1 Birck Nanotechnology Center, Purdue University, West Lafayette, IN 47907, USA
2 School of Mechanical Engineering, Purdue University, West Lafayette, IN 47907, USA
3 School of Electrical and Computer Engineering, Purdue University, West Lafayette, IN 47907, USA
4 School of Materials Engineering, Purdue University, West Lafayette, IN 47907, USA
†These authors contributed equally.
Abstract
Dermal wound infections are a rising source of morbidity and mortality in patients worldwide as new and worsening complications reduce the efficacy of traditional treatments. These challenges in wound care are increasingly caused by comorbidities such as obesity and diabetes as well as surging rates of antibiotic resistance. As a result, there is an urgent need for alternative treatment options. Gaseous ozone has shown great promise as a potential new treatment for infected dermal wounds. In this brief review of current wound therapy techniques found in the literature, an in-depth discussion of the mechanisms, benefits, and results of topical ozone gas as a therapy for infected dermal wounds is presented. This includes studies of ozone applied to wounds performed in vitro, in vivo, and clinical settings, as well as the use of ozone as an adjunct therapy for increasing the efficacy of traditional treatments. The overwhelming evidence suggests that ozone exhibits significant antimicrobial properties and has been shown to promote wound healing factors, especially when applied between 5–60 ppm. As such, this promising alternative therapy warrants a significant investment of time and resources to fully utilize ozone as an effective treatment against antibiotic resistant bacteria and other rising challenges in wound treatment.
Keywords
- wound therapy
- ozone
- topical administration
- adjunct therapy
- antibiotic resistance
Skin is the body’s largest organ and provides a barrier to pathogens, prevents water loss, regulates our temperature, allows us to sense through touch, and many other crucial functions [1]. Much of this barrier function is provided by the epidermis, which constitutes the outer layer of the skin tissue. Disruption of this barrier, in patients with chronic wounds leads to a significant reduction in the skin’s ability to prevent microbes like bacteria from colonizing and causing skin infections [2]. These skin and soft tissue infections (SSTI) are a major concern in the healthcare industry. The wound care market in 2021 was estimated at $17.5 billion and is projected to grow at a compounding rate of 6% [3]. SSTIs cause an estimated 3.5% of emergency room visits in the US, with each visit costing over $8000 [4, 5]. These costs become even more significant for patients with other serious comorbidities, such as diabetes and obesity, which can severely impair the body’s vascular system and lead to poor blood flow. These conditions are widespread as currently, over 30% of Americans are obese, and 10% of the US population has diabetes [6, 7, 8].
One of the most common wound types among this demographic is diabetic foot ulcers (DFU), affecting between 15–25% of patients with diabetes [9]. Due to chronic venous insufficiency, neuropathy, and other complications associated with diabetes, DFUs often become chronic wounds, or wounds that do not progress toward healing at a standard rate (typically within 30 days), leading to a greater chance of reinfection and other serious complications [10]. DFUs that exhibit chronic behavior significantly increases the risk of major complications. According to Apelqvist et al. [11], up to 85% of all lower extremity amputations in diabetic patients stem from chronic wound complications. Infections are the most common complication in DFUs, affecting up to half of all DFUs in healthcare settings [12, 13]. While acute wounds in a healthy adult can often fight off infection through natural defenses, the compromised vascular system and immune system in diabetic patients make this much more difficult and lead to a greater risk of infection [14]. In short, this process occurs as bacteria, either from external contamination or from the over 600 species that typically inhabit the human skin, enter the wound area and begin to proliferate [15]. A healthy body will begin to fight this infection and progress through wound healing through several processes, including the deployment of macrophages to help fight against invading pathogens like bacteria [16]. In cases where underlying conditions prevent these native defenses from being able to eliminate the contamination, the bacteria will proliferate, and the infection will take root. Common bacteria in wound infections include Staphylococcus aureus (S. aureus), Pseudomonas aeruginosa (P. aeruginosa), Proteus mirabilis (P. mirabilis), and Escherichia coli (E. coli) [17].
Once a DFU becomes infected, the ability of the wound to progress toward healing will be significantly reduced [18, 19, 20, 21]. These complications progress as the wound develops along the wound infection continuum (Fig. 1). The wound infection continuum is broadly categorized into five stages: contamination, colonization, local infection, spreading infection, and systemic infection [22, 23]. All open wounds are considered contaminated—hosting small amounts of bacteria without impacting wound healing or the host’s health [23]. Once the bacteria within the wound begin to replicate, the wound is considered to be in the colonization stage, though no detrimental effects to the host are detectable. Here, the body’s defenses must overcome the growth of the bacteria to prevent a traditional infection from occurring.
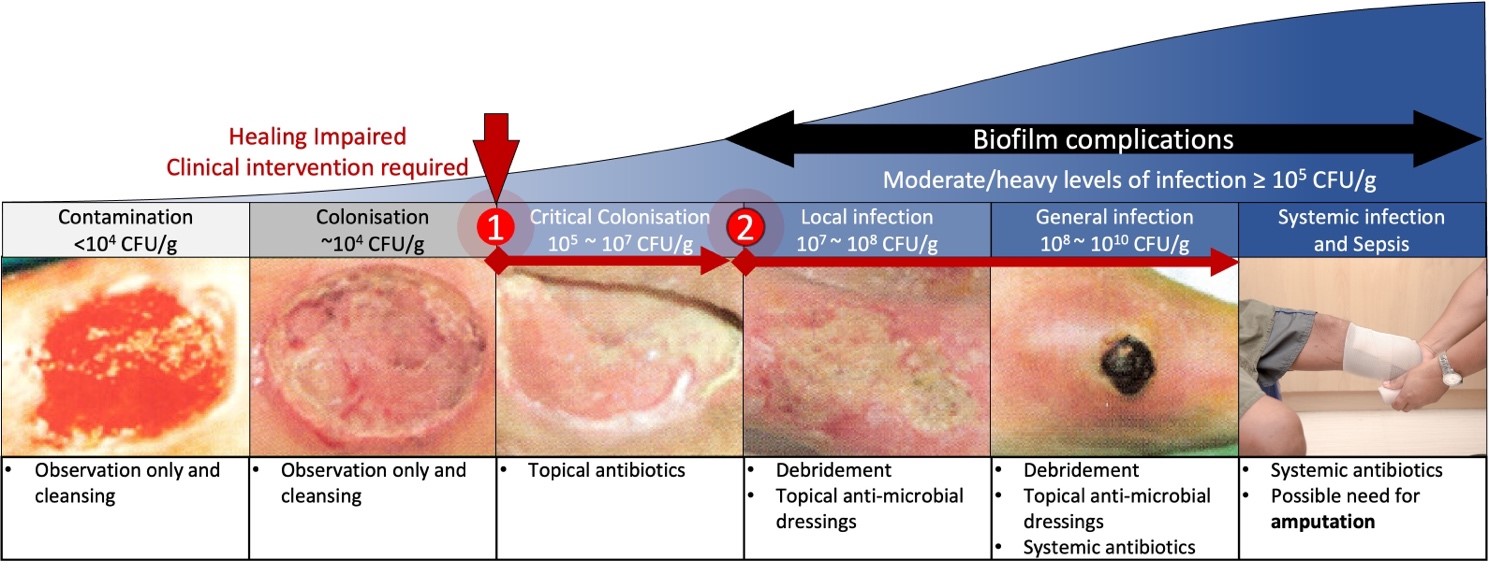 Fig. 1.
