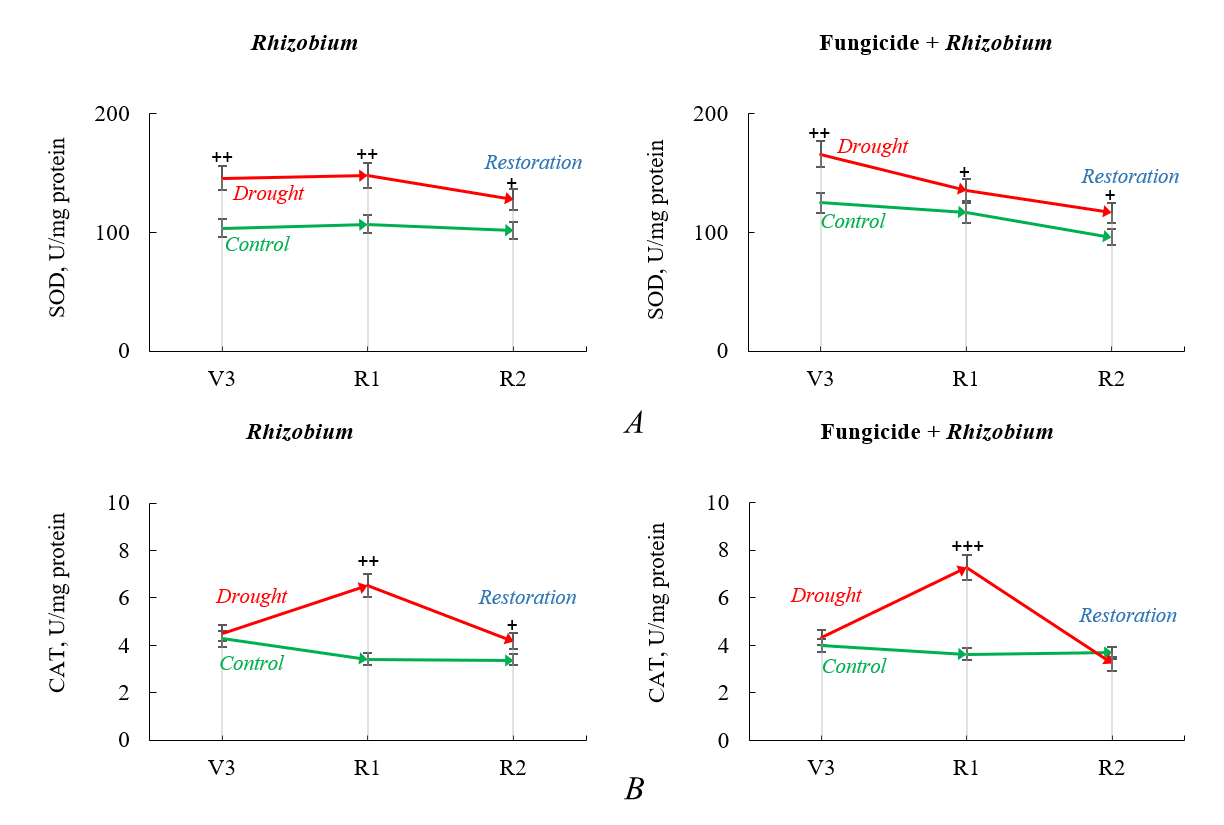 Fig. 4.
Fig. 4.Superoxide dismutase (SOD) (A) and catalase (CAT) (B) activity
in soybean nodules after fungicide seed treatment and Rhizobium
inoculation under optimal watering—control (green) and drought/restoration
(red) conditions. The arrows indicate trends progressing from the third
trifoliate (V3) stage to the beginning flowering (R1) and full bloom (R2) stages.
Data compared to the control (optimal watering) are probable by
At the third trifoliate stage, under moderate stress, there was no statistically significant difference in the levels of CAT activity in soybean nodules between the experimental variants (Fig. 4B). During prolonged drought in the beginning flowering stage, an increase in enzyme activity was observed in the soybean nodules of both experimental variants. The activity of CAT in soybean nodules increased by 100.8% under seed inoculation with rhizobia and fungicide treatment and by 90.6% when using seed bacterization without fungicide, compared to the enzyme activity in soybeans under optimal watering (60% TMC) (Fig. 4B). After restoring watering of the plants, CAT activity was increased by 22.3% in soybean nodules inoculated with rhizobia, but without fungicide seed treatment compared to the enzyme level in soybean nodules not affected by drought (control, 60% TMC) (Fig. 4B). In fungicide- and inoculant-treated soybeans, the level of CAT activity in nodules was restored to the optimal level as in the control group.
