1 College of Chemical and Materials Engineering, Xuchang University, 461000 Xuchang, Henan, China
Abstract
Recently, the preparation of nano-films through chemical vapor deposition (CVD) has been investigated extensively. A suitable precursor plays a key role in a successful CVD process. However, the majority of reported CVD precursors so far suffer from unsatisfactory stability to air and moisture, which severely restricts the further development of film fabrication by CVD technology. Based on this background, this work focuses on Ni-CVD system and develops a nickel precursor (N,N,N′,N′-tetramethylethylenediamine) [bis(2,4-pentanedionato)] nickel(II) [Ni(acac)2(tmeda)] with the characteristics of excellent stability at low temperatures (off) and high reactivity at high temperatures (on), while also possessing high volatility and thermal stability. The applicability of this precursor in CVD was evaluated through thin film deposition experiments, and the morphology, chemical composition, and crystalline structure of the deposited films were studied.
Keywords
- chemical vapor deposition
- nickel oxide films
- off-on
- precursor
Nickel-containing materials have garnered extensive attention due to their remarkable chemical and physical properties [1, 2, 3, 4, 5, 6]. As an important component of nickel-containing materials, nickel-based films, including oxides [7, 8], nitrides [9, 10], sulfides [11], phosphides [12], and alloys [13], have found extensive applications in various scientific and industrial fields. For instance, nickel metal films are used in magnetic random access memory and metal-insulator-metal (MIM) devices owing to their magnetic properties, high oxidation resistance, and elevated work function [14, 15]. Nickel oxides are extensively used as a hole transport layer in solar cells, attributed to their excellent optical transparency and wide band gap [16]. Nickel phosphide and nickel sulfide films are also widely applied in cutting-edge energy technologies, such as batteries and supercapacitors [17, 18]. These applications have made Ni-based films attract extensive attention.
The deposition methods reported for the preparation of nickel-based films primarily include physical vapor deposition (PVD) [19], atomic layer deposition (ALD) [20], electron beam evaporation [21], sol-gel spin coating [22], and chemical vapor deposition (CVD) [23], etc. Among these methods, CVD has become a preferred choice for preparing nano-film materials due to its high efficiency, low cost, and excellent film uniformity [24]. The essence of the CVD process lies in the gas-phase chemical reactions of precursors that occur in the reaction chamber [25]. Undoubtedly, a successful CVD process depends on a suitable precursor, which not only determines the feasibility of film deposition but also influences the performance of the deposited films [25].
To date, the reported CVD nickel precursors mainly consist of metal-organic
complexes such as carbonyls [26],
As CVD precursors, complexes should possess excellent volatility, thermal stability, high chemical reactivity, high purity, and be inexpensive [25]. Additionally, they should exhibit high stability to moisture and air. However, high chemical reactivity and high stability to moisture and air often represent conflicting requirements. These factors make the development of new CVD precursors highly challenging, and the design of excellent CVD precursors has become a primary issue in the advancement of CVD technology.
According to the existing problems in the field of nickel precursors and the requirements of CVD technology, this study draws on research in the field of fluorescent probes to propose a new concept for precursor design [33, 34, 35]: utilizing the change in coordination number of complexes to endow them with an “off-on” characteristic in the CVD process. This type of nickel complex exhibits excellent volatility and thermal stability. At room temperature, it exists in a stable hexacoordinate form with high stability to moisture and air, which is beneficial for synthesis and handling, i.e., in the “off” state. At relatively high deposition temperatures, one coordination bond between the nickel metal center and the neutral ligand is broken, resulting in a pentacoordinate form with higher reactivity, conducive to gas-phase reaction and film deposition, i.e., in the “on” state. This type of nickel precursor is expected to circumvent the common issue of conflicting air and moisture stability and reactivity found in reported precursors. Based on this mind, this study selected the hexacoordinate complex Ni(acac)2(tmeda) as the precursor and its applicability in fabricating Ni-based thin films via low-pressure chemical vapor deposition (LPCVD) was investigated.
Bis(acetylacetonato)nickel [Ni(acac)2] (99.9%) and N,N,N′,N′-tetramethylethylenediamine (99%) were purchased from J&K Scientific Ltd. (Shanghai, China), and other reagents were obtained from Sinopharm Chemical Reagent Co., Ltd. (Zhengzhou, Henan, China). The synthesis of the complex followed the reported literature method [36], and all reagents were used without further purification. The detailed synthesis method is as follows.
