1 School of the Environment and Safety Engineering, Jiangsu University, 212013 Zhenjiang, Jiangsu, China
2 School of Physics and Electronic Engineering, Jiangsu University, 212013 Zhenjiang, Jiangsu, China
Abstract
Photocatalytic hydrogen production represents a highly promising strategy for sustainable energy development. Among conventional photocatalysts, graphitic carbon nitride (g-C3N4) is distinguished by its non-toxicity and cost-effectiveness. However, its inherent limitations, particularly the high surface transfer barrier, result in poor charge-carrier separation. In this study, copper (Cu) was successfully incorporated into the C3N4 framework via a facile in-situ self-assembly method, significantly mitigating the interfacial resistance of the pristine material. The optimized Cu/C3N4 catalyst exhibited a remarkable hydrogen evolution rate of 1.5 mmol·g-1·h-1, which is 3.3-fold that of pure C3N4 (0.45 mmol·g-1·h-1). Mechanistic insights reveal that Cu doping not only facilitates electron transfer between triazine rings but also broadens the light absorption range and promotes carrier separation, thereby markedly augmenting the photocatalytic hydrogen evolution activity.
Keywords
- carbon nitride
- copper doping
- photocatalytic H2 evolution
- carrier separation
The growing global energy demand and massive use of fossil fuels have led to severe environmental issues, including an intensified greenhouse effect (primarily from CO2 emissions), acid rain (from NOx pollutants), and particulate matter (PM2.5) pollution [1, 2, 3, 4, 5]. Consequently, developing clean and renewable energy has become a global imperative [6, 7]. Among alternative energy carriers, hydrogen stands out for its high energy density (up to 142 MJ/kg, roughly triple that of gasoline), along with its zero-carbon emissions and recyclability [8, 9, 10, 11]. Solar-driven photocatalytic hydrogen production offers significant advantages over conventional methods. For instance, the water-gas shift reaction involves high energy consumption and a significant CO2 footprint, whereas water electrolysis is hampered by high energy consumption and high operational costs [12, 13, 14].
The development of efficient photocatalysts is central to advancing photocatalytic hydrogen production technology [15, 16]. Currently, the most extensively studied traditional photocatalysts comprise three primary material systems: titanium dioxide (TiO2), cadmium sulfide (CdS), and carbon nitride (C3N4), each of which possesses inherent limitations [17, 18, 19, 20, 21]. While TiO2 exhibits excellent photostability and is low-cost, its wide bandgap limits its light absorption, thereby severely limiting its practical efficiency [22, 23]. CdS, with a more suitable bandgap of 2.4 eV, responds to visible light but suffers from severe photocorrosion, leading to rapid catalyst deactivation [24, 25]. Conversely, graphitic C3N4 has emerged as a highly promising photocatalyst, owing to its unique two-dimensional layered architecture, sustainable metal-free composition, and robust thermal and chemical stability, which preserves its structural integrity across a wide pH range [26, 27, 28, 29]. As an organic semiconductor, C3N4 possesses a bandgap of 2.7 eV and suitably aligned band positions (a conduction band at approximately –1.1 eV and a valence band at approximately +1.6 eV vs. normal hydrogen electrode [NHE]) [30]. This enables effective visible-light absorption (up to ~460 nm) and provides sufficient thermodynamic driving force for water splitting. However, pristine C3N4 still faces two major challenges: low photocatalytic activity and severe recombination of photogenerated charge carriers (typically evidenced by a fluorescence lifetime of less than 5 ns) [31, 32, 33, 34, 35]. To overcome these bottlenecks, elemental doping has been established as an effective modification strategy. The introduction of appropriate dopant atoms can create specific energy states within the C3N4 band structure [36, 37, 38]. For example, Zhou et al. [39]. reported that individual copper ions form bonds with N-rings, yielding a stable Cu-doped porous graphene carbon nitride (PCN) catalyst. Simultaneously, the unique electronic structure of the Cu-N charge bridge enhances charge-carrier separation efficiency. In such doped systems, these newly introduced states act as effective electron traps, significantly suppressing the charge carrier recombination rate while simultaneously extending the material’s spectral response range.
In this study, Cu-doped C3N4 composite photocatalysts (denoted as
Cu/C3N4) were successfully synthesized via an in situ
template-assisted method. Specifically, using melamine as the precursor and a
copper salt as the dopant, the mixture was subjected to high-temperature
polycondensation under nitrogen (550 °C for 4 hours), yielding
Cu/C3N4 catalysts with a uniform doping structure. The photocatalytic
hydrogen production performance was systematically evaluated under an light emitting diode (LED) light
source (420 nm) to investigate the effects of varying Cu doping levels (1–10
wt%). When the Cu doping amount was 5%, the Cu/C3N4 catalyst (with a
hydrogen evolution reaction [HER] of 1.5
mmol
Melamine (Sinopharm Code: 30112528; Purity:
Solution A was prepared by dissolving melamine (0.5 g) in dimethyl sulfoxide (DMSO) (20 mL), followed by the addition of a specified amount of copper chloride and subsequent ultrasonic dispersion. Solution B was separately prepared by dissolving cyanuric acid (0.51 g) in DMSO (10 mL). Following the slow addition of Solution B to Solution A under continuous magnetic stirring, the resulting mixture was subjected to ultrasonication to ensure thorough blending. The solid precursor was collected, washed with ethanol, and dried overnight in a vacuum oven at 70 °C. The final Cu/C3N4 product was obtained by calcining the dried precursor at 550 °C for 4 hours under a continuous N2 flow. For comparison, the pristine C3N4 was prepared in the absence of Cu salt.
