1 School of Chemistry and Chemical Engineering, Tianjin University of Technology, 300384 Tianjin, China
Abstract
In this article, two subphthalocyanines (SubPcs) derivatives are synthesized and characterized. By photophysical and electrochemical characterisation, the two materials exhibit good green light detection and charge transporting properties. Solution-processed organic photodetectors with a structure of indium tin oxide (ITO)/poly(3,4-ethylenedioxythiophene):polystyrene sulfonate (PEDOT:PSS)/SubPc:poly(9-vinylcarbazole) (PVK)/C60/LiF/Al were prepared. Under 520 nm illumination, the optimal device achieved promising performance metrics, including a specific detectivity (D*) of 1.65 × 1012 Jones and an external quantum efficiency (EQE) of up to 28.34%. This work demonstrates the potential of structurally engineered SubPcs for high-performance monochromatic green light detection.
Keywords
- subphthalocyanine derivative
- photodetector
- green light detection properties
Visible-light photodetectors with outstanding material properties have been shown to enable diverse applications in fields such as medicine, agriculture, environmental monitoring, flexible electronics, optical communications, information processing, and industrial inspection [1, 2, 3, 4, 5]. Driven by rapid advances in material science and device technology, their future application potential appears highly promising. While conventional photodetectors can offer broad spectral coverage from the near-ultraviolet through the visible to the near-infrared, this very breadth often compromises their ability to maintain high quantum efficiency and complicates the fabrication process [6]. In recent years, organic green-light photodetectors have progressed significantly. These devices can selectively absorb specific visible wavelengths, such as green light within the 500–570 nm range. Despite their high selectivity and sensitivity, however, the limited availability of high-performance monochromatic light-absorbing materials hinders their widespread practical adoption. Therefore, developing novel materials for monochromatic photodetection is crucial to overcoming material shortages and improving overall device performance.
Subphthalocyanines (SubPcs) are a class of green-light-absorbing materials.
Their framework comprises three isoindoline units linked by nitrogen atoms to
form a conical, ring-shaped structure. This configuration yields a conjugated
system containing 14
In 2022, Ma et al. [6] synthesised an axially nitro-substituted SubPc
derivative and used a solution-processed method to fabricate a green light
detector based on this compound. At zero bias voltage, the device achieved an
external quantum efficiency (EQE) of 31.91% and a detection efficiency
(D*) of 2.40
The ability of organic molecules to undergo intramolecular charge transfer (ICT)
is crucial for enhancing photoelectric conversion and device performance.
Materials with strong ICT capabilities often have donor-acceptor (D-A)
configurations [12]. For example, donor-substituted tridurylboranes and
1,3,4-oxadiazole derivatives exhibit strong ICT behaviour, resulting in high
quantum efficiency [13, 14]. Drawing inspiration from these designs, our goal is
to develop novel subphthalocyanine materials featuring extended
In this article, we detail the synthesis and comprehensive characterization of two novel nitro/halogen-substituted SubPc derivatives. We elucidated their structure-property relationships, including intramolecular charge transfer and energy levels. Finally, we fabricated and evaluated solution-processed green light photodetectors based on these materials.
The axial ligand (X) in SubPc is determined by the boron trihalide (BX3)
used in the templated cyclotrimerization reaction with 4-nitrobenzonitrile.
Chlorine (Cl) and bromine (Br) were selected to study the influence of halogen
electronegativity on the material properties. The synthetic pathways for
SubPc a and b are shown in Fig. 1. Weigh 2.22 g (12.6 mmol) of
4-nitrobenzonitrile and transfer it to a three-necked, round-bottomed flask. Add
30 mL of o-dichlorobenzene as the reaction solvent, followed by 5 mL of
1M BCl3 toluene solution. Conduct the entire reaction in a nitrogen
atmosphere. Stir and heat to 180 °C, maintaining the reaction for 5
hours. After the reaction has finished, remove the solvent and any excess boron
trichloride by vacuum distillation. Separate the product using silica gel column
chromatography to obtain the red solid SubPc a with a yield of 63.21%. 1H
nuclear magnetic resonance (1H NMR) (400 MHz, CDCl3):
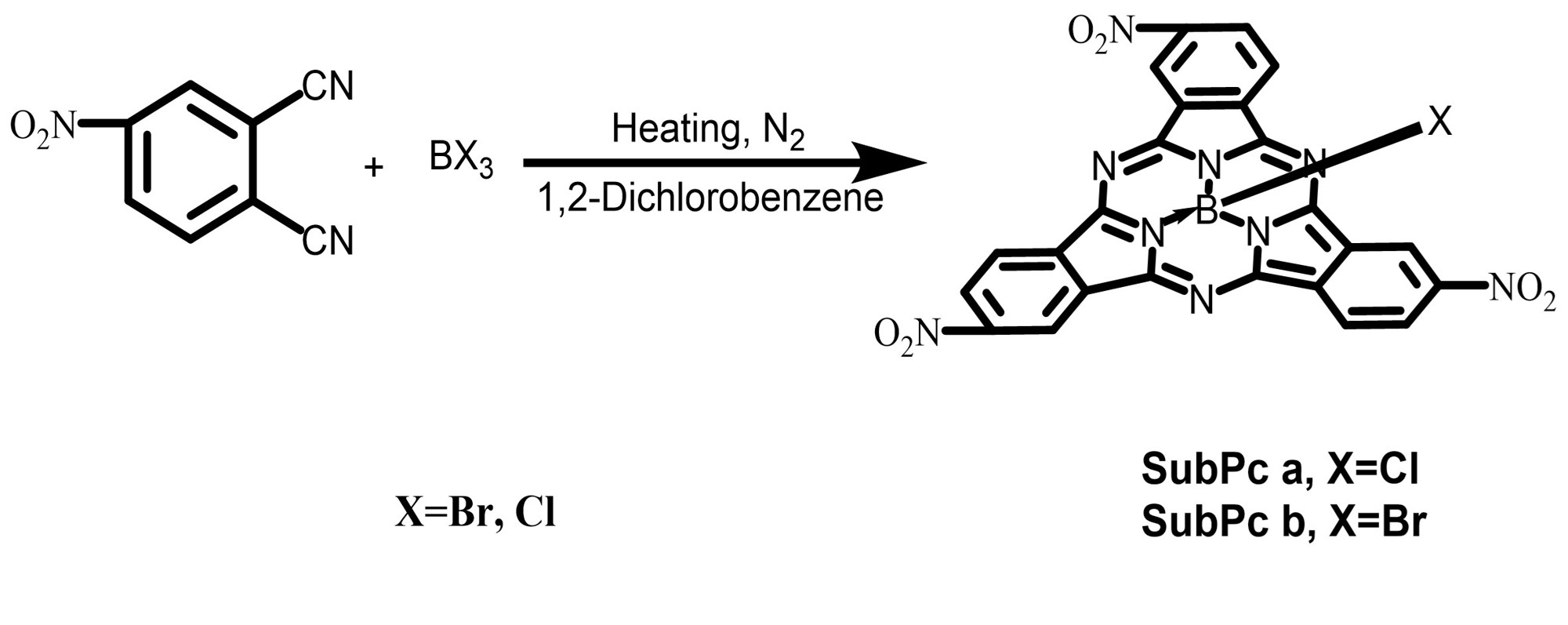 Fig. 1.