Ni(acac)2(tmeda): A 250 mL flask was charged with Ni(acac)2 (7.707 g, 30 mmol) and 100 mL of n-hexane under air. Subsequently, a mixture of N,N,N′,N′-tetramethylethylenediamine (99%) (3.835 g, 33 mmol) and 20 mL of n-hexane was added dropwise at room temperature, and the reaction mixture was stirred at room temperature for 4 h. After filtration and concentration, the reaction mixture was placed at –30 °C for 12 h to yield blue-green crystals of Ni(acac)2(tmeda) (9.738 g, 87%).
The home-made LPCVD apparatus was as described in the literature [37], as shown in Fig. 1. Thin films were deposited on SiO2/Si(100) substrates, and the substrates were evenly arranged in the effective heating zone of the reaction chamber. To remove contaminants from the substrate surface, the substrates were sequentially ultrasonicated in acetone, isopropanol, and deionized water for 10 min each, followed by drying with N2. The complex was heated at 100 °C, with deposition temperatures ranging from 280 to 480 °C and a deposition time of 30 min. The precursor vapor was transported to the reaction chamber via N2. The N2 included 5 mL/min of “N2 + precursor” and 100 mL/min of “N2 alone”, which were used as the dilution gas and carrier gas, respectively. Other parameters included a deposition pressure of 5 torr and an O2 (reactive gas) flow rate of 100 mL/min.
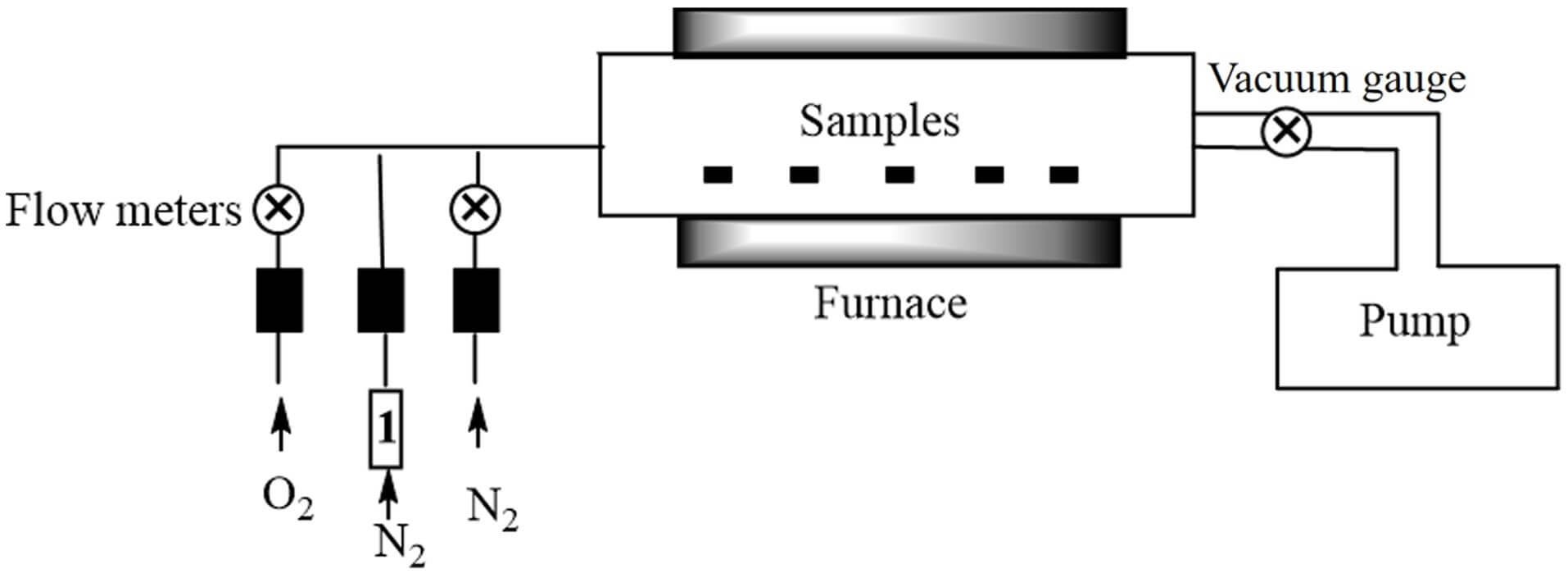 Fig. 1.
Fig. 1.
The LPCVD device. LPCVD, low-pressure chemical vapor deposition.