Into a three-neck flask containing 80 mL of an aqueous solution with 10% triethanolamine (TEOA), 10 mg of the catalyst was added. Then, 80 µL of chloroplatinic acid solution (10 mg/mL) was added, followed by bubbling argon (Ar) for 10 minutes to remove oxygen. Subsequently, under irradiation by an LED light source (420 nm) and using in situ photodeposited Pt (nominal 3 wt%) as a co-catalyst, the hydrogen evolution was monitored hourly using a gas chromatograph (GC9720Plus, Fulli Instrument, Taizhou, Zhejiang, China).
Raman spectra were detected on a Renishaw Invia Raman spectrometer (laser
wavelength 385 nm; Renishaw InVia, Wotton-under-Edge, Gloucestershire, UK). A scanning
electrochemical microscopy (SEM) test was performed on a JSM-7001F (Akishima,
Tokyo, Japan) field emission scanning electron microscope. Transmission electron
microscopy (TEM) images and energy dispersive X-ray spectroscopy (EDS) mapping
were obtained on a Thermo Fisher Scientific microscope (JEM-2100, JEOL, Akishima,
Tokyo, Japan). X-ray photoelectron spectra (XPS) were taken on a Thermo PHI
ESCA-5000C (distributed by Thermo Fisher Scientific Co., Ltd., Shanghai, China)
instrument using S2 Al K
As shown in Fig. 1, C3N4 precursors with a hollow sphere structure were prepared via molecular self-assembly using melamine (C3H6N6) and cyanuric acid (C3H3N3O3) as raw materials. Specifically, these precursors formed through hydrogen bonding in solution. During this process, a copper source was incorporated to achieve a homogeneous distribution of copper ions within the melamine-cyanuric acid supramolecular assembly. The resulting precursor was then calcined at high temperature under a nitrogen atmosphere, during which pyrolytic polycondensation of the organic components occurred. This process led to the escape of elements such as H and O in the form of H2O and CO2, ultimately yielding Cu-doped C3N4 materials.
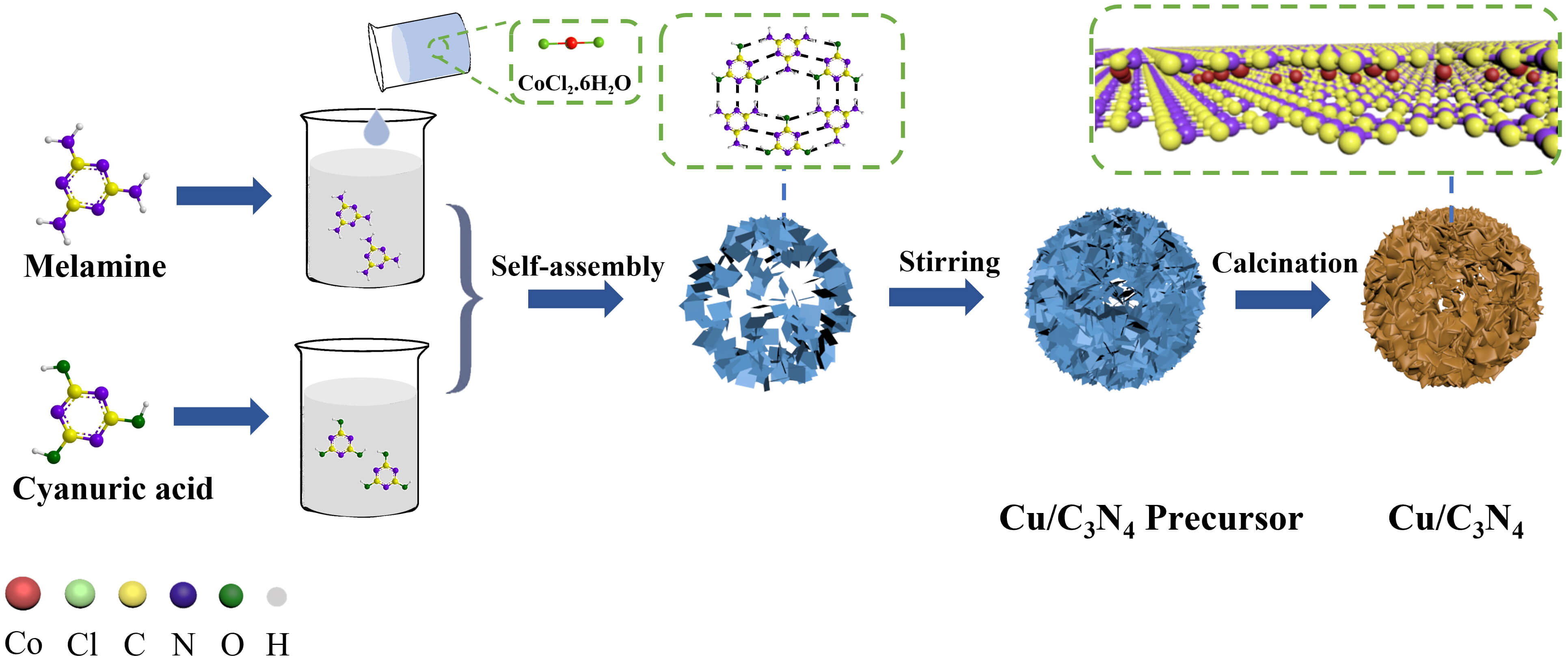 Fig. 1.
