1 Department of Innovative Technologies in Medicine and Dentistry, University of Chieti-Pescara, 66100 Chieti, Italy
2 Department of Biomedical, Surgical and Dental Sciences, School of Dentistry, University of Milan, 20122 Milan, Italy
3 Department of Interdisciplinary Medicine, University of Medicine Aldo Moro, 70124 Bari, Italy
4 Department of Oral Implantology, Dental Research Division, College Ingà, UNINGÁ, 29312 Cachoeiro de Itapemirim, Espirito Santo, Brazil
Academic Editor: Luca Testarelli
Abstract
Background: Peri-implant mucositis is a bacterial and inflammatory
disease of implant surrounding soft tissues. Chlorhexidine (CHX) associated with
PVP-VA (Polyvinylpyrrolidone/vinyl acetate) has an adjuvant function in the
antibacterial activity: it protects the film-forming layer, due to polymer, from
microbial contamination or over-infection, and prolongs its permanence, without a
direct action on the tissues and wounds. Sodium DNA is able to improve gingival
trophism. The aim of the study was to compare a gel with CHX and
antidiscoloration system (A.D.S.) 0.5% with the addition of PVP-VA and sodium
DNA with a placebo gel without chlorexidine in peri-implant mucositis treatment.
Methods: Trial design: A single center randomized controlled pilot
clinical trial was conducted on a total of 24 patients with mucositis, randomly
allocated into two groups. In the group (A) (n = 12) patients were treated with a
gel with CHX + A.D.S. 0.5% while a placebo gel was used in the group (B) (n =
12). Outcome: Plaque Index (PI) (primary outcome), Bleeding on probing (BOP) and
Gingival Index (GI) (secondary outcomes) were assessed at baseline and at 2 weeks
after treatment period. Results: Mean PI score recorded at baseline was
2.4
Keywords
- peri-implant disease
- mucositis
- bacterial biofilms
- plaque
- oral hygiene
- dental implants
The peri-implant mucositis is a dental implant-associated inflammatory state of the soft tissues characterized by the presence of bleeding on probing, in the absence of bone resorption. There may be other signs of inflammation such as swelling, redness, suppuration. Histologically, the peri-implant tissues have been studied mainly in many different animal models, and subsequently confirmed in human studies. The peri-implant mucosa in physiological conditions has a height of 3–4 mm and is made up of connective tissue relatively poor in fibroblasts (3%) covered in part by keratinized epithelium mucosa and in part by non-keratinized epithelium lining mucosa [1]. The main differences with the periodontal tissues are the absence of the cementum and periodontal ligament, and therefore the absence of fibers [2, 3]. The peri-implant tissues are also less vascularized in the area between the bone crest and the junctional epithelium than the corresponding connective tissue surrounding the natural tooth [4, 5]. The implant sites also have a longer epithelium [6].
An increased probing depth is not decisive in formulating the diagnosis of peri-implant mucositis, as it can be determined both by edema and by the reduced resistance to probing of the inflamed tissues. Oral peri-implant mucositis is defined as a reversible multifactorial inflammation of the peri-implant soft tissues with up to 64% prevalence among individuals with implants [7, 8]. This condition affects only the soft marginal peri-implant tissues, not including alveolar bone loss. However it could be an early stage of a subsequent peri-implantitis, which has an incidence between 5–10% [9].
The diagnostic definition of peri-implant mucositis is based on following criteria: (1) presence of peri-implant signs of inflammation (redness, swelling, line or drop of bleeding within 30 seconds following probing), (2) no additional bone loss following initial healing [10].
In literature, the treatment of peri-implant mucositis normally includes mechanical calculus removal by the surface of the implant by curettes, ultrasonic tips, air-abrasives or laser devices [11]. In this way the adjunctive administration of local antibiotics or antiseptics seems to provide a increase of the treatments response, in order to prevent biofilms and calculus adhesion around the implant surfaces [12].