The study of molecular genetic polymorphisms in enzymes is a method used to identify the metabolism specificity of various plant types. The peroxidase family is characterized by polyfunctionality and high heterogeneity of its isoenzyme system, and its isoenzymes can be uniquely represented in different plant genotypes.
Under optimal water supply, no significant differences in the molecular spectra of peroxidases between the investigated variants were recorded. However, under the influence of drought, the electrophoreogram shows a clear increase in peroxidase fractions when the fungicide was applied without rhizobia inoculation, compared to other variants (Fig. 5).
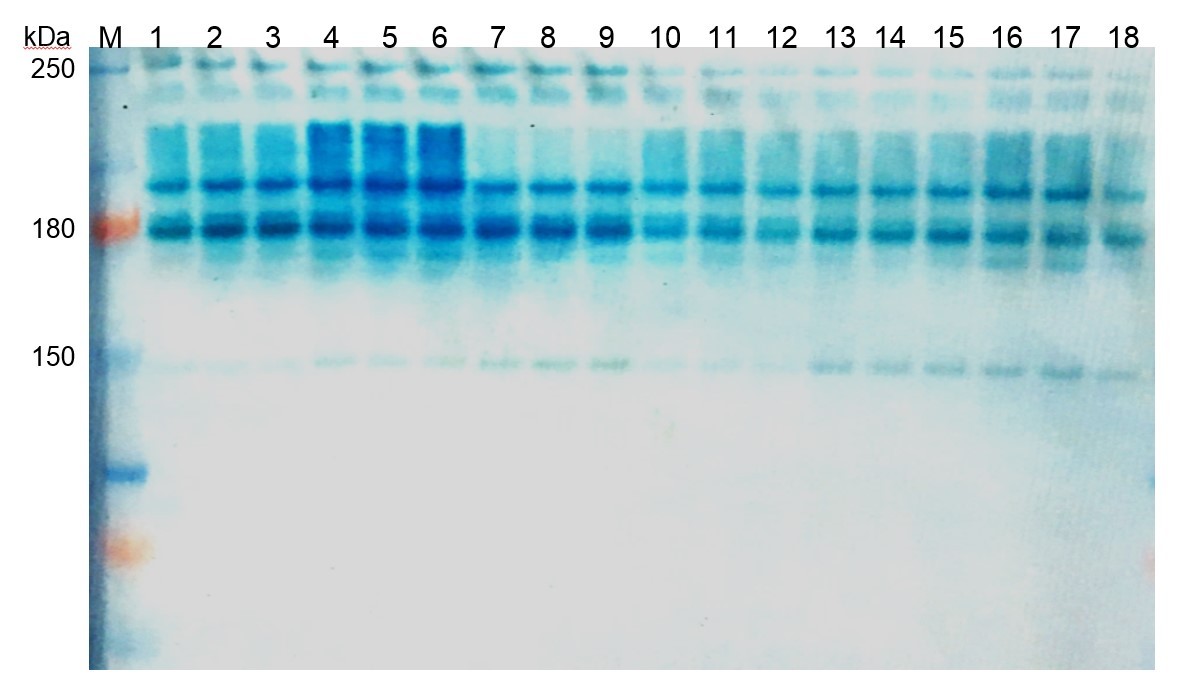 Fig. 5.
Fig. 5.Electrophoretic zymogram of peroxidase in soybean nodules in the third trifoliate stage. M—marker of protein masses (10–250 kDa); 1, 2, 3—rhizobia, 30% TMC; 4, 5, 6—fungicide, 30% TMC; 7, 8, 9—fungicide + rhizobia, 30% TMC; 10, 11, 12—rhizobia, 60% TMC; 13, 14, 15—fungicide, 60% TMC; 16, 17, 18—fungicide + rhizobia, 60% TMC; TMC, total moisture content.
