1 Department of Physics, Faculty of Mathematics and Natural Science, Universitas Negeri Surabaya, 60231 Surabaya, Indonesia
2 Department of Physics, Faculty of Science and Technology, Airlangga University, 60115 Surabaya, Indonesia
3 Department of Mechanical Engineering, Universitas Muhammadiyah Ponorogo, 63471 Ponorogo, Indonesia
Abstract
Critical-sized bone defects exceed the body's natural healing capacity and often require scaffold materials to support bone regeneration. Polyvinylpyrrolidone (PVP) is widely used in biomedical applications due to its biocompatibility; however, its osteoconductive performance and mechanical robustness are limited when used alone. To address these limitations, hydroxyapatite (HA) was incorporated into PVP fibers and fabricated into electrospinning scaffolds. PVP/HA composites were prepared at PVP:HA weight ratios of 100:0 (A), 80:20 (B), 60:40 (C), and 40:60 (D), and characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD), porosity testing, and in vitro degradation in simulated body fluid (SBF) over 21 days. Among all compositions, the 40:60 scaffold (Sample D) showed the most favorable structural features, exhibiting the smallest average fiber diameter (326 ± 95 nm), the highest porosity (86.03%), and an increased crystallinity. In SBF, Sample D displayed controlled degradation, with weight loss increasing from 20.55% (day 7) to 61.06% (day 21). Overall, increasing HA loading improved scaffold microstructure and degradation behavior, suggesting that the 40:60 PVP/HA composition offers an optimal balance for electrospinning scaffold design toward bone tissue engineering applications.
Keywords
- electrospinning
- hydroxyapatite
- polyvinyl pyrrolidone
- scaffold
- biodegradable
Critical-sized bone defects remain a major clinical challenge because their size exceeds the intrinsic regenerative capacity of bone, leading to delayed or non-union without intervention. Such defects commonly arise from high-energy trauma, tumor resection, chronic infection, or congenital abnormalities, and they frequently compromise both function and quality of life [1, 2]. In orthopedics and maxillofacial surgery, restoring structural continuity and biological activity is difficult due to the need for simultaneous mechanical support, vascularization, and osteogenic signaling [3, 4, 5, 6].
Clinically, autografts remain a standard option because they provide both osteoconductive and osteogenic properties. However, their use is constrained by donor-site morbidity, limited graft volume, prolonged surgical time, and variable resorption. Allografts and synthetic implants can reduce donor-site problems but introduce other concerns, such as immune response, infection risk, disease transmission, and incomplete integration [7, 8]. These limitations continue to motivate the development of engineered bone substitutes that can provide temporary support while actively promoting bone regeneration [9, 10].
Bone tissue engineering addresses these challenges by combining cells, bioactive cues, and scaffolds to guide new tissue formation [11, 12]. Among these components, scaffolds serve as temporary three-dimensional matrices that support cell attachment, migration, proliferation, and extracellular matrix deposition [13, 14]. For bone repair, an ideal scaffold should exhibit interconnected porosity for nutrient transport and tissue ingrowth, sufficient mechanical integrity during early healing, biocompatibility, and controlled biodegradability, allowing the scaffold to gradually resorb as new bone forms [15, 16, 17].
Electrospinning has emerged as a widely adopted fabrication technique for bone-regenerative scaffolds because it can produce micro- and nanofibrous structures with high surface area and extracellular-matrix-like morphology, which are favorable for cell adhesion and mass transport. Electrospinning scaffolds also allow tunable fiber diameter, composition, and architecture by adjusting solution properties and processing parameters [18, 19, 20]. Accordingly, numerous electrospinning polymer-based scaffolds and polymer-ceramic composites have been explored for bone repair, aiming to balance processability and workable flexibility with bioactivity and osteoconductivity [21, 22].
Polyvinylpyrrolidone (PVP) was selected as the polymer matrix because it is a synthetic, water-soluble polymer with well-established biocompatibility and low toxicity, and it has been widely used in biomedical applications (e.g., as a binder, hydrogel/wound-dressing component, and drug-delivery carrier) [23, 24, 25, 26]. Importantly for scaffold fabrication, PVP exhibits excellent solubility and spinnability in standard solvent systems, which facilitates stable electrospinning and reproducible fiber formation. Nevertheless, pristine PVP typically shows insufficient osteoconductivity and suboptimal mechanical performance for bone-regenerative scaffolds. To address these limitations, hydroxyapatite (HA), a calcium phosphate bioceramic chemically similar to the mineral phase of bone, is commonly incorporated to introduce a bone-mineral-like phase and enhance osteoconductivity and bioactivity. Recent work on hydroxyapatite-based biomaterials continues to demonstrate their relevance for bone tissue engineering. It motivates further optimization of HA-containing composite scaffolds [27]. However, the performance of electrospinning polymer/HA scaffolds is strongly influenced by HA loading: insufficient HA may not provide adequate bioactivity, whereas excessive HA can promote particle agglomeration, alter solution spinnability, and compromise fiber uniformity [28, 29, 30]. Despite extensive work on electrospinning polymer/HA systems, the PVP/HA combination, and specifically the systematic influence of HA content on fiber morphology, crystallinity, porosity, and degradation behavior, remains underexplored [31, 32, 33].