Fig. 1.
The synthetic route of the SubPc a and b. SubPc, subphthalocyanine.
The synthesis method for SubPc b is identical to that for SubPc a. The reagents
used were 4-nitrobenzonitrile (2.00 g, 11.5 mmol) and boron tribromide (0.49 mL,
57.5 mmol). The yield is 53.46%. 1H NMR (400 MHz, CDCl3):
4-nitrobenzonitrile, boron trichloride and boron tribromide were obtained from Ascender Chemical Technology Company (Shanghai, China). o-Dichlorobenzene was purchased from Tokyo Chemical Industry (Shanghai, China) Chemicals Company (Shanghai, China). The Fourier-transform infrared (FT-IR) spectra were collected in the range 4000–400 cm-1 on a Nicolet Fourier spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA) using KBr pellets. The mass spectra were measured with a matrix-assisted laser desorption/ionization time-of-flight/time-of-flight (MALDI-TOF/TOF Mass spectrometer) (Bruker, Karlsruhe, Baden-Württemberg, Germany). The NMR spectra were recorded using a Bruker Ultrashield Spectrometer (Bruker, Karlsruhe, Baden-Württemberg, Germany).
The indium tin oxide (ITO) glass first underwent sequential ultrasonic cleaning
to remove surface organic matter and particles, followed by ultraviolet (UV)-ozone treatment
for 20 minutes to enhance the hydrophilicity of the ITO surface. A 40 nm thick
layer of poly(3,4-ethylenedioxythiophene) (PEDOT) was spin-coated onto the
ITO glass at 2000 rpm for 30 seconds, followed by heat treatment at 100
°C for 20 minutes to remove residual solvents. The thickness of the
active layer was controlled by adjusting the concentration of the solution and
the spin speed, and was consistently measured to be 40
SubPc a or b was blended with PVK at various weight ratios (1:2, 1:4, 1:6). A preliminary device screening indicated that the 1:4 ratio yielded the optimal balance between film quality and photocurrent response, and was thus selected for all devices reported herein.