Fig. 1.The wound infection continuum. Wound infections
develop and progress as bacteria attach and proliferate within the tissue. There
are two key locations on the continuum associated with wound treatment and
healing. (1) At around 10
If the replication of bacteria within a wound can overcome natural infection
defenses, either due to a virulent strain of replicating bacteria or a weakened
host immune system, the bacterial colonization will reach a critical level,
typically 10
As the wound progresses, many infections develop a protective layer of cells and extracellular matrix called a biofilm. Biofilms form when bacteria can proliferate in the necrotic tissue and debris present in the chronic wound and form a protective colony. A biofilm layer can range between a few micrometers to a millimeter in thickness and significantly impairs the wound-healing process [25]. The protective cell and matrix layer that makes up a biofilm has been shown to significantly impair the diffusion of antibiotics into deep tissues and thereby reduce their efficacy by up to 1000 times [26, 27]. Therefore, treatment of wound infections with biofilm typically requires physical removal of the biofilm layer by a process such as a debridement.
While biofilm formation is a significant complication for local wound treatment, further escalation of infection severity can lead to more harmful conditions in a patient. As a wound infection progresses beyond the local infection stage, it spreads to other body parts. During the spreading infection stage of the wound infection continuum, the bacteria can be found in new surrounding tissues, muscles, and local organs [22]. Once the infection is no longer isolated in a single location, systemic antibiotics are needed to provide treatment. Systemic antibiotic treatments are effective against infections across the body and have long been an effective means to reduce bacterial load when prescribed correctly. Unfortunately, these systemic treatments also present some drawbacks, including the elimination of healthy gut bacteria and a greater chance of new antibiotic resistance developing [28, 29]. Should treatments continue to be ineffective, the infection will continue to spread and become systemic. At this stage, high doses of systemic antibiotics are required for treatment and are typically administered intravenously. Without proper control, the patient is at serious risk of life-threatening complications, including sepsis. Amputation is commonly utilized to reduce the risk of sepsis, though it is not without complications. Alongside the lifelong effects on patient mobility and quality of life, infection of an amputation stump can perpetuate the problem and lead to re-amputation [30].
At each stage of the wound infection continuum, the treatment method chosen by a clinician is critical for improving the patient’s health and reducing the risk of further complications. As a result, treating a wound early is paramount to give the wound the greatest chance to heal. Unique complications such as spreading infections and biofilm that occur at later stages of wound infection require specialized treatments typically necessary as the wound progresses, complicating the course of treatment. This emphasizes the necessity of safe, effective treatments at the early stages of the wound infection continuum.
While each stage of the wound infection continuum has unique needs for effective treatment, it can be seen that topical antibiotics and treatments to target wound infections have many benefits compared to systemic treatments. These include a lower risk of interference with healthy bacteria and the ability to apply higher doses of treatment levels. The topical treatment is even more effective when administered early in the wound treatment process. Unfortunately, the efficacy of many antibiotics has lessened as the emergence of antibiotic resistance in many strains of bacteria continues to rise. In recent years, bacteria with resistance to a greater number of antibiotics have been reported. As a result, there is a unique need to develop alternative therapies. This review is focused on the particular benefit of using gaseous ozone as a topical alternative therapy for the treatment of skin infections.
Currently, there is a limited selection of treatment methods for infected dermal wounds. The following sections describe the benefits and limitations of different methods in detail to provide an understanding of each treatment option’s scope and what improvements in treatment options are still necessary. The results have been summarized in Table 1. Because of the many benefits of topical treatments, such as the reduced risk of systemic toxicity and higher therapeutic index, this section mainly reviews different topical treatment methods used for SSTI.
| Treatment | Applicable uses | Benefits | Drawbacks |
|---|---|---|---|
| Debridement | |||
 |
|||
| Topical Antibiotics | |||
 |
|||
| Antiseptics | |||
 |
|||
| Silver Nanoparticles | |||
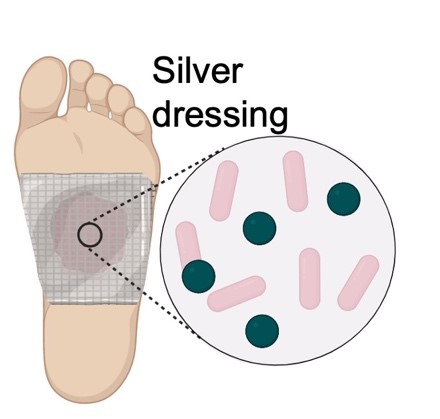 |
|||
| Cold Plasma | |||
 |
The first line of treatment for an infected wound commonly involves cleaning the wound bed to enable further therapies. Debridement is the process of removing inhibitory factors, such as necrotic tissue and biofilm, from a wound to increase treatment efficacy and is part of the wound bed preparation process [31]. Wound bed preparation is an important step in the topical treatment of infected wounds because it removes barriers to wound healing and can stimulate natural progression in a non-advancing wound edge [32]. In chronic wounds, which show slow or no progression toward complete healing, debridement is a necessary step for enabling other topical treatments to be effective [33]. This is especially true when treating wounds infected with biofilm. As a wound infection matures, it often creates a protective matrix layer around outer cells [34]. This biofilm layer, often a few micrometers to a millimeter thick, can significantly affect wound healing and increase resistance to antibiotic treatment by 1000-fold when compared to non-biofilm bacteria while preventing topical treatments from reaching further below [25, 26, 27]. Wound bed preparation through debridement is practiced along with proper dressing, pressure off-loading, and other crucial steps for the care of all wounds. Each wound’s specific conditions are unique, requiring multiple methods of wound bed preparation, including mechanical, surgical, chemical, or enzymatic debridement. Each method is used to promote wound healing through removing inhibiting tissue and is described in greater detail below.
Mechanical and surgical debridement utilize the physical removal of biofilm, slough, necrotic tissue, and other inhibitory factors from the wound bed. Mechanical debridement most commonly uses the practice of wet-to-dry dressings, in which a wet gauze is applied to the wound area and allowed to dry, which embeds the necrotic tissue into the gauze before forceful removal [32, 35]. Surgical debridement uses instruments such as scalpels and ultrasonic applicators to cut away necrotic tissue to achieve a similar result. It is more selective but requires a trained surgeon to perform, so it is often limited to more severe cases [32, 35]. Both mechanical and surgical debridement can quickly remove significant amounts of inhibiting tissues but are painful and can cause damage to surrounding tissue, so they are not suitable for every application.
Chemical debridement, sometimes referred to as enzymatic debridement, utilizes natural enzymes, such as collagenase, to speed up the breakdown of necrotic tissue by placing them in direct contact with the wound bed [35, 36]. Wound debridement is a common practice to promote effective treatment and healing of wounds. Typically, chemical debridement is used in less severe infections, as it is a slower process, with new enzymatic applications occurring daily [35]. While removing necrotic tissue takes longer, chemical debridement is selective and painless for the patient, it’s often considered a relatively slow process compared to mechanical debridement methods [32].
Though each technique has specific benefits and drawbacks, the process of cleaning a wound bed is an essential part of any wound care routine. Despite this necessity, the debridement process on its own does not eliminate infection, but prepares the wound for the application of other antimicrobials [37]. Therefore, many wound treatment procedures often use debridement paired with another treatment, such as antibiotics or antimicrobial dressings, to reduce the rate of bacterial wound infection and improve wound healing.
The treatment of infections with antibiotics has been common since the 1940s, after the discovery of Penicillin by Sir Alexander Flemming in 1928 [38]. Since then, the number of antibiotics available for treating infections has increased dramatically compared to the vast number of bacterial pathogens that can cause infections. Common means of antibiotic treatment include systemic and topical approaches. At the same time, systemic antibiotics are commonly taken orally or intravenously, especially when an infection has begun to spread throughout the body. This widespread dispersion of the antibiotic has been shown to significantly impact the rate at which antibiotic resistance develops in new strains of bacteria [39]. On the other hand, topical antibiotics are useful for treating skin and soft tissue infections due to the localized high concentration that can be applied without reaching toxicity limitations [40]. By localizing the application, topical treatments are able to apply antibiotics without affecting bacteria in healthy parts of the patient’s microbiome and reducing the development and spread of antibiotic-resistant bacteria.