Fig. 1.
Schematic diagram of Cu/C3N4. Cu, copper; C3N4, carbon nitride. The image was created using Blender (Version 4.0, Amsterdam, Netherlands).
The Raman spectra of C3N4 and 5% Cu/C3N4 are shown in Fig. 2a. For 5% Cu/C3N4, a new peak appears near 470 cm-1 [40], which is consistent with the reported characteristic peak for Cu–N bonds, confirming the successful introduction of Cu into the C3N4 matrix. As shown in Fig. 2b, the prepared materials exhibit uniform spherical structures with diameters of 2–3 µm and relatively rough surfaces. The surface roughness likely originated from the decomposition of organic components and the release of gases during the high-temperature calcination. As shown in Fig. 2c, the material possesses a distinct hollow spherical structure with a shell thickness of approximately 100–200 nm and an inner cavity diameter of about 2 µm. The rough morphology observed on both the shell surface and the inner cavity wall is likely caused by gas evolution and the incorporation of Cu species during high-temperature calcination. Moreover, this hollow architecture exhibits a unique light-trapping effect; incident light undergoes multiple internal reflections and scattering within the cavity. This effectively extends the optical path length, thereby augmenting light-harvesting efficiency. These combined advantages enable the Cu/C3N4 hollow spheres to exhibit excellent performance in photocatalytic hydrogen evolution. Fig. 2d presents the high-resolution transmission electron microscopy (HRTEM) results for 5% Cu/C3N4. No distinct phases other than C3N4 are observed, indicating that Cu is incorporated into C3N4 in a doped form.
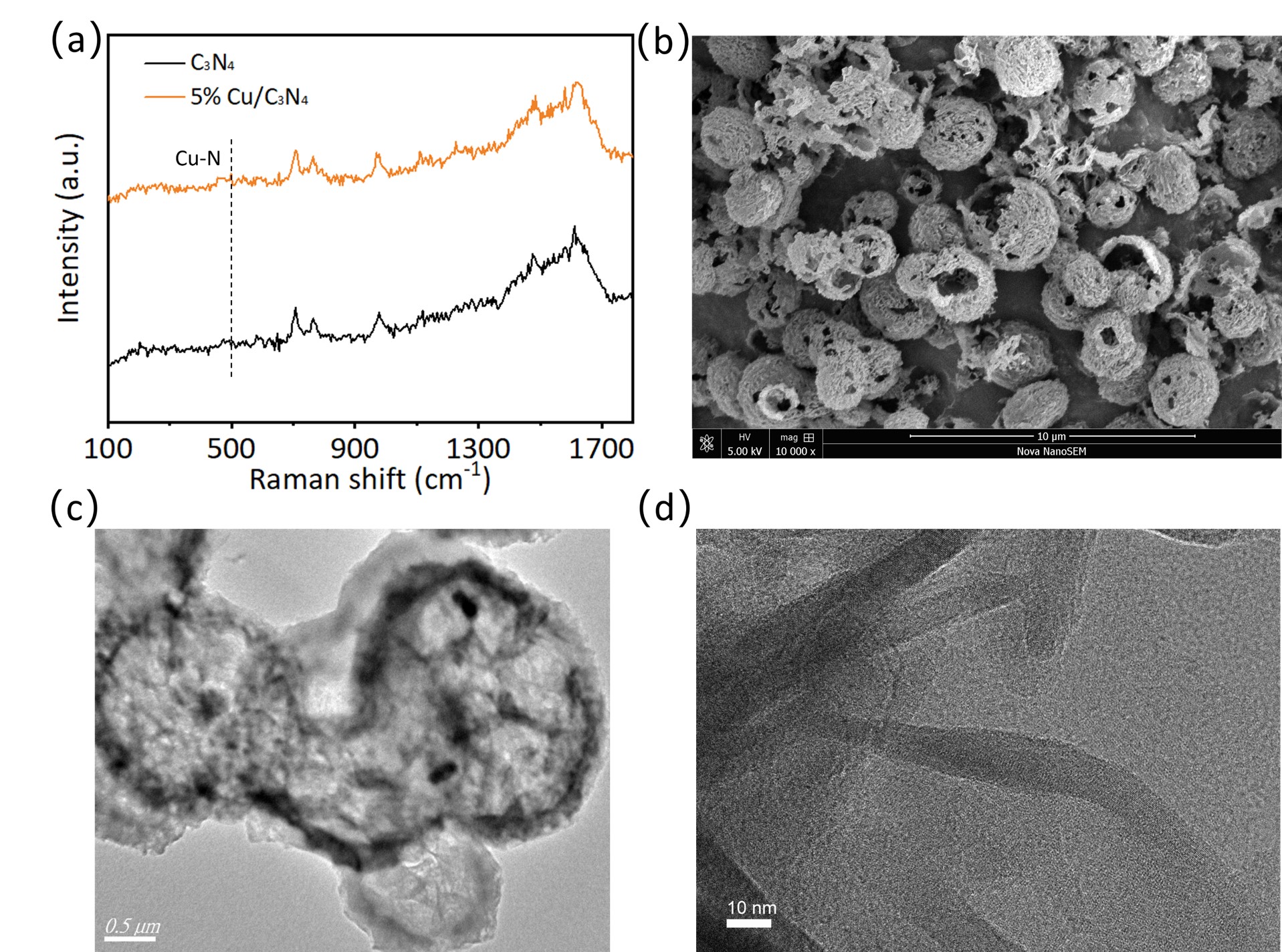 Fig. 2.