The chlorhexidine (CHX) is now known for its optimal antimicrobial and antiplaque properties and its use is considered the Gold Standard for antibacterial action in the oral hygiene [13]. The CHX inhibits the formation of new bacterial plaque and disrupts the already stratified one by acting on its matrix thanks to the competition with Calcium ions [13]. The most common side effect is represented by brownish pigmentations that occur on dental surfaces, restorations, dental prostheses and tongue. This effect, undermining the compliance of the patient, often limits its use [14]. The A.D.S., allows to combine antiplaque with aesthetic action, reducing the the formation of pigmentation on tooth enamel without reducing the CHX effectiveness [15]. The film-forming property of PVP-VA allows the formation of a protective film that shields the underlying tissues from bacterial or chemical attacks. These polymers are able to protect dental surfaces or oral mucosal membranes for several hours with a slow degradation with the release of active ingredients embedded [16]. The layer of PVP-VA blocks the initial aggregation of the plaque, slowing down the creation of the bacterial biofilm and, consequently, the aggression on the tissues and their inflammation [16]. A recent study analyzed the ability of the film-forming layer of PVP-VA contained in a mouthwash compared to a placebo. The results show the effectiveness of PVP-VA in inhibiting bacterial plaque alone compared to placebo, in absence of other antiseptic active substances in the mouthwash [17]. Chlorhexidine, in presence of PVP or PVP-VA, fails to act on oral tissues because its action is shielded by the presence of the film-forming layer of polymers that limit the antiseptic action. For this reason clinical studies that have investigated this association reporting a less effective action of CHX alone [18]. However, it should be noted that the high inhibition to plaque formation offered by the mechanical obstacle of the film-forming layer of PVP and PVP-VA copolymer is to be considered the antiseptic effect offered by the association chlorhexidine-PVP (or PVP-VA) [18, 19, 20]. In fact, if the plaque fails to adhere to the tissues and mature, it cannot exert its own inflammatory action, because it is unable to aggregate in the biofilm and to generate an inflammatory state. The role of chlorhexidine in a PVP-VA association has an adjuvant function: it protects the film-forming layer from contamination or bacterial overinfection, and prolongs its permanence, not acting directly on tissues and wounds, as limited by the mechanical film layer barrier [21]. Therefore, the presence of PVP-VA in a mouthwash or in a periodontal gel is able, alone, to ensure antibacterial protection [22]. Sodium DNA is a natural molecule widely used in cosmetics as an anti-aging product [22]. The using of DNA-sodium has been proved to decrease in vitro the oxidative stress action on the oral tissues and the inflammatory response in a wide spectrum of coltures [23, 24]. This effect has been demonstrated in vivo in a in periodontitis control in association with CHX/PVP-PA mouthwash formulation compared to the placebo [25]. The most frequent side effect of the CHX formulation in form of mouthwash and gels is characterized by brownish pigmentations on dental surfaces, restorations, dental prostheses and tongue [15]. In addition, others side effects are taste alteration, higher tartar formation expecially in case of prolonged administration over 14 days [26, 27, 28, 29]. This effects are able to decrease consistently the patient compliance and often could limits its administration [14]. The Anti Discoloration System (ADS) is able to combine the antiplaque activity with aesthetic effect, reducing the tooth pigmentation with no reducing of the CHX action [18].
The aim of this study was therefore to evaluate the effect of a gel with CHX and
A.D.S. 0.5% with the addition of PVP-VA and sodium DNA and to compare it with a
placebo gel in patients with peri-implant mucositis, that is presence of bleeding
on probing in the absence of bone loss and pathological pockets (
The protocol for this study was designed in accordance with the Helsinki Declaration, with the Good Clinical Practice Guidelines for clinical trials on medicinal products (GCP E6 R2:2017) and UNI EN ISO 14155:2017 and approved by the Ethical Committee for Biomedical Research of Chieti and Pescara (Prot. N. 1776/5.12.2019). The adult patients have been recruited at the Department of Innovative Technologies in Medicine and Dentistry of the University “G.D’Annunzio” of Chieti-Pescara and signed the informed consent.