PEDOT: PSS (Baytron P VP Al 4083) was purchased from Heraeus (Hanau, Hessen, Germany). PVK was purchased from Lumtec Technology Corp. (Hsinchu, Taiwan). C60 was purchased from Sigma-Aldrich (St. Louis, MO, USA). All other reagents, solvents, and chemicals were purchased from commercial sources and used without further purification unless otherwise specified.
The test instrument was a Hitachi 3310 ultraviolet-visible
(UV–Vis) spectrophotometer (Hitachi
Company, Tokyo, Japan), and the samples were dissolved in five solvents to form 1
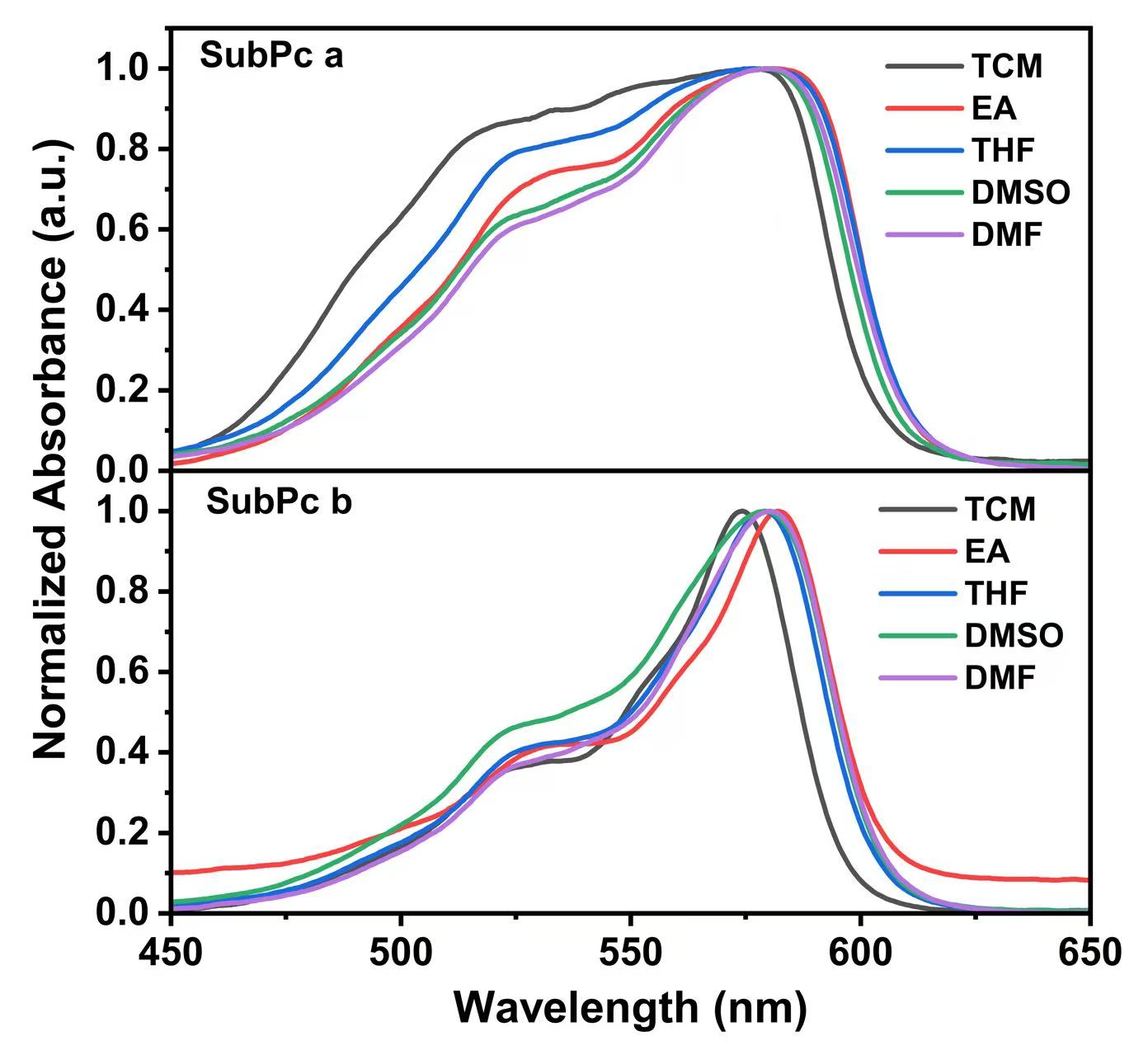 Fig. 2.
Fig. 2.
Absorption spectra of SubPc solutions: (1) TCM; (2) EA; (3) THF; (4) DMSO; (5) DMF. TCM, trichloromethane; EA, ethyl acetate; THF, tetrahydrofuran; DMSO, dimethyl sulfoxide; DMF, N, N-Dimethylformamide.
| Solvents | νa (cm-1) | νf (cm-1) | Δν (cm-1) | f ( | ||
| TCM | 579.0 | 17,241 | 605 | 16,528 | 713 | 0.15 |
| EA | 580.0 | 17,241 | 603 | 16,583 | 658 | 0.20 |
| THF | 580.5 | 17,226 | 610 | 16,393 | 833 | 0.21 |
| DMSO | 581.0 | 17,211 | 615 | 16,260 | 951 | 0.26 |
| DMF | 582.5 | 17,167 | 617 | 16,207 | 960 | 0.27 |
| Solvents | νa (cm-1) | νf (cm-1) | Δν (cm-1) | f ( | ||
| TCM | 574.0 | 17,421 | 599 | 16,694 | 713 | 0.15 |
| EA | 579.0 | 17,271 | 601 | 16,638 | 638 | 0.20 |
| THF | 578.5 | 17,286 | 613 | 16,313 | 833 | 0.21 |
| DMSO | 580.0 | 17,241 | 605 | 16,528 | 951 | 0.26 |
| DMF | 581.0 | 17,211 | 608 | 16,447 | 773 | 0.27 |
Here,
As shown in Fig. 2 and Tables 1,2, the absorption spectra of both SubPc a and SubPc b are centered in the 570.0–590.0 nm range. When measured in the same solvent, the absorption maxima of SubPc a and SubPc b are similar. Upon changing the solvent from TCM to DMF, the absorption maximum of SubPc a undergoes a slight redshift, whereas that of SubPc b exhibits a pronounced redshift.
The test instrument was a Hitachi F-4500 Fluorescence Spectrophotometer (Hitachi Company, Tokyo, Japan), and the results are shown in Fig. 3.