In this study, PVP/HA composite scaffolds were fabricated via electrospinning with varying PVP/HA weight ratios (100:0, 80:20, 60:40, and 40:60). SEM characterized the scaffolds to evaluate fiber morphology and diameter distribution, XRD to assess phase composition and crystallinity, and porosity and in vitro degradation tests in simulated body fluid (SBF) to examine scaffold architecture and resorption behavior. This work aims to identify an appropriate HA loading that yields a structurally robust, porous, and bioactive electrospinning scaffold for potential application in critical-sized bone defect regeneration.
This work provides a formulation-driven optimization of HA loading in electrospinning PVP scaffolds across a wide composition range (0–60 wt% HA), enabling a direct evaluation of how HA content governs scaffold microstructure and performance-relevant properties. In particular, (i) quantify the effects of HA loading on fiber morphology and diameter distribution (SEM), (ii) correlate HA loading with phase/crystallinity characteristics (XRD) and scaffold architecture (porosity), and (iii) assess time-dependent degradation behavior in SBF over 21 days to identify a practical composition window. The resulting structure–property relationships offer a straightforward guideline for selecting HA loading in PVP-based electrospinning scaffolds for bone regeneration-oriented design.
The materials used in this study were polyvinylpyrrolidone (PVP) (Himedia, Maharashtra, India) as the polymer matrix and hydroxyapatite (HA) (Sigma Aldrich, Buchs, Switzerland) as the inorganic bioactive filler. Ethanol (96%) (Merck, Darmstadt, Germany) and distilled water served as solvents to facilitate dissolution and dispersion. All reagents were of analytical grade and used without further purification.
To prepare the spinning solution, 1 g of PVP was dissolved in 8 mL of 96% ethanol and stirred at 900 rpm for 2 hours at 100 °C (hotplate temperature) to form a homogeneous viscous solution. Separately, HA powders were dispersed in a mixture of 1 mL distilled water and 2 mL ethanol using a magnetic stirrer at 500 rpm for 1 hour at room temperature. As shown in Fig. 1, the two solutions were then mixed to obtain three HA-containing PVP:HA solid weight ratios (w/w) of 80:20, 60:40, and 40:60 (PVP:HA); a PVP-only solution (100:0) was prepared as the control. All mixtures were stirred again for 2 hours to ensure homogeneity. Additionally, a solution containing only PVP (100:0, PVP:HA) was prepared as a control to assess the baseline properties of PVP in the absence of HA.
 Fig. 1.
Fig. 1.
Schematic illustration of the preparation of PVP/HA composite solution with different composition ratios: Sample A (100:0), Sample B (80:20), Sample C (60:40), and Sample D (40:60). PVP, polyvinylpyrrolidone; HA, hydroxyapatite. Created with Biorender.com.
Electrospinning was performed using a Nachriebe 601 electrospinning system (CAAI 2601, Bandung, Indonesia) consisting of a high-voltage DC power supply, a syringe pump/feeding unit, and a grounded collector. The final solution was transferred into a 5 mL syringe equipped with a metal needle, and electrospinning was performed at an applied voltage of 16 kV, a solution flow rate of 2.0 mL h⁻1, and a fixed needle-to-collector distance of 10 cm for one hour. Fibers were collected on an aluminum foil-covered grounded collector, as schematically shown in Fig. 2. The as-spun product formed a white, flexible fibrous membrane, indicating successful fiber formation and macroscopically uniform deposition of the composite mat.