Antibiotics can be broken into different classifications, including Penicillin, Cephalosporins, Fluoroquinolones, Monobactams, carbapenems, and more [41]. Each classification of antibiotics is designed to attack the bacterial cells uniquely. These methods of action commonly include inhibition of cell wall synthesis (penicillin, glycol-peptides, cephalosporins), inhibition of protein synthesis (macrolides, lincosamides, oxazolidinones), or inhibition of nucleic acid synthesis (rifampin) among others [41, 42]. While the number of antibiotics available is vast, certain drugs have gained more popularity for the topical treatment of skin infections. These include Bacitracin, Mupirocin, Fusidic Acid, Gentamicin, and Retapamulin. Bacitracin is typically effective against many Gram-positive (G +ve) bacteria such as Staphylococci and Streptococci species and can be purchased as over-the-counter medicine, but resistances to Bacitracin are not uncommon for bacteria to develop, making it less effective over time [33]. Mupirocin is one of the most effective antibiotics used commonly as a 2% topical cream [43]. Mupirocin is used to treat G +ve infections caused by aerobic cocci species (except Enterococci) and is widely tolerated by patients, but extended use can also be in bacteria such as S. aureus [44]. Fusidic Acid is an antibiotic that is more effective at targeting deep tissue infection due to its deeper penetration into the skin than other antibiotics. Unfortunately, Fusidic Acid’s spectrum of activity is limited to G +ve bacteria such as S. aureus and S. epidermidis and can cause severe allergic reactions in some patients [33]. Retapamulin is another antibiotic with wide-ranging efficacy against G +ve bacteria, including strains resistant to common antibiotics [33]. While many of the previously mentioned antibiotics are only effective on G +ve bacteria, Gentamicin has a much wider range of bacteria that it is effective against, including both G +ve strains, such as Staphylococci and Streptococci, and Gram-negative (G-ve) strains such as P. aeruginosa. To maintain effectiveness, Gentamicin must be applied multiple times per day and still has a risk of antibiotic resistance developing, most notably in strains of Enterococci [33, 45]. The increasing prevalence of antibiotic resistance significantly reduces the number of effective treatment options for clinicians. Recently, even antibiotics that are typically used only as a last resort have been shown to develop resistance as well [46, 47]. The development of new drugs has been unable to keep pace with the rate of developing resistance due to the costly and time-consuming process necessary for antibiotics to reach the marketplace [48]. As a result, clinicians are increasingly turning to alternative therapies without antibiotics for treating these infections.
Antiseptics are a broad category of substances that prevent the growth of virulent microorganisms, often with limited selectivity. Antiseptics have been widely used for treating wound-based infections since the early 19th century, with a broader spectrum of activity and lower risk of bacterial resistance development as compared to antibiotics. The most commonly used antiseptic compounds for treating skin infections in clinical practices today include povidone-iodine, alcohols (ethanol), and chlorine compounds [49]. Unlike antibiotics that act selectively on specific targets to eradicate the bacteria, antiseptics have multiple targets and a broader spectrum of activity with a considerably lower risk of resistance to develop against these compounds. Their unique broad-spectrum effect on all vegetative forms of microorganisms enables them to eradicate many strains of antibiotic-resistant bacteria such as MRSA and P. aeruginosa [50]. However, many antiseptics exhibit toxicity to healthy cells which prohibits their suitability for topical application on open wounds [33, 51]. For more than a century, iodine has been used as an antiseptic compound in treating different wounds. Its antibacterial properties were first demonstrated by Davaine in 1882, and in First World War, it was found to reduce the risk of gas gangrene in soldiers’ wounds. Because of its broad use against many microorganisms, iodine has been commonly used as an antiseptic for cleaning skin, wounds, and oral cavities [50].
Iodine’s broad antimicrobial behavior is due to its high level of diffusivity through cellular barriers. It diffuses into the cells, and damage nucleic acids, proteins, and other cellular structures through oxidation [50, 51, 52]. Despite its unique broad-spectrum antibacterial activity, Iodine is highly unstable in its elemental form and is often prepared in the form of a chemical complex containing iodide and an iodate salt with stabilizing agents. This not only improves the shelf life but also its dissolution in aqueous media for more effective and simple topical applications in the form of liquids and ointments. The most common form of these Iodine containing chemical complexes is polyvinylpyrrolidone (PVP) iodide, more commonly called povidone iodide (PVP-I). The PVP enables a sustained release of iodine particles when applied to a wound bed to enable iodine to penetrate microbes effectively for treatment [50, 52]. In addition to improved stability, PVP-I has been shown to have less toxicity and skin irritation than elemental iodine solutions. Finally, although the antibacterial properties of iodine and PVP-I have been demonstrated in different studies, there has been some controversy about their effectiveness in the wound-healing process [49, 53]. Various clinical trials have shown impaired wound healing and reduced wound strength due to the toxic nature of these antiseptic compounds to the fibroblast cells. This trend is also prevalent in antimicrobials like polihexanide and chlorhexidine, commonly used to treat wound infection but are observed to cause high irritation and burns on the wound bed.
Metallic nanoparticles (NPs) are another alternative to traditional antibiotic treatments. These metallic NPs are microscopic particles of metals such as silver, zinc, and copper with dimensions that fall between the 1–100 nm range. NPs are commonly administered through advanced wound dressings for treatment of different SSTI because of their natural antimicrobial properties [54, 55, 56]. These particles are typically generated through physical, chemical, or biological means. Physical generation typically utilizes high temperatures or laser ablation to generate particles in the solution. These processes require significant temperature and can have variations in the size and concentration of particles produced depending on the temperature, but they are the fastest method to produce NPs. Chemical fabrication is achieved through the chemical reduction of the metals at a higher efficiency than physical methods. The final method to generate NPs is biological, which uses microbes to reduce ionic forms of metal compounds into NPs [57, 58].
Once NPs have been fabricated, they are topically applied to the wound area by integrating them into gels and dressings in the form of coatings such as Acticoat® and Silvercel®. Once applied, NPs have multiple methods through which they act against microbes. These include blockage of cell permeability through the aggregation of NPs on the cell wall, oxidative damage to cell structure through the generation of ions or reactive oxygen species (ROS), and prevention of DNA replication through cellular uptake of metallic ions [56]. While there are many beneficial properties of metallic NPs, there are also drawbacks. Like antiseptics, NPs are non-selective and can cause damage to skin cells and have been shown to impair the tissue healing process [59]. Additionally, it is difficult to utilize controlled release mechanisms with NPs, making dosage monitoring difficult [56]. Finally, studies such as the work done by Panáček et al. [60] have frequently found Silver-resistant bacteria in particular strains of P. aeruginosa and E. coli which are amongst the main causes of infections and sepsis in patients with burn wounds.
The term plasma describes a medium of ionized gas that includes disintegrated polyatomic atoms, monoatomic ions, free electrons, ultraviolet (UV) radiation, and heat [61, 62]. Cold or non-thermal atmospheric plasmas are those generated at ambient temperature and pressure that exist briefly in a non-equilibrium state [61, 63]. Cold atmospheric plasma (CAP) wound treatment is an alternative treatment method for surface wounds that utilizes charged and reactive species produced by a high energy source, such as dielectric barrier discharge (DBD), to eradicate bacteria in the wound. Due to technological innovations that enable more efficient production of CAP and its numerous beneficial properties, there has been great interest in developing portable plasma systems for wound care applications [64, 65].
CAP can be generated by an electric discharge that enables the continuous ionization of gas molecules to eradicate bacteria. The most common forms of this are corona discharge (CD) and DBD, which can be used to create plasma jets and other application technologies [62, 63, 66]. In each case, the high electric field causes the dissociation of electrons from the outer shells of gas molecules and causes the breakdown of gas molecules into singular atoms. This process creates a complex mixture of particles and radiation that forms the plasma. This “gaseous mixture” gives CAP its beneficial properties for wound treatment. Of the different components, ROS is the leading contributor to the antimicrobial properties of CAP [61, 62]. ROS exhibit antimicrobial behavior through the oxidation of outer cell structures [67]. Of these species, ozone has been shown to play a foremost role in the inactivation of microbes due to its high reactivity and relatively long lifetime [68, 69]. In addition to sterilization of the wound bed, CAP has also been shown to stimulate wound healing and blood coagulation and help ablate unwanted tissue [70, 71, 72, 73].
