1 Basic Geological Survey Institute of Jiangxi Geological Survey and Exploration Institute, 330030 Nanchang, Jiangxi, China
Abstract
Membrane fouling, especially the composite pollution caused by inorganic scaling and biological fouling, is a key challenge in the treatment of underground karst water with high salinity and high hardness. This study reports a polyethersulfone (PES) ultrafiltration (UF) membrane modified by molybdenum disulfide-tannic acid@copper ions (MoS2-TA@Cu2+) bionanomaterial. By blending MoS2-TA@Cu2+ nanoparticles into the PES matrix, the membrane's structure and comprehensive properties were systematically controlled. Scanning electron microscopy (SEM) and static water contact angle measurements were employed to characterize the morphology and surface properties of the membranes. Characterization analysis confirmed that the hydrophilicity of the modified membrane was enhanced (the water contact angle [WCA] was reduced from 69° to 56°), and it showed high antibacterial properties (the antibacterial rate against Escherichia coli [E. coli] reached 95%). The core contribution of this study is to systematically elucidate the tunable trade-off mechanism between key membrane properties dominated by nanoparticle loading. Specifically, a loading capacity of 10 g/m2 achieved optimal anti-fouling stability, with a flux recovery rate (FRR) as high as 81.5% (40.5% higher than that of the original membrane). In contrast, a loading capacity of 15 g/m2 showed the superior permeation and separation performance, with a turbidity removal rate of 92.9% in actual karst water and a silt density index (SDI) reduction value of 5.3, fully meeting the stringent requirements of reverse osmosis pretreatment. Both static and dynamic anti-fouling experiments confirmed its excellent performance, with the CaCO3 scaling amount reduced by up to 65.2%. This study provides an effective strategy for the development of multifunctional UF membranes that can cope with complex water quality challenges by revealing the inherent trade-off between separation efficiency and anti-fouling stability. This paper provides the key theoretical basis and practical guidance for the precise design of high-performance multifunctional membranes.
Keywords
- ultrafiltration membrane
- underground karst water
- MoS2-TA@Cu2+
- anti-scaling
- antibacterial
- polyethersulfone
- nanoparticles
- membrane formation
- additives
- performance trade-off
In typical karst water distribution areas such as Fengcheng, Jiangxi, China, its groundwater resources are important drinking, industrial and agricultural water sources. However, such water sources often exhibit high salinity and high hardness, which not only affects people’s livelihood and industrial applications, but also poses severe challenges to water treatment technology. Ultrafiltration (UF) technology is an ideal pretreatment process for removing colloids, suspended solids and microorganisms due to its high efficiency and low consumption [1, 2]. Polyethersulfone (PES), as a prominent membrane material, has been extensively utilized in these applications due to its outstanding mechanical strength and chemical stability [3, 4]. However, when dealing with high-hardness karst water (HHKW), UF membranes face the problem of composite fouling (CF) caused by the synergistic effect of inorganic scaling and biological fouling: high concentrations of scaling ions (such as Ca2+, Mg2+) easily form an inorganic scale layer on the membrane surface and in the pores, while the rich nutrients in the water promote the growth of microorganisms to form biofilms. The two are intertwined, resulting in a sharp attenuation of membrane flux and shortened membrane life, which seriously restricts the treatment efficiency [5, 6].
At present, there is still insufficient research specifically on the prevention and control of biological pollution, especially the development of modified membranes (Mf-M) with both high-efficiency anti-fouling and long-lasting antibacterial functions. Moreover, many traditional inorganic nanomaterials have limitations in antibacterial properties and biocompatibility. Research on related membrane modification strategies offers multiple approaches to solving such problems [7, 8].
Chemical modification is a common strategy to improve membrane performance, but it often shows the limitation of “functional singleness” in dealing with CF. For example, Elnagar et al. [9] used triethanolamine and polyethylene glycol (PEG) modified polyvinylidene fluoride UF membrane to achieve a removal rate of 90.4% of textile wastewater and achieve water reuse. However, its design only targeted dye pollutants in textile wastewater, did not consider the impact of inorganic scaling or biological pollution in high-hardness water quality, and had insufficient adaptability to complex water quality such as karst water. Jiang et al. [10] prepared micro-UF membranes by blending PES and PEG through non-solvent-induced and reverse heat-induced phase separation methods. Although the membrane performance was optimized, this strategy was only suitable for conventional drinking water quality and was not designed for CF scenarios of highly mineralized and high hardness water. It lacked synergistic inhibition of inorganic scaling and biological pollution.
Nanoparticle modification provides a new approach to membrane modification. Recently, a diverse array of advanced nanomaterials, such as MXenes [11], Metal-Organic Frameworks (MOFs) [12], and Covalent Organic Frameworks (COFs) [13], have been explored to functionalize membranes. However, existing research also faces the challenge of insufficient functional synergy, making it difficult to balance resistance to inorganic fouling and biological pollution at the same time. Zaremanesh et al. [14] modified polycarbonate UF membrane with Mg-Al layered double hydroxide nanoparticles. By lowering the water contact angle (WCA) and increasing porosity, anti-fouling, and separation properties in refinery wastewater treatment were improved. However, the modification target only targeted organic pollution in oil refining wastewater, lacked antibacterial properties or anti-inorganic scaling design, and was difficult to deal with CF of high-salinity water. The “string of pearls” composite nanoparticle-modified polyvinylidene fluoride UF membrane prepared by Zhang et al. [15] had excellent anti-pollution effect. However, this study only verified its anti-pollution performance and did not involve its ability to inhibit scaling problems caused by calcium and magnesium ions in high-hardness water. The function was relatively single.
To address this challenge, some research has turned to external process combinations, but this has not fundamentally improved the “intrinsic” CF resistance of membrane materials. Lu et al. [16] used magnetotactic bacteria to control membrane fouling in UF treatment of chromium-containing surface water under magnetic field action. Although the removal rate of hexavalent chromium was increased by 14.56%, this method only targeted chromium-containing pollution scenarios, did not involve hardness removal or anti-scaling functions, and could not meet the treatment needs of HHKW. Yapıcıoğlu and Yeşilnacar [17] used pistachio shell biochar to adsorb groundwater and reduced the total energy cost by 44.5%–52.5% through data envelopment analysis. However, its biochar adsorption efficiency was limited and it was not combined with membrane modification, which could not solve the pollution problem of UF membrane in high salinity water. Although the electrochemical softening method proposed by Ulusoy and Şimşek [18] achieved 90% removal rate of groundwater hardness and 92% alkalinity with low energy consumption, it only targeted hardness removal and did not involve biological pollution control, and could not solve the inorganic-biological CF of karst water. Ebrahimi et al. [19] used a ceramic membrane to separate organic micropollutants. Although it had a certain removal efficiency, the risk of scaling of calcium and magnesium ions in high-hardness water was not considered, and there was no antibacterial design. Tunçal and Demirkol [20] used the photocatalytic method of nickel-doped titanium dioxide nanocomposite films to increase the iron and manganese removal rates to 96% and 85%, but only targeted iron and manganese pollution, which was out of touch with the anti-fouling and antibacterial needs of UF membranes. Although the reverse osmosis-forward osmosis-halophyte culture composite system proposed by Park et al. [21] optimized the energy and water consumption of groundwater desalination, it did not improve the anti-pollution performance of the membrane material itself and could not fundamentally solve the CF bottleneck of UF membranes.
In summary, the core challenge for efficient treatment of high-salinity, HHKW is to develop a multifunctional UF membrane that can endogenously and synergistically resist the dual threats of inorganic scaling and biological fouling. However, existing research mostly focuses on the improvement of single performance, leaving the structure-activity relationship within a single material system unclear. To address this gap, this study innovatively designed and prepared a new type of molybdenum disulfide-tannic acid@copper ions (MoS2-TA@Cu2+) bio-nanomaterial modified PES UF membrane. Specifically, the main objectives of this study are stated as follows:
(1) To synthesize MoS2-TA@Cu2+ bionanomaterials and fabricate multifunctional PES UF membranes with systematically controlled physicochemical properties.