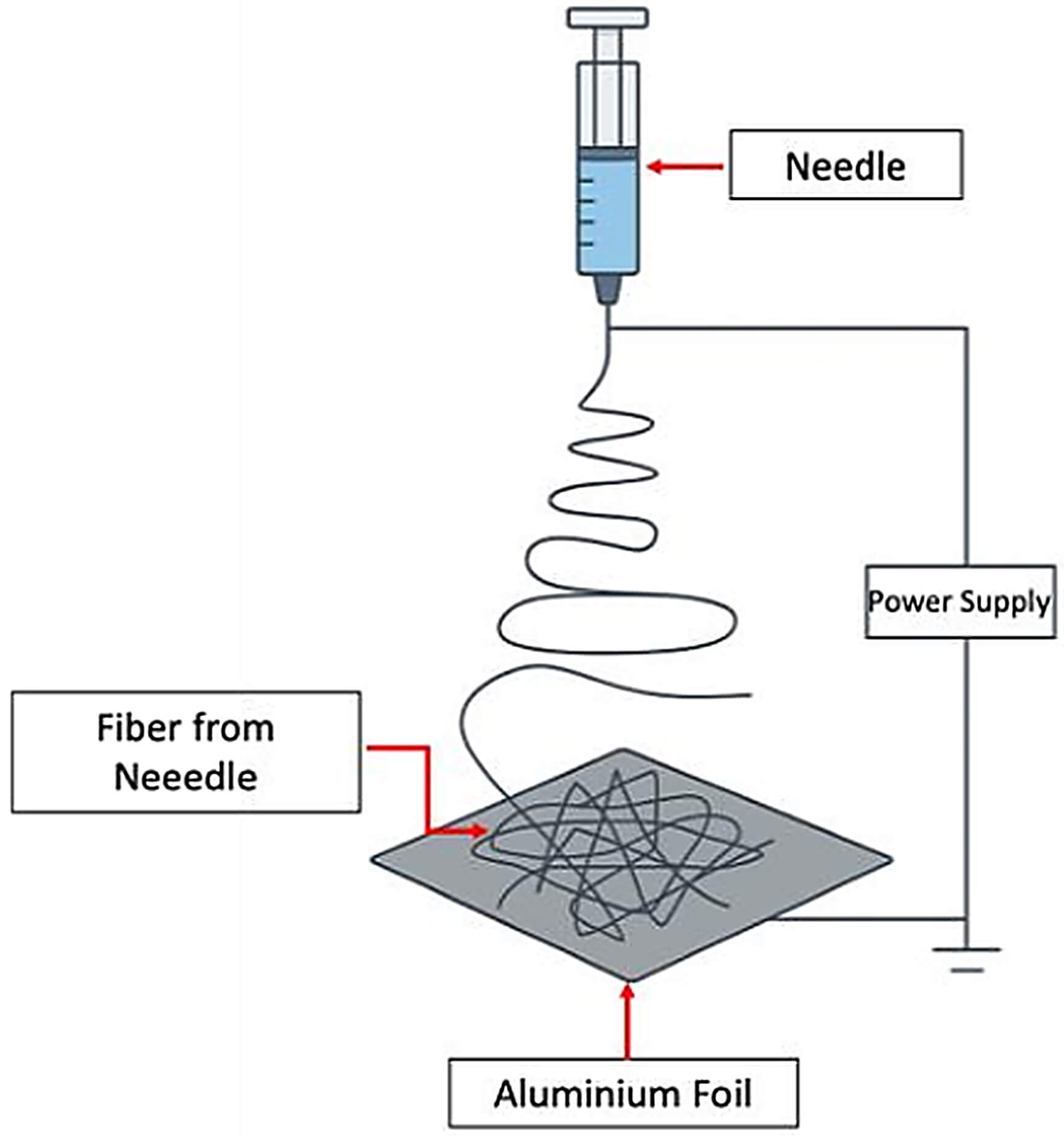 Fig. 2.
Fig. 2.
Schematic illustration of the electrospinning setup used in this study.
Porosity was measured using a liquid displacement method with ethanol as the wetting medium. Briefly, dried electrospinning mats (n = 3 per group) were immersed in a known ethanol volume (V1), and the total volume after infiltration (V2) was recorded. After removing the specimen, the remaining ethanol volume (V3) was recorded. Porosity was calculated as follows:
Time-dependent degradation was evaluated by mass loss during immersion in simulated body fluid (SBF) (sigma Aldrich, Singapore) at 37 °C for 7, 14, and 21 days. Dried specimens were weighed to obtain the initial mass (W0), immersed in SBF, then removed at each time point, gently rinsed with distilled water to remove surface salts, dried again to constant mass, and weighed (Wt). Weight loss was calculated as follows:
The results are reported as mean
To evaluate the structural and functional characteristics of the electrospinning PVP/HA scaffolds, a series of characterizations was performed, including XRD, SEM, porosity analysis, and degradation testing. The XRD analysis was conducted to identify crystalline phases and assess the composite fibers’ crystallinity, particularly to confirm the successful incorporation of hydroxyapatite into the PVP matrix. SEM characterization was used to examine the surface morphology and determine the average fiber diameter of the electrospinning scaffolds. To further assess the suitability of the scaffolds for bone tissue engineering applications, porosity measurements were carried out to estimate the interconnected pore structure, which plays a vital role in cell proliferation and nutrient diffusion. Lastly, degradation studies in SBF were conducted to assess the biodegradability of the scaffolds, an essential property for temporary implants in the healing of critical-sized bone defects.