Fig. 2.
Structural and morphological characterization. (a) Raman spectra of C3N4 and 5% Cu/C3N4. (b) SEM image of 5% Cu/C3N4. (c) TEM image of 5% Cu/C3N4. (d) HRTEM of 5% Cu/C3N4. SEM, scanning electron microscope; TEM, transmission electron microscopy; HRTEM, high-resolution transmission electron microscopy. Fig. 2b scale bar = 10 µm; Fig. 2c scale bar = 0.5 µm; Fig. 2d scale bar = 10 nm.
TEM elemental mapping was performed [41] (Fig. 3). The corresponding maps for C, N, and Cu (Fig. 3b–d) reveal a homogeneous distribution of C and N, consistent with the C3N4 matrix. Crucially, the Cu map also shows a uniform signal, albeit at a lower intensity, confirming the incorporation of Cu species into the structure rather than the formation of large aggregates.
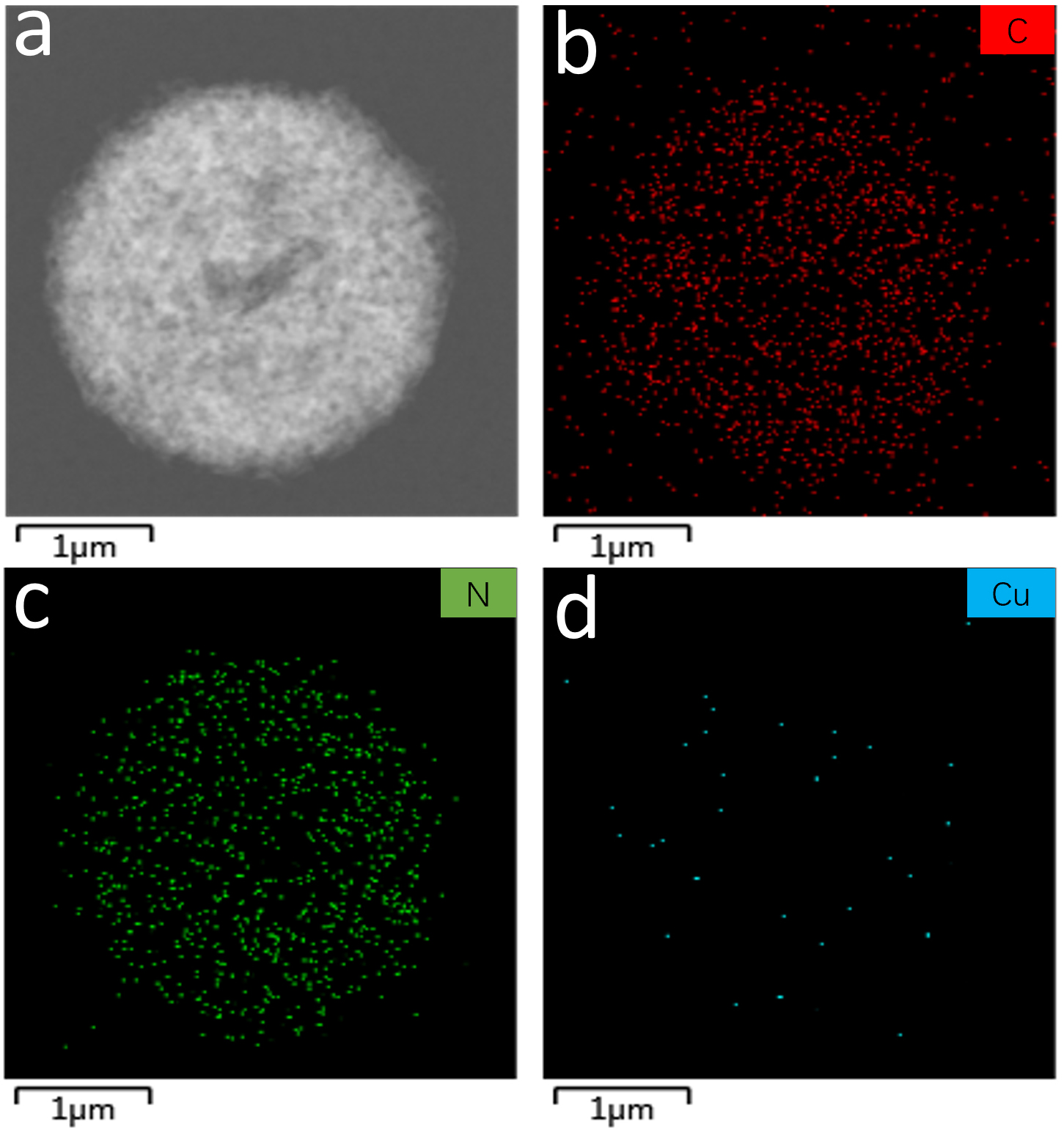 Fig. 3.
Fig. 3.