(2) To comprehensively evaluate the synergistic performance of the modified membranes in terms of hydrophilicity, antibacterial activity against Escherichia coli (E. coli), and resistance to inorganic scaling (CaCO3 and CaSO4).
(3) To reveal and confirm the inherent trade-off mechanism between anti-fouling stability and separation efficiency regulated by nanoparticle loading (identifying the critical thresholds of 10 vs. 15 g/m2).
(4) To validate the practical application potential of the nanocomposite membranes in treating actual high-hardness and high-salinity underground karst water.
The selection of the membrane matrix and nanomaterials was based on the specific treatment requirements for high-salinity and high-hardness underground karst water. Priority was given to materials exhibiting superior hydrophilicity, antibacterial properties, and environmental compatibility. PES was selected as the membrane matrix due to its excellent mechanical strength, chemical stability, and pollution resistance [2]. Molybdenum disulfide (MoS2) was chosen as the nanocore material because its layered structure provides nanochannels to enhance water flux, and its electronegativity aids in electrostatic repulsion of pollutants [22]. Tannic acid (TA) served as the biosource modifier; its abundant phenolic hydroxyl groups offer strong adhesion and reactivity, enabling functionalization through self-polymerization or coordination, aligning with green modification concepts [23]. To introduce antibacterial capabilities, polyethylenimine (PEI) and copper ions (Cu2+) were utilized, targeting bacterial cell membranes and metabolism, respectively.
Polyethersulfone (PES, industrial grade, Ultrason® E 6020 P) was
supplied by BASF (Ludwigshafen, Germany). Scanning electron microscopy (SEM)
grade MoS2 nanoparticles (80 nm, purity
The preparation of antibacterial bionanomaterials adopts a step-by-step modification strategy to endow MoS2 nanoparticles with biocompatibility and antibacterial activity through chemical modification. The specific steps:
Pretreatment of MoS2 suspension: MoS2 nanoparticles were initially washed with DW to remove impurities and dust attached to the surface, and then solid particles were separated by centrifugation. Next, 1.0 g of washed MoS2 was redispersed in 100 mL Tris-HCl buffer solution, and 200 W power ultrasonic treatment was used for more than 30 minutes to break the agglomerates and disperse the particles evenly to improve the stability of the suspension. Afterwards, large particle agglomerates were removed by low-speed centrifugation for further purification to obtain a suspension with a relatively uniform particle size distribution. Finally, the concentration was measured, sealed, and stored. It needed to be lightly stirred or sonicated again before use to avoid particle sedimentation.
Coordination modification of TA/Cu2+: 1.0 g TA was dissolved in 100 mL DW,
adding 0.5 g CuSO4
PEI cross-linking enhancement: The washed particles were redispersed in 50 mL of DW, 2.0 g of PEI (molecular weight 10,000) was added, the pH was adjusted to 8.5, and the reaction was performed with magnetic stirring at 60 °C for 2 h. PEI could combine with the TA-copper coordination layer (TA-Cu2+ layer) through multiple effects: First, the amine group in the PEI molecule forms a hydrogen bond with the phenolic hydroxyl group in the TA molecule that is not involved in coordination, enhancing the interface bonding force; Second, the positive charge carried by PEI electrostatically adsorbs with the negative charge generated by the ionization of phenolic hydroxyl groups in the TA-Cu2+ layer, further stabilizing the coating structure; In addition, the amine group of PEI may undergo a nucleophilic substitution reaction with the phenolic hydroxyl group of TA to form a covalent bond, and ultimately build a 3D network structure to improve material stability. After the reaction was completed, it (10,000 rpm for 10 min) was washed twice with DW and ethanol each to remove residual reagents.
Drying and preservation: The finally obtained MoS2-TA@Cu2+ nanoparticles were dispersed in ethanol, treated with ultrasonics for 5 minutes to break the agglomerates, and then transferred to a vacuum drying oven (VDO) and dried at 60 °C and 0.1 mbar for 24 h. The dried particles were sealed and stored in a desiccator to avoid moisture absorption.
The non-solvent induced phase inversion method (NIPS) was utilized to prepare UF membranes [24, 25]. The process flow mainly includes the steps of film casting liquid preparation, scraping film forming, solidification bath phase separation, and post-processing, as shown in Fig. 1.
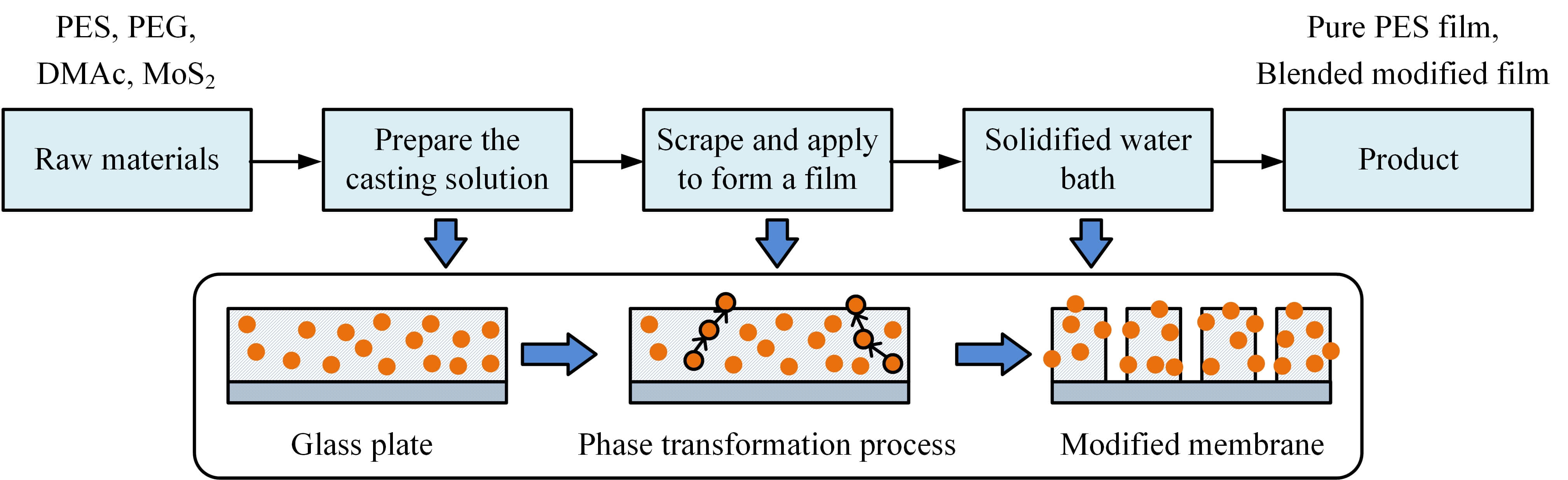 Fig. 1.
Fig. 1.
Membrane preparation process. PES, polyethersulfone; PEG, polyethylene glycol; DMAc, N,N-Dimethylacetamide.