During the drought, nodulation processes in soybeans were inhibited in both experimental variants (Figs. 6,7). However, after the stress period, the root nodulation processes recovered better in the soybean variant treated with fungicide and inoculated with rhizobia. This is evidenced by a decrease of 32.7% in the number of nodules on the roots of the soybeans inoculated with rhizobia without using fungicide compared to the control. Meanwhile, in soybeans under complex treatment with fungicide and rhizobia, the number of nodules on the roots was consistent with the control plants (Fig. 6).
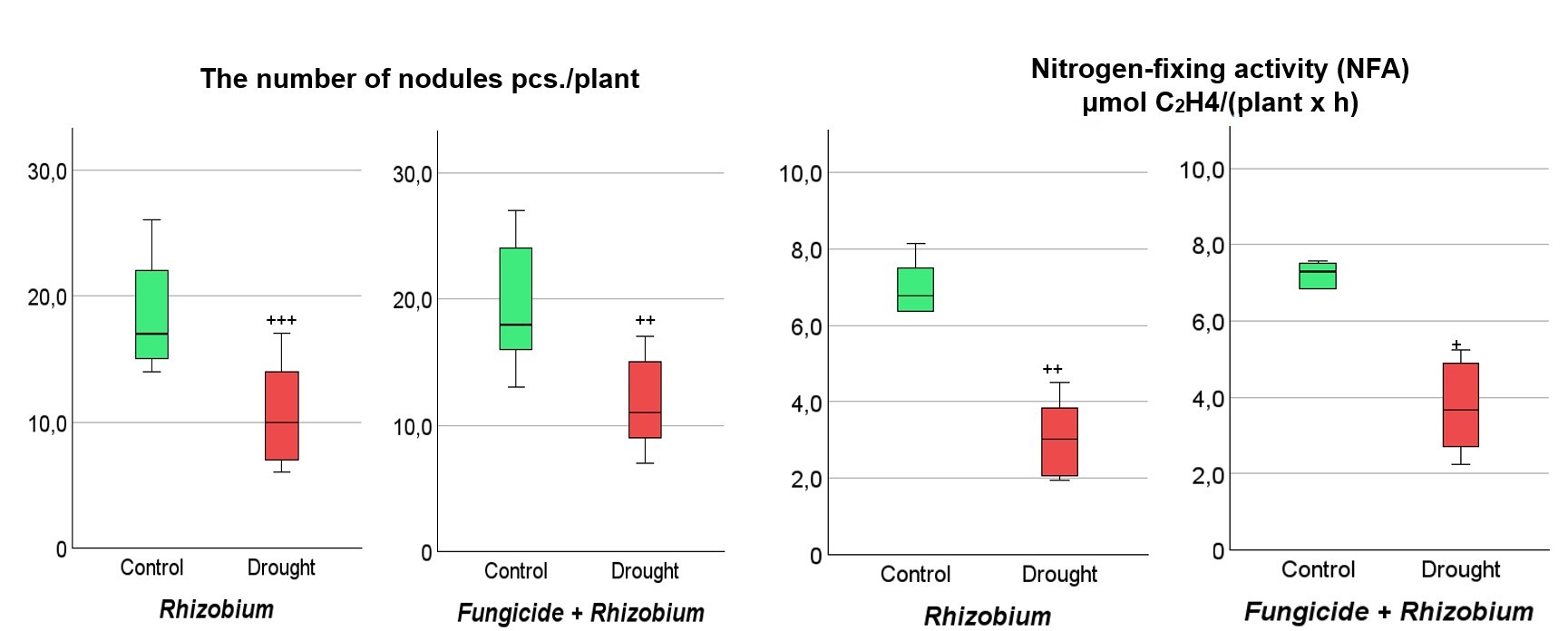 Fig. 6.
Fig. 6.Effect of fungicide seed treatment and Rhizobium on
soybean nodulation and nitrogen-fixing activity (NFA) in nodules. The figure
shows the total value of the indicators analyzed during the third trifoliolate
(V3), beginning flowering (R1), and full bloom (R2) stages. The data compared to
the control (optimal watering) are probable by
 Fig. 7.