Due to the benefits of CAP treatment, several efforts have been conducted toward the commercialization of this technology for wound care, including Terraplasma Plasma Care®, TDK Piezobrush PZ3®, and TPE PlasmaDerm®. Medical personnel often use these systems as point-of-care treatment in clinical settings. Typically, the treatment is applied between 1–3 min once per day for multiple days directly onto the wound bed.
Although several studies have shown the efficacy of many such systems in treating chronic and infected wounds, they often require complex and costly systems, which limits their accessibility to many patients and health care providers. Additionally, the high energy of the plasma and the UV radiation produced in many such systems can cause degenerative changes in the cells and fibrous tissues around the site of treatment [62]. Therefore, many of such therapy methods are often restricted to short durations of treatment (1–3 min) to reduce the toxic effect on healthy cells, which limits the penetration and effective interaction time of the ROS with the wound bed. Due to ozone’s prominent role in providing beneficial properties in CAP, many efforts have been made towards utilizing ozone alone as an antimicrobial treatment to allow for many of the benefits of CAP systems while minimizing complications.
Reactive oxygen species such as ozone are often utilized as an antimicrobial treatment by directly applying ionized gas or oils infused with reactive compounds. Initially used in medicine as a pragmatic technique, ozone therapy has now progressed to be used for antibacterial effects, immunoregulation, antioxidant defenses, and epigenetic alteration. While a 2003 decision by the FDA prohibited commercial use of medical ozone due to “no proof of safety and effectiveness”, continuing research efforts have shown overwhelming results that controlled exposure to ozone can play a significant role in reducing bacterial loads in wounds and thus warrants further investigation [74, 75]. Particularly, microbial inactivation via gaseous ozone exposure has widely been experimented to treat different infected topical wounds [76]. Although ozone can damage crucial cell components at high concentrations and lead to severe irreversible side effects, with optimal application dosing strategies it can be used treat necrotizing skin infections, allergic diseases, and promote wound healing. Though debated, the mechanism of ozone interaction has been generally accepted to be through the generation the oxygen species that damage the pathogenic membranes and promote an immunoregulatory effect. Ozone directly disrupts the nucleic acid and lysosomes of the microbial structure and releases oxygen free radicals, destroying the living environment. Ozone is also observed to accelerate the cell growth cycle and modify growth factors by activating redox transcription factors like nuclear factor kappa B (NFkB) [77]. These nuclear factors are extremely potent proinflammatory activators that promote wound healing. Ozone is also observed to increase the leukocytes, facilitating the formation of monocytes, T-cell signaling, and the phagocytic capacity of granulocytes. As a cumulative effect, the immune system initiates the interferons and interleukin-triggered antibody-dependent cellular cytotoxicity (ADCC) [78, 79]. For example, ozone is used to treat wounds by patients soaking their skin in ozonized water, called Ozonated hydrotherapy. Though it is proven to be an effective treatment against infections and wound healing, the half-life of the ozone in water is just 30 min at room temperature. This has led to new developments towards creating more stable methods for storing ozone at room temperature [80]. Among these efforts, studies have demonstrated that ozone diffused into oil was observed to have an increased lifetime of 10 h at room temperature and around 5 days when stored in a refrigerator [81]. This increase in the shelf life was due to the formation of double bonds between ozone molecules and monounsaturated fatty acids such as oleic acid. This has proven to be an effective means of enhancing the storability and applicability of ozone for patients in a clinical or household scenario. Another novel method for ozone therapy is ozonated autohemotherapy, in which ozone is directly applied to the body through blood infusion and has been shown to promote bactericidal properties [76, 82]. These techniques, in comparison to conventional techniques, are more cost-effective. Though proven effective, such ozonized oil-based treatment techniques and ozonated autohemotherapy require significant infrastructure for developing and storing necessary components. Directly applying gaseous ozone to the wound surface using a portable system can provide a means to administer the promising ozone therapy while eliminating the complex storage requirements and higher efficacy.
Gaseous ozone-based treatment is the process of administering highly unstable gaseous ozone molecules to treat a wound or a disease. The fundamental process involves the generation and application of gaseous directly to the skin surface to promote wound healing, antibacterial effect, and immunoregulation. In fact, a significant portion of the CAP treatment benefits is due to the presence of ozone in the plasma generation process. This was shown in a study where the antimicrobial properties of common plasma components, including ozone, air ions, and electric charges, were individually tested [69]. The authors show that most of the antimicrobial performance was due to the presence of ozone by using designed filters to isolate specific compounds developed using electric discharge. Bacteria of various strains were exposed to each filtered plasma, and it was observed that the ozone alone performed almost identically to the full plasma with air ions. Additionally, because ozone has a long lifetime outside of the plasma region and can be delivered as a gas flow, it can increase the accessibility and application time significantly through simplified systems without the direct application of high-energy plasma that can lead to toxic and harmful effects to the skin tissue.
While gaseous ozone has shown promise as an alternative means for antimicrobial
therapy, it is worthwhile to note that ozone is a highly unstable gas (half-life
of ozone is around 20~60 min) and cannot be stored by any means
directly as a gas under normal working conditions [83]. Thus, it is necessary to
develop a technique to generate ozone for direct delivery of the gas into the
region of infection/wounds. Nevertheless, to consider the applicability of ozone
as a treatment protocol, it is crucial to eliminate the possible inhalation and
other side effects of ozone exposure by monitoring the level of ozone infusion
[76]. It is necessary to note that the existing short-term exposure guidelines
for ozone are about 1 ppm (for 1 h) for humans [84]. With such healthcare
guidelines consideration, it is crucial to develop a simple and swift reliable
technique for controlled and localized delivery of ozone gases into the
administration region. Therefore, there is a crucial need for a localized
wearable ozone delivery system with portable functionality to provide ambulatory
property [85]. In general, ozone can be created by three methods: Ultraviolet
(UV) light, electrolytic generation, and electric discharge (Fig. 2). UV
light-powered ozone generation system utilizes a UV lamp (with a peak radiation
wavelength of 185 nm) to break the oxygen molecules in ambient air and form
highly unstable singlet oxygen radicals that react with another oxygen molecule
to form O
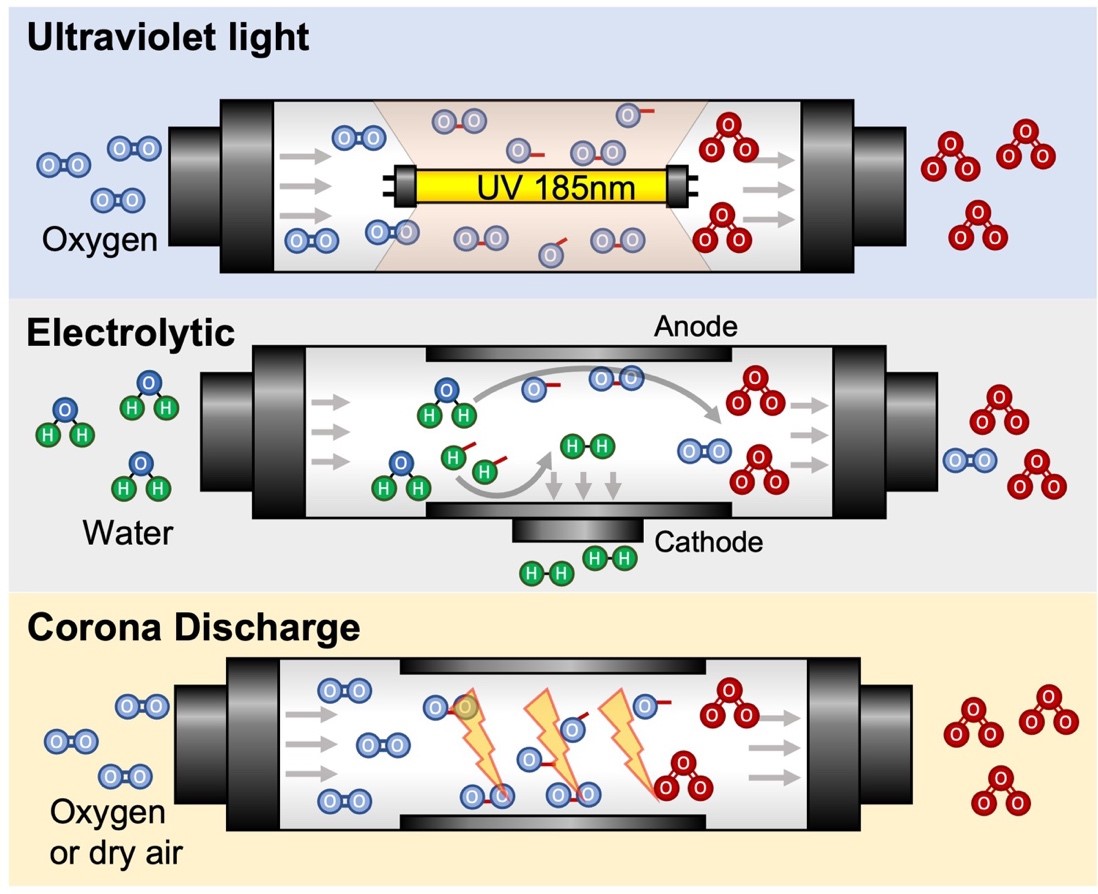 Fig. 2.