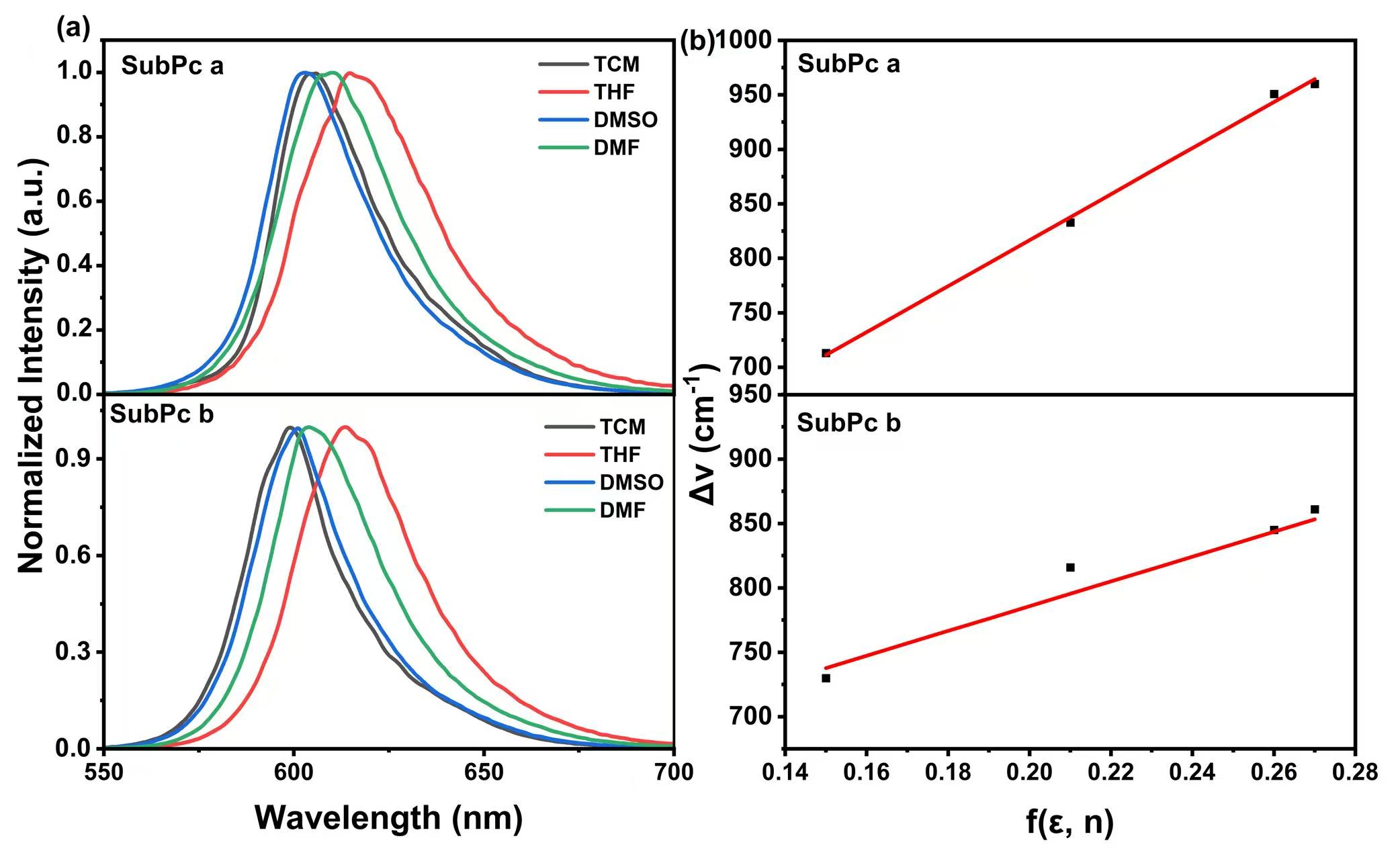 Fig. 3.
Fig. 3.
Fluorescence spectra and Lippert-Mataga plots of SubPc a and SubPc b in various solvents.
(a) Fluorescence spectra of SubPc solutions: (1)
TCM; (2) THF; (3) DMSO; (4) DMF. (b) Lippert-Mataga diagram of solvent polarity
f (
As shown in Fig. 3a and Tables 1,2, the maximum fluorescence emission peak for each compound exhibits a pronounced redshift as the solvent is changed from TCM to DMF. This trend is attributed to enhanced solvent polarity promoting intramolecular charge transfer (ICT), which reduces the energy gap between the excited state and the ground state. This results in the fluorescence emission wavelength shifting to longer wavelengths (red shift).
To estimate the intramolecular charge-transfer capability closely related to
device performance, the dipole moment difference (
In Eqns. 1,2,3,
As analyzed in Fig. 3b, the linear slope of SubPc a is larger than that of SubPc
b. According to the calculation results of Eqns. 1,2,3, the
The service life and environmental stability of organic materials are strongly correlated with their thermal stability. A TG1093F1 thermogravimetric analyzer (NETZSCH Company, Selb, Bavaria, Germany) is employed to analyze the thermal stability of SubPcs. The testing instrument is carried out under nitrogen, and the rate of temperature increase is 10 °C/min. The test results are shown in the figure below.