Elemental distribution analysis. (a) Cu/C3N4. (b–d) The mappings of carbon, nitrogen, and copper elements, respectively. Scale bar = 1 µm.
To investigate the chemical states of various elements in the synthesized Cu/C3N4 and C3N4, XPS analysis was conducted on the samples (Fig. 4). The high-resolution C 1s XPS spectra can be fitted with three peaks at 284.7 eV, 286.37 eV, and 288.16 eV, corresponding to C–C/C=C, C–NH2, and sp2-hybridized carbon in the N–C=N heterocyclic ring, respectively (Fig. 4a) [42]. The high-resolution N 1s XPS spectra can be deconvoluted into three peaks located at 398.69 eV, 400.22 eV, and 401.32 eV, corresponding to sp2-hybridized nitrogen in C–N=C, bridging nitrogen atoms in the tri-s-triazine units, and unreacted amino groups (C–NHx) at the periphery of the tris-triazine rings (Fig. 4b) [42]. Compared with pristine C3N4, the positions of the C 1s and N 1s peaks of Cu/C3N4 exhibit a measurable shift. In the high-resolution Cu 2p XPS spectrum (Fig. 4c), two distinct peaks can be clearly observed at 933.02 eV and 951.73 eV, corresponding to Cu2+, which confirms the successful introduction of Cu into C3N4. The XPS results demonstrate that Cu has been successfully incorporated into C3N4 and that an interaction between Cu and the C3N4 matrix has been established.
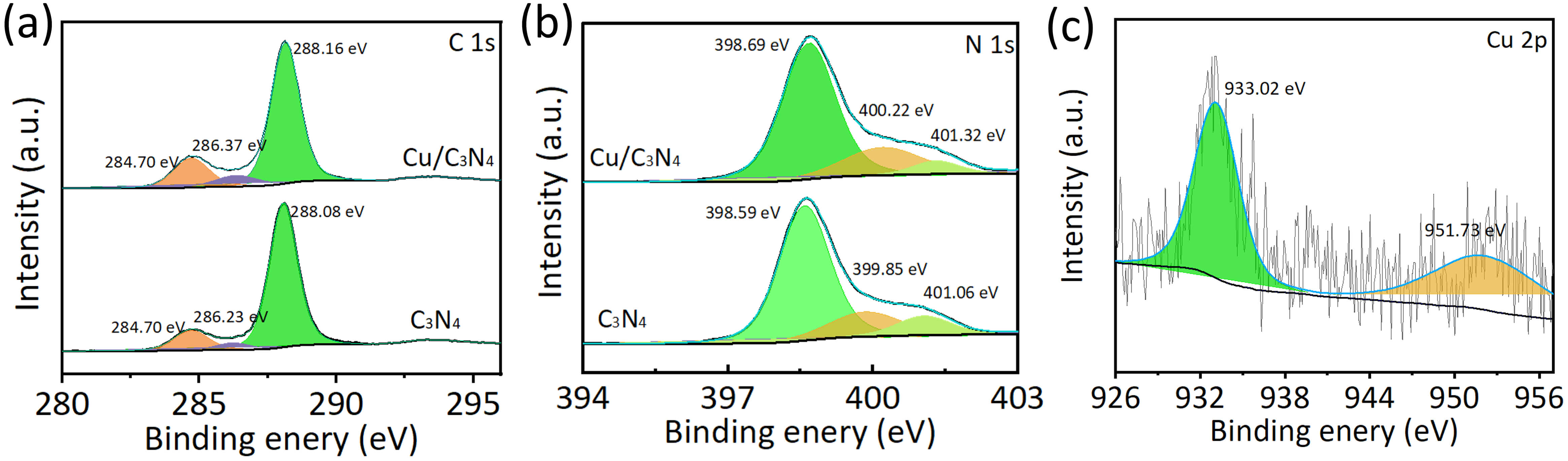 Fig. 4.
Fig. 4.
Chemical environment analysis. (a) C 1s, (b) N 1s XPS high-resolution spectra of Cu/C3N4 and C3N4 samples. (c) Cu 2p XPS high-resolution spectra of Cu/C3N4. XPS, X-ray photoelectron spectra.