Preparation of the film casting liquid: 15% PES and 8% PEG (molecular weight 2000) were weighed according to the total solid mass percentage. The solvent was DMAc. First, PES and PEG were added to DMAc and magnetically stirred in a constant 70 °C water bath for 8 h until they were completely dissolved to form a uniform and transparent base liquid. Subsequently, according to the target loading amount per unit membrane area (0, 5, 10, 15 g/m2), the corresponding mass of MoS2-TA@Cu2+ modified nanoparticles was dispersed in a small amount of DMAc. Ultrasonic treatment with a power of 200 W was used for 30 minutes to break the agglomerates, and then slowly poured into the base liquid, and continued stirring for 4 hours to ensure good compatibility between the nanoparticles and the polymer matrix. Finally, the mixed solution was transferred to VDO and degassed at 40 °C and 0.1 mbar for 12 h to obtain a uniform and stable casting solution. Based on the nanoparticle loading, the un-Mf-M were recorded as PES, and the Mf-Ms were PES-MoS2-TA@Cu2+-5, PES-MoS2-TA@Cu2+-10, and PES-MoS2-TA@Cu2+-15.
Scraping and solidification molding: The degassed casting liquid was poured evenly onto a clean glass plate, and a scraper with 200 µm thickness was used to scrape the film at a constant speed of 3 cm/s to form a liquid film with a uniform thickness. The scraped glass plate was immediately immersed in a DW coagulation bath at 25 °C to induce phase separation of the polymer through rapid exchange of solvent (DMAc) and non-solvent (water). During the coagulation process, DW needed to be replaced every 8 h for 24 h to completely remove residual DMAc and PEG. After solidification was completed, the film was peeled off from the glass plate to obtain a primary film with a finger-like pore structure.
Post-processing and performance stabilization: The primary membrane was soaked in DW for 48 h, during which the water was changed every 12 h to further clean the remaining impurities. Subsequently, the membrane was transferred to VDO and dried at 60 °C and 0.1 mbar for 6 h to remove moisture and avoid membrane pore collapse. The dried membrane needed to be prepressed with pure water before use: pure water was passed in at 0.15 MPa pressure for 30 min to stabilize the membrane structure and activate surface hydrophilic functional groups.
After the membrane sample was quenched by liquid nitrogen, a 5 nm thick platinum layer was sprayed on the surface. The surface and cross-sectional morphology were observed using a scanning electron microscope (Apreo 2, Thermo Fisher Scientific, Waltham, MA, USA) at an acceleration voltage of 10 kV.
The porosity (
where
The membrane samples were cut into 10 cm
The casting liquid was added dropwise to the glass slide and immediately
immersed in a DW coagulation bath. A high-speed camera (Phantom v2512, Vision
Research Inc., Wayne, NJ, USA) with a resolution of 1280
where
The membrane was installed in a cross-flow filtration device and prepressed at
0.1 MPa pressure for 30 min. The pure water permeation volume within 30 min was
recorded, and the pure water flux (
where
The MWCO of the membranes was determined by conducting ultrafiltration
experiments using a series of PEG aqueous solutions with different molecular
weights (e.g., 20, 40, 60, 100, and 200 kDa) at a concentration of 1.0 g/L. The
filtration was performed under a transmembrane pressure of 0.1 MPa. The
concentrations of PEG in the feed solution (
The MWCO was defined as the molecular weight of the solute corresponding to a rejection rate of 90% in the solute rejection curve.
In this study, a mixed solution of humic acid and BSA was used as a pollutant
model, and a cross-flow filtration device was used for testing. Before testing,
the membrane samples were pre-pressed. Under the operating pressure of 0.05 MPa,
the membrane PWF
reversible fouling resistance:
irreversible fouling resistance:
and total fouling resistance:
were used to comprehensively evaluate the anti-fouling performance (AFP) of the membrane.
The membrane sample was immersed in 1
where
Taking the underground karst water in the Fengcheng area of Jiangxi Province as
the feed water sample, a turbidimeter (HK-288, Beijing Huakeyi Technology Co.,
Ltd., Beijing, China) was used to measure the turbidity value before and after
membrane filtration. Before testing, the water samples were pretreated via a 0.45
µm microporous filter membrane (MFM) to remove large particle interference.
Each test was repeated three times to take the average. Turbidity removal rate
(
where
According to the ASTM D4189 standard method, a silt density index (SDI) meter
(Model MRSDI3325-82905, Roddy Technology, Claremont, CA, USA) equipped with a
0.45 µm mixed cellulose ester MFM was used for testing. First, using
underground karst water in the Fengcheng area of Jiangxi Province as the feed
liquid, under a constant pressure of 0.21 MPa, the time
The SDI reduction value was the difference between the raw water SDI and the filtrate SDI.
A total organic carbon (TOC) analyzer (Elab-TOC/DWT, Suzhou Elan Analytical
Instrument Co., Ltd., Suzhou, China) was used to measure the TOC concentration of
water samples before and after membrane filtration. Before testing, the water
samples were filtered through a 0.45 µm filter membrane to eliminate the
interference of suspended organic matter, and each sample was measured three
times in parallel. The TOC removal rate (
where
Static scaling experiment: The membrane samples were cut into 2 cm
Dynamic filtration anti-scaling experiment: The membrane sample was installed in a cross-flow filtration device, and an aqueous solution of humic acid (mixed concentration of 50 mg/L in underground karst water) was continuously filtered three times under 0.1 MPa operating pressure and 1.5 L/h cross-flow rate. After each filtration cycle, a backwash was performed, and the PWF of different membrane materials was measured.
First, the quality of underground karst water taken from the Permian Changxing Formation and Triassic Qinglong Formation limestone aquifers in the Fengcheng area of Jiangxi was analyzed, as shown in Table 1. It showed a typical HCO3-SO4-Ca-Mg water quality type, with a total hardness as high as 380–490 mg/L, which was a typical hard water.
| Inspection items | Result | Inspection items | Result |
| Water quality type | HCO3-SO4-Ca-Mg | SO42– | 60–95 mg/L |
| Total dissolved solids | 170–240 mg/L | Total hardness (calculated as CaCO3) | 380–490 mg/L |
| Ca2+ | 105–140 mg/L | pH | 7.5–8.3 |
| Mg2+ | 32–58 mg/L | Cl– | 8–14 mg/L |
| HCO3– | 210–280 mg/L | Na+ | 12–19 mg/L |
| SiO2 | 5–12 mg/L | / | / |
SEM was utilized to characterize the cross-sectional morphology of the UF membrane before and after modification with MoS2-TA@Cu2+ nanoparticles at different loadings (0, 5, 10, 15 g/m2), as shown in Fig. 2. As shown in Fig. 2A, the unmodified PES membrane exhibits a typical asymmetric structure, consisting of a dense skin layer and a support layer dominated by finger-like macrovoids. While this fundamental asymmetric configuration was preserved across all modified membranes (Fig. 2B–D)—ensuring adequate mechanical stability—a closer inspection reveals certain modifications in the pore architecture. In the pristine PES membrane (Fig. 2A), the macrovoids are relatively narrow and isolated, separated by thick polymer walls. However, upon the incorporation of nanoparticles, these macrovoids showed moderate widening. Specifically, at the highest loading of 15 g/m2 (Fig. 2D), the polymer skeletal walls between the voids appeared slightly thinner, contributing to improved local connectivity compared to the thick-walled structure of the pristine membrane. This phenomenon can be attributed to the role of nanoparticles as hydrophilic pore-forming agents, which accelerated the exchange rate of solvent (DMAc) and non-solvent (water) during the phase transformation process. This accelerated instantaneous liquid-liquid phase separation process facilitated the development of macrovoids in the support layer, enhancing the pore connectivity and providing a structural basis for the subsequently observed increase in water flux.
 Fig. 2.
Fig. 2.
Cross-sectional SEM images of Mf-Ms with different loading amounts of MoS2-TA@Cu2+ nanoparticles. (A) PES. (B) PES-MoS2-TA@Cu2+-5. (C) PES-MoS2-TA@Cu2+-10. (D) PES-MoS2-TA@Cu2+-15. SEM, scanning electron microscopy; Mf-Ms, modified membranes; TA, tannic acid. Scale bar = 50 µm.