As shown in Fig. 4, both SubPc a and SubPc b exhibit good thermal stability
below 100 °C, with a mass loss of less than 10%. A marked decrease in
mass, corresponding to thermal decomposition, occurs at approximately 233
°C and 246 °C (
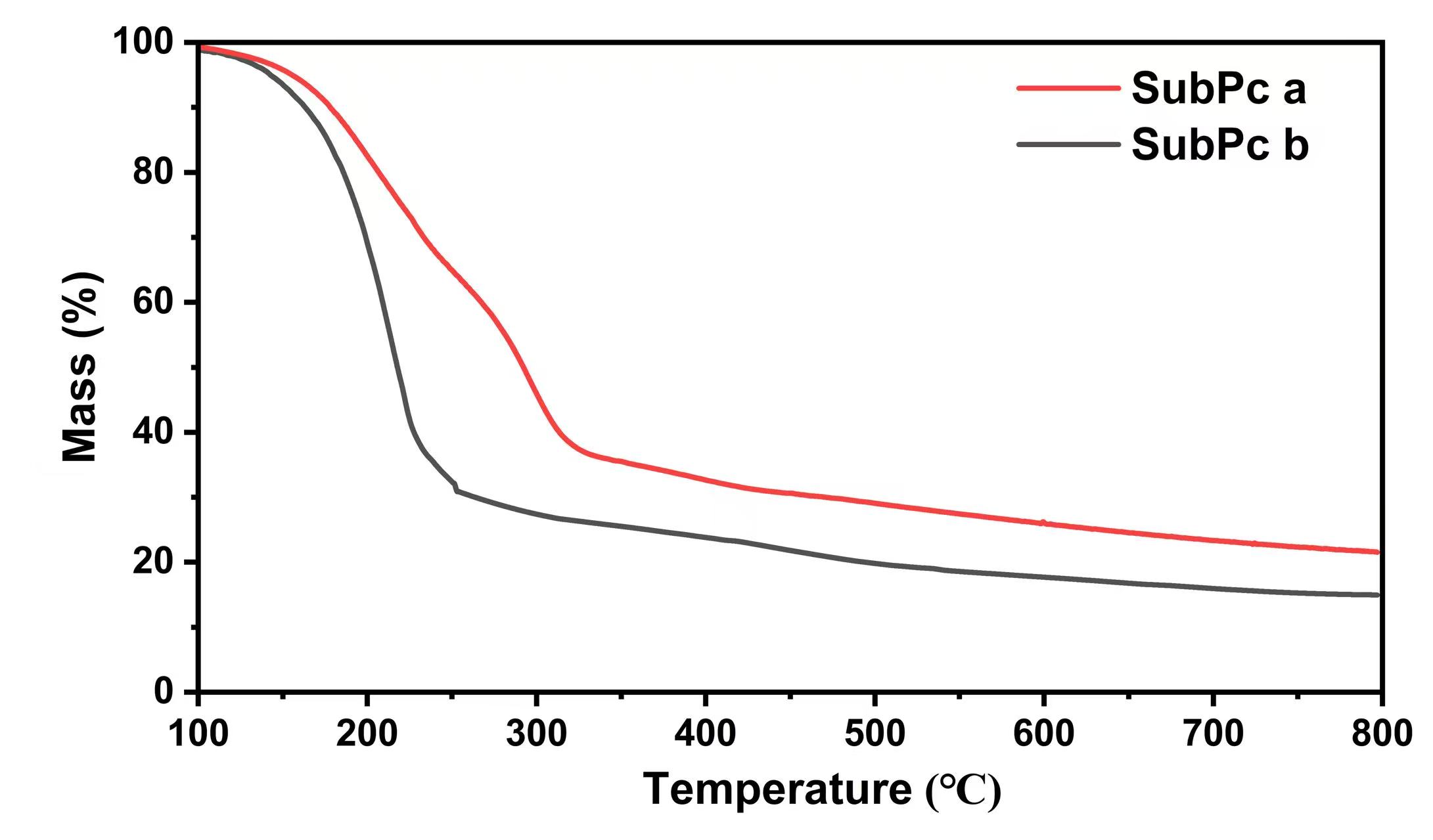 Fig. 4.
Fig. 4.
TGA curves of SubPc a and b. TGA, thermogravimetric analysis.
Cyclic voltammetry (CV) curves are a commonly used research method for electrochemical testing to determine the electrochemical activity of compounds as well as to determine their lowest unoccupied molecular orbital (LUMO) and highest occupied molecular orbital (HOMO) positions [17]. In this paper, a CHI640E electrochemical workstation (Shanghai, China) was used with three electrodes, including a reference electrode, a working electrode, and a counter electrode.
Fig. 5 shows the cyclic voltammetric curves, from which the energy levels of
SubPc materials could be obtained using Eqns. 4,5,6 [18, 19]. EOX denotes the
starting oxidation potential. Eg values can be obtained from the absorption
spectra. Table 3 lists the calculated results. Multiple scanning tests show
deviations of
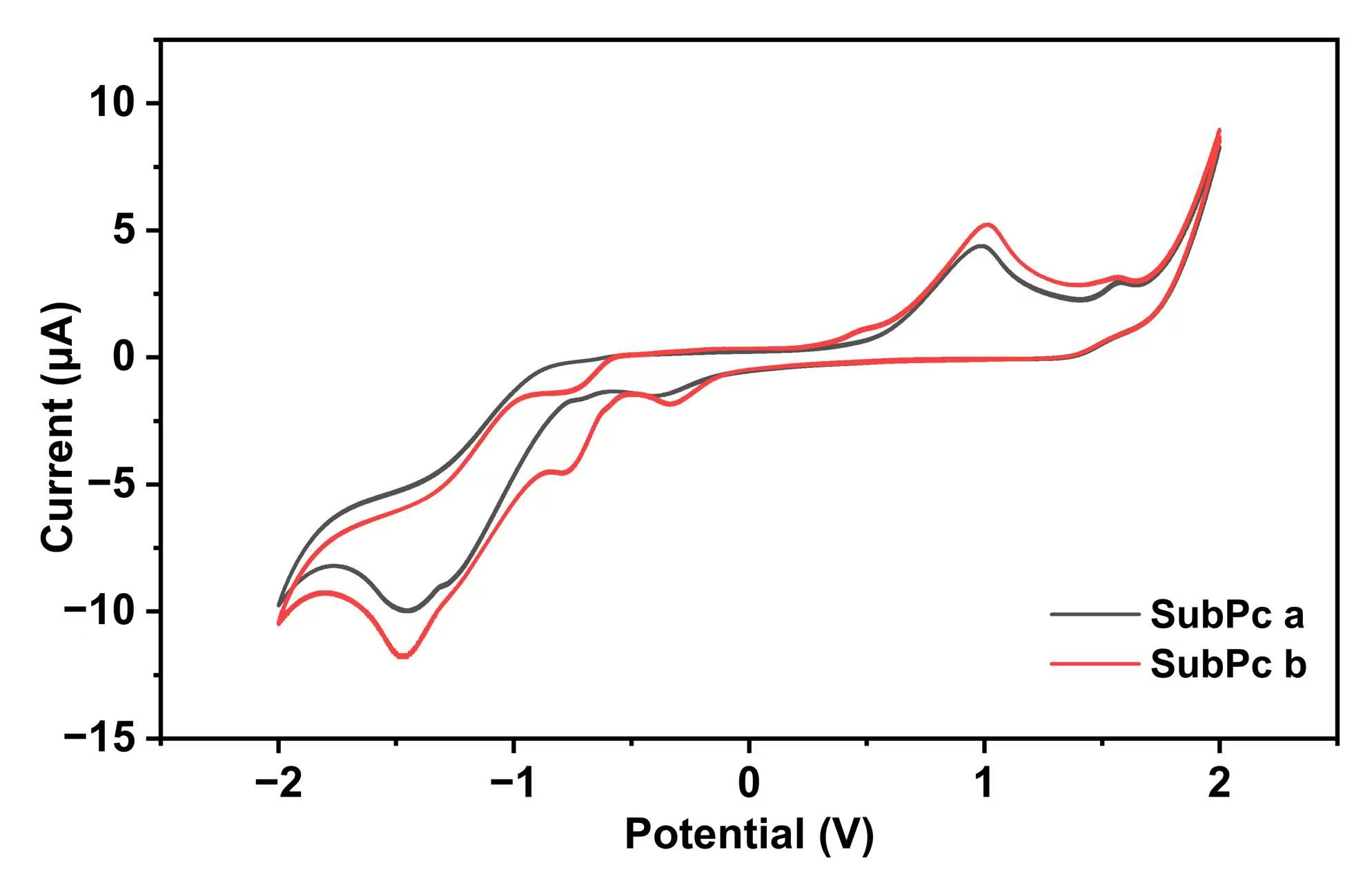 Fig. 5.
