1 Centre for Foundation Studies in Science, Universiti Malaya, 50603 Kuala Lumpur, Malaysia
2 Centre for Ionics Universiti Malaya, Department of Physics, Faculty of Science, Universiti Malaya, 50603 Kuala Lumpur, Malaysia
Abstract
Ultraviolet (UV) radiation poses significant risks to human health and interior materials, creating a demand for effective transparent shielding solutions. While organic polymer-based filters are widely used, they often suffer from poor weatherability and mechanical instability. This study aims to develop a durable, transparent, and hydrophobic UV-shielding coating using a methyltrimethoxysilane (MTMS) matrix reinforced with tungsten oxide (WO3) nanoparticles, specifically designed for scalable retrofitting on glass substrates. Nanocomposite coatings were prepared by dispersing WO3 nanoparticles into MTMS/ethanol solution. Four formulations were synthesized with WO3 loadings of 0, 6.25, 12.5, and 25 wt% relative to the precursor. The coatings were applied to glass slides using a facile sponge-coating technique. Characterization included Field Emission Scanning Electron Microscopy (FESEM) for morphology, X-ray Diffraction (XRD) for crystallinity and Fourier-Transform Infrared (FTIR) spectroscopy for chemical bonding. Optical performance was evaluated via ultraviolet-visible (UV-Vis) spectroscopy (200–900 nm) and surface wettability was assessed using water contact angle measurements. Mechanical durability was tested using the standard pencil hardness test before and after 48 hours of water immersion. XRD and FTIR analyses confirmed the successful covalent anchoring of crystalline WO3 nanoparticles into the siloxane network. FESEM revealed a crack-free surface with homogeneous nanoparticle dispersion. A direct trade-off between UV-shielding and visible transparency was quantified. The 25 wt% sample achieved the highest UV-shielding (81.08%) but suffered from low visible transparency (TVis = 50.44%). The 6.25 wt% formulation was identified as optimal, delivering 63.23% UV-shielding while maintaining high transparency (TVis = 76.32%), surpassing the architectural standard of 70%. All coatings maintained 9H pencil hardness even after water immersion, though hydrophobicity decreased slightly with higher WO3 loading due to surface hydroxyl groups. The MTMS-WO3 coatings demonstrate an excellent balance of optical clarity, UV protection and mechanical hardness compared to conventional soft-polymer films. The 6.25 wt% formulation is proposed as the optimal concentration for durable and transparent UV-shielding applications in architectural and automotive glazing.
Graphical Abstract
Keywords
- coating
- methyltrimethoxysilane
- tungsten oxide
- polymer
- UV-blocking
- visible-blocking
The increasing demand for energy-efficient buildings and the need to protect materials and human eyes from harmful radiation have driven significant research into advanced coating technologies for glass surfaces. Conventional glass offers only limited protection against ultraviolet (UV) radiation, and excessive visible light can increase cooling costs, cause interior fading, and pose potential health risks [1, 2]. While various thin-film technologies and laminated glass products exist, there is still a need for cost-effective, durable, and highly efficient coatings that can be easily applied to both new and existing glass substrates. Developing coatings that can selectively block specific wavelength ranges while maintaining desired aesthetic and functional properties is a key area of interest.
Among the various materials explored for UV-light blocking applications,
tungsten oxide (WO3) nanoparticles are a suitable candidate, as WO3
nanoparticles exhibit strong absorption in the UV [3] regions, making them ideal
for blocking detrimental solar radiation. This originates from the electronic
structures and optical properties of the WO3 nanoparticles, which result in
the band gap of 3.2 eV (
To address these challenges, selecting a suitable binder system is key for a successful nanoparticle-based coating. Several studies report the use of polypyrrole [9], epoxy resin [10], acrylic resin [7], and polyvinyl alcohol [5]. In this study, methyltrimethoxysilane (MTMS) was used as the binder. MTMS is an organosilane precursor that, through a sol-gel process involving hydrolysis and condensation reactions, forms a durable, transparent, and thermally stable cross-linked silica-based (SiOx-based) network [11]. Its methoxy groups allow for strong chemical bonding with the glass substrate via silanol groups, ensuring excellent adhesion [12]. Moreover, the methyl groups provide hydrophobicity and flexibility to the resulting film, and the silane structure can act as a coupling agent, improving the dispersion and integration of the WO3 nanoparticles within the coating matrix [13].
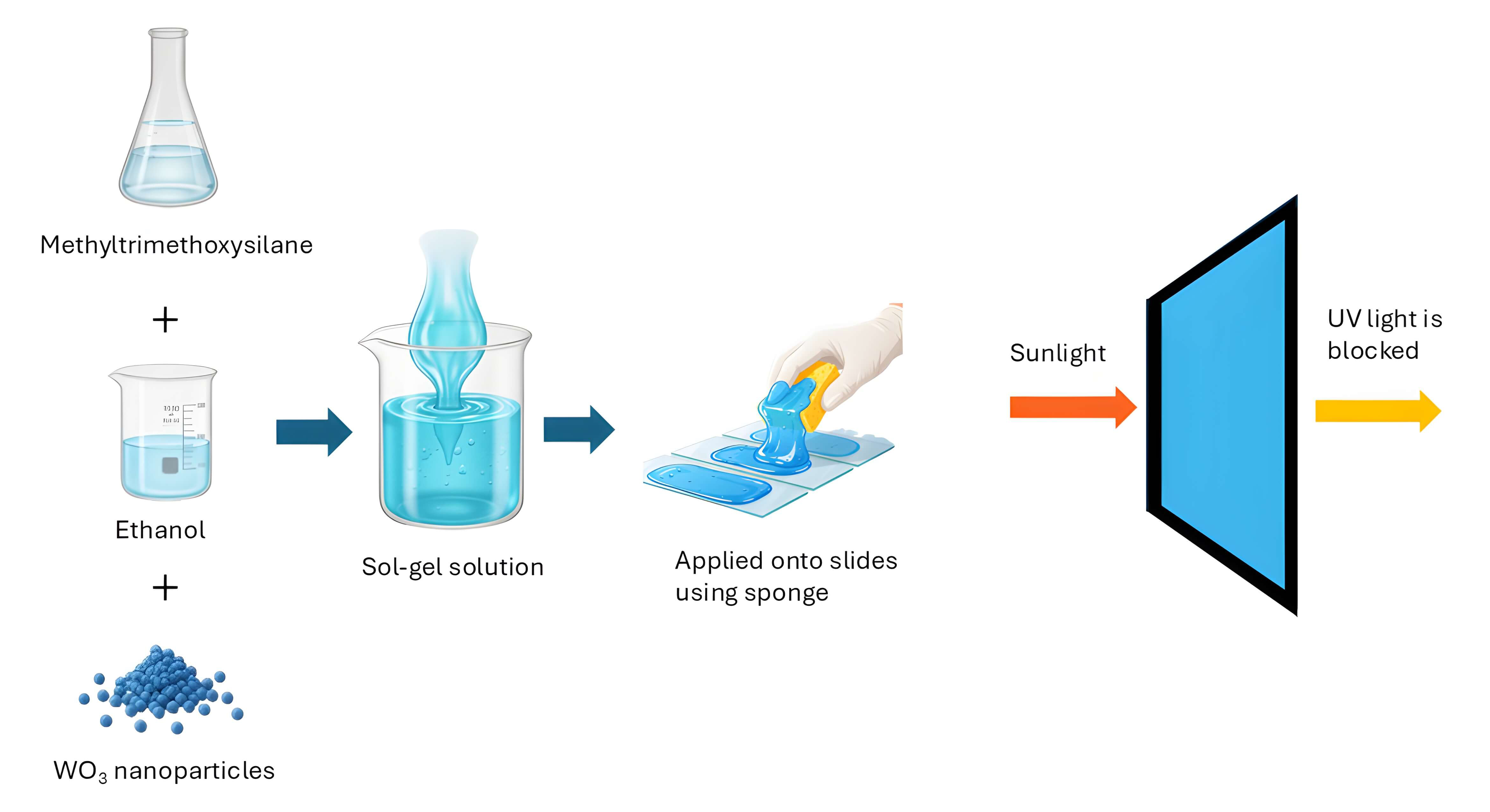
This research develops and characterizes a transparent coating on glass substrates, fabricated using WO3 nanoparticles as a filler and MTMS as a binder. The coating was applied onto glass surfaces using a sponge. This article reports an evaluation of the coating’s effectiveness at blocking UV and visible light while assessing its optical transparency, durability, and wettability. This work aims to provide a simple and scalable approach to producing optical blocking coatings for various applications, such as energy-saving windows and protective eyewear.