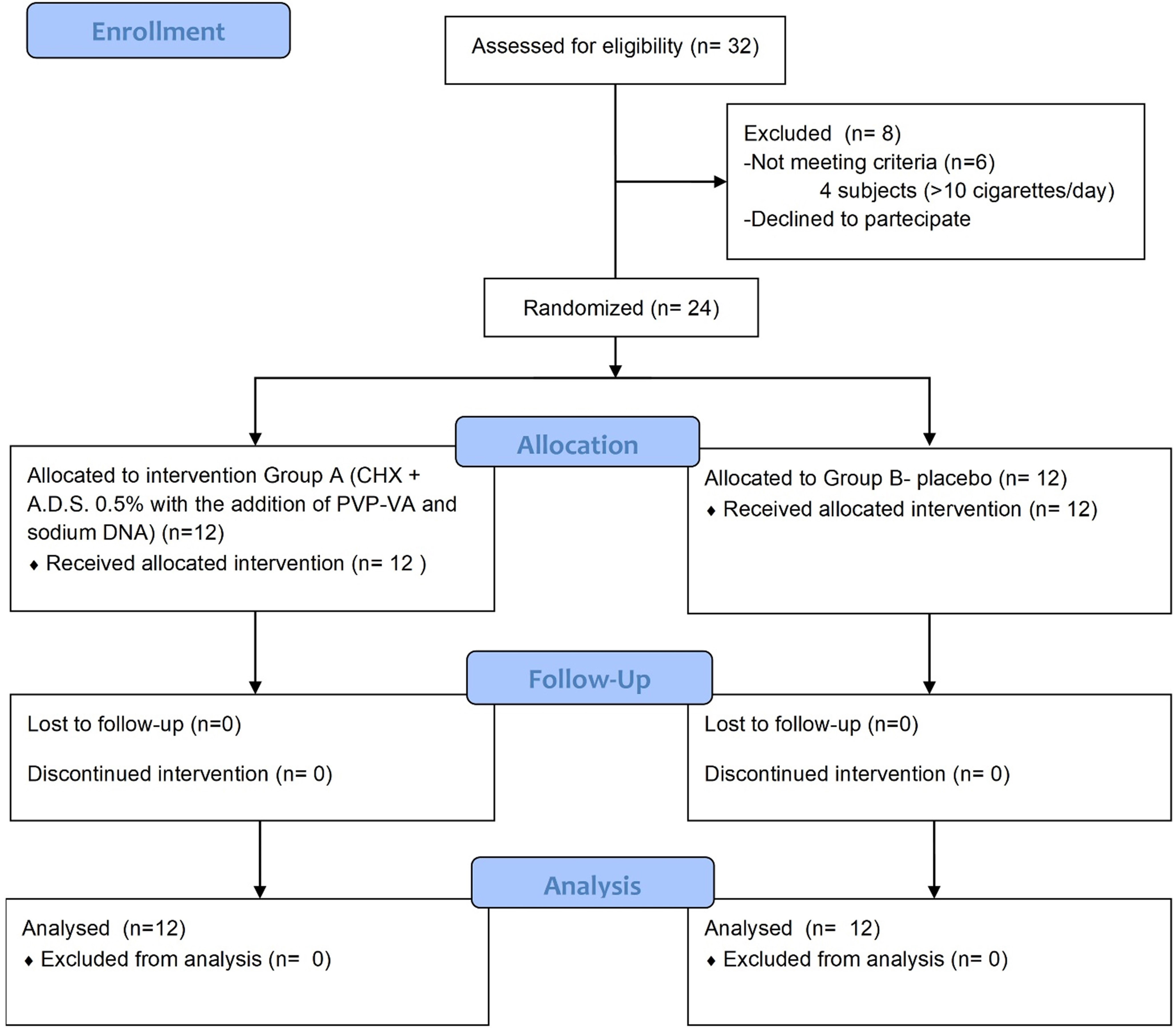
The study was designed as a randomized single-center controlled, triple-blind trial with two parallel groups (Fig. 1, Ref. [30]).