The XRD analysis of pure PVP was conducted as a reference to characterize its
amorphous structure, serving as a baseline for comparing the crystallinity of the
composite PVP/HA scaffolds. The XRD patterns of electrospinning PVP/HA scaffolds
with varying HA content (100:0, 80:20, 60:40, and 40:60 w/w) are presented in
Fig. 3. The pure PVP sample (100:0) exhibits a broad amorphous hump centered at
2
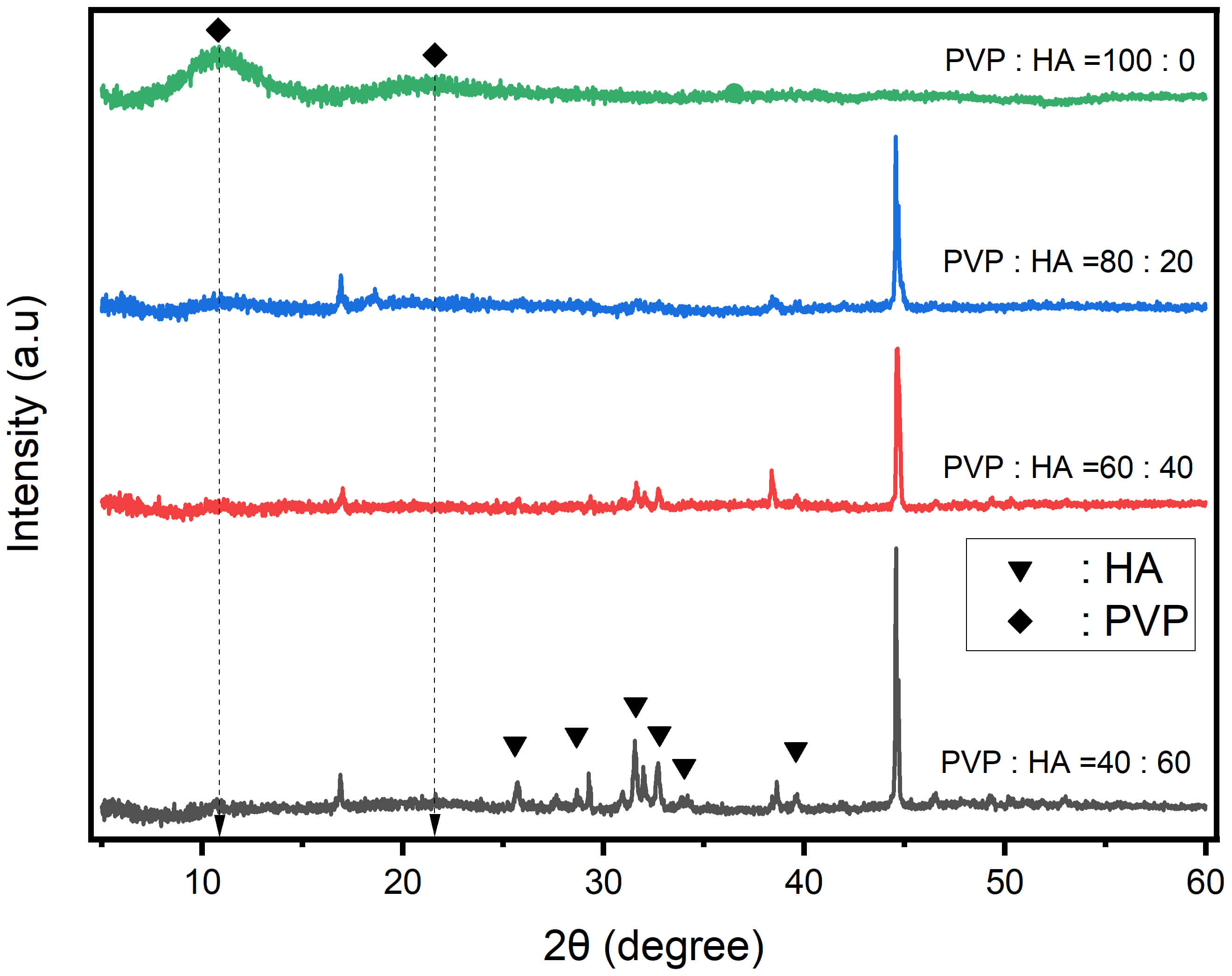 Fig. 3.
Fig. 3.
XRD patterns of electrospinning PVP/HA composite scaffolds at different weight ratios: 100:0, 80:20, 60:40, and 40:60. XRD, X-ray diffraction.
In addition to crystallographic evaluation, the morphological characteristics of the electrospinning PVP/HA scaffolds were investigated using SEM. While XRD confirmed the successful incorporation and increased crystallinity of hydroxyapatite with higher HA content, SEM analysis provided further insight into the composite fibers’ physical structure and surface topography. Understanding fiber morphology—particularly fiber diameter and uniformity—is essential, as it influences cellular behavior, mechanical integrity, and scaffold porosity. The SEM results presented in Fig. 4 below highlight the morphological evolution of the scaffolds with varying HA concentrations and their implications for tissue engineering applications.