WO3 nanoparticles, MTMS, ethanol, and nitric acid were procured from Sigma Aldrich and used without further purification. The size of the WO3 nanoparticles was confirmed using Transmission Electron Microscopy (TEM) (HT7700, Hitachi High-Tech Corporation, Tokyo, Japan), and their crystallinity was studied by X-ray Diffraction (XRD) (Empyrean, PANalytical, Almelo, The Netherlands). No further characterization was carried out because the WO3 was commercially produced.
MTMS was diluted by mixing with an ethanol solution at a 1:1 weight ratio. To prepare the nanocomposites, WO3 nanoparticles were added as a powder to a fixed volume of the solution. 25 wt% WO3 nanoparticles were added to the mixture and were stirred for 2 hours. The resulting mixture was labeled as MTMS-WO3-25%, indicating it contained 25 wt% WO3 nanoparticles by weight. Mixtures containing 12.5 and 6.25 wt% WO3 nanoparticles were also prepared by adjusting the weight accordingly.
To prepare the sol-gel from the mixtures, nitric acid (pH 1) was added as a catalyst. The acid was 10% by weight in the mixtures, and the mixtures were stirred for 2 minutes. The sol-gel was then applied to glass slides using a sponge and allowed them to dry at room temperature. In all, 5 samples were prepared for evaluation in this experiment (Table 1).
| Sample | Label |
| Uncoated glass slide | Uncoated glass |
| Glass coated with only MTMS, no WO3 was added | MTMS-WO3-0% |
| Glass coated with MTMS containing 6.25 wt% of WO3 | MTMS-WO3-6.25% |
| Glass coated with MTMS containing 12.5 wt% of WO3 | MTMS-WO3-12.5% |
| Glass coated with MTMS containing 25 wt% of WO3 | MTMS-WO3-25% |
X-ray Diffraction was used to confirm the crystallinity of WO3 does not change. Ultraviolet-visible (UV-Vis) spectroscopy (200–900 nm) (UV-1900i, Shimadzu Corporation, Kyoto, Japan) was used to measure the transparency of the coating. Fourier Transform Infrared (FTIR) Spectroscopy (Spectrum 400, PerkinElmer, Waltham, MA, USA) was utilized to identify the functional groups, hydrolysis and condensation reactions of the coating. A pencil scratch hardness test was conducted to evaluate the mechanical strength of the developed coatings. The contact angle was measured to evaluate the wettability of the coatings.
Fig. 1 illustrates the X-ray diffraction (XRD) patterns of the as-received WO3 nanoparticles. The diffraction profiles show agreement with the standard monoclinic phase of WO3 (JCPDS Card No. 43-1035). The high intensity and narrow full-width at half-maximum (FWHM) of the diffraction peaks indicate a highly crystalline structure. The XRD patterns for the composite coating (MTMS-WO3-25%) similarly exhibit the characteristic crystalline peaks of WO3. This preservation of crystallinity was expected, as the interaction between the MTMS and WO3 is a surface-limited phenomenon. Since the chemical bonding is restricted to the particle-matrix interface, the bulk crystalline lattice of the WO3 nanoparticles remains unaltered during the coating process.
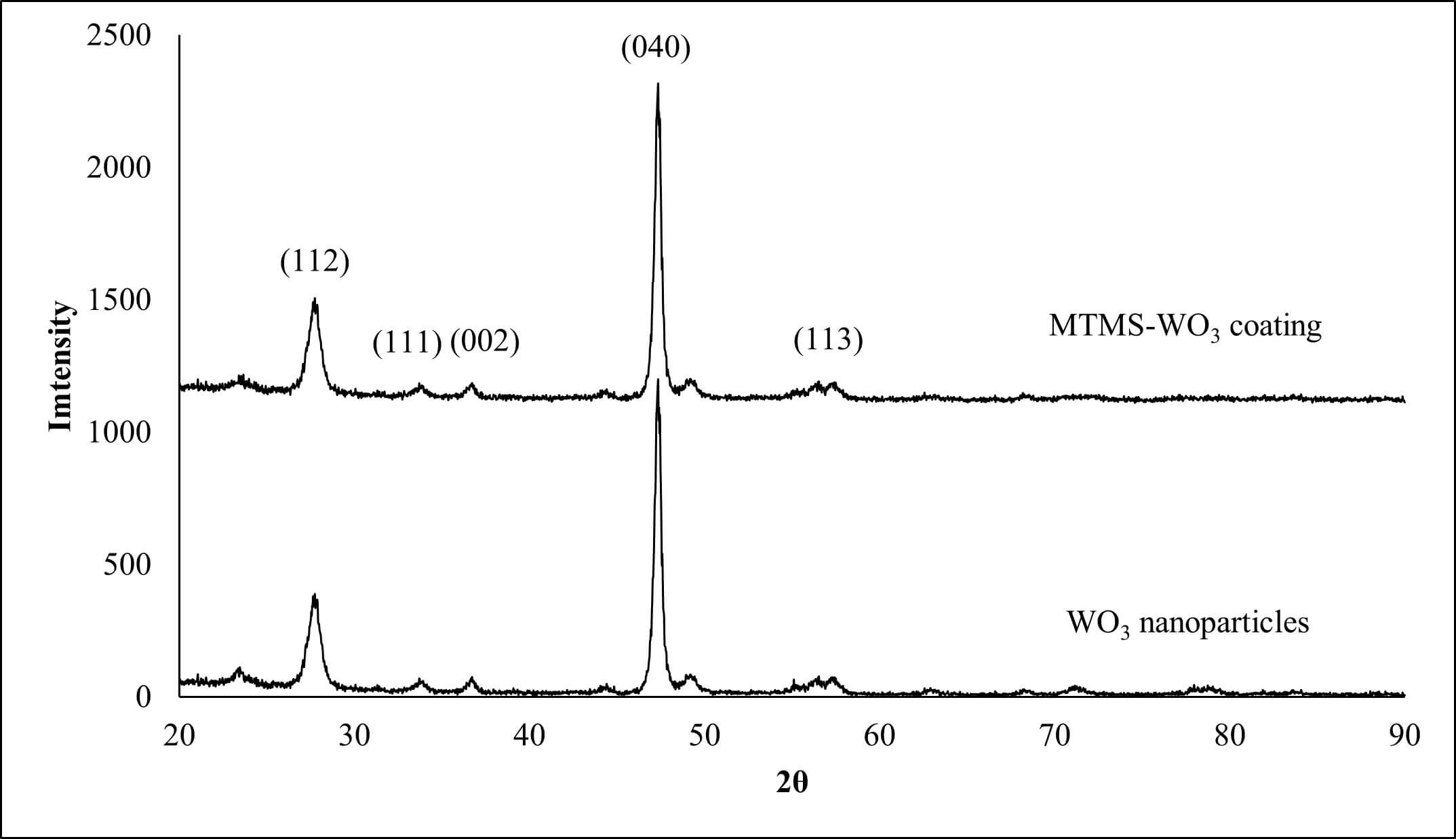 Fig. 1.