Fig. 3 shows the changes in porosity, MWCO, and PWF of the Mf-M. In Fig. 3A, the
porosity of the Mf-M increased from 71.6% of the PES membrane to 82.4% of the
PES-MoS2-TA@Cu2+-15 membrane. It is noteworthy that the porosity
exhibited a sharp jump at the initial loading (0 to 5 g/m2), while the
growth rate slowed down noticeably as the loading further increased to 10 and 15
g/m2. This phenomenon is attributed to the competitive mechanism between
thermodynamic instability and rheological hindrance. Initially, the addition of
hydrophilic nanoparticles significantly lowers the thermodynamic barrier,
triggering rapid instantaneous demixing and creating abundant voids. However, at
higher loadings (
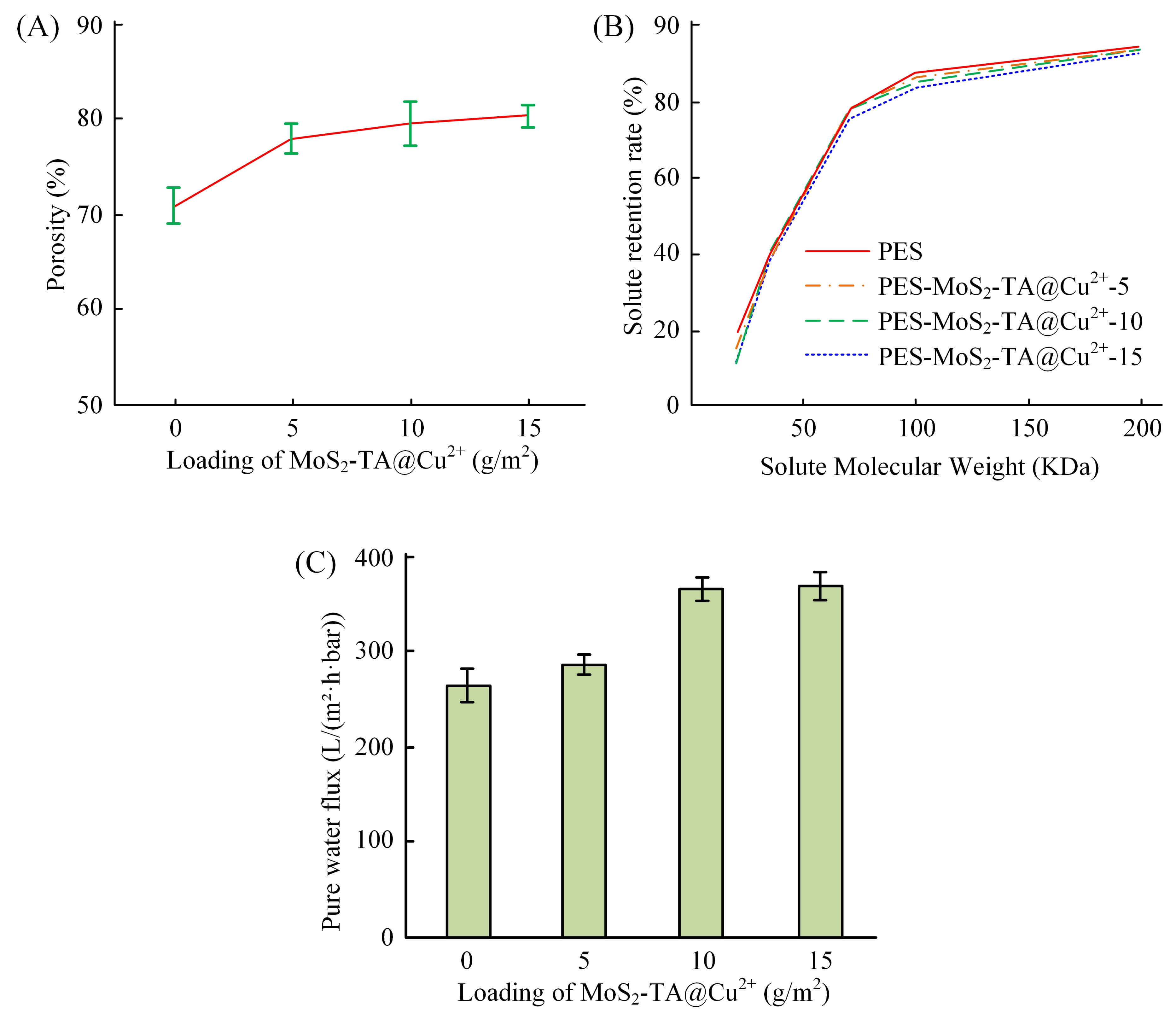 Fig. 3.
Fig. 3.
Changes in membrane properties of the Mf-Ms. (A) Porosity. (B)
MWCO. (C) PWF. PWF, pure water flux; Mf-Ms, modified membranes;
MWCO, molecular weight cutoff. Data are presented as mean
Correspondingly, the PWF of the membrane also increased significantly (Fig. 3C),
from 257.23 L/m2
In addition, the change in MWCO of the UF membrane is exhibited in Fig. 3B. Compared with the PES membrane, the MWCO of the MoS2-TA@Cu2+ Mf-M increased moderately from 127 kDa to 169 kDa. This phenomenon seems to be contrary to the expected separation performance, but its underlying mechanism can be explained as follows: MoS2-TA@Cu2+ nanoparticles acted as hydrophilic “porogens” to accelerate phase transformation and form larger supporting pores (Fig. 2D). At the same time, they may also cause microscopic disturbances in the stacking of polymer chains in the dense cortex of the membrane surface. This perturbation may lead to a slight increase in the mean value of the effective pore size distribution of the cortex, which is manifested as an increase in MWCO. Therefore, the Mf-M achieved a balance between a large increase in water flux and a moderate adjustment of selectivity, rather than a simple performance degradation.
Fig. 4 presents the hydrophilicity test results of MoS2-TA@Cu2+ nanoparticle-modified UF membrane. In Fig. 4A, the contact angle of the unmodified PES film was 69°. As the amount of MoS2-TA@Cu2+ nanoparticles gradually grew from 0 to 15 g/m2, the contact angle of the modified film showed a downward trend, and the contact angle of the PES-MoS2-TA@Cu2+-15 film dropped to 56°, indicating that the hydrophilicity of the film gradually increases. This is because the introduction of MoS2-TA@Cu2+ nanoparticles increases the polar groups on the membrane surface and improves the water wettability of the membrane. Fig. 4B shows the gel displacement curve of the cast film liquid. The gel displacement of all casting solutions gradually increased over time. Among them, the cast liquid gel displacement of the PES-MoS2-TA@Cu2+-15 membrane increased most significantly, reaching 110 µm at 60 seconds, which was significantly higher than the 95 µm of the PES membrane. This demonstrated that as the loading amount of MoS2-TA@Cu2+ nanoparticles increased, the gelation rate of the casting solution accelerated and the gel displacement increased. This change may be due to the introduction of nanoparticles, changing the phase separation process of the casting liquid and promoting the formation and development of gel. Faster gel rates and larger gel displacements contributed to a more uniform and denser membrane structure, which may have a positive impact on membrane performance.