Fig. 2.The three primary methods of ozone generation are UV light, electrolytic reactions, and electrical discharge, such as corona discharge. Corona discharge system has the benefit of portability and high production rate of ozone.
Many corona discharge techniques require a high voltage electrical transformer to initiate this ozone formation effect and a feed gas system to have a constant oxygen flow [91, 92]. Also, simple corona discharge systems create a single discharge location at a point, leading to an inefficient ozone generation. To improve the generation efficiency, alternative discharge methods like Dielectric Barrier Discharge (DBD) are also commonly used to overcome the shortcomings of corona discharge systems. In DBD setups, an electrical discharge is created between two electrodes separated by a dielectric barrier [93]. The presence of the dielectric layer causes the breakdown location to disperse across the surface of the electrodes as a series of short arcs instead of a singular arc discharge [94]. Due to these discharge characteristics, DBD contributes to a greater space efficiency in generating ozone gas and thus is the most common commercial method for ozone generation. DBD route of ozone generation shares many commonalities with plasma treatment discussed previously, as both are generated using electric discharge. The key functionality of DBD-based ozone generation systems is their ability to be powered through simple and low-power electronics, which can aid their portability. Furthermore, many of such ozone-generating setups are highly effective in generating ozone at room temperature with ambient air, which is necessary to develop a portable device.
For applications where large amounts of ozone is needed (
One potential drawback of DBD is that ozone development using this technique will inevitably form reactive nitrogen species, which can be harmful at high concentrations [103]. An integrated circuitry system containing an ozone generator system and a blower circuit could effectively deliver ozone to the region of an infection/wound. One such custom-designed ozone generator system was previously reported by our research group for continuous ozone delivery [75] (Fig. 3, Ref. [75]). The developed system includes a portable ozone delivery unit equipped with a DBD ozone generation device and micro blower. Ozone was locally applied to the wound by using a disposable and low-cost hydrophobic and gas-permeable dressing. This portable system was able to provide a controllable dosage of ozone between 0 to 4 mg/h (0–100 ppm) from ambient air and continuously operate for over 12 h on a standard rechargeable battery. Systematic in vitro studies of the platform with different strains of bacteria indicated the complete removal of P. aeruginosa and a significant reduction in the number of S. epidermidis colonies after 6 h of exposure. Thus, by adapting such techniques, it is possible to develop a portable system that can easily release ozone for the development of wound healing therapeutics.
 Fig. 3.
Fig. 3.A portable ozone generation system for treating dermal wounds as developed by Roth et al. [75]. (a) Multilayered disposable dressing for uniform application of gaseous ozone onto the wound. (b) Photograph of dressing applied onto users’ skin and connected to the portable ozone delivery unit (Reproduced with permission from Ref. [75], copyright Frontiers 2020).
Because ozone therapy for treating superficial wounds is still considered controversial in many regards, the full extent of the technology is still being developed even with extremely promising results. The most important focus of wound treatment with ozone is its ability to eliminate bacterial growth and population. Ozone particularly disrupts the cell walls of bacterium via oxidation of phospholipids and lipoproteins [82, 104] but needs to be limited as it can also cause permanent damage to the cell DNA [105]. It has been postulated that ozone’s disinfectant and antibacterial properties are caused by its ability to induce irreversibly oxidative damage of lipids and lipoproteins in the DNA and cell walls of bacteria [105]. However, ozone at controlled concentrations does not have an as significant effect on the eukaryotic cell wall of healthy skin cells, likely due to the higher ability of eukaryotic cells to compensate for the oxidative as compared to pathogenic microbes [106, 107]. Additionally, the interferon-triggering effect of ozone helps stimulate the body’s immune system to better fight the infection and increase wound healing [108]. Thus, various research groups have conducted experimental studies to treat infections using ozone. To give a perspective of ozone therapy, the following section reviews various studies, including in-vitro, animal, and human clinical trials that have been conducted with ozone for the treatment of infections.