Fig. 1.
X-ray diffraction (XRD) for WO3 nanoparticles and the coating (MTMS-WO3-25%). MTMS, methyltrimethoxysilane; WO3, tungsten oxide.
Fig. 2 presents a TEM micrograph of the WO3 nanoparticles. The particles exhibit a predominantly spherical and ellipsoidal morphology. Analysis of the image reveals a particle diameter of 5.0–15.0 nm.
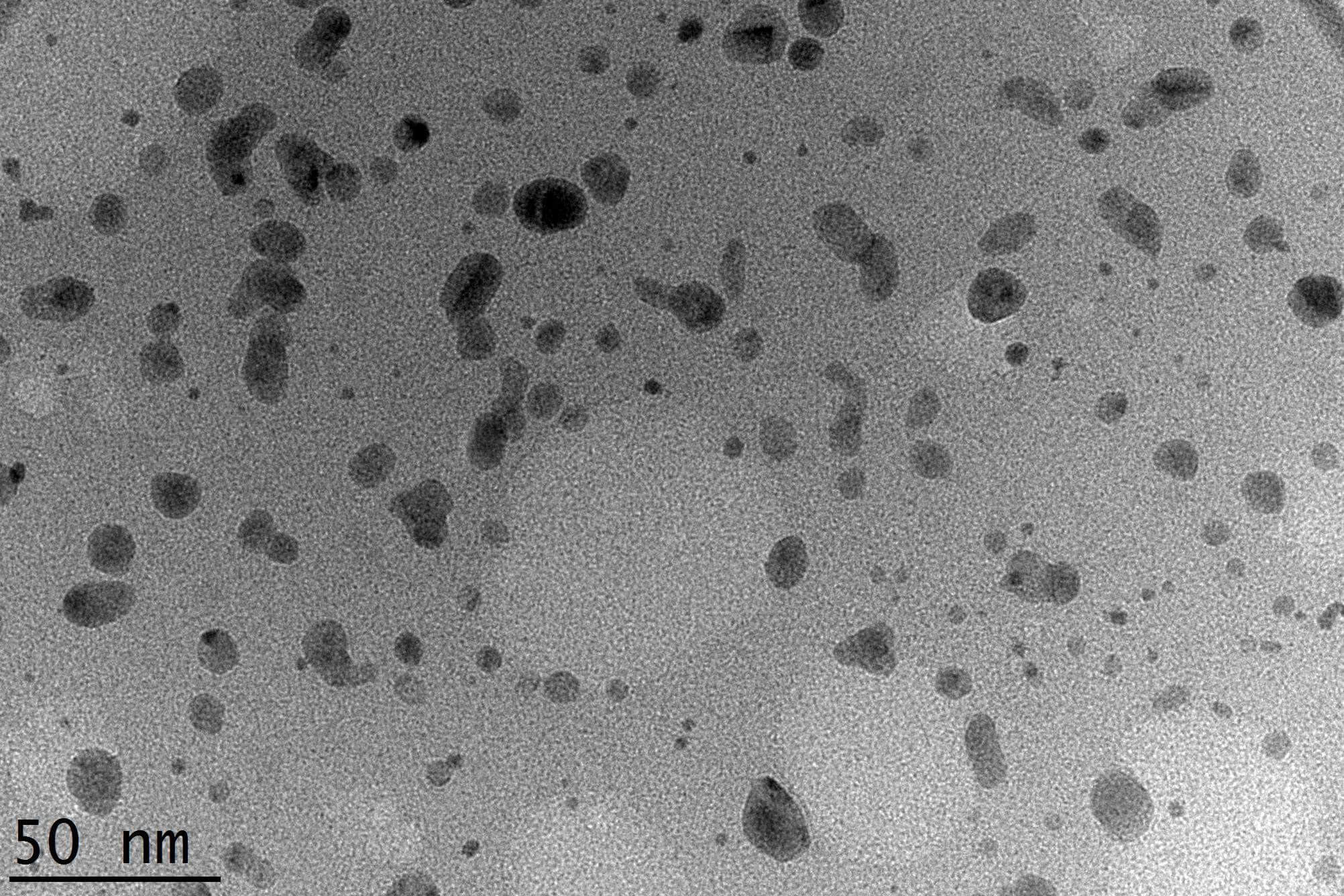 Fig. 2.
Fig. 2.
TEM image of WO3 nanoparticles. Scale bar = 50 nm.
The FESEM (Fig. 3) depicts the MTMS-WO3 coating surface with a characteristic of granular morphology. The WO3 nanoparticles were observed to be well-embedded within the MTMS binder. In some locations, WO3 nanoparticles were observed to form clusters and agglomerates.
 Fig. 3.
Fig. 3.
Field emission scanning electron microscopy (FESEM) of the MTMS-WO3 coating (MTMS-WO3-25%). Scale bar = 1.00 μm
Fourier-transform infrared (FTIR) spectroscopy was performed to investigate the chemical interactions between MTMS and the glass substrates during coating formation. To understand the reaction mechanisms, spectra were acquired for MTMS coating, WO3 nanoparticles, and the MTMS-WO3 coating (Fig. 4).
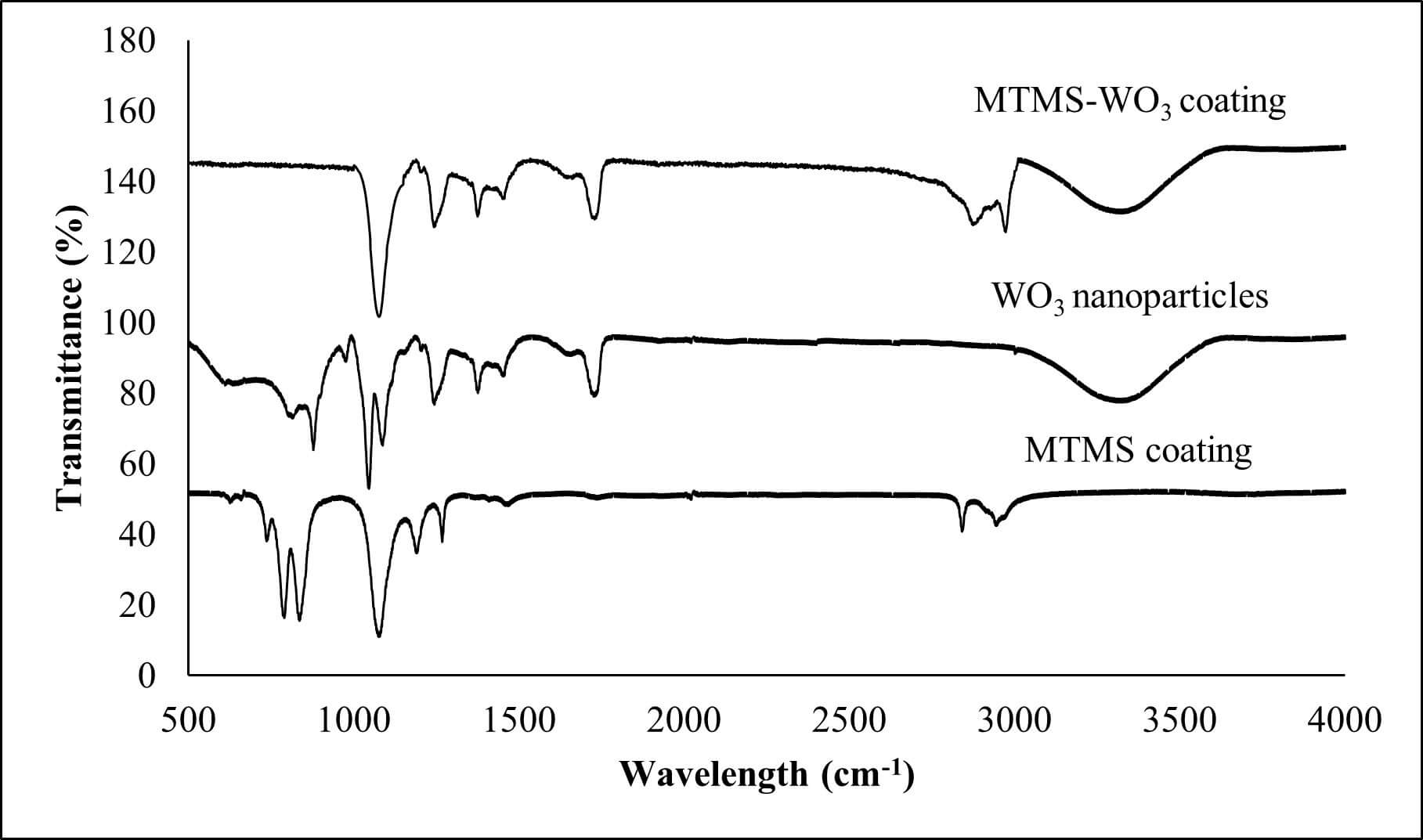 Fig. 4.