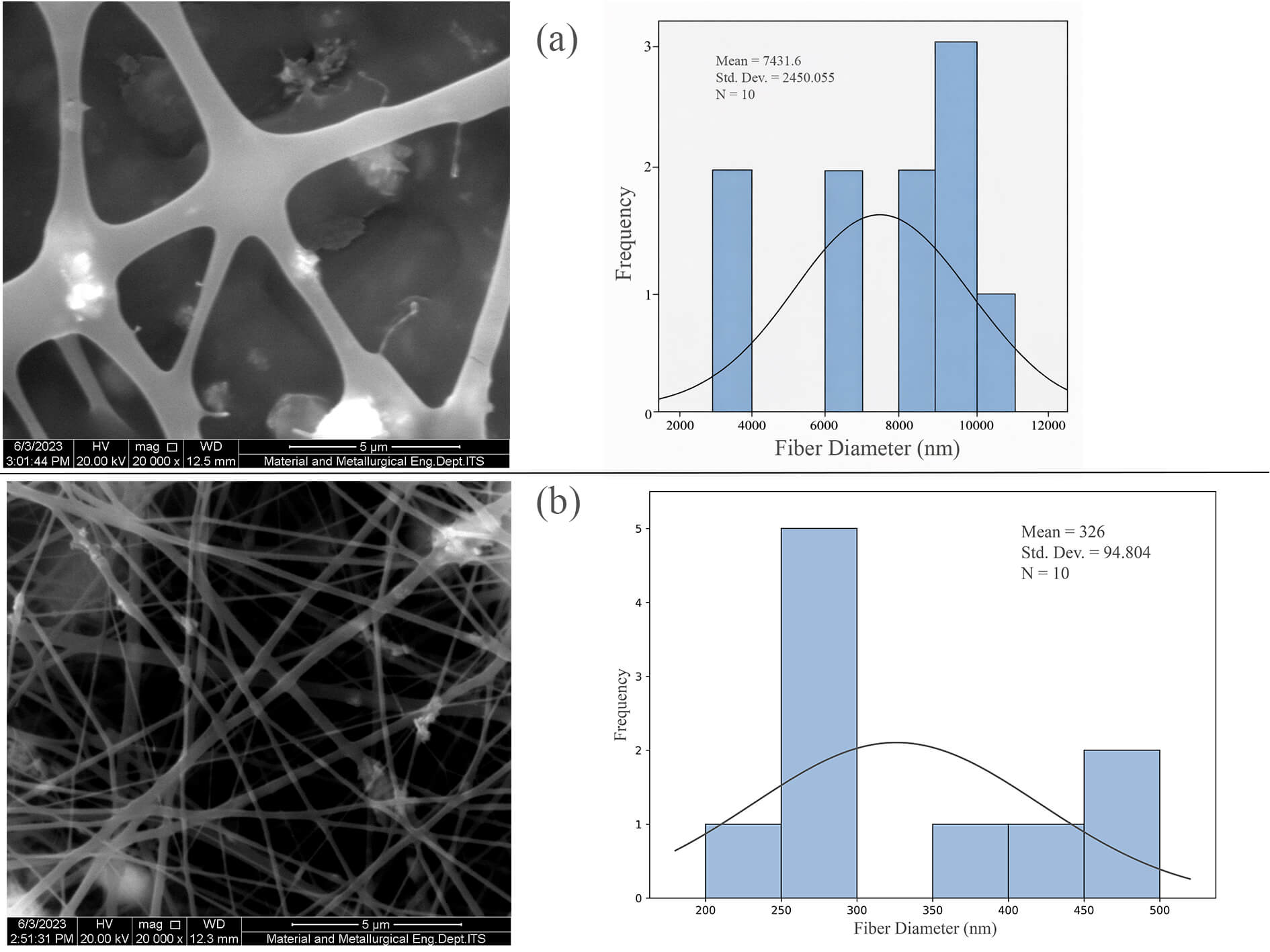 Fig. 4.
Fig. 4.
SEM image and fiber diameter distribution of PVP/HA composite fiber (a) 60:40 and (b) 40:60. Scale bar = 5 µm.
The surface morphology of the electrospinning PVP/HA fibers was analyzed using
SEM at 20,000
In contrast, the PVP/HA 40:60 sample (Fig. 4b) shows a denser fiber network with finer diameters and increased surface roughness, likely due to the higher HA loading. The enhanced dispersion of HA contributes to greater heterogeneity on the fiber surface, which may improve cell adhesion by mimicking the extracellular matrix structure. However, the higher ceramic content also slightly disrupts fiber uniformity, potentially due to increased viscosity or partial agglomeration of HA particles. Overall, the SEM results confirm that increasing HA content alters fiber morphology, with the 40:60 composition achieving a favorable balance of nanofiber structure and bioactive surface features for scaffold applications.