The surface morphology of the deposited films was analyzed using atomic force
microscopy (AFM, Bruker Dimension Icon, CA, USA) using a contact mode and
field-emission scanning electron microscopy (SEM, Nova NanoSEM 450, FEI Company,
Hillsboro, OR, USA) using a SE mode. The thickness of the deposited films was
measured by an EOPTICS SE-100A spectroscopic ellipsometer (Wuhan Eoptics
Technology Co., Ltd., Wuhan, Hubei, China) using the Cauchy optical model with an
incident angle of 65° and a step size of 1 nm, and the value was
calibrated using SEM. The composition of the films was determined using X-ray
photoelectron spectroscopy (XPS, Thermo ESCALAB 250Xi, Thermo Fisher Scientific,
Waltham, MA, USA) using a standard Al K
As described in Experimental section, the complex Ni(acac)2(tmeda) was synthesized via a simple coordination reaction between Ni(acac)2 and N,N,N′,N′-tetramethylethylenediamine at room temperature in air. The straightforward synthesis process ensures a low cost for the synthesis of complex. To further evaluate the stability of the selected complex against moisture and air, an air exposure experiment was conducted. The results indicated that the complex remained unaltered after 5 days, demonstrating its excellent resistance to moisture and air. This characteristic facilitates handling and utilization.
In our previous work, the thermal properties of the selected complex were investigated [32], and the results demonstrated that the synthesized complex possesses excellent volatility and thermal stability, meeting the requirements of CVD precursors.
To directly explore the applicability of the selected complex in fabricating thin films via CVD, this study conducted thin film deposition experiments using Ni(acac)2(tmeda) as the CVD precursor. Through thin film deposition experiments, it was found that nearly no film was formed when the deposition temperature was below 280 °C. Although a film with a thickness of 45.7 nm was obtained at 280 °C, the film