Fig. 4.
Fourier transform infrared (FTIR) spectra of the polymer coating.
The FTIR spectrum of MTMS exhibits characteristic peaks in the high-frequency region at 2945 cm-1 and 2841 cm-1 corresponding to the asymmetric and symmetric C-H stretching vibrations of the methyl (-CH3) and methoxy (-OCH₃) groups, respectively. In the fingerprint region (600–1500 cm-1), a distinct peak at 1268 cm-1 is attributed to the symmetric deformation (scissoring) of the Si-CH₃ bond. The presence of these non-polar methyl groups is significant, as their low surface energy typically increases the coating’s hydrophobicity. A dominant absorption band between 1000 and 1200 cm-1 represents the asymmetric stretching vibrations of the Si-O-Si siloxane network. This provides direct evidence of covalent bonding between the MTMS layer and the glass substrate. Furthermore, the absence of significant methoxy signals in the final coating suggests that the -OCH3 groups underwent complete hydrolysis. Supporting this, the peaks at 837 cm-1 and 790 cm-1 attributed to a combination of Si-O stretching and CH3 rocking have further confirmed the integration of methyl groups within the silicon-oxygen framework.
WO3 nanoparticles display a broad absorption band in the 3100–3500 cm-1 range, which is characteristic of surface hydroxyl (-OH) groups. This broad band was slightly reduced in the MTMS-WO3 coating spectrum, confirming that the MTMS precursors consumed some of the surface hydroxyls to facilitate chemical bonding. Additionally, the Si-O-Si stretching band (1000–1200 cm-1) exhibits a slight shift in the composite sample, indicating that the silicon network has successfully anchored to the heavier tungsten atoms.
The pencil scratch hardness results (Fig. 5) illustrate the mechanical resistance of the MTMS coating (MTMS-WO3-0%) compared to composite coatings with varying WO3 nanoparticle concentrations (6.25, 12.5, and 25 wt%). To evaluate hydrolytic stability, the samples were subjected to a 48 hours water-immersion test, after which their hardness was re-evaluated to determine the impact of moisture exposure on the coating integrity.
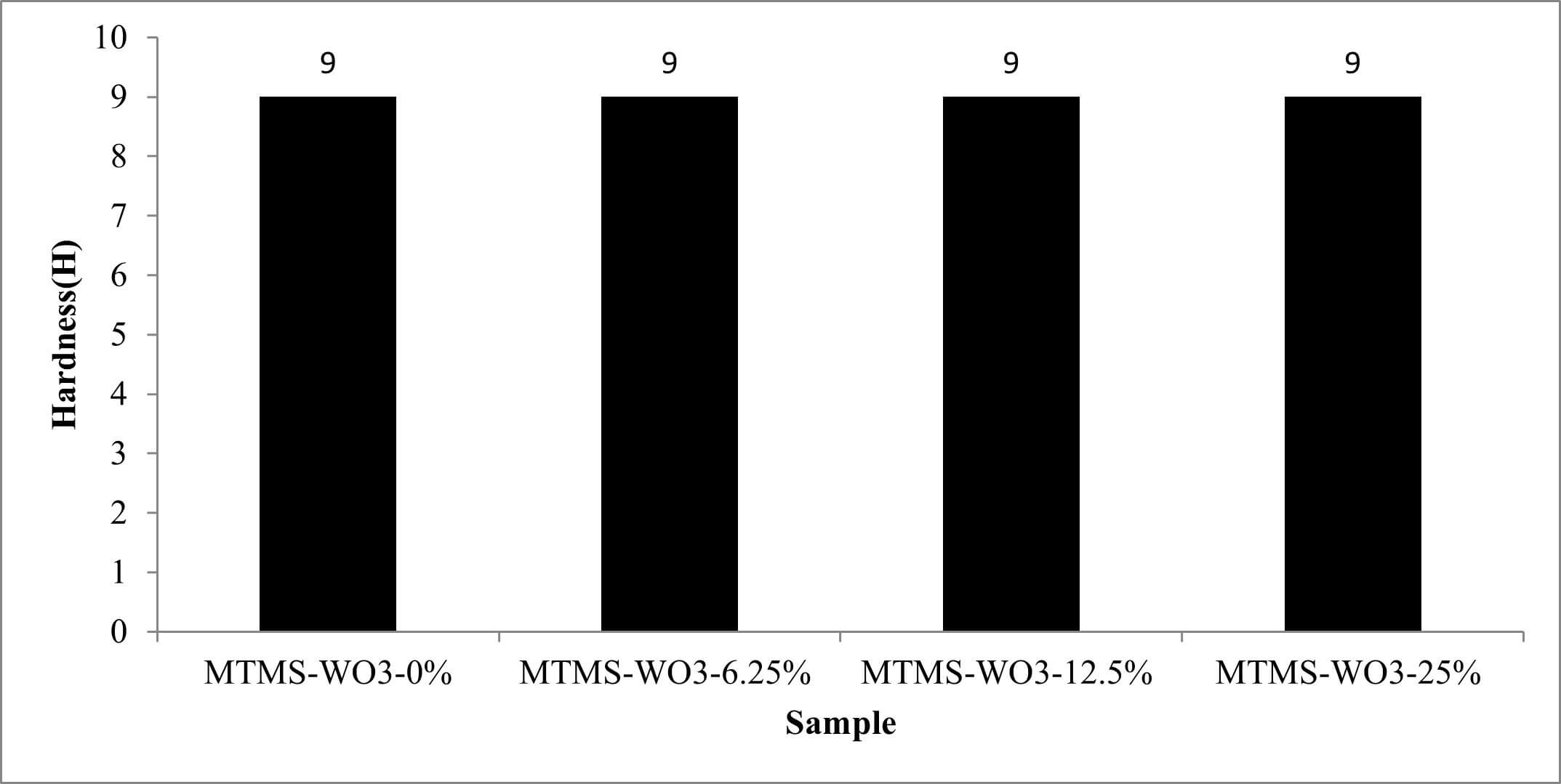 Fig. 5.