In this section, a survey on some of the conducted in-vitro studies to
evaluate the effect of ozone on the toxicity of the cells and the antibacterial
effect is presented. In 1963, Scott et al. [109] reported a 50%
eradication of E. coli bacteria with an ozone concentration of 2
To evaluate the cytotoxic effects of ozone, in another study, two different
ozone gas concentrations of 10 and 35
Similarly, another study evaluated wound healing properties and antimicrobial
effect of fibroblasts (L929) and keratinocytes (HaCaT) cell lines with an
inhibitory analysis towards Candida albicans and S. aureus [113]. Ozonized phosphate buffered saline solutions (8, 4, 2, 1, 0.5, and
0.25
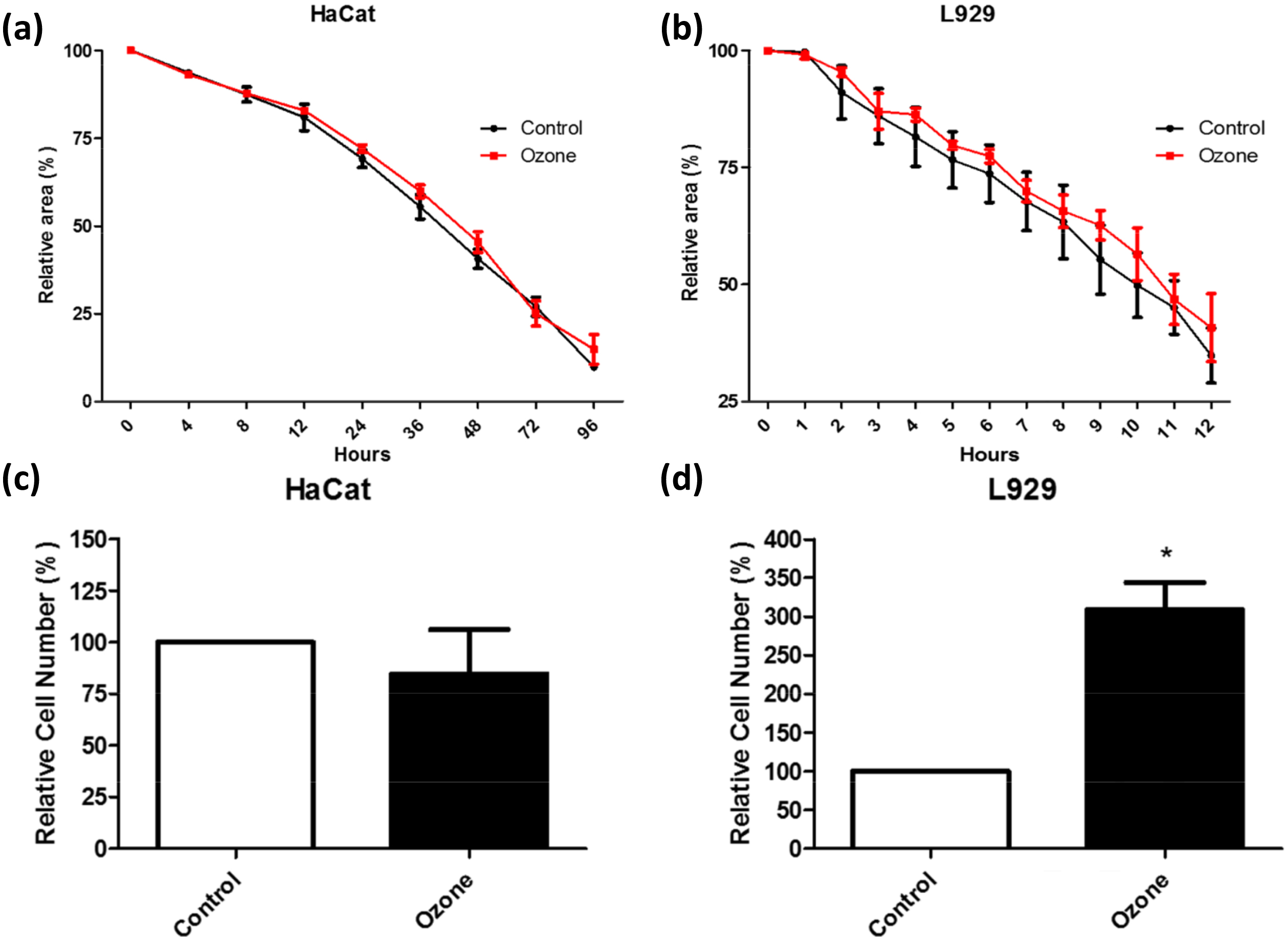 Fig. 4.
Fig. 4.Cytotoxicity of ozone was tested on keratinocytes
(HaCaT) and fibroblasts (L929) using two evaluations methods. Relativistic area
change in % (a) HaCat cells and (b) L929 cells and cell migrated characteristics
in the transwell migration assay for (c) HaCat cells and (d) L929 cells all
showed no significant change between ozone and control groups. This study shows a
significant change in the ozone group elucidating the better remodeling wound
remodeling characteristics *indicates p
With factual in-vitro antimicrobial and biocompatible evidence, many
researchers have extended ozone therapy in various animal models. To
mention a few, Pchepiorka et al. [114] investigated the implications of
ozone treatment on wound healing in buccal mucosa rats in oral cavities. In this
study, surgical wounds were artificially created on the cheeks of Wistar rats
bilaterally, and the wounds were classified into two groups (control and
experiment group). A systemic analysis was performed by exposing 60
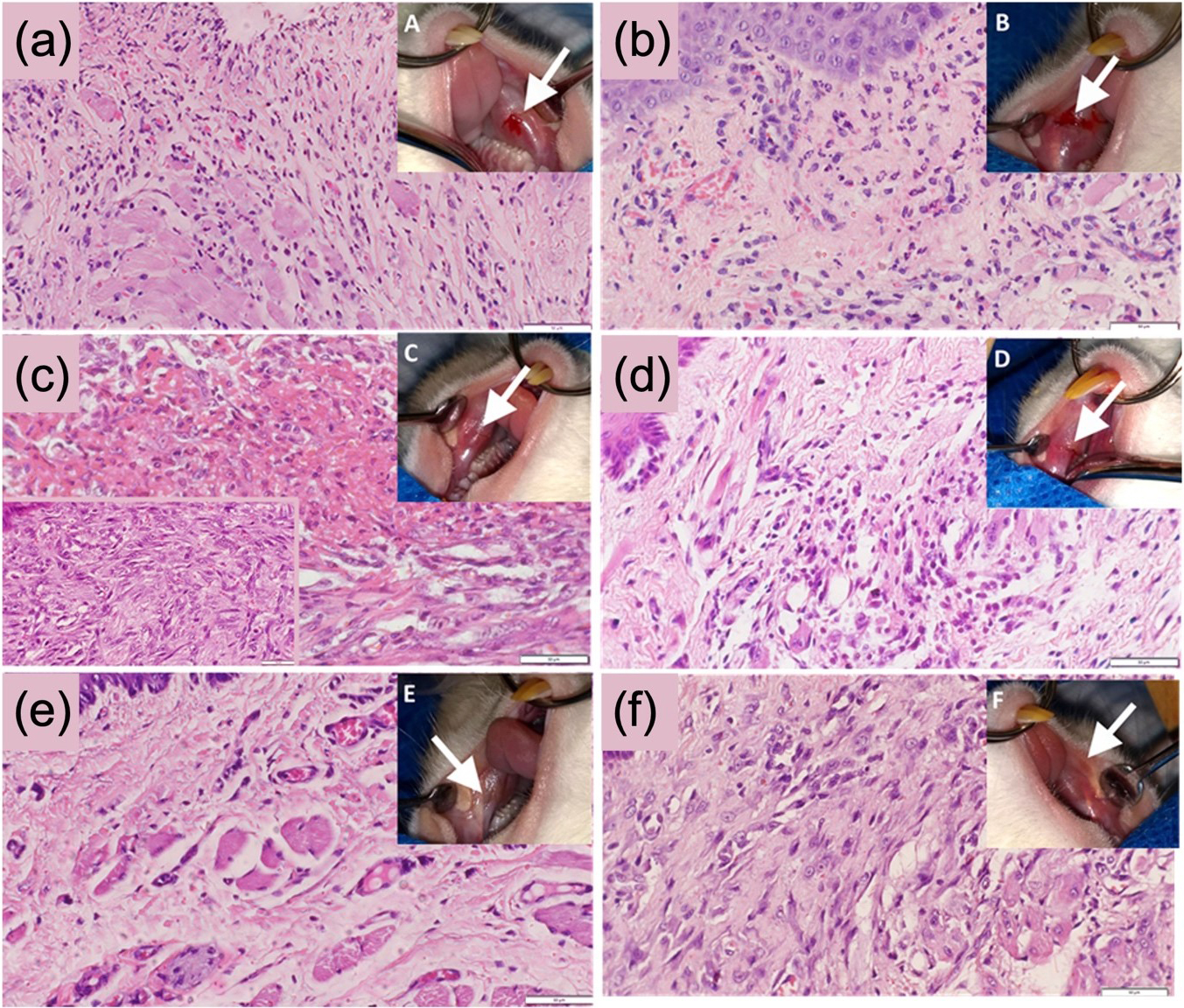 Fig. 5.
Fig. 5.Wound healing characteristics of ozone treatment in mice models. The figure shows the histopathological and macroscopic aspect image of the lesions for the ozone group (a) day 1, (c) day 3, and (e) day 7 and the control group (b) day 1, (d) day 3 and (f) day 7 showing enhanced remodeling of the wounds with the migration of fibroblast cells with significantly higher levels of fibroplasia in the ozone group in comparison to control group (reproduced with permission from Ref. [114], copyright Elsevier 2020).
In another study, a mouse model with excisional wounds was tested with untreated
control and ozonized oil group (99% of ozonide) purchased commercially from an
external source. The granulation tissues were then isolated from the mice, and
the healing efficacy was estimated. Ozone-treated samples were observed to have
better wound-healing properties via fibroblast activation and migration [115].