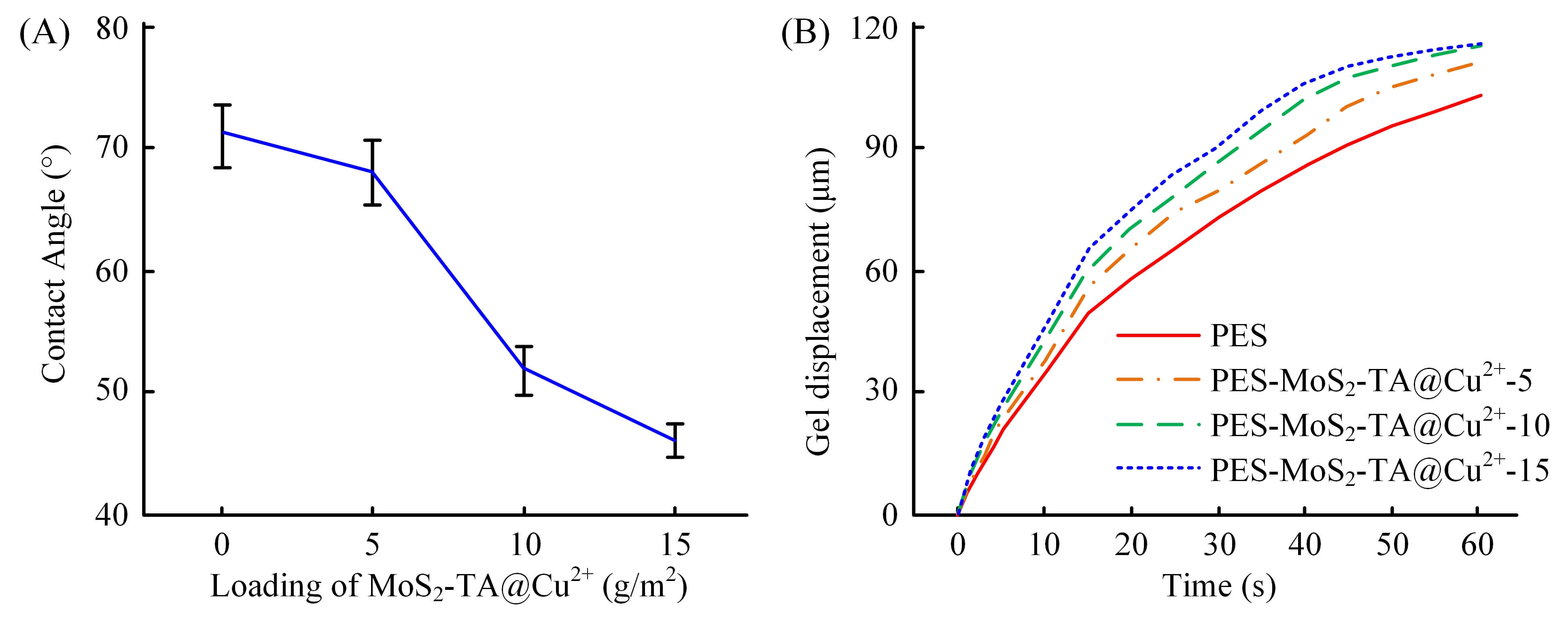 Fig. 4.
Fig. 4.
Membrane hydrophilicity and film formation kinetics. (A) Static WCA. (B) Curve of cast film liquid gel displacement with time. WCA, water contact angle.
A three-cycle filtration test was conducted using a mixed solution of humic acid and BSA as simulated pollutants to evaluate the dynamic anti-fouling stability. The normalized flux changes are presented in Fig. 5. All membranes experienced a typical flux decay during the filtration stage due to the accumulation of foulants. However, the unmodified PES membrane suffered the most severe attenuation, with the normalized flux dropping to 0.58 in the third cycle, indicating serious irreversible fouling caused by hydrophobic adsorption of proteins and organic matter.
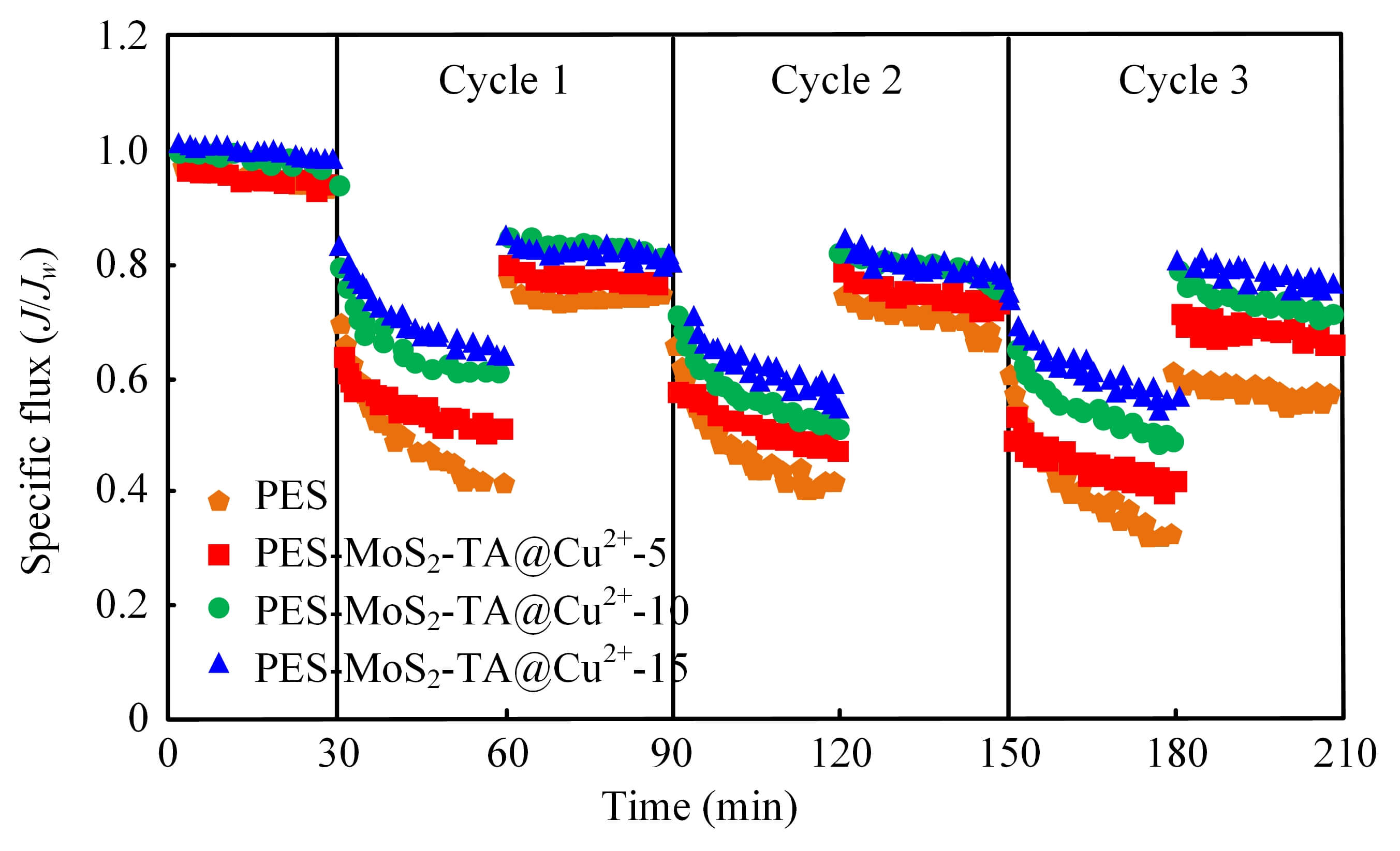 Fig. 5.
Fig. 5.
Normalized flux changes of the membrane in the three-cycle filtration experiment.
In contrast, the Mf-M modified with MoS2-TA@Cu2+ demonstrated significantly enhanced fouling resistance. Specifically, for the PES-MoS2-TA@Cu2+-10 membrane, the normalized flux in the third cycle was maintained at 0.815, representing a 40.5% improvement over the pristine PES membrane. This superior performance can be attributed to two synergistic mechanisms: First, the abundant hydrophilic hydroxyl groups from TA and MoS2 formed a tightly bound hydration layer on the membrane surface, which acts as a physical barrier to prevent the direct contact and adsorption of foulants (steric hindrance effect) [22]. Second, the electronegativity of the modified surface induces a strong electrostatic repulsion against negatively charged foulants (humic acid and BSA) at neutral pH, thereby mitigating the deposition rate [15]. Compared to similar PES-based modification studies where flux recovery rate (FRR) values typically range from 60% to 75% [3], the FRR of 81.5% achieved in this work highlights the exceptional reusability and operational stability of the MoS2-TA@Cu2+ nanomodified membrane.