growth rate (1.52 nm/min) was relatively low. This may be attributed to the lower reactivity of the precursor at the lower deposition temperature. Based on this, the present work further investigated the film growth rates at deposition temperatures of 380 °C and 480 °C, and it is found that the film growth rate significantly increased with the rise in deposition temperature, with the film growth rates being 6.73 nm/min (film thickness of 202 nm) at 380 °C and 6.9 nm/min (film thickness of 207 nm) at 480 °C. Furthermore, the deposition at 380 °C for 60 min successfully prepared a film with a thickness of 423 nm.
The surface morphology of the deposited films was characterized using SEM and AFM. As shown in Fig. 2, the films were composed of granular structures, exhibiting continuous and uniform surfaces without cracks. Additionally, it was observed that the particle size of the deposited films gradually increased with increasing deposition temperature. Similarly, the surface roughness of the films was found to increase with increasing deposition temperature, which is consistent with the results obtained from SEM. This may be attributed to enhanced atomic diffusion and lattice reconstruction at higher deposition temperatures, which leads to an increase in both particle size and surface roughness. Moreover, the deposited film exhibits high adhesion to the substrate, with no film delamination observed after attempting to scratch.
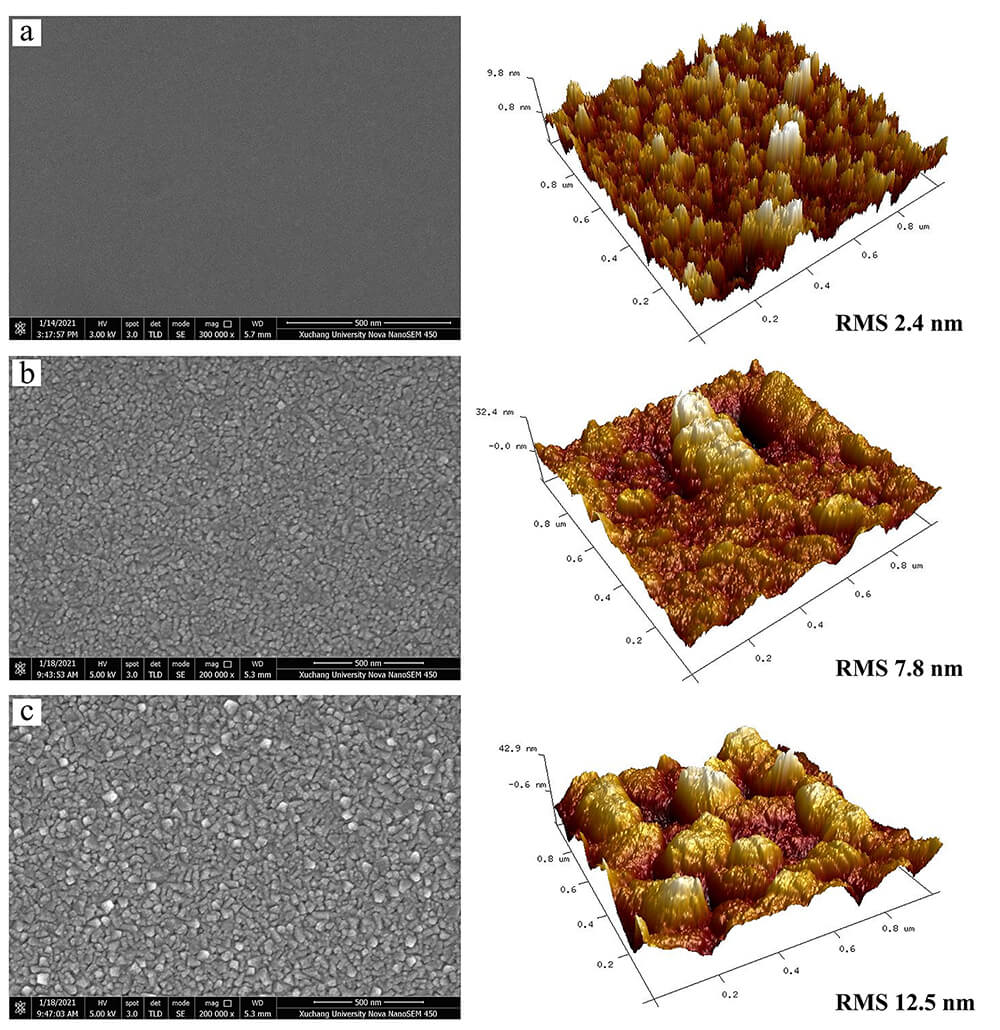 Fig. 2.
Fig. 2.
The SEM and AFM images of the deposited films at 280 °C (a), 380 °C (b), and 480 °C (c). SEM, scanning electron microscopy; AFM, atomic force microscopy; RMS, root mean square roughness. Scale bar = 500 nm.
To obtain the composition and crystalline structure of the deposited films, XRD
analysis was conducted on films deposited at different temperatures. As shown in
Fig. 3, films deposited at 380 °C and 480 °C exhibited diffraction peaks at
2
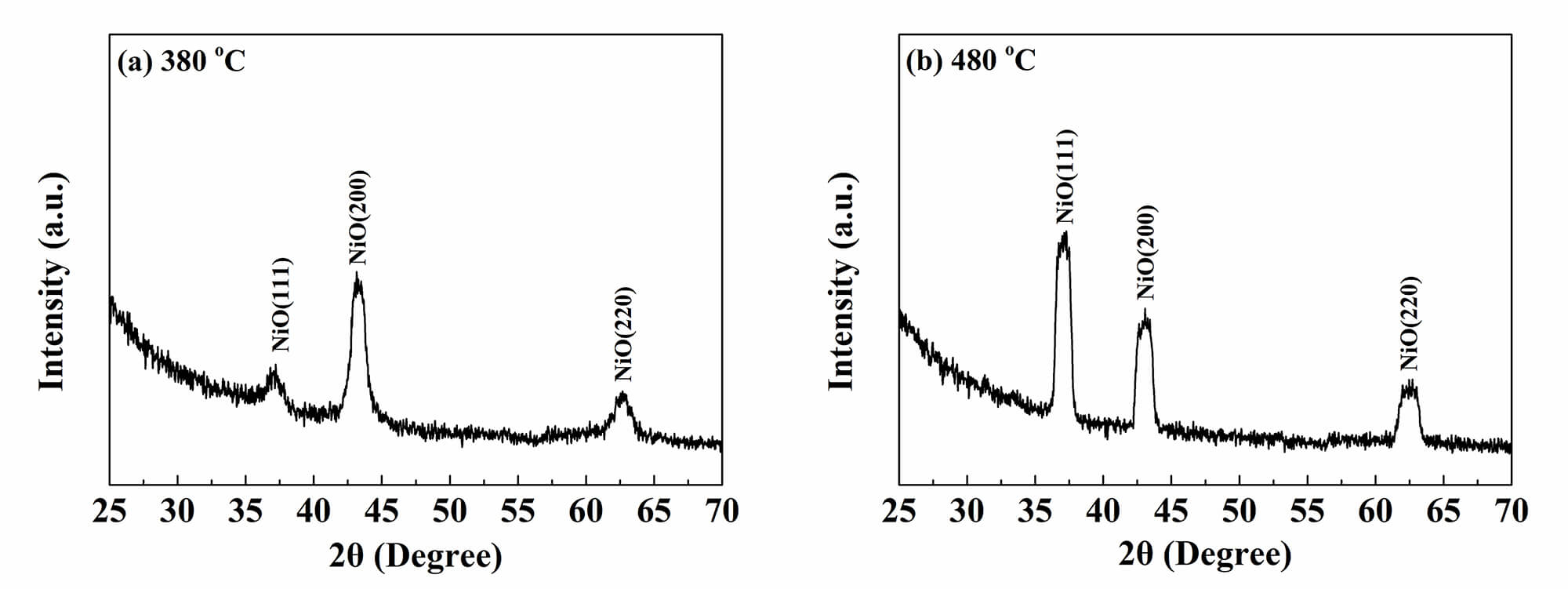 Fig. 3.