Fig. 5.
Pencil scratch hardness test for coatings with varying concentrations of WO3.
All coatings tested exhibited a consistent hardness value of 9H before and after the immersion test. This shows that the MTMS binder forms a very hard and durable film, indicating excellent cross-linking and film formation properties of the silane precursor under the applied curing conditions.
Increasing WO3 nanoparticle concentrations to 6.25, 12.5, and 25 wt% did not result in any observable decrease in hardness. This is an important finding as particulate fillers, especially at higher concentrations, can sometimes compromise the mechanical properties of a polymer matrix due to poor dispersion or agglomeration [14, 15]. The consistent hardness suggests that the WO3 nanoparticles are well-integrated into the MTMS matrix.
Tack-free time is an important evaluation parameter for curing kinetics of a coating. It defines the transition point at which the surface reaches a dry state. The shortest tack-free time was approximately 23 min observed for MTMS-WO3-25%. It was also observed (Fig. 6) that the tack-free time changes with respect to the concentration of WO3 used in the samples. Samples with a higher concentration of WO3 in the mixture tend to have shorter tack-free time and vice versa. The accelerated drying observed at higher WO3 concentrations is attributed to the increased solids content and the high specific surface area of the nanoparticles. The high surface area of the WO3 nanoparticles facilitates faster solvent evaporation.
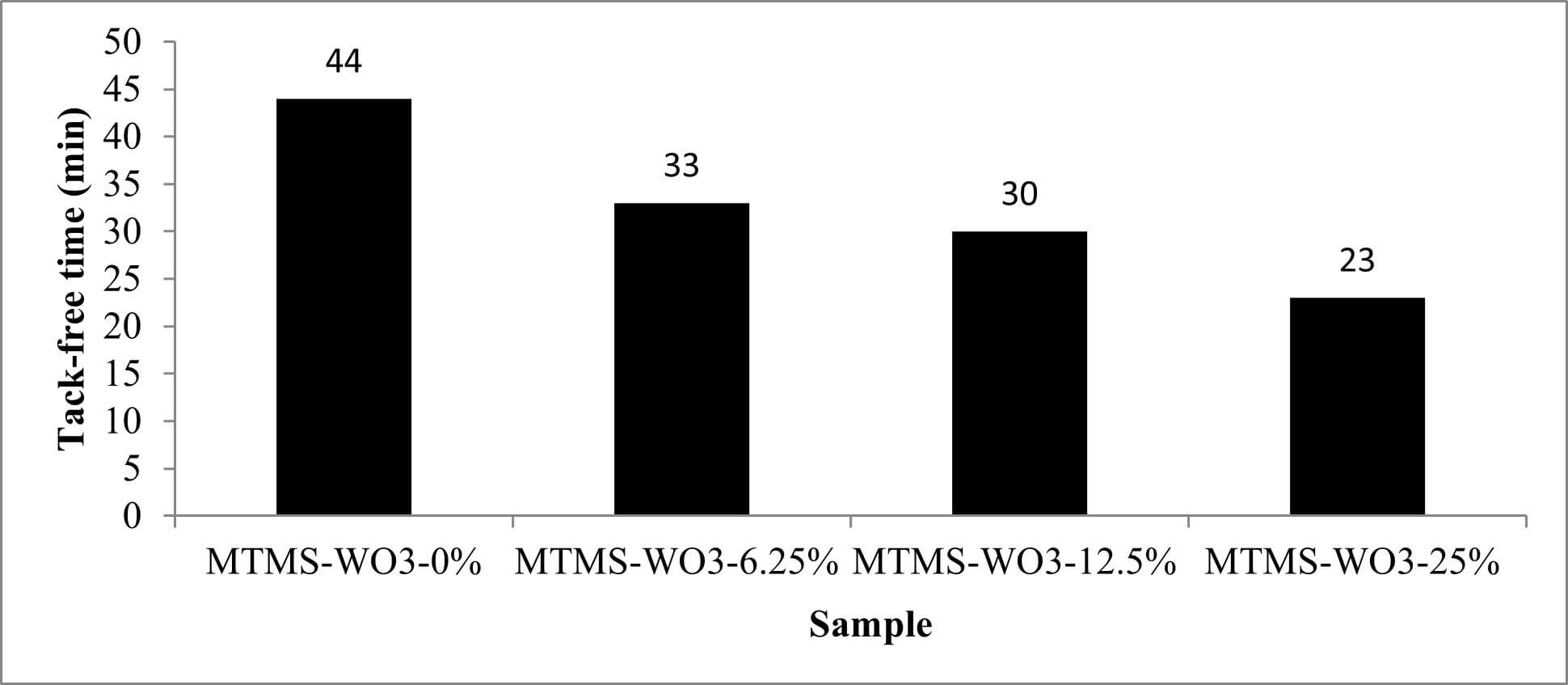 Fig. 6.
Fig. 6.
Tack-free time test for coatings with varying concentrations of WO3.
Fig. 7 illustrates the transmittance spectra of the various coatings. The uncoated glass substrate exhibits a high optical transparency, transmitting approximately 90% of incident light across the 400–900 nm range. In comparison, the MTMS coating (MTMS-WO3-0%) resulted in a slight reduction in transmittance while maintaining a value above 85%.
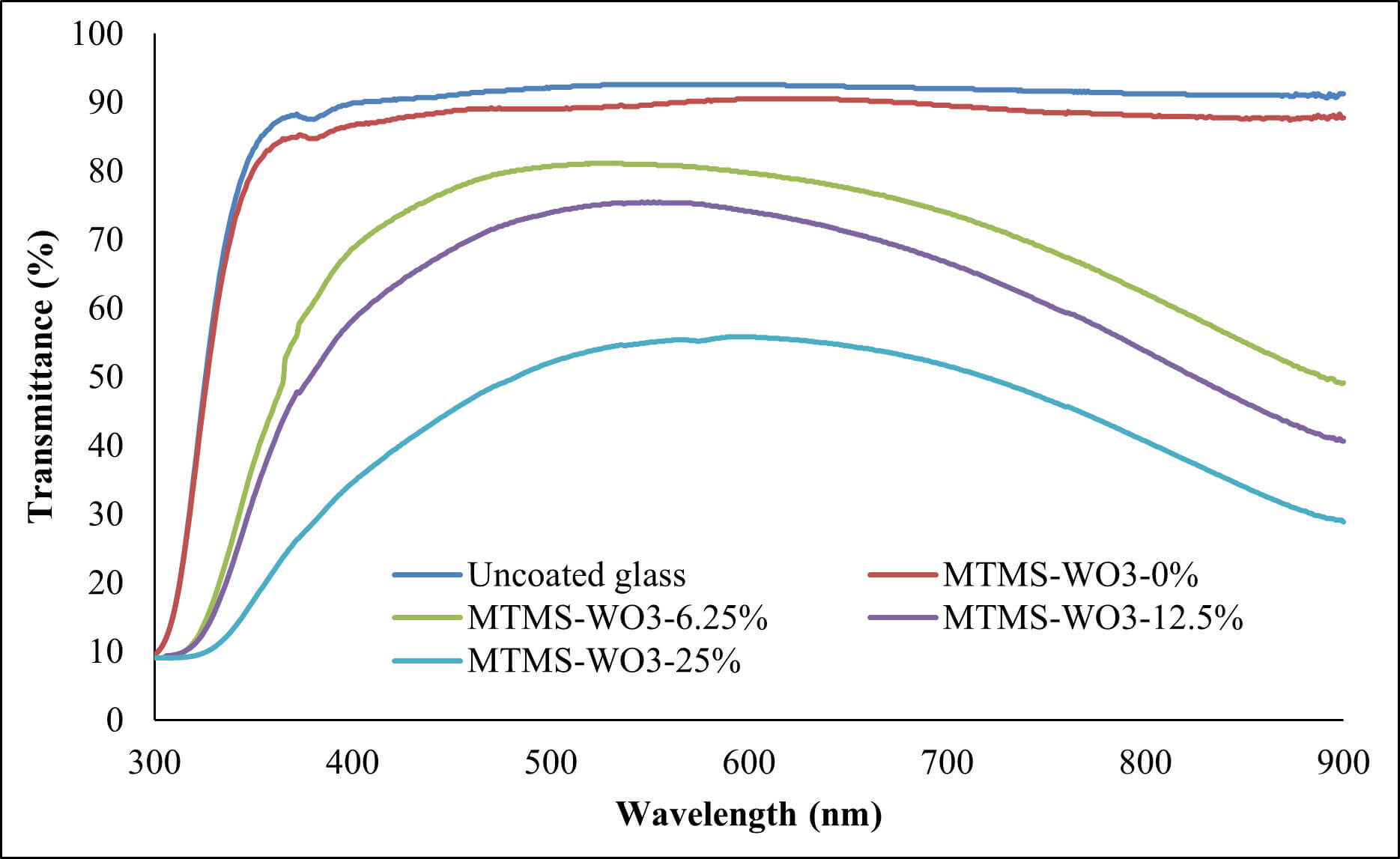 Fig. 7.