The photocatalytic hydrogen evolution activities of pure C3N4 and Cu
doped C3N4 with different doping levels were evaluated using 3wt% Pt
as a co-catalyst (Fig. 5a,b). Under standard conditions, pure C3N4
exhibited a hydrogen evolution rate of approximately 0.45
mmol
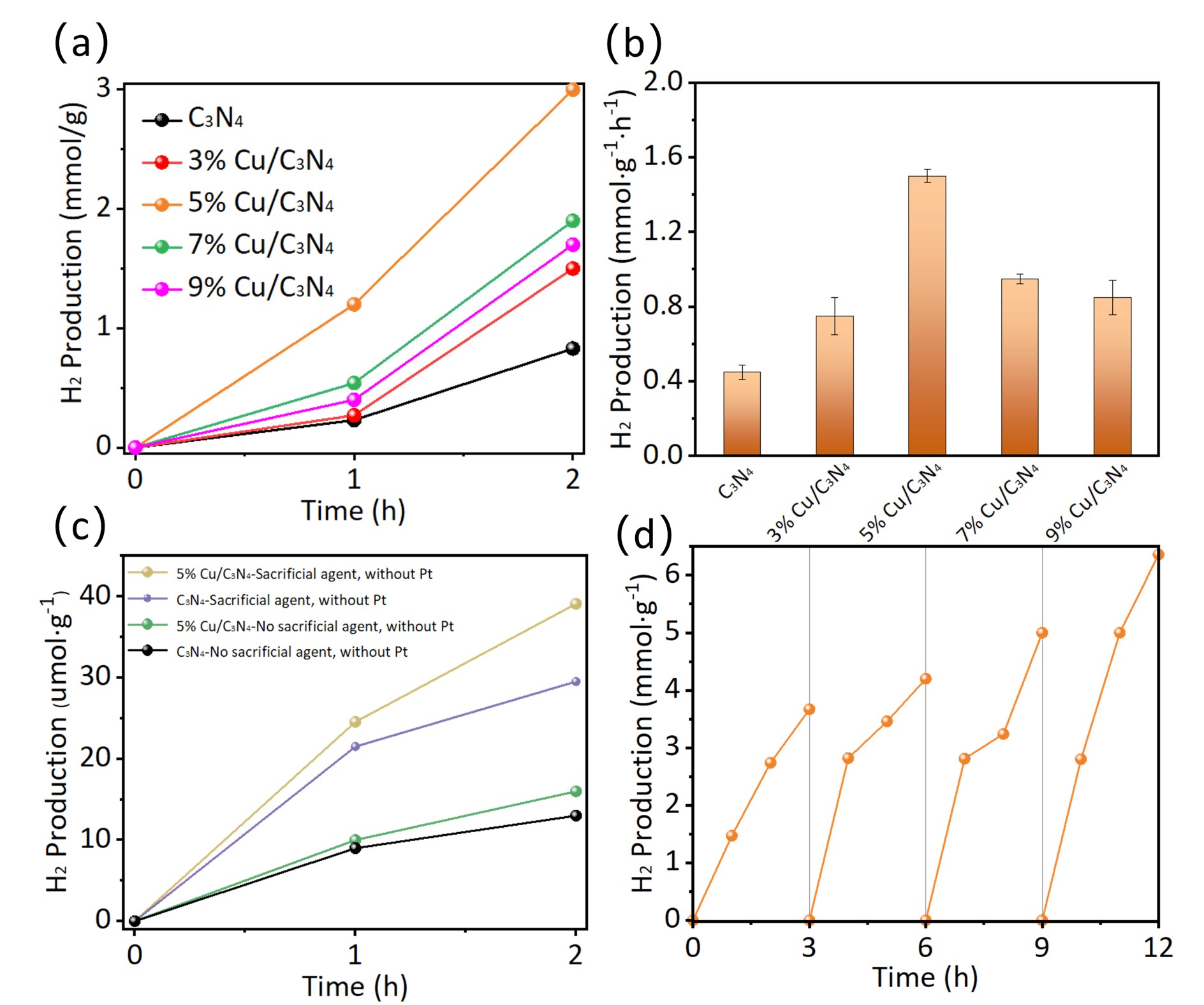 Fig. 5.
Fig. 5.
Hydrogen production performance analysis. (a) Photocatalytic hydrogen production performance of pure C3N4 and Cu/C3N4 with varying Cu contents (3%, 5%, 7%, 9%) under 2-hour illumination. (b) Corresponding hydrogen production rate diagram. (c) Photocatalytic hydrogen production performance of 5% Cu/C3N4 and C3N4 with and without the co-catalyst Pt and sacrificial agents. (d) H2 production recycling experiments of 5% Cu/C3N4.
| Photocatalyst | Light source | Sacrificial agent | Activity mmol |
Ref |
| Cu/C3N4 | LED light source (420 nm) | TEOA | 1.50 | This work |
| CN-NbO | LED light source | lactic acid | 0.75 | [43] |
| O-K-C3N4 | LED light source | TEOA | 0.68 | [44] |
| C60/graphene/g-C3N4 | LED light source | TEOA | 0.55 | [45] |
| 1T-MoS2@C3N4 | 300 W xenon lamp (AM-1.5) | TEOA | 0.57 | [46] |
| CN/MoS2-1 | 300 W xenon lamp ( |
TEOA | 0.45 | [47] |
| C3N4-SnO2-Pt | LED light source (450 nm) | TEOA | 1.06 | [48] |
| Quasi-Honeycombg-C3N4 | 300 W xenon lamp ( |
TEOA | 0.41 | [49] |
| Ni,B-CN | 1000 W xenon lamp | TEOA | 1.31 | [50] |
TEOA, triethanolamine; LED, light emitting diode.
UV-Vis DRS revealed the light absorption properties of the photocatalysts. The
introduction of Cu induced a distinct red shift in the absorption edge of
C3N4. The insets in Fig. 6a show the optical band gaps of
C3N4 and Cu/C3N4, which are 2.3 eV and 2.1 eV, respectively.