Fig. 6A shows the changes in membrane fouling parameters with the loading of MoS2-TA@Cu2+ nanoparticles when a mixed solution of 50 mg/L humic acid and 50 mg/L BSA is used as a model pollutant. As the loading amount of MoS2-TA@Cu2+ nanoparticles increased, the fouling parameters of the membrane changed significantly. Among them, RFR and IFR first decreased and then increased, while FRR first increased and then decreased. Fig. 6B shows the antibacterial performance test results. As the loading amount of MoS2-TA@Cu2+ nanoparticles increased, the antibacterial rate of the membrane significantly increased. When the loading capacity was 15 g/m2, the antibacterial rate reached 95%, which was significantly higher than that of the unmodified PES membrane. This indicated that the addition of MoS2-TA@Cu2+ nanoparticles could effectively enhance the antibacterial performance of the membrane, reduce the attachment and growth of microorganisms on the membrane surface, thereby reducing the risk of membrane fouling. Meanwhile, combined with changes in membrane fouling parameters, appropriately loaded MoS2-TA@Cu2+ nanoparticles could improve the AFP of the membrane while improving the antibacterial performance of the membrane.
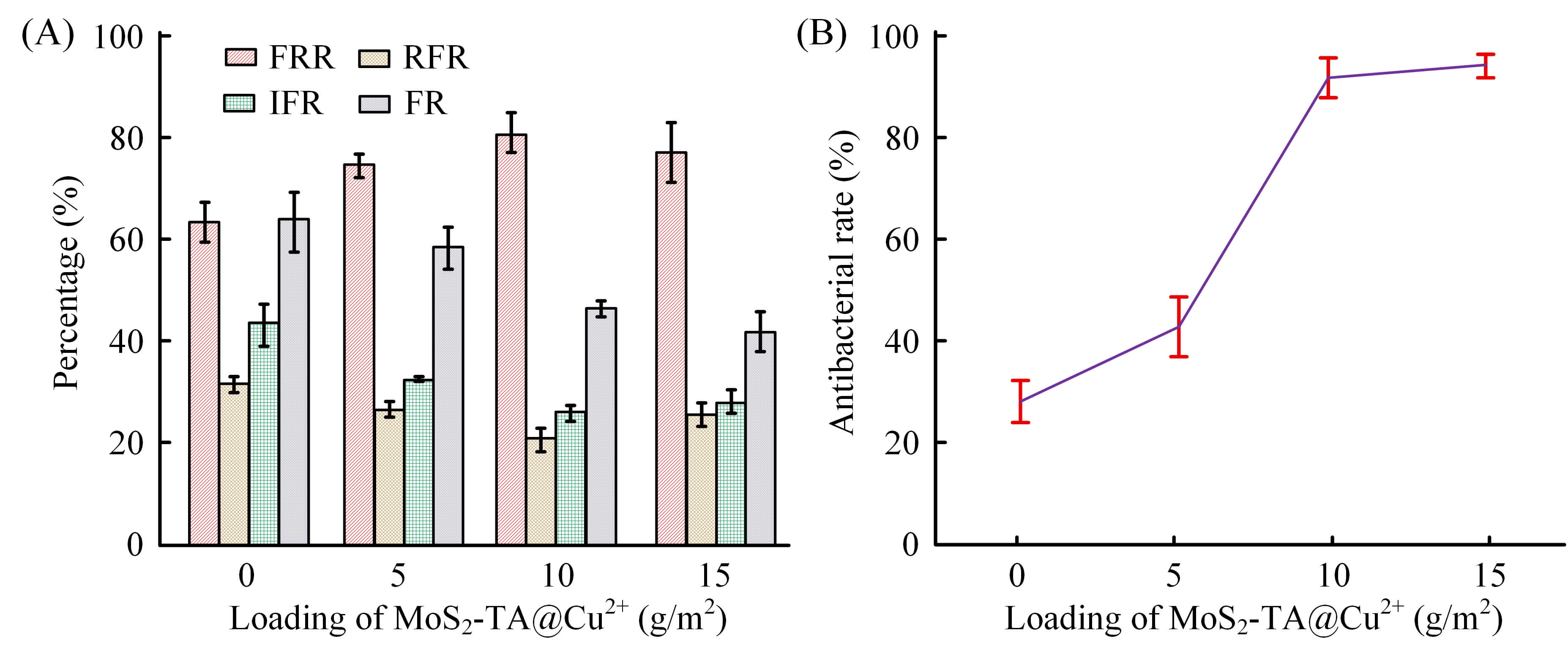 Fig. 6.
Fig. 6.
Anti-fouling and antibacterial properties of membranes. (A) Fouling resistance parameters (RFR, IFR, FR) and FRR of different membranes. (B) Antibacterial rates of different membranes against E. coli. RFR, reversible fouling resistance; IFR, irreversible fouling resistance; FR, fouling resistance; FRR, flux recovery rate; E. coli, Escherichia coli. Error bars represent the standard deviation of three independent replicates.
According to Fig. 7, due to the lack of antibacterial components in the unmodified PES membrane, obvious bacterial proliferation occurred on and around the membrane surface, and no inhibitory zone was formed. The MoS2-TA@Cu2+ Mf-M showed a dose-dependent antibacterial effect: when the nanoparticle loading reached 5 g/m2, the diameter of the inhibition zone was 7.2 mm. When the loading capacity increased to 10 g/m2 and 15 g/m2, the inhibition zone expanded to 9.1 mm and 11.5 mm. This significant antibacterial performance can be attributed to the sustained release of Cu2+, which effectively disrupts microbial cell structures, thus endowing the Mf-M with excellent anti-biofouling properties.
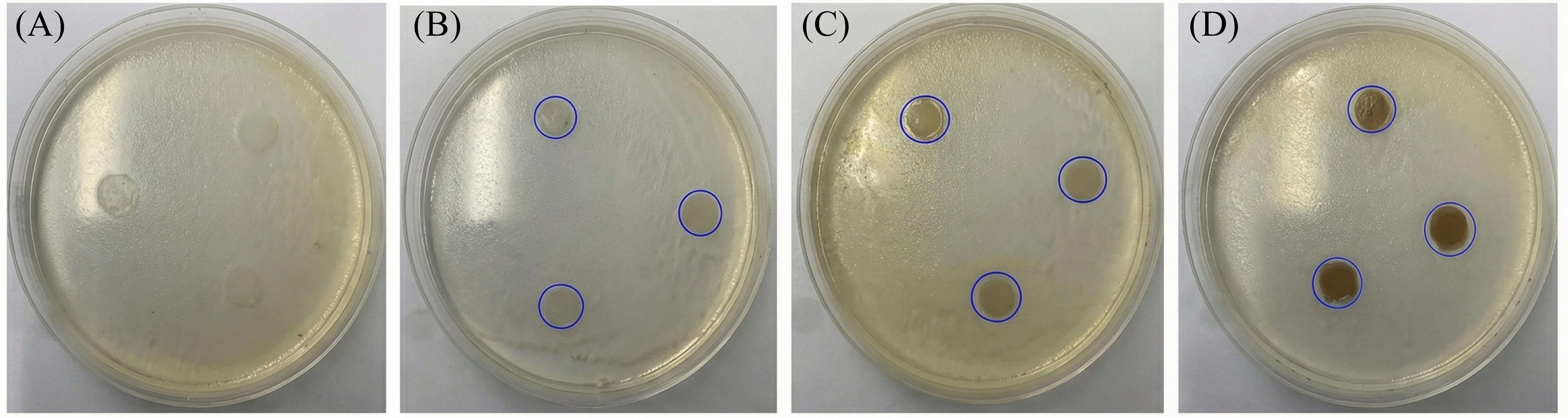 Fig. 7.