Fig. 7.
Transmittance spectra for uncoated glass slide and coated glass slides with varying concentrations.
The incorporation of WO3 nanoparticles significantly enhanced the ultraviolet (UV) reduction of the coatings. It was observed that the degree of UV reduction was proportional to the WO3 filler loading. The sample with the highest WO3 concentration exhibited the most substantial reduction in UV transmission. This trend of concentration-dependent transmittance continued into the visible light region (380–700 nm), where increasing the nanoparticle content led to a decrease in optical transparency.
The reduction in the degree of transmittance of the UV for coatings loaded with WO3 can be attributed to the ability of the WO3 to reflect and absorb UV light. According to absorbance and reflectance spectra (Figs. 8,9), the UV that did not pass through the coating was mostly absorbed by the WO3, with a small percentage being reflected. The same mechanism explains the reduced transmittance of visible light through the coatings.
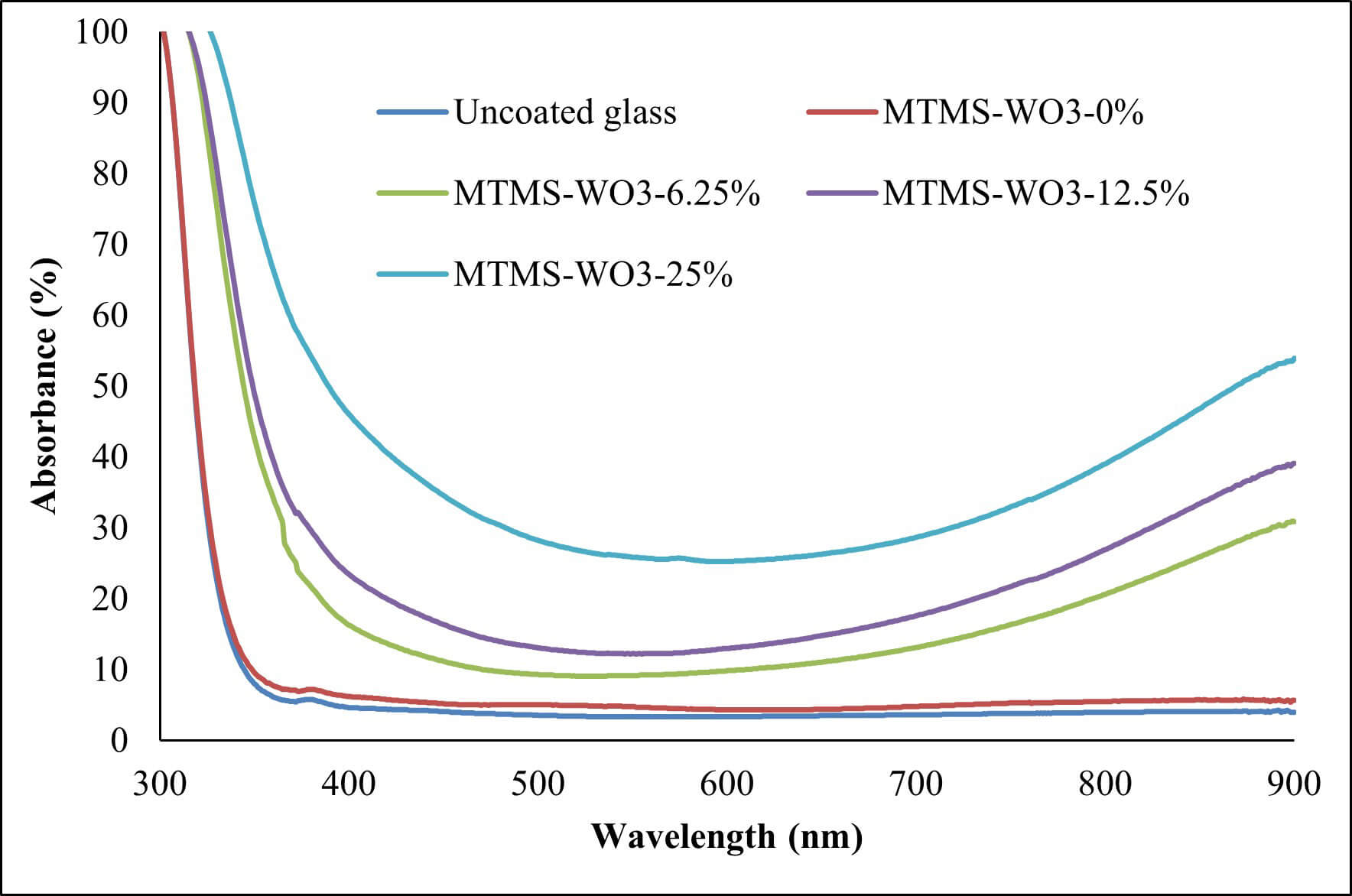 Fig. 8.
Fig. 8.
Absorbance spectra for uncoated glass slides and coated glass slides with varying concentrations.