Fig. 7.Soybean nodules during plant growth at the third trifoliate stage under optimal watering (60% TMC) and drought (30% TMC). TMC, total moisture content.
As a result of drought, a decrease in NFA was observed in soybean nodules of both experimental variants, while in the post-stress period, a more intensive recovery of the nitrogen fixation process to the control level was observed in soybean inoculated with rhizobia and seed treatment with a fungicide. In particular, it was recorded that in soybeans without fungicidal seed treatment, the NFA value in nodules was 51.9% lower and 25.6% lower in soybeans with complex seed treatment with fungicide and rhizobia compared to the control (Fig. 6). Therefore, the fixation of molecular nitrogen by nodules was 26.3% more effective in soybeans under complex fungicide treatment and rhizobia inoculation.
Field results indicate that fungicide seed treatment along with rhizobia inoculation led to a 36% reduction in the number of soybean nodules in the third trifoliate stage (Fig. 8). At the same time, a 28.1% decrease in the levels of NFA nodules formed by B. japonicum was observed when the seeds were treated with a fungicide in the field (Fig. 8). In the full bloom stage of soybeans in the field, there were no statistically significant differences in the levels of nodulation and nitrogen fixation processes between the investigated variants (Fig. 8). In the pod formation stage, the activity of these processes increased in soybeans treated with fungicide and rhizobial inoculant. This is evidenced by a 26.8% increase in the number of soybean nodules, as well as a 71.7% improvement in the NFA of nodules when inoculating seeds with rhizobia and using fungicide treatment compared to similar indicators in soybean without fungicide application (Fig. 8).
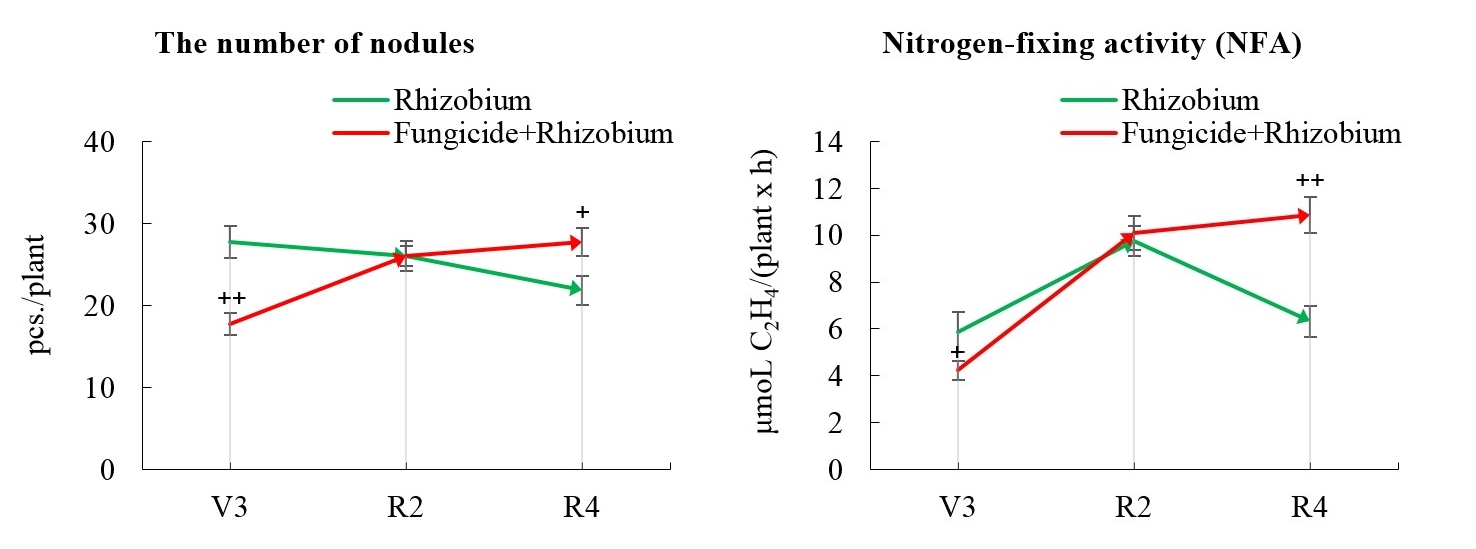 Fig. 8.