The 60:40 sample exhibited relatively thick, smooth, and randomly oriented fibers, with a high variability in fiber diameter (mean: 7431.6 nm; SD: 2450.1 nm), indicating uneven fiber formation likely caused by suboptimal viscosity or poor HA dispersion during electrospinning. In contrast, the 40:60 sample displayed finer, more uniform, and densely packed fibers, forming a well-integrated nanofibrous matrix with a significantly lower, narrower diameter distribution (mean: 326 nm; SD: 94.9 nm). This refined morphology mimics the extracellular matrix, promoting better cell adhesion and proliferation. At the same time, the higher HA content enhances bioactivity through improved surface exposure without compromising fiber integrity. Overall, the increased HA concentration in the 40:60 scaffold results in more favorable topographical features, making it a more suitable candidate for bone tissue engineering applications.
Following evaluation of fiber morphology and diameter distribution, it is essential to assess the scaffold’s internal structure, particularly its porosity.
The porosity analysis of the electrospinning PVP/HA scaffolds, as illustrated in Fig. 5, demonstrates a clear correlation between HA content and scaffold porosity. Sample A, which likely represents pure PVP or low HA content, exhibits the lowest porosity at 66.70%, indicating limited void formation within the fiber matrix. In contrast, Sample D, assumed to be the highest HA loading (e.g., 40:60 PVP/HA), achieves the highest porosity at 86.03%, suggesting that the addition of HA significantly enhances the formation of interconnected pore structures.
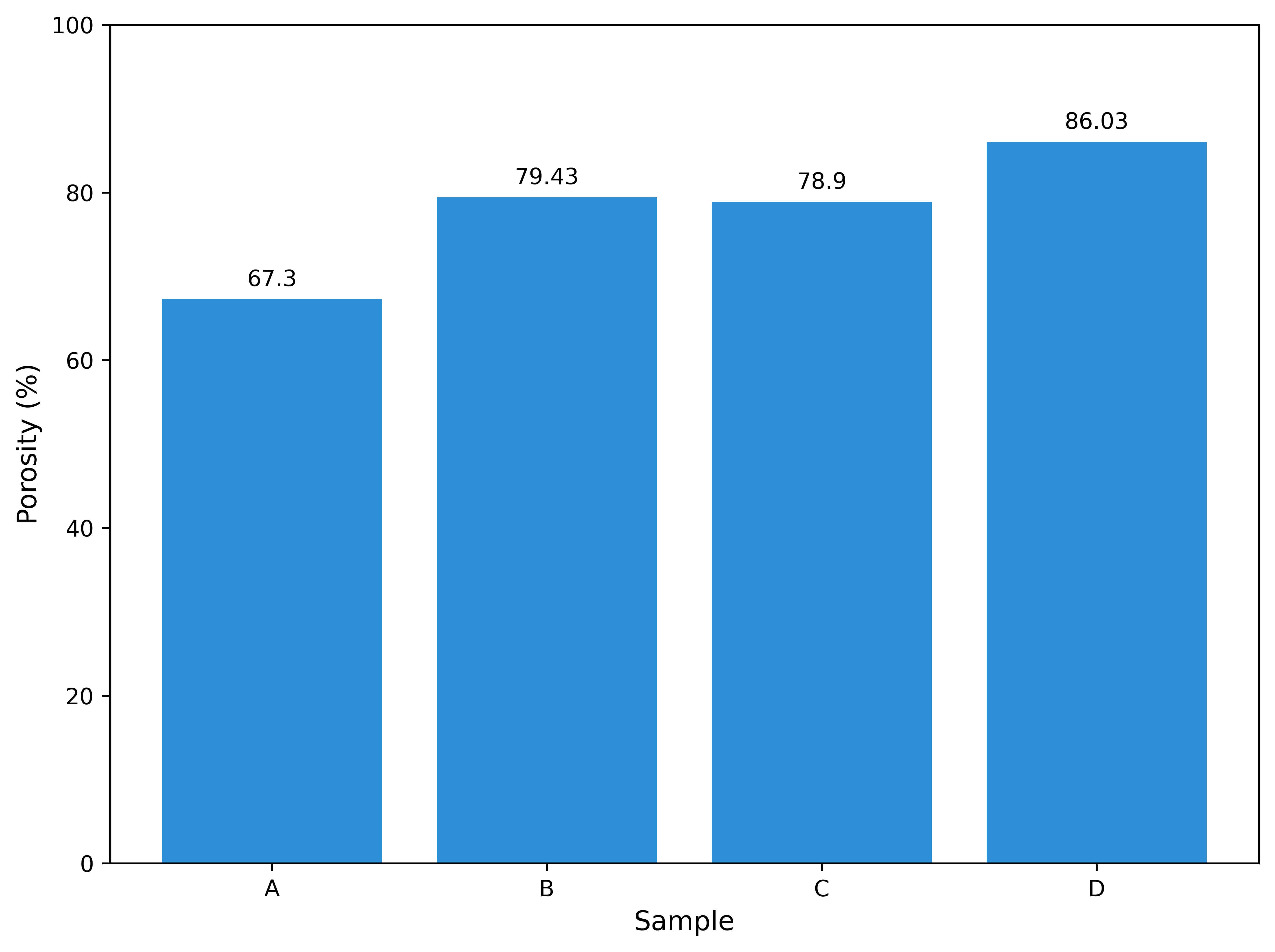 Fig. 5.