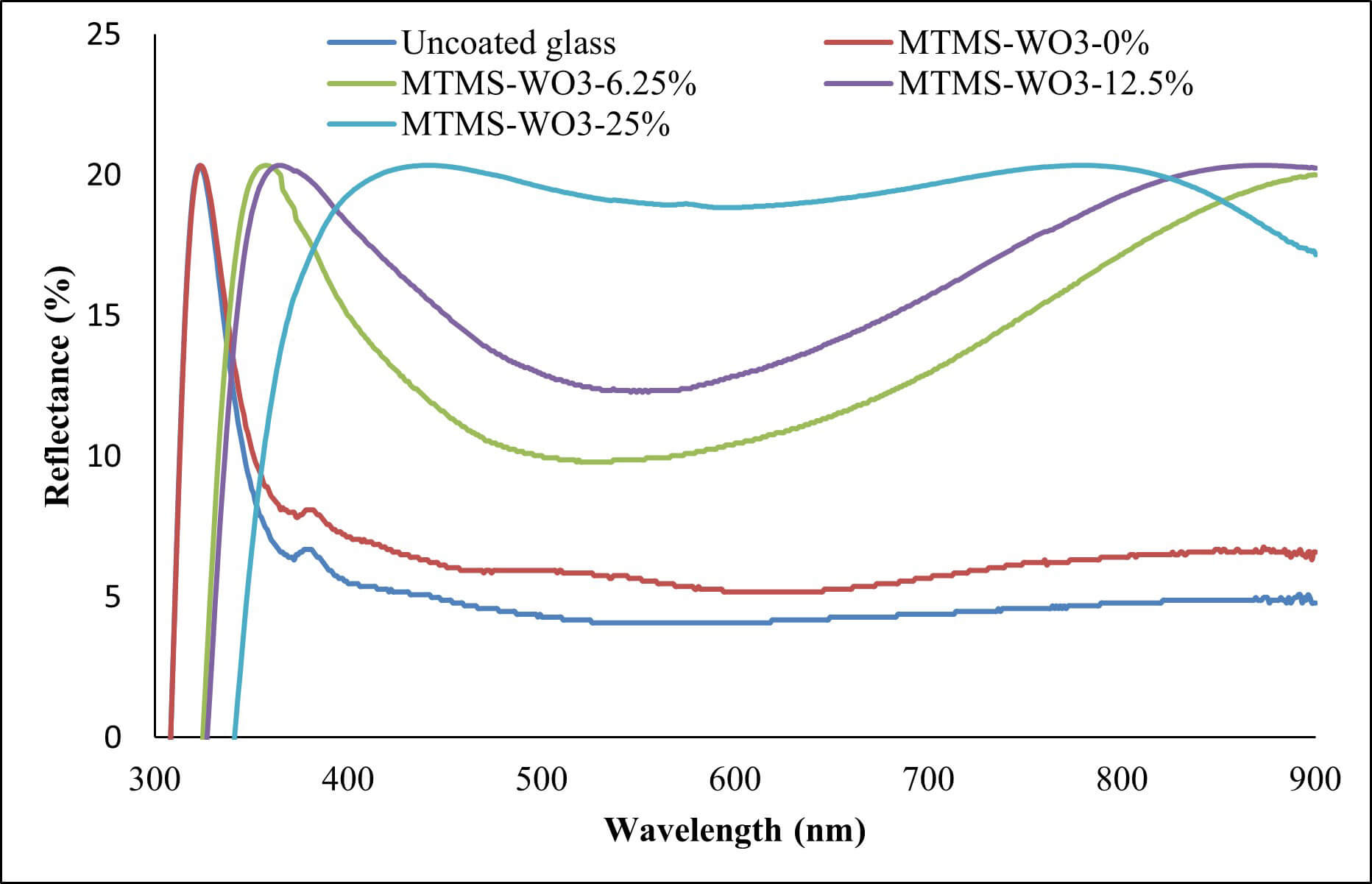 Fig. 9.
Fig. 9.
Reflectance spectra for uncoated glass slides and coated glass slides with varying concentrations.
Table 2 summarizes the UV-shielding performance and transparency of the developed coatings. The uncoated glass substrate exhibited UV-shielding of 33.22% while maintaining high transparency (91.88%). The MTMS coating (MTMS-WO3-0%) caused negligible optical variation, confirming the high quality of the silane binder. Increasing the WO3 concentration revealed a clear trade-off between shielding and transparency:
| Sample | UV-shielding (%) | Transparency (%TVis) |
| Uncoated glass | 33.22 | 91.88 |
| MTMS-WO3-0% | 35.49 | 89.26 |
| MTMS-WO3-6.25% | 63.23 | 76.32 |
| MTMS-WO3-12.5% | 68.30 | 69.25 |
| MTMS-WO3-25% | 81.08 | 50.44 |
UV, ultraviolet; TVis, visible transparency.
(a) At 6.25 wt%: the shielding efficiency nearly doubled (63.23%) compared to the MTMS-WO3-0% (35.49%) while the transparency remained high at 76.32%. This indicates that the nanoparticles were functioning effectively as UV absorbers without causing significant scattering in the visible range.
(b) At 12.5 wt%: the shielding slightly increased to 68.30%, but the transparency dropped to 69.25%. While effective, this formulation falls slightly below the 70% visible light transmission (VLT) standard often required for automotive and architectural windows.
(c) At 25 wt%: a shielding efficiency of 81.08% was achieved but this caused a reduction in clarity (TVis = 50.44%). The sharp drop in transparency suggests the onset of particle agglomeration and Mie scattering, rendering this concentration unsuitable for transparent applications.
Based on this, the 6.25 wt% formulation was considered as the optimal coating
as it resulted in the highest UV protection while strictly satisfying the
transparency criteria (TVis
Fig. 10 displays five glass substrates with an uncoated slide, MTMS coating (MTMS-WO3-0%), and glass slides coated with varying concentrations of 6.25, 12.5, and 25 wt% WO3 content, respectively. A distinct trend can be observed in which, as the concentration of WO3 increases, the transparency of the coating decreases.
 Fig. 10.
Fig. 10.
Images of (a) uncoated glass slides and glass slides coated with varying WO3 content (b) MTMS-WO3-0%, (c) MTMS-WO3-6.25%, (d) MTMS-WO3-12.5%, and (e) MTMS-WO3-25%.
While the MTMS coating (MTMS-WO3-0%) exhibits high transparency, the introduction of 6.25 wt% WO3 results in a noticeable greyish-blue hue and reduces transparency. This effect intensifies at 12.5 wt% WO3, where the coating appears darker and more opaque. At the maximum concentration of 25 wt%, the glass slide is significantly darkened, allowing very limited visible light transmission. A visual comparison of these samples is provided in Fig. 11.
 Fig. 11.
Fig. 11.
Visual comparison of (a) glass slides and coatings with varying WO3 content (b) MTMS-WO3-0%, (c) MTMS-WO3-6.25%, (d) MTMS-WO3-12.5%, and (e) MTMS-WO3-25%.
The observed decrease in transparency is directly related to the increasing
concentration of WO3 nanoparticles in the coatings. WO3 is known for
its strong absorption in the UV region due to its bandgap nature (Eg
The observed decrease in transparency is directly correlated with the increasing
concentration of WO3 nanoparticles within the coatings. This behavior is
attributed to the inherent optical properties of WO3, which is characterized
by strong UV absorption due to its bandgap (Eg
As a higher volume of WO3 nanoparticles is embedded within the coating matrix, the density of these absorbing and scattering centers increases, resulting in more effective attenuation of incident photons across the visible spectrum. This relationship demonstrates a practical approach for tuning visible light transmission by simply modulating the filler concentration.
Fig. 12 presents the water contact angle (
 Fig. 12.
Fig. 12.
Contact angle of water droplets on (a) glass slides and coatings with varying WO3 content (b) MTMS-WO3-0%, (c) MTMS-WO3-6.25%, (d) MTMS-WO3-12.5%, and (e) MTMS-WO3-25%.