Fig. 8.Rhizobium inoculant and seed-applied fungicide effect
on soybean symbiotic apparatus in the field. The figure shows the indicators
analyzed during the third trifoliolate (V3), full bloom (R2) stages, and pod
formation (R4). These data compared to the control (Rhizobium) are
probable by
Evaluation of soybean productivity in the field showed a 12.9% increase in grain yield in the seeds treated with fungicide and rhizobial inoculation compared to the variant without fungicide application (Fig. 9).
 Fig. 9.
Fig. 9.Rhizobium inoculant and seed-applied fungicide effect on soybean productivity in the field.
Plant adaptations to external factors are a complex response involving the perception of the stressor by the plant organism, the initiation of signal transduction pathways, and physiological and biochemical changes in the cell that determine the level of development by the plant organism [25, 26]. At the same time, the pro-oxidant–antioxidant balance in plant cells is disturbed, leading to increased ROS production and activation of peroxidation of cell membrane structures [27]. Activation of the lipoperoxidation process is one of the primary nonspecific links in the general stress response of a plant organism, which can initiate the activation of other defense mechanisms [28]. A significant amount of information is available on the signaling functions of both ROS and lipoperoxidation products. These are considered stress markers and signaling mediators, which are necessary for developing an adaptive response [16, 17]. Antioxidant systems play a decisive role in forming protective reactions and adapting plants to stress factors, among which enzymatic complexes are important [27].
The analysis of the results showed that pre-sowing of fungicide seeds treatment together with rhizobia inoculation under optimal water supply of soybean does not induce statistically significant changes in the course of lipoperoxidation processes, the activity of SOD and CAT enzyme complexes, or the efficiency of molecular nitrogen fixation by nodules in the conditions of a pot experiment, compared with soybean pre-sowed without the use of fungicides. This proves that using such a treatment does not significantly alter plant metabolism and does not negatively affect the functioning of the soybean symbiosis under optimal growing conditions.
Under conditions of drought, the development of antioxidant protection by plants was recorded, as evidenced by an increase in the activity of key antioxidant enzymes (SOD and CAT) in soybean nodules inoculated with rhizobia and treated with a fungicide. At the same time, SOD activity increased more significantly under moderate drought conditions, while CAT activity did likewise under prolonged stress.
Therefore, different enzymatic complexes are included in the work at various intensities, depending on the influence of the stress factor, such as drought. An increase in SOD activity in soybean nodules subjected to moderate dehydration is an apparent consequence of the active involvement of the enzyme in the disposal of excessive ROS production in the cells. This could lead to the regulation of the plant’s pro-oxidant–antioxidant status by activating other antioxidant enzymatic complexes to protect cells from drought-induced oxidative damage. The long-term effect of drought was an increase in CAT activity in soybean nodules inoculated with rhizobia of fungicide-treated seeds, which could be a consequence of the active involvement of the enzyme in the disposal of excessive hydrogen peroxide production—the product of the SOD reaction. Concurrently, an increase in the fractions of the molecular spectra of peroxidases was observed on the electrophoregram of soybean nodules under drought conditions.
We noted changes in the activity of antioxidant enzymes (SOD and CAT) in soybean nodules inoculated with rhizobia after seed treatment with a fungicide, contributed to adaptive changes in soybean metabolism under drought conditions, which in turn induced a slowdown in the development of lipoperoxidation processes during stress. This is evidenced by a lower level of MDA accumulation in nodules formed by rhizobia and treatment of seeds with fungicides, compared to its concentration in soybeans without the use of fungicides. Apparently, the fungicidal treatment of seeds, together with the inoculation of active rhizobia, activates the antioxidant enzymes SOD and CAT in soybean nodules during drought, which contributes to the regulation of the development of the lipoperoxidation process and the protection of cells from oxidative damage. Therefore, in drought conditions, the application of fungicidal treatment to seeds together with inoculating with active rhizobia does not negatively affect soybean metabolism but, on the contrary, promotes adaptive changes in plant metabolism. As proof of this, after exposure to drought, a rapid recovery of the lipoperoxidation process and the activity of antioxidant enzymes to the control level (optimal water supply) was observed in soybean nodules in the variant with fungicide and inoculant seed treatment.