Twenty-four adult patients were enrolled in this trial, they were equally and randomly divided into two groups: group A (n = 12) treated using a gel with CHX + A.D.S. 0.5% with the addition of PVP-VA and sodium DNA and group B (n = 12) administered a placebo, an aqueous inert free of chlorhexidine gel. All patients fulfilled these inclusion criteria: (1) good general health in absence of relevant systemic diseases such as diabetes, immune diseases, haematological diseases; cancer; severe infectious diseases such as HIV or viral hepatitis with signs and/or symptoms of liver failure; cognitive difficulties; intellectual difficulties; motorial deficits, (2) adult age (over 18 years), (3) presence of one implant with mucositis, defined as bleeding on probing with no evidence of peri-implant bone loss, (4) no implant mobility, (5) presence of at least 2 mm of peri-implant keratinized gingiva, (6) implant with a single crown or bridge pillars with a maximum of 3 elements, (7) no overload or occlusal trauma signs.
Exclusion criteria were as follows: pregnant subjects, strong smokers (more than 10 cigarettes/day); fully edentulous patients with exclusively implant rehabilitations; allergy to chlorhexidine gluconate or to other components of the gel formulation and of the placebo.
All patients agreed to adhere to the trial and signed an informed consent.
Simple randomization was conducted at the Department of Innovative Technologies in Medicine and Dentistry of the University “G.D’Annunzio” of Chieti-Pescara, using computer-generated random numbers with an allocation ratio of 1:1. Allocation sequence was concealed using sequentially numbered, opaque, sealed envelopes, which were opened only after the professional hygiene. Blinding of participants, data collectors and data analysts were applied. Operators and patients were not informed about the gel content: the blinded gel had a similar packaging, with no signs or references that could indicate the formulation of the gel.
The enrolled patients with peri-implantitis mucositis, were properly visited and they were subjected to professional oral hygiene session targeted to these sites. Implant sites with mucositis have been cleaned with the aid of titanium or carbon curettes; supragingival calculus removal employing ultrasonic scaler tips and accurate polishing with prophylaxis pastes were also performed in the first session (T0). Baseline parameters of PI, BOP and GI index were properly recorded before the previously described first oral hygiene treatment. Therefore accurate oral hygiene instructions were given, taking care that patients maintained a high level of hygiene throughout the clinical protocol period. No rinses, drugs or medications were prescribed to patients with the only exception of those deemed essential for the health of the patient. Each group was treated with the same protocol of oral hygiene but with a different assigned gel. After the recording of the clinical variables and the oral hygiene session, each patient received an anonymous gel tube, a syringe and an applicator nozzle. During the study the patients were required to use the daily mechanical oral hygiene except for the evaluated implant site. The patient was carefully instructed on the application methods of the assigned gel in the implant site: an application of the gel twice a day, for 14 days was prescribed. In order to reduce inter-operator variability a single dental provider has carried out the oral hygiene, as well as the evaluation and recording of clinical variables. Follow-up evaluations were performed after 14 days (T1). Neither the researchers, therapists nor the patients were aware of the group allocation.
The tested formulations (Curasept Spa, Saronno, VA, Italy) was composed by a solution of 0.5% Chlorexidine Anti Discoloration System (ADS), PVP-VA and Sodium DNA. The ADS system is composed of ascorbic acid and sodium metabilsulfate, while the adding of 0.12% polyvinylpyrrolidone/vinyl acetate proven theirs efficacy to avoid the chlorexidine capability to bind the surface of the tooth and the soft tissue membranes and theirs pigmentation maintaining the anti-plaque efficacy. The 0.2% of Sodium DNA Sodium DNA (Na-DNA) is a biologically active, functional compound obtained by deoxyribonucleic acid of the gonadic tissue of male sturgeons. These molecules submitted a purification, de-polymerization process and finally neutralization sodium (Na+) ions. The PVP-VA co-polymer associated with with sodium DNA improve the gingival tissues trophism and improve their effect on bacterial plaque control. The placebo mouthwash was composed by an aqueous alcohol solution (Betafarma, Cesano Boscone, Milan, Italy).