Fig. 3.
The XRD patterns of the deposited films at 380 °C (a) and 480 °C (b). XRD, X-ray diffraction.
To further analyze the chemical composition and bonding states of the elements in the deposited films, the film deposited at 380 °C was selected as a representative sample for XPS analysis. The XPS survey spectra of the films are shown in Fig. 4. As depicted, the films primarily consist of Ni, O, and a certain amount of C. After argon ion etching of the films to a depth of 10 nm, the carbon content decreased to 2.1%, indicating that the C element mainly originates from environmental contamination.
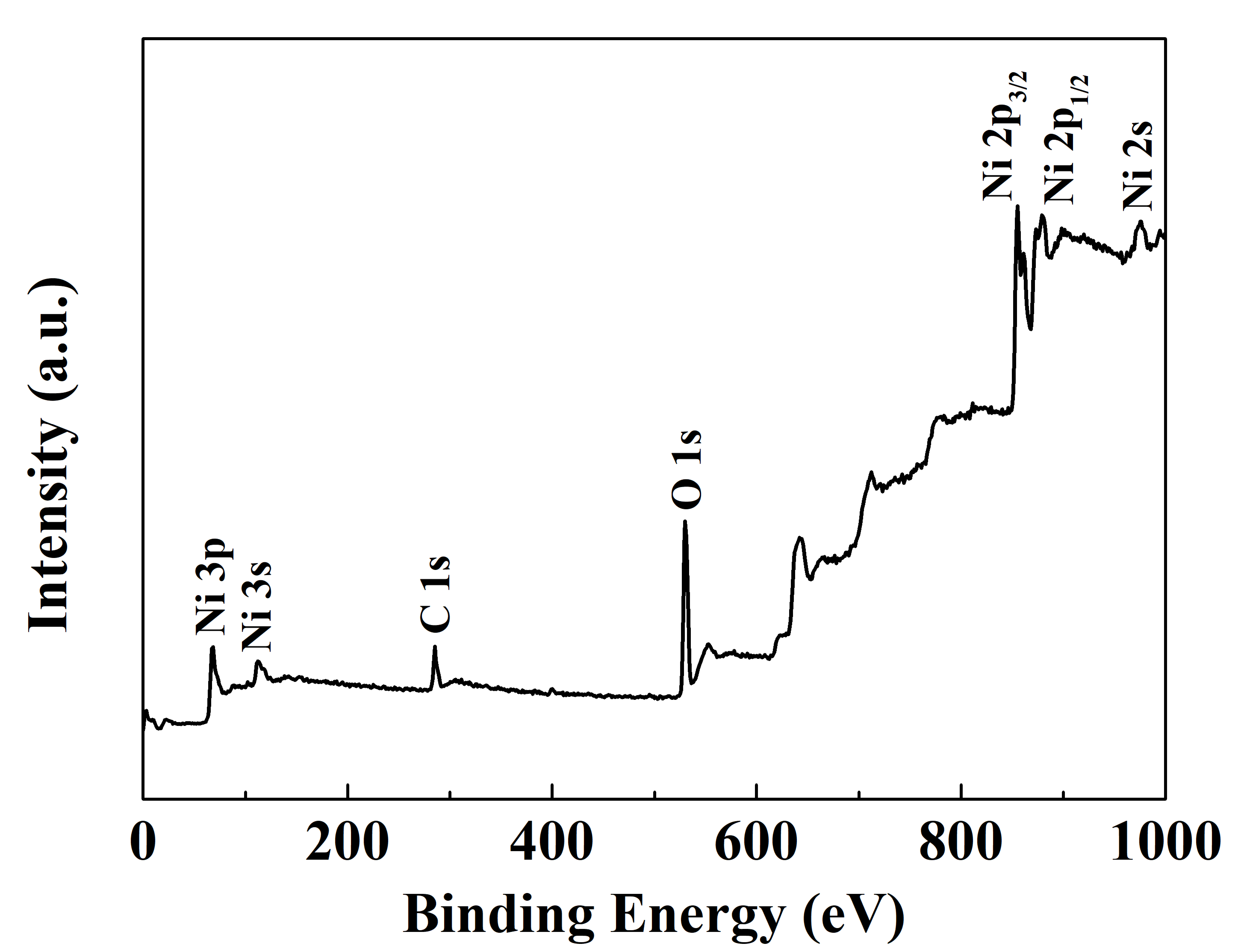 Fig. 4.