Fig. 5.
CV curves of SubPc derivatives in THF. CV, cyclic voltammetry.
| SubPcs | Eox | HOMO (cv) | LUMO (cv) | Eg | HOMO (DFT) | LUMO (DFT) |
| (v) | (ev) | (ev) | (ev) | (ev) | (ev) | |
| SubPc a | 1.06 | –5.80 | –3.67 | 2.13 | –6.71 | –4.10 |
| SubPc b | 1.02 | –5.76 | –3.63 | 2.13 | –6.69 | –4.08 |
HOMO, highest occupied molecular orbital; LUMO, lowest unoccupied molecular orbital; Eox, oxidation potential; Eg, energy gap.
The conformational optimization of two subphthalocyanines was carried out using Gaussian 16W software (Wallingford, CT, USA) based on Density Functional Theory (DFT) with the B3LYP method and the 6-31(G*) basis set [11]. The optimization results are shown in Fig. 6.
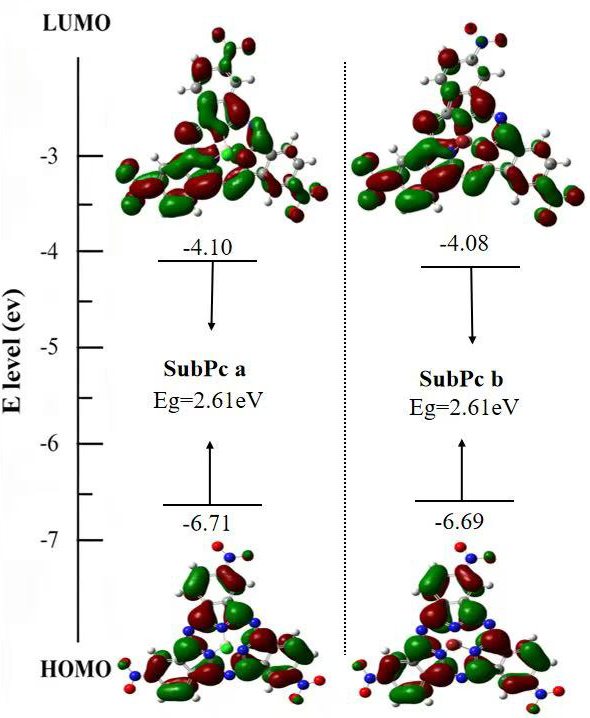 Fig. 6.
Fig. 6.
Effects of two compounds on molecular orbital energy levels.
From Fig. 6, the HOMO energy levels of SubPc a and b are –6.71 eV and –6.69 eV, respectively, and their HOMO orbitals span the entire subphthalocyanine ring. In contrast, the LUMO orbitals are primarily distributed across the benzene rings of the two isoindole units and the nitro groups.
Following material characterization, photodetectors were fabricated. Key parameters, including PVK:SubPc ratio, annealing temperature, and layer thicknesses, were optimized as described in the Experimental Section. Both subphthalocyanine derivatives exhibit strong green light absorption and high HOMO energy levels, which enable them to be used as donor materials in high-performance photodetectors [20, 21]. Two green light photodetectors were prepared using the two SubPc derivatives as photoactive layers in Fig. 7. Fig. 7b presents a schematic energy level diagram of ITO/PEDOT:PSS/C60/Al. The favorable alignment between the ITO/PEDOT:PSS work function and the SubPc HOMO level facilitates hole injection, while the ‘staircase’ alignment of the SubPc LUMO, C60 LUMO, and Al work function promotes efficient electron extraction and blocks hole leakage, contributing to high D*.
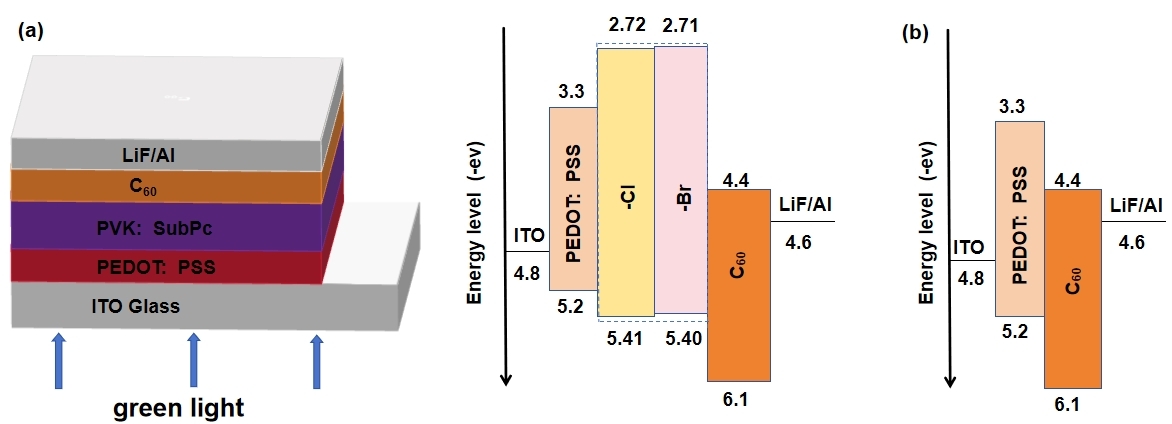 Fig. 7.