It was found that during prolonged drought, both rhizobia nodule activity and NFA in soybean nodules are inhibited by the pre-sowing treatment of seeds with a fungicide. However, after exposure to stress (restoration phase), these indicators were at the control level in the variant with fungicide and inoculant treatment. This indicates that the symbiotic apparatus functioning is preserved during the long-term effects of drought and the post-stress period. Therefore, changes in the activity of SOD and CAT and the intensity of lipid peroxidation in soybean nodules inoculated with rhizobia during the pre-sowing fungicide treatment of seeds indicate the development of plant stress-protective reactions. Subsequently, these contribute to the adaptive changes in metabolism and the preservation of the symbiotic apparatus functioning.
The biological ability of soybeans to the symbiotic type of nutrition, caused by nodule bacteria of the genus Rhizobium, provides plants with fixed atmospheric nitrogen in the form of organic compounds during the most necessary period of plant growth and development, which makes it possible to form stable and environmentally friendly crops [29]. Ultimately, the yield of agricultural crops, including legumes, results from a complex interaction between the plant’s genetic potential and a complex multitude of environmental factors [30]. Grain productivity of leguminous crops is also determined by symbiotic productivity, which depends on the efficiency of symbiosis formed with highly active virulent strains of nodule bacteria [31].
Pre-sowing fungicidal treatment of seeds and inoculation in the field slowed down the nodule formation and nitrogen fixation processes in the third trifoliate leaves stage. However, during the full bloom of soybeans treated with fungicides, the activity of these processes was restored to the control level (rhizobia without fungicides). While in the pod formation stage, when treated with fungicides, there was an increase in both nodulation processes and the level of molecular nitrogen fixation by nodules, which was not observed in soybeans in the field without using fungicides. This positively affected the effectiveness of the symbiosis in the field and also led to a 12.9% increase in soybean productivity when the fungicide was used with the inoculant. In the field, the increase in the mass of the soybean grains (t/ha) was quite significant, considering the uncontrolled growing conditions, where there is an influence by a whole complex of factors of different natures.
Pre-sowing seeds treatment with fungicide (fludioxonil, 25 g/L) and rhizobial inoculant induces the formation of stress-protective reactions by activating the SOD and CAT enzymatic complexes and regulating the lipoperoxidation process in soybean nodules, which contributes to the symbiotic apparatus functioning during drought.
Rhizobium inoculant and seed-applied fungicide (fludioxonil, 25 g/L) effects ensure effective fixation of molecular nitrogen in nodules in the field by extending the period of symbiotic apparatus functionality at the reproductive stage of plant development. This has a positive effect on the soybean yield in the field.
It has been proven that fungicide application and active virulent Rhizobium strains are important for improving the protective properties and nitrogen-fixing potential of soybeans during drought. Our research emphasizes the perspective of such an agroecological approach to growing soybeans under modern climate changes to obtain ecologically and economically safe crop products.
ROS, reactive oxygen species; SOD, superoxide dismutase; CAT, catalase; TMC, total moisture content; NFA, nitrogen-fixing activity; LPO, lipid peroxidation; MDA, malondialdehyde; TCA, trichloroacetic acid; EDTA, ethylenediaminetetraacetic acid.
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reason able request.
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by TN. The first draft of the manuscript was written by TN and PP. Supervision: SK. All authors read and approved the final manuscript. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity. All authors contributed to editorial changes in the manuscript.
Soybeans (Glycine max (L.) Merr.) of the Almaz variety were used in this study. Soybean seeds were kindly provided by Poltava State Agrarian Academy (Poltava, Ukraine). Originator–Poltava State Agrarian Academy, Ukraine. The variety from the 2007 Register of Plant Varieties of Ukraine and recommended for cultivation in the forest steppe of Ukraine.
Not applicable.
The research was carried out within the framework of the state budget topic “Research of the peculiarities of the formation and functioning of symbiotic systems of soybean and alfalfa under the action of external environmental factors” No. 0121U107432, which was funded by the National Academy of Sciences of Ukraine.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