Fig. 5.
Porosity of PVP/HA composite scaffolds for samples A–D.
The degradation profile of electrospinning PVP/HA scaffolds over 21 days in SBF, as shown in Fig. 6, reveals distinct behaviors influenced by HA content. Sample A (100:0), composed entirely of PVP, showed the slowest initial degradation (10.83% at day 7) but underwent a sharp increase by day 14 (46.05%) and continued to rise steadily to 62.94% by day 21. This suggests that pure PVP resists early hydrolysis due to its relatively stable polymeric structure. However, once water absorption increases, the matrix becomes more vulnerable to breakdown. Sample B (80:20) exhibited the highest initial degradation (34.65% at day 7), likely due to the introduction of HA particles that disrupted polymer packing and facilitated water diffusion. However, its degradation plateaued more gradually thereafter, indicating that lower HA content may destabilize the fiber structure early but does not significantly prolong degradation at later stages.
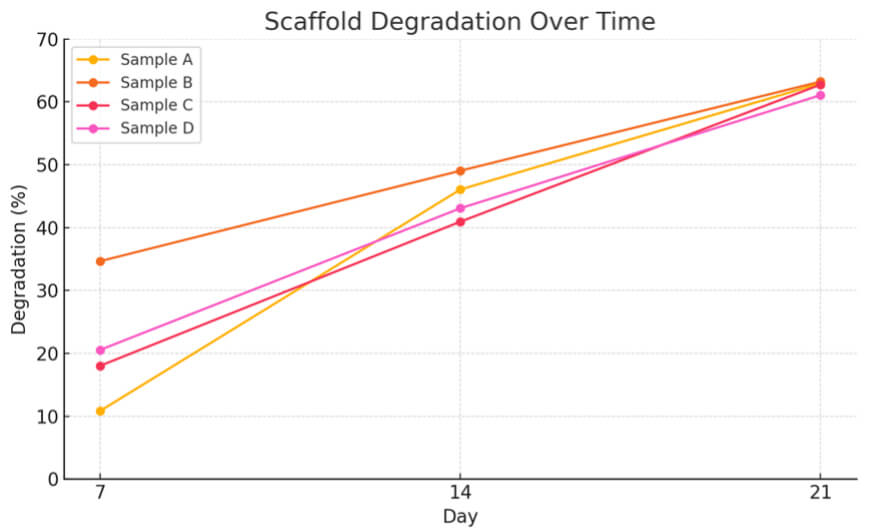 Fig. 6.
Fig. 6.
Time-dependent degradation of electrospinning PVP/HA scaffolds with different PVP:HA ratios.
Samples C (60:40) and D (40:60), which contain higher HA concentrations, demonstrated more controlled degradation across all time points, starting from 18.01% and 20.55% at day 7, respectively, and reaching 62.74% and 61.06% by day 21. This trend suggests that increased HA not only reinforces scaffold structure by introducing a more stable ceramic phase but also slows down water penetration, resulting in more sustained degradation behavior. If the study were extended beyond 21 days, Samples A and B would likely exhibit near-complete degradation due to their higher PVP content, while Samples C and D may continue degrading at a slower rate, allowing for longer scaffold persistence in vivo.
This progressive enhancement in HA crystallinity supports the scaffold’s suitability for bone regeneration, as crystalline HA not only contributes to osteoconductivity but also reinforces the mechanical stability of the composite [1]. The increased intensity and sharpness of HA peaks observed in the 40:60 composition indicate optimal dispersion of HA within the polymer, potentially maximizing its biological function while maintaining the flexibility of the fibrous structure. These results align with the objective of this study to optimize HA loading in electrospinning PVP fibers for the development of structurally and functionally competent scaffolds to repair critical-sized bone defects [4].
The presence of both PVP and HA phases in the composite scaffolds indicates that no new crystalline phases formed during electrospinning, suggesting that HA was physically embedded rather than chemically altered within the polymer matrix [33]. The retained amorphous state of PVP across all compositions indicates that the polymer maintains its film-forming and flexible properties. This biphasic configuration, in which crystalline HA is dispersed in an amorphous polymer matrix, is particularly advantageous, as it balances bioactivity with degradability [6]. Furthermore, the absence of significant peak shifts or broadening indicates minimal chemical interaction at the molecular level, supporting a stable physical blending process and potentially contributing to a predictable degradation profile, critical for scaffold performance in in vivo bone tissue remodeling.
While SEM provides insight into the surface architecture and uniformity of the fibers, porosity analysis offers quantitative information on the scaffold’s capacity to support fluid diffusion, nutrient transport, and cellular infiltration [14]. These properties are fundamental for the scaffold’s performance in tissue regeneration. Therefore, porosity measurements were carried out to further investigate how variations in HA content influence the internal architecture and functional suitability of the electrospinning PVP/HA.