Fig. 7.
Device structure and energy level diagrams of SubPc-based photodetectors. (a) Device with SubPc. (b) Energy levels in the absence of SubPc. PEDOT, poly(3,4-ethylenedioxythiophene); PSS, polystyrene sulfonate; ITO, indium tin oxide; PVK, poly (9-vinylcarbazole).
To further characterize the photodetectors, key performance parameters such as R, D* and EQE were calculated using the following formulas [22, 23, 24]. These parameters are summarized in Table 4.
| Devices | Light intensity | Detectivity | Sensitivity | ||
| light | D* | R | EQE | ||
| (µA) | (×1012 cm·Hz1/2/W) | (×10-2 A/W) | (%) | ||
| SubPc a | 520 nm | 2.97 |
1.65 |
9.35 |
28.34 |
| SubPc b | 0.5 mW/cm2 | 2.82 |
1.52 |
8.79 |
27.13 |
In the formula above,
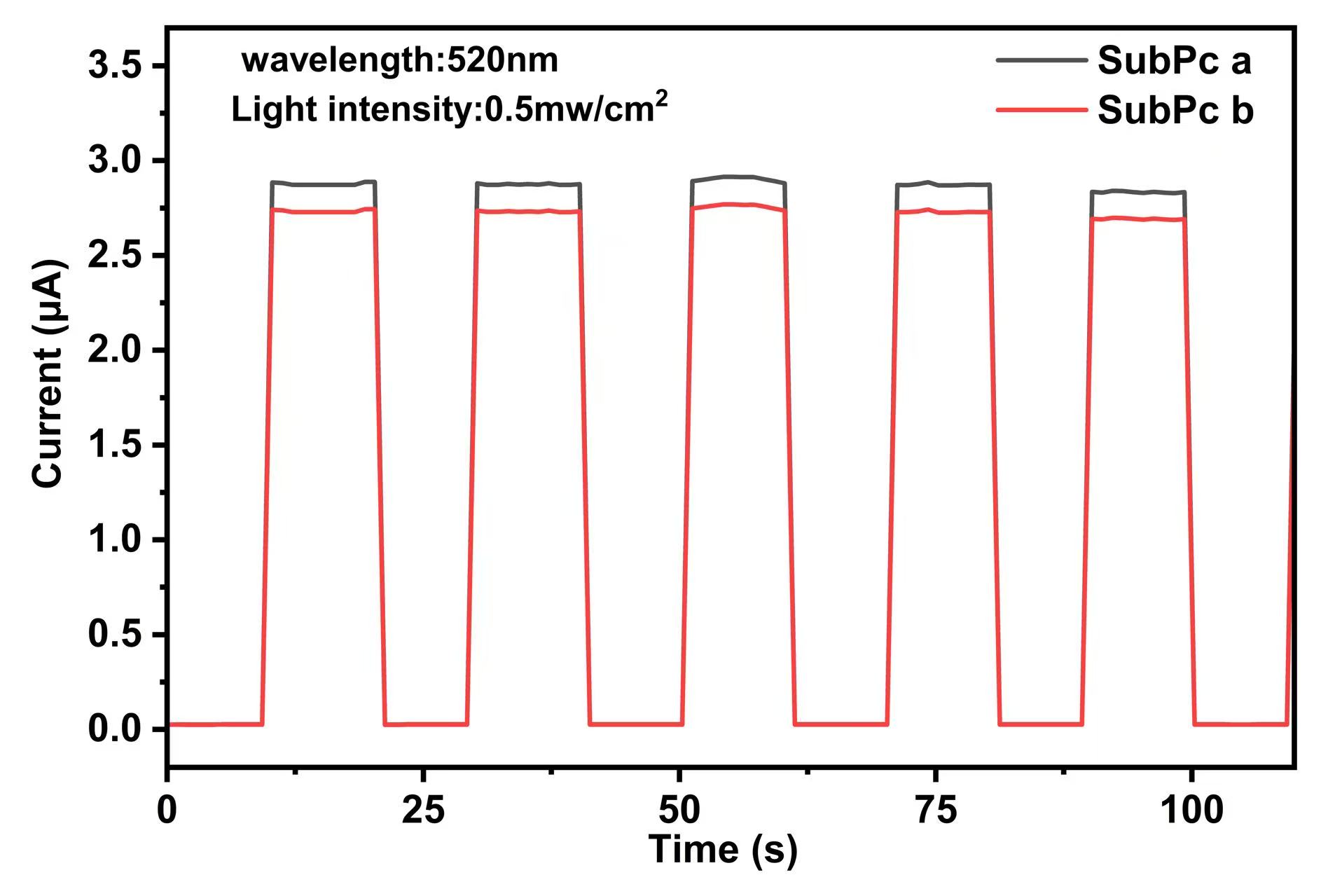 Fig. 8.
Fig. 8.
Switching characteristics of the SubPc-based photodetectors.