Further, these results demonstrate that the ozone oil facilitated wound healing
via increasing fibroblast migration in both in vivo and in
vitro. The cell migration distance was significantly higher, ~50
Similarly, Lim et al. [116] studied the effect of the topical
application of ozone (0.5 ppm
Ozone effects in the treatment of infected wounds were first experimented in World War I by German soldiers to eradicate Clostridium anaerobic [120]. From that instant, various modes of ozone-based treatment like ozonized water and ozonized oil, have been experimented for the treatment of different dermal wounds [120]. However, much of the toxicity effects of ozone were largely unknown by those times and were administered nevertheless until its ban by FDA [76, 82]. However, by leveraging the optimal level of ozone concentrations, the previous sections briefly elucidate the bactericidal and promoted wound healing capabilities of topical ozone treatment. The key idea of ozone treatment is not just to delineate bacterial growth but also to promote anti-inflammatory and wound-healing properties at the site of infection. The presence of ozone ensures a sharp reduction in the microbes and eradicating the infection without any severe patient discomfort.
Various research groups have experimented with the use of gaseous ozone in human
trials for the effective treatment of diabetic patients. For instance, gaseous
ozone therapeutics have been considered by Martinez-Sanchez et al. [121]
to treat chronic wounds in diabetic patients with three control groups of ozone
insufflations (with an ozone dose 50 mg/L approx..0 ppm), topical ozone treatment
(60 mg/L approx..60 ppm), and plaster dressing with ozonized oil. The ozone group
observed a higher wound healing rate and low hospitalization rate than the
control group. Another study showed that five-week administration of ozone
(52
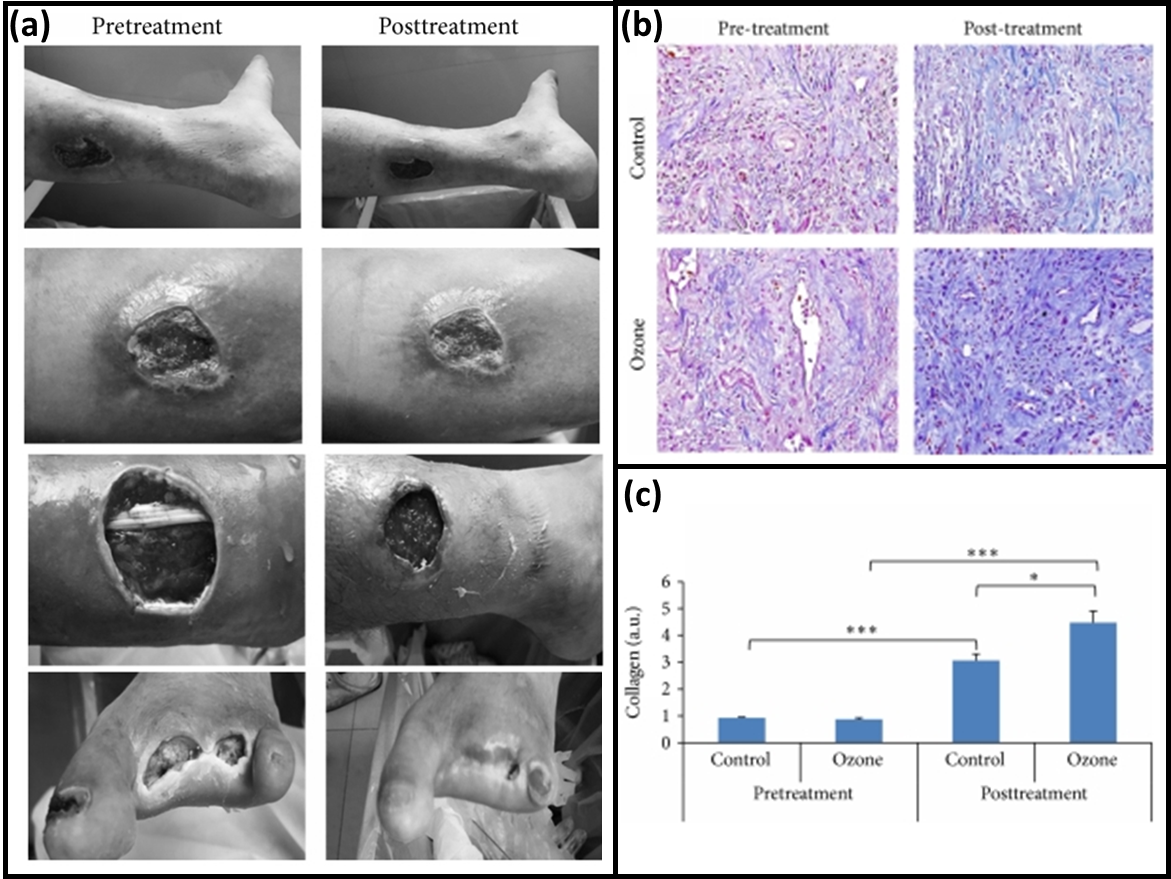 Fig. 6.
Fig. 6.Treatment of DFUs with Ozone Therapy. Oxygen-Ozone therapy with (a) Grading the wound level and the wound size each time of visit before and after treatment, with (b) collagen fibers in tissue specimens by Masson’s staining showing no collagen difference in the pretreatment and significant increase with ozone treatment elucidating its wound healing characteristics, (c) comparative concentration plot for collagen concentration before and after ozone treatment (Reproduced with permission from Ref. [123], copyright Hindawi 2014).
In another case report, ozone oil was tested to treat a 68-year-old patient with
necrotic vasculitis wounds and diagnosed with antiphospholipid syndrome. In this
study, different concentrations of ozone 20, 30, 40, 60, 70
Fitzpatrick et al. [105] reported a systemic review of ozone-based chronic wound treatment studies extracted from various online systems with a set of nine studies (n = 453) patients. The meta-analysis revealed that the results consistently favor ozone treatment in wound healing [105]. Thus, such treatment methods are also receiving considerable funding from various government agencies for larger and more systematic clinical trial studies, including Zekeriya Tasdemir from TC Erciyes University who conducted a triple-blind clinical trial investigating the wound healing properties of ozone on 36 participants who had undergone gingivectomy surgery [125]. For such therapies to be approved by FDA and be potentially used in standard clinical practices, the development of solid evidence backed by clinical trials is necessary. Thus, Erciyes University investigated the effect of Ozone treatment in wound healing using an ozone device procured from Ozonytron XL. The work involved 36 participants with randomized applications and parallel assignments. A split-mouth placebo-controlled design intervention model was used, and the ozone therapeutics efficacy was evaluated [126]. The patients with ozone exposure observed a clear change in stained surface area and lower postoperative pain on the 3rd day of exposure compared to the control, which showed minimal change.
St. John’s Research Institute clinical trials investigated the effect of the
Local Application of Ozone Gas on Infected Ulcers [127]. This study involved 68
patients with a conventional ozone generator applied for 1 h each day and a
placebo alternative [128]. Other trivial processes for ulcer treatment, such as
daily dressings, debridement, and antibiotics, were administered to both groups
throughout the study. These clinical trials prove that ozone is being considered
as a serious alternative to topical antibiotics and other conventional wound
therapy strategies. In another double-blind, randomized clinical trial, the ozone
efficacy in the treatment of diabetic foot ulcers with 61 patients that
participated in the study (32 were randomized to ozone treatment, and 29 to
placebo). The study showed a significantly higher rate of complete wound closure
in the group with ozone treatment (81% vs. 44%, p = 0.03) as compared
to the group with placebo treatment. Wounds with sizes of
Beyond such efforts to develop ozone as a standalone technique, the development
of adjunct therapy with various antibiotics and antimicrobial surfaces could
effectively eradicate microbial-invaded wounds. One serious complication drawback
of ozone therapy is the toxicity of ozone when applied at high concentrations.