Fig. 4.
The XPS survey spectra of the deposited film at 380 °C. XPS, X-ray photoelectron spectroscopy.
Fig. 5 presents the narrow-scan spectra of Ni and O elements in the deposited films. According to the Gaussian-Lorentzian equation, the Ni 2p2/3 region can be divided into seven peaks, corresponding to the compositions of NiO and Ni(OH)2 in the films [39]. The peaks at 854.4 and 856.3 eV, resulting from peak multiplet splitting, can be attributed to Ni2+ in NiO; and the peak at 861.7 eV is a satellite peak of Ni2+ in NiO, arising from the monopolar charge transfer process accompanying the ionization of Ni 2p electrons [39]. Additionally, the peak at 856.6 eV, and the satellite peak at 862.4 eV can be attributed to Ni2+ in Ni(OH)2, also resulting from peak multiplet splitting and the monopolar charge transfer process [39]. The narrow-scan spectrum analysis of the O element is consistent with that of the Ni 2p narrow-scan spectrum. As shown, the O 1s region can be divided into two peaks at 529.8 eV and 531.7 eV, corresponding to O2- in NiO and Ni(OH)2 [39], respectively. It is worth noting that hydroxylation is a common phenomenon for oxide materials [40, 41, 42], and hydroxylation occurs upon exposure of the deposited films to air, resulting in the formation of hydroxides. Obviously, the XPS analysis results are consistent with the XRD analysis results, and all these results strongly demonstrate that the complex Ni(acac)2(tmeda) meets the requirements of a CVD precursor and can successfully fabricate nickel oxide nano-films.
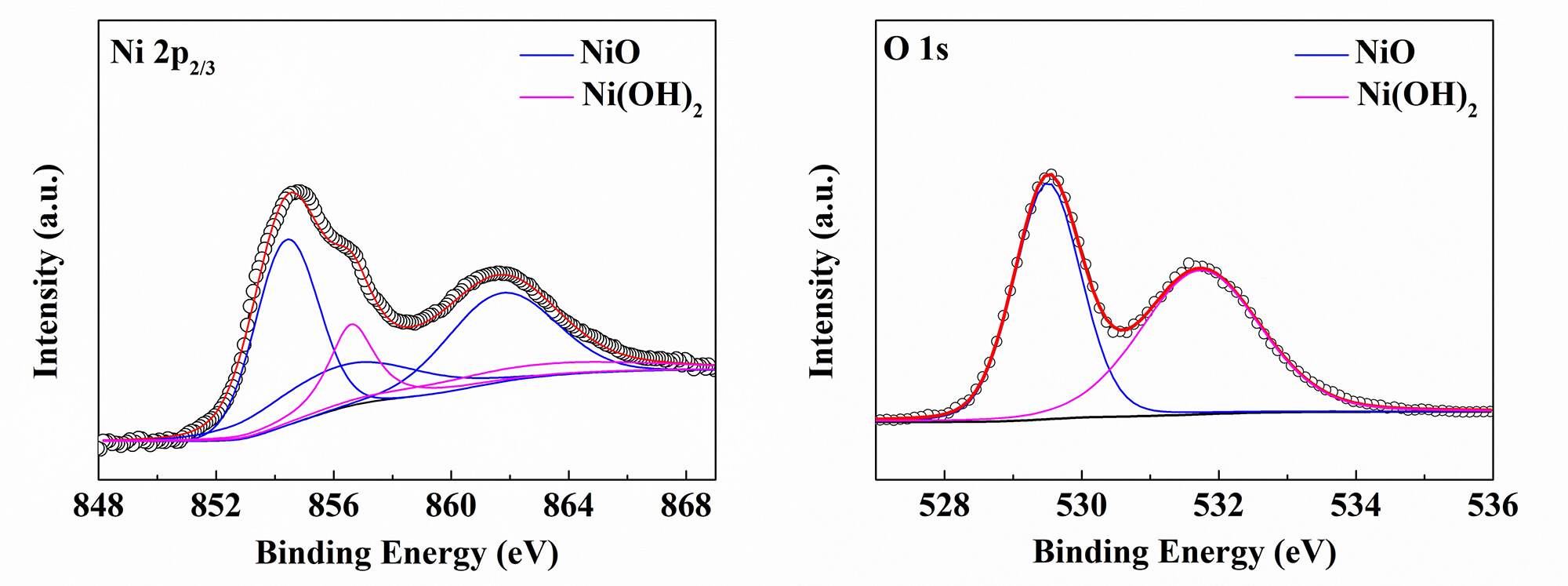 Fig. 5.