Fig. 7.
Inhibition zone test results of different membranes. (A) PES. (B) PES-MoS2-TA@Cu2+-5. (C) PES-MoS2-TA@Cu2+-10. (D) PES-MoS2-TA@Cu2+-15.
It is worth exploring in depth why the membrane with a loading capacity of 10 g/m2 shows the best anti-fouling stability, while the membrane with a loading capacity of 15 g/m2 has better separation performance. It is speculated that this performance trade-off is closely related to the dispersion state of nanoparticles in the polymer matrix and the resulting changes in membrane structure. At lower loadings (such as 10 g/m2), MoS2-TA@Cu2+ nanoparticles may be dispersed in the membrane matrix in a more uniform and isolated state, maximizing the role of their hydrophilic and antibacterial groups, thereby giving the membrane excellent anti-fouling adhesion capabilities. However, when the loading amount is further increased to 15 g/m2, although the macroscopic hydrophilicity continued to increase, the possibility of local micro-agglomeration of the nanoparticles increased. Such microaggregates played a dual role in membrane formation and use. On the one hand, as “porogens”, they induced the formation of larger finger-like pores during the phase transformation process (Fig. 2D), significantly increasing the porosity and PWF of the membrane, which is reflected in the improvement of separation performance. On the other hand, the increased surface roughness caused by these agglomerates would become an “anchor point” for preferential adsorption of pollutants in long-term operation, which in turn slightly weakened its anti-pollution stability in dynamic tests. Therefore, 10 g/m2 was considered the optimal loading to achieve a balance between anti-fouling and separation performance.
To further highlight the superiority of the MoS2-TA@Cu2+ modified
membrane, the comprehensive performance of the optimized membrane in this study
was compared with other reported PES-based nanocomposite ultrafiltration
membranes. As summarized in Table 2 (Ref. [2, 3, 4, 6]), the MoS2-TA@Cu2+
modified membrane exhibits a highly competitive overall performance. Although
Liang et al. [2] achieved higher flux, their membrane required a complex
polyvinylidene fluoride (PVDF)/PES blend and silver nitrate reduction process.
Compared to the pure PES-based membranes modified with inorganic nanoparticles
[3, 4, 6], our work demonstrates a superior balance. Specifically, the
modified membranes demonstrated a superior balance of properties through tunable
nanoparticle loading. The membrane with 15 g/m2 loading achieved a peak pure
water flux of 385.4 L/m2
| Membrane matrix | Modifier/Additive | PWF L/m2 |
FRR (%) | Antibacterial rate (%) | Ref. |
| PES/PVDF | Dopamine/Ag/GO | 650.5 | 84.4 | 96.8 | [2] |
| PES | Tungsten Oxide (WO2.89) | 54.9 | 85.3 | - | [3] |
| PES | MAX Phase (V2GeC) | 249.2 | - | 95.0 | [4] |
| PES | TiO2/MXene | 211.5 | 790* | 95.0 | [6] |
| PES | MoS2-TA@Cu2+ (10 g/m2) | 380.3 | 81.5 | 92.3 | This work |
| PES | MoS2-TA@Cu2+ (15 g/m2) | 385.4 | 76.9 | 95.0 | This work |
Note: PWF, Pure Water Flux; FRR, Flux Recovery Rate. * The FRR of 79.0% reported by Xu et al. [6] was achieved with UV irradiation assistance. “-” indicates data not reported. In this work, the 10 g/m2 loading achieved the highest anti-fouling stability (FRR), while the 15 g/m2 loading exhibited superior antibacterial performance and permeability. PVDF, polyvinylidene fluoride.
Table 3 shows the comparison of turbidity removal rate (TRR), SDI reduction value, and TOC removal rate. The TRR of the Mf-M significantly surpassed that of the unmodified membrane, and increased with the increase of MoS2-TA@Cu2+ loading. The removal rate of the PES-MoS2-TA@Cu2+-15 membrane reached 92.9%, which was 19.1% higher than that of the 73.8% of the unmodified membrane (relative increase of 25.9%). This is related to the increased porosity and enhanced surface hydrophilicity of the Mf-M: a richer pore structure can effectively intercept colloidal particles, and the hydrophilic surface reduces particle adsorption through the hydration layer, thereby improving the turbidity removal effect. The SDI reduction value of the Mf-M outperformed that of the unmodified membrane. The SDI reduction value of the PES-MoS2-TA@Cu2+-15 membrane reached 5.3, and the SDI of the filtrate was reduced to 1.2, which fully met the requirements for reverse osmosis pretreatment and reduced the risk of contamination in the subsequent desalination process. The TOC removal rate of the Mf-M increased with the increase in nanoparticle loading. The removal rate of the PES-MoS2-TA@Cu2+-15 membrane reached 65.7%, which was 25.7% higher than the 40.0% of the unmodified membrane (relative increase of 64.3%). The reason is that the layered structure of MoS2 and the phenolic hydroxyl group of TA can synergistically intercept organic matter through adsorption. At the same time, the optimization of membrane pore size can effectively block the penetration of macromolecular organic matter, thereby reducing the TOC content in water. In summary, MoS2-TA@Cu2+ nanomodification significantly improved the UF membrane’s ability to remove turbidity, colloids, and organic matter in underground karst water, and the SDI reduction effect was particularly prominent. This showed that it could be used as an efficient pretreatment process for high-salinity HHKW to provide high-quality feed water for subsequent deep desalination.
| Membrane type | Turbidity of filtrate | TRR (%) | SDI of filtrate | SDI reduction value | Filtrate TOC (mg/L) | TOC removal rate (%) |
| PES | 1.1 |
73.8 |
3.2 |
3.3 |
2.1 |
40.0 |
| PES-MoS2-TA@Cu2+-5 | 0.7 |
83.3 |
2.3 |
4.2 |
1.7 |
51.4 |
| PES-MoS2-TA@Cu2+-10 | 0.5 |
88.1 |
1.8 |
4.7 |
1.4 |
60.0 |
| PES-MoS2-TA@Cu2+-15 | 0.3 |
92.9 |
1.2 |
5.3 |
1.2 |
65.7 |
TRR, turbidity removal rate; SDI, silt density index; TOC, total organic carbon.
In Fig. 8A, as the loading amount of MoS2-TA@Cu2+ increased, the amount of fouling on the membrane surface showed an obvious downward trend, whether in supersaturated CaCO3 solution or supersaturated CaSO4 solution. In the CaCO3 solution, the scaling amount of the unmodified PES membrane was 0.92 mg/cm2. When the loading reached 15 g/m2, the scaling amount was reduced to 0.32 mg/cm2 corresponding to a reduction rate of approximately 65.2%. In the CaSO4 solution, the initial scaling amount was 0.78 mg/cm2, which dropped to about 0.29 mg/cm2 at 15 g/m2. This showed that MoS2-TA@Cu2+ nanoparticles could effectively inhibit inorganic scaling under static conditions. In Fig. 8B, as the filtration volume increased, the normalized flux attenuation of the unmodified PES membrane was the most significant. When the filtration volume reached 1200 mL, its normalized flux had dropped to 0.1. In contrast, the specific flux attenuation of the Mf-M with MoS2-TA@Cu2+ was significantly slowed down. The PES-MoS2-TA@Cu2+-15 membrane exhibited the strongest resistance to flux attenuation, and the normalized flux remained around 0.5 at the end of filtration. This fully proved that MoS2-TA@Cu2+ nanoparticles could also improve the AFP of the membrane during dynamic filtration.