The incorporation of 6.25, 12.5, and 25 wt% of WO3 (Fig. 12c–e)
significantly enhances the wettability of the surfaces, as evidenced by a sharp
reduction in
While the MTMS matrix provides hydrophobic methyl groups (-CH3), the addition of
WO3 introduces hydroxyl groups (W-OH) as WO3 is a
hydrophilic material [17]. The surface of the WO3 particles is rich in
hydroxyl (-OH) groups. These groups actively attract polar water molecules
through hydrogen bonding, causing the water droplet to spread out and hence
significantly decreasing the water contact angle (
The compatibility between WO3 nanoparticles and the MTMS matrix is the key to coating stability. Preservation of the monoclinic WO3 crystalline structure, confirmed by XRD, shows the synthesis was non-destructive and relied on physical dispersion. Nevertheless, the nanoparticle-matrix interface is chemically active. FTIR analysis reveals evidence of a condensation reaction. The hydrolyzed silanol groups (Si-OH) from the MTMS precursor reacted with surface hydroxyl groups (W-OH) on WO3 nanoparticles. This is shown by the reduced broad hydroxyl absorption band in the MTMS-WO3 spectra and the shift in Si-O-Si network vibrations. This bonding ensures nanoparticles are covalently attached to the siloxane network, not just physically trapped. Anchoring prevents leaching and improves structural strength and uniformity seen in FESEM analysis. This effective chemical anchoring is evident in the coating’s mechanical properties. Typically, high particulate fillers in sol-gel matrices cause brittleness or poor adhesion. In contrast, this study consistently found 9H hardness, regardless of WO3 concentration, even after water immersion. This suggests that the MTMS binder forms a highly cross-linked, dense siloxane network that encapsulates the nanoparticles. Faster tack-free time observed with higher WO3 loadings supports this. The nanoparticles’ high surface area likely provides sites for solvent evaporation and condensation, speeding up film formation without weakening cohesive strength.
The main function of the coating is to balance UV protection and visible
transparency. The UV-blocking mechanism functions through a combination of
physical scattering and the natural bandgap absorption of WO3 (Eg
At lower concentrations (6.25 wt%), absorption is the main factor. The high
transparency (
The wetting behavior of the coatings shows the importance of surface chemistry. Pure MTMS coatings are usually hydrophobic in nature due to their nonpolar methyl groups. However, adding WO3 makes the surface more hydrophilic. This change suggests that the surface energy comes from the exposed hydroxyl groups on the WO3 nanoparticles. Even with the covalent bonding of the silane, many W-OH groups remain active at the surface, as seen in the broad hydroxyl (-OH) band at 3100–3500 cm⁻1 in the FTIR spectra of the MTMS-WO3 coating. These groups engage in hydrogen bonding with water molecules. This adjustable wettability, which can be controlled by nanoparticle concentration, indicates potential uses for anti-fogging features alongside UV protection.
To compare the performance of the MTMS-WO3 coating, the optimal MTMS-WO3-6.25% concentration was compared with UV-shielding systems reported in the literature (Table 3, Ref. [18, 19, 20, 21]). Some systems, such as dual-absorber ethylene-vinyl acetate (EVA) films [18] or specialized sulfur-selenium polymers [19], achieve nearly total UV blocking, exceeding 95%. However, these systems often require complicated synthesis methods, expensive materials, or lower visible transparency. For example, pristine tungsten oxide/polyvinyl butyral (WO3/PVB) systems [20, 22] require higher loadings to exceed 90% shielding, which reduces visible transparency. In contrast, this study shows a better balance. The MTMS-WO3-6.25% coating achieves a UV-shielding efficiency of 63.23% while maintaining a high visible transparency of 76.32%. Nevertheless, the greyish-blue coloration of the WO3 nanoparticles may limit applications where a certain aesthetic is required.
| System/Matrix | Filler | Method | Transparency (TVis) | UV-Shielding | Remark | Ref. |
| EVA (Polymer) | UV326/UV531/PY14) | Laminating | 73.90% | 100% | Multiple filler | [18] |
| S-Se Polymer | - | Polymerization | 95.10% | High | Specialized synthesis | [19] |
| PVB (Interlayer) | WO3 | Casting | 60%–80% | Moderate surface hardness | [20] | |
| PMMA/Cellulose | WO3 | Casting | 88% | Hydrophobic surface property | [21] | |
| MTMS (Silane) | WO3 (6.25%) | Sponge | 76.32% | 63.23% | - Simpler single-filler system | This Work |
| - Uses standard, low-cost reagents | ||||||
| - Superior surface hardness (9H) | ||||||
| - Hydrophobic surface property | ||||||
| - Facile application & Durability |
EVA, ethylene vinyl acetate; PVB, polyvinyl butyral; PMMA, polymethyl methacrylate; PY, pigment yellow 14.
Additionally, unlike soft polymer matrices such as PVB, polymethyl methacrylate (PMMA), or EVA [18, 20, 21], the MTMS silane matrix used here offers superior surface hardness (9H) and chemical stability. The ability to achieve significant UV protection, greater than 60%, using a simple sponge application method instead of complex vacuum deposition or multi-step polymerization shows the potential of this coating for scalable as well as cost-effective retrofitting. This method prioritizes ease of application and mechanical durability over complete UV blockage.
Despite promising results, this study has several limitations. The manual sponge-coating technique is simple and scalable but lacks the precise thickness control of automated methods like spin-coating or dip-coating. Secondly, while WO3 nanoparticles successfully enhance UV-blocking, they make the surface more hydrophilic, hence preventing the achievement of a self-cleaning surface, which could potentially broaden the coating’s application. Lastly, haze at high concentrations (25 wt%) caused by particle agglomeration has put a limit to applications that demand strict color neutrality.
This study has successfully developed a transparent and effective UV-shielding coating by dispersing WO3 nanoparticles in a MTMS matrix. The characterization confirms that the sponge-application method produces a microscopically homogeneous film suitable for practical glass retrofitting.
The high loadings (25 wt%) caused excessive darkening (TVis
In terms of processing and surface characteristics, the inclusion of WO3 accelerated the curing process, achieving a tack-free time of 23 minutes at the highest loading, which is highly advantageous for large-scale processing. Additionally, the addition of WO3 has tuned the surface wettability from hydrophobic to hydrophilic, owing to the increase in surface hydroxyl groups. This demonstrates a potential application in anti-fogging applications.
This work also demonstrates that a simple and non-vacuum coating technique can produce high-performance optical filters comparable to those of complex commercial systems. The combination of 9H hardness, rapid curing, and balanced optical properties makes this MTMS-WO3 coating a promising solution for energy-efficient glazing.
To further optimize the surface functionality, future work will investigate the functionalization of WO3 nanoparticles with silane coupling agents. This surface modification aims to minimize the active hydroxyl groups, thereby reducing water spreading and enhancing hydrophobicity. Simultaneously, to address agglomeration at higher concentrations, optimization of the dispersion process through high-shear mixing or ultrasonication will be considered to increase the limit beyond 25 wt% without causing optical haze.
From a validation standpoint, weathering tests will also be considered to confirm the coating’s resistance to UV degradation and environmental pollutants in outdoor conditions. Finally, future studies will not only explore bio-based matrices but will also evaluate the potential reduction in volatile organic compounds (VOC) emissions compared to the current solvent-based silane system, thereby aligning the technology with the principles of green chemistry.
All data collected or analyzed during this study are included in this published article. If any additional information, underlying materials, or further clarification of the procedures is needed, it can be provided by the corresponding author upon reasonable request.
ZAA designed the study, conducted the experiments, analyzed the data, and wrote and revised the manuscript. The author read and approved the final manuscript. The author has participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The author would like to thank Universiti Malaya for providing access to the necessary facilities and technical support for this research. The author would also like to thank Dr. Wan Ahliah Wan Ismail for her technical and industrial guidance.
This research received no external funding.
The author declares no conflict of interest.
AI assistance (Gemini) was used solely to rephrase sentences for better clarity and flow. After using this tool, the author reviewed and edited the content as needed and took full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.