The PL spectra (Fig. 6b) were employed to investigate the separation of
photogenerated electrons and holes. It was observed that the introduction of Cu
into C3N4 led to a decrease in the PL spectral intensity, indicating
the effective separation of electron-hole pairs. To probe the effect of Cu doping
on C3N4 charge carrier transport, transient photocurrent response (i-t)
and EIS measurements were conducted. As shown in Fig. 6c, under periodic LED
light irradiation (
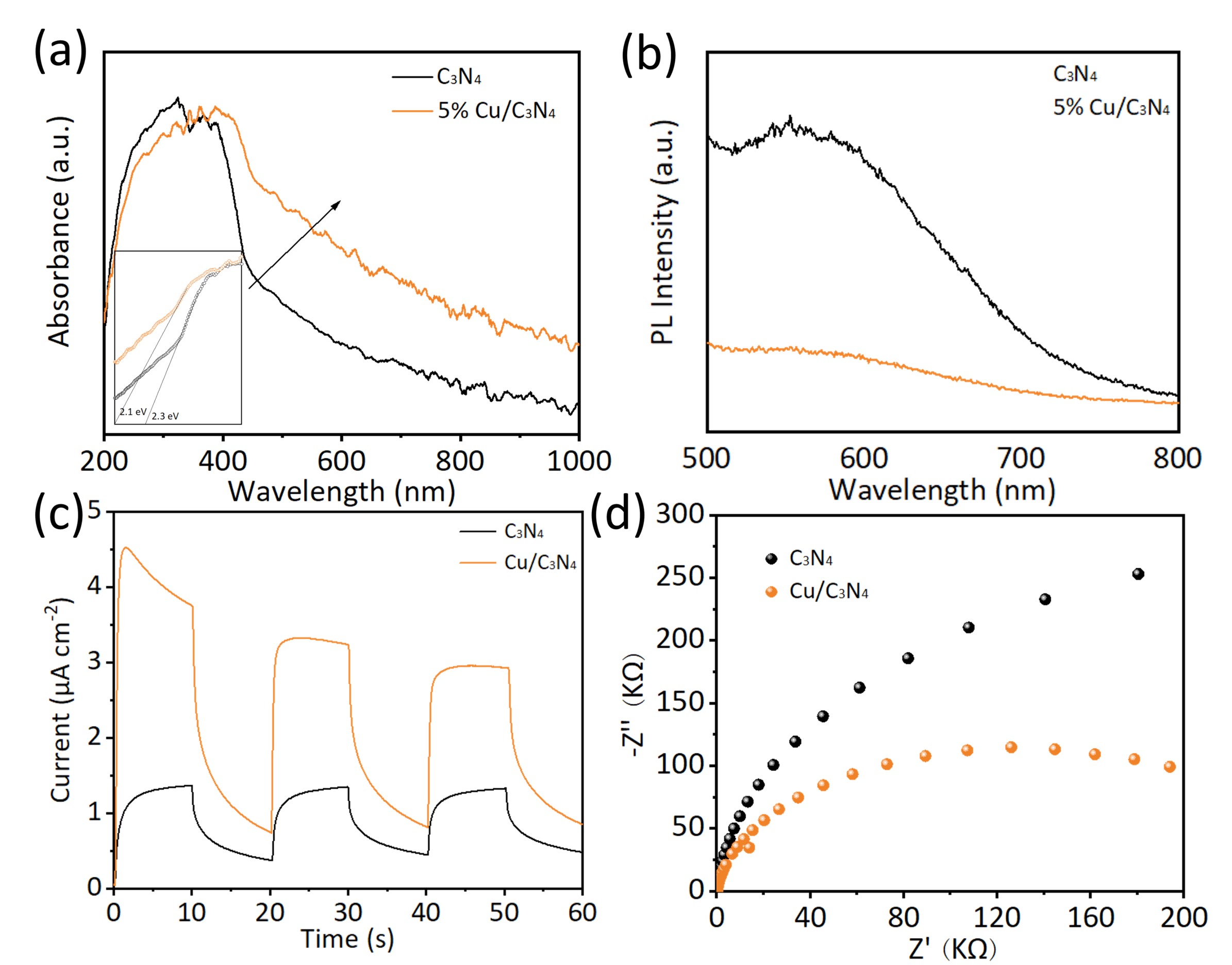 Fig. 6.
Fig. 6.
Light absorption and electrochemical analysis. (a) UV-vis absorption spectra of C3N4 and 5% Cu/C3N4. (b) PL spectra of C3N4 and 5% Cu/C3N4. (c) Photocurrent diagram of C3N4 and 5% Cu/C3N4. (d) Impedance diagram of C3N4 and 5% Cu/C3N4. PL, photoluminescence; UV-vis, ultraviolet- visible.
| Sample | C3N4 | 5% Cu/C3N4 |
| Rct (Ω) | 316,920 | 249,390 |
Based on the above results, a photocatalytic H2 evolution mechanism for the Cu/C3N4 composites is proposed (Fig. 7). The successful doping of Cu atoms introduces intermediate energy levels within the band gap of C3N4. These levels serve multiple critical functions: (1) they act as electron traps, effectively suppressing the recombination of photogenerated carriers; (2) they broaden the light absorption range; and (3) they provide additional pathways for charge transport. Upon light irradiation, photogenerated electrons rapidly transferred through these Cu-induced intermediate levels to the catalyst surface, where they reduce adsorbed protons (H+) to form H2. Simultaneously, the sacrificial agent TEOA efficiently scavenges the photogenerated holes in the valence band.