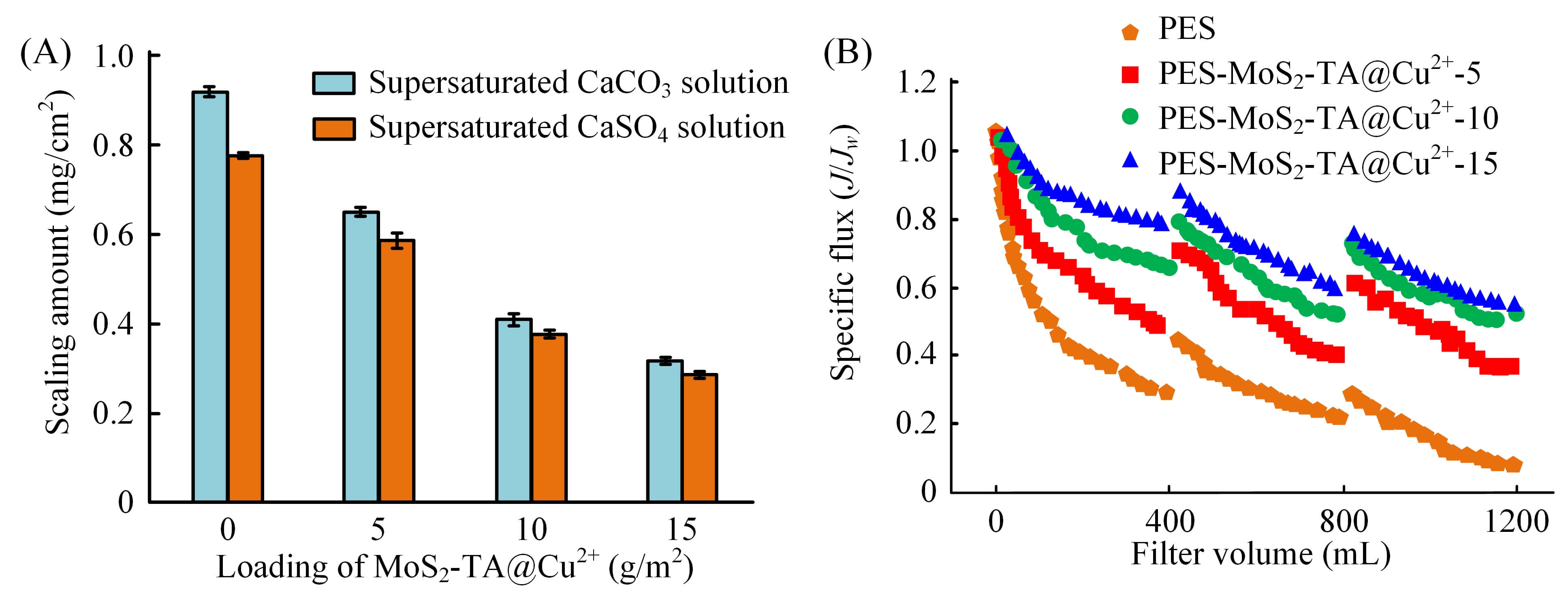 Fig. 8.
Fig. 8.
Membrane AFP test results. (A) The amount of fouling per unit area in the static fouling experiment. (B) The change of specific flux with filtration volume in the dynamic filtration experiment. AFP, anti-fouling performance.
A core finding of this study is that the membrane with a loading capacity of 10 g/m2 exhibits optimal anti-fouling recoverability and long-term stability, whereas the membrane with a loading capacity of 15 g/m2 is superior in permeability and instantaneous separation efficiency. This performance trade-off can be attributed to the dispersion state of the nanoparticles in the polymer matrix and their dual regulatory role on the membrane structure and surface properties.
On the one hand, acting as efficient “porogens”, especially at a high loading of 15 g/m2, the local micro-agglomeration of nanoparticles induces the formation of larger and more interconnected finger-like pores during the phase transformation process (macro pore structure in Fig. 2D). This significantly increases the overall porosity and PWF of the membrane, resulting in optimal separation performance.
On the other hand, these microaggregates also alter the micromorphology of the membrane surface. At a lower loading (10 g/m2), the nanoparticles are dispersed more uniformly, maximizing the exposure of hydrophilic and antibacterial groups, thereby endowing the membrane with excellent anti-fouling adhesion capabilities. However, at higher loading (15 g/m2), microagglomerates may increase surface roughness at the nanometer scale. These raised structures can act as “anchoring sites” for the preferential adsorption of pollutants during long-term operation. Although the macroscopic hydrophilicity remains strong, the presence of these “anchors” slightly increases the difficulty of foulant detachment during backwashing, which explains why the FRR (76.9%) at this loading is slightly lower than the optimal value achieved at 10 g/m2.
Therefore, 10 g/m2 is considered the optimal loading amount to achieve a balance between anti-pollution and separation performance, maximizing the anti-pollution potential while ensuring excellent separation efficiency.
While the elucidation of this tunable trade-off mechanism provides a precise
theoretical guideline for balancing membrane permeability and stability in
controlled environments, the transition from laboratory synthesis to practical
industrial application still faces several constraints that must be acknowledged.
First, the experiments were conducted on a laboratory scale using dead-end and
cross-flow filtration modules. While these results provide a strong
proof-of-concept, they may not fully predict the hydrodynamic behavior and
fouling distribution in industrial-scale spiral-wound membrane modules. Second,
although the 72-hour static scaling and three-cycle dynamic filtration tests
confirmed the short-term stability of the membrane, the long-term durability
(
In this study, a multifunctional MoS2-TA@Cu2+ modified PES ultrafiltration membrane was successfully developed to address the critical challenges of inorganic scaling and biological fouling in high-hardness karst water treatment. The scientific significance of this work lies in revealing the inherent tunable trade-off mechanism between anti-fouling stability and separation efficiency, which is precisely regulated by nanoparticle loading (identifying 10 g/m2 for stability vs. 15 g/m2 for permeability).
Quantitatively, this study confirmed a critical performance trade-off regulated by nanoparticle loading, where the 10 g/m2 loading achieved optimal operational stability, while the 15 g/m2 loading prioritized permeability and separation efficiency:
(1) Permeability (Separation-Dominant): The membrane with 15 g/m2 loading
achieved a peak pure water flux of 385.4 L/m2
(2) Anti-fouling & Antibacterial Stability (Stability-Dominant): The membrane with 10 g/m2 loading achieved the highest FRR of 81.5% solely through physical hydraulic washing, surpassing the typical range (60–75%) of conventional nanocomposite membranes. Simultaneously, the membrane system demonstrated potent biological resistance, with the antibacterial rate reaching up to 95% against E. coli (at 15 g/m2 loading), competitive with silver-based membranes but with lower cost and toxicity risks.
(3) Practical Engineering Value: In the treatment of actual underground karst
water, the membrane exhibited a turbidity removal rate of 92.9% and, most
importantly, reduced the SDI of the filtrate to 1.2. This value is far below the
stringent threshold (SDI
To bridge the gap between laboratory success and industrial application, future
work will focus on scaling up the fabrication process to pilot-scale rolled
membrane modules. Priority will be given to evaluating the mechanical integrity
and chemical stability of the membranes under real-world operating conditions
over extended periods (
All data generated or analyzed during this study are included in this published article.
MZ designed the research study, performed the research, and analyzed the data. MZ drafted the manuscript and contributed to the critical revision for important intellectual content. The author read and approved the final manuscript. The author has participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The author would like to acknowledge the Basic Geological Survey Institute of Jiangxi Geological Survey and Exploration Institute for providing the necessary facilities to conduct this research.
This research received no external funding.
The author declares no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