The plaque index (PI) was recorded at all timepoints to evaluate the presence or
absence of dental plaque along the mucosal margin was recorded. The recording has
been performed by two blinded calibrated operators and the mean value was
considered for further statistical evaluations. Moreover, the bleeding on probing
(BOP) was recorded to assess the presence/absence of bleeding after probing of
the gingiva. Moderate force (
The descriptive analysis of the data was carried out: the quantitative variables
are presented with mean and standard deviation. The statistical evaluations were
carried out by means of two sample t test to compare for each group of
patients analyzed, the values detected at time T0 and the values measured at time
T1. The level of statistical significance was set at 5% (p
Twenty-four patients, with a mean age of 64.2
| General characteristics | |
| Total sample size | n = 24 subjects |
| 12 subjects (Group A); 12 subjects (Group B) | |
| Average age of subjects | 64.2 |
| Age range | 41–74 years |
| Gender | Male/Female ratio 0.92/1 (Group A); 0.95/1 (Group B) |
| Total of rated implant with mucosistis | N = 62 implants |
| 32 implants (Group A); 26 implants (Group B) | |
| Incisal/canine region implants | 82.26% (51 implants) |
| Molar/premolar region implants | 17.74% (11 implants) |
| Mean pocket depth (DS) | Group A: 2.2 |
The results for the gingival index at baseline and at T1 are summarized in Table 2.
| Plaque index | Group A | Group B | p value |
| (mean, SD) | Test | Control | |
| Baseline | 2.4 |
2.2 |
p |
| [95% CI: –2.682/7.482] | [95% CI: –4.153/8.553] | ||
| 2 weeks | 0.5 |
1.7 |
p |
| [95% CI: –4,582/5,582] | [95% CI: –9,736/13,14] | ||
| BOP score | Group A | Group B | p value |
| (mean, SD) | Test | Control | |
| Baseline | 57.1% |
55.3% |
p |
| [95% CI: –136,0/250,2] | [95% CI: –93,36/204,0] | ||
| 2 weeks | 14.3% |
45.4% |
p |
| [95% CI: –69,56/98,16] | [95% CI: –79,12/169,9] |
At T0 time, the baseline related to the Plaque Index detected was 2.4
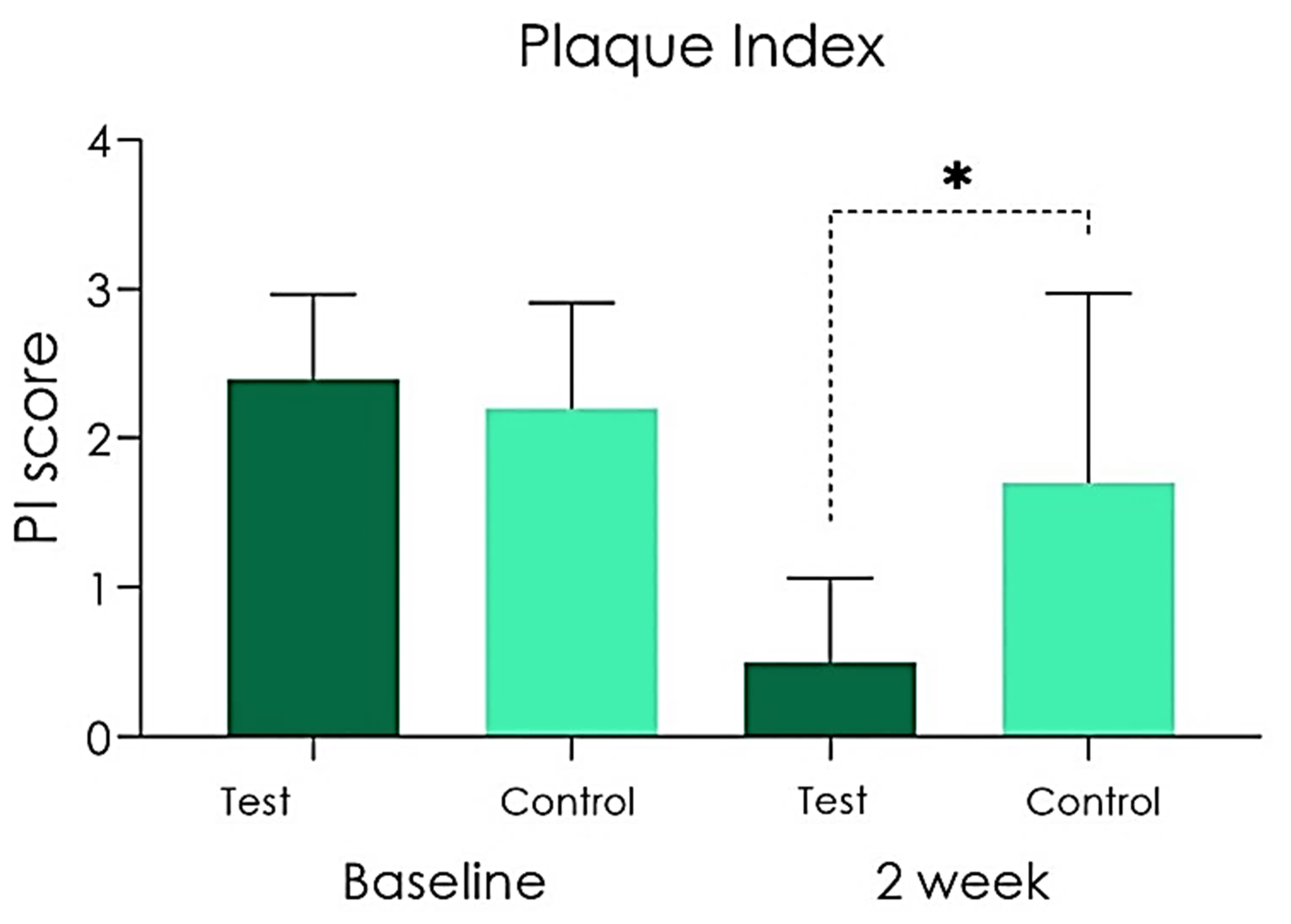 Fig. 2.
Fig. 2.
Plaque Index scored at baseline and after 2 weeks [*p
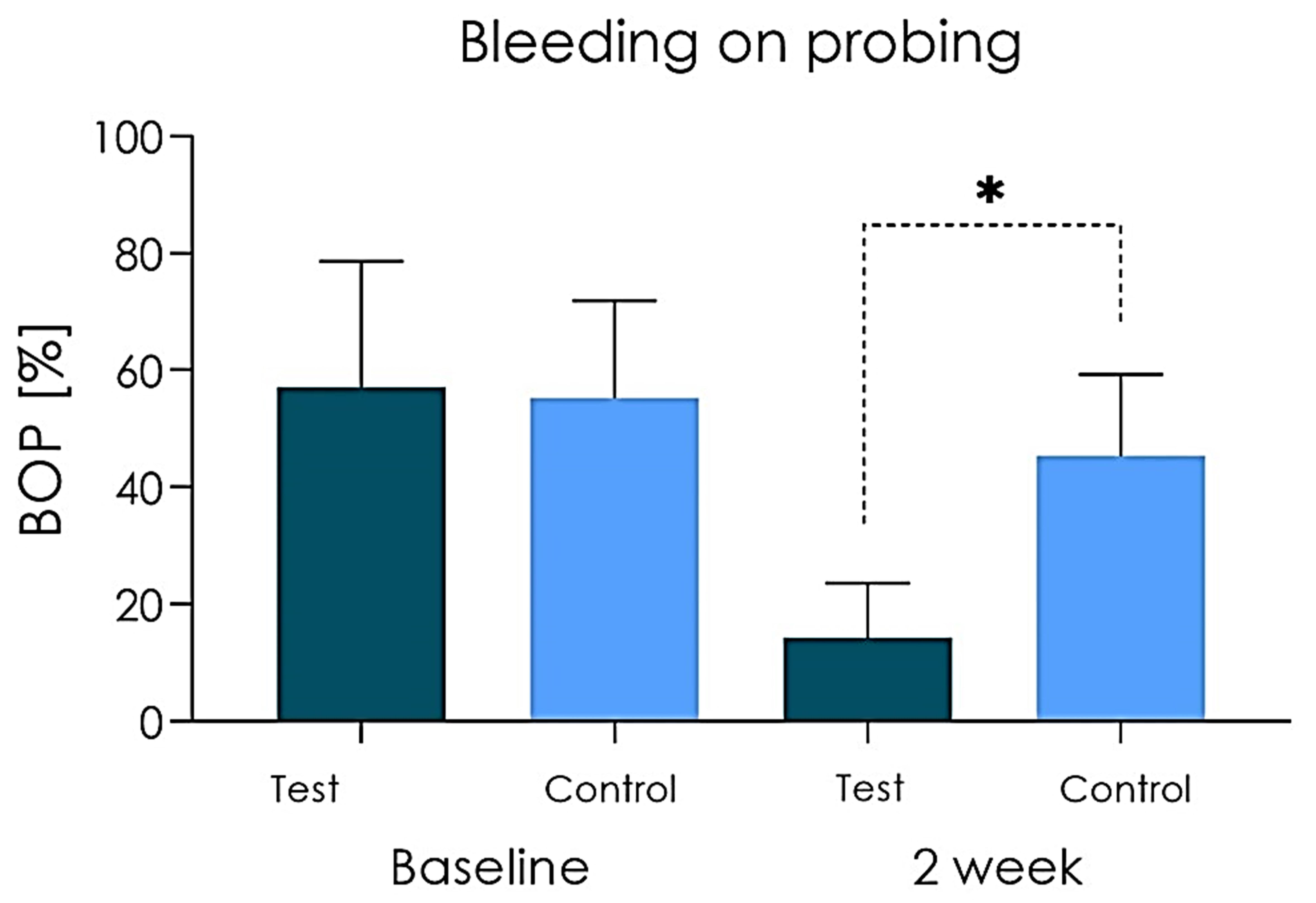 Fig. 3.
Fig. 3.BOP (%) scored at baseline and 2 weeks from baseline.
The chlorhexidine (CHX) has been investigated for several years as an antimicrobial agent for the chemical control of plaque formation and it is unanimously recognized as one of the most effective cationic antiseptic compound currently used in oral hygiene [13, 14]. The film-forming properties of PVP-VA (Polyvinylpyrrolidone/vinyl acetate) in protecting dental surfaces or oral mucosal membranes, even if not intact, are known [16]. Moreover, the sodium DNA in form of topical formulation is able to improve gingival trophism and human gingival epithelium repair [24]. In our work we have analyzed, as primary outcome, the antiplaque activity of a gel containing Chlorexidine and A.D.S. 0.5% with the addition of PVP-VA and Sodium DNA compared to a placebo gel in the peri-implant mucositis treatment [22]. From a histological point of view, the inflammatory lesion characteristic of peri-implant mucositis is well defined and confined to the most coronal connective tissue, lateral to the junctional or pocket epithelium [2, 10, 31]. It contains an inflammatory infiltrate rich in vascular structures, plasma cells and lymphocytes, which does not extend apically in the supracrestal connective area [32, 33]. Strong scientific evidence identifies plaque as the etiological factor of peri-implant mucositis [34]. However, there is interindividual variability in the immune response, and factors such as smoking, diabetes mellitus, and radiation therapy may be modifying factors [35, 36].