Fig. 5.
The Ni 2p2/3 (left) and O 1s (right) XPS survey spectra of the deposited film at 380 °C.
This work systematically investigated the synthesis, chemical properties of Ni(acac)2(tmeda), and its performance as a CVD precursor for thin film deposition. All results indicate that Ni(acac)2(tmeda) satisfies the requirements of CVD precursors regarding volatility, thermal stability, stability to moisture and air, and manufacturing cost, and has successfully enabled the preparation of NiO thin film materials via LPCVD. To more directly demonstrate the application advantages of the selected complex as a CVD precursor, a brief table comparing key properties (volatility, stability, cost) of Ni(acac)2(tmeda) with other common Ni precursors is compiled. As shown in Table 1, the selected complex exhibits remarkable application advantages across three key parameters: volatility, stability, and manufacturing cost.
| Ni Precursor | Ni(CO)4 | Ni(dmamb)2 | Ni(acac)2 | Ni(acac)2(tmeda) |
| Volatility | excellent | excellent | poor | excellent |
| Stability | poor | moderation | moderation | excellent |
| Cost | expensive | expensive | inexpensive | inexpensive |
Ni(dmamb)2, Bis(dimethylamino-2-methyl-2-butoxo)nickel; Ni(acac)2, Bis(acetylacetonato)nickel; Ni(acac)2(tmeda), (N,N,N′,N′-tetramethylethylenediamine) (bis(2,4-pentanedionato)) nickel(II).
To elucidate the “off-on” structural characteristics of the selected complex,
a speculative structural transformation process is proposed. As shown in Fig. 6,
the Gibbs free energy change (
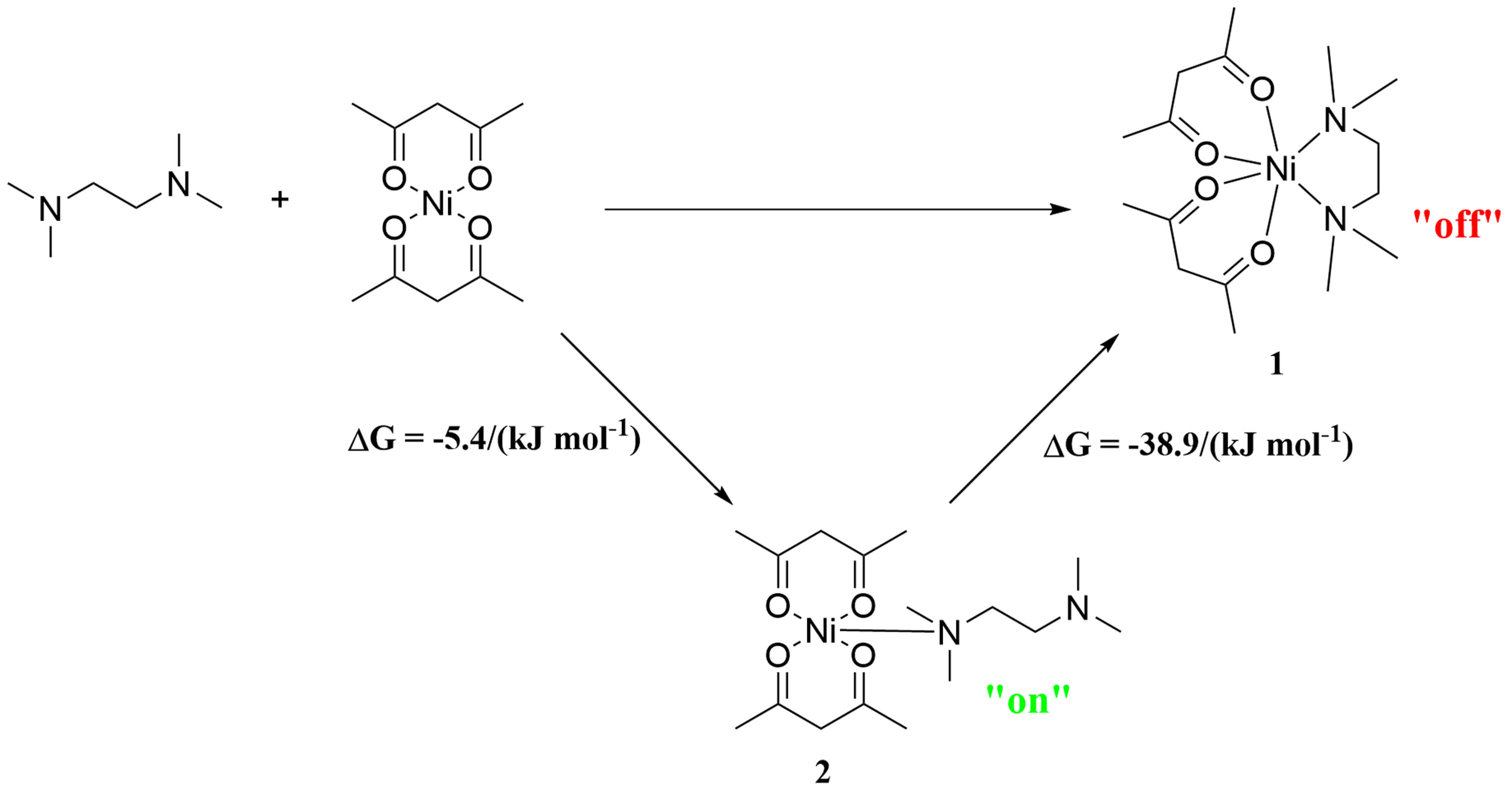 Fig. 6.