This increase in porosity is advantageous for bone tissue engineering, as it facilitates better fluid exchange, cell migration, and vascularization—critical factors for tissue integration and regeneration. The observed trend aligns with prior findings that inorganic fillers, such as hydroxyapatite, disrupt polymer packing during electrospinning, thereby promoting micro-void formation [29]. Interestingly, while Samples B and C show relatively similar porosity levels (~79%), the slight differences may result from varying fiber orientations or localized HA agglomeration. Overall, the results indicate that higher HA content contributes positively to scaffold architecture, with Sample D offering the most favorable internal structure for mimicking cancellous bone and supporting effective tissue growth.
Where structural support is required during the early stages of healing the porosity is beneficial for bone tissue regenerationand correlated with degradation process [20]. The data indicate that higher HA loading moderates the degradation rate without compromising full resorbability, making the 40:60 composition (Sample D) an optimal balance between mechanical stability and biodegradability for critical-sized bone defect applications. From a translational perspective, the fabrication route used in this study is intentionally designed to be relatively cost-effective and scalable. The system relies on widely available materials (PVP and hydroxyapatite). It uses a simple solvent system (ethanol/water). At the same time, the scaffold morphology and properties are primarily tuned by adjusting the PVP:HA ratio rather than introducing complex chemistries. In addition, electrospinning provides a single-step approach to generate highly porous fibrous matrices that mimic extracellular-matrix-like architecture without requiring high-temperature processing or multi-stage manufacturing. These costs and process efficiency are relevant for early-stage development because they enable rapid formulation screening (identifying the 40:60 composition as the most promising balance between porosity and controlled degradation) and support future scale-up using established electrospinning configurations. Therefore, beyond improving bioactivity, optimizing HA loading within an electrospinning PVP matrix also provides a practical pathway toward accessible scaffold manufacturing for bone-regeneration applications.
Several limitations should be acknowledged. First, this work examined four PVP/HA weight ratios (100:0, 80:20, 60:40, and 40:60) fabricated under a single electrospinning configuration. The effects of key processing variables (e.g., applied voltage, flow rate, tip-to-collector distance, and ambient conditions) were not the primary focus of this study and were therefore kept constant rather than systematically varied. Second, scaffold characterization relied mainly on SEM (morphology and fiber diameter) and XRD (phase identification). Quantitative assessment of HA distribution and surface chemistry (e.g., elemental/chemical mapping) as well as three-dimensional pore architecture was not performed, which limits a comprehensive description of filler dispersion and pore interconnectivity. Third, porosity was estimated using ethanol liquid displacement, providing a bulk value without resolving pore-size distribution or distinguishing open and closed pores. Degradation was assessed by mass loss during static immersion in SBF for up to 21 days. This approach does not quantify mineral/apatite deposition, ion release, pH changes, or the retention of mechanical integrity during immersion, and it does not replicate the enzymatic and cell-mediated processes present in vivo. Finally, direct mechanical testing and biological evaluations (e.g., cytocompatibility, osteogenic response, and in vivo performance) were beyond the scope of this study and are required to validate the optimized composition for translational applications.
This study demonstrated that electrospinning of PVP/HA composite fibers with varying HA content significantly influences scaffold morphology, porosity, and degradation behavior—key factors for bone tissue engineering applications. Increasing HA loading led to enhanced fiber uniformity, higher crystallinity, and improved porosity, particularly in the 40:60 PVP/HA composition, which exhibited a nanofibrous structure closely resembling the extracellular matrix. Furthermore, the scaffold showed controlled degradation over 21 days in simulated body fluid, indicating its suitability for temporary support in bone regeneration. Overall, the optimized PVP/HA 40:60 scaffold presents a promising balance between mechanical integrity, bioactivity, and biodegradability, warranting further investigation in biological environments for potential clinical translation.
The data that support the findings of this study are not publicly available due to confidentiality and institutional restrictions.
ES: investigation, writing-original draft, data curation; EEY: data curation, writing-review and editing; RA: data curation, data calculating, formal analysis, critical revision of the manuscript; WM: methodology development, validation, visualization, and critical revision of the manuscript; NPP: supervision, project administration, and investigation, formal analysis, critical revision of the manuscript; F: validation, project administration, writing-review and editing; M: supervision, formal analysis, conceptualization, methodology, formal analysis, critical revision of the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors would like to thank the LPPM of Universitas Negeri Surabaya, the Department of Physics at Universitas Negeri Surabaya for their support.
This research was funded by Penelitian dasar Kolaborasi (LPPM) Universitas Negeri Surabaya, with contract number B/35011/UN38.III.1/LK.04.00/2025.
The authors declare no conflict of interest.
During the preparation of this work, the author used ChatGPT (Open AI), Grammarly in order to improve the clarity of language, grammar, and overall readability of the manuscript. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publications.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