Certainly, the oral environment is exposed to oxidative stress and free- radicals action occurred by the inflammation state [37, 38]. The state of chronicity induces an unbalanced connection between these exogenous agents and the physiological response to the damage repair [39, 40, 41]. The hydrolyzed DNA is reported to a protective action against the oxidative stress in the oral cavity [23, 24] with a very high strong versatility in its formulation. In fact, it could be find in combination with anti-microbial agents in form of topical gels and mouthwash solution [18, 42]. In vitro, the hydrolyzed DNA a significant effect of the proliferation and activity of the fibroblast cells due to the activation of A2 purinergic receptors [43]. The repair activity seems to be induced following a synergic action to the growth factors cascade including epidermal growth factor (EGF) platelet-derived growth factor (PDGF) and fibroblast growth factor (FGF) [44].
Experimental studies have shown that peri-implant mucositis is reversible, and that it is therefore possible to restore health through plaque control procedures, while a resolution of clinical signs may take more than 3 weeks after treatment [31]. The present study evaluated the short-term effect after two weeks of treatment, in order to detect the effect of the teraphies on marginal peri-implant inflammation. Gingival inflammation can be assessed by different methods including gingival indices increaseing its specificity if associated to the bleeding on probing index. In this study, there was no statistically significant differences in the Plaque Index (primary outcome) between the Test and Placebo groups at baseline while a significant difference was found between the two groups after 14 days of treatment, with a decreased bacterial plaque in the Group A (test). As for secondary outcomes, less bleeding (Bleeding on Probing Score) emerged at 2 weeks of treatment in the Test group compared to Control group. According to Mish et al. [45], the clinical Bleeding on Probing index, represents a simple and accurate indicator of the health of peri-implant tissues. The Bleeding on probing (BOP) is a basic clinical exploratory sign: a positive bleeding on probing is indicative of gingival inflammation. The diagnostic utility of BOP is high, although its prognostic value is debated. Many authors concluded that the presence of BOP does not indicate an increased risk of tissue destruction and bone support, as this clinical sign has a low positive predictive value [2, 3, 21]. Conversely, the negative predictive value of the bleeding on probing is high. A prospective study on implants confirmed that, similar to the situation of natural teeth, the absence of bleeding on probing has high negative predictive values and therefore can be interpreted as a sign of stability of peri-implant soft tissues [46, 47]. Within a periodontal treatment, it is essential to ensure healthy gums, without inflammation and therefore in the absence of BOP [8, 48, 49]. In the present study, at T1 evaluation an absent/mild degree of inflammation (Gingival Index) was present in patients that used CHX gel while a mild/moderate degree of inflammation was found in the placebo group at the same time. According to the literature peri-implant mucositis is considered as a superficial clinical phase of peri-implantitis, that is characterized by a progressive involvement of deep tissues with bone resorption. The control of many factors such as bacterial plaque, tartar deposits and home and professional maintenance are fundamental aspects for the long-term functional survival of supported implant rehabilitations [3, 50, 51]. At experimental time T1 a statistically significant difference emerges between the test group and the placebo group with regard to the primary parameter Plaque Score. The beneficial effect induced by the CHX and ADS 0.5%, PVP-VA and sodium DNA gel on the peri-implant soft tissues are also evident in relation to the secondary outcomes of BOP and GI, which showed a statistically significant difference between patients in favour of the Test group compared to the placebo gel group.
According to our results, the use of a Chlorexidine gel with Anti Discoloration Syestem (ADS) 0.5%, PVP-VA and Sodium DNA, tested for two weeks in peri-implant mucositis treatment, appears to be effective in leading to a reduction in the clinical parameters of mucous inflammation and reduced deposits of bacterial plaque, without adverse effects and risks associated with treatment.
Conceptualization—AS; Methodology—AS, FL; Investigation—AS, FL; Data curation—FL, Writing and reviewing of the manuscript—AS, FL; Supervision—GT, FI; Validation—AS, FL, GT, FI.
Ethical Committee for Biomedical Research of Chieti and Pescara (Prot. N. 1776/5.12.2019).
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. FL is serving as one of the Editorial Board members and Guest editors of this journal. We declare that FL had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to LT.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.