Fig. 6.
The transformation process of the complex Ni(acac)2(tmeda) from hexacoordinate to pentacoordinate.
To further verify the “off-on” structural characteristics of the selected complex, a series of experiments was designed. Using Ni(acac)2(tmeda) as the precursor and O2 as the reactant gas, a film thickness of 45.7 nm (corresponding to film growth rate of 1.52 nm/min) was obtained at a deposition temperature of 280 °C (Fig. 7a). In contrast, no film deposition was observed when Ni(acac)2(tmeda) was used as the precursor in the absence of O2. This demonstrates that the CVD process at 280 °C primarily involves the gas-phase chemical reaction between Ni(acac)2(tmeda) and O2, and that Ni(acac)2(tmeda) does not undergo thermal decomposition at this temperature. Further experiments were conducted under the same deposition conditions using Ni(acac)2 as the precursor and O2 as the reactant gas, resulting in a film thickness of 30.8 nm (corresponding to a film growth rate of 1.03 nm/min, Fig. 7b). This thickness is significantly lower than that obtained with Ni(acac)2(tmeda), demonstrating that Ni(acac)2(tmeda) exhibits higher reactivity than Ni(acac)2 during the CVD process at 280 °C. This enhanced reactivity of Ni(acac)2(tmeda) may be attributed to the fact that Ni(acac)2 is a trimeric structure [36], which introduces significant steric hindrance around the nickel metal center, thereby reducing its reactivity. In contrast, Ni(acac)2(tmeda) has a pentacoordinate, unsaturated intermediate structure, which enhances its reactivity. At room temperature, Ni(acac)2(tmeda) exists in a hexacoordinate structure, exhibiting significantly lower reactivity than Ni(acac)2. These results on some content demonstrate that the selected precursor Ni(acac)2(tmeda) possesses high stability to moisture and air at room temperature (off) and high reactivity at elevated deposition temperatures (on).
 Fig. 7.
Fig. 7.
The cross-section SEM images of the deposited films at 280 °C using Ni(acac)2(tmeda) (a) and Ni(acac)2 (b) as precursor. Scale bar = 500 nm.
In the present work, the proposed “off-on” structural characteristics of the selected compound were preliminarily investigated based on the comparative experiments and the literature reported structural transformation process. However, an in-depth research on “off-on” structural characteristics, simulation of deposition, optimization of conditions, and quantitative analysis of the film component and mechanism cannot be completed at this stage, and this will be carried out as the core work of the subsequent phase.
In this study, the applicability of Ni(acac)2(tmeda) as a CVD precursor for depositing nickel-based films is investigated, and the results demonstrate that the selected precursor meets the requirements of CVD technology. By utilizing the change in coordination number of the complex, it exhibits high stability to moisture and air at room temperature (off) and high reactivity at elevated deposition temperatures (on), significantly facilitating the synthesis, handling, and usage of the precursor. Moreover, NiO thin films are successfully deposited using this precursor via LPCVD and the deposited films are continuous, uniform, and of high purity.
The datasets used and analyzed during the current study are available from the corresponding authors on reasonable request.
Yu Z and Ya Z designed the research study. QH and FZ performed the research. DT and SL analyzed the data. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by the Excellent Young Backbone Teachers Funding Project of Xuchang University, the Natural Science Foundation of Henan Province (No. 252300420812), and the Key Scientific Research Project of Colleges and Universities in Henan Province (No. 24A530008).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