This limits the maximum application of ozone therapy to low concentrations. By
utilizing a novel approach, such as ozone adjunct therapy with antibiotics, new
treatments could provide a more effective treatment without relying on high doses
of ozone or drugs. Particularly, as discussed before, the microbial cell walls,
on oxidation with the ozone molecules, compromises the structural integrity of
the cells by collapsing/weakening the lining membranes and creating pores. The
created pores in the membrane facilitate passive drug transport into the
bacteria/fungus and enhanced the effectiveness of antibiotics. For example,
Faraji et al. [129] developed a combination therapy based on
silver-containing wound dressing combined with ozone as an adjunct therapy for
diabetic foot ulcers (70
 Fig. 7.
Fig. 7.Adjunct silver and ozone therapy of a patient with a diabetic foot ulcer. (a) Stitched diabetic foot ulcer after discharge from hospital. (b) Diabetic foot ulcer after removing the stitches. (c) The silver dressing was applied, followed by ozone therapy. (d) After 6 sessions of ozone therapy. (e) After 1 month of ozone therapy. (f) Diabetic foot ulcer of the patient after about 4 months of therapy (reproduced with permission from Ref. [129], copyright Springer nature 2021).
Similarly, our group investigated this hypothesis by electrospinning
PVA-dissolved vancomycin and linezolid antibiotics-based nanofiber integrated
with an engineered wound patch. The study showed that the adjunct treatment of
ozone and antibiotics successfully extended the applicability of antibiotics
typically only effective on Gram-positive bacteria to treat P.
aeruginosa, a G-ve species with significant natural resistances. The treatment
was successfully administered in full by a portable ozone generation system and
wound patch coated with drug-eluting nanofibers, showing both the strength and
applicability of the adjunct therapy (Fig. 8). As proposed by the hypothesis, a
remarkable reduction in bacterial colonies (up to 5.5 log CFU/cm
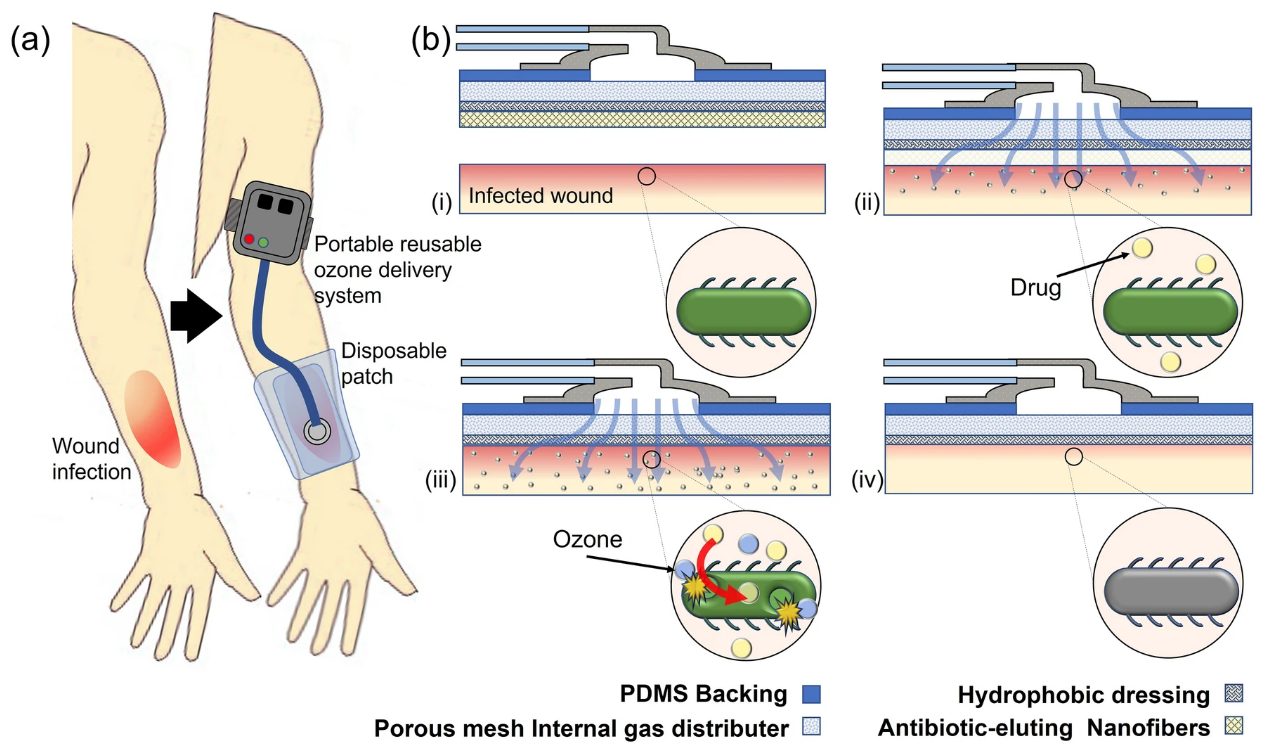 Fig. 8.
Fig. 8.A portable system was developed by Roth et al. for the topical administration of adjunct ozone and antibiotic therapy to treat G-ve bacteria. (a) Illustration of the application process of the ozone and antibiotic combination therapy. (b) Working principle: (i) the disposable patch coated with drug eluded water soluble nanofibers are applied to the wound bed, (ii) the nanofibers dissolve upon contact with the wound, releasing their drug-containing payload, (iii) as the antibiotics are released from the nanofibers, gaseous ozone at a localized concentration of 100 ppm is applied to the wound bed, leading to adjunct interactions between ozone and drug helping in eliminating bacterial infections (iv).
Ozone is a highly debated gas to be used in a therapeutic scenario. However, with such overwhelming proof by the research community, ozone could be a key therapeutic technique for topical wound healing which does not rely on antibiotics. In this setup, the concentration of the ozone administration is quite not clear, with various research groups administering different gas concentrations. Based on this review, it is observed that the concentration ranging between 5 and 60 mg/L ozone has been identified as a safe and effective working range for treating dermal wounds by medical standards [107]. Furthermore, quite a few research articles report a good therapeutic effect of ozone beyond 60 mg/L (~60 ppm) concentration which might call upon more intensive studies to analyze the toxicity effects. While no current studies have provided a definitive understanding of why low ozone concentrations are ineffective on human cells, one plausible explanation is that the ozone does not disrupt the eukaryotic cell wall of the host. This is likely due to the ability of the cells to compensate for the oxidative burden better than other pathogenic microbes for a therapeutic scale of 5–60 ppm. With proper safety guidelines and ozone-generating systems developed, this treatment could revolutionize the standard care for wound treatment by providing a safe and effective alternative therapy for dermal wounds without reliance on antibiotics and provide clinicians and patients with new options as antibiotic resistance continues to reduce the number of effective treatments.
While the safety concerns of gaseous ozone therapy are merited, well-engineered solutions to utilize this novel treatment have proven to exhibit extremely promising results. The data presented in this review indicates that ozone as a topical treatment for infected dermal wounds deserves serious consideration and further investigation as an alternative treatment to traditional therapies due to the broadly antimicrobial and cost-effective nature of the treatment, as well as stimulating wound healing. New advancements in ozone adjunct therapy with antibiotics is a promising area for significant research as the problem of antibiotic resistance continues to plague the healthcare industry. It is the belief of the authors that ozone is a promising treatment option that presents significant evidence for further investigation into treatment methods and technology that could safely and effectively change the lives of millions of patients and their families by providing new solutions to the significant burden of clinical wound care.
AR and AK contributed to the literature search and wrote the manuscript. RR provided oversight and direction to the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
The authors would like to thank the staff of the Birck Nanotechnology Center at Purdue University for their assistance throughout the project, along with the Purdue Mechanical Engineering Department, the Purdue Electrical and Computer Engineering Department, and the Purdue Materials Engineering Department.
School of Materials Engineering at Purdue University provided financial support for this effort.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