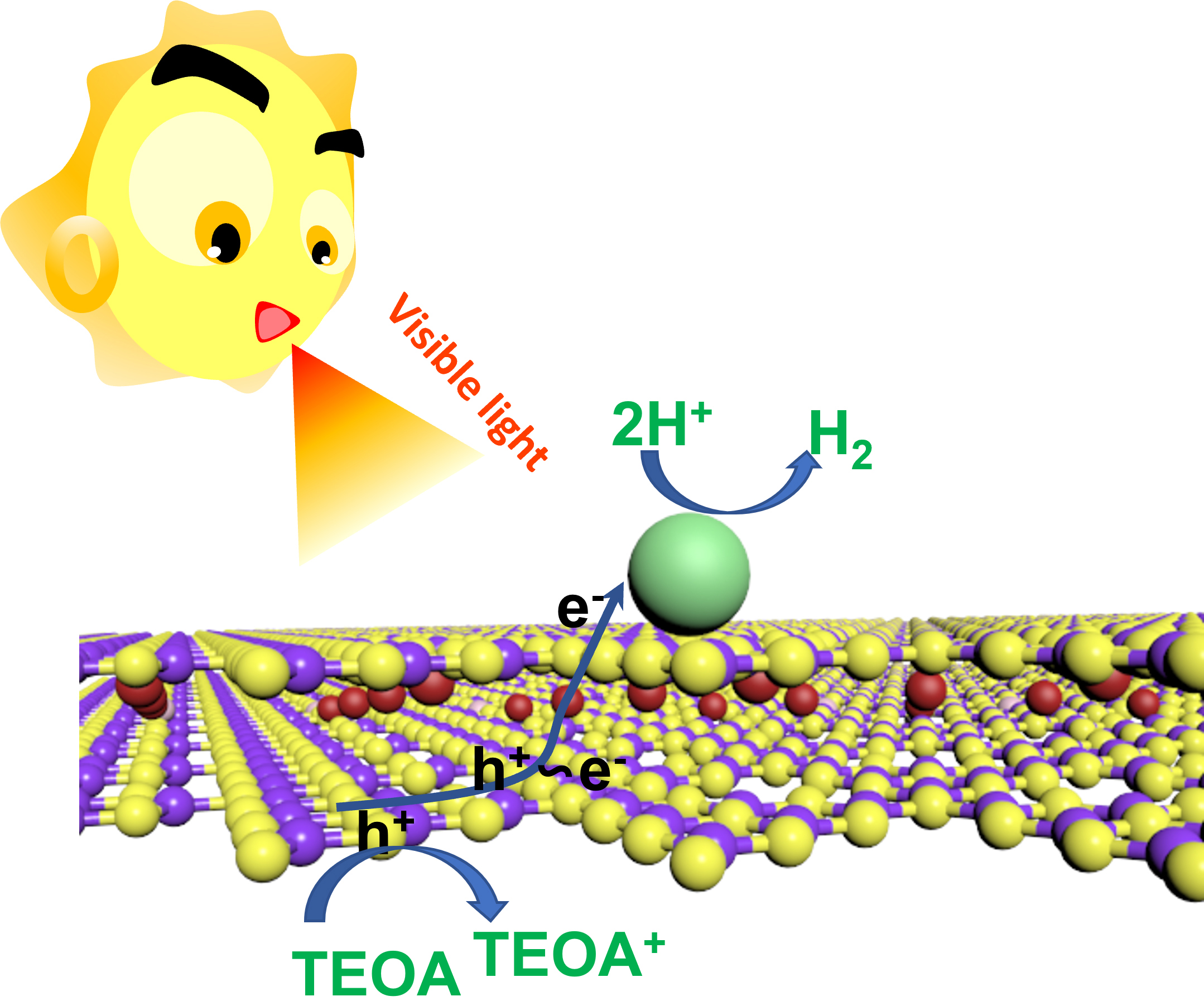 Fig. 7.
Fig. 7.
Mechanism diagram of Cu/C3N4 photocatalytic hydrogen production. The image was created using Blender (Version 4.0, Amsterdam, Netherlands).
While this study demonstrates the efficacy of Cu doping in enhancing the photocatalytic performance of C3N4, several limitations should be acknowledged.
Mechanistic Insight Depth: DRS and electrochemical tests strongly support the mechanism whereby copper doping enhances electron transfer between triazine rings and reduces carrier transport barriers. However, this study lacks more detailed spectroscopic and theoretical analysis at the microscopic level (e.g., density functional theory [DFT] calculations).
Scope of Reaction Conditions: The photocatalytic evaluation appears to be conducted under idealized laboratory conditions (e.g., using LED light). The performance under more challenging or realistic scenarios, such as under natural sunlight or in the presence of potential impurities, is unknown and represents a significant gap for practical implementation.
In summary, copper was successfully incorporated into the graphitic carbon
nitride (g-C3N4) lattice by introducing a copper precursor during the
supramolecular self-assembly stage. This approach promoted effective coordination
between Cu atoms and the molecular building blocks. The Cu/C3N4
composite was subsequently obtained through thermal condensation. The
introduction of Cu significantly enhanced the photocatalytic hydrogen evolution
activity. Under optimal doping conditions, the hydrogen evolution rate of the
Cu/C3N4 catalyst reached 1.5 mmol
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
WY designed the research study. CX and JL performed the research. YZ and SH participated in the data analysis and interpretation for the work. CX analyzed the data. CX and JL drafted the manuscript. CX managed the data curation. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors would like to express their sincere gratitude to Jiangsu University.
This study was supported by the Jiangsu Province College Students’ Innovation and Entrepreneurship Training Program (Project No. 202510299087z).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