As presented in Table 4, the device based on SubPc a demonstrated
superior photodetection performance under 520 nm light illumination (0.5
mW/cm2), with a specific detectivity (D*) of 1.65
Response time (
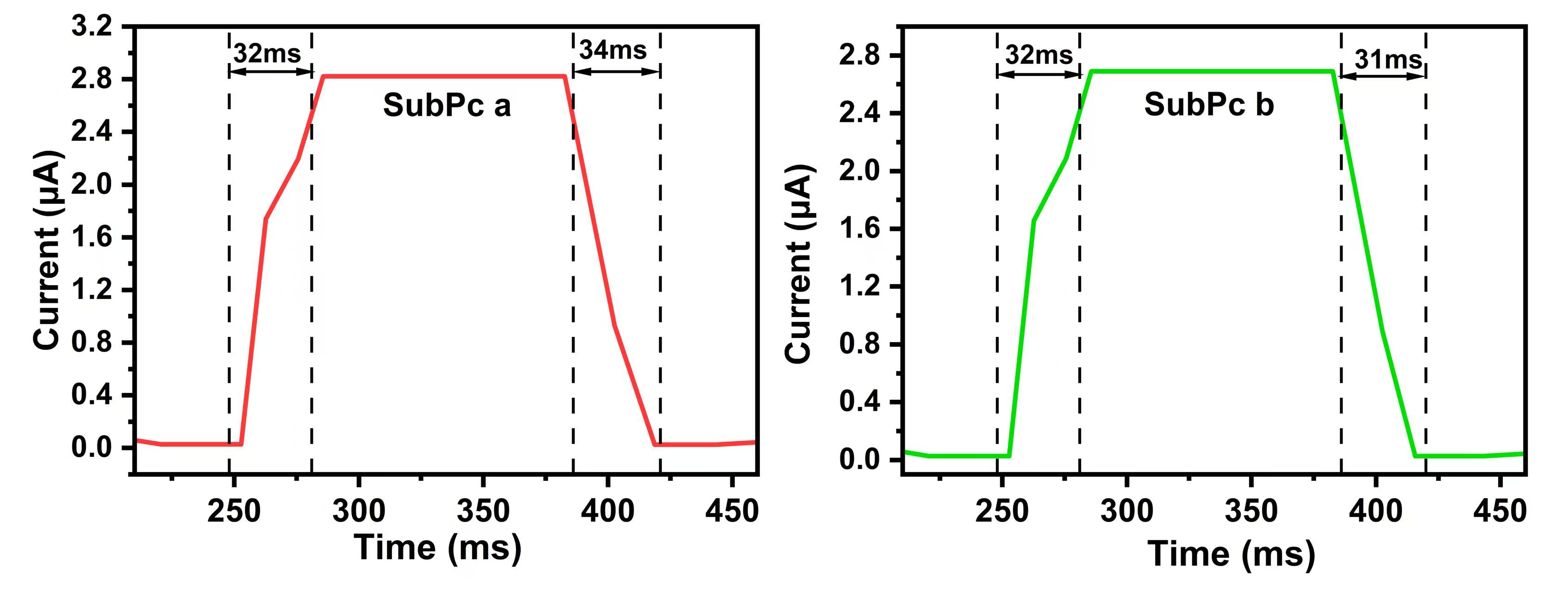 Fig. 9.
Fig. 9.
The diagrams of the light response times of the two photodetectors based on SubPcs.
The absorption maxima of both SubPc derivatives in the 570.0–590.0 nm region
correspond to the characteristic Q-band arising from the C3v symmetric
structure of the SubPc core [27]. The minimal shift in absorption maxima upon
changing the axial substituent indicates that axial modification does not
substantially perturb the core
The charge transfer capability of organic semiconductor materials under
photoexcitation can be estimated by the dipole moment difference
(
The high onset decomposition temperatures indicate excellent intrinsic thermal stability. This property is crucial for the long-term operational stability of OPDs, as it mitigates performance degradation caused by thermal stress during device operation or processing.
The HOMO energy level values of the compounds were calculated via the CV curve to be –5.80 eV and –5.76 eV, which are similar to the typical energy level values of donor materials. This indicates that both SubPc a and SubPc b can serve as donor materials in optoelectronic devices [31]. Additionally, the DFT calculation yielded a HOMO energy level approximately 0.9 eV lower than that obtained from CV testing. This discrepancy is typical, as DFT models an isolated molecule in vacuum, while CV measurements reflect solvated species and involve kinetic processes [32]. Nevertheless, both methods confirm the materials’ suitability as donors and show the same trend (SubPc a has a slightly deeper HOMO than SubPc b).
In photodetector devices, this favorable energy level alignment in the
photodetector devices plays a key role in achieving high detectivity.
Specifically, the matching between the work function of ITO/PEDOT:PSS and the
HOMO of SubPc facilitates efficient hole injection, while the cascade alignment
among the LUMO of SubPc, the LUMO of C60, and the work function of Al
enables effective electron extraction. The superior D* and R of
the SubPc a-based device correlate directly with its larger excited-state dipole
moment change (
Both devices show comparable response times (
We have synthesised novel phthalocyanine semiconductor materials and conducted in-depth investigations into their optoelectronic properties and photogenerated charge separation capabilities. We have also achieved preliminary results in their application within organic photodetector devices. However, the transport mechanism of photogenerated carriers at semiconductor heterojunction interfaces remains to be thoroughly explored. This will be our primary research focus in the next phase.
In summary, two novel SubPc derivatives (SubPc a and b) with nitro/peripheral
and axial halogen substitutions were synthesized. SubPc a exhibited a stronger
charge-transfer character (
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
JT performed the research and drafted the manuscript. SY provided help and advice on the device fabrication and performance testing experiments, and analyzed the data. FM designed the research study and revised the manuscript. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We are deeply grateful to Xiaoming Wu research ream for their assistance and guidance in device testing.
This research was partially funded by the College Student Innovation and Entrepreneurship Training Program Project (No. 202410060014).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









